Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
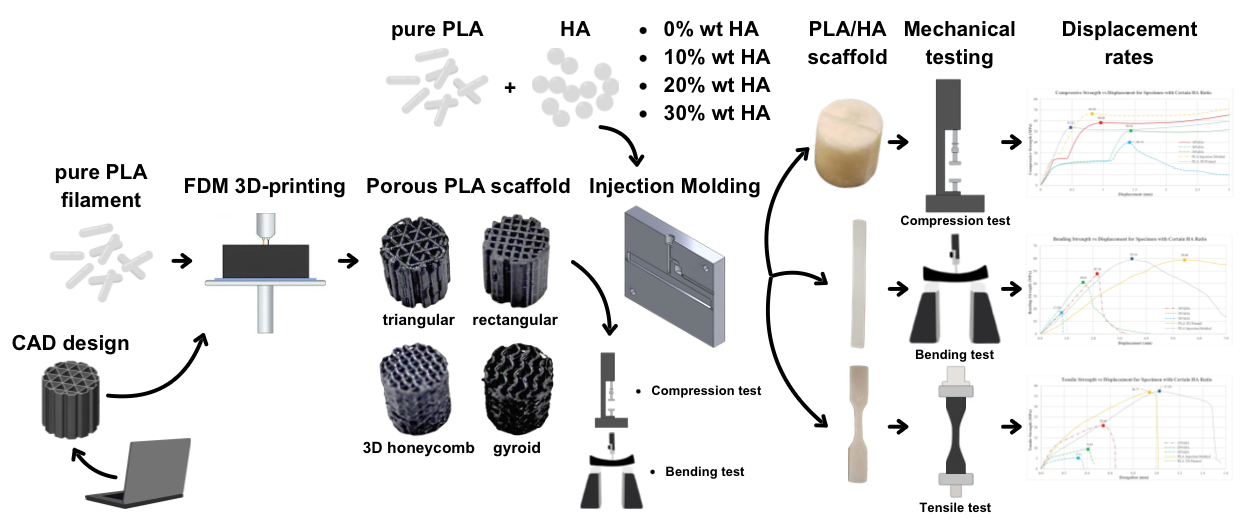
2.1. Materials
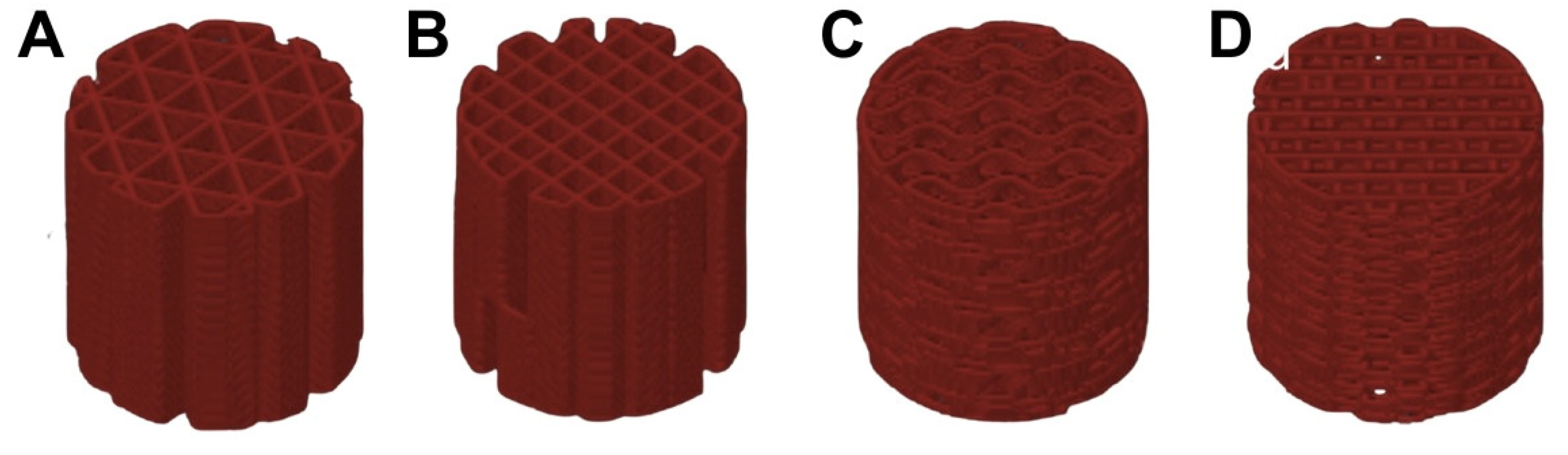
2.2. Scaffold Design
2.3. Fabrication of Scaffolds
2.4. Determination of Mechanical Properties
3. Results and Discussion
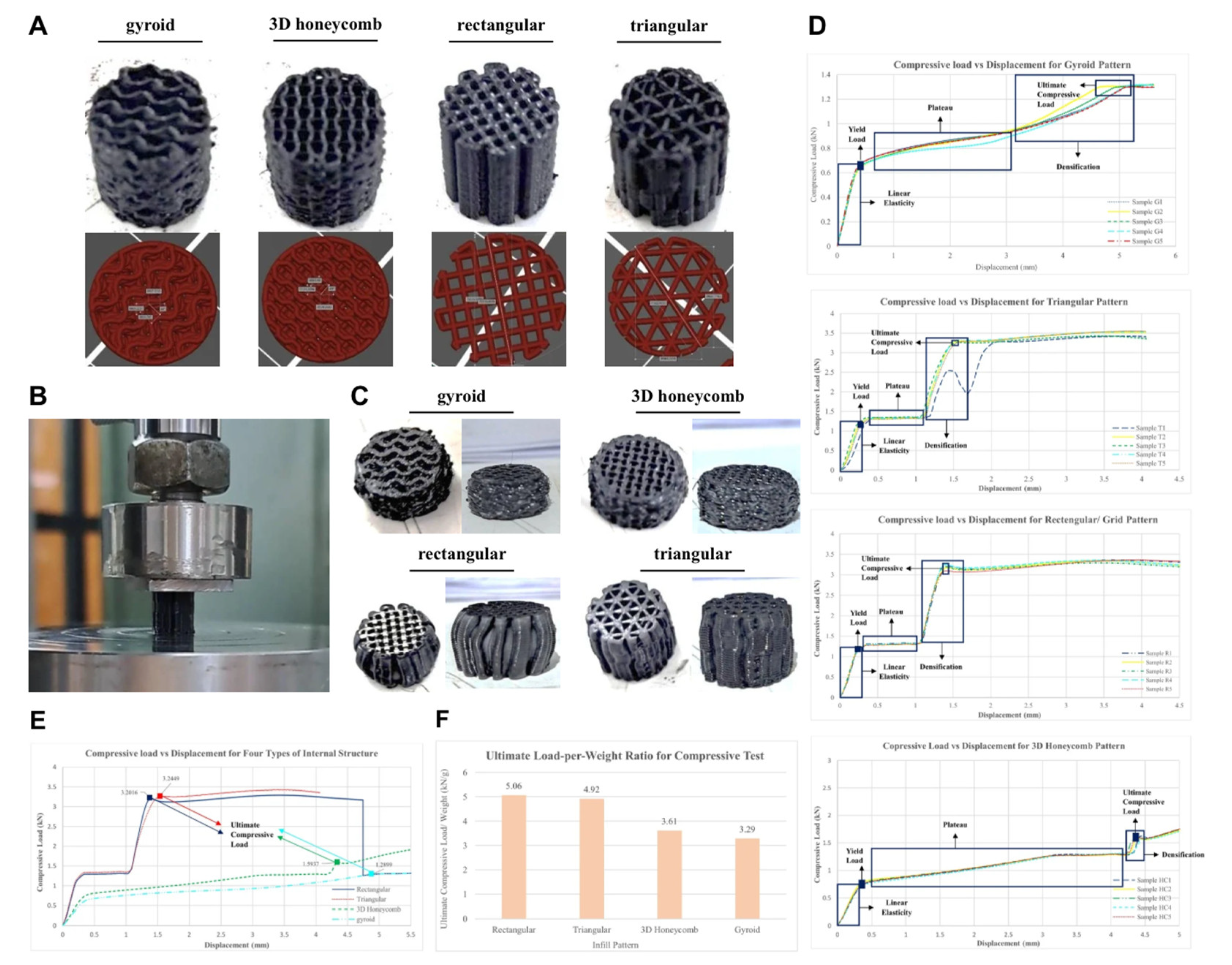
3.1. Fabrication and Characterization of 3D-Printed PLA/HA Composite Scaffolds
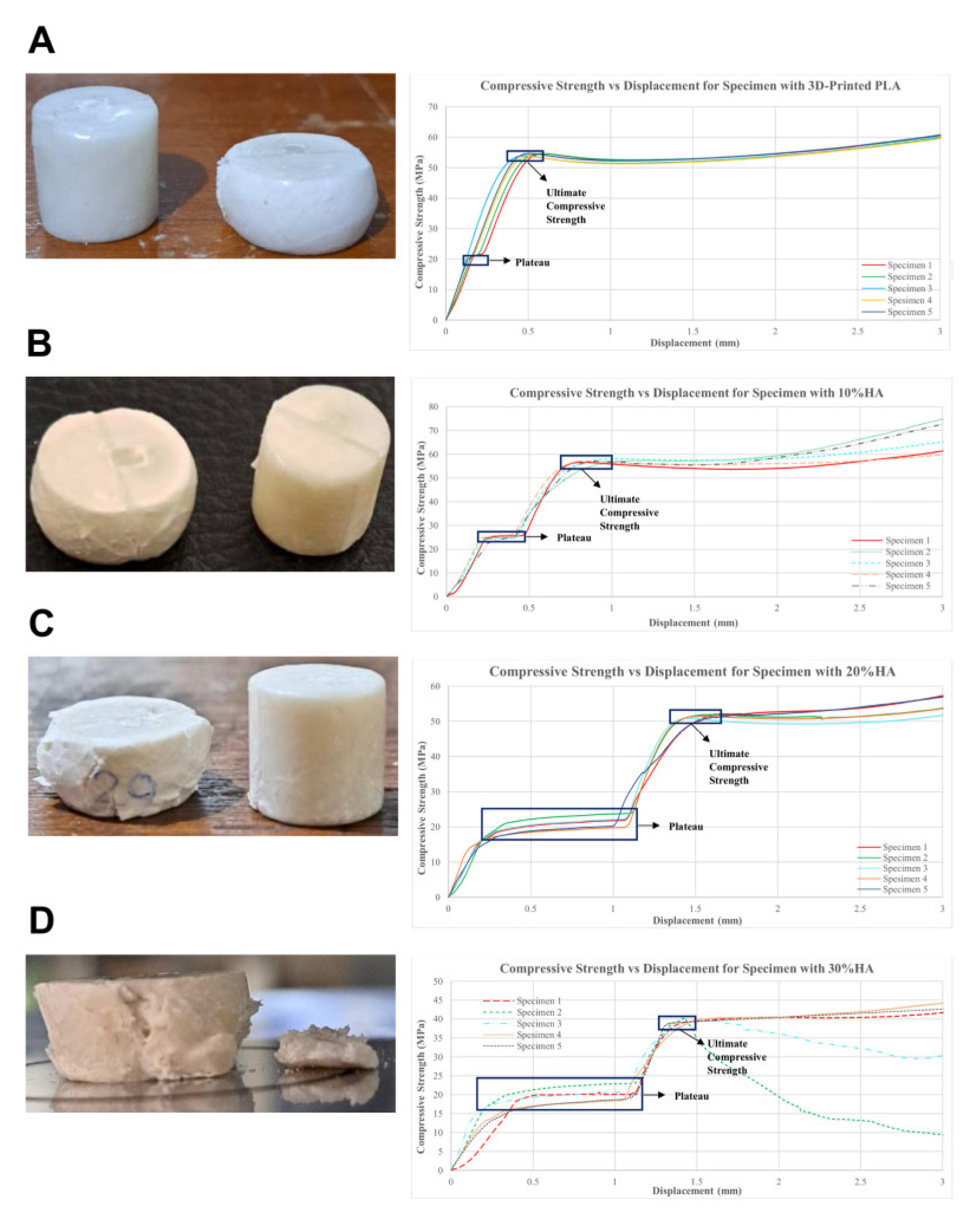
3.2. Compression Test Results
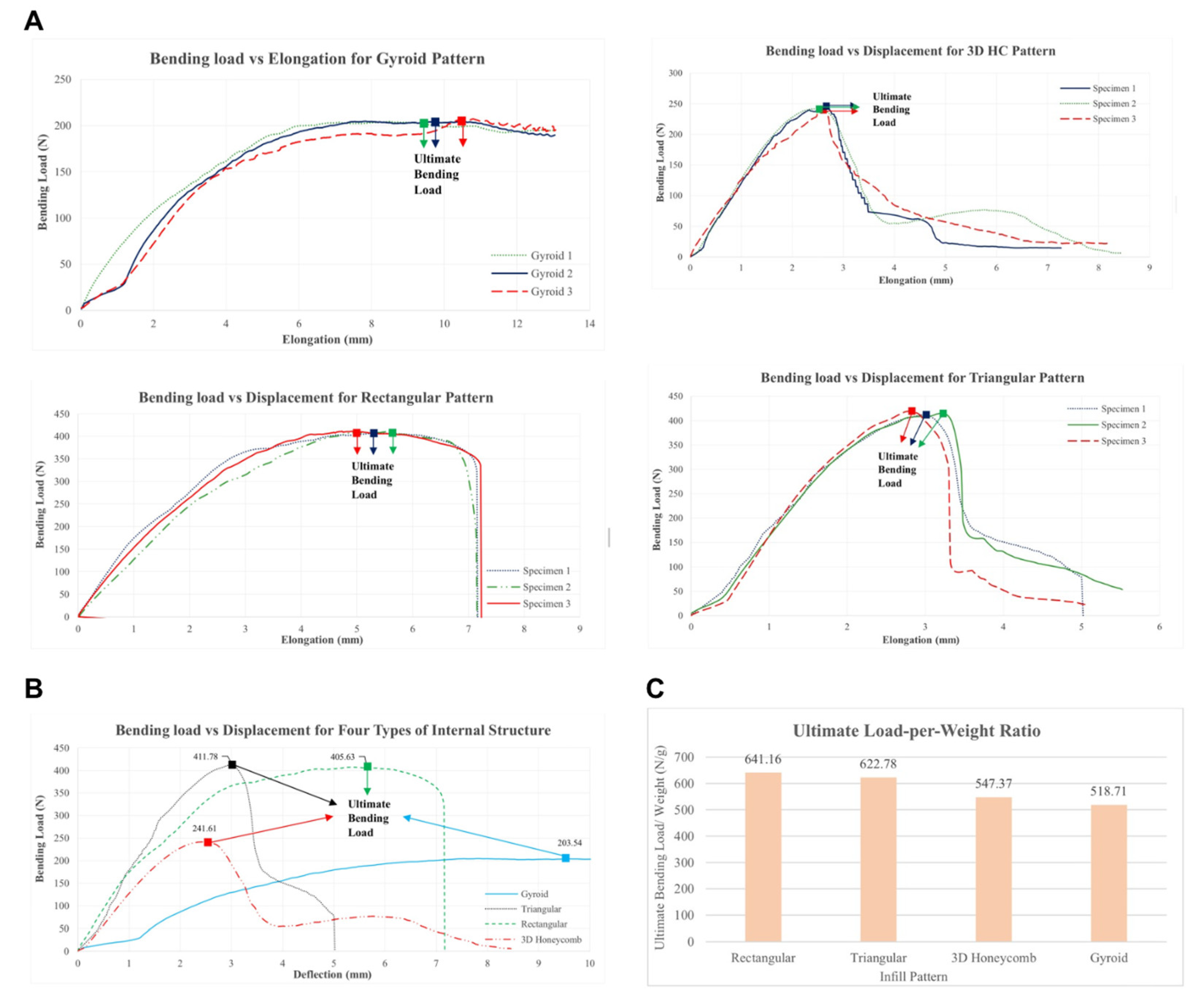
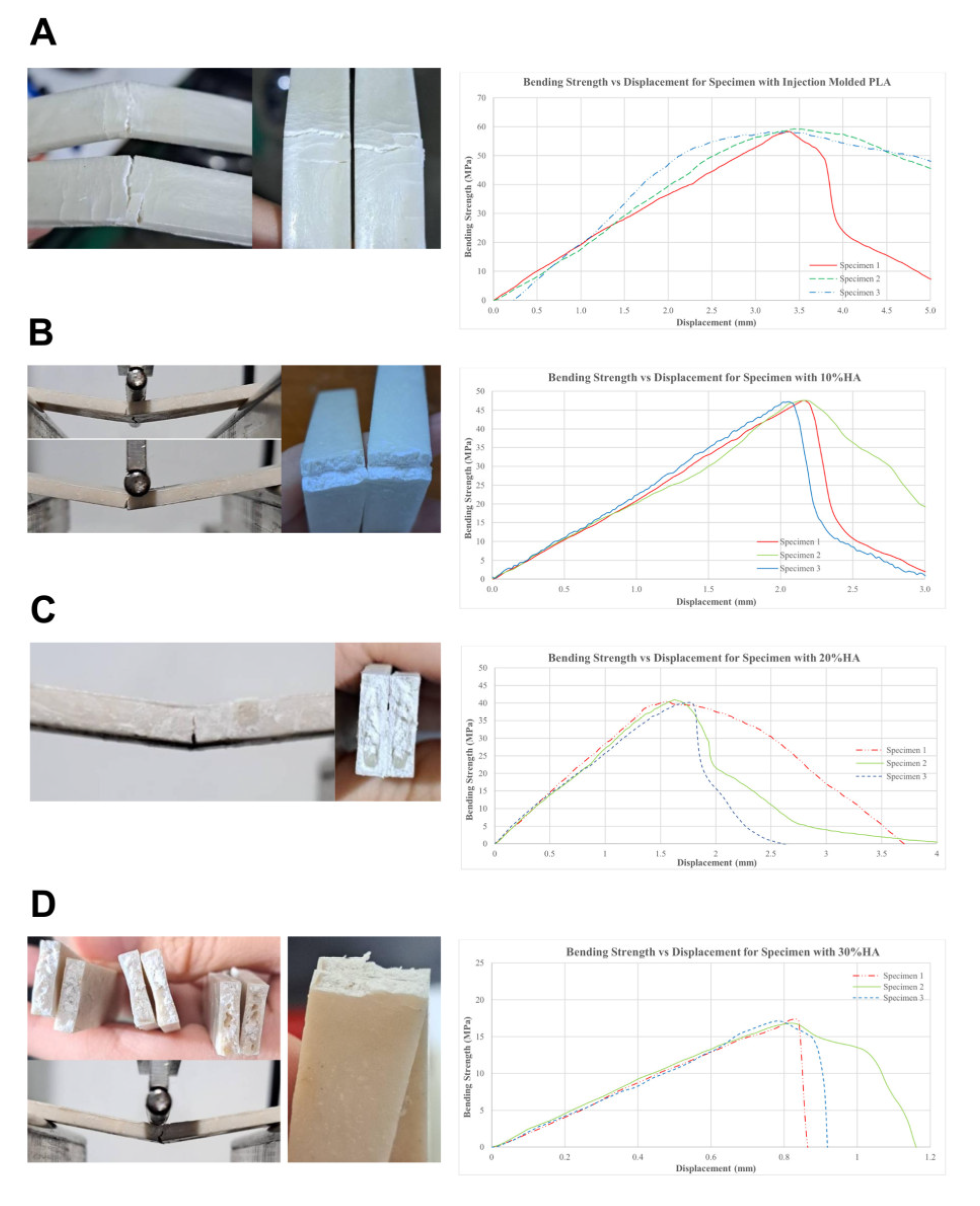
3.3. Bending Test Results
3.4. Effects of HA on Mechanical Properties of PLA scaffolds
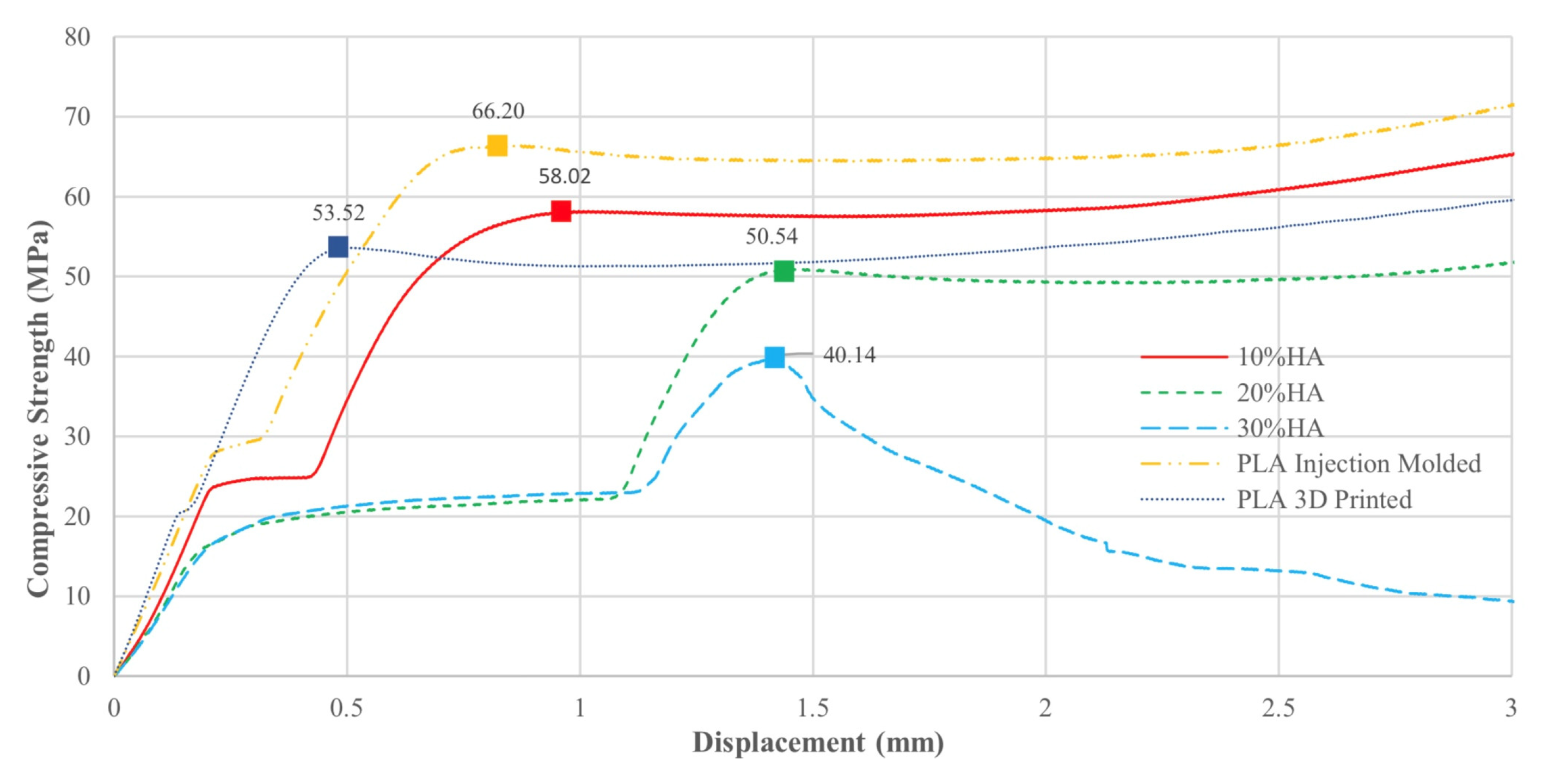
3.4.1. Compressive Test
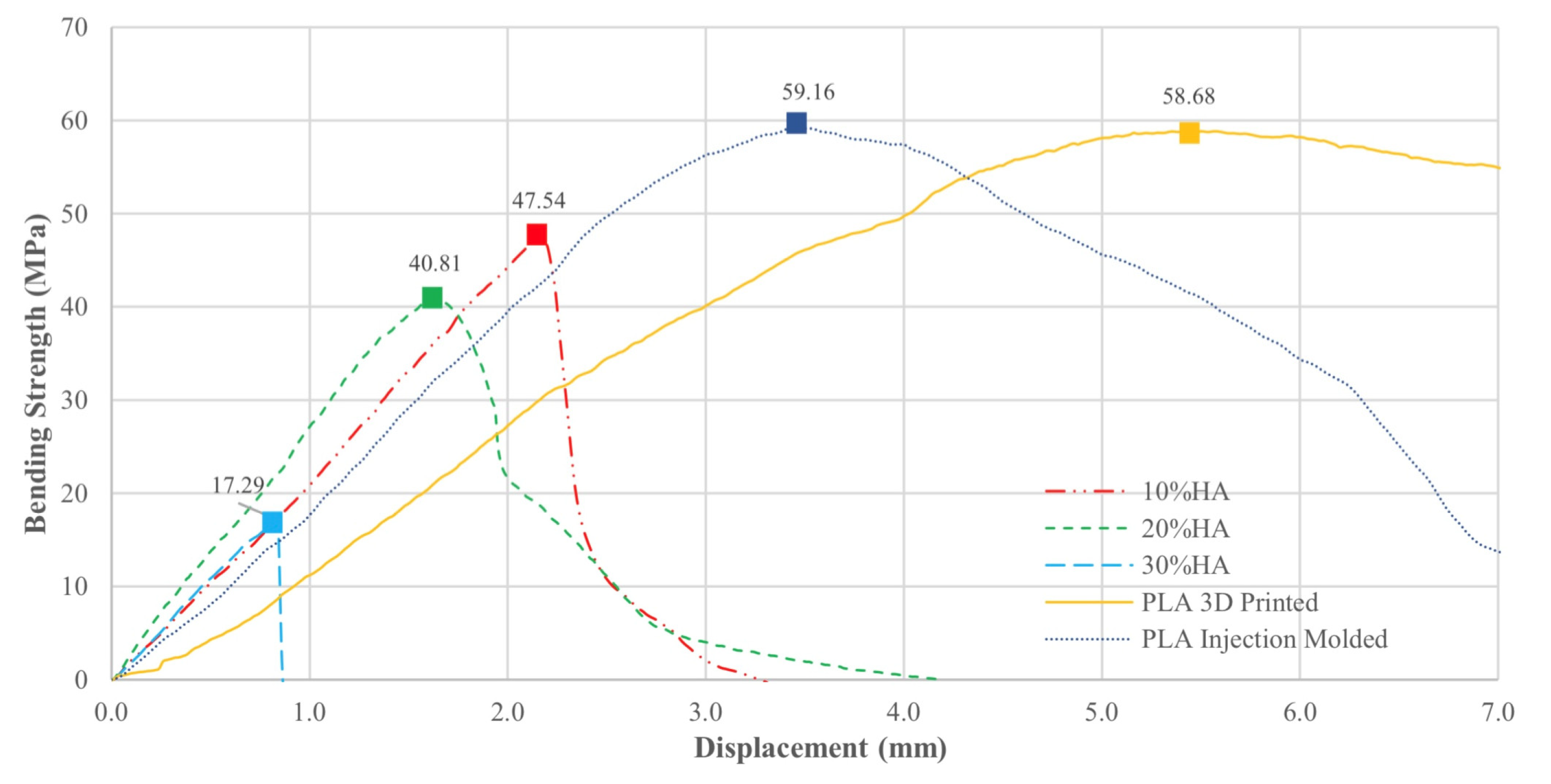
3.4.2. Bending Test
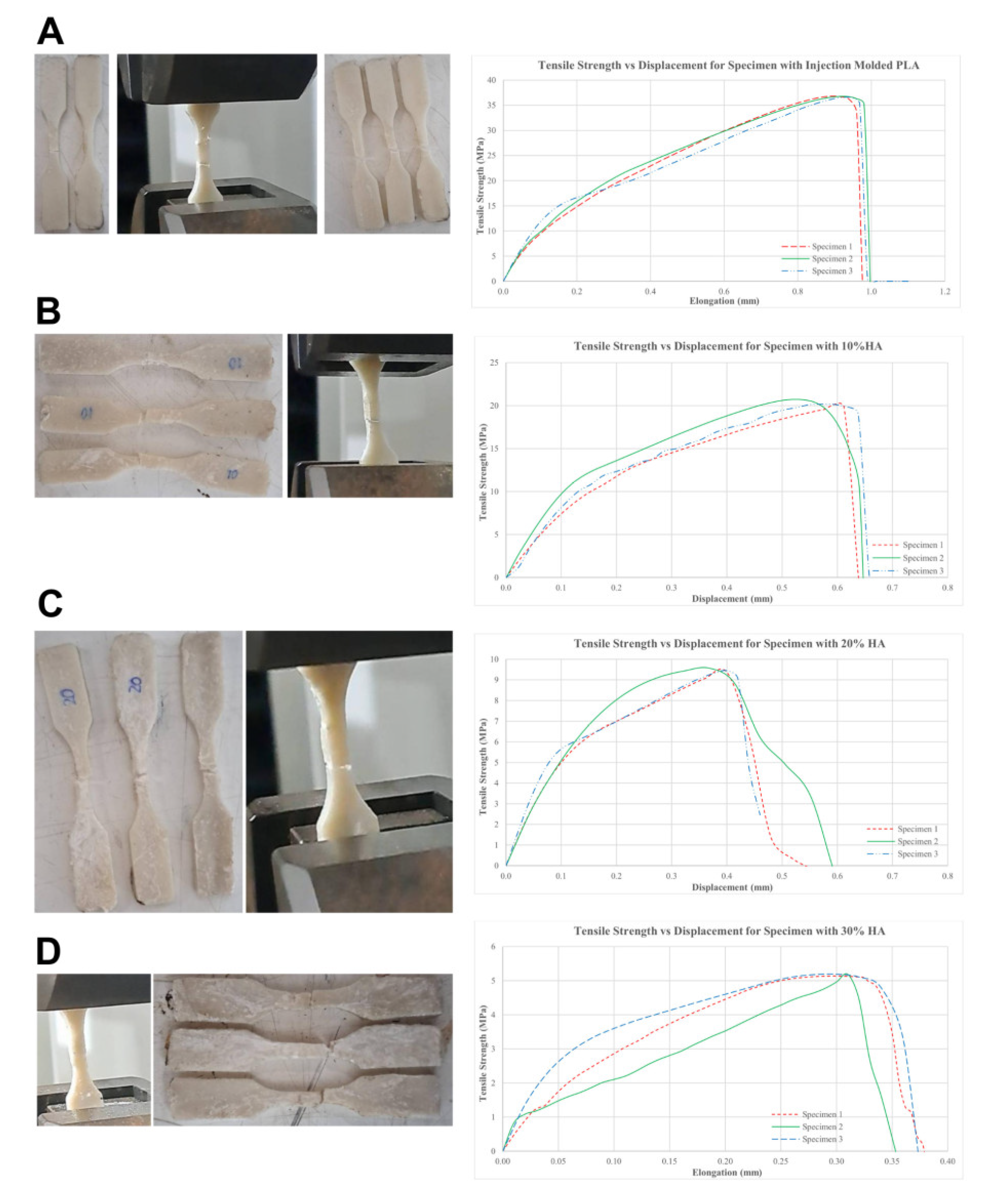
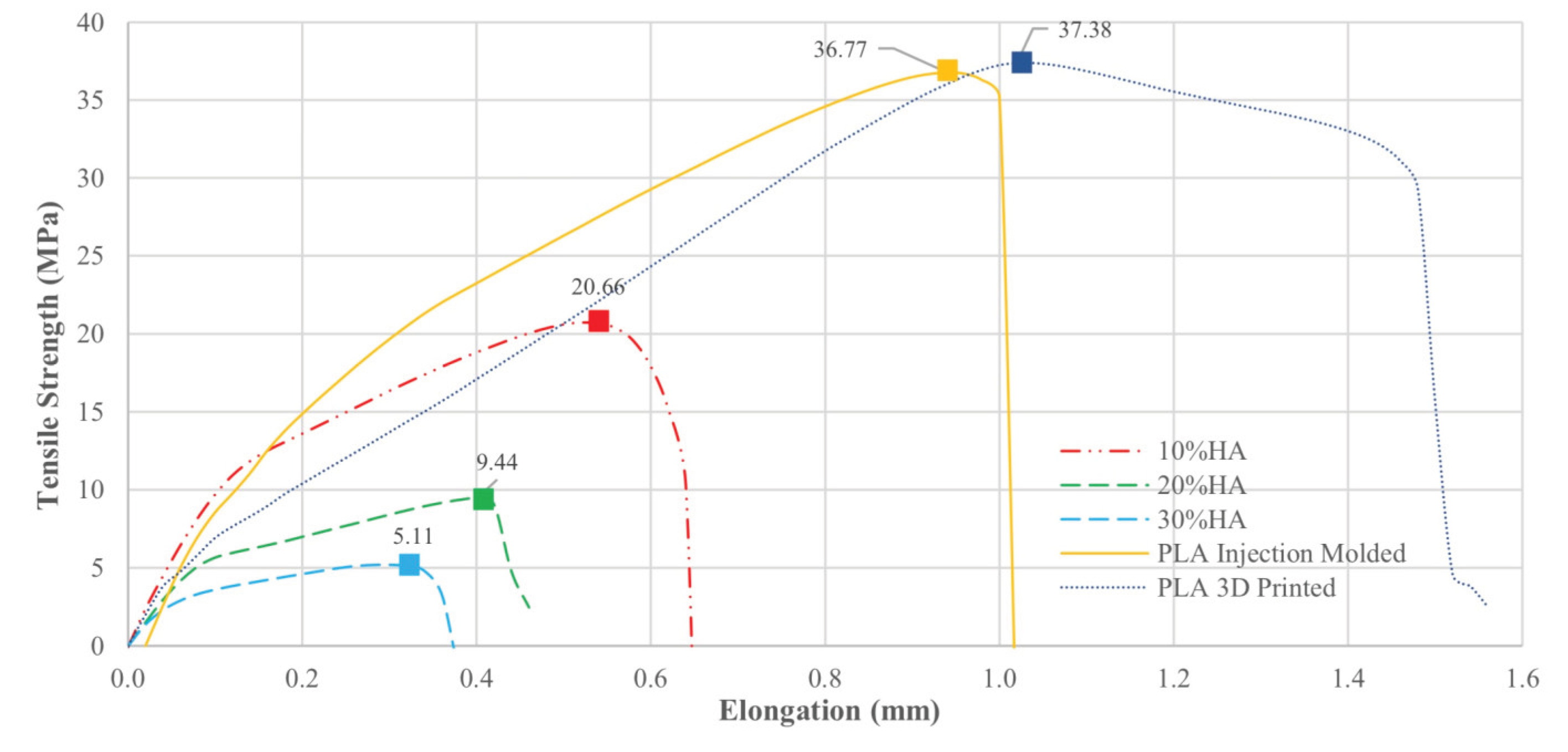
3.4.3. Tensile Test
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PLA | Polylactic acid |
| HA | Hydroxyapatite |
| n-HA | Nano-hydroxyapatite |
| FDM | Fused-deposition modeling |
| 3D | Three-dimensional |
| TPMS | Triply periodic minimal surface |
References
- Qin, L.; Yang, S.; Zhao, C.; Yang, J.; Li, F.; Xu, Z.; Yang, Y.; Zhou, H.; Li, K.; Xiong, C.; et al. Prospects and Challenges for the Application of Tissue Engineering Technologies in the Treatment of Bone Infections. Bone Res 2024, 12, 28. [Google Scholar] [CrossRef]
- Heng, B.C.; Bai, Y.; Li, X.; Lim, L.W.; Li, W.; Ge, Z.; Zhang, X.; Deng, X. Electroactive Biomaterials for Facilitating Bone Defect Repair under Pathological Conditions. Advanced Science 2023, 10, 2204502. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Yue, W.; Qin, W.; Zhao, Y.; Xu, G. Research Progress of Bone Grafting: A Comprehensive Review. IJN 2025, Volume 20, 4729–4757. [Google Scholar] [CrossRef] [PubMed]
- Archunan, M.W.; Petronis, S. Bone Grafts in Trauma and Orthopaedics. Cureus 2021. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, P.; Li, D.J.; Auston, D.A.; Mir, H.S.; Yoon, R.S.; Koval, K.J. Autograft, Allograft, and Bone Graft Substitutes: Clinical Evidence and Indications for Use in the Setting of Orthopaedic Trauma Surgery. Journal of Orthopaedic Trauma 2019, 33, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yeung, K.W.K. Bone Grafts and Biomaterials Substitutes for Bone Defect Repair: A Review. Bioactive Materials 2017, 2, 224–247. [Google Scholar] [CrossRef]
- Tournier, P.; Guicheux, J.; Paré, A.; Maltezeanu, A.; Blondy, T.; Veziers, J.; Vignes, C.; André, M.; Lesoeur, J.; Barbeito, A.; et al. A Partially Demineralized Allogeneic Bone Graft: In Vitro Osteogenic Potential and Preclinical Evaluation in Two Different Intramembranous Bone Healing Models. Sci Rep 2021, 11, 4907. [Google Scholar] [CrossRef]
- Sohn, H.-S.; Oh, J.-K. Review of Bone Graft and Bone Substitutes with an Emphasis on Fracture Surgeries. Biomater Res 2019, 23, 9. [Google Scholar] [CrossRef]
- Bexkens, R.; Ogink, P.T.; Doornberg, J.N.; Kerkhoffs, G.M.M.J.; Eygendaal, D.; Oh, L.S.; Van Den Bekerom, M.P.J. Donor-Site Morbidity after Osteochondral Autologous Transplantation for Osteochondritis Dissecans of the Capitellum: A Systematic Review and Meta-Analysis. Knee Surg Sports Traumatol Arthrosc 2017, 25, 2237–2246. [Google Scholar] [CrossRef]
- Percival, K.M.; Paul, V.; Husseini, G.A. Recent Advancements in Bone Tissue Engineering: Integrating Smart Scaffold Technologies and Bio-Responsive Systems for Enhanced Regeneration. IJMS 2024, 25, 6012. [Google Scholar] [CrossRef]
- Qi, J.; Yu, T.; Hu, B.; Wu, H.; Ouyang, H. Current Biomaterial-Based Bone Tissue Engineering and Translational Medicine. IJMS 2021, 22, 10233. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, G.; Clarke, J.; Picard, F.; Riches, P.; Jia, L.; Han, F.; Li, B.; Shu, W. 3D Bioactive Composite Scaffolds for Bone Tissue Engineering. Bioactive Materials 2018, 3, 278–314. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Fernández, I.; Haugen, H.J.; López-Peña, M.; González-Cantalapiedra, A.; Muñoz, F. Use of 3D-Printed Polylactic Acid/Bioceramic Composite Scaffolds for Bone Tissue Engineering in Preclinical in Vivo Studies: A Systematic Review. Acta Biomaterialia 2023, 168, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, L.; Song, P.; Pei, X.; Sun, H.; Wu, L.; Zhou, C.; Wang, K.; Fan, Y.; Zhang, X. 3D Printed Bone Tissue Regenerative PLA/HA Scaffolds with Comprehensive Performance Optimizations. Materials & Design 2021, 201, 109490. [Google Scholar] [CrossRef]
- Yang, Z.; Yin, G.; Sun, S.; Xu, P. Medical Applications and Prospects of Polylactic Acid Materials. iScience 2024, 27, 111512. [Google Scholar] [CrossRef]
- Bernardo, M.P.; Da Silva, B.C.R.; Hamouda, A.E.I.; De Toledo, M.A.S.; Schalla, C.; Rütten, S.; Goetzke, R.; Mattoso, L.H.C.; Zenke, M.; Sechi, A. PLA/Hydroxyapatite Scaffolds Exhibit in Vitro Immunological Inertness and Promote Robust Osteogenic Differentiation of Human Mesenchymal Stem Cells without Osteogenic Stimuli. Sci Rep 2022, 12, 2333. [Google Scholar] [CrossRef]
- Chi, M.; Li, N.; Cui, J.; Karlin, S.; Rohr, N.; Sharma, N.; Thieringer, F.M. Biomimetic, Mussel-Inspired Surface Modification of 3D-Printed Biodegradable Polylactic Acid Scaffolds with Nano-Hydroxyapatite for Bone Tissue Engineering. Front. Bioeng. Biotechnol. 2022, 10, 989729. [Google Scholar] [CrossRef]
- Gendviliene, I.; Simoliunas, E.; Alksne, M.; Dibart, S.; Jasiuniene, E.; Cicenas, V.; Jacobs, R.; Bukelskiene, V.; Rutkunas, V. Effect of Extracellular Matrix and Dental Pulp Stem Cells on Bone Regeneration with 3D Printed PLA/HA Composite Scaffolds. eCM 2021, 41, 204–215. [Google Scholar] [CrossRef]
- Fiume, E.; Magnaterra, G.; Rahdar, A.; Verné, E.; Baino, F. Hydroxyapatite for Biomedical Applications: A Short Overview. Ceramics 2021, 4, 542–563. [Google Scholar] [CrossRef]
- Ramesh, N.; Moratti, S.C.; Dias, G.J. Hydroxyapatite–Polymer Biocomposites for Bone Regeneration: A Review of Current Trends. J Biomed Mater Res 2018, 106, 2046–2057. [Google Scholar] [CrossRef]
- Shariful Islam, M.; Abdulla-Al-Mamun, M.; Khan, A.; Todo, M. Excellency of Hydroxyapatite Composite Scaffolds for Bone Tissue Engineering. In Biomaterials; Vizureanu, P., Manuela Da Cunha Ferreira Botelho, C., Eds.; IntechOpen, 2020; ISBN 978-1-78984-464-1. [Google Scholar]
- Feng, C.; Zhang, K.; He, R.; Ding, G.; Xia, M.; Jin, X.; Xie, C. Additive Manufacturing of Hydroxyapatite Bioceramic Scaffolds: Dispersion, Digital Light Processing, Sintering, Mechanical Properties, and Biocompatibility. J Adv Ceram 2020, 9, 360–373. [Google Scholar] [CrossRef]
- del-Mazo-Barbara, L.; Johansson, L.; Tampieri, F.; Ginebra, M.-P. Toughening 3D Printed Biomimetic Hydroxyapatite Scaffolds: Polycaprolactone-Based Self-Hardening Inks. Acta Biomaterialia 2024, 177, 506–524. [Google Scholar] [CrossRef] [PubMed]
- Soleymani, S.; Naghib, S.M. 3D and 4D Printing Hydroxyapatite-Based Scaffolds for Bone Tissue Engineering and Regeneration. Heliyon 2023, 9, e19363. [Google Scholar] [CrossRef] [PubMed]
- Mo, X.; Zhang, D.; Liu, K.; Zhao, X.; Li, X.; Wang, W. Nano-Hydroxyapatite Composite Scaffolds Loaded with Bioactive Factors and Drugs for Bone Tissue Engineering. IJMS 2023, 24, 1291. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Mao, X.; Du, Z.; Jiang, W.; Han, X.; Zhao, D.; Han, D.; Li, Q. Three Dimensional Printed Macroporous Polylactic Acid/Hydroxyapatite Composite Scaffolds for Promoting Bone Formation in a Critical-Size Rat Calvarial Defect Model. Science and Technology of Advanced Materials 2016, 17, 136–148. [Google Scholar] [CrossRef]
- Vega, G.; Paz, R.; Monzón, M.; Donate, R.; Gleadall, A. Curve-Based Infill Pattern Optimization for 3D Printed Polymeric Scaffolds for Trabecular Bone Applications. Materials 2025, 18, 4055. [Google Scholar] [CrossRef]
- Adel, A.; Adly, M.; Reda, R.; Abdelkawy, A. Comparative Structural and Mechanical Characterization of FDM 3D -Printed PLA Scaffolds for Bone Tissue Engineering Applications. Polymers for Advanced Technologies 2025, 36. [Google Scholar] [CrossRef]
- Guo, W.; Yang, Y.; Liu, C.; Bu, W.; Guo, F.; Li, J.; Wang, E.; Peng, Z.; Mai, H.; You, H.; et al. 3D Printed TPMS Structural PLA/GO Scaffold: Process Parameter Optimization, Porous Structure, Mechanical and Biological Properties. Journal of the Mechanical Behavior of Biomedical Materials 2023, 142, 105848. [Google Scholar] [CrossRef]
- Winarso, R.; Anggoro, P.W.; Ismail, R.; Jamari, J.; Bayuseno, A.P. Application of Fused Deposition Modeling (FDM) on Bone Scaffold Manufacturing Process: A Review. Heliyon 2022, 8, e11701. [Google Scholar] [CrossRef]
- Qu, H. Additive Manufacturing for Bone Tissue Engineering Scaffolds. Materials Today Communications 2020, 24, 101024. [Google Scholar] [CrossRef]
- Grémare, A.; Guduric, V.; Bareille, R.; Heroguez, V.; Latour, S.; L’heureux, N.; Fricain, J.-C.; Catros, S.; Le Nihouannen, D. Characterization of Printed PLA Scaffolds for Bone Tissue Engineering. Journal of Biomedical Materials Research Part A 2018, 106, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Pernet, B.; Nagel, J.; Zhang, H. Compressive Strength Assessment of 3D Printing Infill Patterns. Procedia CIRP 2022, 105, 682–687. [Google Scholar] [CrossRef]
- Ma, Q.; Rejab, R.; Kumar, A.P.; Fu, H.; Nallapaneni, M.K.; Tang, J. Effect of Infill Pattern, Density and Material Type of 3D Printed Cubic Structure under Quasi-Static Loading. Proceedings of the Institution of Mechanical Engineers, Part C: Journal of Mechanical Engineering Science 2020, 1, 1–19. [Google Scholar] [CrossRef]
- Guan, R.; Smith, D. Influence of Infill Parameters on the Tensile Mechanical Properties of 3D Printed Parts. J Emerg Invest 2020. [Google Scholar] [CrossRef]
- Lu, Y.; Cheng, L.; Yang, Z.; Li, J.; Zhu, H. Relationship between the Morphological, Mechanical and Permeability Properties of Porous Bone Scaffolds and the Underlying Microstructure. PLoS ONE 2020, 15, e0238471. [Google Scholar] [CrossRef]
- Garcia Garcia, A.; Hébraud, A.; Duval, J.-L.; Wittmer, C.R.; Gaut, L.; Duprez, D.; Egles, C.; Bedoui, F.; Schlatter, G.; Legallais, C. Poly(ε-Caprolactone)/Hydroxyapatite 3D Honeycomb Scaffolds for a Cellular Microenvironment Adapted to Maxillofacial Bone Reconstruction. ACS Biomater. Sci. Eng. 2018, 4, 3317–3326. [Google Scholar] [CrossRef]
- Zhao, H.; Li, L.; Ding, S.; Liu, C.; Ai, J. Effect of Porous Structure and Pore Size on Mechanical Strength of 3D-Printed Comby Scaffolds. Materials Letters 2018, 223. [Google Scholar] [CrossRef]
- Swanson, W.B.; Omi, M.; Zhang, Z.; Nam, H.K.; Jung, Y.; Wang, G.; Ma, P.X.; Hatch, N.E.; Mishina, Y. Macropore Design of Tissue Engineering Scaffolds Regulates Mesenchymal Stem Cell Differentiation Fate. Biomaterials 2021, 272, 120769. [Google Scholar] [CrossRef]
- Huang, J.; Liu, W.; Liang, Y.; Li, L.; Duan, L.; Chen, J.; Zhu, F.; Lai, Y.; Zhu, W.; You, W.; et al. Preparation and Biocompatibility of Diphasic Magnetic Nanocomposite Scaffold. Materials Science and Engineering: C 2018, 87, 70–77. [Google Scholar] [CrossRef]
- Xu, Y.; Xu, G.; Tang, C.; Wei, B.; Pei, X.; Gui, J.; Min, B.-H.; Jin, C.; Wang, L. Preparation and Characterization of Bone Marrow Mesenchymal Stem Cell-Derived Extracellular Matrix Scaffolds. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2015, 103, 670–678. [Google Scholar] [CrossRef]
- Cheng, Y.-J.; Wu, T.-H.; Tseng, Y.-S.; Chen, W.-F. Development of Hybrid 3D Printing Approach for Fabrication of High-Strength Hydroxyapatite Bioscaffold Using FDM and DLP Techniques. Biofabrication 2024, 16, 025003. [Google Scholar] [CrossRef]
- Tithito, T.; Sillapaprayoon, S.; Pimtong, W.; Thongbunchoo, J.; Charoenphandhu, N.; Krishnamra, N.; Lert-itthiporn, A.; Maneeprakorn, W.; Pon-On, W. Development of Biomaterials Based on Biomimetic Trace Elements Co-Doped Hydroxyapatite: Physical, In Vitro Osteoblast-like Cell Growth and In Vivo Cytotoxicity in Zebrafish Studies. Nanomaterials 2023, 13, 255. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Aziz, A.M.; El-Maghraby, A.; Ewald, A.; Kandil, S.H. In-Vitro Cytotoxicity Study: Cell Viability and Cell Morphology of Carbon Nanofibrous Scaffold/Hydroxyapatite Nanocomposites. Molecules 2021, 26, 1552. [Google Scholar] [CrossRef] [PubMed]
- Zimmerling, A.; Yazdanpanah, Z.; Cooper, D.M.L.; Johnston, J.D.; Chen, X. 3D Printing PCL/nHA Bone Scaffolds: Exploring the Influence of Material Synthesis Techniques. Biomater Res 2021, 25, 3. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Man, Y.; Li, W.; Li, L.; Xu, J.; Parungao, R.; Wang, Y.; Zheng, S.; Nie, Y.; Liu, T.; et al. 3D Bio-Printing of CS/Gel/HA/Gr Hybrid Osteochondral Scaffolds. Polymers 2019, 11, 1601. [Google Scholar] [CrossRef]
- Einhorn, T.A.; Gerstenfeld, L.C. Fracture Healing: Mechanisms and Interventions. Nat Rev Rheumatol 2015, 11, 45–54. [Google Scholar] [CrossRef]
- Dimitriou, R.; Jones, E.; McGonagle, D.; Giannoudis, P.V. Bone Regeneration: Current Concepts and Future Directions. BMC Med 2011, 9, 66. [Google Scholar] [CrossRef]
- Hinsenkamp, M.; Muylle, L.; Eastlund, T.; Fehily, D.; Noël, L.; Strong, D.M. Adverse Reactions and Events Related to Musculoskeletal Allografts: Reviewed by the World Health Organisation Project NOTIFY. International Orthopaedics (SICOT) 2012, 36, 633–641. [Google Scholar] [CrossRef]
- Mushtaq, R.T.; Askari, G.H.; Bao, C.; Wang, Y.; Ahmed, K.; Khan, A.M.; Sharma, S.; Alkahtani, M. Optimization of 3D-Printed Bio-Based Super Tough PLA (ST-PLA) Scaffolds for Cancellous Bone Regeneration: Mechanical Properties, Lattice Architecture, and Osseointegration Potential. International Journal of Biological Macromolecules 2025, 316, 144466. [Google Scholar] [CrossRef]
- Koupaei, N.; Karkhaneh, A. Porous Crosslinked Polycaprolactone Hydroxyapatite Networks for Bone Tissue Engineering. Tissue Eng Regen Med 2016, 13, 251–260. [Google Scholar] [CrossRef]
- Lin, K.; Sheikh, R.; Romanazzo, S.; Roohani, I. 3D Printing of Bioceramic Scaffolds—Barriers to the Clinical Translation: From Promise to Reality, and Future Perspectives. Materials 2019, 12, 2660. [Google Scholar] [CrossRef]
- Mondschein, R.J.; Kanitkar, A.; Williams, C.B.; Verbridge, S.S.; Long, T.E. Polymer Structure-Property Requirements for Stereolithographic 3D Printing of Soft Tissue Engineering Scaffolds. Biomaterials 2017, 140, 170–188. [Google Scholar] [CrossRef] [PubMed]
- Zimina, A.; Senatov, F.; Choudhary, R.; Kolesnikov, E.; Anisimova, N.; Kiselevskiy, M.; Orlova, P.; Strukova, N.; Generalova, M.; Manskikh, V.; et al. Biocompatibility and Physico-Chemical Properties of Highly Porous PLA/HA Scaffolds for Bone Reconstruction. Polymers 2020, 12, 2938. [Google Scholar] [CrossRef] [PubMed]
- Wüster, J.; Neckel, N.; Sterzik, F.; Xiang-Tischhauser, L.; Barnewitz, D.; Genzel, A.; Koerdt, S.; Rendenbach, C.; Müller-Mai, C.; Heiland, M.; et al. Effect of a Synthetic Hydroxyapatite-Based Bone Grafting Material Compared to Established Bone Substitute Materials on Regeneration of Critical-Size Bone Defects in the Ovine Scapula. Regenerative Biomaterials 2024, 11, rbae041. [Google Scholar] [CrossRef] [PubMed]
- Birosz, M.; Dániel, L.; Ando, M. Effect of FDM Infill Patterns on Mechanical Properties. Polymer Testing 2022, 113, 107654. [Google Scholar] [CrossRef]
- Aboelella, M.G.; Ebeid, S.J.; Sayed, M.M. Layer Combination of Similar Infill Patterns on the Tensile and Compression Behavior of 3D Printed PLA. Scientific Reports 2025, 15, 11759. [Google Scholar] [CrossRef]
- Ambati, S.; Ambatipudi, R. Effect of Infill Density and Infill Pattern on the Mechanical Properties of 3D Printed PLA Parts. Materials Today: Proceedings 2022, 64. [Google Scholar] [CrossRef]
- Khaliq, J.; Gurrapu, D.R.; Elfakhri, F. Effects of Infill Line Multiplier and Patterns on Mechanical Properties of Lightweight and Resilient Hollow Section Products Manufactured Using Fused Filament Fabrication. Polymers 2023, 15, 2585. [Google Scholar] [CrossRef]
- Mondal, S.; Pal, U.; Dey, A. Natural Origin Hydroxyapatite Scaffold as Potential Bone Tissue Engineering Substitute. Ceramics International 2016, 42. [Google Scholar] [CrossRef]
- Zhou, H.; Lee, J. Nanoscale Hydroxyapatite Particles for Bone Tissue Engineering. Acta Biomaterialia 2011, 7, 2769–2781. [Google Scholar] [CrossRef]


| Parameter | Specification |
|---|---|
| Nozzle diameter | 0.4 mm |
| Printing speed | 60 mm/s |
| Bed temperature (PLA) | 60°C |
| Nozzle temperature (PLA) | 215°C |
| Layer thickness | 0.3 mm |
| First layer height | 0.2 mm |
| Shell thickness | 0 perimeters |
| Bottom thickness | 0.5 mm (3 layers) |
| Top thickness | 0.7 mm (4 layers) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




