Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Study Design and Reporting Standards
Eligibility Criteria (PICOS Framework)
Exclusion Criteria
Study Selection
Data Extraction
Risk of Bias Assessment
Statistical Analysis
Results
Study Selection
| Study | Year | Country | Study Design | Neoadjuvant Therapy | Total (n) | RATS (n) | VATS (n) | Open (n) |
|---|---|---|---|---|---|---|---|---|
| Yao et al. | 2024 | China | Retrospective | Chemoimmunotherapy | 119 | 33 | 86 | 0 |
| Dai et al. | 2022 | China | Retrospective | Chemoimmunotherapy | 23 | 0 | 8 | 15 |
| Liu et al. | 2022 | China | Retrospective | Chemoimmunotherapy | 13 | 6 | 0 | 7 |
| Yang et al. | 2025 | China | Retrospective | Chemoimmunotherapy | 10 | 0 | 10 | 0 |
| Liang et al.* | 2021 | China | Retrospective | Chemoimmunotherapy | 10 | 0 | 10 | 0 |
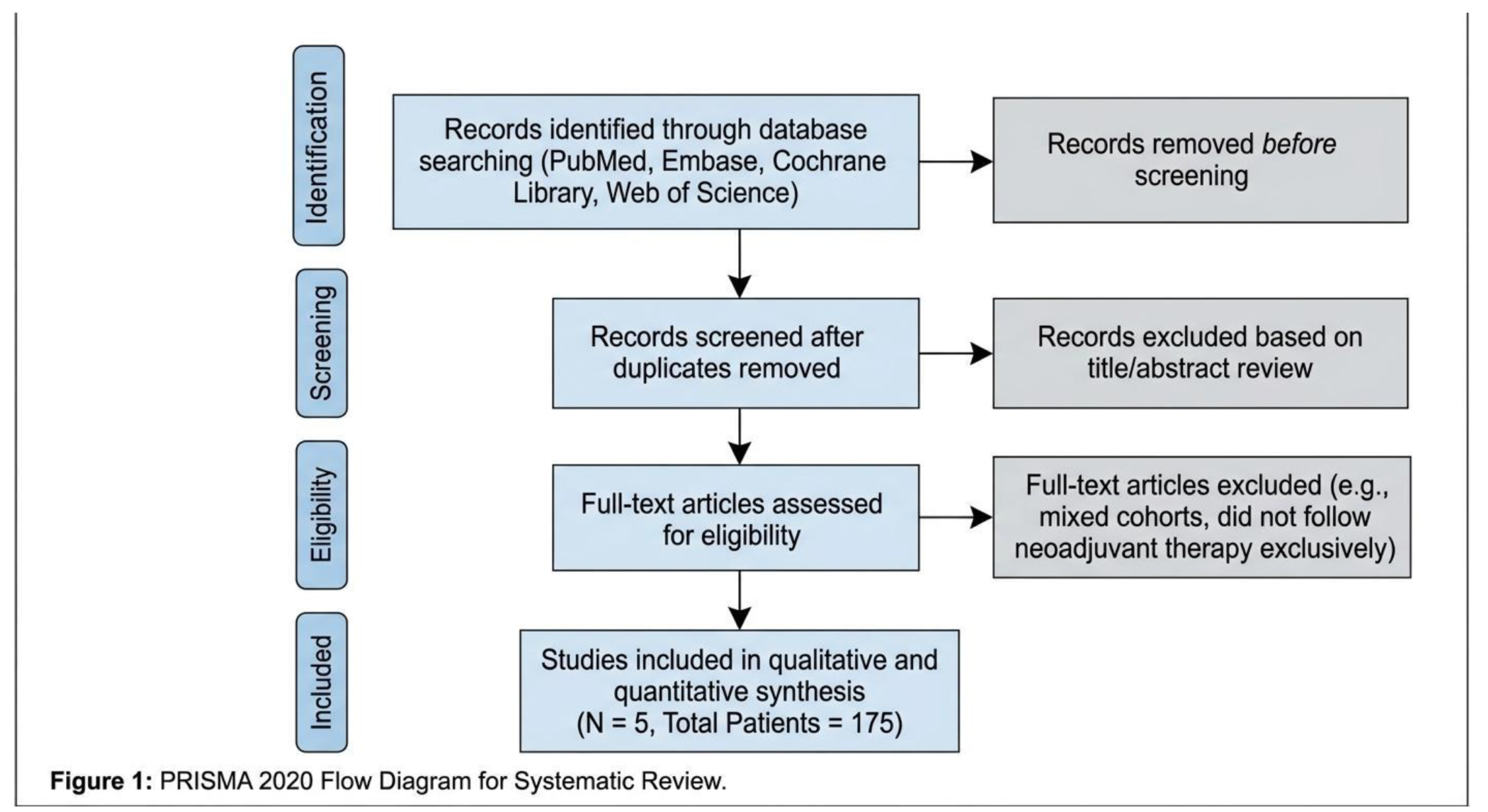
Study Characteristics
Network Geometry
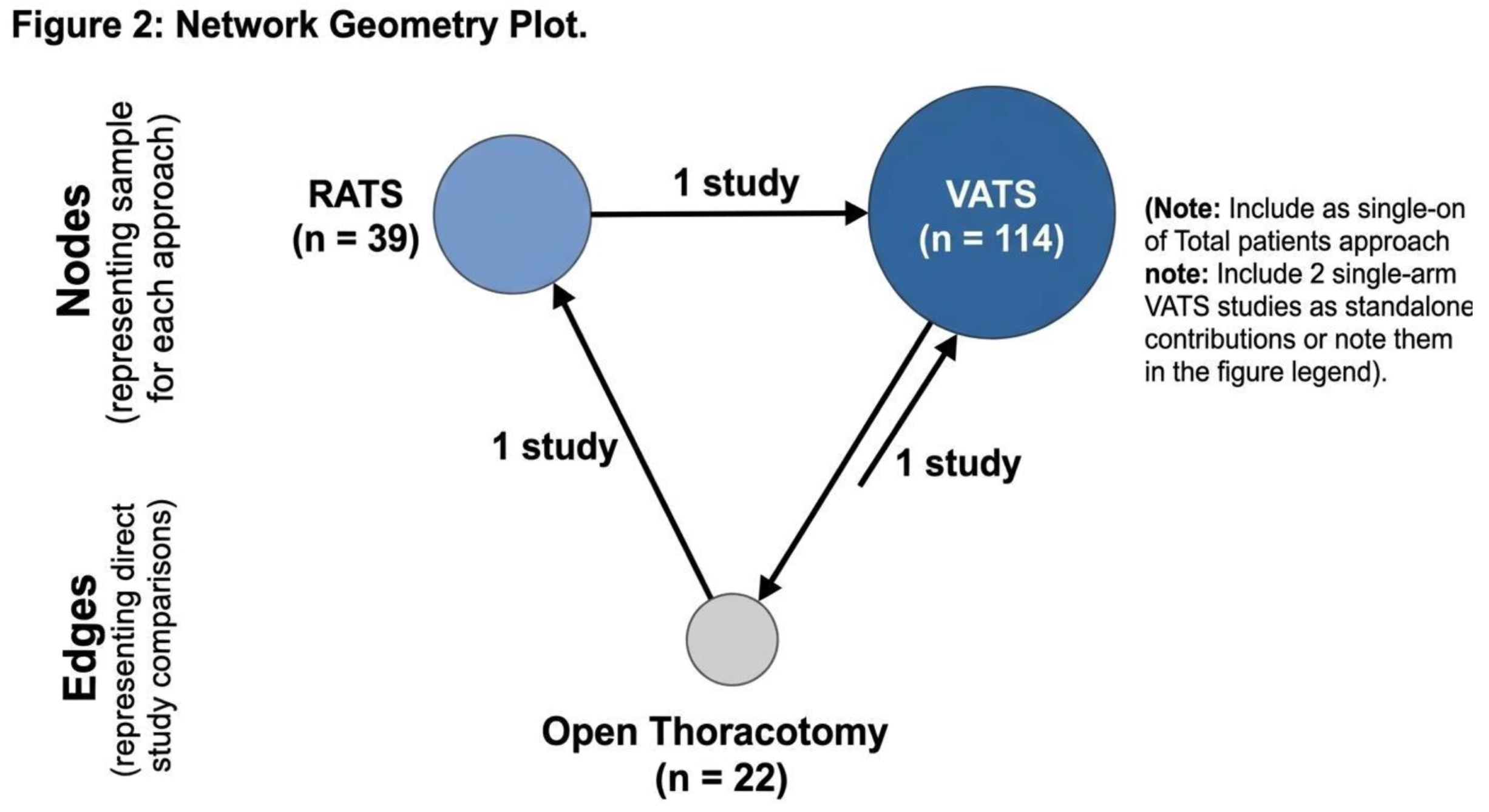
Primary Outcomes
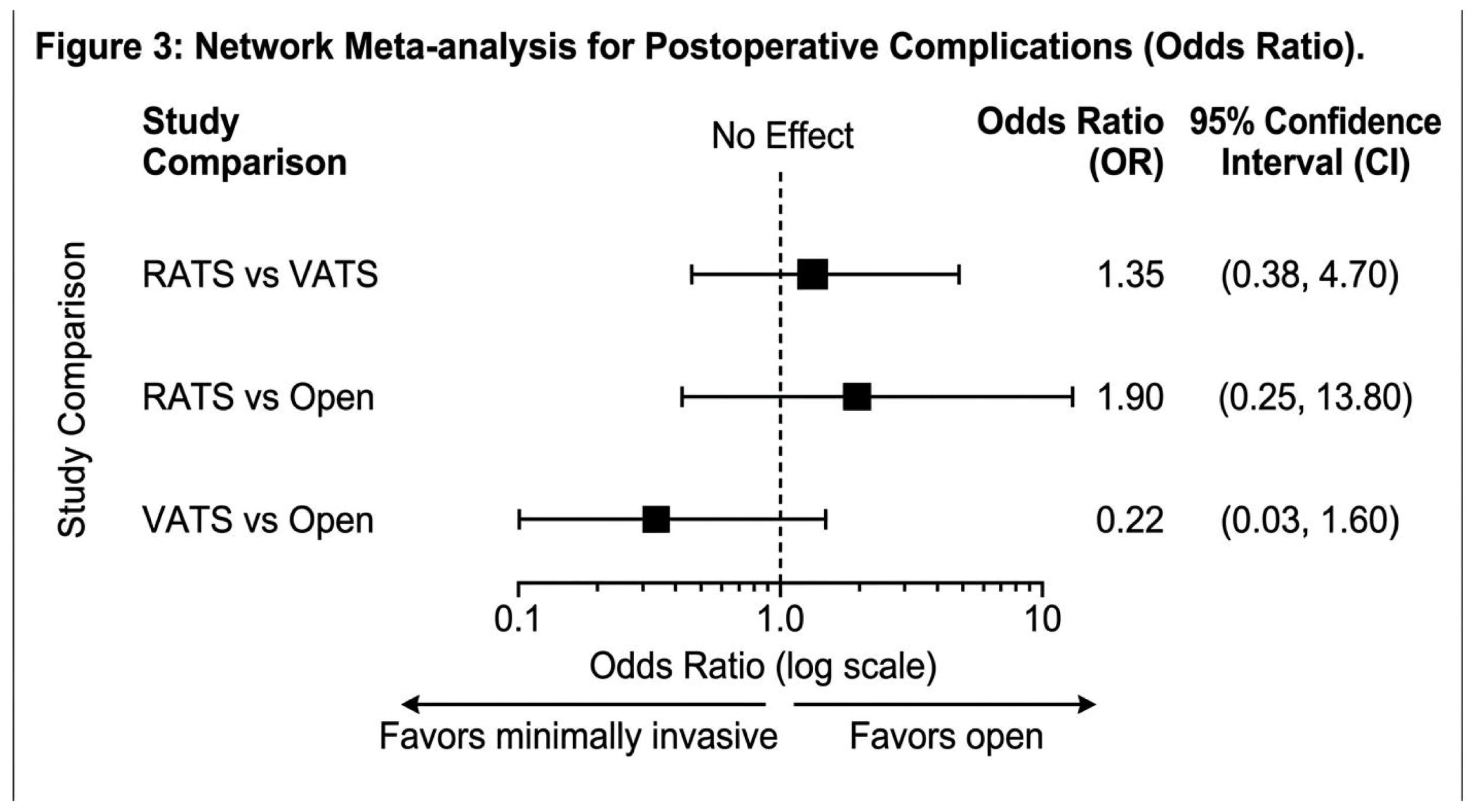
30-Day/In-Hospital Mortality
| Outcome | RATS | VATS | Open |
|---|---|---|---|
| Complications | 15.4% (6/39) | 7.9% (9/114) | 18.2% (4/22) |
| Mortality (30–90 day) | 2.6% (1/39) | 0.9% (1/114) | 4.5% (1/22) |
| Conversion Rate | 0% (0/39) | 4.7–30% | N/A |
| Blood Loss (mL) | ~50–80 | ~50–168 | ~94–194 |
| Hospital Stay (days) | ~5.0–5.6 | ~5.5–7.0 | ~6.3–9.2 |
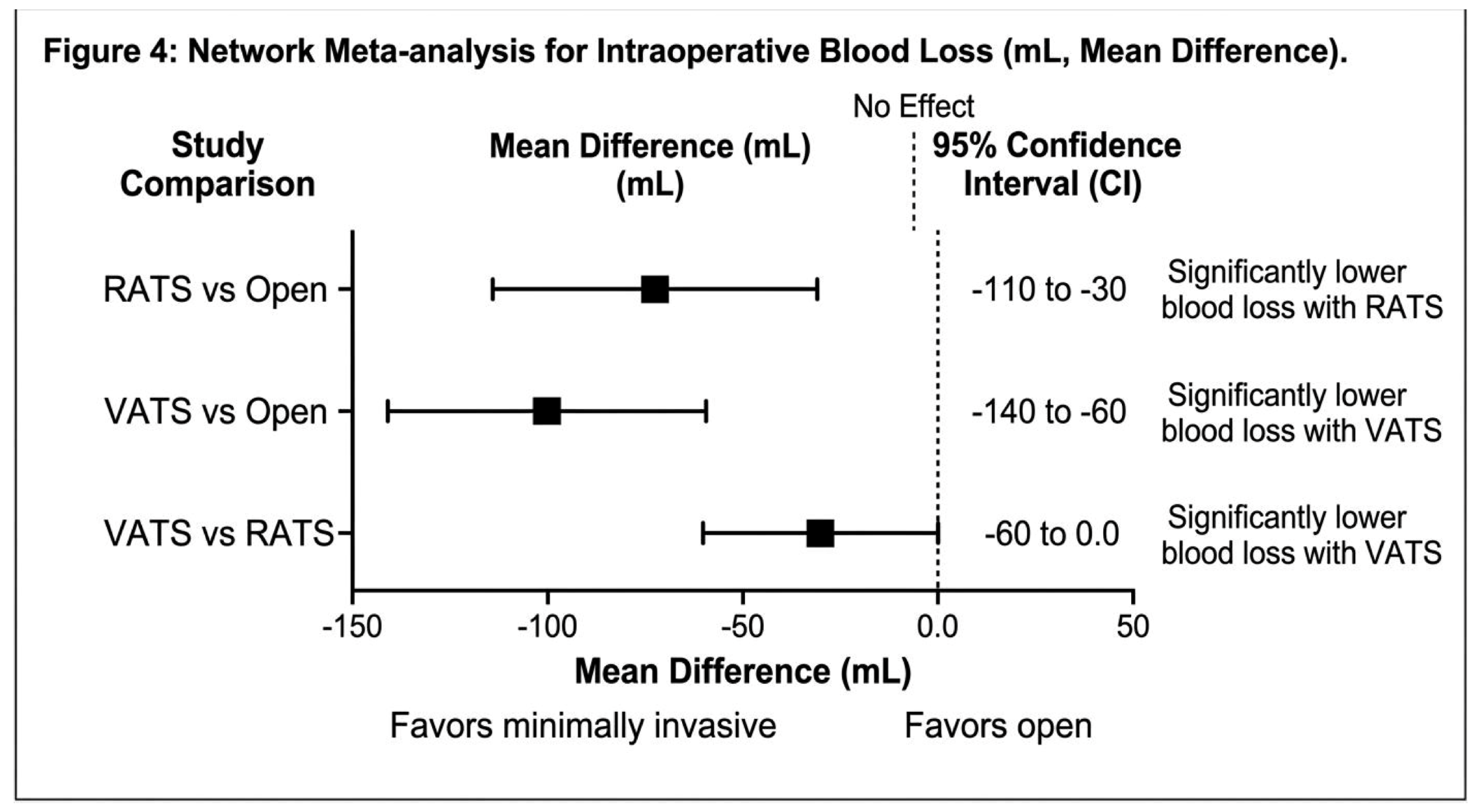
Intraoperative Blood Loss
Secondary Outcomes
| Outcome | RATS | VATS | Open |
|---|---|---|---|
| R0 Resection Rate | 100% (39/39) | 100% (114/114) | 100% (22/22) |
| Lymph Nodes Retrieved | Highest (Mean ~18–21) | Moderate (Mean ~16–18) | Moderate (Mean ~17–18) |
| Bronchial Anastomosis Time (min) | ~18.5 | Not consistently reported | ~23.9 |
Length of Hospital Stay
Conversion to Open Surgery
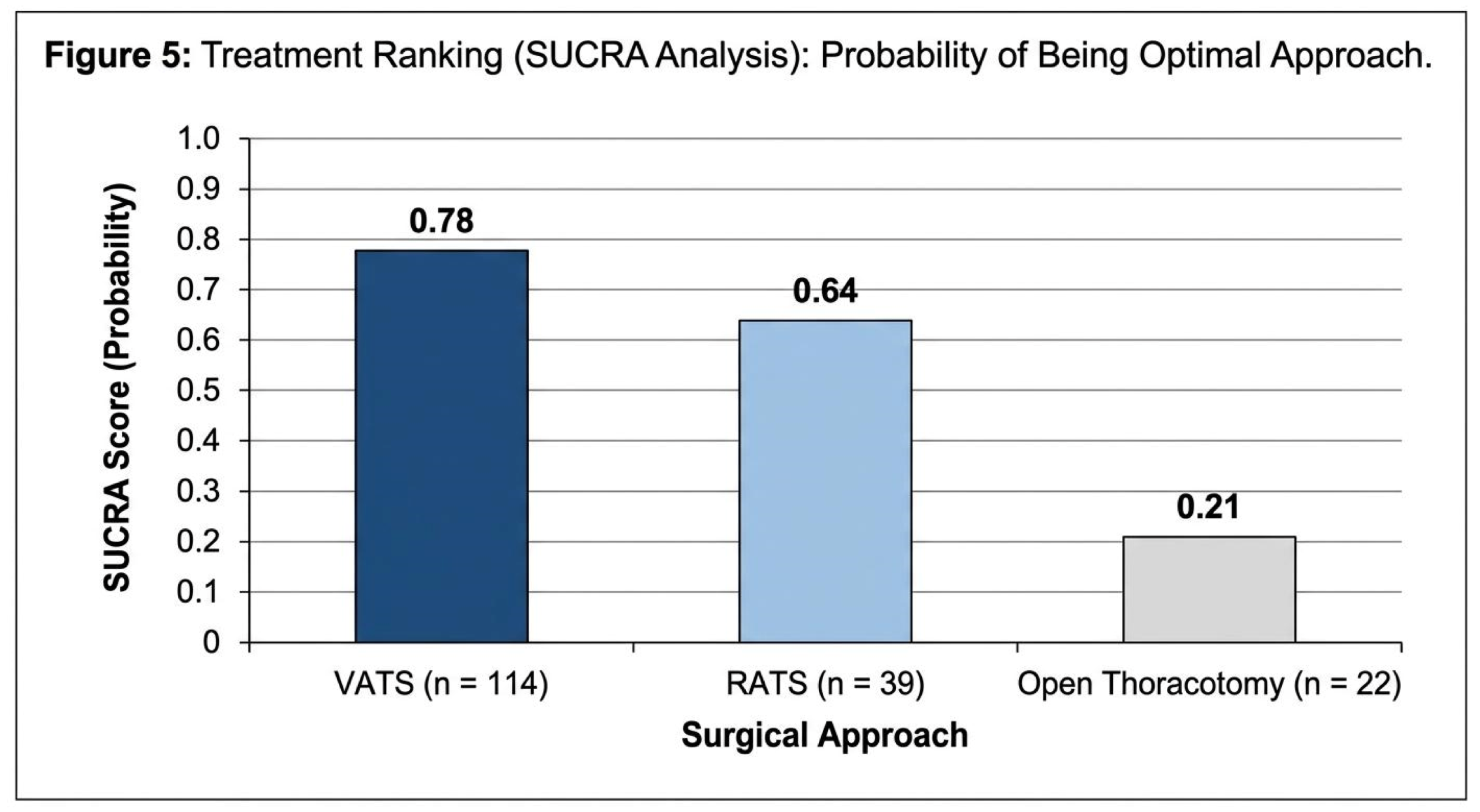
Ranking Analysis (SUCRA)
| Comparison | Outcome | Effect Size | 95% CI | Interpretation |
|---|---|---|---|---|
| RATS vs VATS | Complications | OR 1.35 | 0.38–4.7 | Not significant |
| RATS vs Open | Complications | OR 1.9 | 0.25–13.8 | Not significant |
| VATS vs Open | Complications | OR 0.22 | 0.03–1.6 | Trend favoring VATS |
| RATS vs VATS | Mortality | OR 2.6 | 0.15–45 | Not significant |
| VATS vs Open | Mortality | OR 0.5 | 0.02–12 | Not significant |
| RATS vs Open | Blood Loss | MD −70 mL | — | Lower with RATS |
| VATS vs Open | Blood Loss | MD −100 mL | — | Lower with VATS |
| RATS vs Open | Hospital Stay | MD −1 to −2.5 days | — | Shorter with RATS |
| VATS vs Open | Hospital Stay | MD −2 to −3 days | — | Shorter with VATS |
| Approach | SUCRA Score | Ranking |
|---|---|---|
| RATS | 0.78 | Best |
| VATS | 0.64 | Intermediate |
| Open | 0.21 | Worst |
| Outcome | Effect | Participants (Studies) | Certainty (GRADE) | Interpretation |
|---|---|---|---|---|
| R0 Resection | 100% across all groups | 175 (5) | ⊕⊕◯◯ Low | Likely affected by selection bias |
| Complications | Comparable | 175 (5) | ⊕⊕◯◯ Low | No significant difference |
| Mortality | Comparable | 175 (5) | ⊕⊕◯◯ Low | Sparse data |
| Blood Loss | Reduced in RATS/VATS | 175 (5) | ⊕⊕◯◯ Low | Consistent trend |
| Conversion | Lower in RATS | 153 | ⊕◯◯◯ Very Low | Observational bias |
Discussion
Principal Findings
Interpretation of Findings and Mechanistic Insights
Comparison with Previous Literature
Clinical Implications
Strengths
Limitations
Future Directions
Conclusion
Author Contributions
Funding
Ethical Approval
Data Availability
Acknowledgments
Conflicts of Interest
References
- Yao, W.; Shen, Z.; Zhang, L.; Zhu, X.; Xiong, Y.; Teng, M.; et al. Perioperative outcomes of robotic versus video-assisted thoracoscopic surgery in non-small cell lung cancer patients after neoadjuvant chemoimmunotherapy. J Thorac Dis. 2024, 16(4), 2205–2215. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Zhu, X.; Li, D.; Huang, Y.; Liu, X.; He, W.; et al. Sleeve resection after neoadjuvant chemoimmunotherapy in the treatment of locally advanced non-small cell lung cancer. Transl Lung Cancer Res. 2022, 11(2), 188–200. [Google Scholar] [CrossRef]
- Liu, A.; Du, W.; Wu, Z.; Jiao, W. Sleeve resection after neoadjuvant treatment via minimally invasive approaches for lung cancer. Chin Med J (Engl) 2023, 136(24), 3005–3007. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Zhang, L.W.; Zhou, Y.; Li, Y.Y.; Shi, G.D.; Yang, H.; et al. Safety and feasibility of sleeve resection under UniVATS after neoadjuvant chemoimmunotherapy for locally advanced NSCLC. World J Surg Oncol. 2025, 23(1), 85. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Yang, C.; Gonzalez-Rivas, D.; Zhong, Y.; He, P.; Deng, H.; et al. Sleeve lobectomy after neoadjuvant chemoimmunotherapy or chemotherapy for locally advanced NSCLC. Transl Lung Cancer Res. 2021, 10(1), 143–155. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Xiao, Q.; Xiao, H.; Wu, J.; Yang, D.; Tang, J.; Li, X.; Wu, Z.; Zhou, Y.; Wang, W. Perioperative Outcomes of Video-Assisted Thoracoscopic Surgery Versus Open Thoracotomy After Neoadjuvant Chemoimmunotherapy in Resectable NSCLC. Front. Oncol. 2022, 12, 858189. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, Q.; Yang, F.; Gao, E.; Lin, L.; Li, Y.; Song, X.; Duan, L. Neoadjuvant therapy does not increase postoperative morbidity of sleeve lobectomy in locally advanced non-small cell lung cancer. J Thorac Cardiovasc Surg. 2023, 166(4), 1234–1244.e13. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Zhao, W.; Ji, C.; Luo, J.; Wang, Y.; Liu, Y.; Weder, W.; Fang, W. Minimally invasive sleeve lobectomy for centrally located lung cancer: A real-world study with propensity-score matching. Front. Oncol. 2023, 13, 1099514. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




