Submitted:
13 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
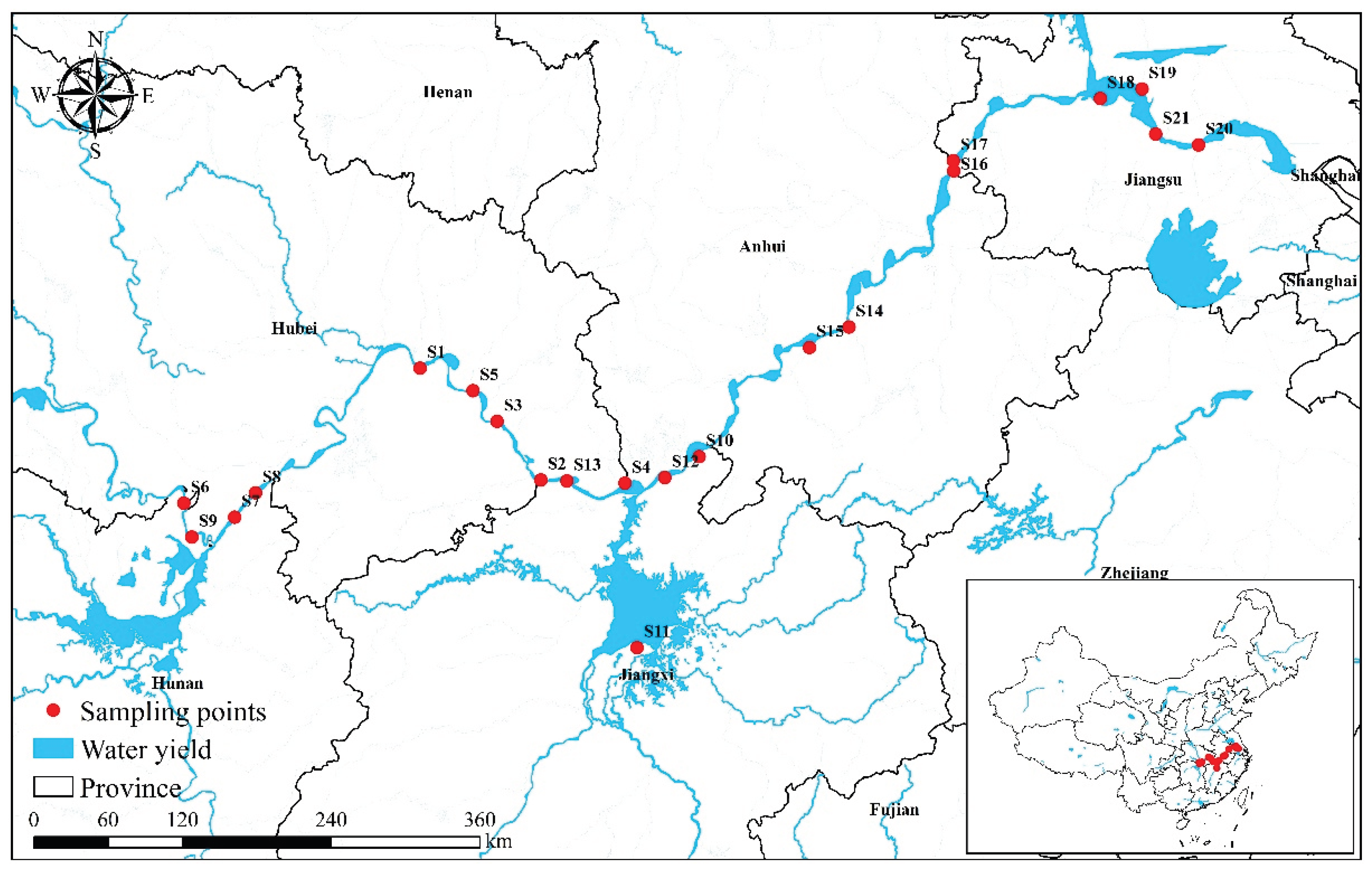
2.1. Study Area and Sampling Design
2.2. Sample Collection and Identification
2.2.1. Physicochemical Parameters
2.2.2. Benthic Macroinvertebrates
2.3. Data Analysis
2.3.1. Diversity Indices
2.3.2. Statistical Analysis
3. Results
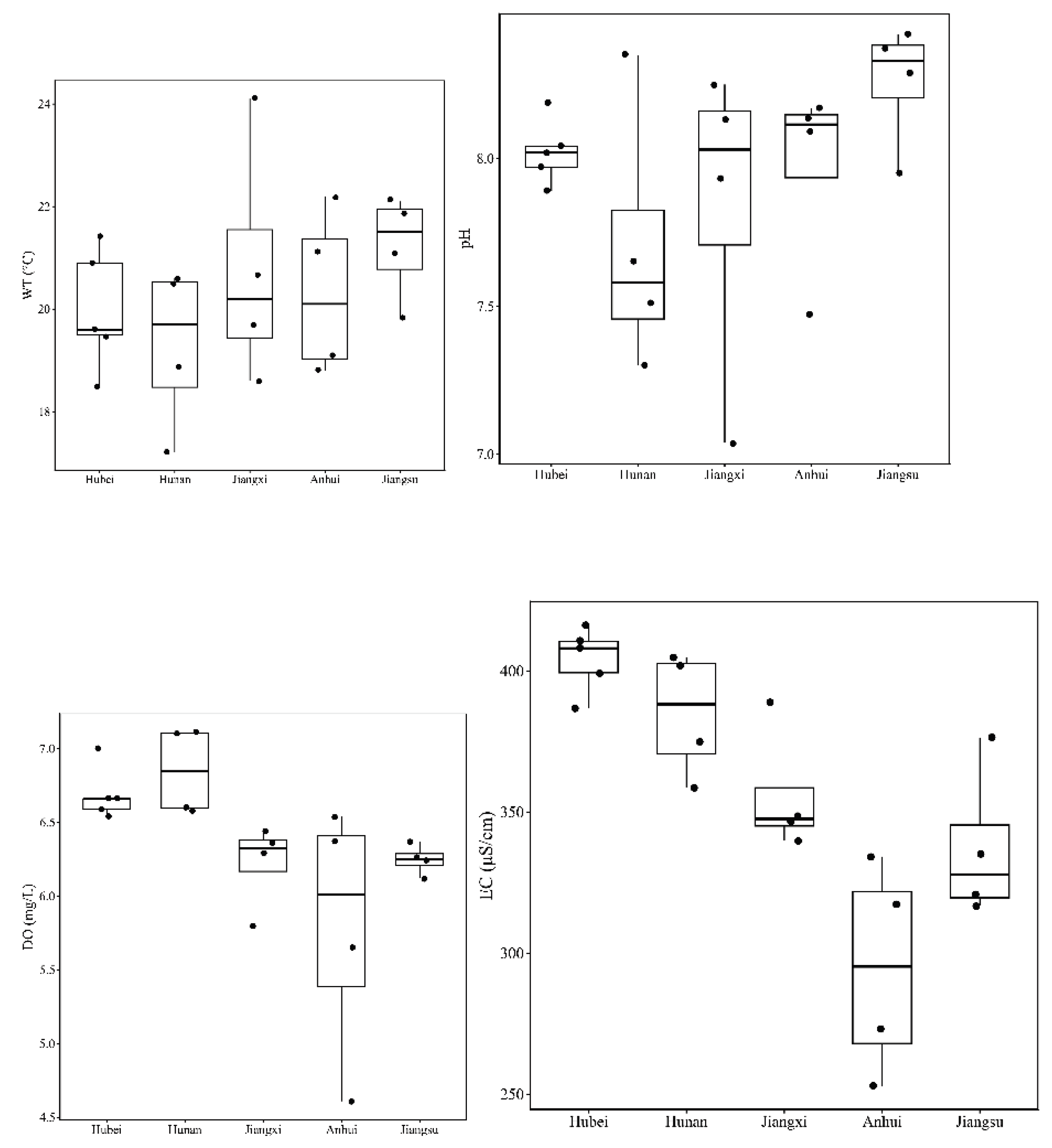
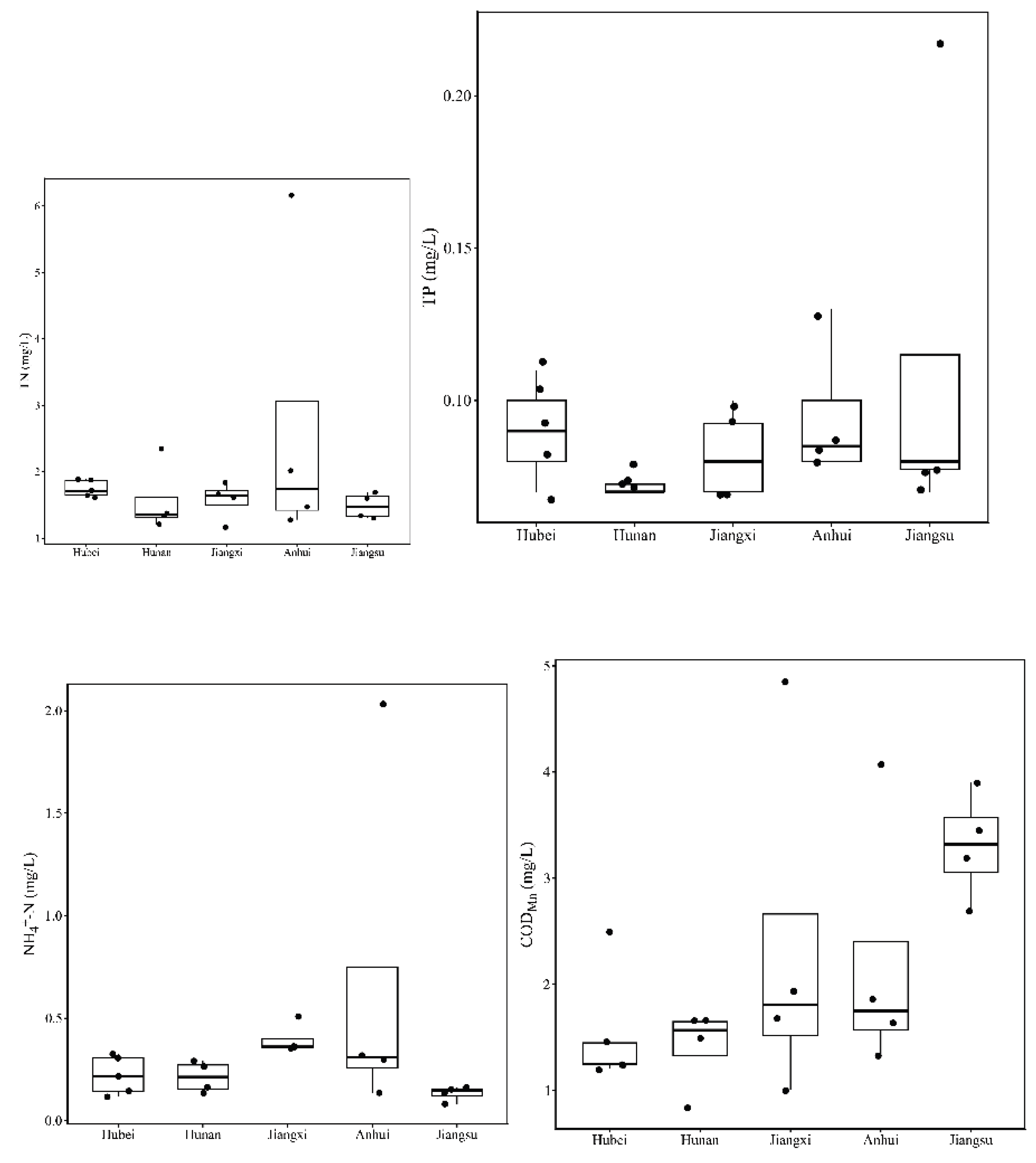
3.1. Spatial Variation in Physicochemical Parameters
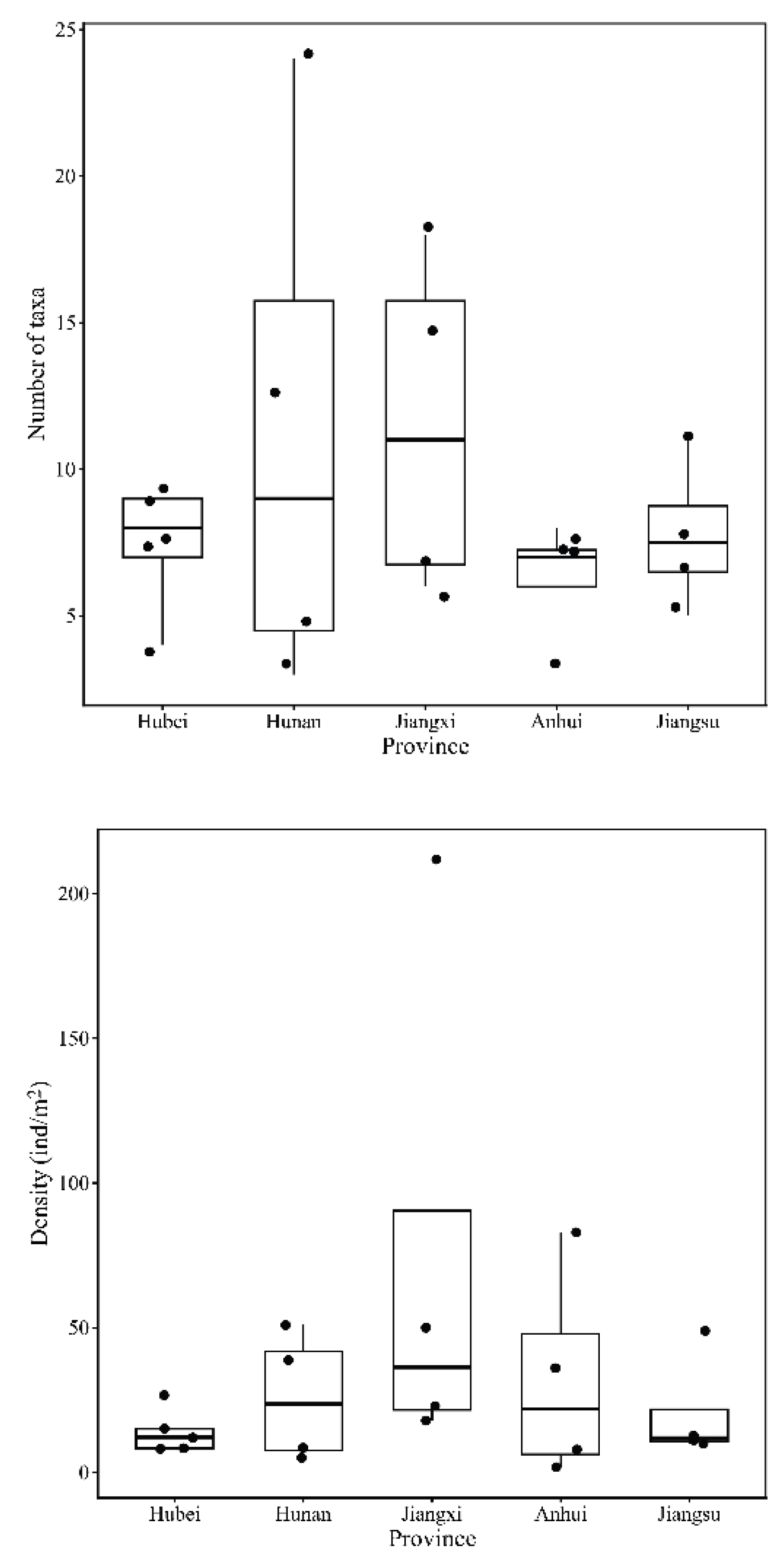
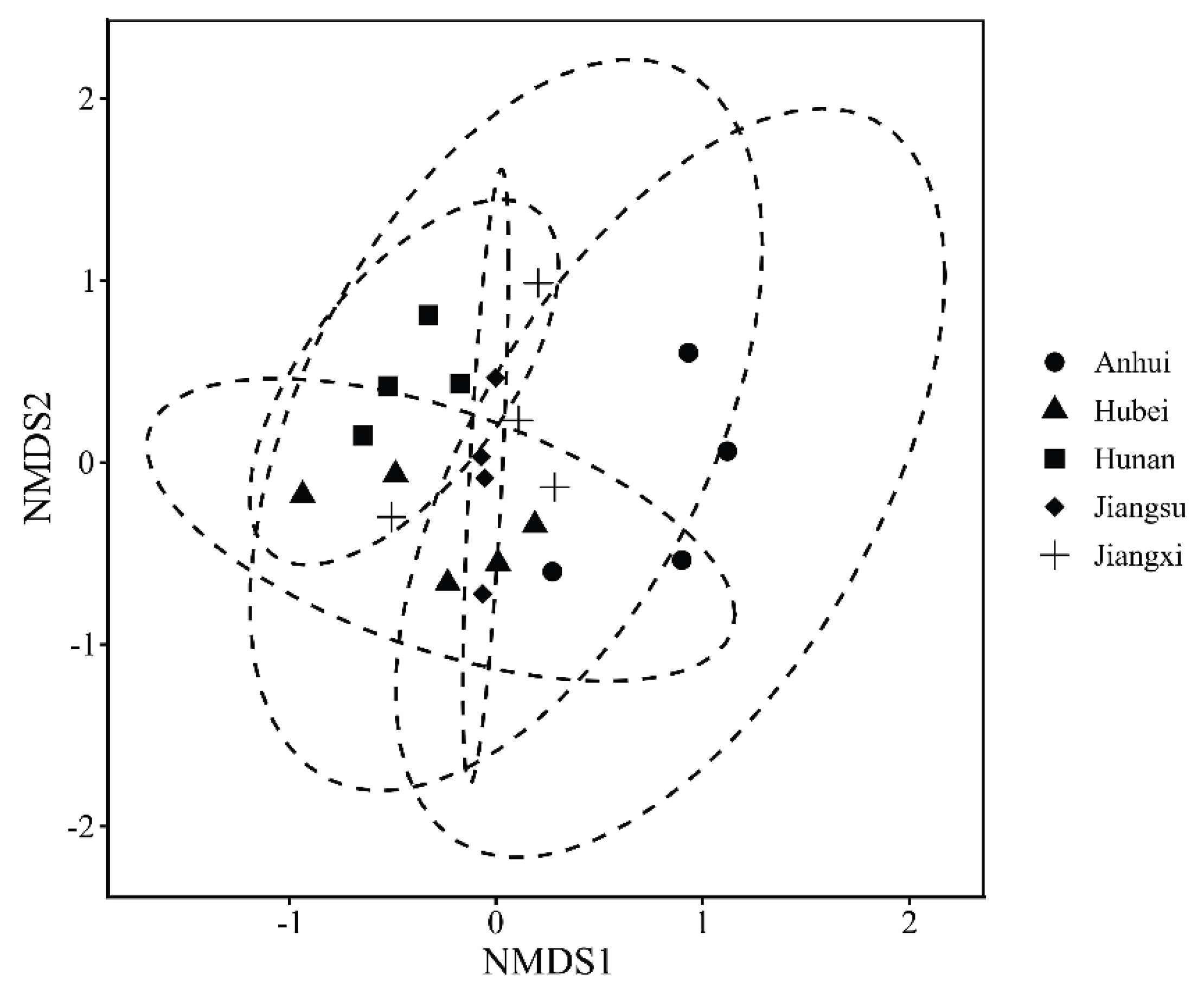
3.2. Community Composition and Structure of Benthic Macroinvertebrates
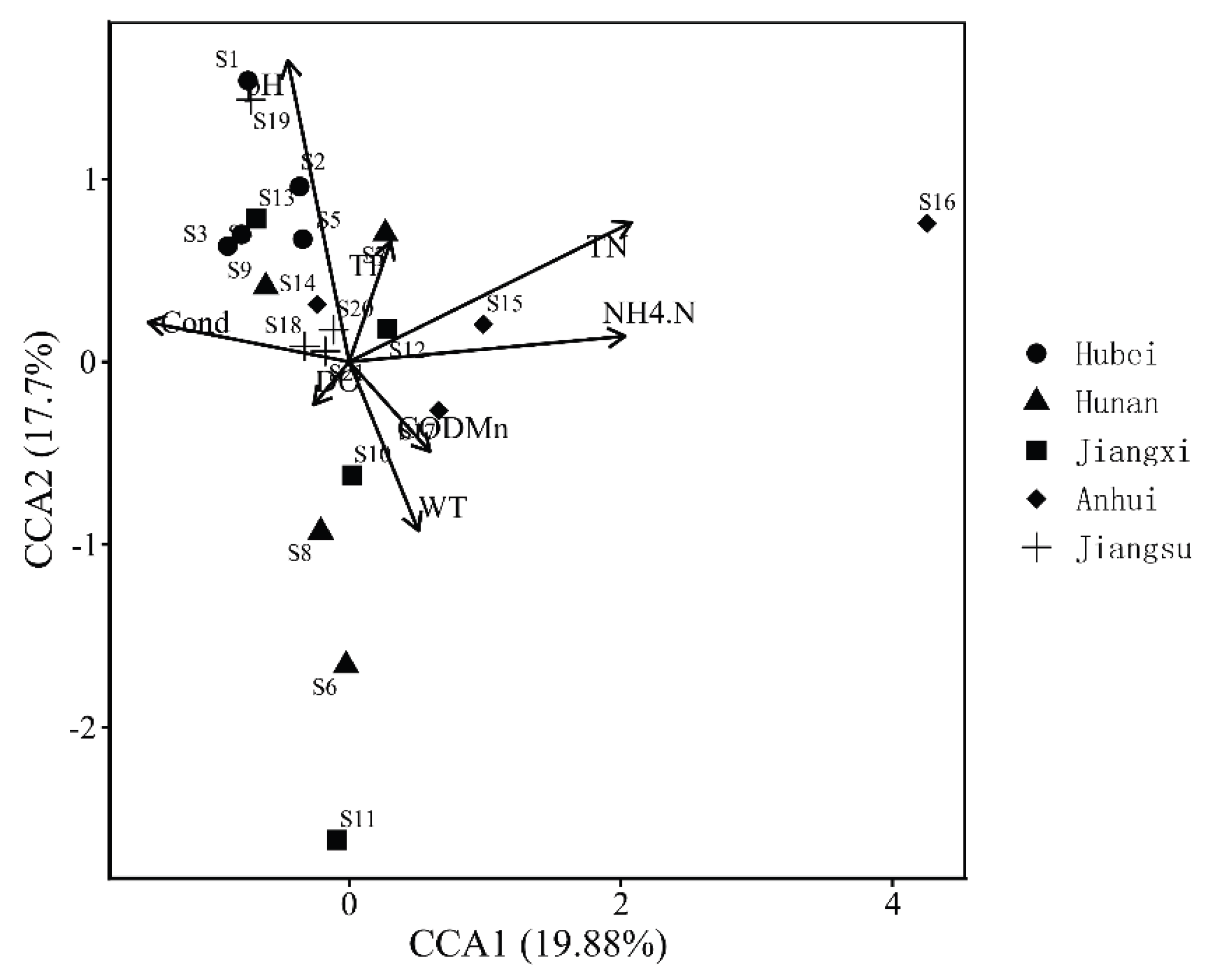
3.3. Relationships between Benthic Communities and Environmental Factors
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Wu, W.L.; Chen, W.X. Study on synergy and differential convergence of inclusive green development in the Yangtze River economic belt. Review of Economic Research 2022, (6), 103–118. [CrossRef]
- Ye, X.C.; Xu, C.Y.; Zhang, D.; et al. Variation of summer precipitation and its connection with Asian monsoon system in the middle-lower Yangtze River Basin. Sci. Geogr. Sin. 2018, 38, 1174–1182. [CrossRef]
- Liu, F.; Lin, P.C.; Li, M.Z.; et al. Situations and conservation strategies of fish resources in the Yangtze River Basin. Acta Hydrobiol. Sin. 2019, 43, 144–156.
- Wang, J.N.; Sun, H.L.; Xu, Y.X.; et al. Water eco-environment protection framework in the Yangtze River Basin during the 14th five-year plan period. Res. Environ. Sci. 2020, 33, 1075–1080. [CrossRef]
- Chen, J. Ecological redline identification in the Yangtze River Basin. Technol. Econ. Changjiang 2018, 2, 30–36. [CrossRef]
- Xie, P. Biodiversity crisis in the Yangtze River: The culprit was dams, followed by overfishing. J. Lake Sci. 2017, 29, 1279–1299. [CrossRef]
- Sun, L.T.; Zhao, F.; Zhang, T.; et al. Review on research progress of population resources and genetic diversity of Chinese sturgeon ( Acipenser sinensis Gray 1835 ). Fish. Inf. Strateg. 2017, 32, 112–117. [CrossRef]
- Yang, H.L.; Shen, L.; He, Y.F.; et al. Status of aquatic organisms resources and their environments in the Yangtze River system (2017—2021). J. Fish. China 2023, 47, 3–30. [CrossRef]
- Resh, V.H.; Jackson, J.K. Rapid assessment approaches to biomonitoring using benthic macroinvertebrates. In Freshwater Biomonitoring and Benthic Macroinvertebrates; Rosenberg, D.M., Resh, V.H., Eds.; Chapman & Hall: New York, NY, USA, 1993; pp. 195–233.
- Hu, Z.Y.; Bao, Y.X.; Cheng, H.Y.; et al. Research progress on ecology of natural wetland zoobenthos in China. Chin. J. Ecol. 2009, 28, 959–968. [CrossRef]
- Lamberti, G.A.; Gregory, S.V.; Ashkenas, L.R.; et al. Productive capacity of periphyton as a determinant of plant-herbivore interactions in streams. Ecology 1989, 70, 1840–1856. [CrossRef]
- Sundermann, A.; Stoll, S.; Haase, P. River restoration success depends on the species pool of the immediate surroundings. Ecol. Appl. 2011, 21, 1962–1971. [CrossRef]
- Sun, G.; Fang, Y.; Li, H.S. Ecology of benthic animals; Tsinghua University Press: Beijing, China, 2024.
- Mermillod-Blondin, F. The functional significance of bioturbation and biodeposition on biogeochemical processes at the water-sediment interface in freshwater and marine ecosystems. J. N. Am. Benthol. Soc. 2011, 30, 770-778. [CrossRef]
- Heino, J.; Louhi, P.; Muotka, T. Identifying the scales of variability in stream macroinvertebrate abundance, functional composition and assemblage structure. Freshw. Biol. 2004, 49, 1230–1239. [CrossRef]
- Ma, T.; Huang, Q.; Wang, H.; et al. Selection of benthic macroinvertebrate-based multimetrics and preliminary establishment of biocriteria for the bioassessment of the water quality of Taihu Lake, China. Acta Ecol. Sin. 2008, 28, 1192–1200. [CrossRef]
- Duan, X.H. Ecological impacts of water and sediment transportation on macroinvertebrate community in rivers. Ph.D. Thesis, Tsinghua University, Beijing, China, 2009.
- Xu, M.B.; Jiang, F.J.; Lai, J.X.; et al. Study on the ecological characteristics of benthic macrofauna in the Qinzhou bay of Cuangxi autonomous region and their correlation with sediment factors. Journal of Ocean Technology 2015, 34, 62–68.
- Real, M.; Prat, N. Factors influencing the distribution of chironomids and oligochaetes in profundal areas of Spanish reservoirs. Neth. J. Aquat. Ecol. 1992, 26, 405-410. [CrossRef]
- Chi, S.Y.; Zhao, X.F.; Gao, S.B.; et al. The spatial distribution pattern of autumn macroinvertebrates in relation to environmental factors in Danjiangkou Reservoir. Acta Ecol. Sin. 2021, 41, 1229–1241. [CrossRef]
- Marcus, W.B.; Lorin, K.H. A review of research on the development of lake indices of biotic integrity. Environ. Rev. 2009, 17, 21–44. https://www.jstor.org/stable/envirevi.17.21.
- Zhang, Y.H.; Qu, X.D.; Wang, S.M.; et al. River health assessment of hun river basin based on benthic index of biological integrity. Resour. Environ. Yangtze Basin 2020, 29, 1374–1386.
- Jin, X.W.; Wang, Y.Y.; Wang, B.X.; et al. Methods development for monitoring and assessment of ecological integrity of surface waters in China. Environ. Monit. China 2017, 33, 75–81. [CrossRef]
- Li, D.; Niu, Z.C.; Wang, X.; et al. Distribution and diversity of macrobenthic communities in the Jiangsu Reach of the Yangtze River. Chin. J. Appl. Environ. Biol. 2015, 21, 96–100. [CrossRef]
- Ma, Y.X.; Yao, W.L.; Yuan, S.B.; et al. Community structure and environmental analysis of macrozoobenthos in Yichang-Anqing reaches of the Yangtze mainstem. Acta Hydrobiol. Sin. 2019, 43, 634–642. [CrossRef]
- Sun, J.M.; Mei, Z.H.; Pi, Y.Z.; et al. Community characteristics and correlation of environmental factors of benthos in Nanjing section of Changjiang River. Environmental Monitoring and Forewarning 2023, 15, 23-29. [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.D.; et al. Rapid bioassessment protocols for use in streams and wadeable rivers: Periphyton, benthic macroinvertebrates, and fish, 2nd edition. USEPA: Washington, DC, USA, 1999.
- Liu, Y.Y. Economic Fauna of China: Freshwater Mollusks; Science Press: Beijing, China, 1979.
- Wang, Z.R. Fauna Sinica: Mollusca, Bivalvia, Mytiloida; Science Press: Beijing, China, 1997.
- Tang, H.Q. Biosystematic study on the chironomid larvae in China(Diptera: Chironomidae). Ph.D. Thesis, Nankai University, Tianjin, China, 2006.
- Wang, J.C.; Wang, X.H. Chironomid Larvae in Northern China; China Yan Shi Press: Beijing, China, 2011.
- Morse, J.C.; Yang, L.; Tian, L. Aquatic insects of China useful for monitoring water quality; Hohai University Press: Nanjing, China, 1984.
- Zhang, Y.H.; Zhang, M.; Peng, W.Q.; et al. Distribution pattern of macroinvertebrate community and its relationships with environmental factors in the Yongding River Basin. Chin. J. Appl. Ecol. 2020, 31, 3880-3888. [CrossRef]
- Zhao, R.; Zhao, X.M.; Gou, T.; et al. Spatial and temporal distributions of macroinvertebrate communities in the Beijiang River, Guangxi. Environ. Monit. China 2019, 35, 40-49. [CrossRef]
- Ma, K.; Shi, X.; You, X.G.; et al. Relationship of the heavy metals distribution in sediments to vegetation diversity and macroinvertebrate communities in riparian wetlands. Acta Ecol. Sin. 2021, 41, 2001-2010. [CrossRef]
- Bai, H.F. Spatio-temporal distribution characteristics and driving mechanism of aquatic communities and water ecosystem health assessment in the Weihe River Basin, Northwestern, China. Ph.D. Thesis, Northwest University, Xi’an, China, 2022.
- Chen, H.M.; Qu, X.D.; Wang, F. Research progress of river dynamic influences on the distribution of macroinvertebrates. Res. Environ. Sci. 2019, 32, 758-765. [CrossRef]
- Chen, L.; Zhang, L.H.; Huang, D.J.; et al. Water quality status and characteristics of aquatic community structure in Dahuofang Reservoir and its tributaries. Environ. Pollut. Control. 2021, 43, 834-838. [CrossRef]
- Wen, S.K.; Peng, K.; Gong, Z.J.; et al. Succession of macrozoobenthic communities and its drivers in Meiliang Bay of Lake Taihu during the past 40 year. J. Lake Sci. 2023, 35, 599-611. [CrossRef]
- Boltovskoy, D.; Sylvester, F.; Otaegui, A.; et al. Environmental modulation of reproductive activity of the invasive mussel Limnoperna fortunei: implications for antifouling strategies. Austral Ecol. 2009, 34, 719-730. [CrossRef]
- Zhang, D.P.; Liu, Y.; Bai, X.; et al. Species diversity and drivers of macroinvertebrate assemblages in the mainstream of Xijiang River. J. Hydroecol. 2025, 46, 171-183. [CrossRef]
- Heino, J.; Tolonen, K.T. Ecological drivers of multiple facets of beta diversity in a lentic macroinvertebrate metacommunity. Limnol. Oceanogr. 2017, 62, 2431-2444. [CrossRef]
- Mejía, G.A.; Su, C.; Allen, D.; et al. Land-use legacies affect the composition and distribution of tree species and their belowground functions in a succession from old-field to mature temperate forest. For. Ecosyst. 2024, 11, 960-973. [CrossRef]
- Zou, L.; Ma, H.Y.; Zhang, T.T.; et al. Macrozoobenthic community structure and niche analysis in the riparian zone of the mainstem of the middle and lower Yangtze River. Environ. Ecol. 2026, 8, 106-114. [CrossRef]
- Wang, Y.N.; Tian, Y.L.; Liu, Y.W.; et al. The relationship between macrobenthic community structure and environmental factors during dry and wet seasons in the Gongnaisi River, Xinjiang. Ecol. Sci. 2022, 41, 208-218. [CrossRef]
- Ru, H.J.; Zhang, Y.; Wu, X.X.; et al. Macrozoobenthos community structure and its relationship with environmental factors in the riparian zone of the lower Jinsha River. J. Fish. Sci. China 2022, 29, 1679-1692. [CrossRef]
- Zhang, X.T.; Li, W.M.; Zhang, K.; et al. Spatiotemporal distribution of macroinvertebrate functional feeding groups in Qiaobian River, a tributary of Yangtze River in Yichang. Acta Ecol. Sin. 2022, 42, 2559-2570. [CrossRef]
- Dou, Q.M.; Du, X.; Wang, L.; et al. Effects of land use and aquatic environmental factors on secondary productivity of macroinvertebrates : A case of Lake Lianhuan Group, northeast China. J.Lake Sci. 2024, 36, 846-857. [CrossRef]
- Tian, W.; Yang, Z.S.; Shao, K.Q.; et al. Effect of a comprehensive improvement project on water quality in urban lakes: A case study of water quality variation in Lihu Lake over the past 30 years. Environ. Sci. 2020, 41, 183-193. [CrossRef]
- Xu, H.; Paerl, H.W.; Qin, B.; et al. Nitrogen and phosphorus inputs control phytoplankton growth in eutrophic Lake Taihu, China. Limnol. Oceanogr. 2010, 55, 420-432. [CrossRef]
- Perga, M.E.; Desmet, M.; Enters, D.; et al. A century of bottom-up and top-down driven changes on a lake planktonic food web: A paleoecological and paleoisotopic study of Lake Annecy, France. Limnol. Oceanogr. 2010, 55, 803-816. [CrossRef]
- Liu, W. A study on carbon migration processes in reservoirs with different geological settings in subtropical areas, SW China. Ph.D. Thesis, Southwest University, Chongqing, China, 2015.
- Liu, J.T.; Li, Y.Z.; Gao, Z.J.; et al. Hydrochemistry and relationship between groundwater and surface water in the middle and lower reaches of Lhasa river basin. J. Shandong Univ. Sci. Technol. (Nat. Sci.) 2020, 39, 10-20. [CrossRef]
- Liu, C.; Liu, X.Y.; Zhou, J.C.; et al. Response of stream benthic algal diversity to urbanization: A case study in Shenzhen. Acta Ecol. Sin. 2022, 42, 10041-10050. [CrossRef]

| Physicochemical parameters | Hubei | Hunan | Jiangxi | Anhui | Jiangsu | P | |
|---|---|---|---|---|---|---|---|
| WT (°C) | Mean ± SD | 19.98 ± 1.16 | 19.30 ± 1.60 | 20.78 ± 2.38 | 20.33 ± 1.64 | 21.23 ± 1.04 | 0.51 |
| Min – Max | 18.50 - 21.40 | 17.20 - 20.60 | 18.60 - 24.10 | 18.80 - 22.20 | 19.80 - 22.10 | ||
| pH | Mean ± SD | 8.02 ± 0.11 | 7.70 ± 0.46 | 7.84 ± 0.55 | 7.97 ± 0.33 | 8.26 ± 0.21 | 0.29 |
| Min – Max | 7.89 - 8.19 | 7.30 - 8.35 | 7.04 – 8.25 | 7.47 - 8.17 | 7.95 - 8.42 | ||
| DO (mg/L) | Mean ± SD | 6.69 ± 0.18 | 6.85 ± 0.30 | 6.22 ± 0.29 | 5.79 ± 0.88 | 6.25 ± 0.10 | 0.02 |
| Min – Max | 6.54 - 7.0 | 6.58 - 7.11 | 5.80 - 6.44 | 4.61 - 6.54 | 6.12 - 6.37 | ||
| EC (μS/cm) | Mean ± SD | 404.24 ± 11.56 | 385.00 ± 22.22 | 356.11 ± 22.35 | 294.41 ± 37.92 | 337.22 ± 27.21 | < 0.001 |
| Min – Max | 386.72 - 416.52 | 358.59 - 404.87 | 339.99 - 389.19 | 252.73 - 334.24 | 316.87 - 376.28 | ||
| TN (mg/L) | Mean ± SD | 1.74 ± 0.13 | 1.57 ± 0.52 | 1.57 ± 0.29 | 2.74 ± 2.31 | 1.49 ± 0.19 | 0.43 |
| Min – Max | 1.61 - 1.89 | 1.21 - 2.34 | 1.17 - 1.84 | 1.28 - 6.17 | 1.30 - 1.69 | ||
| TP (mg/L) | Mean ± SD | 0.09 ± 0.02 | 0.08 ± 0.01 | 0.08 ± 0.02 | 0.09 ± 0.02 | 0.11 ± 0.07 | 0.59 |
| Min – Max | 0.07 - 0.11 | 0.07 - 0.08 | 0.07 - 0.10 | 0.08 - 0.13 | 0.07 - 0.22 | ||
| NH4+-N (mg/L) | Mean ± SD | 0.22 ± 0.09 | 0.21 ± 0.08 | 0.40 ± 0.08 | 0.70 ± 0.89 | 0.13 ± 0.04 | 0.30 |
| Min – Max | 0.12 - 0.32 | 0.13 - 0.29 | 0.35 - 0.51 | 0.14 - 2.03 | 0.08 - 0.16 | ||
| CODMn (mg/L) | Mean ± SD | 1.53 ± 0.55 | 1.41 ± 0.38 | 2.37 ± 1.71 | 2.22 ± 1.24 | 3.31 ± 0.51 | 0.09 |
| Min – Max | 1.21 - 2.49 | 0.84 - 1.65 | 1.01 - 4.86 | 1.33 - 4.06 | 2.69 - 3.90 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




