5. Discussion
- 1)
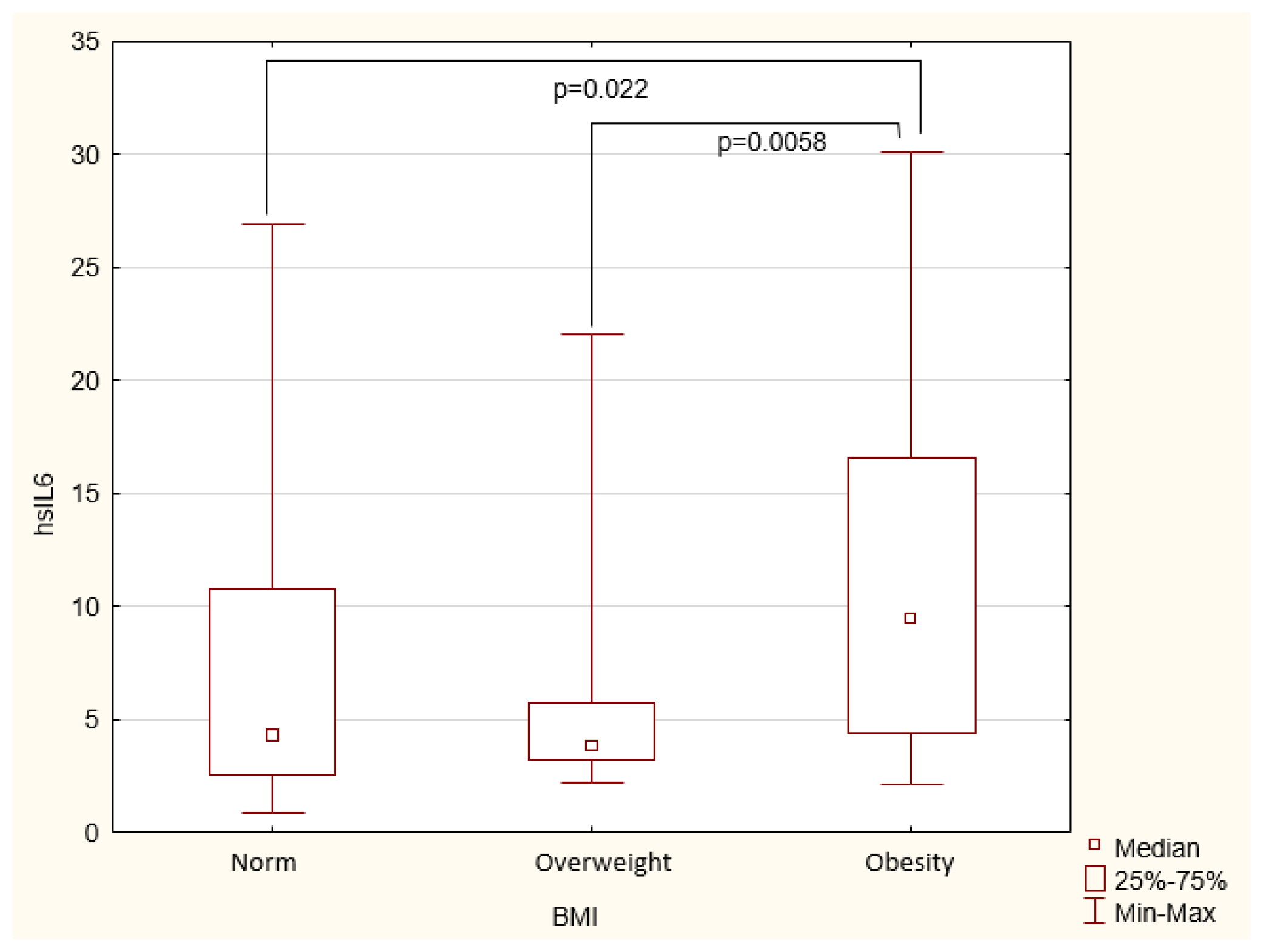
BMI does not correlate with any inflammatory markers or nutritional status markers, such as albumin, transferrin, and cholesterol.
Obesity is becoming an increasingly serious problem worldwide (7). Excess body weight is associated with an increased risk of CKD in the general population (8). In our study group, BMI proved insufficient for a comprehensive characterization of the participants. No significant correlations were found between BMI and inflammatory markers or nutritional status markers. When measured separately, this parameter proved to be clinically irrelevant and did not affect the significance of the results obtained. This view is supported by the findings of Stenvinkel et al. (9), who noted that raw BMI did not reflect the severity of inflammation.
In the general population, the risk of death increases as BMI rises. However, in the HD population, factors commonly considered to indicate cardiovascular risk - such as obesity, high cholesterol, and hypertension - paradoxically appear to have a beneficial effect on the patients (9). One possible explanation for this paradox is the malnutrition-inflammation complex syndrome (MICS), which is common among patients with end-stage renal disease and is the leading cause of cardiovascular mortality in this group, rather than the traditional cardiovascular risk factors. It has also been suggested that this reverse epidemiology may be associated with higher albumin levels, a lower risk of energy deficiency, and higher cholesterol and lipoprotein concentrations in patients with higher body weight (10–12).
On the other hand, this phenomenon may be due to the lack of accuracy in the BMI measurement itself. A study by Lin et al. (13) highlighted the superiority of body composition measurements - and in particular body fat percentage - as a predictor of mortality over BMI, the latter failing to account for muscle atrophy, a common occurrence in CKD patients. It may incorrectly classify individuals with sarcopenic obesity as normal weight patients. Therefore, the MIS score proves to be more useful; as an inflammatory marker in HD patients, it is a very good predictor of mortality in this group (14), while low MIS score patients reported a significantly better quality of life (15). Another useful tool is the bioelectrical impedance method (16).
- 2)
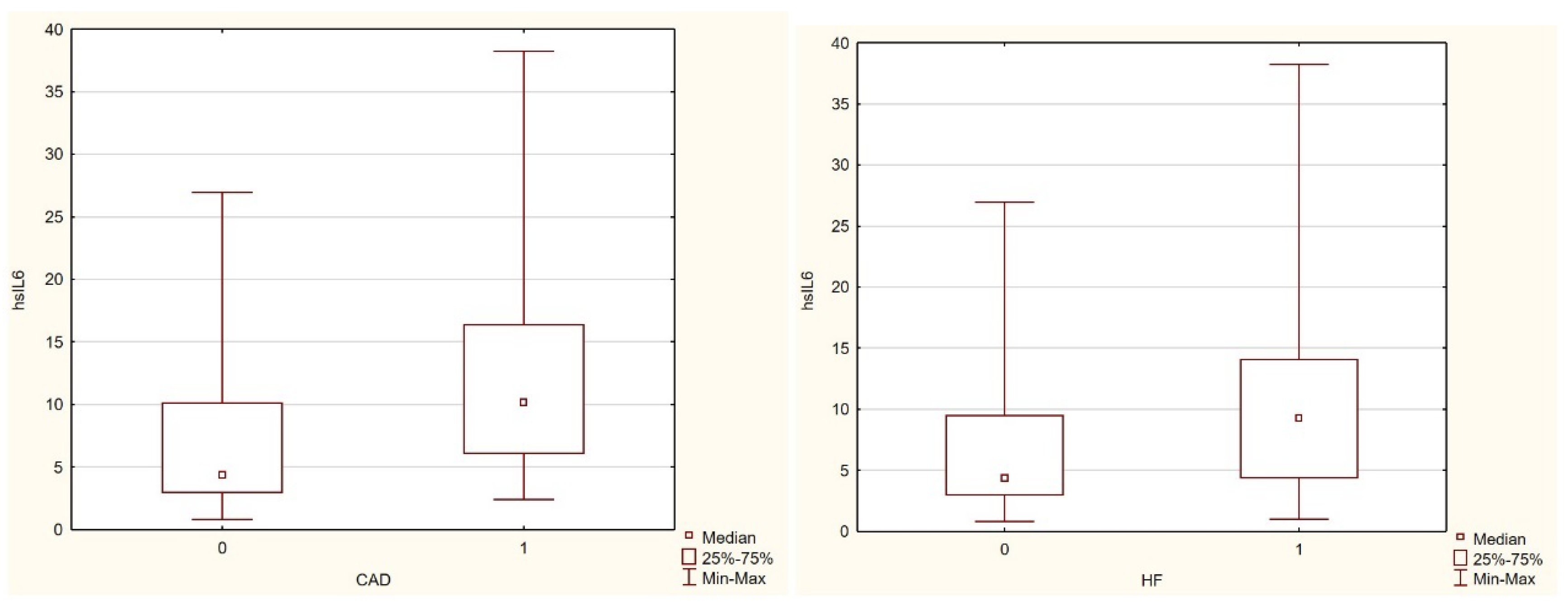
Body composition has a significant impact on two inflammatory markers: hs-CRP and IL-6.
- 3)
CRP and IL-6 levels are higher in patients with more adipose tissue and lower in patients with more muscle mass.
Inflammation is a physiological defense response of the human body (17), but it becomes a pathological condition when it is excessive, uncontrolled, and chronically progressive (18–19). One indicator of inflammation may be interleukin-6, a key cytokine of the immune system that coordinates inflammatory processes. Its excessive synthesis leads to an increased and generalized inflammatory process, which plays a significant role in CKD (20). Another important and easily monitored marker is high-sensitivity C-reactive protein (hs-CRP), elevated levels of which have a negative impact on survival in both the general population (21) and among HD patients (22).
Our research showed that body composition significantly influenced the levels of both of the aforementioned inflammatory markers. The results indicated that hs-CRP and IL-6 levels were higher in patients with greater body fat (FMI [kg,%]) and lower in those with greater muscle mass (SMM [kg,%]). As the percentage of body fat increased, so did the levels of hs-CRP and IL-6 in the study group. In contrast, as muscle mass increased, the levels of these inflammatory markers decrease significantly.
Similar conclusions were drawn in a 2025 single-center study, which found that patients with higher body fat mass had higher levels of hs-CRP (23). A study by Ishimura et al. (24) conducted on a group of 425 HD patients indicated that high-sensitivity CRP was significantly higher in individuals with more adipose tissue, particularly visceral fat. Furthermore, CRP and IL-6 are good indicators of excessive inflammation, and their levels are higher in patients with a higher BMI, as indicated in the report by Delgado et al. (25).
As for IL-6, our results were consistent with those of Kaizu et al. (26) of 2003, which was one of the first studies to demonstrate a direct link between pro-inflammatory cytokines and muscle loss in HD patients. Patients with higher muscle mass percentages had lower IL-6 and CRP levels. In addition, CRP is independently associated with muscle mass in HD patients, making it an independent risk factor for muscle atrophy, which translates into its higher levels in sarcopenia (27). This directly confirms a link between sarcopenia and excessive inflammation.
Through the results of our study, we aim to highlight the critical importance of muscle tissue in this patient population. Using a professional medical body composition analyzer, our patients were divided into four subgroups. Patients with increasing sarcopenic obesity versus those with increasing muscle mass had significantly higher levels of CRP (10.5 vs. 3.7, p < 0.002) and IL-6 (10.14 vs. 5.8, p < 0.029). A similar pattern emerged when comparing patients with increasing obesity to those with increasing muscle mass - with inflammatory markers being higher in the former of the two groups (CRP: 11.4 vs 3.68 p<0.0035; IL-6: 10 vs 5.75 p<0.025). These results indicate that muscle tissue plays a protective, anti-inflammatory role in the HD group.
- 4)
Higher visceral fat levels increase IL-6 concentrations.
With hemodialysis patients, it is important to distinguish between obesity defined as an elevated BMI and visceral obesity, the latter being characterized by an increased amount of visceral fat and occurring even in individuals with a normal BMI. When comparing these two groups, Baberashvili et al. (28) noted that patients with visceral obesity and a normal BMI exhibited a more atherogenic profile in terms of inflammatory markers and adipokine expression versus patients with an elevated BMI (29). Adipose tissue exhibits pro-inflammatory activity, leading to an increase in chronic inflammation through the production of cytokines (30.31).
In our study, we found that IL-6 was positively correlated with the amount of visceral fat (VF[L]). This was confirmed by a study by Zhang et al. (32), where a positive correlation between visceral fat mass and cytokine levels were demonstrated in older adults. This topic was further explored by Dilaver et al. in 2025 (33) with their paper focusing work on intramuscular adipose tissue (IMAT). Their team noted that HD patients had higher IMAT levels compared to individuals without CKD, highlighting a positive correlation between IMAT, body composition, and elevated levels of inflammatory markers such as IL-6. The more IMAT, the lower the handgrip strength, which is a good and reliable indicator of muscle function and, consequently, of progressive sarcopenia (34). Also, elevated IL-6 levels are known to be associated with sarcopenia (35), a finding supported by reports showing that a higher MIS score is positively correlated with elevated IL-6 levels (36). Istanbuly et al. (37) highlighted the association between this cytokine and all-cause mortality in HD patients. Furthermore, higher levels of this interleukin are also associated with an increased incidence of cardiovascular events in this group due to accelerated progression of coronary artery calcification (38).
- 5)
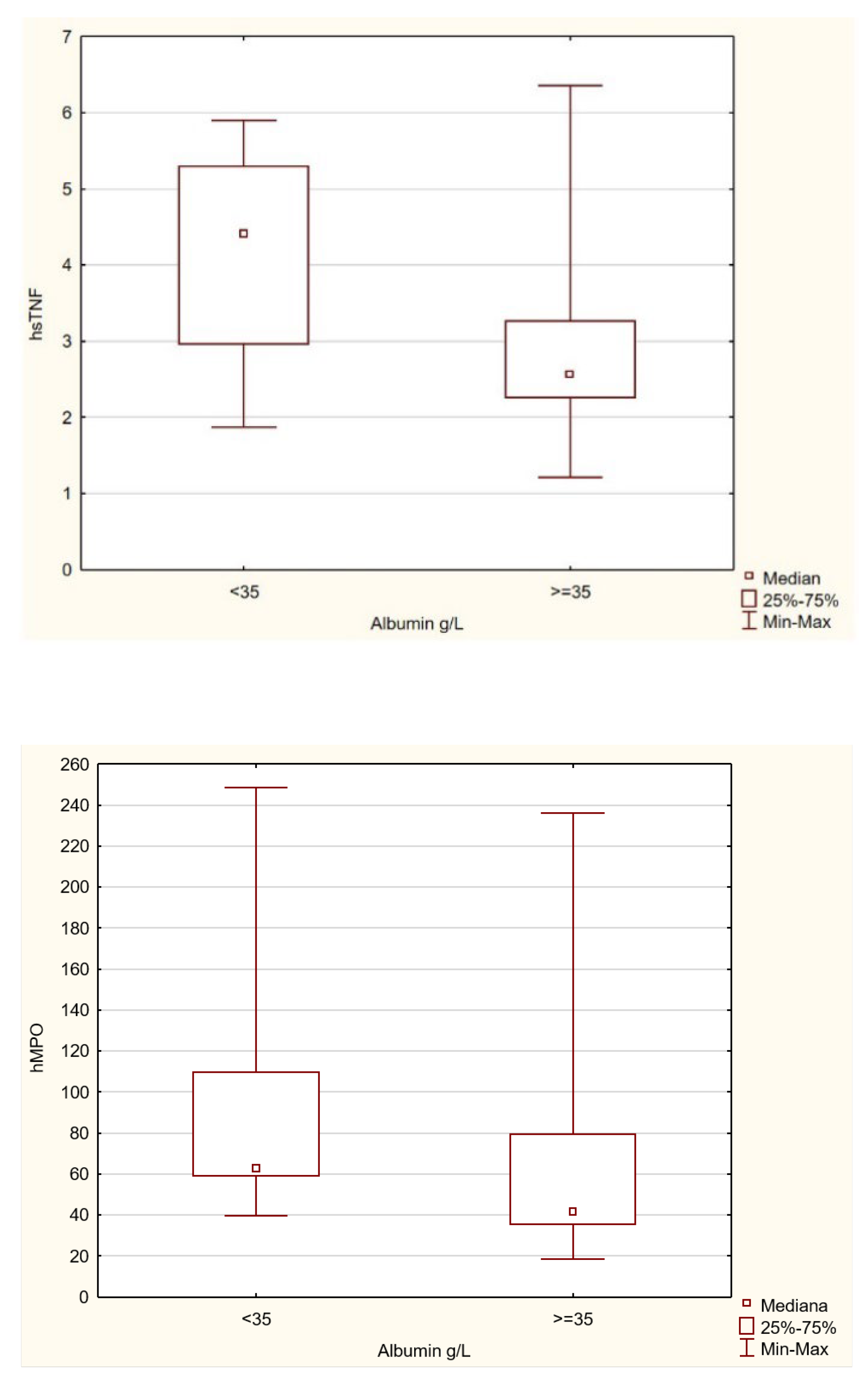
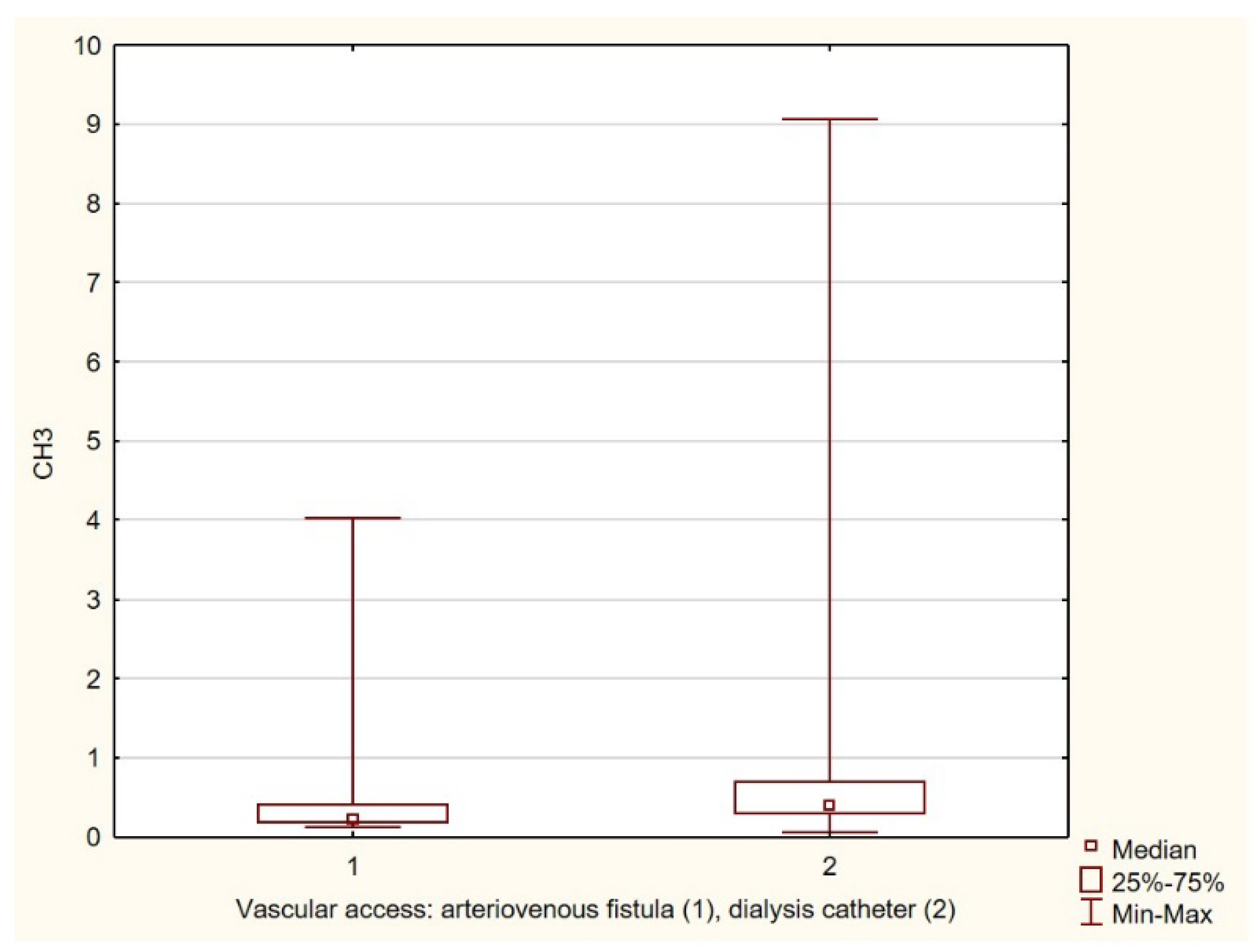
NET activation markers do not have a significant effect on body composition. NET formation grows with the duration of HD treatment and in patients with vascular access via a dialysis catheter. High CH3 levels are associated with low albumin levels.
In our study, we took an unconventional approach to investigating inflammation in hemodialysis patients. In addition to standard inflammatory markers (hs-CRP, IL-1B, IL-6, TNF-α), the following markers of NET activity were assessed, as well: CH3 (citrullinated histones), MPO (myeloperoxidase), and PMN elastase (neutrophil elastase). We were guided by the research on chronic inflammation, neutrophil activation, NETosis, and its complications in hemodialysis patients [
40,
41,
42]. The chronic inflammation that develops in this group has a complex etiology, comprising factors related to both renal replacement therapy and other agents [
42,
43,
44,
45,
46]. Constant stimulation of the immune system is not without significance in activating neutrophils and the NETs they produce.
Our study pointed to a positive correlation between NET formation and renal dialysis vintage. Bieber et al., in their study of the effects of modern dialysis membranes on the immune system, demonstrated that a single hemodialysis session alone led to neutrophil activation and the release of their extracellular traps [
47]. This finding was supported by a 2024 cross-sectional study by Emana R. Edris et al. [
48]. It should be noted that both studies indicated a higher baseline level of NET activity markers in CKD patients compared to the control group; however, they highlighted a significant increase in neutrophil activity following a single HD session.
Our study, too, confirmed the finding reported in the literature regarding the effect of the type of vascular access on inflammation, demonstrating a positive correlation between the presence of a dialysis catheter and neutrophil activity. Stuart L. Goldstein et al. found that the mere presence of an uninfected dialysis catheter was responsible for elevated CRP levels. Furthermore, in the group of patients who successfully developed an AVF during the study, there was a significant decrease in CRP levels (by as much as 82%, p < 0.0001) and an increase in albumin and hemoglobin levels [
49]. Another study by Mihaly B. Tapolyai et al. showed that patients with a dialysis catheter not only had higher CRP levels but also a higher neutrophil-to-lymphocyte ratio (NLR) compared to those with an AVF, which demonstrated that the dialysis catheter kept neutrophils in a state of constant mobilization [
50].
Chronic inflammation is accompanied by persistently high levels of pro-inflammatory cytokines, including IL-6 and TNF-α, which are believed to have anorexigenic effects [
51,
52]. A study by Kamyar Kalantar-Zadeh’s team demonstrated that a lack of appetite in HD patients was one of the simplest and least expensive indicators suggesting severe inflammation [
4]. Anorexia, meanwhile, was positively correlated with hypoalbuminemia and higher MIS scores, indicating the patient’s worsening cachexia (MICS) [
4]. It was therefore not surprising that our study found a significant negative correlation between CH3, a marker of NETosis, and albumin levels. In a study by Yue Zheng et al., albumin demonstrated antioxidant properties against mtROS, thereby inhibiting the NET activation cascade [
53]. Albumin at physiological concentrations is believed to act as an inhibitor of extracellular trap release [
54,
55]. However, one cannot overlook the finding reported in the study by Daria V. Grigorieva et al. regarding the effect of MPO on albumin [
55]. This enzyme, released during NETosis, causes the formation of HOCl, a powerful oxidizing agent that also oxidizes albumin. The resultant HOCl-modified HSA become neutrophil activators. When these factors are considered together, a vicious cycle becomes apparent: HD patients suffer from chronic inflammation and malnutrition. The associated hypoalbuminemia correlates with increased NET activity, which, in turn, can modify albumin so that it activates additional neutrophils.
Our study rendered an interesting result regarding the NETosis process and body composition. Patients with increasing obesity, compared to the group with increasing muscle mass, were characterized by significantly higher levels of PMN - an indicator of NETosis (49.74 vs. 35.43, p 0.002). This indicated that muscle tissue played a protective, anti-inflammatory role in the HD group, as described earlier in this paper.
- 6)
The main predictors of death during a 12-month follow-up in HD patients are albumin and TNF-α levels.
In our study, we demonstrated that lower albumin levels were associated with an increased risk of death during the 12-month follow-up period. An important study in this area is the research conducted by Kamyar Kalantar-Zadeh’s team, which demonstrated how the risk of death increased dramatically as albumin levels declined in HD patients [
56]. This study emphasized that it was the dynamic analysis of albumin levels - specifically, the direction of change in albumin concentration (i.e., an increase or decrease) - that helped predict survival. This indicated that, in this group of patients, it was crucial to know not only the current albumin level but also the levels from previous months. Even more importantly, this retrospective cohort study of over 58,000 patients receiving chronic hemodialysis showed that as many as 19% of deaths in this population could potentially be prevented by maintaining albumin levels above 3.8 g/dL. The association between low albumin levels and mortality was confirmed by a study by George A. Kaysen et al., which also explained the physiological mechanisms responsible for the decrease in albumin levels among patients undergoing renal replacement therapy [
57]. In addition to the impact of diet, the researchers emphasized the impact that inflammation had on hypoalbuminemia. It was shown that inflammation was responsible for the increased catabolism of these proteins.
According to our findings, TNF-α levels also proved to predict death in a 12-month follow-up period. Le Viet Thang’s team reached a similar conclusion after observing 319 patients undergoing low-flow dialysis over a 3-year period [
58]. In the study population, which was divided into three subgroups based on TNF-α levels, the group with the highest concentration of this pro-inflammatory cytokine was associated with significantly higher overall mortality compared to the other two groups. One article worth mentioning in the context of the relationship between TNF-α and mortality is the study by Beata Marie R. Quinto et al. [
59], which examined the effects of removing inflammatory factors from the blood in critically ill AKI patients. The main finding of this study was the established association between the filter’s high capacity to remove TNF-α and a better survival rate. Furthermore, a low capacity of the filter to remove TNF-α was found to be an independent risk factor for death, even after adjusting for the patient’s condition. Although this study focused on a population other than patients on chronic dialysis, it highlighted the significant role of tumor necrosis factor-alpha in the progression of a patient’s condition.
- 7)
Factors affecting albumin levels included those that increased it (percentage of muscle mass, a high phase angle) and those that decreased it (CH3, IL-6, TNF-α).
As mentioned above, in our study, albumin levels were one of the main predictors of death. Therefore, it appears important to understand the relationships between albumin and other parameters. Our observations indicated that both a higher percentage of muscle mass and a high phase angle increased albumin levels.
The phase angle is determined based on the resistance and reactance values obtained from the bioelectrical impedance analysis. This parameter, which is influenced by the integrity and functionality of cell membranes, has become an indicator of cellular health, a surrogate marker of muscle quality [
60,
61], a predictor of sarcopenia [
62], and a predictor of PEW in patients on chronic dialysis [
63]. In this group of patients, an acute (low) phase angle may indicate malnutrition, overhydration, or a combination of both. A sufficiently high phase angle is associated with good nutritional status, greater muscle mass, lower overhydration, and better prognosis. This was reflected in our results, which indicated a correlation between a high phase angle and higher albumin concentrations, and thus better nutritional status, reduced inflammation, and a more favorable prognosis.
In the human body, there are two main categories of protein: somatic protein (skeletal muscle) and visceral protein. In their paper, D. Fouque et al. highlighted the term PEW (protein-energy wasting), using it to replace the concept of malnutrition itself [
64]. In patients with ESRD, this is a complex issue. In addition to a loss of appetite, the depletion of protein and energy reserves is also a result of chronic inflammation. Pro-inflammatory cytokines reduce the production in the liver of so-called "negative" acute-phase proteins, including albumin and prealbumin, leading to a decrease in visceral protein levels. It is only as a result of prolonged catabolism that the ubiquitin-proteasome pathways are activated, leading to the catabolism of skeletal muscle. Thus, it can be concluded that patients with a higher percentage of muscle mass - and therefore higher albumin levels - exhibit less severe inflammation.
Therefore, further results of our study indicating that CH3, IL-6, and TNF-α contributed to a decrease in albumin levels did not come as a surprise. These findings were consistent with the latest ASPEN guidelines (David C. Evans et al.), which emphasize that visceral proteins are primarily negative acute-phase proteins and should not be used in isolation in diagnosing malnutrition, as their serum concentrations correlate more strongly with the intensity of the inflammatory response than with nutritional status itself [
65]. This phenomenon can be explained by the liver’s prioritization of inflammatory protein synthesis and plasma protein redistribution resulting from increased vascular permeability during inflammation.
- 8)
Factors affecting TNF-α levels: inflammatory markers such as elastase, IL-1β, hs-CRP, and IL-6 increase these levels.
TNF-α is an inflammatory cytokine that plays a key role in inflammation, apoptosis, and anticancer defense. In our study, we demonstrated that inflammatory markers such as neutrophil elastase, IL-1β, IL-6, and hs-CRP increased levels of tumor necrosis factor-alpha.
It is worth noting that cytokines are components of a complex signaling network in the body, in which target cells are exposed to various combinations of cytokines that can exert additive, inhibitory, or synergistic effects, as appropriate [
66]. The aforementioned markers of inflammation can be considered from a similar standpoint. Contact between blood and the dialysis membrane triggers neutrophil activation and the release of NETs, one of the components of which is neutrophil elastase [
38]. A study by Mężyk-Kopeć et al. suggested that proteolytic enzymes - neutrophil elastase and cathepsin G - might release the biologically active form of s-TNF-α circulating in serum through their ability to ‘cleave’ m-TNF-α, initially anchored in the cell membrane [
67]. This cascade of inflammation could then lead to an increased expression of IL-1 and IL-6 by TNF-α. A study by Turner et al. identified intracellular signal transduction pathways through which TNF-α induced the expression of IL-1 and IL-6 in human cardiac fibroblasts, contributing to cardiac remodeling [
68]. In the context of patients on chronic dialysis, this study illustrates how inflammatory cytokines are responsible for the clinical implications observed in this patient group, such as progressive circulatory failure.
Furthermore, patients undergoing renal replacement therapy showed a significant increase in endogenous inhibitors of pro-inflammatory mediators; paradoxically, these inhibitors were unable to fully neutralize the biological activity of IL-1 and TNF-α, which evidenced immune dysregulation in these patients [
69]. Tumor necrosis factor-alpha causes an increase in CRP levels indirectly, via IL-6 [
70]. In turn, the CRP protein leads to an increase in the levels of TNF-α, IL-1beta, and IL-6 [
71]. The above illustrates the self-amplifying vicious cycle of inflammation in HD patients: neutrophil elastase, emerging through NETosis, leads to increased levels of TNF-α, which, via IL-6, causes the liver to prioritize the synthesis of inflammatory proteins, which in turn escalate inflammation through a positive feedback loop. Consequently, this leads to the aforementioned MICS.
- 9)
No association was found between body composition indices and survival during the 12-month follow-up period.
One method of analyzing body composition in adults involves the use of so-called body composition indices, which are calculated using fat mass (FM), fat-free mass (FFM), and total body water (TBW) [
72]. They enable metabolic and endocrine characterization, contributing to patient profiling and the identification of risks associated with abnormal values of these indices [
73]. The available literature contains ample evidence supporting their high usefulness. One example is a study conducted by Kang et al. [
74], which showed that fat-free mass (FFM) was useful in determining bone mineral density (BMD). This was described from another perspective by Baldessari et al. [
75], who demonstrated that a comprehensive assessment of the patient, including their nutritional status, could have a positive impact on further treatment. For this reason, there are serious grounds for a widespread use of indices such as FM, FMI, FFM, and TBW, as they help to account for the highly variable body composition despite a similar interpretation of the body mass index (BMI) in different patients [
76].
This topic is widely discussed nowadays, particularly with regard to patient mortality. Overweight and obesity are well-known to be significant and key risk factors for cardiovascular disease (CVD) [
77], which is associated with a higher risk of death from the disease [
78]. In contrast, however, the term “obesity paradox” has emerged to describe an aspect that manifests itself repeatedly in certain medical conditions as based on the protective association between high BMI values and mortality [
79]. Nevertheless, there is a growing recognition of the limitations and shortcomings of this method as a measure of obesity (BMI), with suggestions that the assessment of body fat and fat-free mass - the latter of which has often been overlooked - is crucial for the overall results of the tests [
80]. One example is a study conducted by Graf et al. [
81], which led to the conclusion that a decrease in the fat-free mass index (FFMI) was associated with increased mortality among older adults.
Our study, in turn, allowed us to assess the relationship between body composition indices and their impact on HD patient survival. Based on the results obtained, it can be concluded that the indicators in question do not have a direct impact on the survival of patients undergoing HD during the 12-month follow-up period. The main predictors of death in the study patients are albumin and TNF-α levels (cf. Point 6 of the conclusions). It is worth noting that the narrow time frame available for our study significantly limited our ability to conduct a broader analysis of the impact of body composition indices on patient survival. The above must lead to the obvious conclusion that continuing the research over the long term could yield interesting results - which may differ significantly from the conclusion reached so far.
- 10)
No association was found between NET markers and survival during the 12-month follow-up period.
NETosis is described as the process of forming a specialized network composed of DNA and antimicrobial proteins released by activated neutrophils, which helps neutralize pathogens but also has pro-inflammatory and pro-thrombotic potential [
82]. Hemodialysis, which is used to treat chronic kidney disease (CKD), is one of the factors contributing to exacerbation of inflammation in the patients. This occurs through the activation of the innate immune system, particularly the complement system, as well as through the recruitment of neutrophils [
83]. It is because of them that the NETosis markers described earlier- such as CH3-citrullinated histones, MPO (myeloperoxidase), and PMN elastase - are elevated in patients undergoing chronic hemodialysis [
41].
NET indices, as described in the available literature - particularly during dialysis sessions - have been identified as predictors of poorer treatment outcomes [
84]. In addition, a link is increasingly being reported between an exacerbated NETosis and the pathogenesis of many diseases, including sepsis, systemic lupus erythematosus, rheumatoid arthritis, and small-vessel vasculitis [
85]. A study by Lin et al. [
86] demonstrated that NETosis was significantly correlated with disseminated intravascular coagulation (DIC) caused by septic shock. This is an important observation regarding patients on chronic hemodialysis who, as a result of treatment for their underlying condition (CKD), are at risk of developing chronic inflammation manifested by elevated levels of C-reactive protein (CRP) or endothelial dysfunction [
87]. These components contribute to the multifaceted activation of NETs [
88].
The regulation of NET processes remains the focus of research exploring inhibitory and stimulatory mechanisms [
89]. The available literature emphasizes the importance and relevance of research on NETosis and its markers in order to better understand the process that is a key host defense mechanism [
90]. Hence, an important conclusion drawn from our study is that we found no association between NET markers and survival in HD patients; however, we did find a significant correlation between NET markers and muscle mass and body composition (Cf. Points 4 and 5). It is important to note the relatively short 12-month follow-up period for patients undergoing HD in this study. Perhaps a long-term observation would be more beneficial.