Submitted:
14 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Definitions and Clinically Actionable Endotypes
3.1. Structural Coronary Microvascular Dysfunction (CMD)
3.2. Functional/Endothelial CMD
3.3. Epicardial Vasospasm (Vasospastic Angina)
3.4. Microvascular Spasm
4. Pathophysiological Rationale
4.1. Coronary Flow as the Final Common Pathway
4.2. Pressure-Based Versus Flow-Based Physiology
4.3. Mechanisms of Coronary Microvascular Dysfunction
4.4. Vasomotor Disorders and Dynamic Flow Limitation
4.5. Prognostic Implications of Flow Impairment
4.6. From Pathophysiology to Mechanism-Based Care
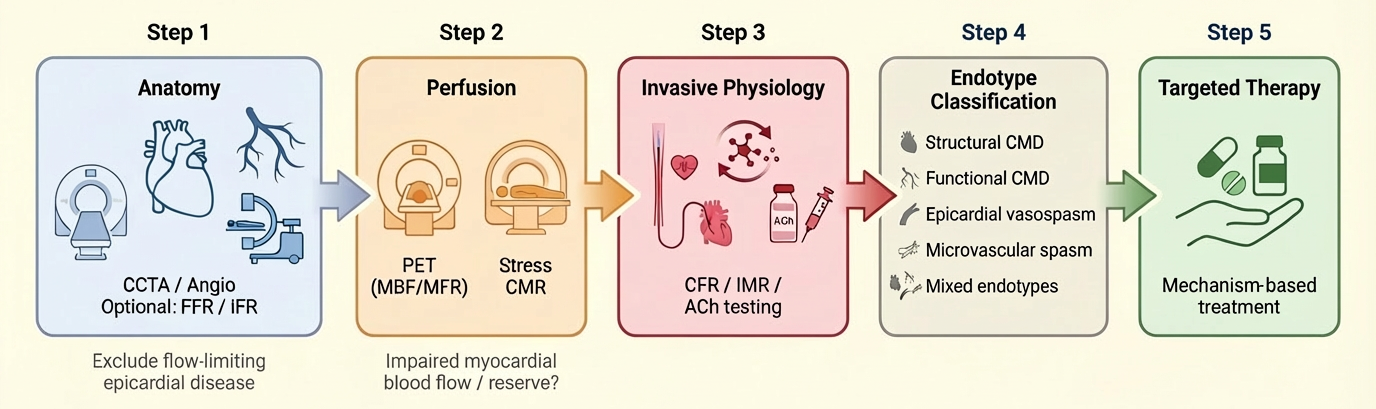
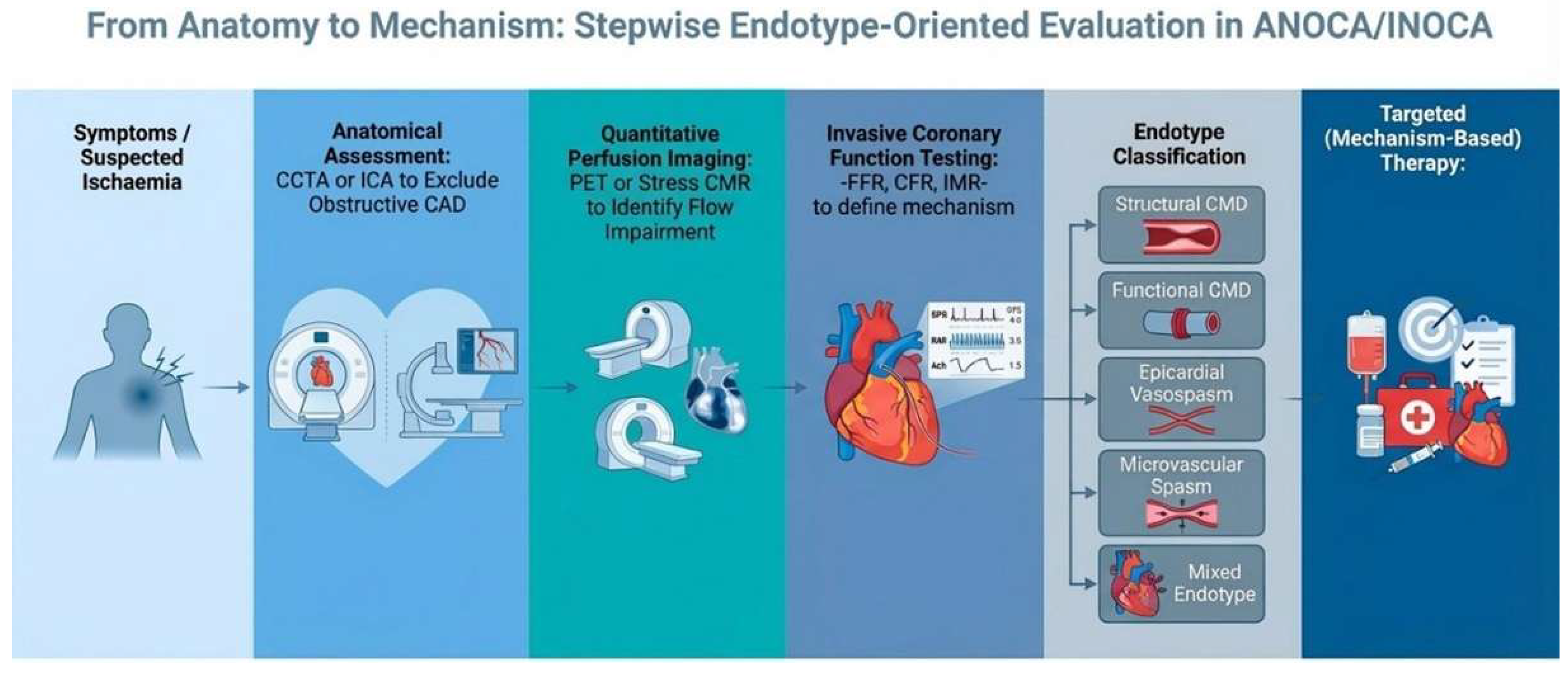
5. Non-Invasive Testing: From Ischaemia Detection to Functional Endotyping
5.1. CCTA: An Anatomic Gatekeeper and Plaque Phenotype Tool
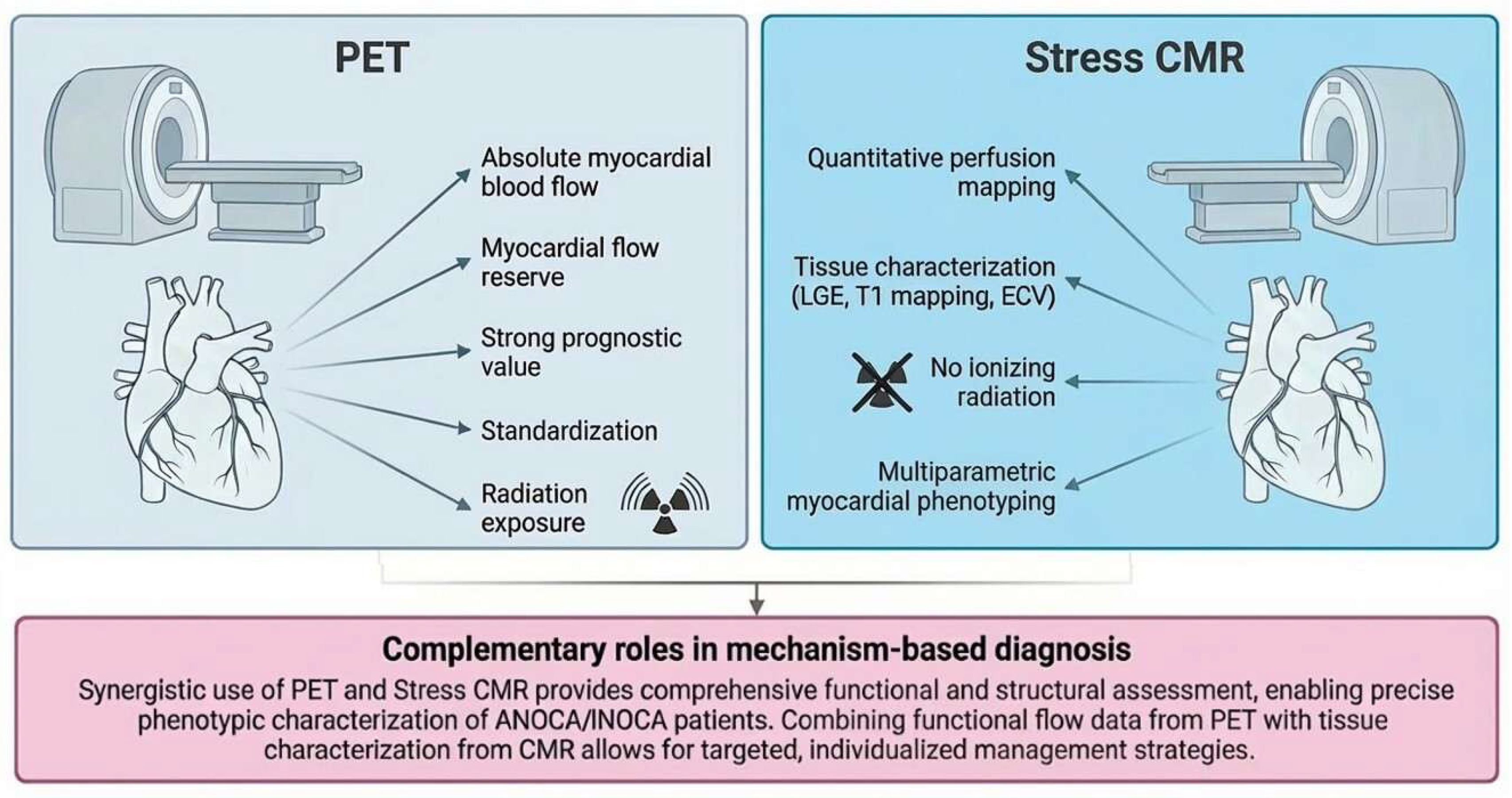
5.2. PET: Absolute Myocardial Blood Flow and Flow Reserve
- epicardial flow-limiting disease,
- diffuse atherosclerosis,
- structural or functional coronary microvascular dysfunction,
- or combinations thereof
5.2.1. Diagnostic Patterns and Endotype Inference
5.2.3. Thresholds and Standardisation Challenges
5.2.4. Prognostic Implications
5.2.5. Limitations
5.3. Stress CMR: Quantitative Perfusion and Myocardial Phenotyping
5.3.1. Quantitative Perfusion Mapping
5.3.2. Integrated Myocardial Characterisation
5.3.3. Diagnostic Performance and Incremental Value
5.3.4. Limitations and Technical Considerations
5.4. PET Versus CMR: Complementary Rather than Competitive
5.5. Bridge: from Anatomy to Physiology to Mechanism
6. Invasive Coronary Function testing: ESC-Aligned “Complete the Endotype” Strategy
6.1. Why ICFT Is Increasingly Central: Concept and Rationale
6.2. Core Invasive Measurements: Epicardial Physiology, CFR, and Microvascular Resistance
6.3. Acetylcholine Provocation Testing: Epicardial vs Microvascular Spasm
6.4. Integrated ICFT: Completing the Endotype
6.5. Evidence of Clinical Utility: CorMicA and Stratified Therapy
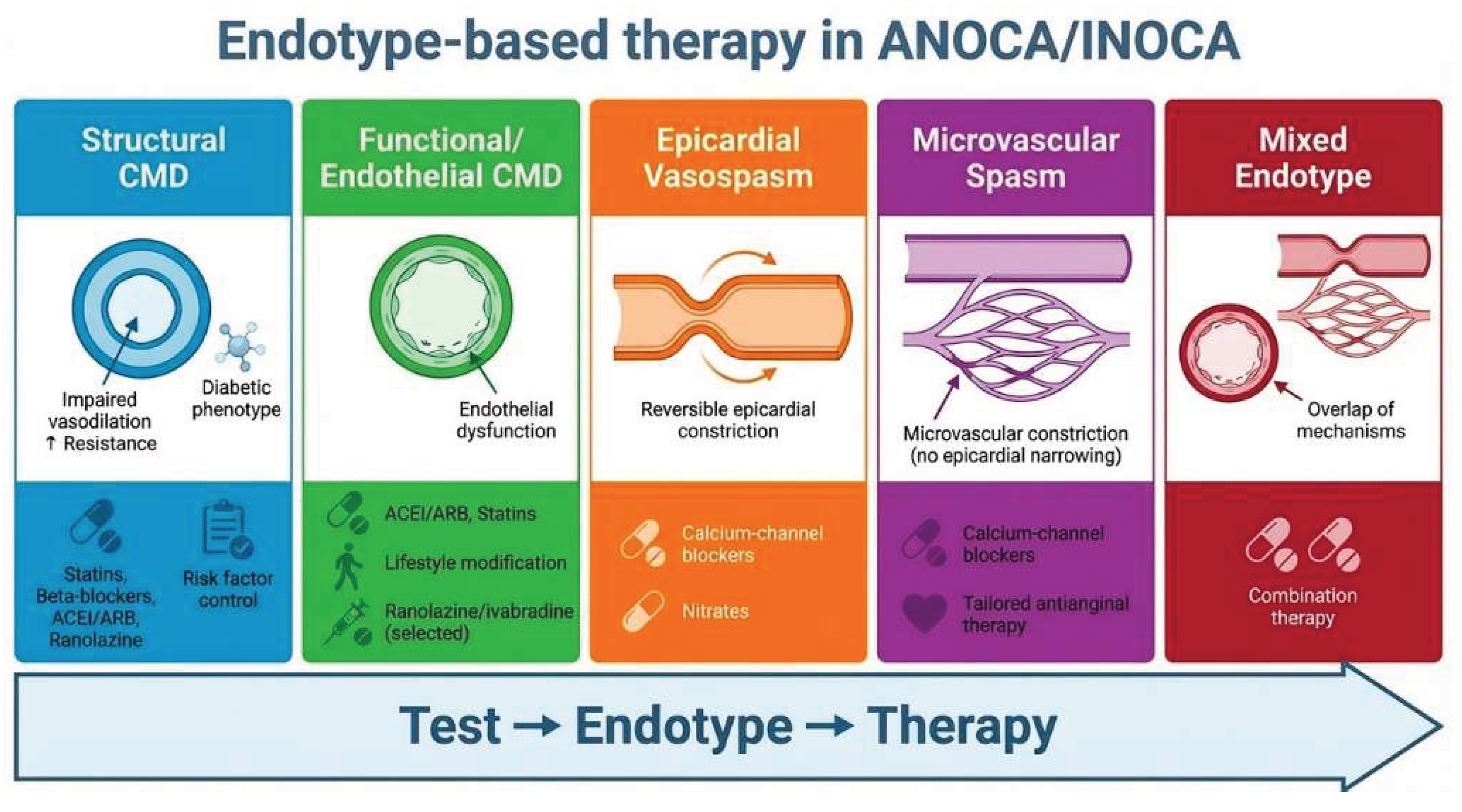
7. Linking Endotype to Therapy
7.1. From Mechanism to Management: A Paradigm Shift
7.2. Structural Coronary Microvascular Dysfunction: A Systemic Cardiometabolic Target
7.3. Functional CMD: Targeting Endothelial Dysfunction
7.4. Vasospastic Angina and Microvascular Spasm: Suppressing Abnormal Vasoconstriction
7.5. Mixed Endotypes: The Rule Rather than the Exception
7.6. Evidence Supporting Stratified Therapy
7.7. Toward Precision Medicine in ANOCA/INOCA
8. Future Prospects and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACh, acetylcholine; ACEi, angiotensin-converting enzyme inhibitors; ANOCA, angina with non-obstructive coronary arteries; ARB, angiotensin receptor blockers; CAD, coronary artery disease; CCB, calcium-channel blockers; CCS, chronic coronary syndromes; CCTA, coronary computed tomography angiography; CFR, coronary flow reserve; CMD, coronary microvascular dysfunction; CMR, cardiovascular magnetic resonance; COVADIS, Coronary Vasomotion Disorders International Study Group; ECG, electrocardiogram; ECV, extracellular volume; ESC, European Society of Cardiology; FFR, fractional flow reserve; FFR-CT, computed tomography-derived fractional flow reserve; ICA, invasive coronary angiography; ICFT, invasive coronary function testing; iFR, instantaneous wave-free ratio; IMR, index of microcirculatory resistance; INOCA, ischaemia with non-obstructive coronary arteries; LGE, late gadolinium enhancement; MBF, myocardial blood flow; MFR, myocardial flow reserve; MPR, myocardial perfusion reserve; NO, nitric oxide; PET, positron emission tomography; VSA, vasospastic angina. |
References
- Vrints, C.; Andreotti, F.; Koskinas, K.C.; Rossello, X.; Adamo, M.; Ainslie, J.; Banning, A.P.; Budaj, A.; Buechel, R.R.; Chiariello, G.A.; et al.; ESC Scientific Document Group 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur Heart J 2024, 45(36), 3415–3537. [Google Scholar] [CrossRef]
- Samuels, B.A.; Shah, S.M.; Widmer, R.J.; Kobayashi, Y.; Miner, S.E.S.; Taqueti, V.R.; Jeremias, A.; Albadri, A.; Blair, J.A.; Kearney, K.E.; et al. Microvascular Network (MVN). Comprehensive Management of ANOCA, Part 1-Definition, Patient Population, and Diagnosis: JACC State-of-the-Art Review. J Am Coll Cardiol. 2023, 82(12), 1245–1263. [Google Scholar] [CrossRef]
- Kunadian, V.; Chieffo, A.; Camici, P.G.; Berry, C.; Escaned, J.; Maas, A.H.E.M.; Prescott, E.; Karam, N.; Appelman, Y.; Fraccaro, C.; et al. An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries in Collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group. Eur Heart J 2020, 41(37), 3504–3520. [Google Scholar] [CrossRef]
- Beltrame, J.F.; Crea, F.; Kaski, J.C.; Ogawa, H.; Ong, P.; Sechtem, U.; Shimokawa, H.; Bairey Merz, C.N. Coronary Vasomotion Disorders International Study Group (COVADIS). International standardization of diagnostic criteria for vasospastic angina. Eur Heart J 2017, 38(33), 2565–2568. [Google Scholar] [CrossRef]
- Ong, P.; Camici, P.G.; Beltrame, J.F.; Crea, F.; Shimokawa, H.; Sechtem, U.; Kaski, J.C.; Bairey Merz, C.N. Coronary Vasomotion Disorders International Study Group (COVADIS). International standardization of diagnostic criteria for microvascular angina. Int J Cardiol 2018, 250, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.; Rahman, H.; Perera, D. Reply: Rethinking False Positive Exercise Electrocardiographic Stress Tests by Assessing Coronary Microvascular Function. J Am Coll Cardiol. 2024, 83(20), e195–6. [Google Scholar] [CrossRef]
- Leone, A.M.; Galante, D.; Viceré, A.; Marrone, A.; Verardi, F.M.; Giuliana, C.; Pollio Benvenuto, C.; Viccaro, V.; Todisco, S.; Erriquez, A.; G.; et al. Functional coronary assessment in angina with intermediate coronary stenosis: the #FullPhysiology approach. Eur Heart J 2025, 46(10), 978–980. [Google Scholar] [CrossRef] [PubMed]
- van de Hoef, T.P.; Lee, J.M.; Boerhout, C.K.M.; de Waard, G.A.; Jung, J.H.; Lee, S.H.; Mejía-Rentería, H.; Hoshino, M.; Echavarria-Pinto, M.; Meuwissen, M.; et al. Combined Assessment of FFR and CFR for Decision Making in Coronary Revascularization: From the Multicenter International ILIAS Registry. JACC Cardiovasc Interv. 2022, 15(10), 1047–1056. [Google Scholar] [CrossRef] [PubMed]
- Seitz, A.; Martínez Pereyra, V.; Sechtem, U.; Ong, P. Update on coronary artery spasm 2022 - A narrative review. Int J Cardiol 2022, 359, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Suda, A.; Takahashi, J.; Hao, K.; Kikuchi, Y.; Shindo, T.; Ikeda, S.; Sato, K.; Sugisawa, J.; Matsumoto, Y.; Miyata, S.; et al. Coronary Functional Abnormalities in Patients With Angina and Nonobstructive Coronary Artery Disease. J Am Coll Cardiol. 2019, 74(19), 2350–2360. [Google Scholar] [CrossRef] [PubMed]
- Woudstra, J.; Dimitriu-Leen, A.C.; Crooijmans, C.; Yosofi, B.; Meeder, J.G.; Meuwissen, M.; De Vos, A.M.J.; Paradies, V.; Bijvank, E.G.M.O.; Winkler, P. for NL-CFT. Microvascular vasomotor dysfunction is more prevalent in female versus epicardial vasomotor dysfunction in male ANOCA patients. Int J Cardiol. 2026, 450, 134226. [Google Scholar] [CrossRef]
- Del Buono, M.G.; Montone, R.A.; Camilli, M.; Carbone, S.; Narula, J.; Lavie, C.J.; Niccoli, G.; Crea, F. Coronary Microvascular Dysfunction Across the Spectrum of Cardiovascular Diseases: JACC State-of-the-Art Review. J Am Coll Cardiol. 2021, 78(13), 1352–1371. [Google Scholar] [CrossRef]
- Pijls, N.H.; De Bruyne, B.; Peels, K.; Van Der Voort, P.H.; Bonnier, H.J.; Bartunek, J.; Koolen, J.J.; Koolen, J.J. Measurement of fractional flow reserve to assess the functional severity of coronary-artery stenoses. N Engl J Med. 1996, 334(26), 1703–8. [Google Scholar] [CrossRef]
- Parlati, A.L.M.; Nardi, E.; Sucato, V.; Madaudo, C.; Leo, G.; Rajah, T.; Marzano, F.; Prastaro, M.; Gargiulo, P.; Paolillo, S.; Vadalà, G.; et al. ANOCA, INOCA, MINOCA: The New Frontier of Coronary Syndromes. J Cardiovasc Dev Dis. 2025, 12(2), 64. [Google Scholar] [CrossRef] [PubMed]
- Duncker, D.J.; Koller, A.; Merkus, D.; Canty, J.M. Regulation of coronary blood flow in health and ischemic heart disease. Prog Cardiovasc Dis. 2015, 57(5), 409–22. [Google Scholar] [CrossRef] [PubMed]
- Camici, P.G.; Crea, F. Coronary microvascular dysfunction. N Engl J Med 2007, 356(8), 830–40. [Google Scholar] [CrossRef]
- Gould, K.L. Does coronary flow trump coronary anatomy? JACC Cardiovasc Imaging 2009, 2(8), 1009–23. [Google Scholar] [CrossRef]
- Tonino, P.A.; De Bruyne, B.; Pijls, N.H.; Siebert, U.; Ikeno, F.; van’ t Veer, M.; Klauss, V.; Manoharan, G.; Engstrøm, T.; Oldroyd, K.G.; et al. FAME Study Investigators. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009, 360(3), 213–24. [Google Scholar] [CrossRef]
- Gould, K.L.; Kirkeeide, R.L.; Buchi, M. Coronary flow reserve as a physiologic measure of stenosis severity. J Am Coll Cardiol. 1990, 15(2), 459–74. [Google Scholar] [CrossRef]
- Berry, C.; Corcoran, D.; Hennigan, B.; Watkins, S.; Layland, J.; Oldroyd, K.G. Fractional flow reserve-guided management in stable coronary disease and acute myocardial infarction: recent developments. Eur Heart J 2015, 36(45), 3155–64. [Google Scholar] [CrossRef] [PubMed]
- Stegehuis, V.E.; Wijntjens, G.W.; Piek, J.J.; van de Hoef, T.P. Fractional Flow Reserve or Coronary Flow Reserve for the Assessment of Myocardial Perfusion : Implications of FFR as an Imperfect Reference Standard for Myocardial Ischemia. Curr Cardiol Rep. 2018, 20(9), 77. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.P.; Kirkeeide, R.L.; Gould, K.L. Is discordance of coronary flow reserve and fractional flow reserve due to methodology or clinically relevant coronary pathophysiology? JACC Cardiovasc Imaging 2012, 5(2), 193–202. [Google Scholar] [CrossRef]
- Ahn, S.G.; Suh, J.; Hung, O.Y.; Lee, H.S.; Bouchi, Y.H.; Zeng, W.; Gandhi, R.; Eshtehardi, P.; Gogas, B.D.; Samady, H. Discordance Between Fractional Flow Reserve and Coronary Flow Reserve: Insights From Intracoronary Imaging and Physiological Assessment. JACC Cardiovasc Interv. 2017, 10(10), 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Kelshiker, M.A.; Seligman, H.; Howard, J.P.; Rahman, H.; Foley, M.; Nowbar, A.N.; Rajkumar, C.A.; Shun-Shin, M.J.; Ahmad, Y.; Sen, S. Coronary Flow Outcomes Reviewing Committee. Coronary flow reserve and cardiovascular outcomes: a systematic review and meta-analysis. Eur Heart J 2022, 43(16), 1582–1593. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Jung, J.H.; Hwang, D.; Park, J.; Fan, Y.; Na, S.H.; Doh, J.H.; Nam, C.W.; Shin, E.S.; Koo, B.K. Coronary Flow Reserve and Microcirculatory Resistance in Patients With Intermediate Coronary Stenosis. J Am Coll Cardiol. 2016, 67(10), 1158–1169. [Google Scholar] [CrossRef] [PubMed]
- Rahman, H.; Demir, O.M.; Khan, F.; Ryan, M.; Ellis, H.; Mills, M.T.; Chiribiri, A.; Webb, A.; Perera, D. Physiological Stratification of Patients With Angina Due to Coronary Microvascular Dysfunction. J Am Coll Cardiol. 2020, 75(20), 2538–2549. [Google Scholar] [CrossRef]
- Crea, F.; Camici, P.G.; Bairey Merz, C.N. Coronary microvascular dysfunction: an update. Eur Heart J 2014, 35(17), 1101–11. [Google Scholar] [CrossRef] [PubMed]
- Pelliccia, F.; Cecchi, F.; Olivotto, I.; Camici, P.G. Microvascular Dysfunction in Hypertrophic Cardiomyopathy. J Clin Med. 2022, 11(21), 6560. [Google Scholar] [CrossRef] [PubMed]
- Collet, C.; Yong, A.; Munhoz, D.; Akasaka, T.; Berry, C.; Blair, J.E.A.; Collison, D.; Engstrøm, T.; Escaned, J.; Fearon, W.F.; et al. A Systematic Approach to the Evaluation of the Coronary Microcirculation Using Bolus Thermodilution: CATH CMD. J Soc Cardiovasc Angiogr Interv. 2024, 3(7), 101934. [Google Scholar] [CrossRef]
- Lanza, G.A.; Careri, G.; Crea, F. Mechanisms of coronary artery spasm. Circulation 2011, 124(16), 1774–82. [Google Scholar] [CrossRef]
- Hubert, A.; Seitz, A.; Pereyra, V.M.; Bekeredjian, R.; Sechtem, U.; Ong, P. Coronary Artery Spasm: The Interplay Between Endothelial Dysfunction and Vascular Smooth Muscle Cell Hyperreactivity. Eur Cardiol 2020, 15, e12. [Google Scholar] [CrossRef]
- Picard, F.; Sayah, N.; Spagnoli, V.; Adjedj, J.; Varenne, O. Vasospastic angina: A literature review of current evidence. Arch Cardiovasc Dis 2019, 112(1), 44–55. [Google Scholar] [CrossRef]
- Takahashi, J.; Suda, A.; Nishimiya, K.; Godo, S.; Yasuda, S.; Shimokawa, H. Pathophysiology and Diagnosis of Coronary Functional Abnormalities. Eur Cardiol 2021, 16, e30. [Google Scholar] [CrossRef]
- Masumoto, A.; Mohri, M.; Shimokawa, H.; Urakami, L.; Usui, M.; Takeshita, A. Suppression of coronary artery spasm by the Rho-kinase inhibitor fasudil in patients with vasospastic angina. Circulation 2002, 105(13), 1545–7. [Google Scholar] [CrossRef]
- Matta, A.; Bouisset, F.; Lhermusier, T.; Campelo-Parada, F.; Elbaz, M.; Carrié, D.; Roncalli, J. Coronary Artery Spasm: New Insights. J Interv Cardiol. 2020, 2020, 5894586. [Google Scholar] [CrossRef] [PubMed]
- Mileva, N.; Nagumo, S.; Mizukami, T.; Sonck, J.; Berry, C.; Gallinoro, E.; et al. Prevalence of Coronary Microvascular Disease and Coronary Vasospasm in Patients With Nonobstructive Coronary Artery Disease: Systematic Review and Meta-Analysis. J Am Heart Assoc. 2022, 11(7), e023207. [Google Scholar] [CrossRef] [PubMed]
- Ziadi, M.C.; Dekemp, R.A.; Williams, K.A.; Guo, A.; Chow, B.J.; Renaud, J.M.; Ruddy, T.D.; Sarveswaran, N.; Tee, R.E.; Beanlands, R.S. Impaired myocardial flow reserve on rubidium-82 positron emission tomography imaging predicts adverse outcomes in patients assessed for myocardial ischemia. J Am Coll Cardiol. 2011, 58(7), 740–8. [Google Scholar] [CrossRef] [PubMed]
- Højstrup, S.; Hansen, K.W.; Talleruphuus, U.; Marner, L.; Bjerking, L.; Jakobsen, L.; Christiansen, E.H.; Bouchelouche, K.; Wiinberg, N.; Guldbrandsen, K.; et al. Myocardial Flow Reserve, an Independent Prognostic Marker of All-Cause Mortality Assessed by 82Rb PET Myocardial Perfusion Imaging: A Danish Multicenter Study. Circ Cardiovasc Imaging 2023, 16(8), e015184. [Google Scholar] [CrossRef]
- Boerhout, C.K.M.; de Waard, G.A.; Lee, J.M.; Mejia-Renteria, H.; Lee, S.H.; Jung, J.H.; Hoshino, M.; Echavarria-Pinto, M.; Meuwissen, M.; Matsuo, H.; et al. Prognostic value of structural and functional coronary microvascular dysfunction in patients with non-obstructive coronary artery disease; from the multicentre international ILIAS registry. EuroIntervention 2022, 18(9), 719–728. [Google Scholar] [CrossRef]
- Hwang, D.; Park, S.; Koo, B.K. Reply: Ischemia With Nonobstructive Coronary Artery Disease. JACC Asia;PubMed 2023, 3(4), 687. [Google Scholar] [CrossRef] [PubMed]
- Miner, S.; Mejia-Renteria, H.; Leone, A.M.; Velollari, O.; Sykes, R.; Biscaglia, S.; Esposito, G.; Galante, D.; Oreglia, J.; Ang, D.; et al. Endotypes of angina with non-obstructive coronary arteries: a prospective multicentre study. Eur Heart J. 2026, 47(5), 594–604. [Google Scholar] [CrossRef] [PubMed]
- Hokimoto, S.; Kaikita, K.; Yasuda, S.; Tsujita, K.; Ishihara, M.; Matoba, T.; Matsuzawa, Y.; Mitsutake, Y.; Mitani, Y.; Murohara, T. Japanese Circulation Society and Japanese Association of Cardiovascular Intervention and Therapeutics and Japanese College of Cardiology Joint Working Group. Electronic address: jcsGL@j-circ.or.jp. JCS/CVIT/JCC 2023 guideline focused update on diagnosis and treatment of vasospastic angina (coronary spastic angina) and coronary microvascular dysfunction. J Cardiol. 2023, 82(4), 293–341. [Google Scholar] [CrossRef]
- Jenkins, K.; Pompei, G.; Ganzorig, N.; Brown, S.; Beltrame, J.; Kunadian, V. Vasospastic angina: a review on diagnostic approach and management. Ther Adv Cardiovasc Dis. 2024, 18, 17539447241230400. [Google Scholar] [CrossRef]
- Welch, I.; Case, B. Treatment options for patients with coronary microvascular dysfunction. Future Cardiol 2025, 21(10), 735–8. [Google Scholar] [CrossRef]
- Ford, T.J.; Stanley, B.; Good, R.; Rocchiccioli, P.; McEntegart, M.; Watkins, S.; Eteiba, H.; Shaukat, A.; Lindsay, M.; Robertson, K.; et al. Stratified Medical Therapy Using Invasive Coronary Function Testing in Angina: The CorMicA Trial. J Am Coll Cardiol. 2018, 72 23 Pt A, 2841–2855. [Google Scholar] [CrossRef]
- Ford, T.J.; Stanley, B.; Sidik, N.; Good, R.; Rocchiccioli, P.; McEntegart, M.; Watkins, S.; Eteiba, H.; Shaukat, A.; Lindsay, M.; et al. 1-Year Outcomes of Angina Management Guided by Invasive Coronary Function Testing (CorMicA). JACC Cardiovasc Interv. 2020, 13(1), 33–45. [Google Scholar] [CrossRef] [PubMed]
- Hansen, B.; Holtzman, J.N.; Juszczynski, C.; Khan, N.; Kaur, G.; Varma, B.; Gulati, M. Ischemia with No Obstructive Arteries (INOCA): A Review of the Prevalence, Diagnosis and Management. Curr Probl Cardiol 2023, 48(1), 101420. [Google Scholar] [CrossRef]
- Kolossváry, M.; Szilveszter, B.; Merkely, B.; Maurovich-Horvat, P. Plaque imaging with CT-a comprehensive review on coronary CT angiography based risk assessment. Cardiovasc Diagn Ther 2017, 7(5), 489–506. [Google Scholar] [CrossRef] [PubMed]
- Nieman, K.; García-García, H.M.; Hideo-Kajita, A.; Collet, C.; Dey, D.; Pugliese, F.; Weissman, G.; Tijssen, J.G.P.; Leipsic, J.; Opolski, M.P. Standards for quantitative assessments by coronary computed tomography angiography (CCTA): An expert consensus document of the society of cardiovascular computed tomography (SCCT). J Cardiovasc Comput Tomogr. 2024, 18(5), 429–443. [Google Scholar] [CrossRef] [PubMed]
- Motoyama, S.; Sarai, M.; Harigaya, H.; Anno, H.; Inoue, K.; Hara, T.; Naruse, H.; Ishii, J.; Hishida, H.; Wong, N.D.; et al. Computed tomographic angiography characteristics of atherosclerotic plaques subsequently resulting in acute coronary syndrome. J Am Coll Cardiol. 2009, 54(1), 49–57. [Google Scholar] [CrossRef] [PubMed]
- Murthy, V.L.; Lee, B.C.; Sitek, A.; Naya, M.; Moody, J.; Polavarapu, V.; Ficaro, E.P.; Di Carli, M.F. Comparison and prognostic validation of multiple methods of quantification of myocardial blood flow with 82Rb PET. J Nucl Med. 2014, 55(12), 1952–8. [Google Scholar] [CrossRef]
- Schindler, T.H.; Fearon, W.F.; Pelletier-Galarneau, M.; Ambrosio, G.; Sechtem, U.; Ruddy, T.D.; Patel, K.K.; Bhatt, D.L.; Bateman, T.M.; Gewirtz, H.; et al. Myocardial Perfusion PET for the Detection and Reporting of Coronary Microvascular Dysfunction: A JACC: Cardiovascular Imaging Expert Panel Statement. JACC Cardiovasc Imaging 2023, 16(4), 536–548. [Google Scholar] [CrossRef]
- Vohra, S.; Sakul, S.; Agarwal, V.; Patel, K.K. Quantification of myocardial blood flow using PET: Current status, clinical applications, and future directions. Prog Cardiovasc Dis. 2025, 93, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Chehab, O.; Ambale-Venkatesh, B.; Lima, J.A.C. Quantitative myocardial perfusion imaging across PET, SPECT, CMR, and CT. Front Radiol. 2026, 6, 1760241. [Google Scholar] [CrossRef] [PubMed]
- Sciagrà, R.; Lubberink, M.; Hyafil, F.; Saraste, A.; Slart, R.H.J.A.; Agostini, D.; Nappi, C.; Georgoulias, P.; Bucerius, J.; Rischpler, C.; Verberne, H.J.; Cardiovascular Committee of the European Association of Nuclear Medicine (EANM). EANM procedural guidelines for PET/CT quantitative myocardial perfusion imaging. Eur J Nucl Med Mol Imaging 2021, 48(4), 1040–1069. [Google Scholar] [CrossRef]
- Taqueti, V.R.; Di Carli, M.F. Coronary Microvascular Disease Pathogenic Mechanisms and Therapeutic Options: JACC State-of-the-Art Review. J Am Coll Cardiol. 2018, 72(21), 2625–41. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, G.; Builoff, V.; Miller, R.J.H.; Lemley, M.; Carvajal-Juarez, I.; Alexanderson, E.; Rosamond, T.L.; Song, N.; Travin, M.I.; Slipczuk, L.; et al. Multicenter Evaluation of Myocardial Flow Reserve as a Prognostic Marker for Mortality in 13N-Ammonia PET Myocardial Perfusion Imaging. Circ Cardiovasc Imaging 2026, e018729. [Google Scholar] [CrossRef]
- Valenta, I.; Quercioli, A.; Schindler, T.H. Diagnostic value of PET-measured longitudinal flow gradient for the identification of coronary artery disease. JACC Cardiovasc Imaging PubMed. 2014, 7(4), 387–96. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, K.; Javadi, M.S.; Higuchi, T.; Bravo, P.E.; Chien, D.; Lautamäki, R.; Merrill, J.; Nekolla, S.G.; Bengel, F.M. Impaired global myocardial flow dynamics despite normal left ventricular function and regional perfusion in chronic kidney disease: a quantitative analysis of clinical 82Rb PET/CT studies. J Nucl Med. 2012, 53(6), 887–93. [Google Scholar] [CrossRef] [PubMed]
- deKemp, R.A. Toward improved standardization of PET myocardial blood flow. J Nucl Cardiol. 2023, 30(4), 1297–9. [Google Scholar] [CrossRef] [PubMed]
- Czernin, J.; Müller, P.; Chan, S.; Brunken, R.C.; Porenta, G.; Krivokapich, J.; Chen, K.; Chan, A.; Phelps, M.E.; Schelbert, H.R. Influence of age and hemodynamics on myocardial blood flow and flow reserve. Circulation 1993, 88(1), 62–9. [Google Scholar] [CrossRef]
- Huck, D.M.; Weber, B.N.; Brown, J.M.; Lopez, D.; Hainer, J.; Blankstein, R.; Dorbala, S.; Divakaran, S.; Di Carli, M.F. Prognostic value of myocardial flow reserve vs corrected myocardial flow reserve in patients without obstructive coronary artery disease. J Nucl Cardiol. 2024, 37, 101854. [Google Scholar] [CrossRef] [PubMed]
- Sperry, B.W.; Metzinger, M.P.; Ibrahim, A.O.; Thompson, R.C.; Cho, Y.J.; Jones, P.G.; McGhie, A.I.; Bateman, T.M. Age- and Sex-Specific Myocardial Blood Flow Values in Patients Without Coronary Atherosclerosis on Rb-82 PET Myocardial Perfusion Imaging. Circ Cardiovasc Imaging 2024, 17(7), e016577. [Google Scholar] [CrossRef] [PubMed]
- Packard, R.R.S.; Maddahi, J.; Pelletier-Galarneau, M.; Al-Mallah, M.H.; Coelho, M.; Dorbala, S.; Galt, J.; Hyun, M.; Menon, N.; Miller, E.J.; et al. SNMMI/EANM/ASNC/ACNM Procedure Standard/Practice Guideline for 18F-Flurpiridaz PET Myocardial Perfusion Imaging and Blood Flow Quantitation. J Nucl Med. 2025, 66(10), 1538–1554. [Google Scholar] [CrossRef] [PubMed]
- Bijloo, I.; Woudstra, J.; Aribas, E.; Elias-Smale, S.E.; Vink, C.; Piek, J.J.; Duncker, D.J.; Roeters van Lennep, J.E.; Kavousi, M.; Appelman, Y. Non-invasive and invasive diagnostic modalities for the assessment of coronary vasomotor dysfunction in ANOCA patients. EuroIntervention 2026, 22(5), e283–e291. [Google Scholar] [CrossRef]
- Kotecha, T.; Martinez-Naharro, A.; Boldrini, M.; Knight, D.; Hawkins, P.; Kalra, S.; Patel, D.; Coghlan, G.; Moon, J.; Plein, S.; et al. Automated Pixel-Wise Quantitative Myocardial Perfusion Mapping by CMR to Detect Obstructive Coronary Artery Disease and Coronary Microvascular Dysfunction: Validation Against Invasive Coronary Physiology. JACC Cardiovasc Imaging 2019, 12(10), 1958–1969. [Google Scholar] [CrossRef]
- Hsu, L.Y.; Jacobs, M.; Benovoy, M.; Ta, A.D.; Conn, H.M.; Winkler, S.; Greve, A.M.; Chen, M.Y.; Shanbhag, S.M.; Bandettini, W.P.; et al. Diagnostic Performance of Fully Automated Pixel-Wise Quantitative Myocardial Perfusion Imaging by Cardiovascular Magnetic Resonance. JACC Cardiovasc Imaging 2018, 11(5), 697–707. [Google Scholar] [CrossRef]
- Crawley, R.; Kunze, K.P.; Milidonis, X.; Highton, J.; McElroy, S.; Frey, S.M.; Hoefler, D.; Karamanli, C.; Wong, N.C.K.; Backhaus, S.J.; et al. High-resolution free-breathing automated quantitative myocardial perfusion by cardiovascular magnetic resonance for the detection of functionally significant coronary artery disease. Eur Heart J Cardiovasc Imaging 2024, 25(7), 914–925. [Google Scholar] [CrossRef] [PubMed]
- Messroghli, D.R.; Moon, J.C.; Ferreira, V.M.; Grosse-Wortmann, L.; He, T.; Kellman, P.; Mascherbauer, J.; Nezafat, R.; Salerno, M.; Schelbert, E.B.; et al. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: A consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI). J Cardiovasc Magn Reson. Erratum in: J Cardiovasc Magn Reson. 2018 Feb 7;20(1):9. doi: 10.1186/s12968-017-0408-9. 2017, 19(1), 75. [Google Scholar] [CrossRef] [PubMed]
- Dorfman, A.L.; Geva, T.; Samyn, M.M.; Greil, G.; Krishnamurthy, R.; Messroghli, D.; Festa, P.; Secinaro, A.; Soriano, B.; Taylor, A.; et al. SCMR expert consensus statement for cardiovascular magnetic resonance of acquired and non-structural pediatric heart disease. J Cardiovasc Magn Reson. 2022, 24(1), 44. [Google Scholar] [CrossRef]
- Plein, S.; Kozerke, S.; Suerder, D.; Luescher, T.F.; Greenwood, J.P.; Boesiger, P.; Schwitter, J. High spatial resolution myocardial perfusion cardiac magnetic resonance for the detection of coronary artery disease. Eur Heart J 2008, 29(17), 2148–55. [Google Scholar] [CrossRef]
- Nagel, E.; Greenwood, J.P.; McCann, G.P.; Bettencourt, N.; Shah, A.M.; Hussain, S.T.; Perera, D.; Plein, S.; Bucciarelli-Ducci, C.; Paul, M.; et al. MR-INFORM Investigators. Magnetic Resonance Perfusion or Fractional Flow Reserve in Coronary Disease. N Engl J Med. 2019, 380(25), 2418–2428. [Google Scholar] [CrossRef]
- Ge, Y.; Antiochos, P.; Steel, K.; Bingham, S.; Abdullah, S.; Chen, Y.Y.; Mikolich, J.R.; Arai, A.E.; Bandettini, W.P.; Shanbhag, S.M.; et al. Prognostic Value of Stress CMR Perfusion Imaging in Patients With Reduced Left Ventricular Function. JACC Cardiovasc Imaging 2020, 13(10), 2132–2145. [Google Scholar] [CrossRef]
- Wang, S.; Kim, P.; Wang, H.; Ng, M.Y.; Arai, A.E.; Singh, A.; Mushtaq, S.; Sin, T.H.; Tada, Y.; Hillier, E.; et al. Myocardial Blood Flow Quantification Using Stress Cardiac Magnetic Resonance Improves Detection of Coronary Artery Disease. JACC Cardiovasc Imaging 2024, 17(12), 1428–1441. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Kang, N.; Chung, J.; Gupta, A.R.; Parwani, P. Evaluation of Ischemia with No Obstructive Coronary Arteries (INOCA) and Contemporary Applications of Cardiac Magnetic Resonance (CMR). Medicina (Kaunas) 2023, 59(9), 1570. [Google Scholar] [CrossRef] [PubMed]
- Chiribiri, A.; Arai, A.E.; DiBella, E.; Hsu, L.Y.; Ishida, M.; Jerosch-Herold, M.; Kozerke, S.; Milidonis, X.; Nezafat, R.; Plein, S.; et al. Society for cardiovascular magnetic resonance expert consensus statement on quantitative myocardial perfusion cardiovascular magnetic resonance imaging. J Cardiovasc Magn Reson. 2025, 27(2), 101940. [Google Scholar] [CrossRef]
- Ford, T.J.; Yii, E.; Sidik, N.; Good, R.; Rocchiccioli, P.; McEntegart, M.; Watkins, S.; Eteiba, H.; Shaukat, A.; Lindsay, M.; et al. Ischemia and No Obstructive Coronary Artery Disease: Prevalence and Correlates of Coronary Vasomotion Disorders. Circ Cardiovasc Interv. 2019, 12(12), e008126. [Google Scholar] [CrossRef]
- Namba, H.F.; Boerhout, C.K.M.; Damman, P.; Kunadian, V.; Escaned, J.; Ong, P.; Perera, D.; Berry, C.; van de Hoef, T.P.; Piek, J.J. Invasive coronary function testing in clinical practice: Implementing the 2024 ESC guidelines on chronic coronary syndromes. Int J Cardiol 2025, 430, 133176. [Google Scholar] [CrossRef] [PubMed]
- Fearon, W.F.; Kobayashi, Y. Invasive Assessment of the Coronary Microvasculature: The Index of Microcirculatory Resistance. Circ Cardiovasc Interv 2017, 10(12), e005361. [Google Scholar] [CrossRef]
- Gallinoro, E.; Bertolone, D.T.; Fernandez-Peregrina, E.; Paolisso, P.; Bermpeis, K.; Esposito, G.; Gomez-Lopez, A.; Candreva, A.; Mileva, N.; Belmonte, M.; et al. Reproducibility of bolus versus continuous thermodilution for assessment of coronary microvascular function in patients with ANOCA. EuroIntervention 2023, 19(2), e155–e166. [Google Scholar] [CrossRef]
- Rahman, H.; Ryan, M.; Lumley, M.; Modi, B.; McConkey, H.; Ellis, H.; Scannell, C.; Clapp, B.; Marber, M.; Webb, A.; et al. Coronary Microvascular Dysfunction Is Associated With Myocardial Ischemia and Abnormal Coronary Perfusion During Exercise. Circulation 2019, 140(22), 1805–1816. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, X.; Feng, W.; Zhou, H.; Peng, W.; Wang, X. Diagnostic and prognostic value of angiography-derived index of microvascular resistance: a systematic review and meta-analysis. Front Cardiovasc Med. 2024, 11, 1360648. [Google Scholar] [CrossRef] [PubMed]
- Montone, R.A.; Rinaldi, R.; Del Buono, M.G.; Gurgoglione, F.; La Vecchia, G.; Russo, M.; Caffè, A.; Burzotta, F.; Leone, A.M.; Romagnoli, E.; et al. Safety and prognostic relevance of acetylcholine testing in patients with stable myocardial ischaemia or myocardial infarction and non-obstructive coronary arteries. EuroIntervention 2022, 18(8), e666–e676. [Google Scholar] [CrossRef] [PubMed]
- Ashokprabhu, N.D.; Quesada, O.; Alvarez, Y.R.; Henry, T.D. INOCA/ANOCA: Mechanisms and novel treatments. Am Heart J Plus 2023, 30, 100302. [Google Scholar] [CrossRef]
- Montone, R.A.; Rinaldi, R.; Niccoli, G.; Andò, G.; Gragnano, F.; Piccolo, R.; Pelliccia, F.; Moscarella, E.; Zimarino, M.; Fabris, E.; et al. Interventional Cardiology Working Group of the Italian Society of Cardiology. Optimizing Management of Stable Angina: A Patient-Centered Approach Integrating Revascularization, Medical Therapy, and Lifestyle Interventions. J Am Coll Cardiol. 2024, 84(8), 744–760. [Google Scholar] [CrossRef] [PubMed]
- Masiero, G.; Aurigemma, C.; Biscaglia, S.; Bruno, F.; Burzotta, F.; Campo, G.; Cangemi, S.; Chieffo, A.; Di Mario, C.; Esposito, G.; et al. Implementazione delle nuove raccomandazioni sull’impiego dei test funzionali coronarici e dell’imaging intravascolare nell’angioplastica coronarica per le sindromi coronariche croniche: documento di consenso GISE/ANMCO/SIC [GISE/ANMCO/SIC Consensus document on the implementation of the new recommendations for the use of coronary functional testing and intravascular imaging in coronary angioplasty for chronic coronary syndromes]. G Ital Cardiol (Rome);Italian 2025, 26(12), 941–953. [Google Scholar] [CrossRef]
| Modality/Test | Key outputs | Endotype signal | Clinical role | Limitations | Therapeutic implications |
| CCTA | Stenosis severity; plaque burden and phenotype | Non-obstructive plaque → atherosclerotic substrate; normal CCTA does not exclude CMD or vasospasm | Anatomical gatekeeper; selection for functional testing | Heavy calcification; motion artefacts | Intensify preventive therapy; proceed to functional testing if symptoms persist |
| Stress PET (MBF/MFR) | Rest and stress MBF; global and regional MFR | Global reduction in MFR/MBF → structural CMD or diffuse coronary dysfunction | First-/second-line functional assessment | Haemodynamic variability; tracer and software differences; limited detection of spasm | Supports CMD diagnosis; risk stratification; guides cardiometabolic optimisation |
| Stress CMR (quantitative) | Stress MBF/MPR; regional perfusion; LGE; T1/ECV | Extesnvie perfusion impairment → CMD; tissue abnormalities may suggest alternative myocardial pathology” | Alternative to PET; integrated “one-stop” evaluation | Arrhythmias; motion artefacts; variability in analysis pipelines | Guides CMD management; avoids misclassification; identifies alternative myocardial pathology |
| ICA + FFR/iFR | Epicardial physiology | Flow-limiting epicardial disease | Assessment of obstructive CAD prior to ICFT; excludes focal flow-limiting epicardial disease | Does not directly assess coronary blood flow or microvascular dysfunction; non-focal atherosclerotic disease may affect interpretation | Guides revascularisation decisions; a normal FFR/iFR does not exclude coronary vascular dysfunction |
| CFR (wire-based) | Coronary flow reserve | Reduced CFR suggests CMD but lacks specificity | Component of ICFT | Influenced by haemodynamics and epicardial disease | Requires integration with resistance indices and vasoreactivity testing |
| Microvascular resistance (IMR) | Microvascular resistance index | Elevated resistance → structural CMD | Core component of microvascular assessment | Technique-dependent | Supports CMD-targeted therapy; avoids inappropriate vasodilator-only strategies |
| Acetylcholine provocation | Vasomotor response; symptoms; ECG changes | Epicardial vs microvascular spasm; mixed phenotypes | Diagnosis of vasospastic disorders | Requires expertise; protocol variability | Calcium-channel blockers first-line; nitrates and trigger modification |
| Integrated ICFT | Comprehensive physiological assessment | CMD, vasospasm, or mixed endotypes | ESC-recommended in persistent symptoms | Requires dedicated infrastructure | Enables stratified therapy; improves symptoms and quality of life |
| Endotype | Pathophysiology | ICFT findings (with cut-offs) (FFR ≤0.80 indicates haemodynamically significant epicardial disease) | Clinical implication | Treatment approach |
| Structural CMD | Microvascular remodelling, increased minimal resistance | ↓ CFR (<2.0–2.5 depending on methodology) + ↑ IMR (≥25) | Impaired vasodilatory capacity | Risk factor optimisation; ACEi/ARB; statins |
| Functional CMD | Endothelial dysfunction, impaired vasomotor regulation | ↓ CFR (<2.0–2.5 depending on methodology) + normal IMR (<25); ± abnormal vasoreactivity | Dynamic microvascular dysfunction | Endothelial-targeted therapy; antianginal agents |
| Epicardial vasospasm | Vascular smooth muscle hyperreactivity | ACh-induced epicardial constriction ≥90% + symptoms and/or ECG changes | Variant angina phenotype | Calcium-channel blockers; nitrates |
| Microvascular spasm | Microvascular hyperreactivity | ACh-induced symptoms + ECG changes without epicardial constriction (<90%) | INOCA with vasomotor dysfunction | Calcium-channel blockers; tailored antianginal therapy |
| Mixed endotype | Overlapping CMD and vasospastic mechanisms | Combination of ↓ CFR and/or ↑ IMR with abnormal ACh response | Complex and heterogeneous phenotype | Combination therapy targeting CMD and vasospasm |
| Endotype | Key diagnostic features | Pathophysiology | Mechanistic target | First-line therapy | Adjunctive therapy | Clinical pearls / pitfalls |
| Structural CMD | ↓ MFR (PET/CMR); ↓ CFR + ↑ IMR | Impaired vasodilatory capacity; microvascular remodelling; increased minimal resistance | Improve microvascular vasodilation and reduce resistance | Risk factor control; beta-blockers | ACEi/ARB; statins; ranolazine (selected patients) | Systemic cardiometabolic disease; prioritise risk factor optimisation over escalation of antianginal therapy; avoid reassurance based on “normal angiogram” |
| Functional / endothelial CMD | ↓ CFR with normal or mildly ↑ IMR; ± abnormal vasoreactivity | Endothelial dysfunction; impaired NO-mediated vasodilation; altered vascular tone regulation | Restore endothelial function and nitric oxide bioavailability | Risk factor control | ACEi/ARB; statins; lifestyle intervention; ranolazine or ivabradine (selected patients) | Heterogeneous phenotype; frequent overlap with vasospasm; variable therapeutic response; reassess if symptoms persist |
| Epicardial vasospasm (VSA) | ACh-induced epicardial constriction ≥90% + symptoms and/or ECG changes | Vascular smooth muscle hyperreactivity; endothelial dysfunction | Suppress abnormal vasoconstriction and smooth muscle hyperreactivity | Calcium-channel blockers | Nitrates; nicorandil (selected cases) | Avoid triggers (smoking, stress, sympathomimetics); avoid non-selective beta-blockers; high response to vasodilator therapy |
| Microvascular spasm | ACh-induced symptoms and ECG changes without epicardial constriction (<90%) | Microvascular hyperreactivity; abnormal vasomotor regulation | Reduce microvascular vasoconstriction | Calcium-channel blockers | Nitrates; tailored antianginal therapy | Diagnosis requires invasive testing; often underdiagnosed; symptoms may be disproportionate to imaging findings; response less predictable |
| Mixed endotypes | Combination of ↓ CFR and/or ↑ IMR with abnormal ACh response | Overlapping CMD and vasospastic mechanisms | Combined modulation of vasodilation and vasoconstriction pathways | Combination therapy (CCB + CMD-directed therapy) | Individualised strategy | Most common phenotype; avoid single-mechanism treatment; requires integrated interpretation of physiology |
| Non-obstructive atherosclerosis (modifier) | Plaque on CCTA/ICA without flow-limiting stenosis | Diffuse atherosclerotic burden; endothelial dysfunction; inflammatory activation | Stabilise plaque and reduce atherogenic and inflammatory burden | Lipid-lowering therapy; risk factor control | Antiplatelet therapy (selected cases) | Major prognostic driver even without stenosis; do not undertreat; integrate with CMD management |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




