Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Computational Details
3. Results and Discussion
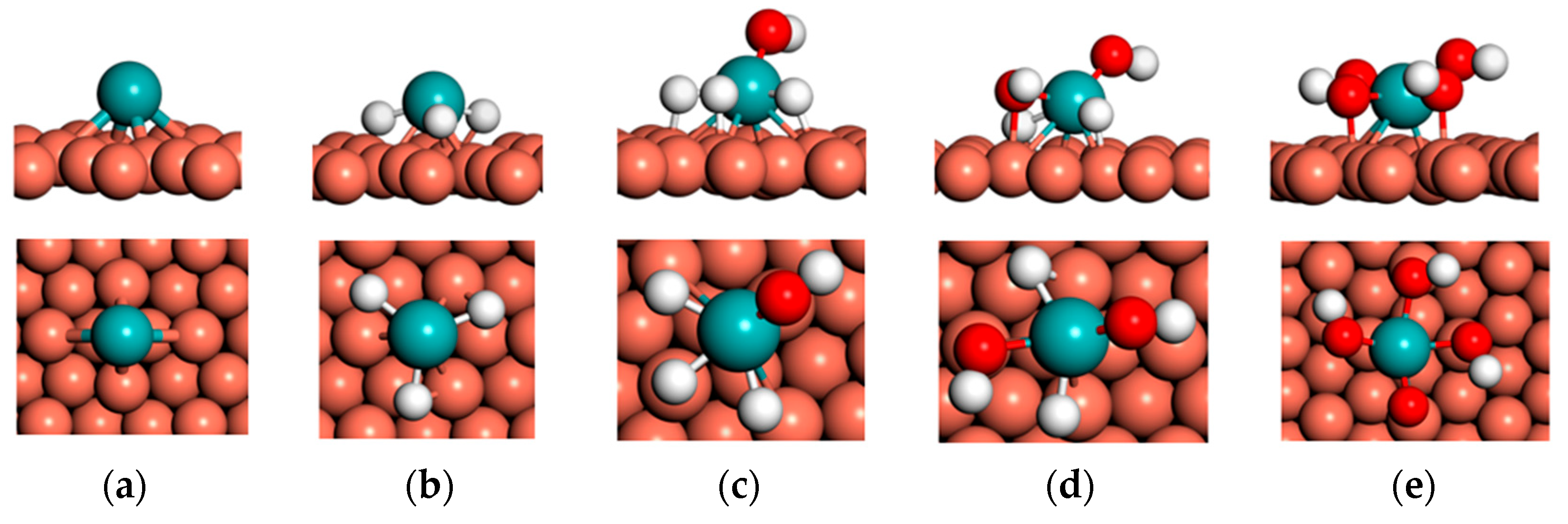
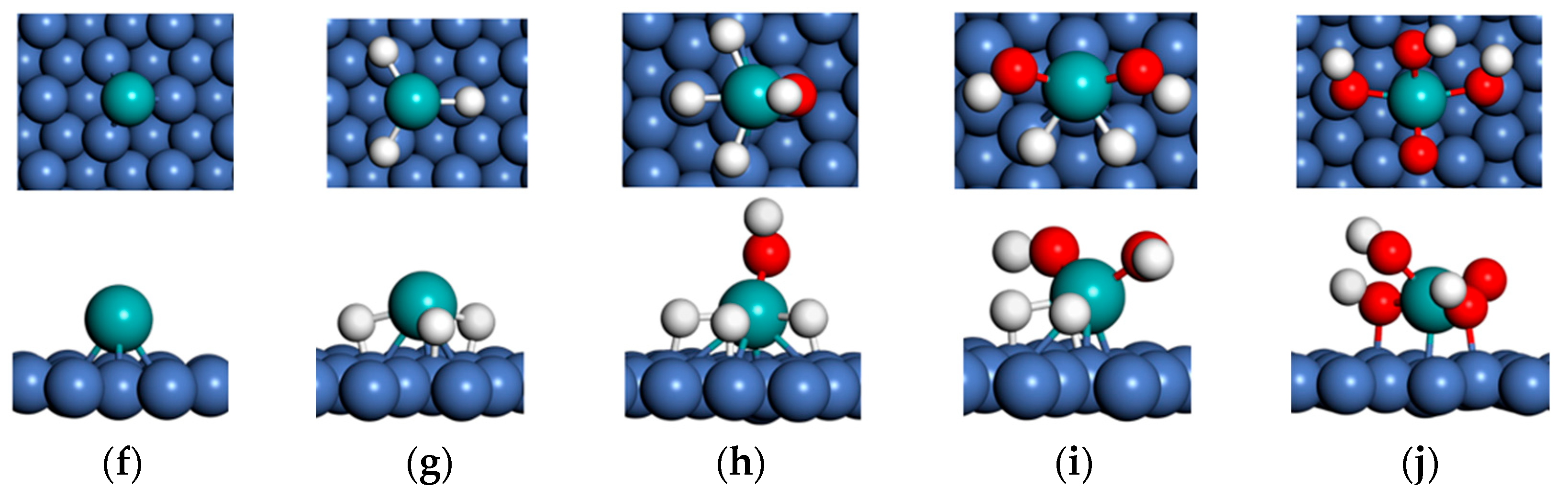
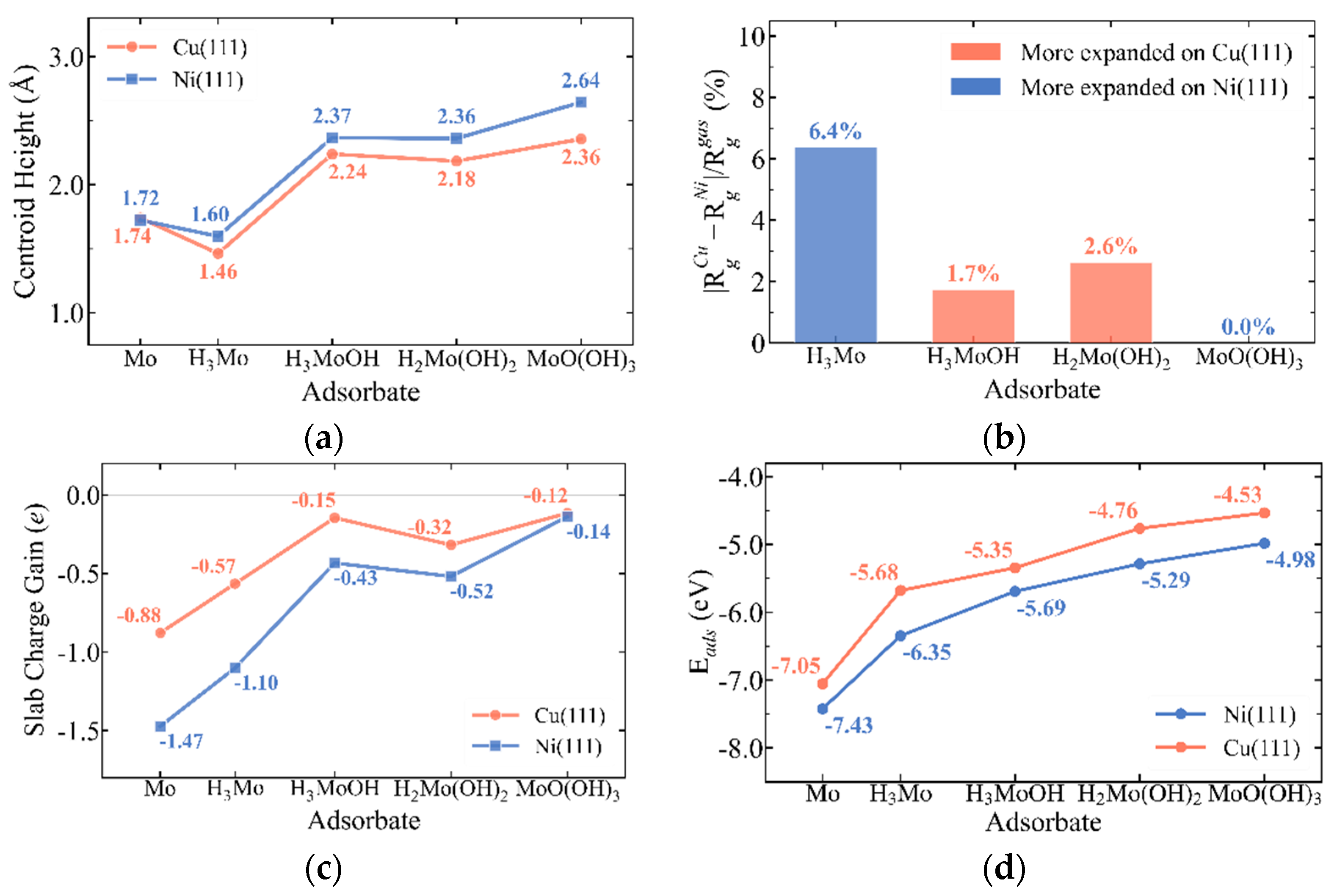
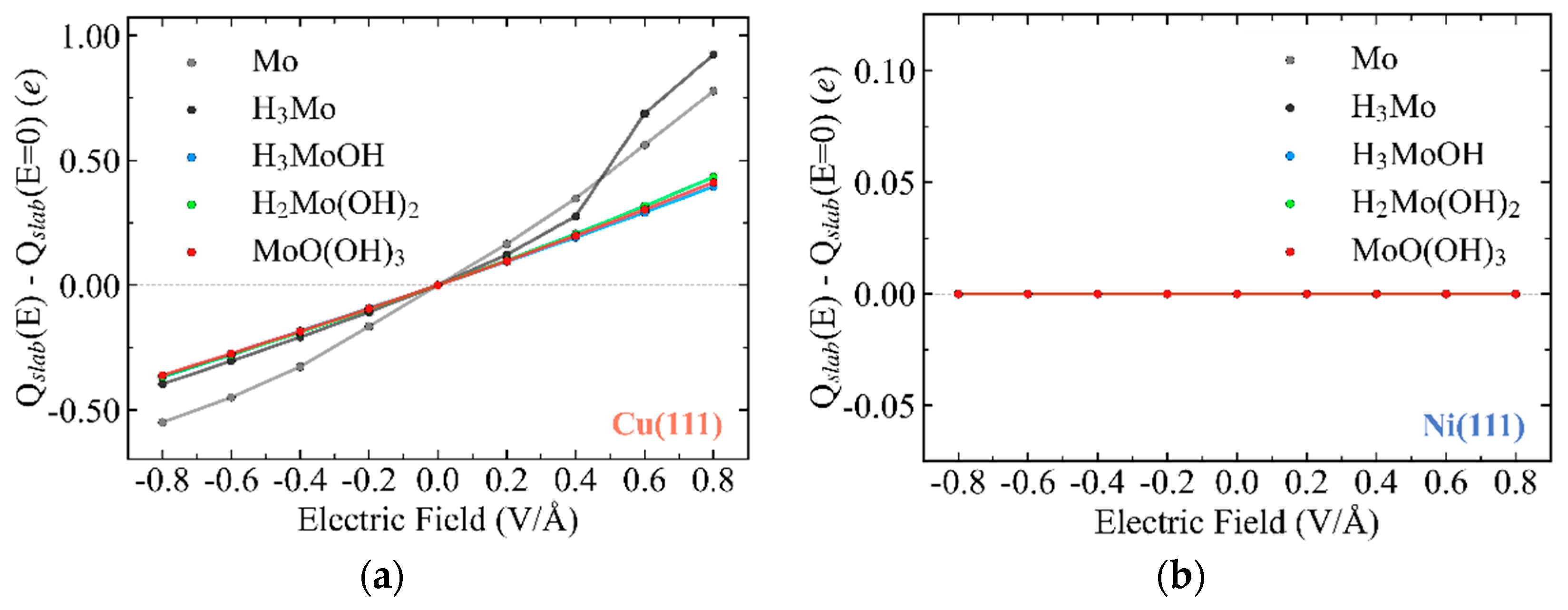
3.1. Substrate-Dependent Adsorbate–Metal Hybridization Governs Structure and Energetics of Mo Complexes on Cu(111) and Ni(111)
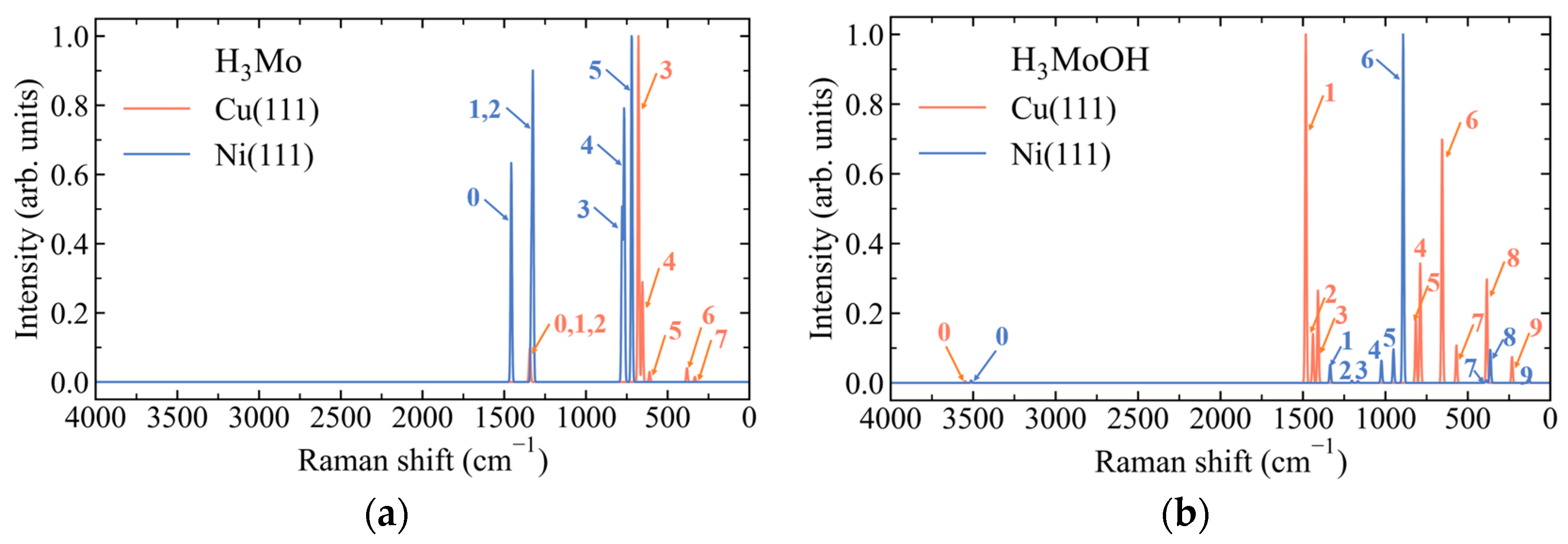
3.2. Substrate-Dependent Weakening and Reorganization of Mo–H Bonding in H3Mo on Cu(111) and Ni(111)
3.3. Substrate-Dependent Redistribution of Mo–H Bonding and Weakly Perturbed O–H Modes in H3MoOH on Cu(111) and Ni(111)
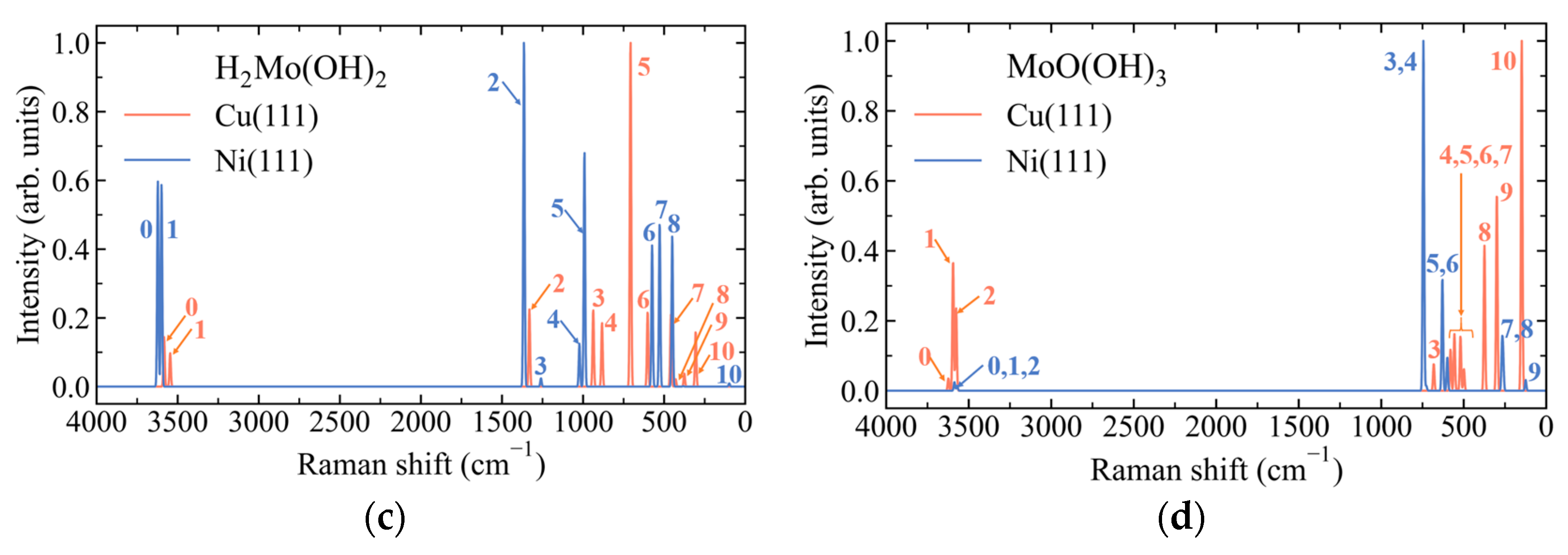
3.4. Substrate-Dependent Symmetry Breaking and Loss of Vibrational Correspondence in H2Mo(OH)2 on Cu(111) and Ni(111)
3.5. Substrate-Dependent Redistribution of Mo–OH Bonding and Mode-Dependent O–H Vibrational Response in MoO(OH)3 on Cu(111) and Ni(111)
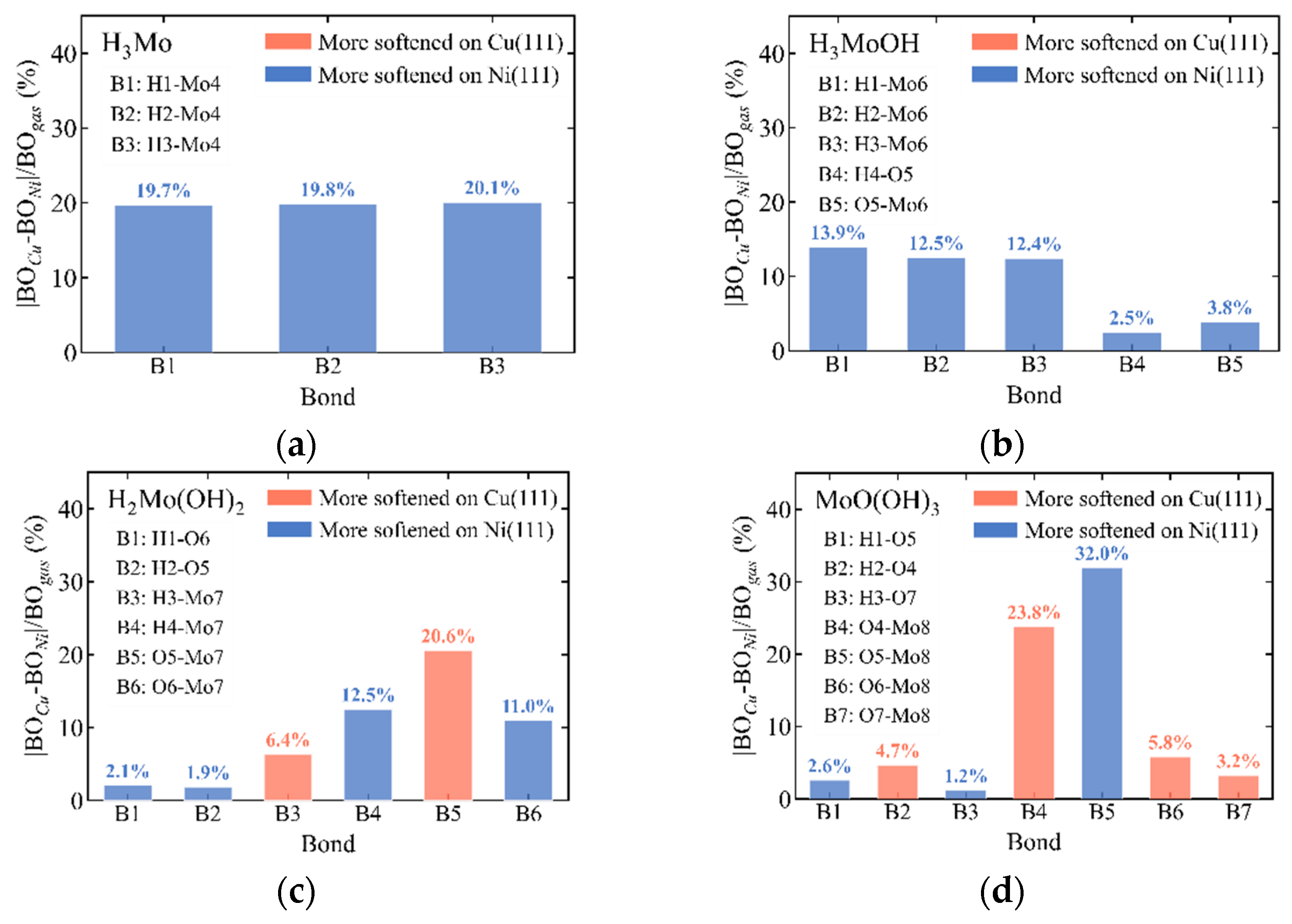
3.6. Substrate-Dependent Bonding and Vibrational Trends Across Mo Complexes on Cu(111) and Ni(111)
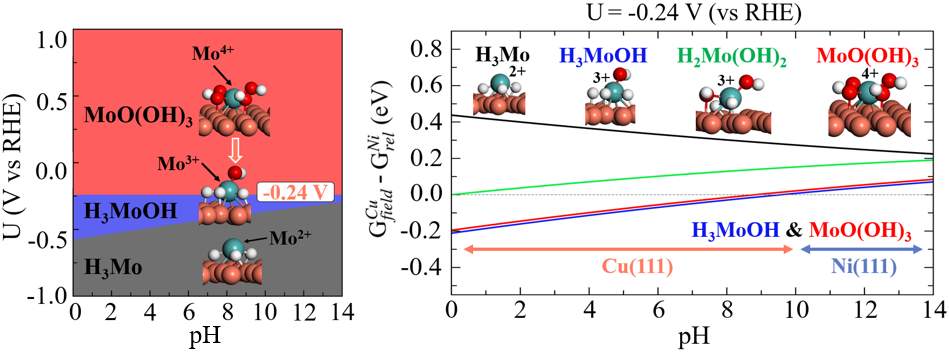
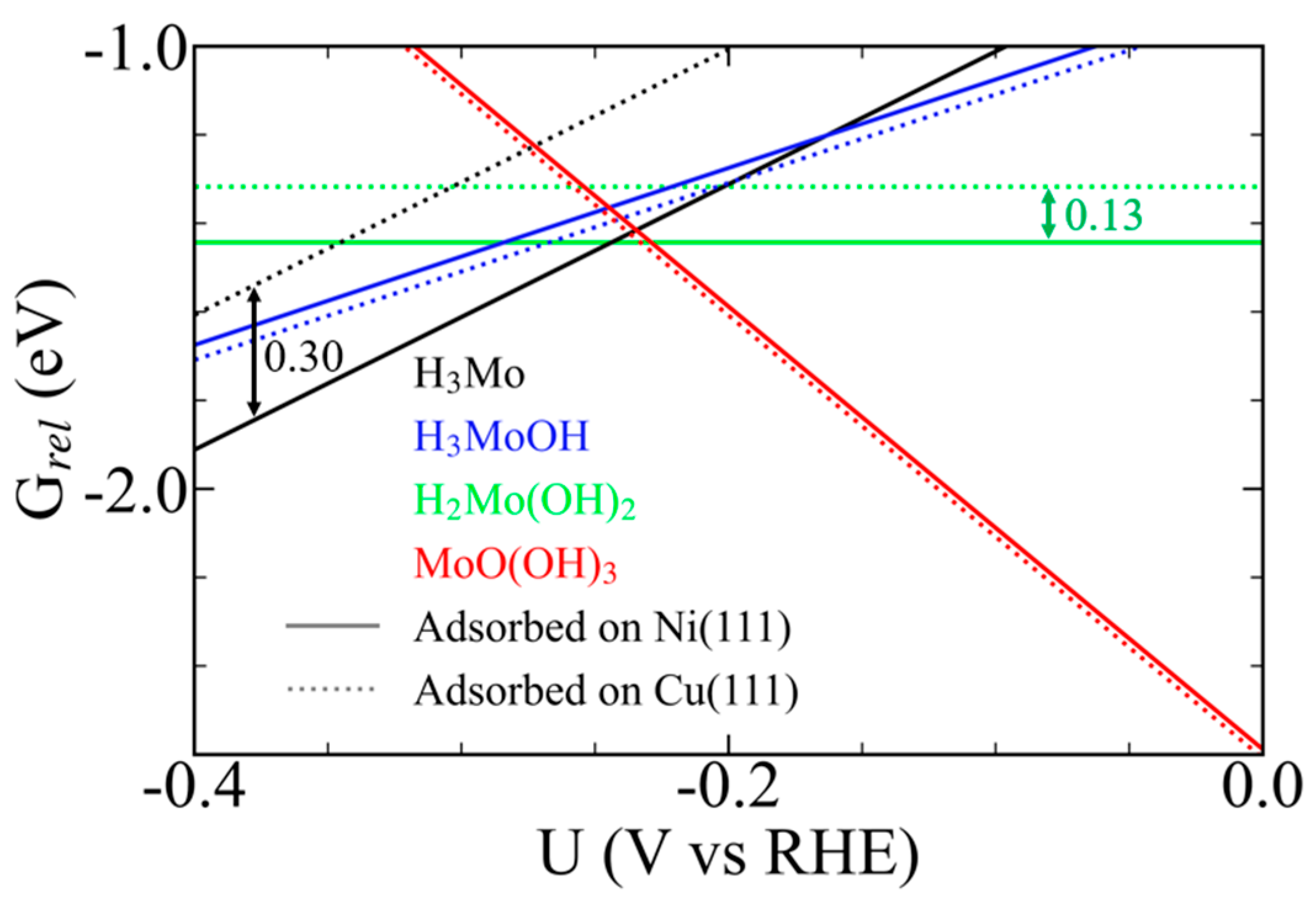
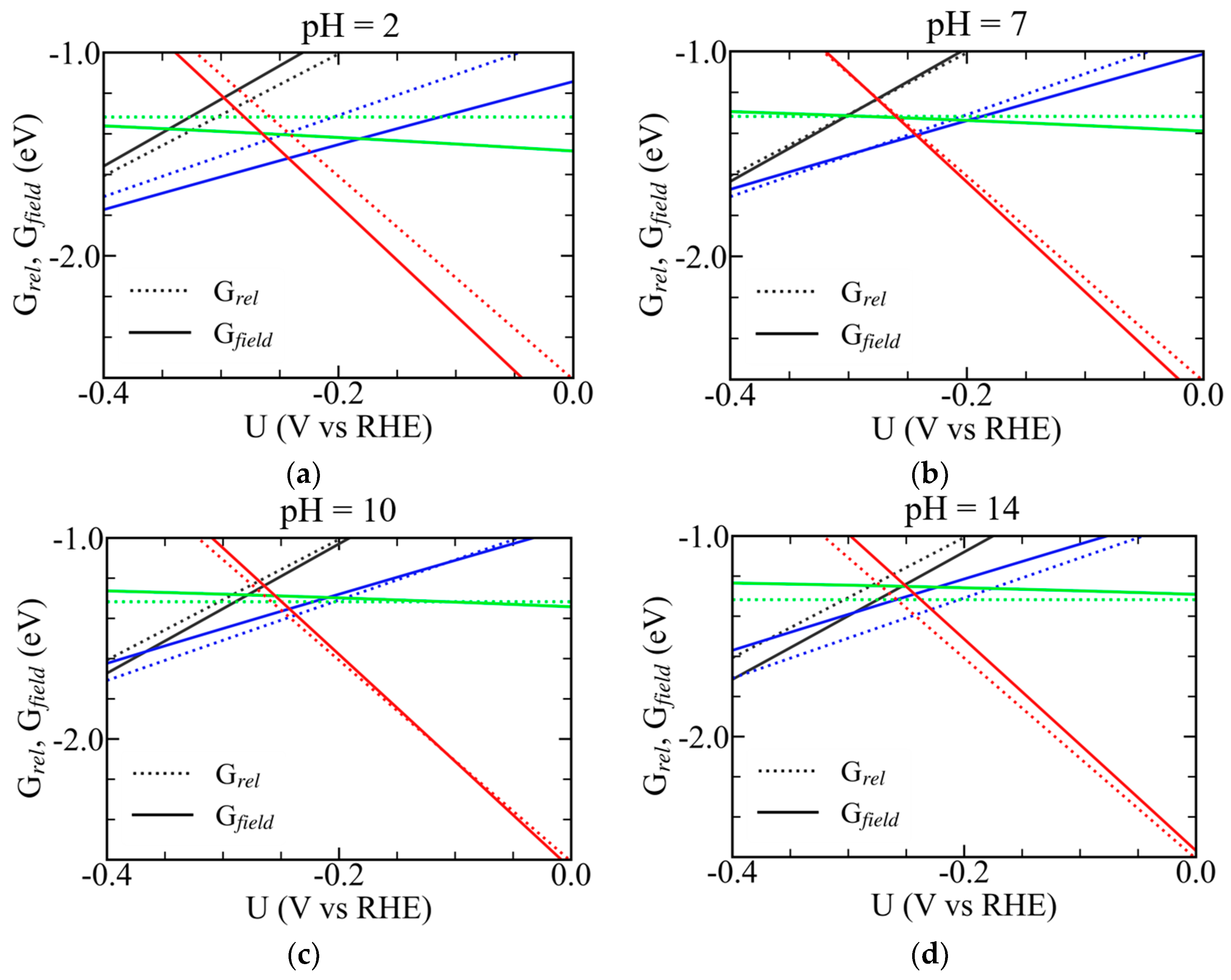
3.7. Field-Dependent Thermodynamics and Electrochemical Stability of Mo Complexes on Cu(111) and Ni(111)
3.8. Contrasting Roles of Ni and Cu in Governing Mo-Centered Redox Thermodynamics and Reactivity in Ni–Mo HER
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 1D | One-dimensional |
| 2D | Two-dimensional |
| CHE | Computational Hydrogen Electrode |
| DDEC | Density Derived Electrostatic and Chemical (method) |
| DFT | Density Functional Theory (method) |
| HER | Hydrogen Evolution Reaction |
| PBE | Perdew–Burke–Ernzerhof (functional) |
| RHE | Reversible Hydrogen Electrode |
| RPBE | Revised Perdew–Burke–Ernzerhof (functional) |
| SHE | Standard Hydrogen Electrode |
| VASP | Vienna Ab initio Simulation Package |
References
- Nishimoto, T.; Obata, K.; Komiya, H.; Naito, T.; Harada, K.; Yoshida, M.; Takanabe, K. Oxidized Copper and Molybdenum Species Exclusively Boosting Electrocatalytic Hydrogen Evolution in Non-Extreme pH Carbonate Buffer Electrolyte. ACS Catal. 2023, 13, 14725–14736. [Google Scholar] [CrossRef]
- Miao, F.; Cui, P.; Yu, S.; Gu, T. In situ fabrication of a 3D self-supported porous Ni–Mo–Cu catalyst for an efficient hydrogen evolution reaction. Dalton Trans. 2023, 52, 8654–8660. [Google Scholar] [CrossRef]
- Wu, X.-F.; Li, Z.-Y.; Wang, H.; Wang, J.-C.; Xi, G.-Q.; Zhao, X.-J.; Zhang, C.-X.; Liao, W.-G.; Ho, J.C. Defect-Engineered Multi-Intermetallic Heterostructures as Multisite Electrocatalysts for Efficient Water Splitting. Advanced Science 2025, 12, 2502244. [Google Scholar] [CrossRef]
- Luo, M.; Yang, J.; Li, X.; Eguchi, M.; Yamauchi, Y.; Wang, Z.-L. Insights into alloy/oxide or hydroxide interfaces in Ni–Mo-based electrocatalysts for hydrogen evolution under alkaline conditions. Chem. Sci. 2023, 14, 3400–3414. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Shi, Y.; Zhou, W.; Yu, Y.; Zhang, B. Unveiling the In Situ Dissolution and Polymerization of Mo in Ni4 Mo Alloy for Promoting the Hydrogen Evolution Reaction. Angew Chem Int Ed 2021, 60, 7051–7055. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Shi, C.; Guo, X.; He, Z.; Pan, L.; Huang, Z.-F.; Zhang, X.; Zou, J.-J. Rational Design of Better Hydrogen Evolution Electrocatalysts for Water Splitting: A Review. Advanced Science 2022, 9, 2200307. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, Y.; Zhao, Z.; Zhang, Q.-H.; Huang, L.-B.; Gu, L.; Lu, G.; Hu, J.-S.; Wan, L.-J. Steering elementary steps towards efficient alkaline hydrogen evolution via size-dependent Ni/NiO nanoscale heterosurfaces. Natl Sci Rev 2020, 7, 27–36. [Google Scholar] [CrossRef]
- Shen, L.; Lu, B.; Li, Y.; Liu, J.; Huang-fu, Z.; Peng, H.; Ye, J.; Qu, X.; Zhang, J.; Li, G.; Cai, W.; Jiang, Y.; Sun, S. Interfacial Structure of Water as a New Descriptor of the Hydrogen Evolution Reaction. Angewandte Chemie International Edition 2020, 59, 22397–22402. [Google Scholar] [CrossRef]
- Liu, E.; Qin, X.; Delmo, E.P.; Shao, M.; Vegge, T.; Drisdell, W.S. Discovering the Catalytic Role of Interfacial Water Towards Hydrogen Evolution/Oxidation Reactions in Aqueous Solutions, Meet. Abstr. MA2024-02. 2024, 2822. [Google Scholar] [CrossRef]
- Sun, M.; Staykov, A.; Yamauchi, M. Understanding the Roles of Hydroxide in CO2 Electroreduction on a Cu Electrode for Achieving Variable Selectivity. ACS Catal. 2022, 12, 14856–14863. [Google Scholar] [CrossRef]
- Guha, A.; Sahoo, M.; Alam, K.; Rao, D.K.; Sen, P.; Narayanan, T.N. Role of water structure in alkaline water electrolysis. iScience 2022, 25, 104835. [Google Scholar] [CrossRef]
- Zhang; Yao, J.; Liu, J.; Zhang, T.; Wan, H.; Wang, H. Reducing the pH dependence of hydrogen evolution kinetics via surface reactivity diversity in medium-entropy alloys. EES Catal. 2023, 1, 1017–1024. [Google Scholar] [CrossRef]
- Bau, J.A.; Kozlov, S.M.; Azofra, L.M.; Ould-Chikh, S.; Emwas, A.-H.; Idriss, H.; Cavallo, L.; Takanabe, K. Role of Oxidized Mo Species on the Active Surface of Ni–Mo Electrocatalysts for Hydrogen Evolution under Alkaline Conditions. ACS Catal. 2020, 10, 12858–12866. [Google Scholar] [CrossRef]
- Bau, J.A.; Ahmad, R.; Cavallo, L.; Rueping, M. A Unified Theory for H2 Evolution on Mo-Based Electrocatalysts. ACS Energy Lett. 2022, 7, 3695–3702. [Google Scholar] [CrossRef]
- Bau, J.A.; Emwas, A.-H.; Nikolaienko, P.; Aljarb, A.A.; Tung, V.; Rueping, M. Mo3+ hydride as the common origin of H2 evolution and selective NADH regeneration in molybdenum sulfide electrocatalysts. Nat Catal 2022, 5, 397–404. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, J.; Zhang, W.; Wu, A. Growth of Ni/Mo/Cu on carbon fiber paper: An efficient electrocatalyst for hydrogen evolution reaction. International Journal of Hydrogen Energy 2021, 46, 35550–35558. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, Z.; Oliveira, A.M.; Xi, S.; Zhiani, M.; Zhang, J.; Tu, Z.; Xiao, F.; Wang, S.; Yan, Y.; Xiao, J. Accelerating Hydrogen Desorption of Nickel Molybdenum Cathode via Copper Modulation for Pure-Water-Fed Hydroxide Exchange Membrane Electrolyzer. Advanced Functional Materials 2024, 34, 2313275. [Google Scholar] [CrossRef]
- Hansen, H.A.; Rossmeisl, J.; Nørskov, J.K. Surface Pourbaix diagrams and oxygen reduction activity of Pt, Ag and Ni(111) surfaces studied by DFT. Phys. Chem. Chem. Phys. 2008, 10, 3722–3730. [Google Scholar] [CrossRef]
- Nørskov, J.K.; Rossmeisl, J.; Logadottir, A.; Lindqvist, L.; Kitchin, J.R.; Bligaard, T.; Jónsson, H. Origin of the Overpotential for Oxygen Reduction at a Fuel-Cell Cathode. J. Phys. Chem. B 2004, 108, 17886–17892. [Google Scholar] [CrossRef]
- Kelly, S.R.; Kirk, C.; Chan, K.; Nørskov, J.K. Electric Field Effects in Oxygen Reduction Kinetics: Rationalizing pH Dependence at the Pt(111), Au(111), and Au(100) Electrodes. J. Phys. Chem. C 2020, 124, 14581–14591. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, D.; Wang, Y.; Li, H. Reversible Hydrogen Electrode (RHE) Scale Dependent Surface Pourbaix Diagram at Different pH. Langmuir 2024, 40, 7632–7638. [Google Scholar] [CrossRef] [PubMed]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169–11186. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Computational Materials Science 1996, 6, 15–50. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Hammer, B.; Hansen, L.B.; Nørskov, J.K. Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals. Phys. Rev. B 1999, 59, 7413–7421. [Google Scholar] [CrossRef]
- Wellendorff, J.; Silbaugh, T.L.; Garcia-Pintos, D.; Nørskov, J.K.; Bligaard, T.; Studt, F.; Campbell, C.T. A benchmark database for adsorption bond energies to transition metal surfaces and comparison to selected DFT functionals. Surface Science 2015, 640, 36–44. [Google Scholar] [CrossRef]
- Tonigold, K.; Groß, A. Dispersive interactions in water bilayers at metallic surfaces: A comparison of the PBE and RPBE functional including semiempirical dispersion corrections. J Comput Chem 2012, 33, 695–701. [Google Scholar] [CrossRef]
- Blöchl, P.E. Projector augmented-wave method. Phys. Rev. B 1994, 50, 17953–17979. [Google Scholar] [CrossRef]
- Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 1999, 59, 1758–1775. [Google Scholar] [CrossRef]
- Dudarev, S.L.; Botton, G.A.; Savrasov, S.Y.; Humphreys, C.J.; Sutton, A.P. Electron-energy-loss spectra and the structural stability of nickel oxide: An LSDA+U study. Phys. Rev. B 1998, 57, 1505–1509. [Google Scholar] [CrossRef]
- Lutfalla, S.; Shapovalov, V.; Bell, A.T. Calibration of the DFT/GGA+U Method for Determination of Reduction Energies for Transition and Rare Earth Metal Oxides of Ti, V, Mo, and Ce. J. Chem. Theory Comput. 2011, 7, 2218–2223. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Maxisch, T.; Ceder, G. Oxidation energies of transition metal oxides within the GGA + U framework. Phys. Rev. B 2006, 73, 195107. [Google Scholar] [CrossRef]
- Monkhorst, H.J.; Pack, J.D. Special points for Brillouin-zone integrations. Phys. Rev. B 1976, 13, 5188–5192. [Google Scholar] [CrossRef]
- Methfessel, M.; Paxton, A.T. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 1989, 40, 3616–3621. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J Chem Phys 2010, 132, 154104. [Google Scholar] [CrossRef]
- Mathew, K.; Sundararaman, R.; Letchworth-Weaver, K.; Arias, T.A.; Hennig, R.G. Implicit solvation model for density-functional study of nanocrystal surfaces and reaction pathways. J. Chem. Phys. 2014, 140, 084106. [Google Scholar] [CrossRef] [PubMed]
- Mathew, K.; Kolluru, V.S.C.; Mula, S.; Steinmann, S.N.; Hennig, R.G. Implicit self-consistent electrolyte model in plane-wave density-functional theory. J. Chem. Phys. 2019, 151, 234101. [Google Scholar] [CrossRef] [PubMed]
- Archer, D.G.; Wang, P. The Dielectric Constant of Water and Debye-Hückel Limiting Law Slopes. Journal of Physical and Chemical Reference Data 1990, 19, 371–411. [Google Scholar] [CrossRef]
- Manz, T.A.; Limas, N.G. DDEC6: A Method for Computing Even-Tempered Net Atomic Charges in Periodic and Nonperiodic Materials. 2015. [Google Scholar] [CrossRef]
- Limas, N.G.; Manz, T.A. Introducing DDEC6 atomic population analysis: part 4. Efficient parallel computation of net atomic charges, atomic spin moments, bond orders, and more. RSC Adv. 2018, 8, 2678–2707. [Google Scholar] [CrossRef] [PubMed]
- Wang, V.; Xu, N.; Liu, J.-C.; Tang, G.; Geng, W.-T. VASPKIT: A user-friendly interface facilitating high-throughput computing and analysis using VASP code. Computer Physics Communications 2021, 267, 108033. [Google Scholar] [CrossRef]
- Trasatti, S. The Absolute Electrode Potential: An Explanatory Note: (Recommendations 1986). n.d. [Google Scholar] [CrossRef]
- Jerkiewicz, G. Standard and Reversible Hydrogen Electrodes: Theory, Design, Operation, and Applications. ACS Catal. 2020, 10, 8409–8417. [Google Scholar] [CrossRef]
- Singh, A.K.; Zhou, L.; Shinde, A.; Suram, S.K.; Montoya, J.H.; Winston, D.; Gregoire, J.M.; Persson, K.A. Electrochemical Stability of Metastable Materials. Chem. Mater. 2017, 29, 10159–10167. [Google Scholar] [CrossRef]
- Łukomska, A.; Sobkowski, J. Potential of zero charge of monocrystalline copper electrodes in perchlorate solutions. Journal of Electroanalytical Chemistry 2004, 567, 95–102. [Google Scholar] [CrossRef]
- Ohmori, T. MEASUREMENT OF THE POTENTIAL OF ZERO-CHARGE ON NICKEL ELECTRODE BY THE GALVANOSTATIC TRANSIENT METHOD. n.d. [Google Scholar]
- Porezag, D.; Pederson, M.R. Infrared intensities and Raman-scattering activities within density-functional theory. Phys. Rev. B 1996, 54, 7830–7836. [Google Scholar] [CrossRef]
- Baroni, S.; De Gironcoli, S.; Dal Corso, A.; Giannozzi, P. Phonons and related crystal properties from density-functional perturbation theory. Rev. Mod. Phys. 2001, 73, 515–562. [Google Scholar] [CrossRef]
- Gonze, X. Perturbation expansion of variational principles at arbitrary order. Phys. Rev. A 1995, 52, 1086–1095. [Google Scholar] [CrossRef]
- Fonari, A.; Stauffer, S. vasp_raman.py. 2013. Available online: https://github.com/raman-sc/VASP/.
- Lazzeri, M.; Mauri, F. First-Principles Calculation of Vibrational Raman Spectra in Large Systems: Signature of Small Rings in Crystalline ${\mathrm{S}\mathrm{i}\mathrm{O}}_{2}$. Phys. Rev. Lett. 2003, 90, 036401. [Google Scholar] [CrossRef]
- Kabsch, W. A solution for the best rotation to relate two sets of vectors. Acta Cryst A 1976, 32, 922–923. [Google Scholar] [CrossRef]
- Yu, M.; Moayedpour, S.; Yang, S.; Dardzinski, D.; Wu, C.; Pribiag, V.S.; Marom, N. Dependence of the electronic structure of the EuS/InAs interface on the bonding configuration. Phys. Rev. Materials 2021, 5, 064606. [Google Scholar] [CrossRef]
- Ruban, A.; Hammer, B.; Stoltze, P.; Skriver, H.L.; Nørskov, J.K. Surface electronic structure and reactivity of transition and noble metals1. Journal of Molecular Catalysis A: Chemical 1997, 115, 421–429. [Google Scholar] [CrossRef]
- Hammer, J.K. Norskov, Why gold is the noblest of all the metals. Nature 1995, 376, 238–240. [Google Scholar] [CrossRef]
- Hammer; Morikawa, Y.; Nørskov, J.K. CO Chemisorption at Metal Surfaces and Overlayers. Phys. Rev. Lett. 1996, 76, 2141–2144. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, S.; Waghmare, U.V.; Lee, S.-C. An improved d-band model of the catalytic activity of magnetic transition metal surfaces. Sci Rep 2016, 6, 35916. [Google Scholar] [CrossRef]
- Hammer, B.; Nørskov, J.K. Electronic factors determining the reactivity of metal surfaces. Surface Science 1995, 343, 211–220. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



