Submitted:
13 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
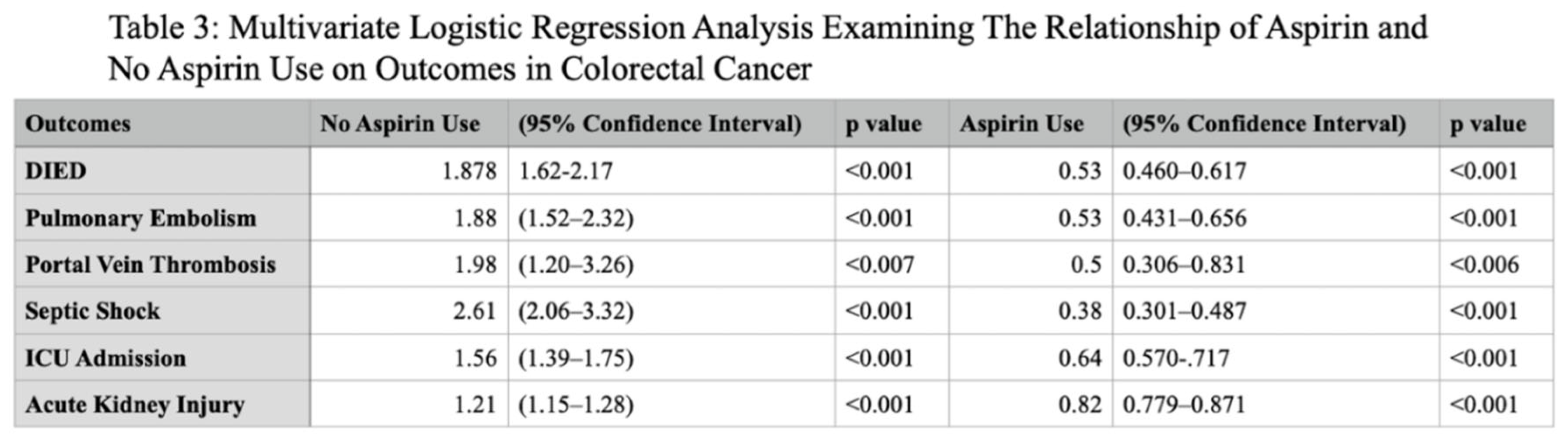
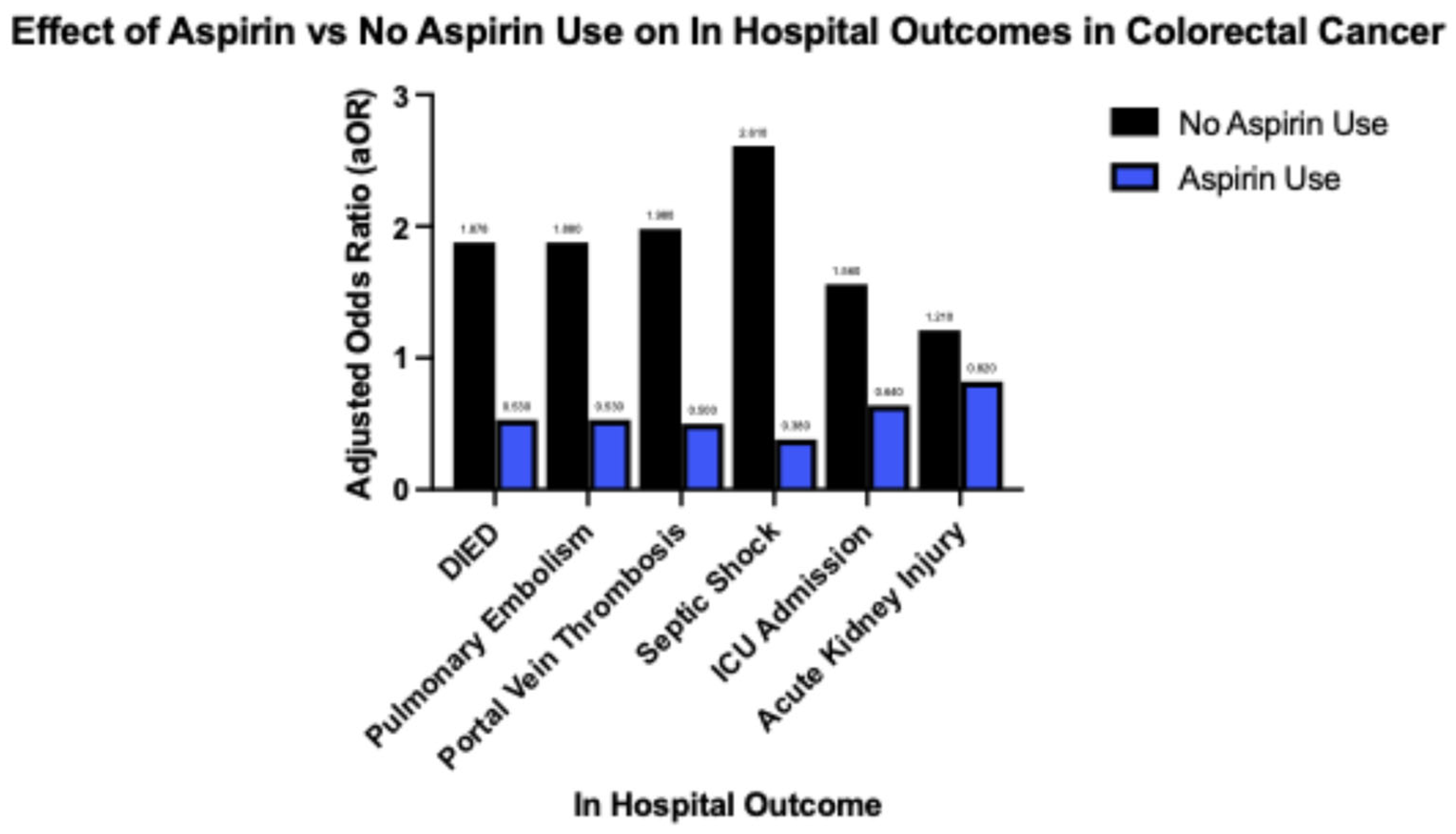
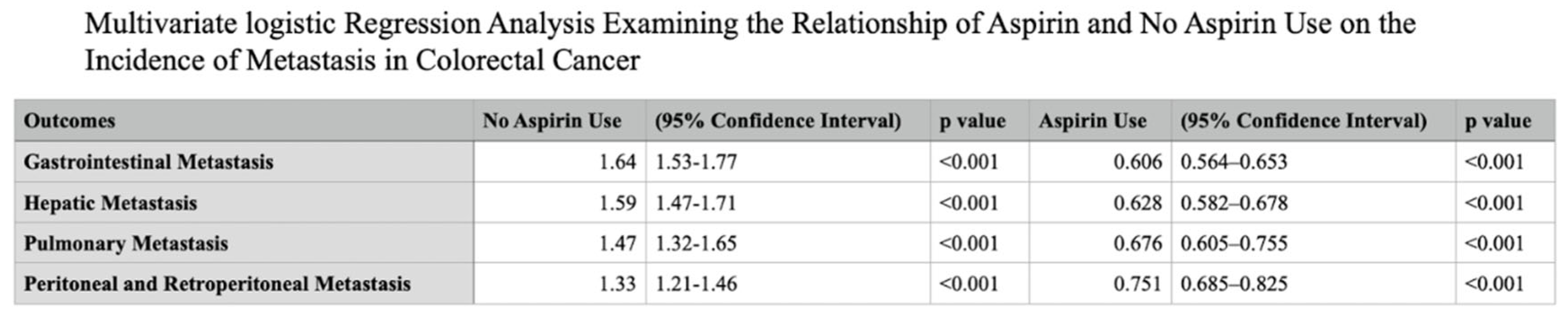
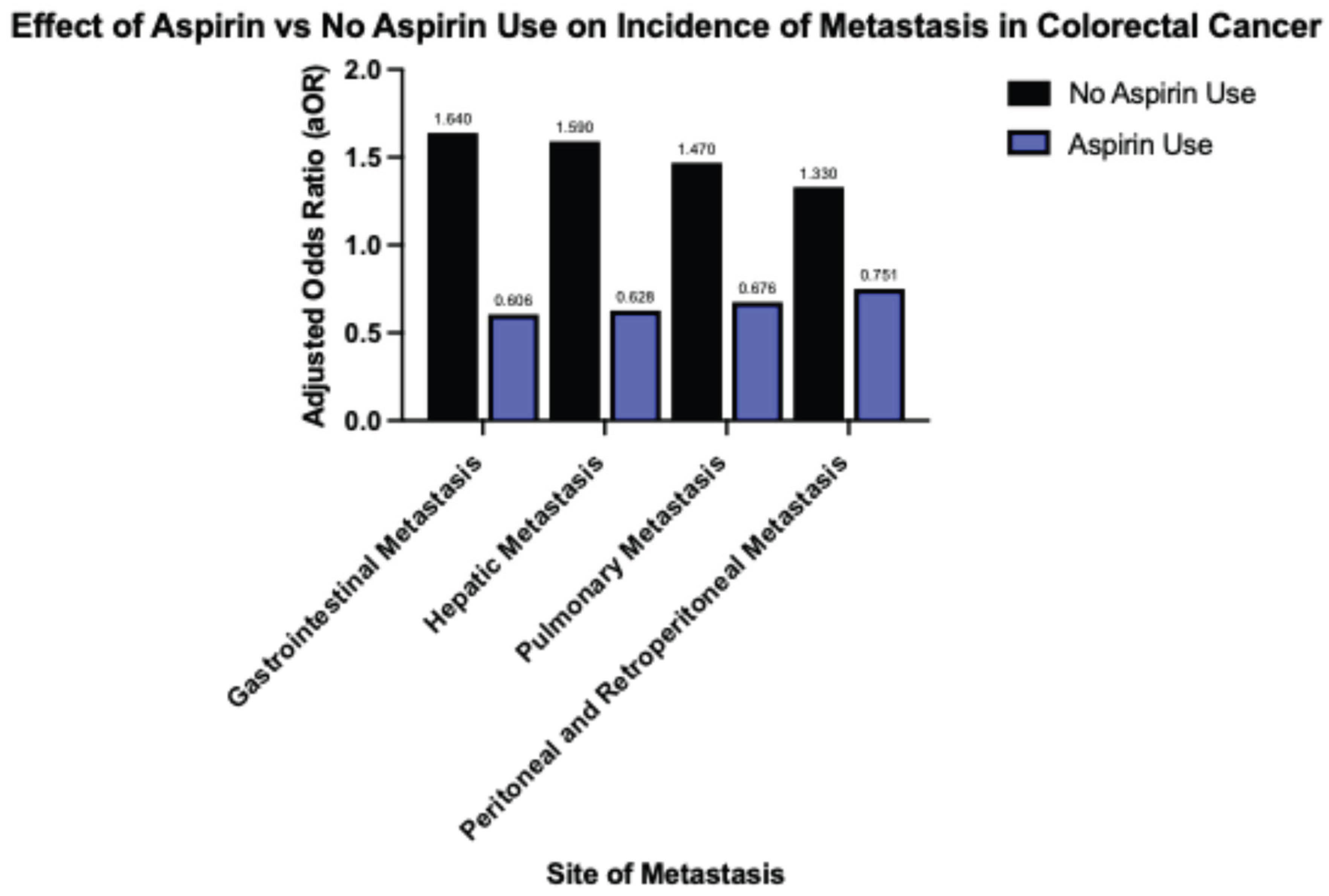
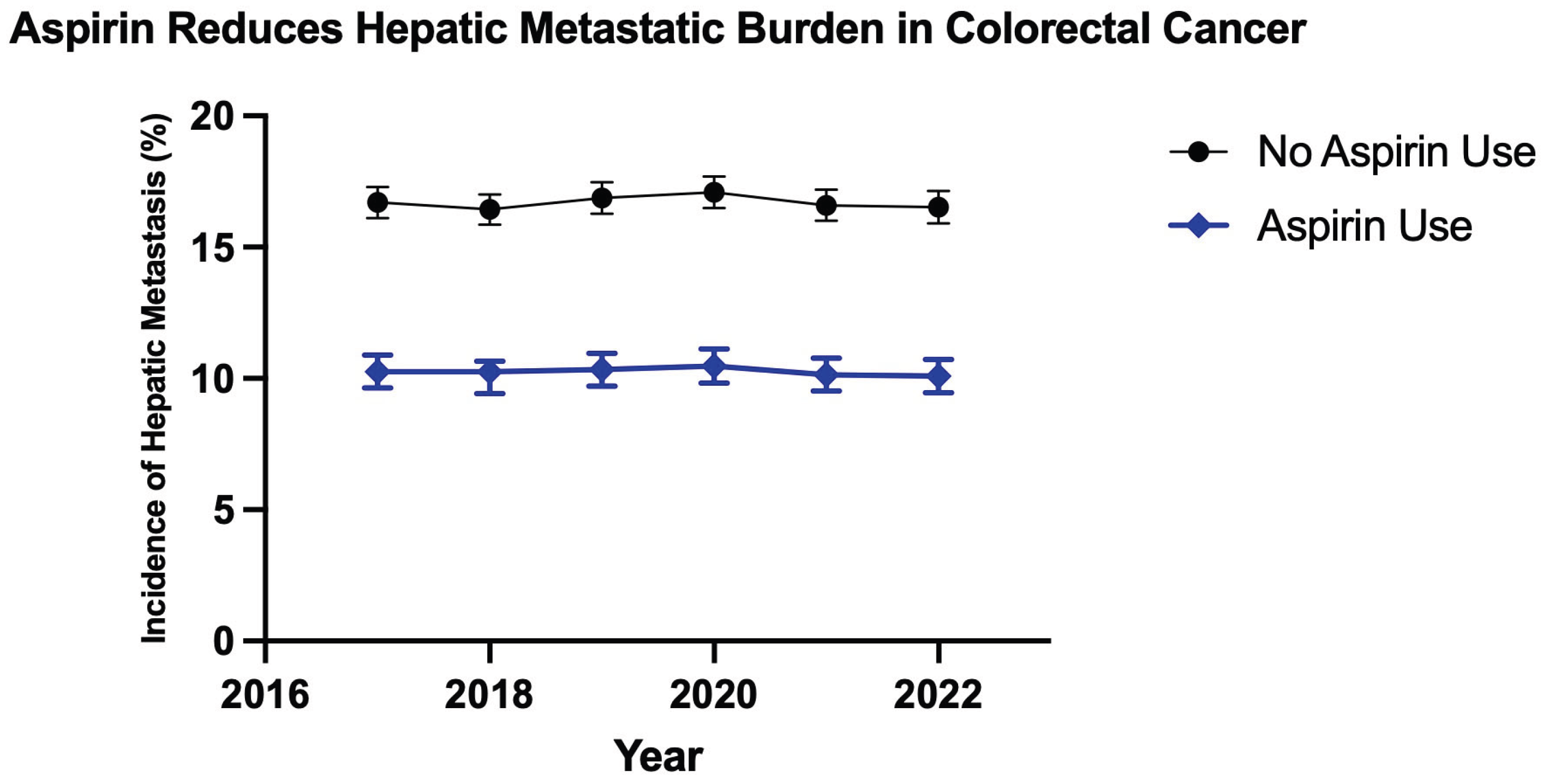
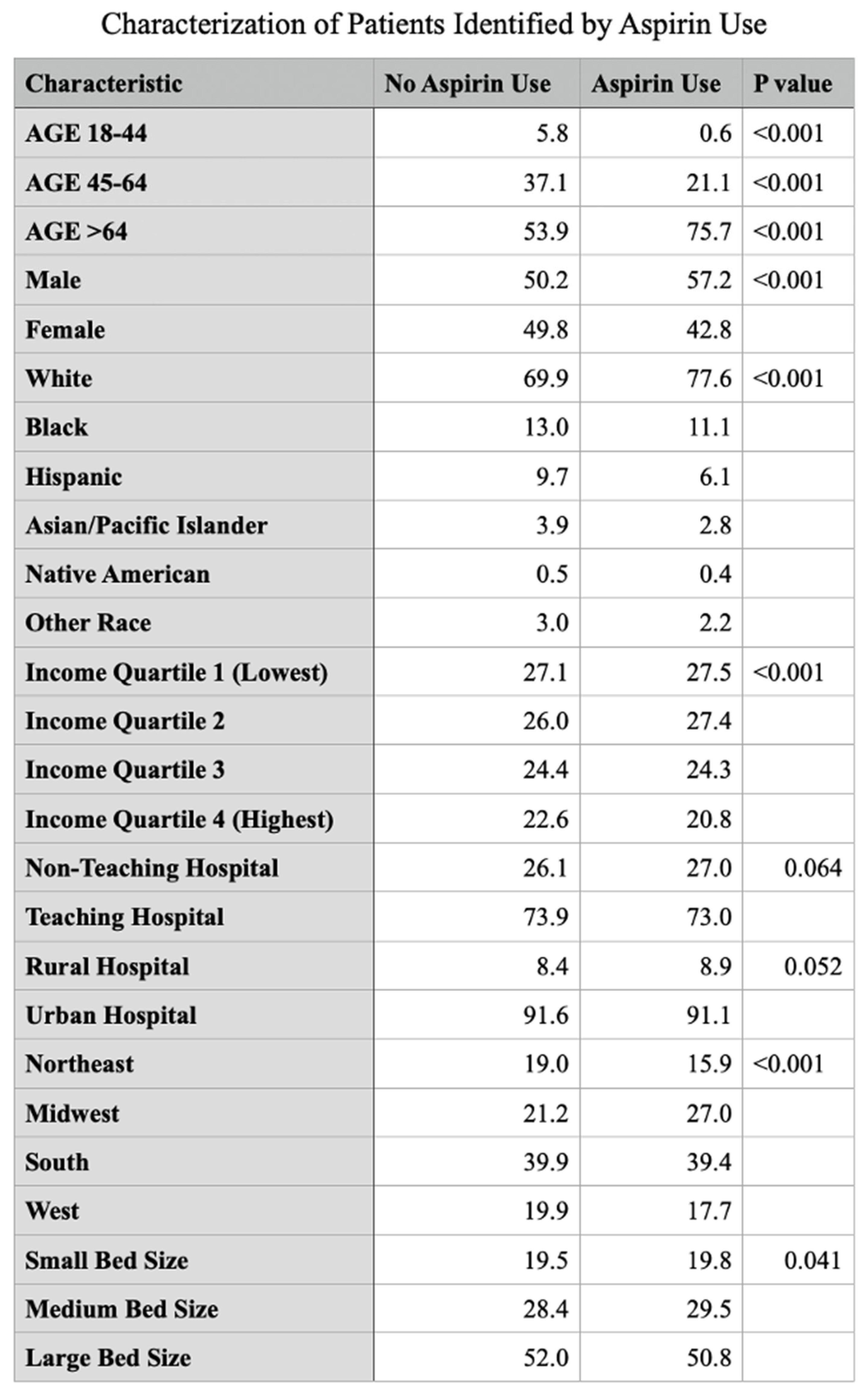
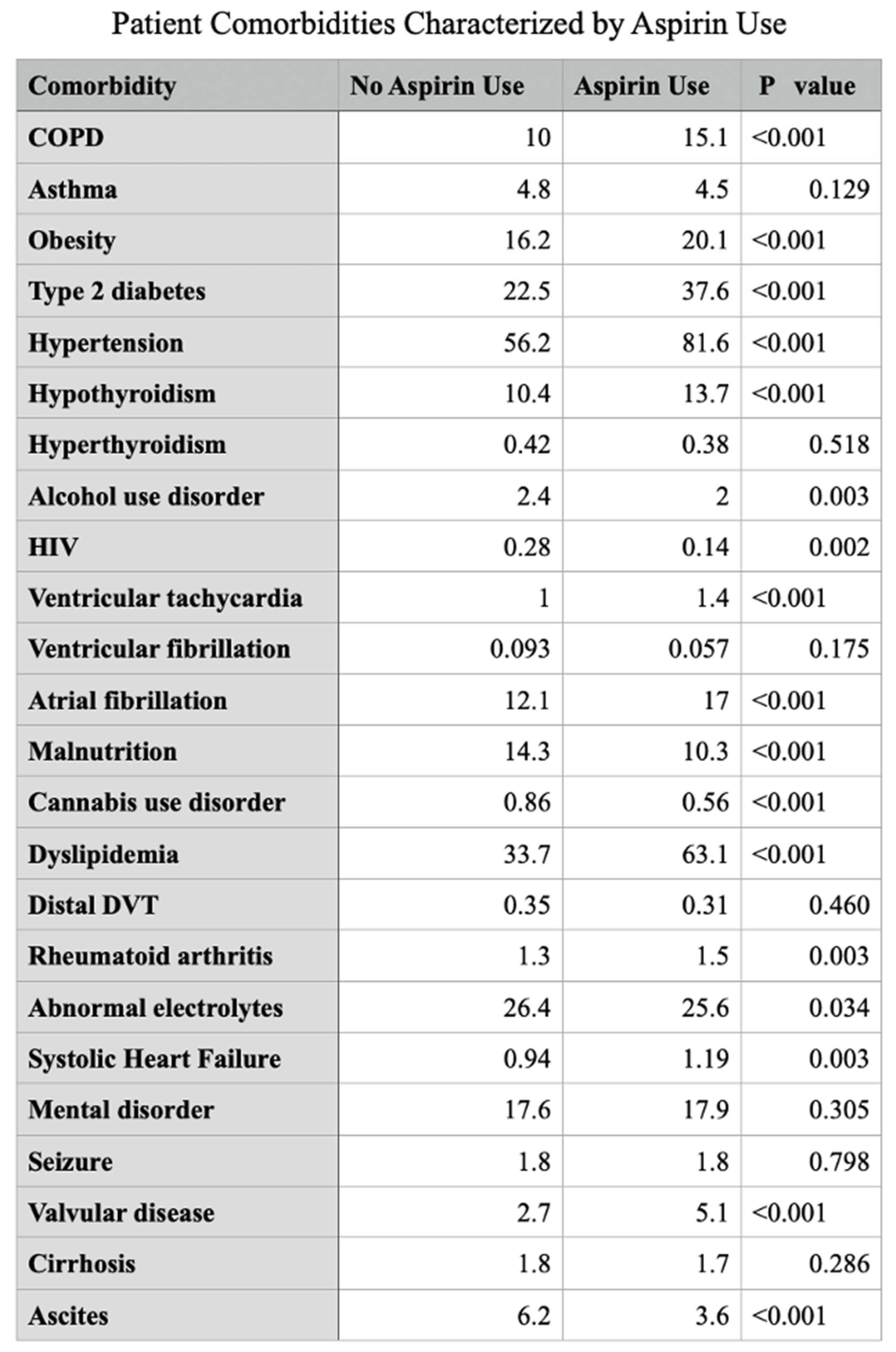
Introduction Aspirin initially recognized for its anti-inflammatory, antipyretic and analgesic properties hold a prominent role in the treatment of cardiovascular disease. The utility of aspirin in cancer therapeutics has been explored and stratified into COX dependent and independent mechanisms. COX2 gene expression has been demonstrated to be significantly upregulated in colorectal cancer and various other gastrointestinal malignancies including pancreatic, esophageal, and gastric cancer. This study investigates the relationship of aspirin use and outcomes in patients with colorectal cancer. Methodology The Nationwide Inpatient Sample (NIS) database from 2017 to 2022 was analyzed for patients age >18 who were hospitalized for colorectal cancer and its decompensations using ICD-10 diagnostic codes. These patients were further stratified based on the long term use of aspirin. The principal outcome of this investigation is in-hospital mortality, with secondary outcomes including rates of pulmonary embolism, portal vein thrombosis, acute kidney injury and need for renal replacement therapy, septic shock and rates of hepatic, pulmonary, gastrointestinal and peritoneal or retroperitoneal metastatic disease. Multivariate logistic regression accounting for hospital and patient characteristics was implemented for analysis, with the Charlson Comorbidity Index used to adjust for coexisting comorbidity burden; a p-value (p) of <0.05 was considered statistically significant. Results In our analysis of the NIS, 569,306 patients were identified with colorectal cancer and 11.7% (66,608) of this population were identified with long term use of aspirin. Aspirin use was identified to have a significantly reduced odds of in-patient mortality (adjusted odds ratio) [aOR] 0.530, p value <0.001 95% CI (confidence interval): 0.460 – 0.617. Patients with aspirin use also demonstrated significantly reduced odds of gastrointestinal, hepatic, pulmonary and retroperitoneal/peritoneal metastasis; (aOR 0.606, 95% CI: 0.564-0.653, P<0.001), (aOR 0.628, 95% CI: 0.582 – 0.678, P<0.001), (aOR 0.676, 95% CI: 0.605 – 0.755, P<0.001) and (aOR 0.751, 95% CI: 0.685 – 0.825, P<0.001) respectively. Conclusion In recent years there has been an alarming increase in incidence of colorectal cancer, particularly amongst younger individuals with increased associated mortality. This mortality increase, albeit alarming, is a driving force for treatment innovation with continual examination of our repertoire of medications for possible repurposed applications. COX2 mediated signaling serves as a key promotor of tumorigenic molecular signaling that directly contribute to tumor cell proliferation, angiogenesis and metastasis in colorectal cancer. Aspirin use and its inhibitory action on COX2 demonstrated a significantly reduced risk of in-hospital mortality. Aspirin use is also linked to a significant reduction in odds of developing metastatic disease to the liver, gastrointestinal system, lungs and peritoneum in patients with colorectal cancer. These findings reveal that aspirin exerts shielding effects against in-hospital mortality and protects patients with colorectal cancer from the development of major comorbid conditions and metastatic disease as compared to those who do not use aspirin.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
 |
|---|
 |
|---|
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miret Durazo, C.I.; Zachariah Saji, S.; Rawat, A.; Motiño Villanueva, A.L.; Bhandari, A.; Nurjanah, T.; Ryali, N.; Zepeda Martínez, I.G.; Cruz Santiago, J.A. Exploring Aspirin’s Potential in Cancer Prevention: A Comprehensive Review of the Current Evidence. Cureus 2024, 16, e70005. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Siegel, R. L.; Kratzer, T. B.; Giaquinto, A. N.; Sung, H.; Jemal, A. Cancer statistics, 2025. CA: A Cancer Journal for Clinicians 2025, 75, 10–45. [Google Scholar] [CrossRef]
- National Cancer Institute. Surveillance, epidemiology, and end results (SEER) program. Cancer Statistics, SEER Data & Software, Registry Operations 2018. [Google Scholar]
- Gurpinar, E.; Grizzle, W. E.; Piazza, G. A. COX-Independent Mechanisms of Cancer Chemoprevention by Anti-Inflammatory Drugs. Frontiers in Oncology 2013, 3, 55079. [Google Scholar] [CrossRef] [PubMed]
- Lim, S. C.; Lee, T. B.; Choi, C. H.; Ryu, S. Y.; Kim, K. J.; Min, Y. D. Expression of Cyclooxygenase-2 and its Relationship to p53 Accumulation in Colorectal Cancers. Yonsei Medical Journal 2007, 48, 495. [Google Scholar] [CrossRef]
- Pang, L. Y.; Hurst, E. A.; Argyle, D. J. Cyclooxygenase-2: A Role in Cancer Stem Cell Survival and Repopulation of Cancer Cells during Therapy. Stem Cells International 2016, 2016, 2048731. [Google Scholar] [CrossRef]
- Chan, A. T.; Ogino, S.; Fuchs, C. S. Aspirin Use and Survival After Diagnosis of Colorectal Cancer. JAMA: The Journal of the American Medical Association 2009, 302, 649. [Google Scholar] [CrossRef]
- Elwood, P. C.; Morgan, G.; Delon, C.; Protty, M.; Galante, J.; Pickering, J.; Watkins, J.; Weightman, A.; Morris, D. Aspirin and cancer survival: A systematic review and meta-analyses of 118 observational studies of aspirin and 18 cancers. Ecancermedicalscience 2021, 15, 1258. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wu, H.; Zhang, H.; et al. Aspirin use after diagnosis but not prediagnosis improves established colorectal cancer survival: a meta-analysis. Gut 2015, 64, 1419–1425. [Google Scholar] [CrossRef] [PubMed]
- Rothwell, P. M.; Wilson, M.; Elwin, C. E.; Norrving, B.; Algra, A.; Warlow, C. P.; Meade, T. W. Long-term effect of aspirin on colorectal cancer incidence and mortality: 20-year follow-up of five randomised trials. The Lancet 2010, 376, 1741–1750. [Google Scholar] [CrossRef]
- Chan, A.T.; Giovannucci, E.L.; Meyerhardt, J.A.; Schernhammer, E.S.; Curhan, G.C.; Fuchs, C.S. Aspirin Dose and Duration of Use and Risk of Colorectal Cancer in Men and Women. JAMA. 2005, 294, 914–923. [Google Scholar] [CrossRef]
- Albandar, H. J.; Markert, R.; Agrawal, S. The relationship between aspirin use and mortality in colorectal cancer. Journal of Gastrointestinal Oncology 2018, 9, 1133. [Google Scholar] [CrossRef] [PubMed]
- Negi, R. R.; Rana, S. V.; Gupta, V.; Gupta, R.; Chadha, V. D.; Prasad, K. K.; Dhawan, D. K. Over-Expression of Cyclooxygenase-2 in Colorectal Cancer Patients. Asian Pacific Journal of Cancer Prevention: APJCP 2019, 20, 1675. [Google Scholar] [CrossRef]
- Wu, Q. B.; Sun, G. P. Expression of COX-2 and HER-2 in colorectal cancer and their correlation. World Journal of Gastroenterology: WJG 2015, 21, 6206. [Google Scholar] [CrossRef]
- Roelofs, H. M.; Nagengast, F. M.; Peters, W. H. Over-expression of COX-2 mRNA in colorectal cancer. BMC Gastroenterology 2014, 14, 1. [Google Scholar] [CrossRef]
- Qu, D.; Shen, L.; Liu, S.; Li, H.; Ma, Y.; Zhang, R.; Wu, K.; Yao, L.; Li, J.; Zhang, J. Chronic inflammation confers to the metabolic reprogramming associated with tumorigenesis of colorectal cancer. Cancer Biology & Therapy 2017, 18, 237–244. [Google Scholar] [CrossRef]
- Fajardo, A.M.; Piazza, G.A. Chemoprevention in gastrointestinal physiology and disease. Anti-inflammatory approaches for colorectal cancer chemoprevention. Am J Physiol Gastrointest Liver Physiol. 2015, 309, G59–G70. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liebl, M.C.; Hofmann, T.G. The Role of p53 Signaling in Colorectal Cancer. Cancers (Basel) 2021, 13, 2125. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ai, G.; Dachineni, R.; Kumar, D.R.; Marimuthu, S.; Alfonso, L.F.; Bhat, G.J. Aspirin acetylates wild type and mutant p53 in colon cancer cells: identification of aspirin acetylated sites on recombinant p53. Tumour Biol. 2016, 37, 6007–6016. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Shen, X.; Chen, G.; Du, J. Drug Resistance in Colorectal Cancer: From Mechanism to Clinic. Cancers 2022, 14, 2928. [Google Scholar] [CrossRef]
- Ashique, S.; Bhowmick, M.; Pal, R.; Khatoon, H.; Kumar, P.; Sharma, H.; Garg, A.; Kumar, S.; Das, U. Multi drug resistance in Colorectal Cancer- approaches to overcome, advancements and future success. Advances in Cancer Biology—Metastasis 2024, 10, 100114. [Google Scholar] [CrossRef]
- Chen, L.; Yang, F.; Chen, S.; Tai, J. Mechanisms on chemotherapy resistance of colorectal cancer stem cells and research progress of reverse transformation: A mini-review. Frontiers in Medicine 2022, 9, 995882. [Google Scholar] [CrossRef] [PubMed]
- Ades, S.; Pulluri, B.; Holmes, C.E.; Lal, I.; Kumar, S.; Littenberg, B. Risk factors for venous thromboembolism in metastatic colorectal cancer with contemporary treatment: A SEER-Medicare analysis. Cancer Med 2022, 11, 1817–1826. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aonuma, A.O.; Nakamura, M.; Sakamaki, K.; Murai, T.; Matsuda, C.; Itaya, K.; Sone, T.; Yagisawa, M.; Koike, Y.; Endo, A.; Tsukuda, Y.; Ono, Y.; Nagasaka, A.; Nishikawa, S.; Yamanaka, T.; Sakamoto, N. Incidence of cancer-associated thromboembolism in Japanese gastric and colorectal cancer patients receiving chemotherapy: a single-institutional retrospective cohort analysis (Sapporo CAT study). BMJ Open 2019, 9, e028563. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rees, P.A.; Clouston, H.W.; Duff, S.; Kirwan, C.C. Colorectal cancer and thrombosis. Int J Colorectal Dis 2018, 33, 105–108. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rucker, D.; Dhamoon, A.S. Physiology, Thromboxane A2. [Updated 2022 Sep 12]; In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 2025; Available online: https://www.ncbi.nlm.nih.gov/books/NBK539817/.
- Patrono, C.; Rocca, B. Measurement of Thromboxane Biosynthesis in Health and Disease. Frontiers in Pharmacology 2019, 10, 476355. [Google Scholar] [CrossRef]
- Undas, A.; Undas, R.; Musiał, J.; Szczeklik, A. A low dose of aspirin (75 mg/day) lowers thrombin generation to a similar extent as a high dose of aspirin (300 mg/day). Blood Coagul Fibrinolysis 2000, 11, 231–234. [Google Scholar] [PubMed]
- Otani, K.; Ishihara, S.; Hata, K.; Murono, K.; Sasaki, K.; Yasuda, K.; Nishikawa, T.; Tanaka, T.; Kiyomatsu, T.; Kawai, K.; Nozawa, H.; Yamaguchi, H.; Watanabe, T. Colorectal cancer with venous tumor thrombosis. Asian Journal of Surgery 2018, 41, 197–202. [Google Scholar] [CrossRef]
- Shah, D. Diagnosis of portal vein tumor thrombosis in colorectal carcinoma in fluorodeoxyglucose positron emission tomography–computed tomography scan and its clinical implication. World Journal of Nuclear Medicine 2020, 19, 296. [Google Scholar] [CrossRef]
- Ginjupalli, M.; Hotwani, P.; Jayakumar, J.; et al. Portal vein thrombosis in gastrointestinal cancers: Insights into prevalence and prognosis—A nationwide inpatient analysis 2018–2020. J Clin Oncol. 2024, 42, e16382. [Google Scholar] [CrossRef]
- Xu, B.; Wang, Y.; Yang, J.; Zhang, Z.; Zhang, Y.; Du, H. Celecoxib induces apoptosis but up-regulates VEGF via endoplasmic reticulum stress in human colorectal cancer in vitro and in vivo. Cancer Chemother Pharmacol. 2016, 77, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Tsujii, M.; Kawano, S.; Tsuji, S.; Sawaoka, H.; Hori, M.; DuBois, R.N. Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell. 1998, 93, 705–716. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Teng, Y.; Zhang, R.; Luo, L. Antitumor effect of the selective COX-2 inhibitor celecoxib on endometrial adenocarcinoma in vitro and in vivo. Oncol Lett. 2012, 4, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Ranger, G.S. Current concepts in colorectal cancer prevention with cyclooxygenase inhibitors. Anticancer Res 2014, 34, 6277–6282. [Google Scholar] [PubMed]
- Wang, D.; DuBois, R. N. The Role of COX-2 in Intestinal Inflammation and Colorectal Cancer. Oncogene 2009, 29, 781. [Google Scholar] [CrossRef]
- Sheng, J.; Sun, H.; Yu, F. B.; Li, B.; Zhang, Y.; Zhu, Y. T. The Role of Cyclooxygenase-2 in Colorectal cancer. International Journal of Medical Sciences 2020, 17, 1095. [Google Scholar] [CrossRef]
- Chen, L.; Yang, F.; Chen, S.; Tai, J. Mechanisms on chemotherapy resistance of colorectal cancer stem cells and research progress of reverse transformation: A mini-review. Frontiers in Medicine 2022, 9, 995882. [Google Scholar] [CrossRef]
- Voutilainen, S.; Heikkilä, P.; Sampo, M.; Nevanlinna, H.; Blomqvist, C.; Mattson, J. Expression of markers of stem cell characteristics, epithelial-mesenchymal transition, basal-like phenotype, proliferation, and androgen receptor in metaplastic breast cancer and their prognostic impact. Acta Oncologica 2021, 60, 1233–1239. [Google Scholar] [CrossRef]
- Tang, D.; Yang, Z.; Long, F.; Luo, L.; Yang, B.; Zhu, R.; Sang, X.; Cao, G.; Wang, K. Long noncoding RNA MALAT1 mediates stem cell-like properties in human colorectal cancer cells by regulating miR-20b-5p/Oct4 axis. Journal of Cellular Physiology 2019, 234, 20816–20828. [Google Scholar] [CrossRef]
- Algra, A.M.; Rothwell, P.M. Effects of regular aspirin on long-term cancer incidence and metastasis: a systematic comparison of evidence from observational studies versus randomised trials. Lancet Oncol 2012, 13, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yamashita-Kanemaru, Y.; Morris, B. I.; Contursi, A.; Trajkovski, D.; Xu, J.; Patrascan, I.; Benson, J.; Evans, A. C.; Conti, A. G.; et al. Aspirin prevents metastasis by limiting platelet TXA2 suppression of T cell immunity. Nature 2025, 640, 1052–1061. [Google Scholar] [CrossRef] [PubMed]
- El Omari, N.; El Fessikh, M.; Aboulaghras, S.; Bakrim, S.; Khalid, A.; Abdalla, A. N.; Goh, K. W.; Bouyahya, A. The role of inflammation in colorectal Cancer and the preventive potential of natural compounds. Journal of Functional Foods 2025, 129, 106857. [Google Scholar] [CrossRef]
- Li, Q.; Geng, S.; Luo, H.; Wang, W.; Mo, Y. Q.; Luo, Q.; Wang, L.; Song, G. B.; Sheng, J. P.; Xu, B. Signaling pathways involved in colorectal cancer: Pathogenesis and targeted therapy. Signal Transduction and Targeted Therapy 2024, 9, 266. [Google Scholar] [CrossRef]
- Li, J.; Huang, L.; Zhao, H.; Yan, Y.; Lu, J. The Role of Interleukins in Colorectal Cancer. International Journal of Biological Sciences 2020, 16, 2323. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, C.; Wang, S.; et al. Wnt5a-induced M2 polarization of tumor-associated macrophages via IL-10 promotes colorectal cancer progression. Cell Commun Signal 2020, 18, 51. [Google Scholar] [CrossRef] [PubMed]
- Townsend, M. H.; Felsted, A. M.; Piccolo, S. R.; Robison, R. A. Metastatic colon adenocarcinoma has a significantly elevated expression of IL-10 compared with primary colon adenocarcinoma tumors. Cancer Biology & Therapy 2018, 19, 913. [Google Scholar] [CrossRef]
- Waldner, M.J.; Neurath, M.F. TGFβ and the Tumor Microenvironment in Colorectal Cancer. Cells 2023, 12, 1139. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, W.; Zhang, N.; Jin, C.; Long, M.D.; Rajabi, H.; Yasumizu, Y.; Fushimi, A.; Yamashita, N.; Hagiwara, M.; Zheng, R.; Wang, J.; Kui, L.; Singh, H.; Kharbanda, S.; Hu, Q.; Liu, S.; Kufe, D. MUC1-C drives stemness in progression of colitis to colorectal cancer. JCI Insight 2020, 5, e137112. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pothuraju, R.; Rachagani, S.; Krishn, S.R.; et al. Molecular implications of MUC5AC-CD44 axis in colorectal cancer progression and chemoresistance. Mol Cancer 2020, 19, 37. [Google Scholar] [CrossRef]
- Betge, J.; Schneider, N.I.; Harbaum, L.; Pollheimer, M.J.; Lindtner, R.A.; Kornprat, P.; Ebert, M.P.; Langner, C. MUC1, MUC2, MUC5AC, and MUC6 in colorectal cancer: expression profiles and clinical significance. Virchows Arch 2016, 469, 255–265. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zheng, Y.; Chen, Y.; Yu, K.; Yang, Y.; Wang, X.; Yang, X.; Qian, J.; Liu, X.; Wu, B. Fatal Infections Among Cancer Patients: A Population-Based Study in the United States. Infectious Diseases and Therapy 2021, 10, 871. [Google Scholar] [CrossRef] [PubMed]
- Homsi, J.; Walsh, D.; Panta, R.; et al. Infectious complications of advanced cancer. Support Care Cancer 2000, 8, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Andresen, K.; Carreira, H.; Strongman, H.; McDonald, H.I.; Benitez-Majano, S.; Mansfield, K.E.; Nitsch, D.; Tomlinson, L.A.; Bhaskaran, K. The risk of acute kidney injury in colorectal cancer survivors: an english population-based matched cohort study. BMC Cancer 2023, 23, 839. [Google Scholar] [CrossRef]
- Cosmai, L.; Porta, C.; Foramitti, M.; Perrone, V.; Mollica, L.; Gallieni, M.; Capasso, G. Preventive strategies for acute kidney injury in cancer patients. Clinical Kidney Journal 2020, 14, 70. [Google Scholar] [CrossRef]
- Rosner, M. H.; Perazella, M. A. Acute kidney injury in the patient with cancer. Kidney Research and Clinical Practice 2019, 38, 295. [Google Scholar] [CrossRef]
- Jesus Pereira, I.; Santos, M.; Sganzerla, D.; Robinson, C. C.; De Souza, D.; Kochhann, R.; Falavigna, M.; Azevedo, L.; Bozza, F.; Sharshar, T.; Goulart Rosa, R.; Granja, C.; Teixeira, C. Long term cognitive dysfunction among critical care survivors: Associated factors and quality of life—A multicenter cohort study. Annals of Intensive Care 2024, 14, 1–13. [Google Scholar] [CrossRef]
- Yao, L.; Li, Y.; Yin, R.; Yang, L.; Ding, N.; Li, B.; Shen, X.; Zhang, Z. Incidence and influencing factors of post-intensive care cognitive impairment. Intensive and Critical Care Nursing 2021, 67, 103106. [Google Scholar] [CrossRef]
- Pandharipande, P.P.; Girard, T.D.; Jackson, J.C.; Morandi, A.; Thompson, J.L.; Pun, B.T.; Brummel, N.E.; Hughes, C.G.; Vasilevskis, E.E.; Shintani, A.K.; Moons, K.G.; Geevarghese, S.K.; Canonico, A.; Hopkins, R.O.; Bernard, G.R.; Dittus, R.S.; Ely, E.W. BRAIN-ICU Study Investigators. Long-term cognitive impairment after critical illness. N Engl J Med. 2013, 369, 1306–1316. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Inoue, S.; Nakanishi, N.; Amaya, F.; Fujinami, Y.; Hatakeyama, J.; Hifumi, T.; Iida, Y.; Kawakami, D.; Kawai, Y.; Kondo, Y.; et al. Post-intensive care syndrome: Recent advances and future directions. Acute Medicine & Surgery 2024, 11, e929. [Google Scholar] [CrossRef]
 |
|---|
 |
|---|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




