Submitted:
15 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Literature Collection
3. Discussion
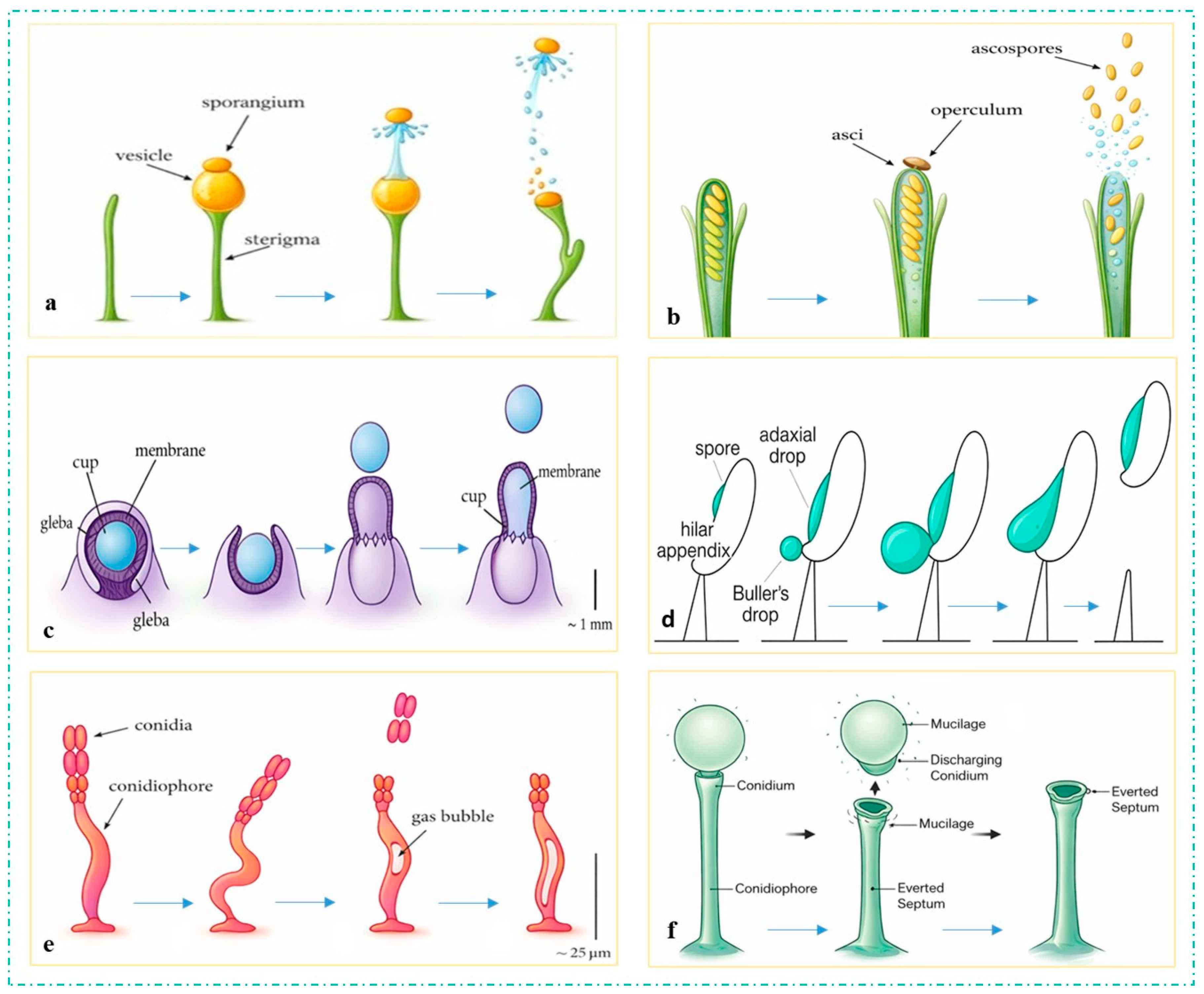
3.1. Ballistic Spore Launching from a Biophysical Perspective
3.1.1. Turgor Pressure Generation
3.1.2. Energy Storage and Transformation
3.1.3. Rapture and Launch Mechanics
| Species | Number of spores released per sporangium |
Initial velocity (m s 1): range, mean ± s.e.m. (n) |
Peak launch velocity | Peak launch acceleration | Calculated discharge distance (cm): range, mean | Reference |
| Pilobolus kleinii | 30,000–90,000 | 0.01–0.03 | 16 m/s (mean: 9 m/s) | 21,407g | 2.5m | [5,26] |
| Basidiobolus ranarum | 1 conidium | - | 9 m/s (mean: 4 m/s) | 152,905g | 0.02- 0.05m | [5,26] |
| Species | Number of spores released per ascus |
Initial velocity (m s 1): range, mean ± s.e.m. (n) |
Launch acceleration | Calculated discharge distance (cm): range, mean | Reference |
| Ascobolus immersus | 8 | 5–18, 14 ±1.3 (12) | 183,486 peak | 9–33, 26 | [7,26] |
| Podospora anserina | 4 | 10–25, 21 ±1.1 (17) | - | 7–17, 14 | [7,26] |
| Morchella semilibera | 8 | 16–25, 19 ±1.9 (4) | - | 17–27, 21 (5 cm for spores flying singly) | [7] |
|
Neurospora tetrasperma |
4 | 4–32, 16 ±0.6 (58) | - | 2–16, 8 | [5,7] |
| Sordaria macrospora | 8 | 5–30, 15 ±0.1 (43) | - | 4–24, 12 | [7] |
| Sporormiella australis | one at a time | 15–16, 16 ±0.1 (5) | - | 0.80–0.85, 0.85 | [7] |
| Leptosphaeria acuta | one at a time | 4–5, 4 ±0.5 (3) | - | 0.24–0.31, 0.24 | [7] |
| Neolecta vitellina | stream of multiple spores | 13–19, 16 ±1.6 (3) | - | 0.12–0.18, 0.15 | [7] |
| Macrospora scirpicolac | one at a time | 3–7, 5 ±0.5 (10) | - | 0.02–0.05, 0.04 | [7] |
| Leptosphaeria acutac | one at a time | 1–2, 1 ±0.3 (4) | - | 0.001–0.004, 0.003 | [7] |
| Gibberella zeae | - | 34.5 | 870,000 | 2.810−3 - 8.510−3 | [5] |
| Species | Radius of Buller’s drop | Initial velocity (m s −1) : range, mean ± s.e.m. (n) |
Launch acceleration (g) | Calculated discharge distance (cm): range, mean | Reference |
| Sphaerobolus stellatus | - | 9.16 ± 0.79 | - | 4 to 7 m (average 6m) | [20] |
| Gymnosporangium juniperi virginianae | 5.2 mm (19) | 0.66–1.35, 1.1160.06 (18) | - | 1.2660.06 mm | [24] |
| Tilletia caries | 5.2 mm (28) | 0.32–1.53, 1.1060.07 (21) | 0.6660.04 mm | [24] | |
| Sporobolomyces salmonicolor | 3.8 mm (14) | 1.08–1.83, 1.4260.12 (6) | - | 0.5460.04 mm | [24] |
| Auricularia auricula | 3.1 mm (15) | 0.87–1.62, 1.2560.06 (13) | - | 0.4560.02 mm | [5,24] |
| Polyporus squamosus | 2.6 mm (4) | 0.45–0.68, 0.5860.08 (6) | - | 0.1460.02 mm [0.1360.02 mm (6)] | [24] |
| Armillaria tabescens | 1.5 mm (16) | 0.12–0.91, 0.6460.08 (9) | - | 0.1060.01 mm [0.0660.01 mm (4)] | [24] |
| Clavicorona pyxidata | 1.2 mm (6) | 0.52–0.87, 0.6960.06 (5) | - | 0.04260.004 mm [0.03660.003 mm (5)] | [24] |
| Itersonilia perplexans | - | 0.67 | ~ 25,484 | 1.10−3mm,- | [5] |
| Hyphodontia latitans | - | 1.05 | - | 4.10−6 peak | [5] |
| Aleurodiscus gigasporus | - | - | - | 2 mm,- | [32] |
| Hyphodontia latitans | - | - | - | 4 µm,- | [32] |
3.2. Acceleration and Force
3.3. Projectile Motion, Air Resistance, and Drag
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oneto, D.L.; Golan, J.; Mazzino, A.; et al. Timing of fungal spore release dictates survival during atmospheric transport. Proc Natl Acad Sci USA 2020, 117, 5134–5143. [Google Scholar] [CrossRef] [PubMed]
- Roper, M.; Seminara, A.; Bandi, M.M.; et al. Dispersal of fungal spores on a cooperatively generated wind. Proc. Natl. Acad. Sci. USA 2010, 107, 17474–17479. [Google Scholar] [CrossRef] [PubMed]
- Hassett, M.O.; Fischer, M.W.F.; Sugawara, Z.T.; et al. Splash and grab: Biomechanics of peridiole ejection and function of the funicular cord in bird’s nest fungi. Fungal Biol 2013, 117, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Trail, F. Fungal cannons: explosive spore discharge in the Ascomycota. FEMS Microbiol Lett. 2007, 276, 12–18. [Google Scholar] [CrossRef]
- Sakes, A.; Wiel, MDer; Van Henselmans, P.W.J.; et al. Shooting mechanisms in nature: A systematic review. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Bueno-Sancho, V.; Orton, E.S.; Gerrity, M.; et al. Aeciospore ejection in the rust pathogen Puccinia graminis is driven by moisture ingress. Commun Biol. 2021, 4. [Google Scholar] [CrossRef]
- Money, N.P.; Stolze-Rybczynski, J.; Smith, B.E.; et al. Ascus function: From squirt guns to ooze tubes. Fungal Biology 2023, 127, 1491–1504. [Google Scholar] [CrossRef]
- Pringle, A.; Patek, S.N.; Fischer, M.; et al. The captured launch of a ballistospore. Mycologia 2005, 97, 866–871. [Google Scholar] [CrossRef]
- Viriato, A. Pilobolus species found on herbivore dung from the São Paulo Zoological Park, Brazil. Acta Bot Bras. 2008, 22, 614–620. [Google Scholar] [CrossRef]
- Mukherjee, R.; Gruszewski, H.A.; Bilyeu, L.T.; Schmale, D.G.I.I.I.; Boreyko, J.B. Synergistic dispersal of plant pathogen spores by jumping-droplet condensation and wind. Proc. Natl. Acad. Sci. USA 2021, 24, 118. [Google Scholar] [CrossRef]
- Money, N.P. Chapter 3 - Spore Production, Discharge, and Dispersal. In The Fungi (Third Edition); Watkinson, S.C., Boddy, L., Money, N.P., Eds.; Academic Press: Boston, 2016; pp. 67–97. [Google Scholar] [CrossRef]
- Aluoch, A.M.; Otiende, M.Y.; Obonyo, M.A.; et al. First genetic identification of Pilobolus (Mucoromycotina, Mucorales) from Africa (Nairobi National Park, Kenya). South African Journal of Botany 2017, 111, 182–8. [Google Scholar] [CrossRef]
- Lee, C.M.; van Geel, B.; Gosling, W.D. On the Use of Spores of Coprophilous Fungi Preserved in Sediments to Indicate Past Herbivore Presence. Quaternary 2022, 5. [Google Scholar] [CrossRef]
- Money, N.P. Goldilocks mushrooms: How ballistospory has shaped basidiomycete evolution. Fungal Biology 2023, 127, 975–84. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Chavez, R.L.; Patek, S.N.; et al. Asymmetric drop coalescence launches fungal ballistospores with directionality. J R Soc Interface 2017, 14. [Google Scholar] [CrossRef]
- Roper, M.; Pepper, R.E.; Brenner, M.P.; et al. Explosively launched spores of ascomycete fungi have drag-minimizing shapes. Proc Natl Acad Sci USA 2008, 105, 20583–20588. [Google Scholar] [CrossRef] [PubMed]
- Corona Ramirez, A.; Bregnard, D.; Junier, T.; et al. Assessment of fungal spores and spore-like diversity in environmental samples by targeted lysis. BMC Microbiol. 2023, 23. [Google Scholar] [CrossRef]
- Fischer, M.; Cox, J.; Davis, D.J.; et al. New information on the mechanism of forcible ascospore discharge from Ascobolus immersus. Fungal Genetics and Biology 2004, 41, 698–707. [Google Scholar] [CrossRef]
- Iapichino, M.; Wang, Y.W.; Gentry, S.; et al. A precise relationship among Buller’s drop, ballistospore, and gill morphologies enables maximum packing of spores within gilled mushrooms. Mycologia 2021, 113, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Zohuri, B. Chapter 5 - First Law of Thermodynamics. In Physics of Cryogenics; Zohuri, B., Ed.; Elsevier, 2018; pp. 119–63. [Google Scholar] [CrossRef]
- Nobliri, X.; Yang, S.; Dumais, J. Surface tension propulsion of fungal spores. Journal of Experimental Biology 2009, 212, 2835–2843. [Google Scholar] [CrossRef] [PubMed]
- Money, N.P.; Stolze, J.; Fischer, M.W.F. Mechanics of the artillery fungus. Fungal Biol. 2024, 128, 2334–2340. [Google Scholar] [CrossRef]
- Fischer, M.W.F.; Stolze-Rybczynski, J.L.; Davis, D.J.; et al. Solving the aerodynamics of fungal flight: How air viscosity slows spore motion. Fungal Biol. 2010, 114, 943–8. [Google Scholar] [CrossRef] [PubMed]
- Ruiter JDe Arnbjerg-Nielsen, S.F.; Herren, P.; et al. Fungal artillery of zombie flies: Infectious spore dispersal using a soft water cannon. J R Soc Interface 2019, 16. [Google Scholar] [CrossRef]
- Challita, E.J.; Rohilla, P.; Saad Bhamla, M. Fluid ejections in nature. Annu Rev Chem Biomol Eng. 2024, 15, 187–217. [Google Scholar] [CrossRef] [PubMed]
- Stolze-Rybczynski, J.L.; Cui, Y.; Stevens, M.H.H.; et al. Adaptation of the spore discharge mechanism in the Basidiomycota. PLoS One 2017, 4. [Google Scholar] [CrossRef]
- Coelho, M.A.; Bakkeren, G.; Sun, S.; et al. Fungal Sex: The Basidiomycota. Microbiol Spectr. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Yafetto, L.; Carroll, L.; Cui, Y.; et al. The fastest flights in nature: High-speed spore discharge mechanisms among fungi. PLoS One 2008, 3. [Google Scholar] [CrossRef] [PubMed]
- MEREDITHDS. Spore Discharge in Deightoniella torulosa (Syd.) Ellis. Ann Bot. 1961. [Google Scholar] [CrossRef]
- PMKirk, P.F.C.D.W.M.; JAS. DICTIONARY OF THE FUNGI, 10th ed.; Cromwell Press: Wallingford, UK; Trowbridge, 2008; Available online: www.indexfungorum.org.
- Money, N.P.; Fischer, M.W.F. 6 Biomechanics of Spore Release in Phytopathogens. In The Mycota, 2nd ed. n.p.; Karl Esser, H.B.D., Ed.; Springer-Verlag: Berlin Heidelberg, 2009. [Google Scholar]
- Fischer, M.W.F.; Stolze-Rybczynski, J.L.; Cui, Y.; et al. How far and how fast can mushroom spores fly? Physical limits on ballistospore size and discharge distance in the Basidiomycota. Fungal Biol. 2010, 114, 669–675. [Google Scholar] [CrossRef]
- Segers, F.J.J.; Dijksterhuis, J.; Giesbers, M.; et al. Natural folding of airborne fungal spores: a mechanism for dispersal and long-term survival? In Fungal Biology Reviews; Elsevier Ltd, 2023; vol. 44. Preprint. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




