Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Quantum-Chemical Calculations
2.2. Synthesis and Physicochemical Characterization
| Indicator | KC-246 | KC-248 |
|---|---|---|
| Iodide concentration, mg/L | 382.99 ± 0.76 | 238.80 ± 0.62 |
| Peak area, S (mEA×s ) | 1008.97 | 629.13 |
| Indicator | KC-246 | KC-248 |
|---|---|---|
| Cation concentration, mg/L | 11.43 ± 0.11 | 9.15 ± 0.04 |
| Peak area, S (mEA×s) | 14.005 | 11.215 |
| Compound name | Solubility | рН | Melting point, °С | Iodine, g/kg | Iodide, g/kg |
|---|---|---|---|---|---|
| Titration | |||||
| KC-246 | Water, DMSO, acetone 1g/10mL (soluble) |
2.31 ± 0.02 | 57-58 ± 0.58 | 99.78 ± 1.05 | 286.95 ± 0.98 |
| KC-248 | Water 1g/5mL (freely soluble) |
3.92 ± 0.05 | 62-64 ± 0.25 | 99.47 ± 2.13 | 168.17 ± 1.97 |
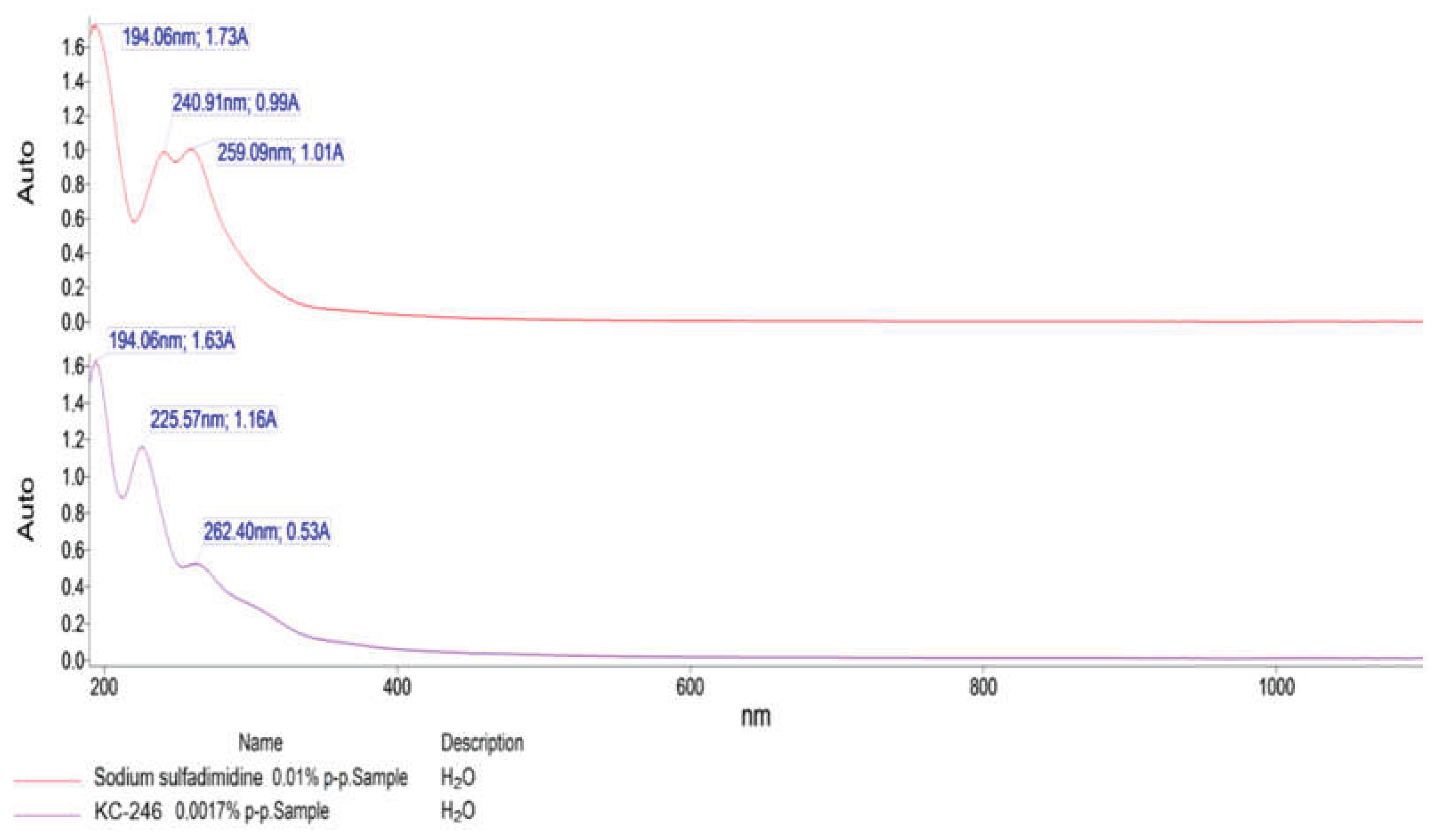
2.3. UV Spectroscopic Methods of Analysis
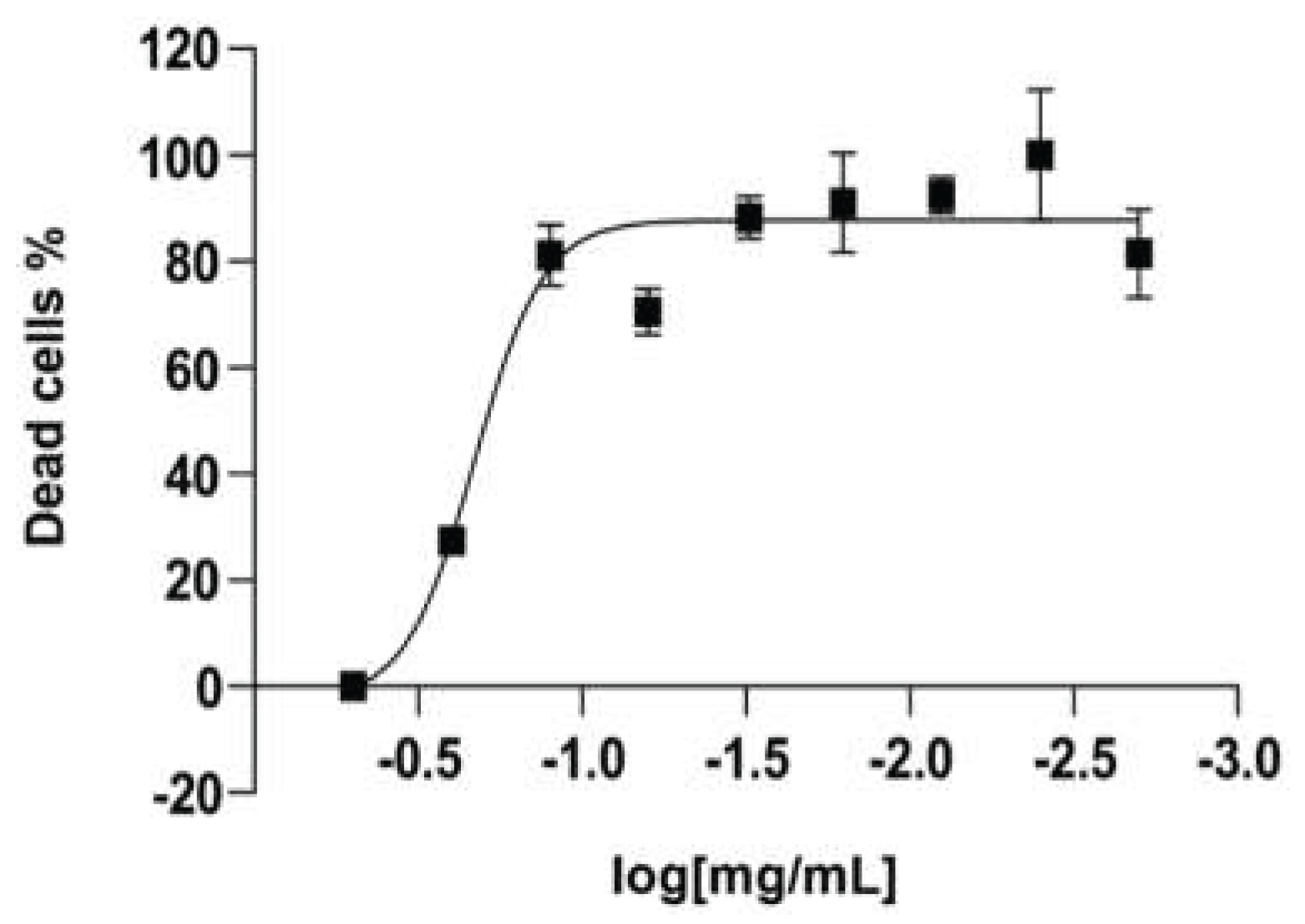
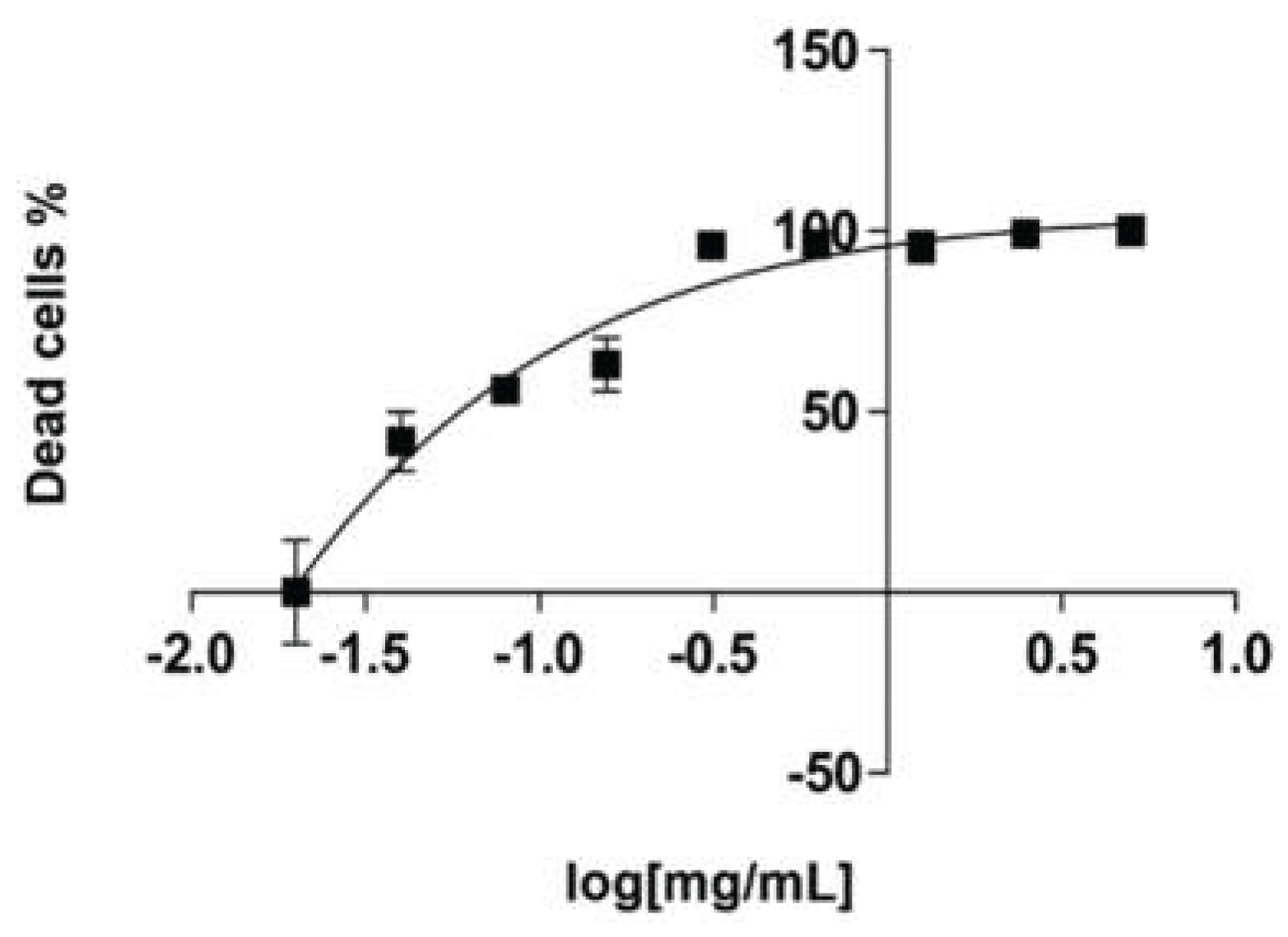
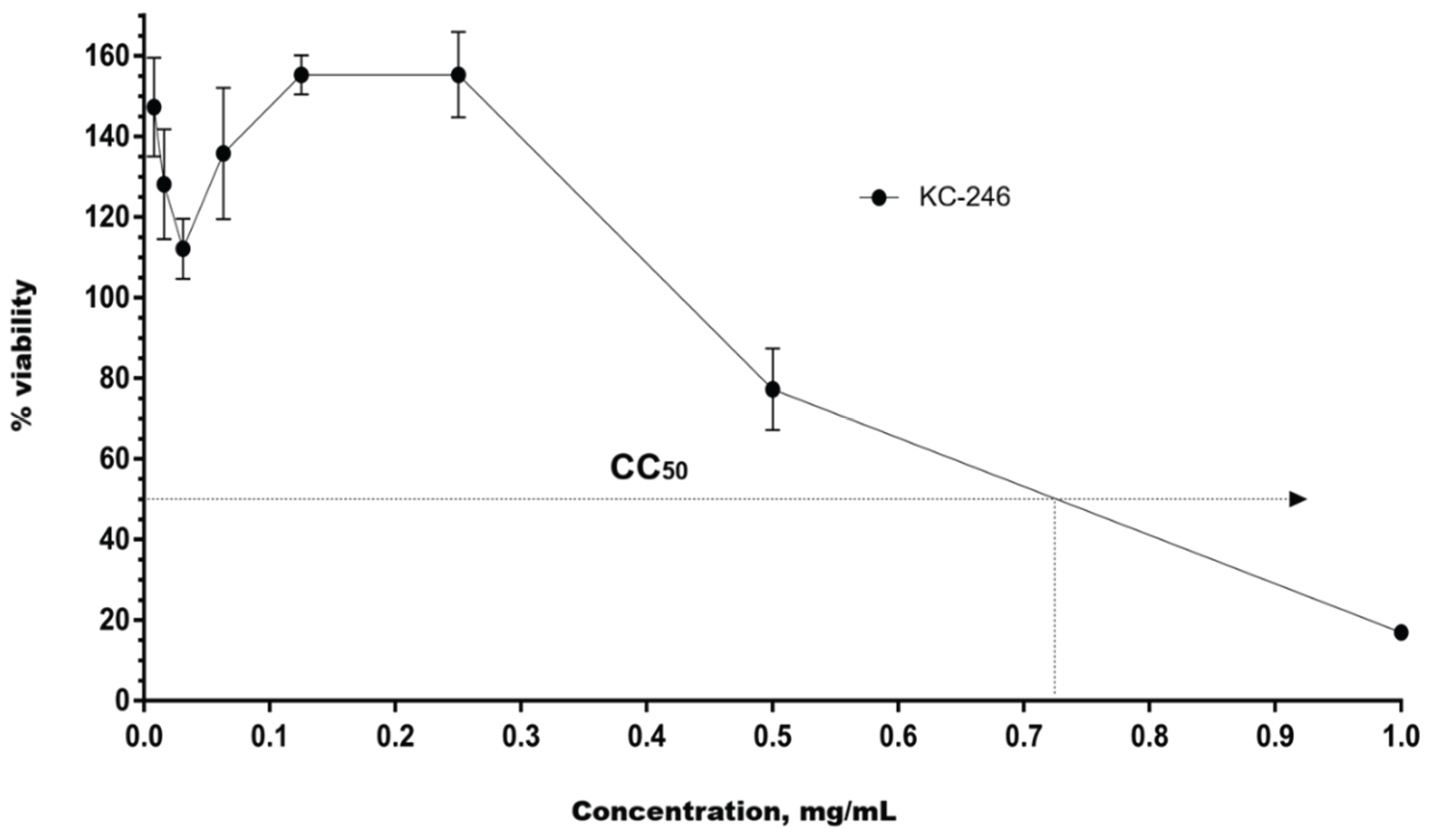
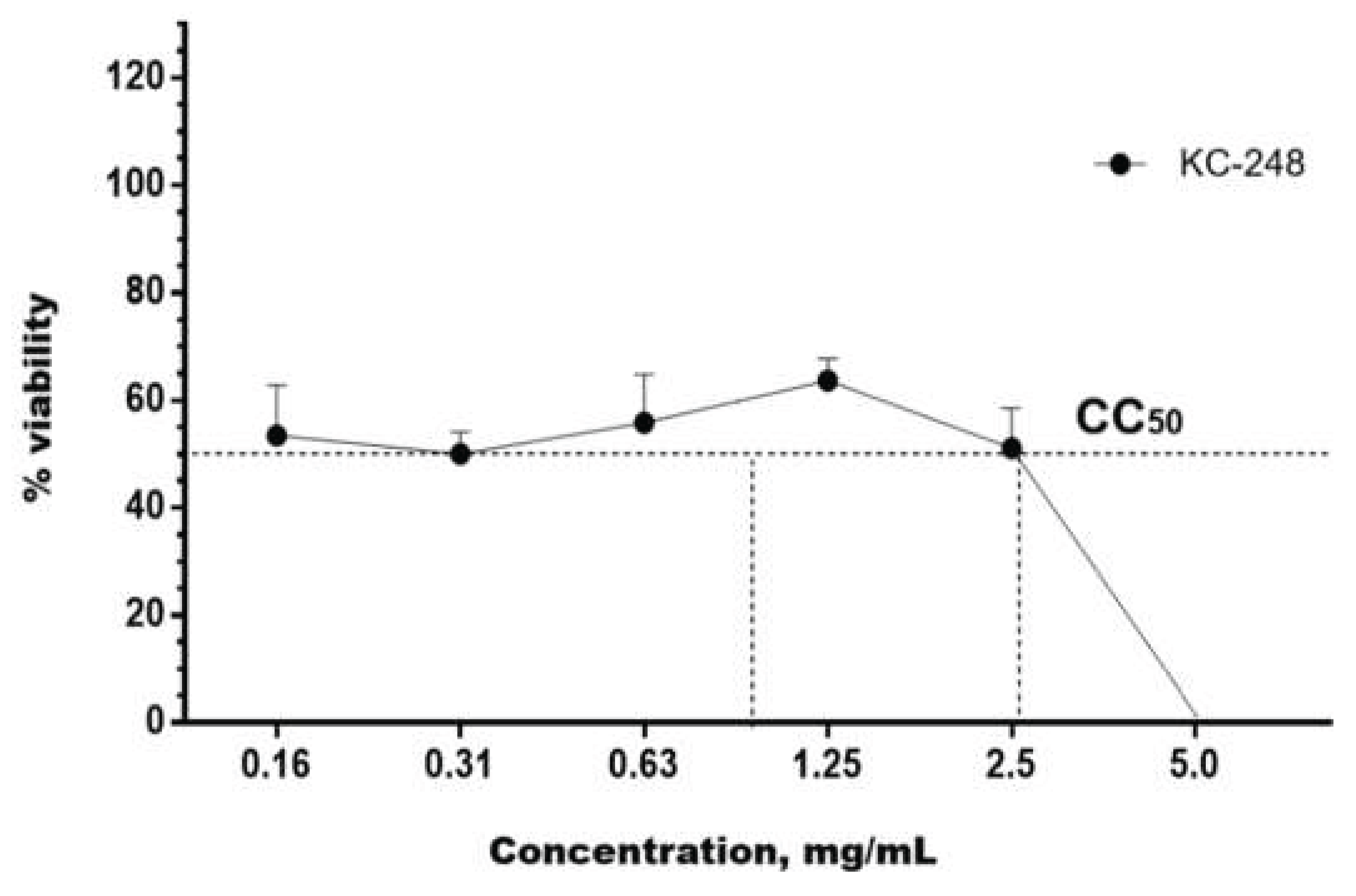
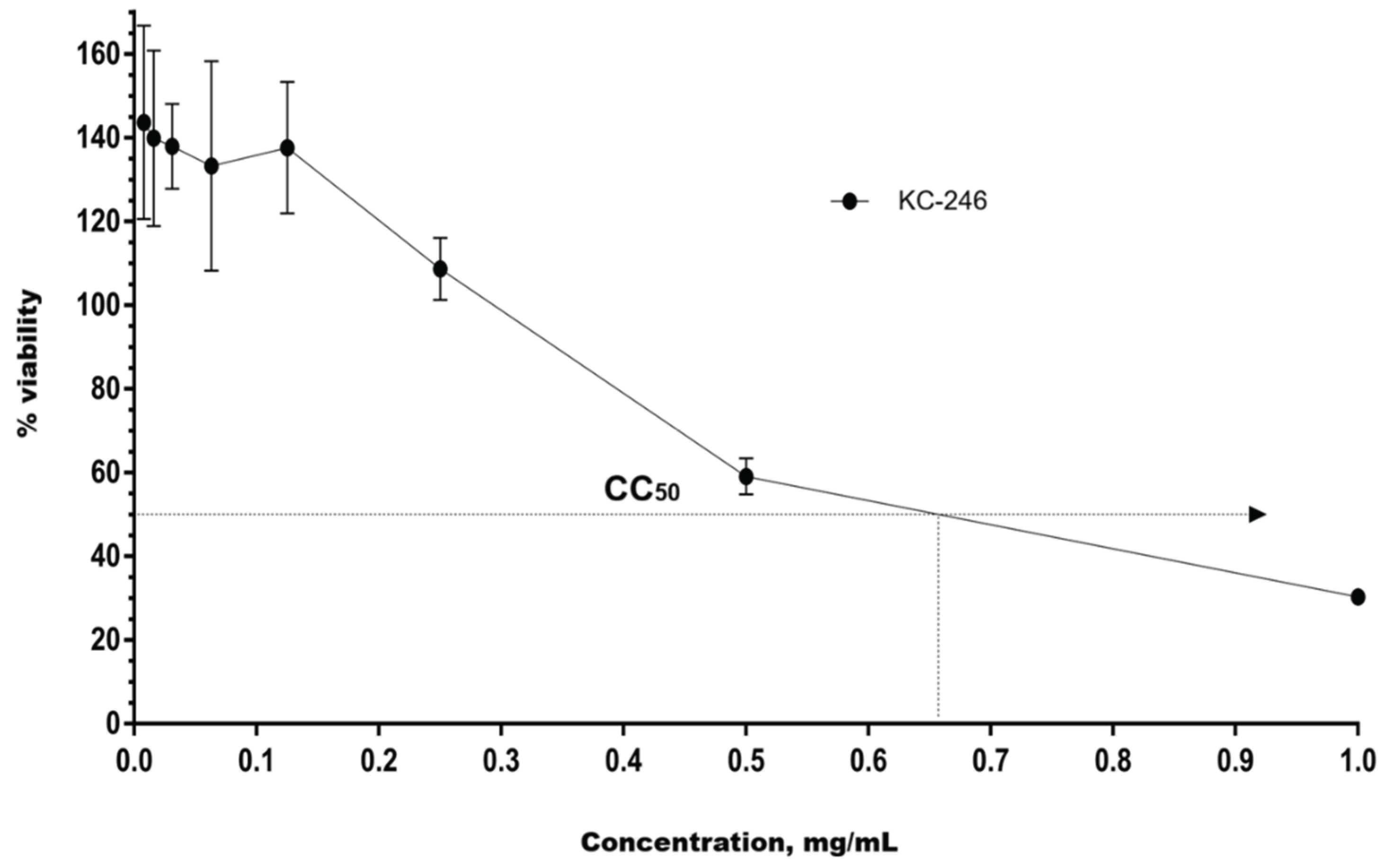
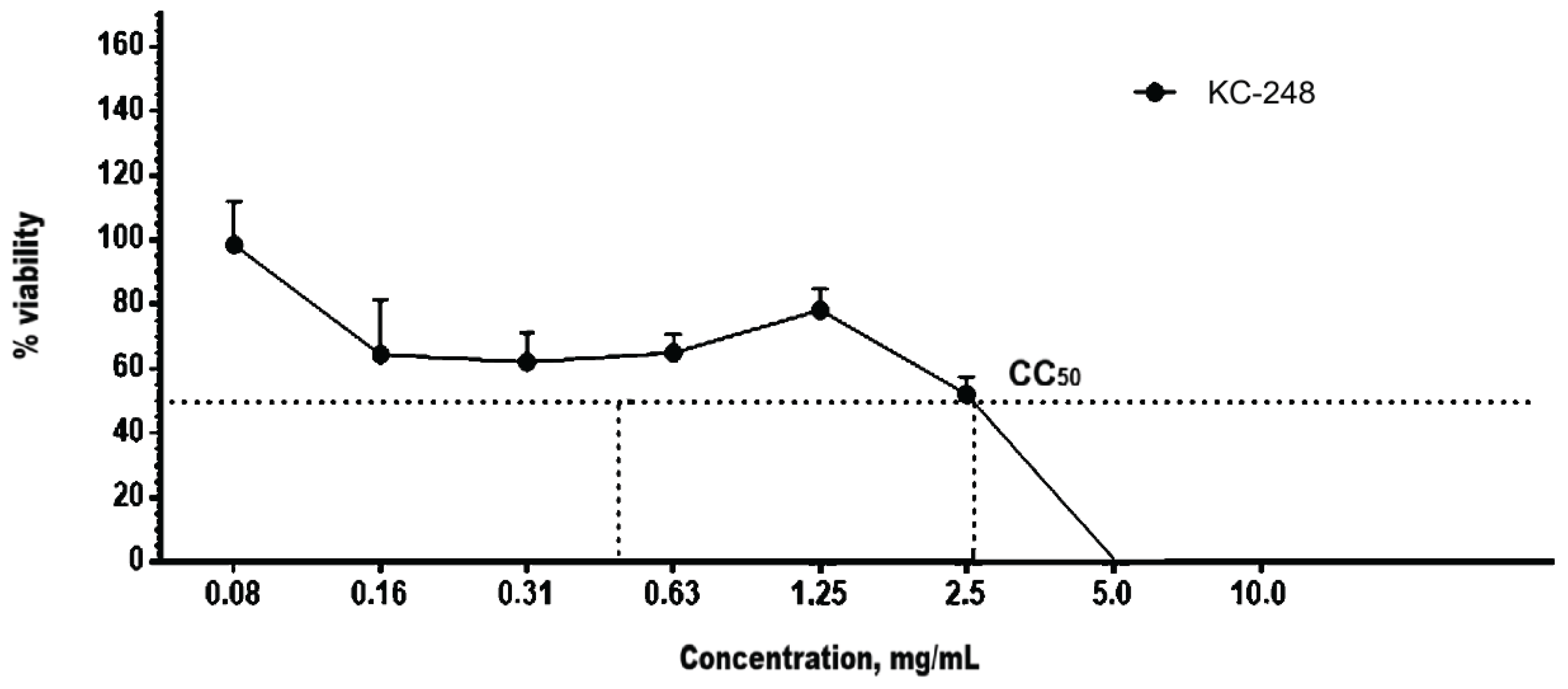
2.4. In Vitro Safety Testing
| Compound name | CC50, on MDCK cell celture, (mg/mL) | CC50, on RD cell culture, (mg/mL) |
|---|---|---|
| KC-246 | 0.730 ± 0.06 | 0.660 ± 0.3 |
| KC-248 | 2.550 ± 3.30 | 2.600 ± 1.60 |
2.5. Antimicrobial Activity Screening
2.6. Virus-Inhibitory Activity Screening
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Test-Systems
- Staphylococcus aureus ATCC 6538-P (museum, sensitive strain)
- Staphylococcus aureus ATCC 333591 (museum, multidrug-resistant strain)
- Escherichia coli ATCC 8739 (museum, sensitive strain)
- Escherichia coli ATCC BAA-2523 (museum, multidrug-resistant strain)
- Pseudomonas aeruginosa ATCC 9027 (museum, sensitive strain)
- Pseudomonas aeruginosa TA2 (clinical, multidrug-resistant strain)
- Klebsiella pneumoniae ATCC 10031 (museum, sensitive strain)
- Klebsiella pneumoniae ATCC 2524 (museum, multidrug-resistant strain)
- Salmonella enterica ATCC 14028 (museum, sensitive strain)
- Salmonella enterica ATCC 35988 (museum, multidrug-resistant strain)
- Enterococcus hirae ATCC 10541 (museum, sensitive strain)
- Enterococcus faecalis ATCC 51575 (museum, multidrug-resistant strain)
- Candida albicans ATCC 10231 (museum, sensitive strain)
- Candida albicans SCID PHRX 1-2019 (clinical, multidrug-resistant strain)
4.3. Preparation of the APSs
4.4. Quantum-Chemical Calculations
4.5. UV-Vis Spectroscopy
4.6. Capillary Electrophoresis (CE)
4.7. Quantitive Analysis of Iodine and Iodide
4.8. Serial Dilution Method
4.9. Endpoint Dilution Assay for Infectious Titer Determination
4.10. MTT Cytotoxicity Assay
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| APS | Active Pharmaceutical Substance |
| AMR | Antimicrobial Resistance |
| ATCC | American Type Culture Collection |
| BGE | Background Electrolyte |
| CC₅₀ | Half-maximal Cytotoxic Concentration |
| CDC | Centers for Disease Control and Prevention |
| CE | Capillary Electrophoresis |
| CLSI | Clinical and Laboratory Standards Institute |
| CPE | Cytopathic Effect |
| CRS | Certified Reference Standard |
| DAD | Diode-Array Detector |
| DFT | Density Functional Theory |
| DMSO | Dimethyl Sulfoxide |
| FBS | Fetal Bovine Serum |
| GLP | Good Laboratory Practice |
| GMP | Good Manufacturing Practice |
| HSV-1 | Herpes Simplex Virus Type 1 |
| MBC | Minimum Bactericidal Concentration |
| MDCK | Madin-Darby Canine Kidney |
| MES | Ministry of Education and Science |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| OD | Optical Density |
| PBMC | Peripheral Blood Mononuclear Cell |
| RD | Rhabdomyosarcoma |
| RT | Room Temperature |
| UV-Vis | Ultraviolet-Visible (spectroscopy) |
References
- Curren, E.J.; Lutgring, J.D.; Kabbani, S.; Diekema, D.J.; Gitterman, S.; Lautenbach, E.; Morgan, D.J.; Rock, C.; Salerno, R.M.; McDonald, L.C. Advancing diagnostic stewardship for healthcare-associated infections, antibiotic resistance, and sepsis. Clin. Infect. Dis. 2022, 74, 723–728. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Regional Office for Europe. Tackling Antibiotic Resistance from a Food Safety Perspective in Europe; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 2011. [Google Scholar]
- Naghavi, M.; et al. Global burden of bacterial antimicrobial resistance 1990–2021: A systematic analysis with forecasts to 2050. Lancet 2024, 404, 1199–1226. [Google Scholar] [CrossRef] [PubMed]
- Poirel, L.; Jayol, A.; Nordmann, P. Polymyxins: Antimicrobial activity, susceptibility testing, and resistance mechanisms encoded by plasmids or chromosomes. Clin. Microbiol. Rev. 2017, 30, 557–596. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Kasiakou, S.K.; Tsiodras, S. Colistin: The revival of polymyxins for the management of multidrug-resistant gram-negative bacterial infections. Clin. Infect. Dis. 2019, 40, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Turganbay, S.; Kenesheva, S.; Jumagaziyeva, А. et al. Synthesis, physicochemical properties and antimicrobial activity of a di-aminopropionic acid hydrogen tri-iodide coordination compound. BMC Res Notes 2024, 17, 384. [Google Scholar] [CrossRef] [PubMed]
- Doszhanov, Y.; Atamanov, M.; Jandosov, J.; Saurykova, K.; Bassygarayev, Zh.; Orazbayev, A.; Turganbay, S.; Sabitov, A. Preparation of Granular Organic Iodine and Selenium Complex Fertilizer Based on Biochar for Biofortification of Parsley. Scientifica 2024, 6601899, 14 pages. [Google Scholar] [CrossRef] [PubMed]
- Sirimulla, S.; Bailey, J.B.; Vegesna, R.; Narayan, M. Halogen interactions in protein–ligand complexes: Implications of halogen bonding for rational drug design. J. Chem. Inf. Model. 2013, 53, 2781–2791. [Google Scholar] [CrossRef] [PubMed]
- Shepilov, D.; Turganbay, S.; Jumagaziyeva, A.; Bukeyeva, T.; Askarova, D.; Bolatova, D.; Doszhanov, Y.; Zhumazhanov, A.; Kerimkulova, A.; Saurykova, K.; et al. The Iodine–Dextrin–Lithium Complex: Morphology, Antibacterial Activity, and Cytotoxicity. Molecules 2025, 30, 4822. [Google Scholar] [CrossRef] [PubMed]
- Turganbay, S.; Ilin, A.; Sabitov, A.; Askarova, D.; Jumagaziyeva, A.; Iskakbayeva, Z.; Seisembekova, A.; Bukeyeva, T.; Azembayev, A. Dextrin/Polyvinyl Alcohol/Iodine Complexes: Preparation, Characterization, and Antibacterial, Cytotoxic Activity. Eng. Sci. 2025, 35, 1595. [Google Scholar] [CrossRef]
- Aluker, N.L.; Mitrofanov, A.Yu.; Suzdaltseva, Ya.M. Spektrofotometricheskoe issledovanie vodnykh rastvorov soedinenii ioda [Spectrophotometric Study of Aqueous Solutions of Iodine Compounds]. 1964.
- Vardanyan, R.S.; Hruby, V.J. Antimicrobial Drugs. In Synthesis of Essential Drugs; Elsevier: Amsterdam, The Netherlands, 2006; pp. 499–523. [Google Scholar] [CrossRef]
- Plumlee, K.H. Pharmaceuticals. In Clinical Veterinary Toxicology; Mosby: St. Louis, MO, USA, 2004; pp. 282–336. [Google Scholar] [CrossRef]
- Hodiamont, C.J.; van den Broek, A.K.; de Vroom, S.L.; Prins, J.M.; Mathôt, R.A.A.; van Hest, R.M. Clinical pharmacokinetics of gentamicin in various patient populations and consequences for optimal dosing for gram-negative infections: An updated review. Clin. Pharmacokinet. 2022, 61, 1075–1094. [Google Scholar] [CrossRef] [PubMed]
- Garneau-Tsodikova, S.; Labby, K.J. Mechanisms of resistance to aminoglycoside antibiotics: Overview and perspectives. MedChemComm 2016, 7, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Voloshchuk, A.M. Rukovodstvo po kapillyarnomu elektroforezu [Guide to Capillary Electrophoresis]; Moscow, Russia, 1996.
- Agilent Technologies. Instruction Manual for the Capillary Electrophoresis System; Agilent Technologies, 2000. [Google Scholar]
- Council of Europe. European Pharmacopoeia, 7th ed.; Council of Europe: Strasbourg, France, 2011; Vol. 1, pp. 77–82. [Google Scholar]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing, 29th ed.; CLSI Supplement M100; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2019.
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 10th ed.; CLSI Document M07-A10; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2015.
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing, 28th ed.; CLSI Supplement M100; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018.
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 2001, 48, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Freshney, R.I. Culture of Animal Cells: A Manual of Basic Technique, 5th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Gibaldi, M.; Perrier, D. Pharmacokinetics, 2nd ed.; Informa Healthcare: New York, NY, USA, 2007. [Google Scholar] [CrossRef]
- Reed, L.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Ershov, F.I.; Tazulakhova, E.B.; Narovlyanskii, A.N.; Mironov, A.N.; Merkulov, V.A.; Leneva, I.A.; Ospelnikova, T.P.; Vasilev, A.N. Metodicheskie rekomendatsii po doklinicheskomu izucheniyu spetsificheskoi protivovirusnoi aktivnosti lekarstvennykh sredstv [Guidelines for the Preclinical Study of Specific Antiviral Activity of Medicinal Products]. In Rukovodstvo po provedeniyu doklinicheskikh issledovanii lekarstvennykh sredstv [Guidelines for Conducting Preclinical Studies of Medicinal Products]; Ministerstvo zdravookhraneniya RF: Moscow, Russia, 2012; pp. 525–549.
- Loginova, S.Ya.; Borisevich, S.V.; Semenova, I.V.; MaKCimov, V.A.; Bondarev, V.P.; Nebolsin, V.E. Izuchenie protivovirusnoi aktivnosti Ingavirina v otnoshenii vozbuditelya grippa A (H3N2) in vitro [Study of the Antiviral Activity of Ingavirin against Influenza A (H3N2) in Vitro]. Antibiot. Khimioter. 2009, 54, 19–22. [Google Scholar]
- Atkins, P.W.; Friedman, R.S. Solutions Manual for Molecular Quantum Mechanics; Oxford University Press: Oxford, UK, 1997. [Google Scholar]
- Levine, I.N. Quantum Chemistry, 5th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999. [Google Scholar]
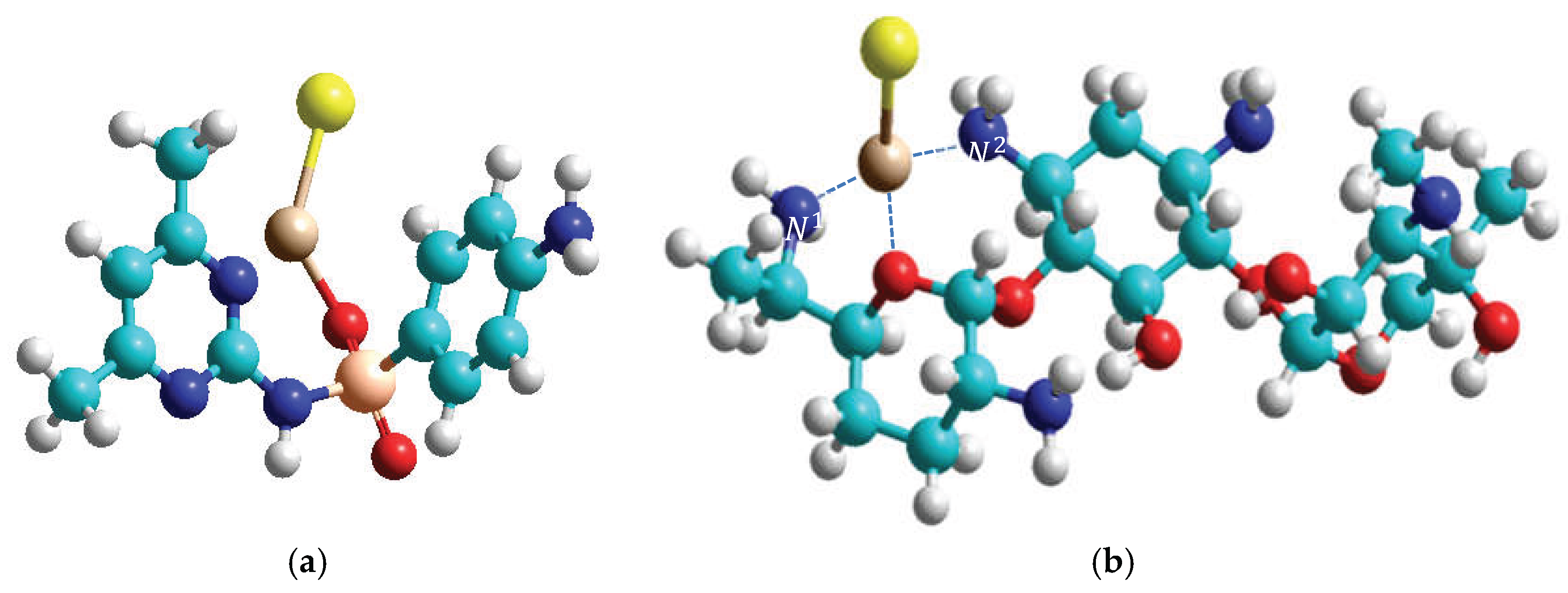
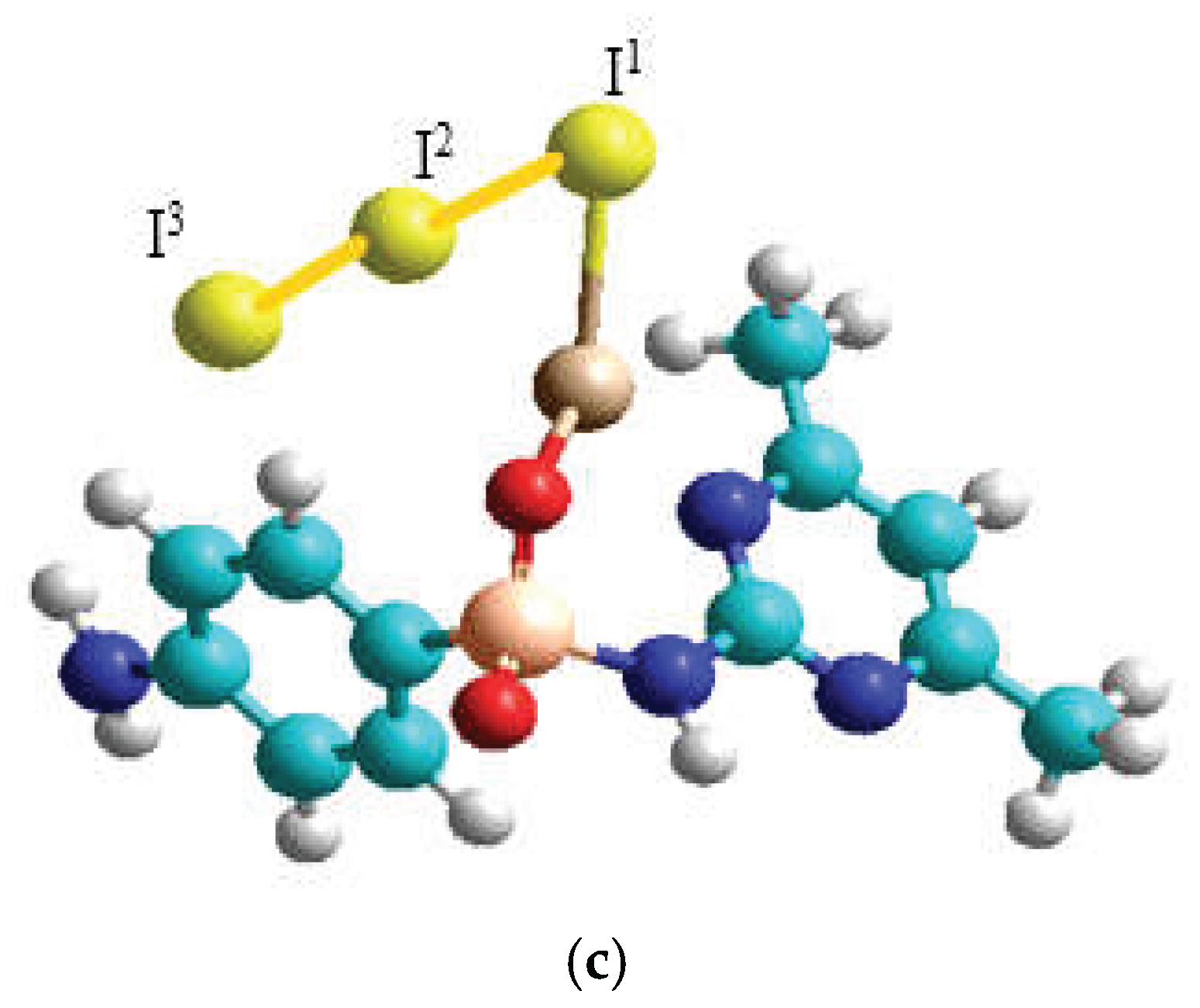
| Structure | -∆Е | Me-O | Me-N |
|---|---|---|---|
| A | 72.39 | 1.93 | 2.12 |
| B | 84.44 | 2.00 | 2.10 |
| Structure | -∆Е | -∆Е1 | Li-O | Li-I1 | Li-I2 | I1-I2 | I2-I3 |
|---|---|---|---|---|---|---|---|
| c | 27.23 | 20.48 | 1.92 | 2.62 | 2.96 | 3.07 | 2.91 |
| Compound name | Solubility | рН | Melting point, °С | Iodine, g/kg | Iodide, g/kg |
|---|---|---|---|---|---|
| Titration | |||||
| KC-246 | Water, DMSO, acetone 1g/10ml (soluble) |
2.35 ± 0.01 | 56-57 ±0.71 | 101.67 ± 0.57 | 266.68 ± 0.82 |
| KC-248 | Water 1g/5ml (freely soluble) |
2.35± 0.01 | 60-61 ± 1.0 | 100.78 ± 2.26 | 166.87 ± 1.29 |
| Compound name | λmax, nm | Amax | Transitions |
|---|---|---|---|
| KC-246 | 194.06 | 1.63 | σ → σ* |
| 225.57 | 1.16 | n → π* | |
| 262.40 | 0.53 | n → π* | |
| Sodium sulfadimidine | 194.06 | 1.73 | n → π* |
| 240.91 | 0.99 | n → π* | |
| 259.09 | 1.01 | n → π* or π → π* | |
| KC-248 | 194.18 | 1.91 | σ → σ* |
| 225.13 | 1.46 | π → π* | |
| 283.80 | 0.18 | n → π* | |
| Gentamicin sulfate | - | - | - |
| Concentrations, mg/mL | KC-246 M ± SD (n = 4) |
Concentrations, mg/mL | KC-248 M ± SD (n = 4) |
|---|---|---|---|
| 0.50 | 28.7 ± 2.6 | 5.00 | 18.0 ± 4.2 |
| 0.25 | 50.9 ± 2.3 | 2.50 | 18.7 ± 1.3 |
| 0.125 | 87.4 ± 7.9 | 1.25 | 22.7 ± 4.5 |
| 0.063 | 84.0 ± 8.8 | 0.62 | 21.8 ± 1.9 |
| 0.031 | 98.5 ± 8.6 | 0.31 | 22.2 ± 1.6 |
| 0.016 | 97.8 ± 10.7 | 0.15 | 56.7 ± 7.7 |
| 0.008 | 99.3 ± 3.4 | 0.08 | 63.9 ± 2.8 |
| 0.004 | 102.2 ± 10.9 | 0.04 | 78.9 ± 8.5 |
| 0.002 | 96.4 ± 14.7 | 0.02 | 122.5 ± 15.1 |
| CC50, mg/mL | 0.179 | CC50, mg/mL | 0.072 |
| Test strains | Compound name | |
|---|---|---|
| KC-246 | KC-248 | |
| Minimum bactericidal concentration (MBC) values, μg/mL, expressed as I2 | ||
| S.aureus ATCC 6538 | 0.198 | 0.787 |
| S.aureus ATCC 33591 | 0.397 | 0.393 |
| E.coli ATCC 8739 | 0.794 | 0.787 |
| E.coli ATCC 2523 | 0.794 | 0.787 |
| S.enterica ATCC 14028 | 0.794 | 3.149 |
| S.enterica ATCC 35988 | 0.397 | 0.787 |
| Kl.pneumoniae ATCC 10031 | 0.794 | 0.787 |
| Kl.pneumoniae ATCC 2524 | 0.794 | 0.393 |
| P.aeruginosa ATCC 9027 | 0.397 | 1.574 |
| P.aeruginosa ТА2 | 0.794 | 3.149 |
| Ent.hirae ATCC 10541 | 0.794 | 0.787 |
| Ent.faecalis ATCC 51575 | 0.397 | 0.787 |
| C.albicans ATCC 10231 | 0.794 | 3.149 |
| C.albicans SCAID PHPX 1-2019 | 0.794 | 3.149 |
| Compound name | Concentration, mg/mL | Ku, % |
|---|---|---|
| KC-246 | 0.365 | 100 |
| 0.183 | 100 | |
| 0.091 | 21.7 | |
| KC-248 | 1.275 | 100.0 |
| 0.638 | 66.7 | |
| 0.319 | 4.5 |
| Compound name | Concentration, mg/mL | Ku, % |
|---|---|---|
| KC-246 | 0.330 | 100 |
| 0.165 | 100 | |
| 0.083 | 33.3 | |
| KC-248 | 1.300 | 100 |
| 0.650 | 4.8 | |
| 0.325 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




