Submitted:
13 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Evolution of PVS Animal Models
2.1. Large Animal Models: Pioneering Contributions and Limitations
2.2. Rodent Models: A Paradigm Shift
2.2.1. Neonatal Rat PVS Models
2.2.2. PH-LHD Rat Models
2.3. Genetic Models and Future Directions
3. Therapeutic Targets Identified Through Animal Models
3.1. mTOR Signaling and Sirolimus
3.2. Renin-Angiotensin System and Losartan
3.3. PDGF and VEGF Pathways
3.4. Emerging Targets: FAP and YAP/β-Catenin
3.5. Iron Metabolism and Ferroptosis: Lessons from PVOD
4. Integrating Mechanistic Insights: A Multi-Pathway Framework
5. Choosing the Right Model for the Right Question
6. Translating Insights from Animals to Patients
6.1. Successes and Limitations of Translation
6.2. Towards Precision Medicine in PVS
6.3. Future Directions
7. Conclusion
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chinese Society of Cardiology; Chinese Medical Association; Editorial Board of Chinese Journal of Cardiology. Chinese expert consensus on the diagnosis and treatment of pulmonary vein stenosis. Chinese Journal of Cardiology 2023, 51, 930–943. [Google Scholar]
- Simard, T.; Sarma, D.; Miranda, W.R.; Jain, C.C.; Anderson, J.H.; Collins, J.D.; El Sabbagh, A.; Jhand, A.; Peikert, T.; Reeder, G.S.; et al. Pathogenesis, Evaluation, and Management of Pulmonary Vein Stenosis. JACC 2023, 81, 2361–2373. [Google Scholar] [CrossRef]
- Takajo, D.; Critser, P.J.; Cash, M.; Magness, M.; Hirsch, R. Mortality Patterns in Pediatric Pulmonary Vein Stenosis: Insights Into Right Ventricular Systolic Pressure Associations. J. Am. Heart Assoc. 2025, 14, e037908. [Google Scholar] [CrossRef]
- Backes, C.H.; Nealon, E.; Armstrong, A.K.; Cua, C.L.; Mitchell, C.; Krishnan, U.; Vanderlaan, R.D.; Song, M.K.; Viola, N.; Smith, C.V.; et al. Pulmonary Vein Stenosis in Infants: A Systematic Review, Meta-Analysis, and Meta-Regression. J. Pediatr. 2018, 198, 36–45.e3. [Google Scholar] [CrossRef] [PubMed]
- Kalustian, A.B.; Ji, P.; Wadhwa, L.; Caldarone, C.A.; Bansal, M.; Qureshi, A.M.; Heinle, J.S.; Birla, R.K. Insights into the Molecular Mechanism of Pulmonary Vein Stenosis in Pediatric Patients with Congenital Heart Disease. JTCVS Open 2025, 26, 166–181. [Google Scholar] [CrossRef]
- Nasr, V.G.; Callahan, R.; Wichner, Z.; Odegard, K.C.; DiNardo, J.A. Intraluminal Pulmonary Vein Stenosis in Children: A “New” Lesion. Anesth. Analg. 2019, 129, 27–40. [Google Scholar] [CrossRef]
- Prieto, L.R. In Search of the Holy Grail for Pediatric Pulmonary Vein Stenosis. J Am Coll Cardiol.. 2021, 77, 2819–2821. [Google Scholar] [CrossRef]
- Jenkins, K.J. Evaluation of Treatment Effect of Sirolimus on Pediatric Pulmonary Vein Stenosis Using a Neonatal Rat Model Induced by Bilateral Pulmonary Vein Banding. JACC Basic Transl. Sci. 2025, 10, 101296. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Marshall, A.C.; Gauvreau, K.; Peng, L.F.; Nugent, A.W.; Lock, J.E.; McElhinney, D.B. Outcomes after Stent Implantation for the Treatment of Congenital and Postoperative Pulmonary Vein Stenosis in Children. Circ. Cardiovasc. Interv. 2012, 5, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Kovach, A.E.; Magcalas, P.M.; Ireland, C.; McEnany, K.; Oliveira, A.M.; Kieran, M.W.; Baird, C.W.; Jenkins, K.; Vargas, S.O. Paucicellular Fibrointimal Proliferation Characterizes Pediatric Pulmonary Vein Stenosis: Clinicopathologic Analysis of 213 Samples From 97 Patients. Am. J. Surg. Pathol. 2017, 41, 1198–1204. [Google Scholar] [CrossRef] [PubMed]
- Suntharos, P.; Satawiriya, M.; Prieto, L.R. Pulmonary Vein Stenosis: Future Optimism. Curr. Opin. Cardiol. 2025, 40, 298–304. [Google Scholar] [CrossRef]
- Almakadma, A.H.; Sarma, D.; Hassett, L.; Miranda, W.; Alkhouli, M.; Reeder, G.S.; Munger, T.M.; Packer, D.L.; Simard, T.; Holmes, D.R. Pulmonary Vein Stenosis-Balloon Angioplasty Versus Stenting: A Systematic Review and Meta-Analysis. JACC Clin. Electrophysiol. 2022, 8, 1323–1333. [Google Scholar] [CrossRef]
- Mansour, M.; Gerstenfeld, E.P.; Patel, C.; Natale, A.; Whang, W.; Cuoco, F.A.; Mountantonakis, S.E.; Gibson, D.N.; Harding, J.D.; Holland, S.K.; et al. Pulmonary Vein Narrowing after Pulsed Field versus Thermal Ablation. Eur. Eur. Pacing Arrhythm. Card. Electrophysiol. J. Work. Groups Card. Pacing Arrhythm. Card. Cell. Electrophysiol. Eur. Soc. Cardiol. 2024, 26, euae038. [Google Scholar] [CrossRef]
- Cory, M.J.; Ooi, Y.K.; Kelleman, M.S.; Vincent, R.N.; Kim, D.W.; Petit, C.J. Reintervention Is Associated With Improved Survival in Pediatric Patients With Pulmonary Vein Stenosis. JACC Cardiovasc. Interv. 2017, 10, 1788–1798. [Google Scholar] [CrossRef]
- Riedlinger, W.F.J.; Juraszek, A.L.; Jenkins, K.J.; Nugent, A.W.; Balasubramanian, S.; Calicchio, M.L.; Kieran, M.W.; Collins, T. Pulmonary Vein Stenosis: Expression of Receptor Tyrosine Kinases by Lesional Cells. Cardiovasc. Pathol. Off. J. Soc. Cardiovasc. Pathol. 2006, 15, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Sadr, I.M.; Tan, P.E.; Kieran, M.W.; Jenkins, K.J. Mechanism of Pulmonary Vein Stenosis in Infants with Normally Connected Veins. Am. J. Cardiol. 2000, 86, 577–579, A10. [Google Scholar] [CrossRef]
- Kato, H.; Fu, Y.Y.; Zhu, J.; Wang, L.; Aafaqi, S.; Rahkonen, O.; Slorach, C.; Traister, A.; Leung, C.H.; Chiasson, D.; et al. Pulmonary Vein Stenosis and the Pathophysiology of “Upstream” Pulmonary Veins. J. Thorac. Cardiovasc. Surg. 2014, 148, 245–253. [Google Scholar] [CrossRef]
- LaBourene, J.I.; Coles, J.G.; Johnson, D.J.; Mehra, A.; Keeley, F.W.; Rabinovitch, M. Alterations in Elastin and Collagen Related to the Mechanism of Progressive Pulmonary Venous Obstruction in a Piglet Model. A Hemodynamic, Ultrastructural, and Biochemical Study. Circ. Res. 1990, 66, 438–456. [Google Scholar] [CrossRef]
- Sun, C.C.; Doyle, T.; Ringel, R.E. Pulmonary Vein Stenosis. Hum. Pathol. 1995, 26, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Crick, S.J.; Sheppard, M.N.; Ho, S.Y.; Gebstein, L.; Anderson, R.H. Anatomy of the Pig Heart: Comparisons with Normal Human Cardiac Structure. J. Anat. 1998, 193, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, G.C.; Devine, W.; Redel, B.K.; Whitworth, K.M.; Samuel, M.; Spate, L.D.; Cecil, R.F.; Prather, R.S.; Wu, Y.; Wells, K.D.; et al. Cardiovascular Development and Congenital Heart Disease Modeling in the Pig. J. Am. Heart Assoc. 2021, 10, e021631. [Google Scholar] [CrossRef]
- Zhu, J.; Ide, H.; Fu, Y.Y.; Teichert, A.-M.; Kato, H.; Weisel, R.D.; Maynes, J.T.; Coles, J.G.; Caldarone, C.A. Losartan Ameliorates “Upstream” Pulmonary Vein Vasculopathy in a Piglet Model of Pulmonary Vein Stenosis. J. Thorac. Cardiovasc. Surg. 2014, 148, 2550–2557. [Google Scholar] [CrossRef] [PubMed]
- Masaki, N.; Adachi, O.; Katahira, S.; Saiki, Y.; Horii, A.; Kawamoto, S.; Saiki, Y. Progression of Vascular Remodeling in Pulmonary Vein Obstruction. J. Thorac. Cardiovasc. Surg. 2020, 160, 777–790.e5. [Google Scholar] [CrossRef]
- Li, D.; Xiao, Y.; Ruan, P.; Wan, Z.; Hu, Y.; Sun, S.; Wang, Z.; Zheng, S.; Yan, Y.; Chen, H.; et al. Evaluation of Treatment Effect of Sirolimus on Pediatric Pulmonary Vein Stenosis Using a Neonatal Rat Model. JACC Basic Transl. Sci. 2025, 10, 101229. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Gonzalez, I.; Mühleder, S.; Fernández-Chacón, M.; Benedito, R. Genetic Tools to Study Cardiovascular Biology. Front. Physiol. 2020, 11. [Google Scholar] [CrossRef]
- Jia, T.; Wang, C.; Han, Z.; Wang, X.; Ding, M.; Wang, Q. Experimental Rodent Models of Cardiovascular Diseases. Front. Cardiovasc. Med. 2020, 7, 588075. [Google Scholar] [CrossRef]
- Doetschman, T.; Azhar, M. Cardiac-Specific Inducible and Conditional Gene Targeting in Mice. Circ. Res. 2012, 110. [Google Scholar] [CrossRef]
- Li, D.; Qiu, L.; Hong, H.; Chen, H.; Zhao, P.; Xiao, Y.; Zhang, H.; Sun, Q.; Ye, L. A Neonatal Rat Model of Pulmonary Vein Stenosis. Cell Biosci. 2023, 13, 112. [Google Scholar] [CrossRef]
- Patel, J.D.; Briones, M.; Mandhani, M.; Jones, S.; Suthar, D.; Gray, R.; Pettus, J.; McCracken, C.; Thomas, A.; Petit, C.J. Systemic Sirolimus Therapy for Infants and Children With Pulmonary Vein Stenosis. J. Am. Coll. Cardiol. 2021, 77, 2807–2818. [Google Scholar] [CrossRef]
- Kalustian, A.B.; Hagan, J.L.; Brlecic, P.E.; Iacobas, I.; Vanderlaan, R.D.; Burns, J.; Wu, T.T.; Birla, R.; Gowda, S.; Bansal, M.; et al. Systemic Sirolimus Therapy Is Associated With Reduced Intervention Frequency in Pulmonary Vein Stenosis. JACC Adv. 2024, 3, 101401. [Google Scholar] [CrossRef] [PubMed]
- Callahan, R.; Esch, J.J.; Wang, G.; Ireland, C.M.; Gauvreau, K.; Jenkins, K.J. Systemic Sirolimus to Prevent In-Stent Stenosis in Pediatric Pulmonary Vein Stenosis. Pediatr. Cardiol. 2020, 41, 282–289. [Google Scholar] [CrossRef]
- Münks, J.; Yogeswaran, A.; Antoine, T.K.; Blumrich, L.A.; Dorfmüller, P.; Ghofrani, H.A.; Assmus, B.; Schermuly, R.T.; Sydykov, A. A Novel Rat Model of Mild Pulmonary Hypertension Associated with Pulmonary Venous Congestion Induced by Left Pulmonary Vein Banding. Int. J. Mol. Sci. 2024, 25, 2827. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yan, Y. Animal Models of Pulmonary Hypertension Due to Left Heart Disease. Anim. Models Exp. Med. 2022, 5, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Shentu, J.; Dai, W.; Chen, C.; Huang, J.; Chen, L.; Yan, Y.; Zhang, H.; Zhu, Z.; Shi, G.; Chen, H. A Novel Rat Model for Group 2 Pulmonary Hypertension by Total Pulmonary Vein Banding: Multi-Omics Insights into Pathophysiological Mechanisms. J. Thorac. Cardiovasc. Surg. 2026, S0022-5223(26)00085-1. [Google Scholar] [CrossRef]
- Li, D.; Hong, H.; Li, M.; Xu, X.; Wang, S.; Xiao, Y.; Zheng, S.; Wang, Z.; Yan, Y.; Chen, H.; et al. A Surgical Mouse Model of Neonatal Right Ventricular Outflow Tract Obstruction by Pulmonary Artery Banding. J. Heart Lung Transplant. 2024, 43, 496–507. [Google Scholar] [CrossRef]
- Sengupta, S.; Peterson, T.R.; Sabatini, D.M. Regulation of the mTOR Complex 1 Pathway by Nutrients, Growth Factors, and Stress. Mol. Cell 2010, 40, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Laplante, M.; Sabatini, D.M. mTOR Signaling in Growth Control and Disease. Cell 2012, 149, 274–293. [Google Scholar] [CrossRef]
- Saxton, R.A.; Sabatini, D.M. mTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960–976. [Google Scholar] [CrossRef]
- Marx, S.O.; Marks, A.R. Bench to Bedside: The Development of Rapamycin and Its Application to Stent Restenosis. Circulation 2001, 104, 852–855. [Google Scholar] [CrossRef]
- Panwar, V.; Singh, A.; Bhatt, M.; Tonk, R.K.; Azizov, S.; Raza, A.S.; Sengupta, S.; Kumar, D.; Garg, M. Multifaceted Role of mTOR (Mammalian Target of Rapamycin) Signaling Pathway in Human Health and Disease. Signal Transduct. Target. Ther. 2023, 8, 375. [Google Scholar] [CrossRef]
- Kuo, J.A.; Petit, C.J. Pulmonary Vein Stenosis in Children: A Programmatic Approach Employing Primary and Anatomic Therapy. Children 2021, 8. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Zou, L.-P.; Xu, K.-F.; Xu, W.-S.; Zhang, M.-N.; Lu, Q.; Tian, X.-L.; Pang, L.-Y.; He, W.; Wang, Q.-H.; et al. Long-Term Safety and Influence on Growth in Patients Receiving Sirolimus: A Pooled Analysis. Orphanet J. Rare Dis. 2024, 19, 299. [Google Scholar] [CrossRef]
- Wehming, M.; Krebs-Schmitt, D.; Briem-Richter, A.; Hegen, B.; Brinkert, F.; Fischer, L.; Grabhorn, E. Experience with the mTOR Inhibitor Everolimus in Pediatric Liver Graft Recipients. Children 2023, 10. [Google Scholar] [CrossRef]
- Ekholm, M.; Kahan, T. The Impact of the Renin-Angiotensin-Aldosterone System on Inflammation, Coagulation, and Atherothrombotic Complications, and to Aggravated COVID-19. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef]
- AlQudah, M.; Hale, T.M.; Czubryt, M.P. Targeting the Renin-Angiotensin-Aldosterone System in Fibrosis. Matrix Biol. J. Int. Soc. Matrix Biol. 2020, 91–92, 92–108. [Google Scholar] [CrossRef]
- Laghlam, D.; Jozwiak, M.; Nguyen, L.S. Renin–Angiotensin–Aldosterone System and Immunomodulation: A State-of-the-Art Review. Cells 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Forrester, S.J.; Booz, G.W.; Sigmund, C.D.; Coffman, T.M.; Kawai, T.; Rizzo, V.; Scalia, R.; Eguchi, S. Angiotensin II Signal Transduction: An Update on Mechanisms of Physiology and Pathophysiology. Physiol. Rev. 2018, 98, 1627–1738. [Google Scholar] [CrossRef] [PubMed]
- Habashi, J.P.; Judge, D.P.; Holm, T.M.; Cohn, R.D.; Loeys, B.L.; Cooper, T.K.; Myers, L.; Klein, E.C.; Liu, G.; Calvi, C.; et al. Losartan, an AT1 Antagonist, Prevents Aortic Aneurysm in a Mouse Model of Marfan Syndrome. Science 2006, 312, 117–121. [Google Scholar] [CrossRef]
- Vanderlaan, R.; Fu, Y.; Pan, J. Mechanistic Insights into the Pathophysiology of Pulmonary Vein Stenosis; Baltimore, MD, USA, 16 May 2016. [Google Scholar]
- Münzel, T.; Daiber, A. Vascular Redox Signaling, Endothelial Nitric Oxide Synthase Uncoupling, and Endothelial Dysfunction in the Setting of Transportation Noise Exposure or Chronic Treatment with Organic Nitrates. Antioxid. Redox Signal. 2023, 38, 1001–1021. [Google Scholar] [CrossRef]
- Vanderlaan, R.D.; Rome, J.; Hirsch, R.; Ivy, D.; Caldarone, C.A. Pulmonary Vein Stenosis: Treatment and Challenges. J. Thorac. Cardiovasc. Surg. 2021, 161, 2169–2176. [Google Scholar] [CrossRef] [PubMed]
- Shentu, J.; Zhang, M.; Xu, Z.; Wen, C.; Zhang, H.; Zhu, Z.; Chen, H.; Shi, G. Valsartan to Prevent Acquired Pulmonary Vein Stenosis in Pediatric Patients After Total Anomalous Pulmonary Venous Connection Surgery. J. Am. Heart Assoc. Cardiovasc. Cerebrovasc. Dis. 2025, 14, e036911. [Google Scholar] [CrossRef]
- Zeng, W.; Liu, S.; Xu, Z.; Ding, F.; Mei, J.; Zhu, J. Losartan Attenuates Upstream Vasculopathy in a Modified Piglet Model of Pulmonary Vein Stenosis: Contribution of the Hippo Pathway. Ann. Transl. Med. 2022, 10, 1153. [Google Scholar] [CrossRef]
- Dey, A.; Varelas, X.; Guan, K.-L. Targeting the Hippo Pathway in Cancer, Fibrosis, Wound Healing and Regenerative Medicine. Nat. Rev. Drug Discov. 2020, 19, 480–494. [Google Scholar] [CrossRef]
- Mia, M.M.; Singh, M.K. New Insights into Hippo/YAP Signaling in Fibrotic Diseases. Cells 2022, 11, 2065. [Google Scholar] [CrossRef]
- Callahan, R.; Kieran, M.W.; Baird, C.W.; Colan, S.D.; Gauvreau, K.; Ireland, C.M.; Marshall, A.C.; Sena, L.M.; Vargas, S.O.; Jenkins, K.J. Adjunct Targeted Biologic Inhibition Agents to Treat Aggressive Multivessel Intraluminal Pediatric Pulmonary Vein Stenosis. J. Pediatr. 2018, 198, 29–35.e5. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Shu, S.; Peng, Z.; Liu, X.; Chen, X.; Zeng, Z.; Yang, Y.; Cui, H.; Zhao, R.; Wang, X.; et al. Single-Cell RNA Sequencing of Coronary Perivascular Adipose Tissue From End-Stage Heart Failure Patients Identifies SPP1+ Macrophage Subpopulation as a Target for Alleviating Fibrosis. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 2143–2164. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Hou, Z.; Ding, Z.; Wang, P.; Pan, X.; Li, X. Single Cell RNA-Seq Identifies Cell Subpopulations Contributing to Idiopathic Pulmonary Fibrosis in Humans. J. Cell. Mol. Med. 2025, 29, e70402. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, M.; Cao, D.; Zheng, A.; Zhang, Y.; Su, Y.; Wang, J.; Xu, Y.; Zhou, M.; Tang, Y.; et al. Inhibition of Fap Promotes Cardiac Repair by Stabilizing BNP. Circ. Res. 2023, 132, 586–600. [Google Scholar] [CrossRef]
- Kimura, T.; Monslow, J.; Klampatsa, A.; Leibowitz, M.; Sun, J.; Liousia, M.; Woodruff, P.; Moon, E.; Todd, L.; Puré, E.; et al. Loss of Cells Expressing Fibroblast Activation Protein Has Variable Effects in Models of TGF-β and Chronic Bleomycin-Induced Fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 317, L271–L282. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.E.; Hamson, E.J.; Koczorowska, M.M.; Tholen, S.; Chowdhury, S.; Bailey, C.G.; Lay, A.J.; Twigg, S.M.; Lee, Q.; Roediger, B.; et al. Identification of Novel Natural Substrates of Fibroblast Activation Protein-Alpha by Differential Degradomics and Proteomics. Mol. Cell. Proteomics MCP 2019, 18, 65–85. [Google Scholar] [CrossRef]
- Edosada, C.Y.; Quan, C.; Wiesmann, C.; Tran, T.; Sutherlin, D.; Reynolds, M.; Elliott, J.M.; Raab, H.; Fairbrother, W.; Wolf, B.B. Selective Inhibition of Fibroblast Activation Protein Protease Based on Dipeptide Substrate Specificity*. J. Biol. Chem. 2006, 281, 7437–7444. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Hayashi, H.; Kawahara, T.; Katsuki, S.; Kimura, M.; Hino, R.; Sun, J.; Nakamaru, R.; Tenma, A.; Toyoura, M.; et al. A Vaccine Against Fibroblast Activation Protein Improves Murine Cardiac Fibrosis by Preventing the Accumulation of Myofibroblasts. Circ. Res. 2025, 136, 26–40. [Google Scholar] [CrossRef]
- Liu, T.; Zhou, L.; Yang, K.; Iwasawa, K.; Kadekaro, A.L.; Takebe, T.; Andl, T.; Zhang, Y. The β-Catenin/YAP Signaling Axis Is a Key Regulator of Melanoma-Associated Fibroblasts. Signal Transduct. Target. Ther. 2019, 4, 63. [Google Scholar] [CrossRef]
- Francisco, J.; Zhang, Y.; Jeong, J.I.; Mizushima, W.; Ikeda, S.; Ivessa, A.; Oka, S.; Zhai, P.; Tallquist, M.D.; Del Re, D.P. Blockade of Fibroblast YAP Attenuates Cardiac Fibrosis and Dysfunction Through MRTF-A Inhibition. JACC Basic Transl. Sci. 2020, 5, 931–945. [Google Scholar] [CrossRef]
- Pang, Z.-D.; Sun, X.; Bai, R.-Y.; Han, M.-Z.; Zhang, Y.-J.; Wu, W.; Zhang, Y.; Lai, B.-C.; Zhang, Y.; Wang, Y.; et al. YAP-Galectin-3 Signaling Mediates Endothelial Dysfunction in Angiotensin II-Induced Hypertension in Mice. Cell. Mol. Life Sci. CMLS 2023, 80, 38. [Google Scholar] [CrossRef]
- Eyries, M.; Montani, D.; Girerd, B.; Perret, C.; Leroy, A.; Lonjou, C.; Chelghoum, N.; Coulet, F.; Bonnet, D.; Dorfmüller, P.; et al. EIF2AK4 Mutations Cause Pulmonary Veno-Occlusive Disease, a Recessive Form of Pulmonary Hypertension. Nat. Genet. 2014, 46, 65–69. [Google Scholar] [CrossRef]
- Zhang, J.; Mao, P.; Zhou, T.; Yue, B.; Li, Y.; Qiu, Y.; Ying, K.; Wang, F.; Chen, J.; Yang, J. Macrophage Ferroptosis Potentiates GCN2 Deficiency Induced Pulmonary Venous Arterialization. Nat. Commun. 2025, 16, 8335. [Google Scholar] [CrossRef]
- Zurawska, G.; Jończy, A.; Niklewicz, M.; Sas, Z.; Rumieńczyk, I.; Kulecka, M.; Piwocka, K.; Rygiel, T.P.; Mikula, M.; Mleczko-Sanecka, K. Iron-triggered signaling via ETS1 and the p38/JNK MAPK pathway regulates Bmp6 expression. American journal of hematology. 2024, 99, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Hammer, P.E.; McEnaney, K.; Callahan, R.; Baird, C.W.; Hoganson, D.M.; Jenkins, K.J. The Role of Elevated Wall Shear Stress in Progression of Pulmonary Vein Stenosis: Evidence from Two Case Studies. Children 2021, 8, 729. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, A.H.; Yamada, J.M.; Miller, D.T.; McEnaney, K.; Ireland, C.; Roberts, A.E.; Gauvreau, K.; Jenkins, K.J.; Chen, M.H. Clinical Syndromic Phenotypes and the Potential Role of Genetics in Pulmonary Vein Stenosis. Children 2021, 8, 128. [Google Scholar] [CrossRef]
- Hall, I.F.; Aikawa, E.; Sluimer, J.; Baker, A.H.; Kovacic, J.C. Endothelial to Mesenchymal Transition in Cardiovascular Diseases: Molecular Insights and Clinical Perspectives. Eur. Heart J. 2026, 47, 1144–1158. [Google Scholar] [CrossRef]
- Wendler, A.; Wehling, M. The Translatability of Animal Models for Clinical Development: Biomarkers and Disease Models. Curr. Opin. Pharmacol. 2010, 10, 601–606. [Google Scholar] [CrossRef]
- Loewa, A.; Feng, J.J.; Hedtrich, S. Human Disease Models in Drug Development. Nat. Rev. Bioeng. 2023, 1, 545–559. [Google Scholar] [CrossRef] [PubMed]
- Callahan, R.; Gauvreau, K.; Marshall, A.C.; Sena, L.M.; Baird, C.W.; Ireland, C.M.; McEnaney, K.; Bjornlund, E.C.; Mendonca, J.T.; Jenkins, K.J. Outcomes in Establishing Individual Vessel Patency for Pediatric Pulmonary Vein Stenosis. Children 2021, 8, 210. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Novruzov, E.; Schmitt, D.; Cardinale, J.; Watabe, T.; Choyke, P.L.; Alavi, A.; Haberkorn, U.; Giesel, F.L. Clinical Applications of Fibroblast Activation Protein Inhibitor Positron Emission Tomography (FAPI-PET). Npj Imaging 2024, 2, 48. [Google Scholar] [CrossRef] [PubMed]
- Lavis, P.; Garabet, A.; Cardozo, A.K.; Bondue, B. The Fibroblast Activation Protein Alpha as a Biomarker of Pulmonary Fibrosis. Front. Med. 2024, 11, 1393778. [Google Scholar] [CrossRef]
- van Genuchten, W.J.; Averesch, H.; van Dieren, Q.M.; Bonnet, D.; Odermarsky, M.; Beghetti, M.; Roos-Hesselink, J.W.; Reinhardt, Z.; Male, C.; Naumburg, E.; et al. Clinical Impact of Circulating Biomarkers in Prediction of Adverse Cardiac Events in Patients with Congenital Heart Disease. A Systematic Review. Int. J. Cardiol. 2025, 421, 132723. [Google Scholar] [CrossRef]
- Wang, R.-S.; Maron, B.A.; Loscalzo, J. Multiomics Network Medicine Approaches to Precision Medicine and Therapeutics in Cardiovascular Diseases. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 493–503. [Google Scholar] [CrossRef]
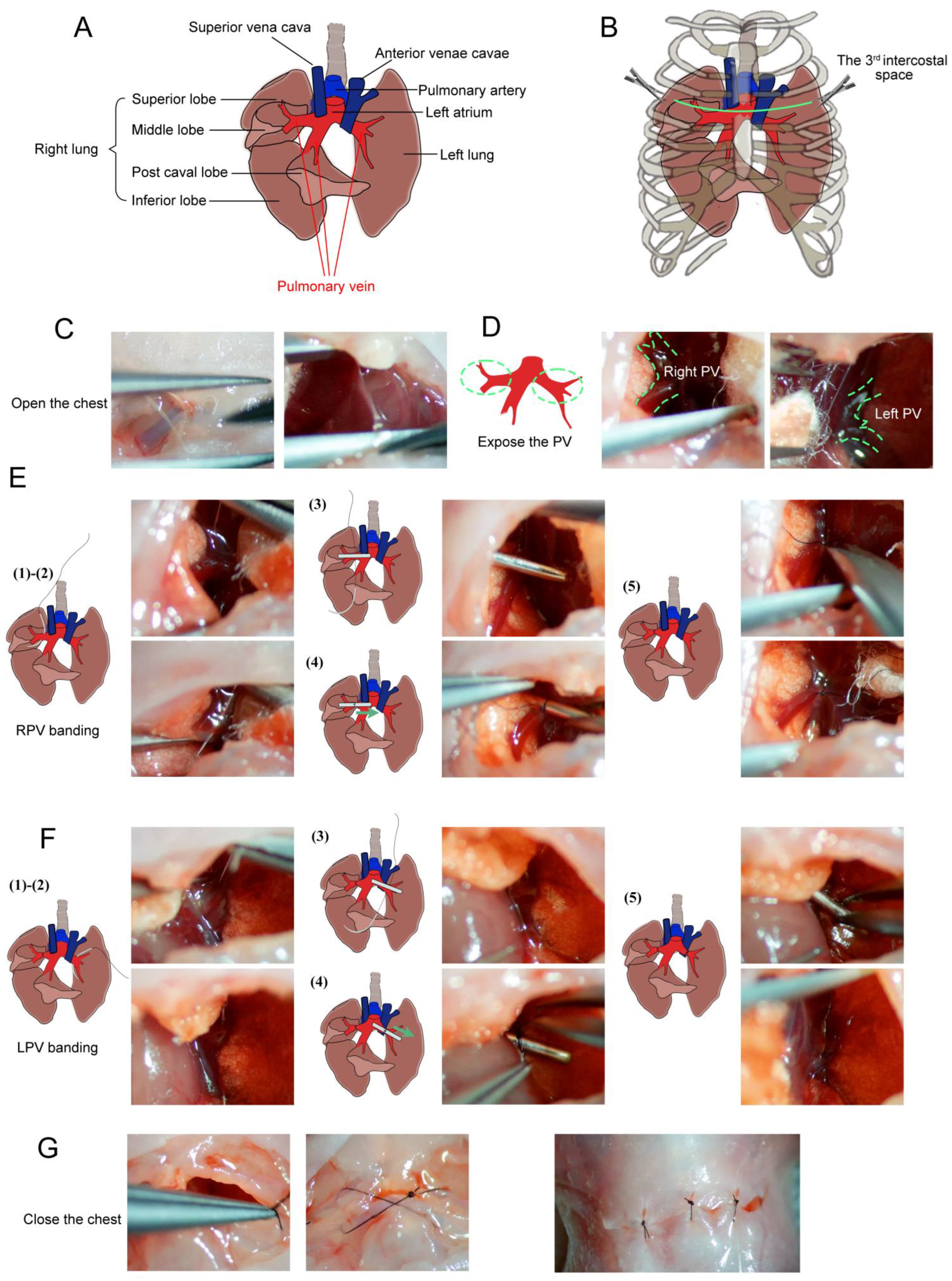
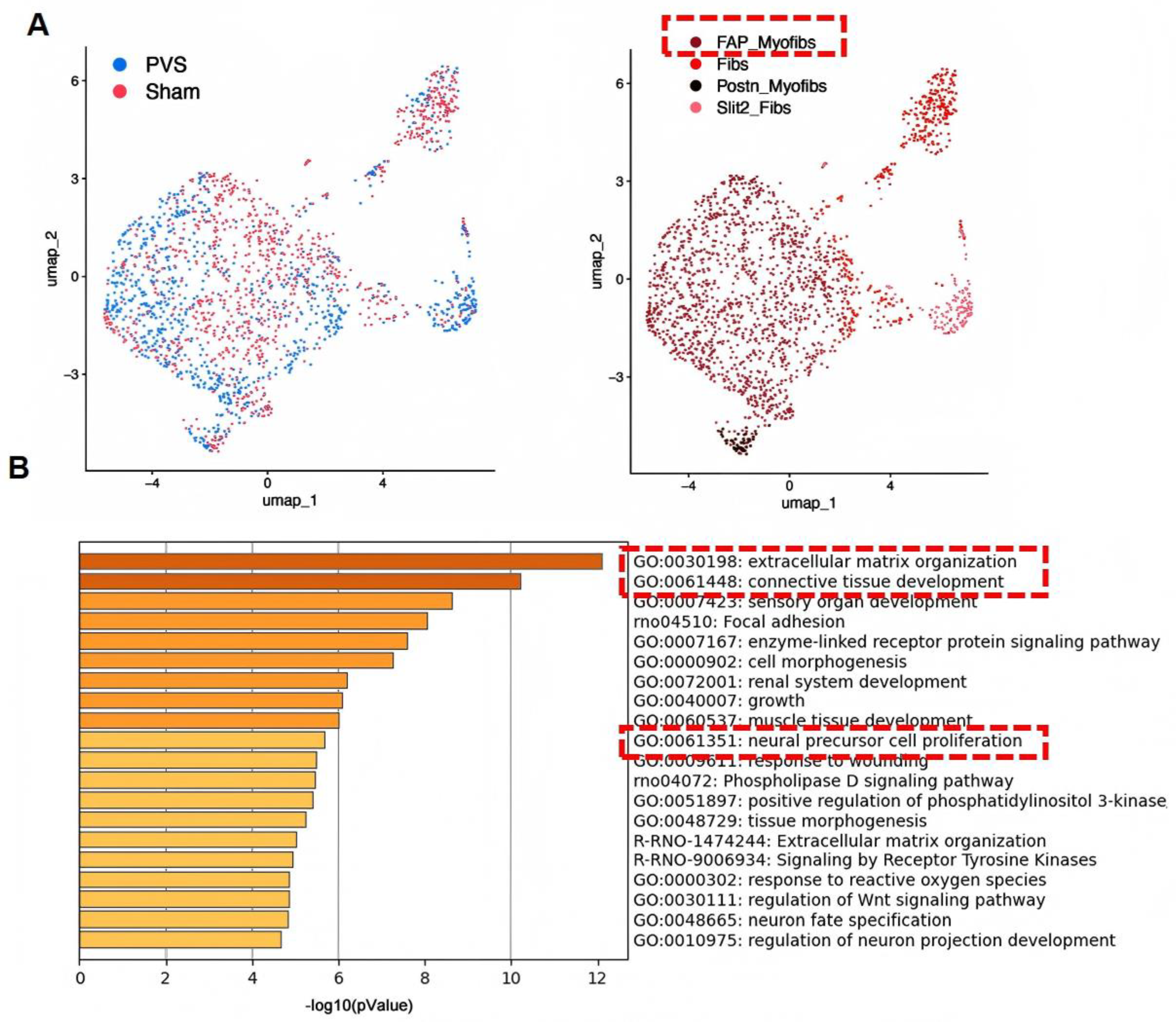
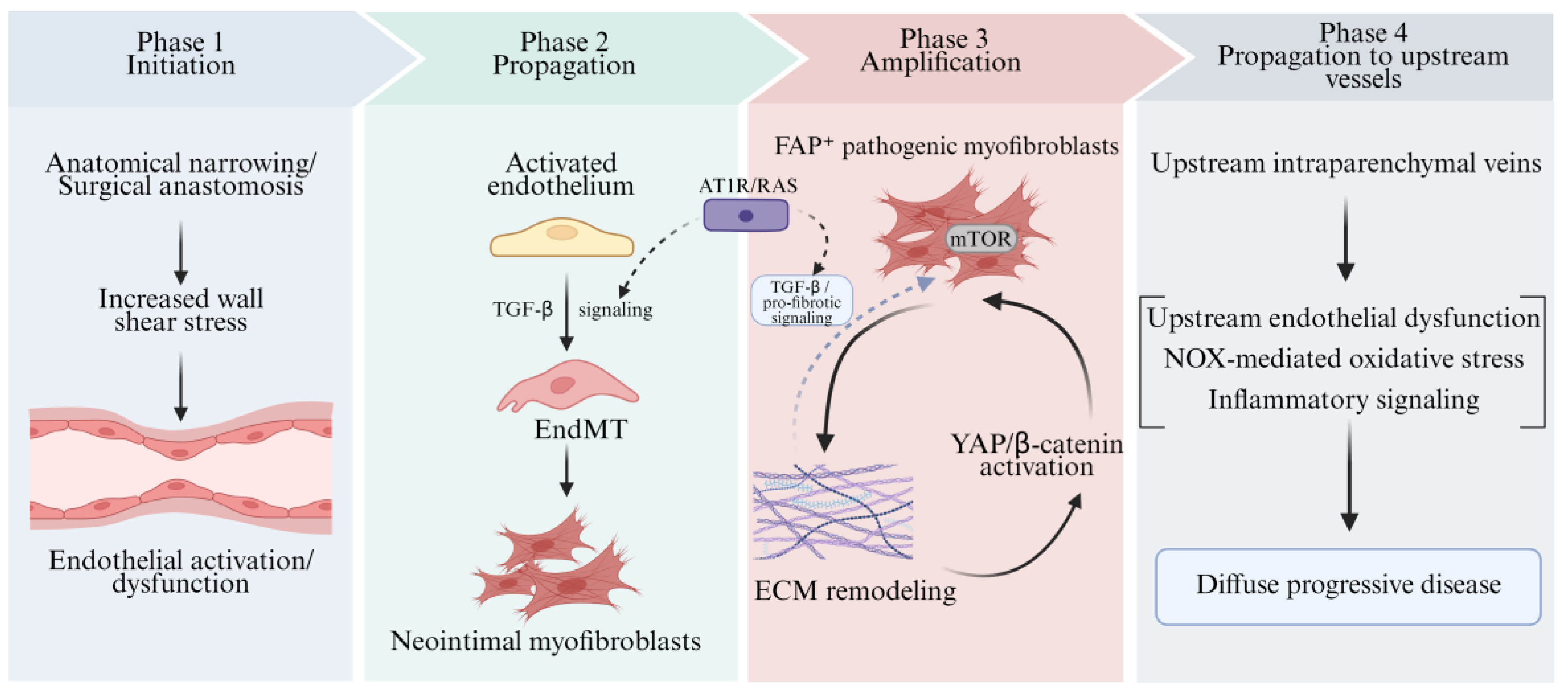
| Feature | Piglet Model | Neonatal Rat Model |
|---|---|---|
| Cost per animal experiment | ¥5,000-10,000 | ¥50-100 |
| Time to disease | 8-12 weeks | 3-4 weeks |
| Surgical complexity | High | Very high (microsurgery) |
| Genetic tools | Limited | Extensive |
| Throughput | Low | High |
| Hemodynamic assessment | Comprehensive | Limited by size |
| Human relevance | High (anatomy) | High (pathology) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




