Submitted:
13 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- A)
- Patient recruitment
- B)
- PET-CT
- C)
- Regular follow-up
- D)
- Statistical Analysis
- Age (≤77 vs. >77 year old)
- Gender (male vs. female)
- ECOG performance status (≤1 vs. >1)
- Smoking history (never vs. ever smoker)
- Number of co-morbidities (≤2 vs. >2)
- History of treated non-pulmonary malignancy (no vs. yes)
- Histology (adenocarcinoma / squamous cell carcinoma vs. NSCLC not otherwise specified)
- Primary tumor maximum length (≤50 vs >50mm)
- Primary tumor SUVmax (≤13 vs >13)
- Number of metastatic organs (≤1 vs. >1) and presence of extra-nodal metastases (yes vs. no)
- Presence of pleural and/or pericardial effusion (absent vs. present)
- Serum CEA level (≤10 vs. >10 ng/mL)
- E)
- Approval by IRB
3. Results
3.1. Demographics
3.2. Tumor Characteristics
3.3. Statistical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Filho AM, Laversanne M, Ferlay J, et al.: The GLOBOCAN 2022 cancer estimates: data sources, methods, and a snapshot of the cancer burden worldwide. Int J Cancer. 2025, 156:1336-1346. [CrossRef]
- SEER Cancer Statistics Review 1975-2014. https://seer.cancer.gov/archive/csr/1975_2014/index.html.
- Hong Kong Cancer Registry [Internet]. Hong Kong: Hospital Authority; [cited 2025 Feb 13]. Available from. https://www3.ha.org.hk/cancereg/.
- Hendriks LE, Remon J, Faivre-Finn C, et al.: Non-small-cell lung cancer. Nat Rev Dis Primers. 2024, 10:71. [CrossRef]
- Alduais Y, Zhang H, Fan F, et al.: Non-small cell lung cancer (NSCLC): A review of risk factors, diagnosis, and treatment. Medicine :e32899. [CrossRef]
- Abu Rous F, Singhi EK, Sridhar A, Faisal MS, Desai A: Lung cancer treatment advances in 2022. Cancer Invest. 2023, 41:12-24. [CrossRef]
- Hirsch FR, Spreafico A, Novello S, Wood MD, Simms L, Papotti M: The prognostic and predictive role of histology in advanced non-small cell lung cancer: a literature review. J Thorac Oncol. 2008, 3:1468-1481. [CrossRef]
- Garinet S, Wang P, Mansuet-Lupo A, Fournel L, Wislez M, Blons H: Updated prognostic factors in localized NSCLC. Cancers. 2022, 9:1400. [CrossRef]
- Alamri S, Badah MZ, Zorgi S, et al.: Disease prognosis and therapeutic strategies in patients with advanced non-small cell lung cancer (NSCLC): a 6-year epidemiological study between 2015-2021. Transl Cancer Res. 2024, 13:762-770. [CrossRef]
- SEER Cancer Statistics Review (CSR) 1975-2017. https://seer.cancer.gov/archive/csr/1975_2017/index.html.
- Abbasi S, Badheeb A: Prognostic factors in advanced non-small-cell lung cancer patients: patient characteristics and type of chemotherapy. Lung Cancer Int. 2011:152125. [CrossRef]
- Paesmans M: Prognostic and predictive factors for lung cancer. Breathe :112-121. [CrossRef]
- Zhu J, Branstetter S, Lazarus P, Muscat JE: Smoking, lung cancer stage, and prognostic factors— findings from the National Lung Screening Trial. International journal of environmental research and public health. 2024, 26:400. [CrossRef]
- Fu XL, Zhu XZ, Shi DR, et al.: Study of prognostic predictors for non-small cell lung cancer. Lung cancer. 1999, 1:143-52. [CrossRef]
- Hu Q, Li B, Garfield D, Ren S, Li A, Chen X, Zhou C: Prognostic factors for survival in a Chinese population presenting with advanced non-small cell lung cancer with an emphasis on smoking status: a regional, single-institution, retrospective analysis of 4552 patients. Thoracic cancer. 2012, 3:162-8. [CrossRef]
- Zuo S, Wei M, Zhang H, Chen A, Wu J, Wei J, Dong J: A robust six-gene prognostic signature for prediction of both disease-free and overall survival in non-small cell lung cancer. Journal of translational medicine. 2019, 14:152. [CrossRef]
- Chen P, Liu Y, Wen Y, Zhou C: Non-small cell lung cancer in China. Cancer Commun :937-970. [CrossRef]
- Zhou W, Christiani DC: East meets West: ethnic differences in epidemiology and clinical behaviors of lung cancer between. East Asians and Caucasians. Chin J Cancer. 2011, 30:287-292. [CrossRef]
- Izumi M, Suzumura T, Ogawa K, et al.: Differences in molecular epidemiology of lung cancer among ethnicities (Asian vs. Caucasian). Caucasian). J Thorac Dis. 2020, 12:3776-3784. [CrossRef]
- Kitajima K, Nakamoto Y, Okizuka H, et al.: Accuracy of whole-body FDG-PET/CT for detecting brain metastases from non-central nervous system tumors. Ann Nucl Med. 2008, 22:595-602. [CrossRef]
- Riely GJ, Wood DE, Ettinger DS, et al.: Non-small cell lung cancer, version 4.2024, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2024, 22:249-274. [CrossRef]
- Pinto JA, Vallejos CS, Raez LE, et al.: Gender and outcomes in non-small cell lung cancer: an old prognostic variable comes back for targeted therapy and immunotherapy?. ESMO open. 20181, 3:000344. [CrossRef]
- Radkiewicz C, Dickman PW, Johansson AL, Wagenius G, Edgren G, Lambe M: Sex and survival in non-small cell lung cancer: A nationwide cohort study. PloS one. 2019, 27:0219206. [CrossRef]
- Asamura H, Nishimura KK, Giroux DJ, et al.: IASLC lung cancer staging project: the new database to inform revisions in the ninth edition of the TNM classification of lung cancer. J Thorac Oncol. 2023, 18:564-575. [CrossRef]
| Characteristic | n (%) |
|---|---|
| Age: 54 to 93 (Median 77) | |
| ≤77 | 39 (48.1) |
| >77 | 42 (51.9) |
| Gender | |
| Male | 62 (76.5) |
| Female | 19 (23.5) |
| Performance Status | |
| 0 | 9 (11.1) |
| 1 | 46 (56.8) |
| 2 | 19 (23.5) |
| 3 | 7 (8.6) |
| Smoking History | |
| Never | 29 (35.8) |
| Active Smoker | 20 (24.7) |
| Ex-smoker | 32 (39.5) |
| Number of Co-morbidities: 1 to 10 (Median 2) | |
| History of Cancers | |
| No | 69 (85.2) |
| Yes | 12 (14.8) |
| Overall Survival: 38 to 2072 days (Mean 485 days) | |
| Tumor Characteristics | n (%) |
|---|---|
| Histology | |
| Adenocarinoma | 43 (53.1) |
| Squamous Cell Carcinoma | 22 (27.2) |
| Adenosquamous | 1 (1.2) |
| Poorly Differentiated | 2 (2.5) |
| NSCLC Not Otherwise Specified | 13 (16.0) |
| Maximum Tumor Length: 13 to 119mm (Mean 52.9mm) | |
| Primary Tumor SUVmax: 1.3 to 34.1 (Mean 13.3) | |
| Number of Metastatic Organ: 0 to 6 (Median 1) | |
| Lymph node alone | 25 (30.9) |
| Extra-nodal Metastases | 49 (60.5) |
| Effusion | |
| No | 61 (75.3) |
| Pleural | 17 (21.0) |
| Pericardial | 1 (1.2) |
| Both | 2 (2.5) |
| Serum CEA: 1 to 1464ng/mL (Median 10ng/mL) | |
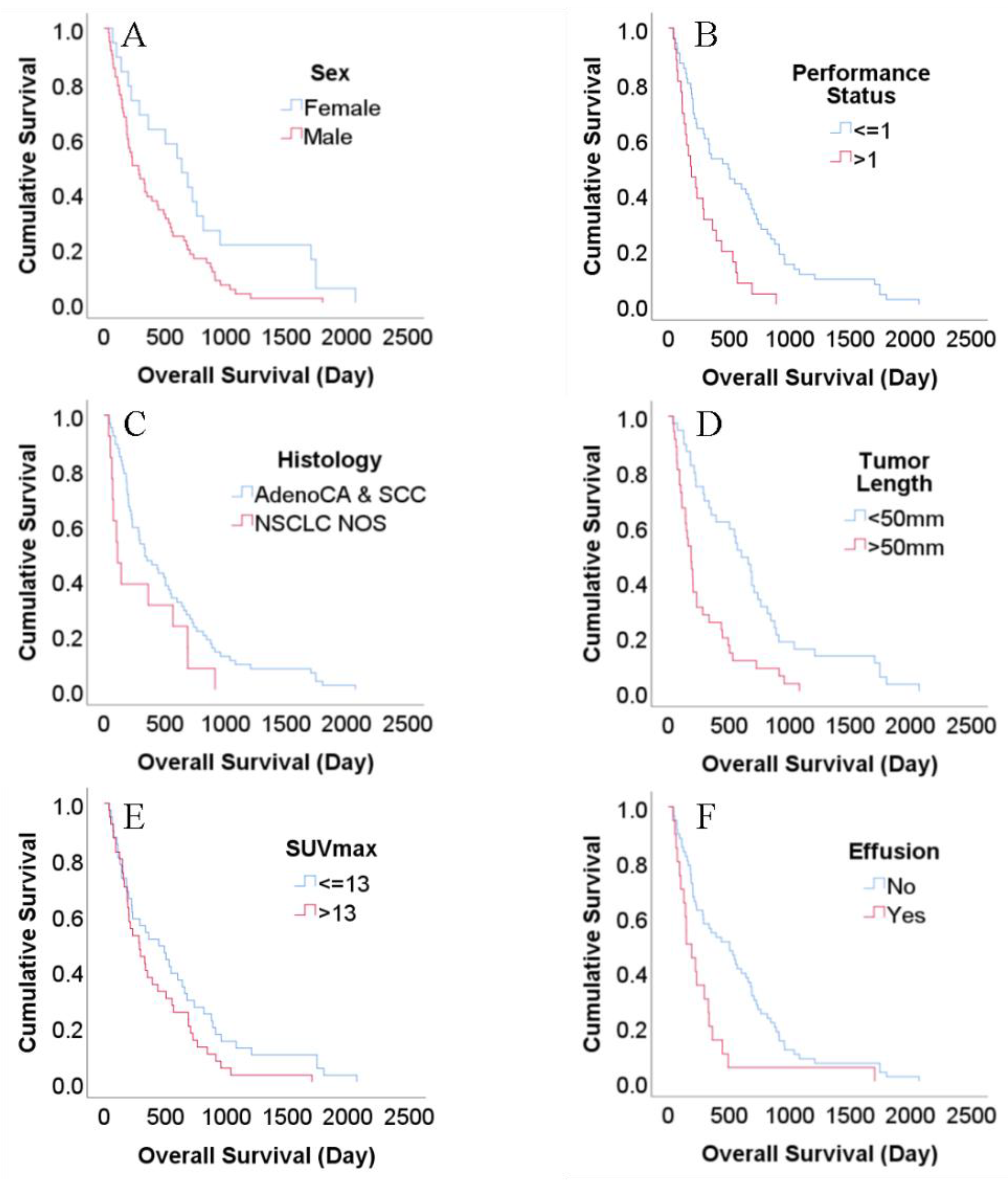
| Subgroup | OS (Day) | p |
|---|---|---|
| Age | ||
| ≤77 | 486 | 0.822 |
| >77 | 484 | |
| Gender | ||
| Male | 402 | 0.015 |
| Female | 756 | |
| Performance Status | ||
| ≤1 | 586 | <0.001 |
| >1 | 271 | |
| Smoking History | ||
| Never | 612 | 0.119 |
| Ever Smoker | 431 | |
| Number of Co-morbidities | ||
| ≤2 | 436 | 0.297 |
| >2 | 552 | |
| History of Cancers | ||
| No | 463 | 0.255 |
| Yes | 609 | |
| Histology | ||
| Adenocarinoma | 542 | |
| Squamous Cell Carcinoma | 474 | |
| NSCLC - not otherwise specified | 299 | p = 0.049* |
| Maximum Tumor Length (mm) | ||
| ≤50mm | 682 | <0.001 |
| >50mm | 279 | |
| Primary Tumor SUVmax | ||
| ≤13 | 567 | 0.09 |
| >13 | 401 | |
| Number of Metastatic Organ | ||
| ≤1 | 544 | 0.22 |
| >1 | 430 | |
| Presence of extra-nodal metastases | ||
| No | 514 | 0.687 |
| Yes | 467 | |
| Effusion | ||
| No | 552 | 0.002 |
| Yes | 281 | |
| Serum CEA (ng/mL) | ||
| ≤10 | 530 | 0.229 |
| >10 | 420 | |
| Subgroup | Hazard Ratio (95% CI) | p |
|---|---|---|
| Gender | ||
| Male | 1.91 (1 – 3.68) | 0.05 |
| Female | Reference | |
| Performance Status | ||
| ≤1 | Reference | 0.001 |
| >1 | 2.57 (1.47 – 4.48) | |
| Histology | ||
| Adenocarinoma & Squamous Cell Carcinoma | Reference | 0.399 |
| NSCLC Without Further Classification | 1.34 (0.68 – 2.65) | |
| Maximum Tumor Length (mm) | ||
| ≤50mm | Reference | 0.003 |
| >50mm | 2.25 (1.31 – 3.86) | |
| Primary Tumor SUVmax | ||
| ≤13 | Reference | 0.539 |
| >13 | 1.17 (0.71 – 1.93) | |
| Effusion | ||
| No | Reference | 0.011 |
| Yes | 2.27 (1.20 – 4.27) | |
| Reasons | n | % |
|---|---|---|
| Treatment Side-effect | 26 | 32.1 |
| Advanced Age | 21 | 25.9 |
| Low Performance Status | 14 | 17.3 |
| Treatment Cost | 7 | 8.6 |
| Asymptomatic | 2 | 2.5 |
| Unknown | 14 | 17.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




