Submitted:
13 April 2026
Posted:
14 April 2026
You are already at the latest version
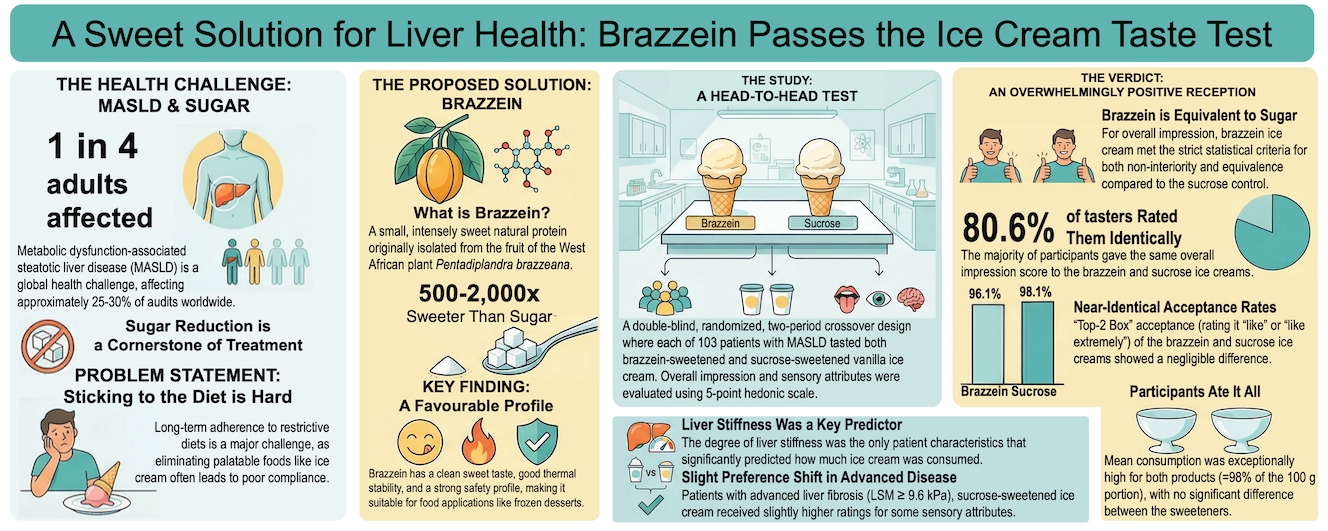
Abstract
Keywords:
1. Introduction
2. Materials and Methods
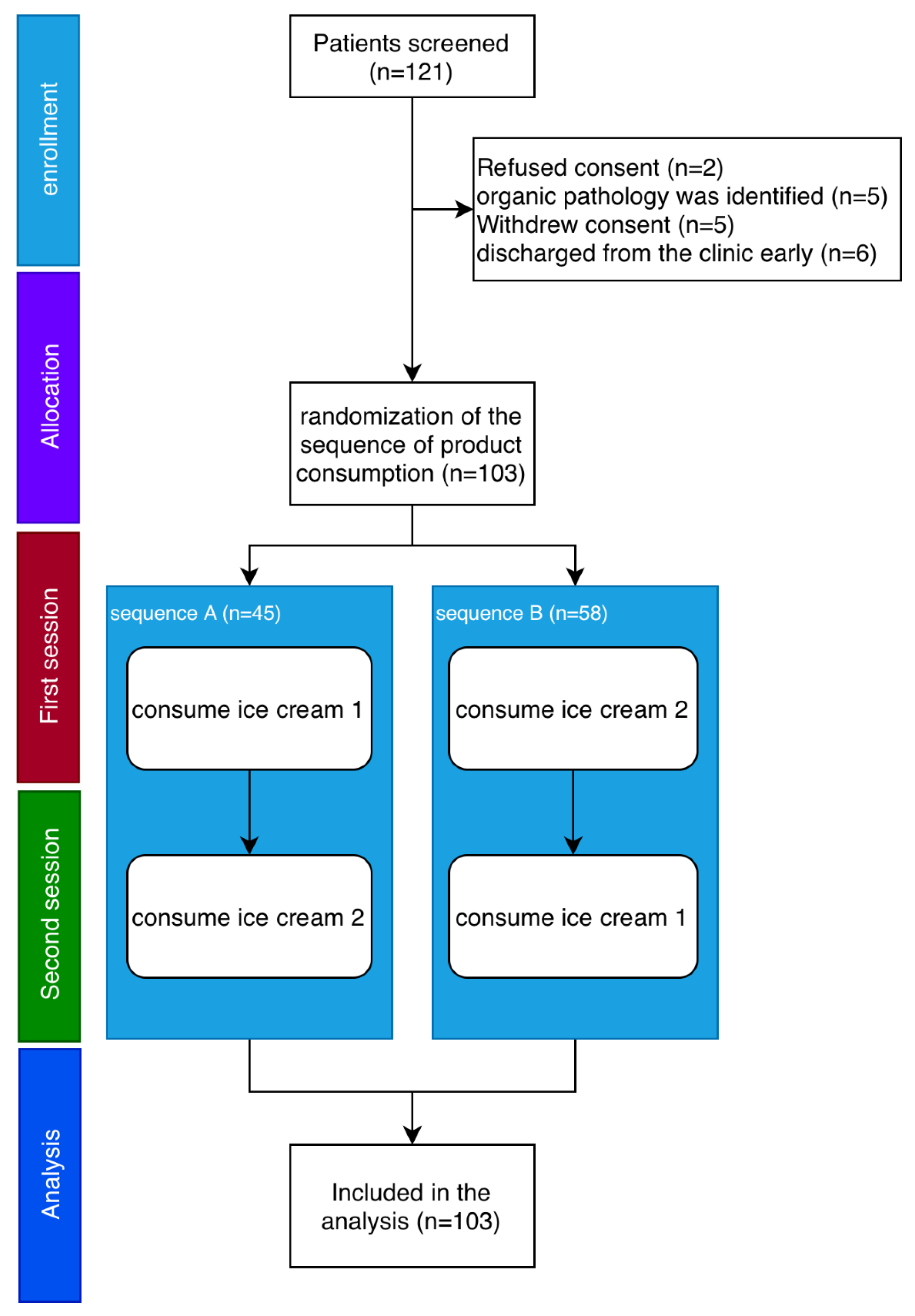
2.1. Participants
2.2. Study Design
2.3. Ice Cream Formulation
2.4. Sensory Evaluation Procedures
2.5. Data Collection and Management
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Evaluation of Period and Carryover Effects
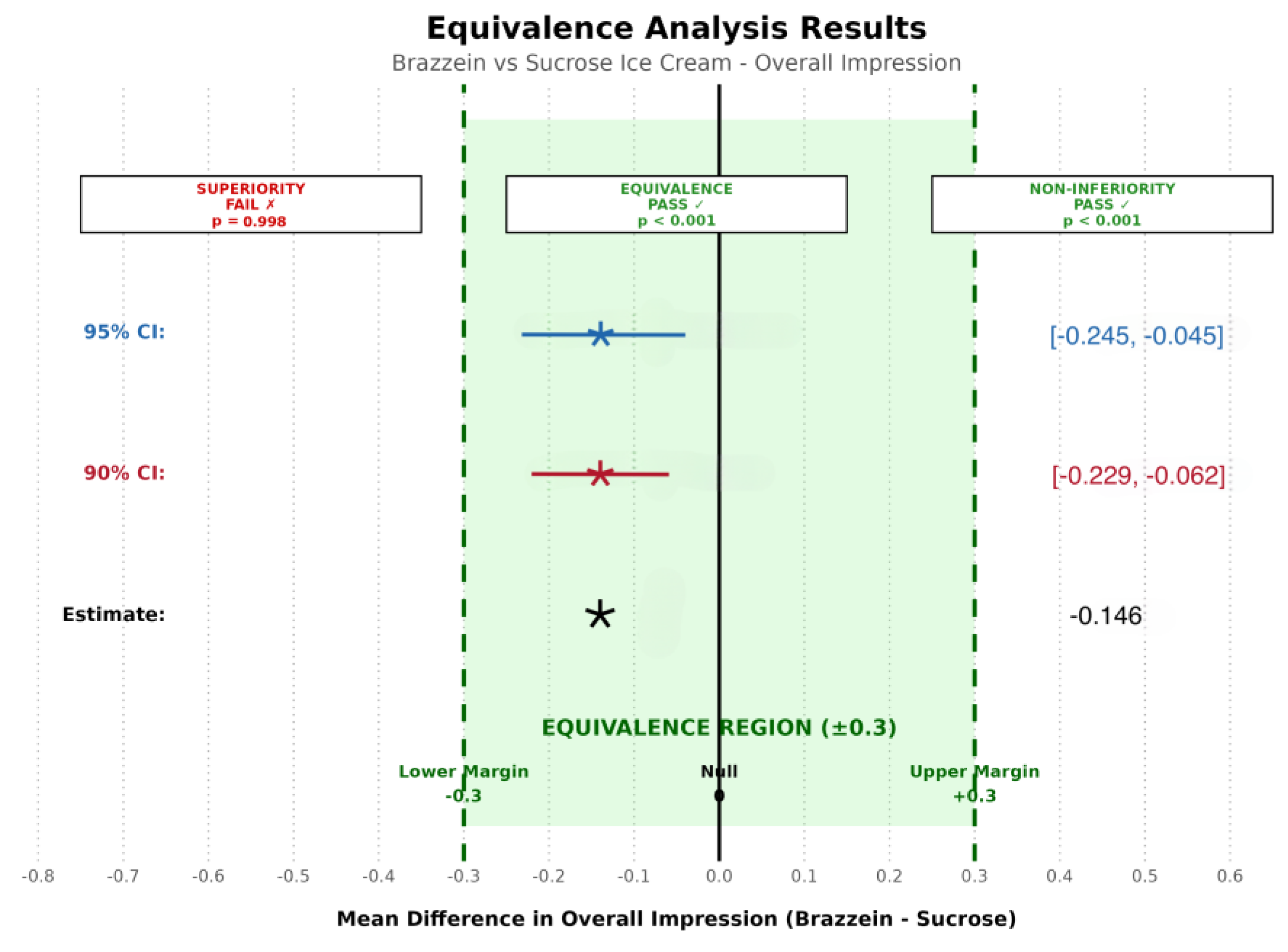
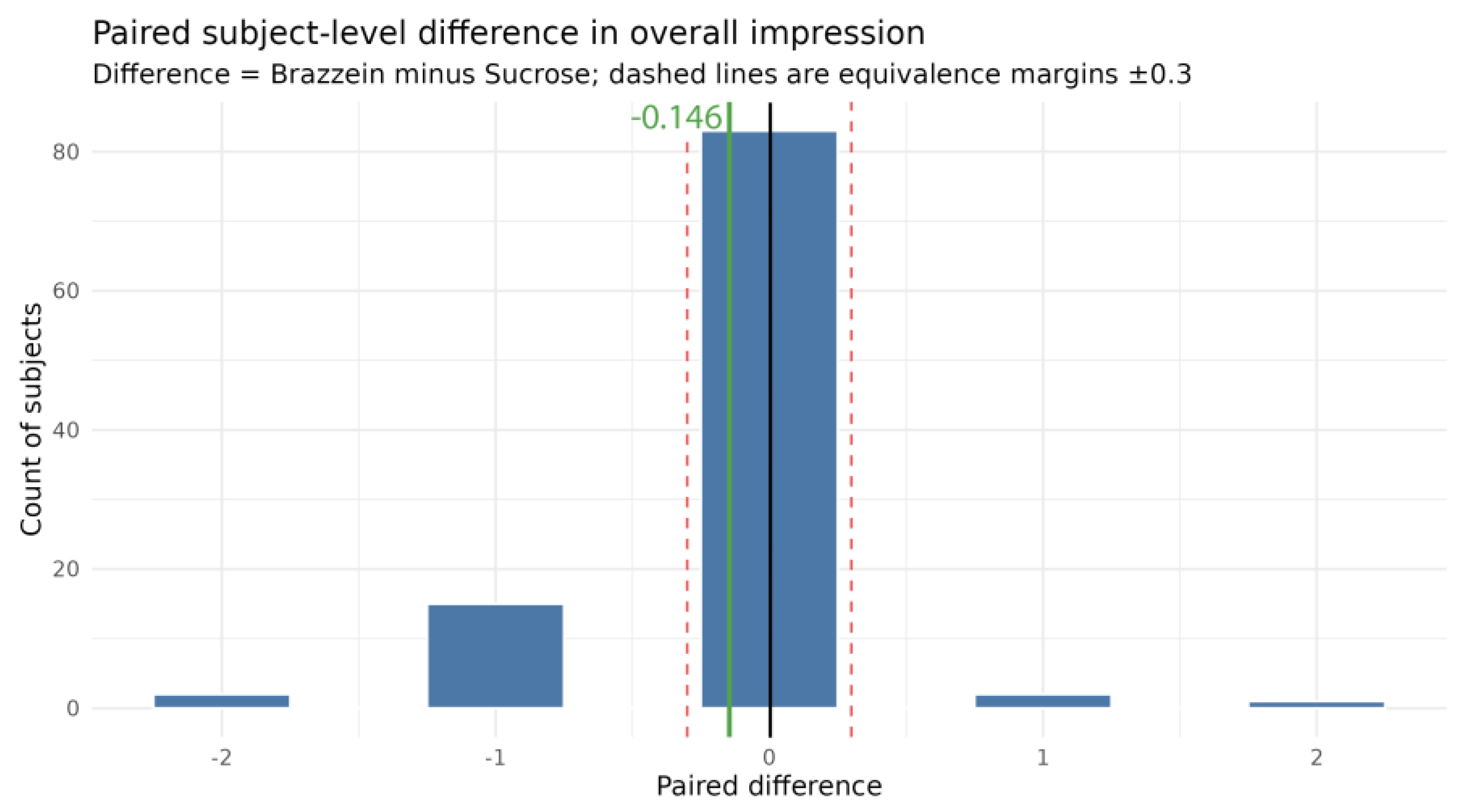
3.3. Primary Analysis Results
3.4. Secondary Analyses
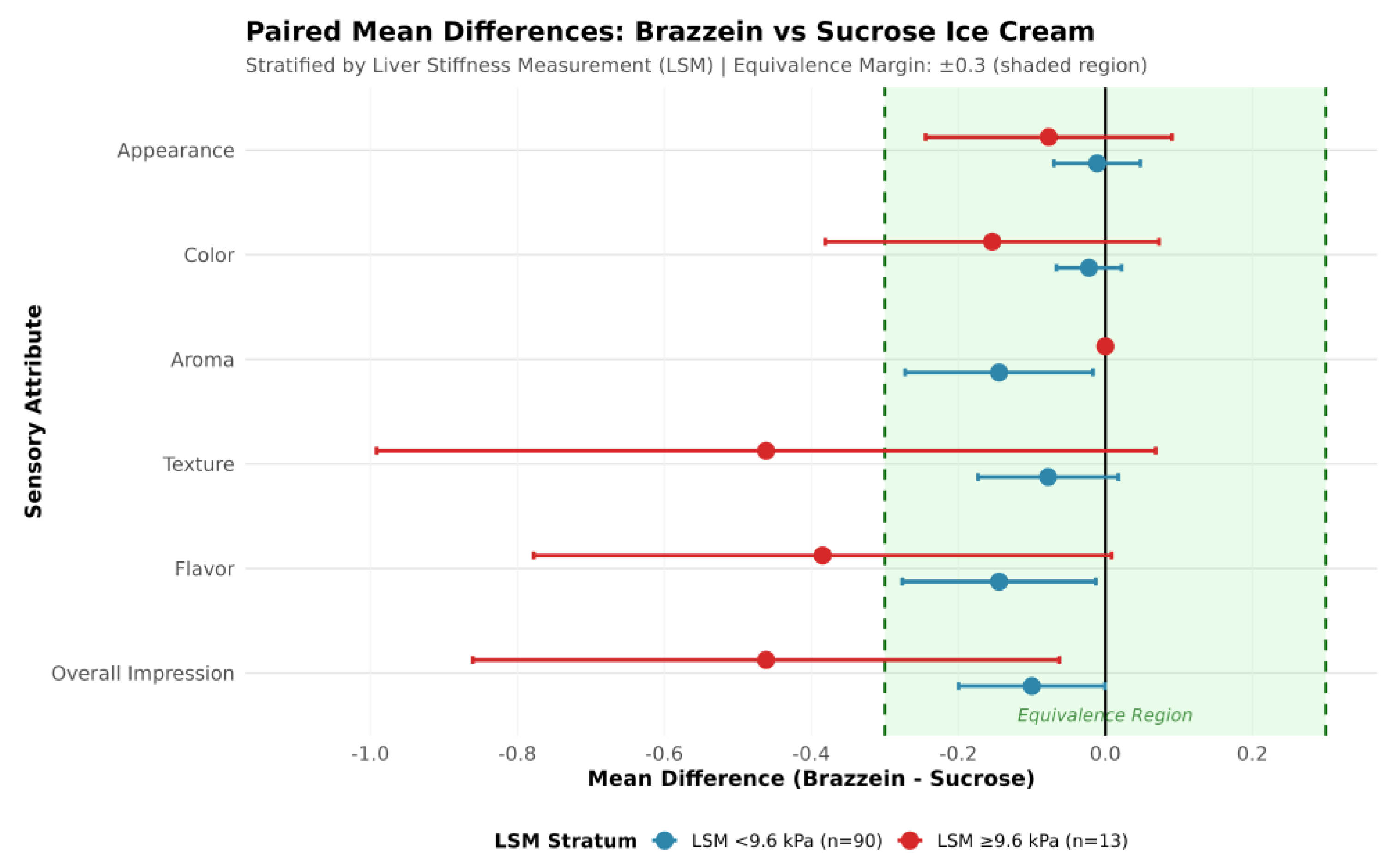
3.4.1. Individual Sensory Attributes
3.4.2. Top-2 Box Acceptance
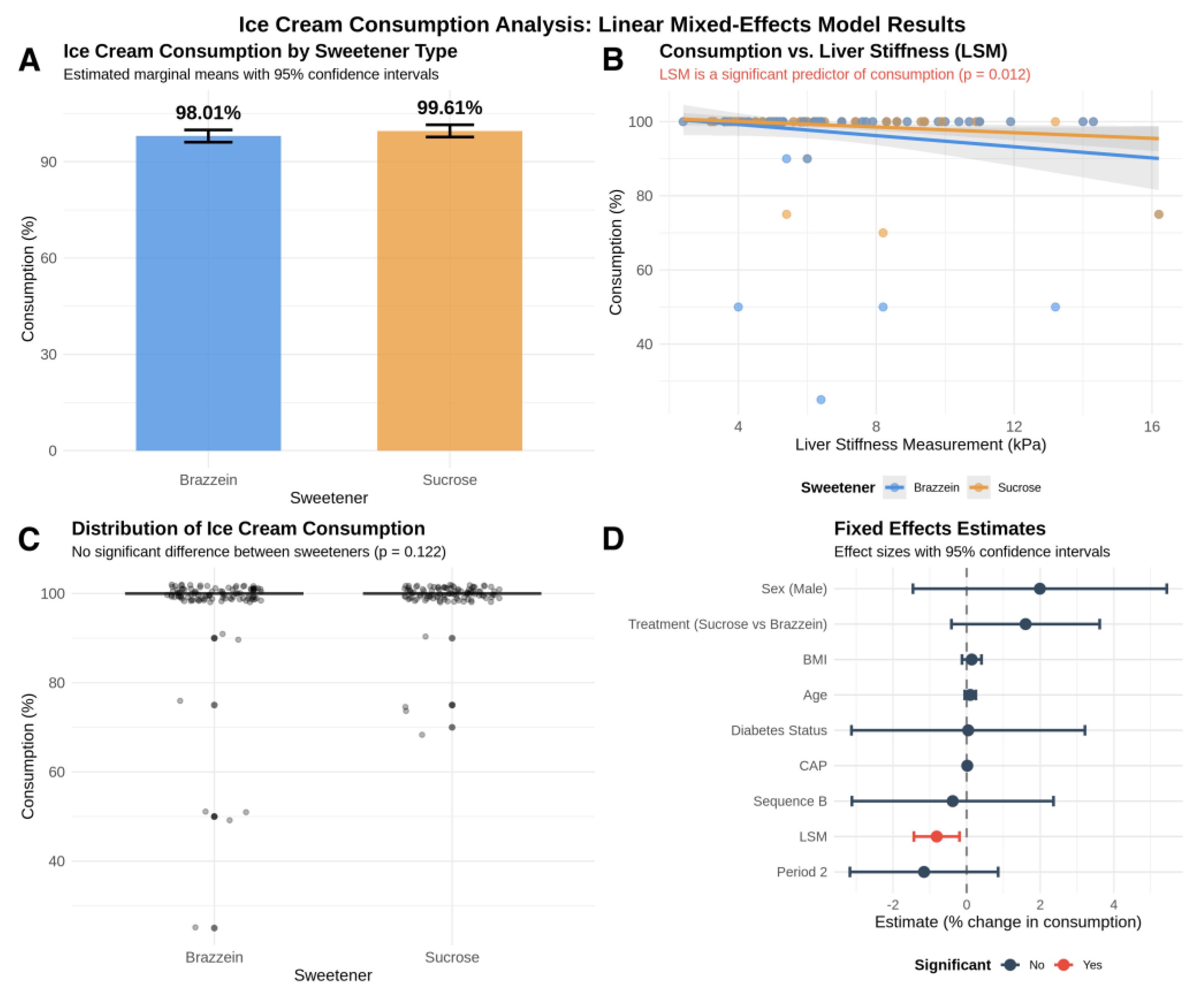
3.4.3. Ice Cream Consumption Analysis
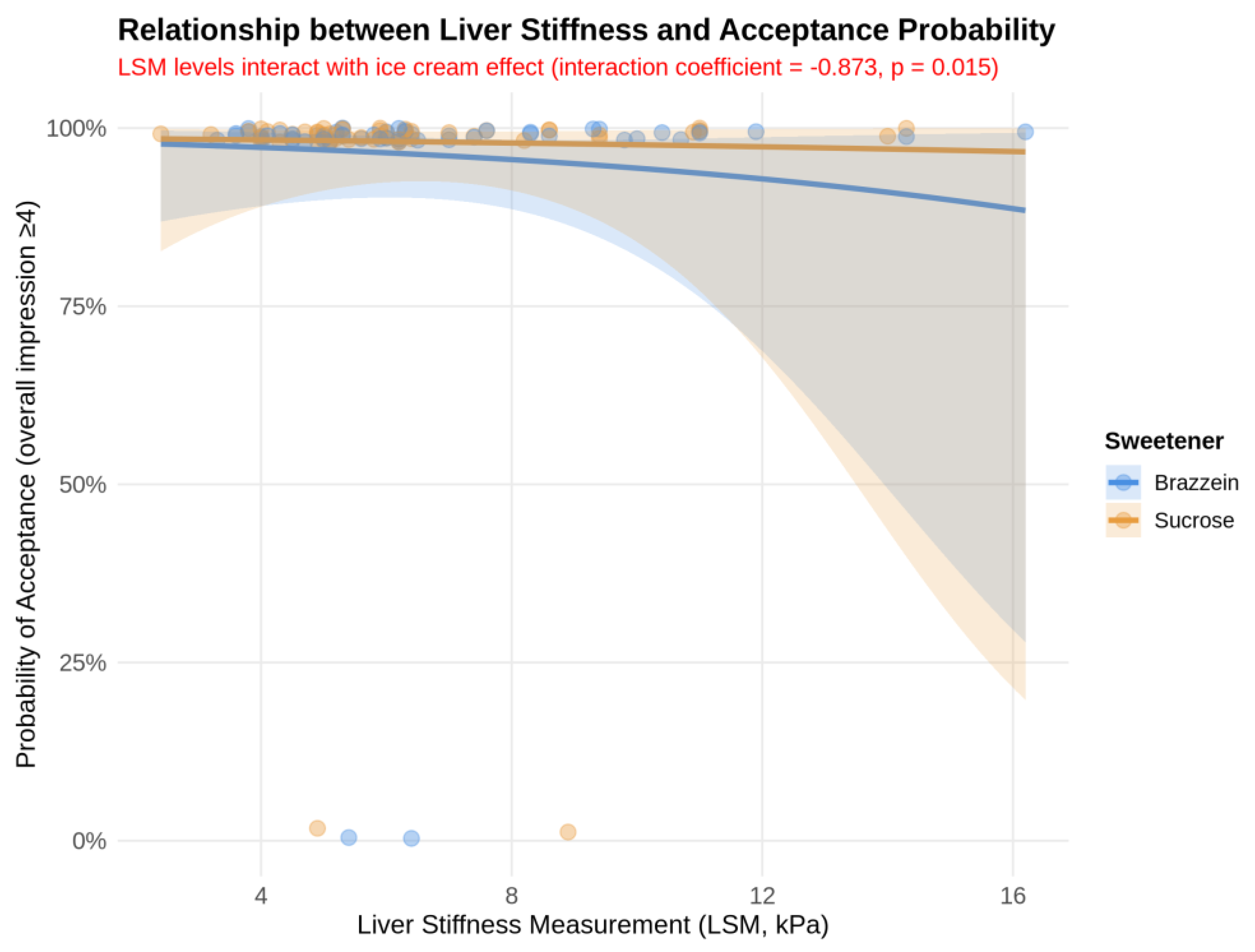
3.4.4. Subgroup Analyses
4. Discussion
4.1. Summary of Main Findings
4.2. Brazzein vs Sucrose in Ice Cream: Positioning Within Sensory Literature
4.3. Implications for Sugar Reduction and Dietary Management in MASLD
4.4. Disease-Related Differences in Sensory Perception: The Liver Stiffness Signal
4.5. Methodological and Sensometric Considerations
4.6. Strengths and Limitations
4.7. Practical Implications and Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAP | Controlled attenuation parameter |
| CLMM | Cumulative link mixed model |
| GLMM | Generalized linear mixed model |
| LSM | Liver stiffness measurement |
| MASL | Metabolic dysfunction-associated steatotic liver |
| MASLD | Metabolic dysfunction-associated steatotic liver disease |
| MASH | Metabolic dysfunction-associated steatohepatitis |
| MCA | Multiple Correspondence Analysis |
| REML | Restricted maximum likelihood |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
| TOST | Two one-sided tests |
| VCTE | Vibration-controlled transient elastography |
Appendix A
| Criteria | |
|---|---|
| Inclusion criteria |
|
| Exclusion criteria |
|
| Attribute | W_statistic | p_value | Interpretation |
|---|---|---|---|
| appearance | 5.0 | 0.5716 | No significant carryover |
| color | 3.5 | 0.1294 | No significant carryover |
| aroma | 18.0 | 0.0238 | Significant carryover detected |
| texture | 30.0 | 0.0197 | Significant carryover detected |
| flavor | 85.0 | 0.0071 | Significant carryover detected |
| Attribute | Brazzein mean | Sucrose mean | Odds ratio (95% CI) | p value |
|---|---|---|---|---|
| Appearance | 4.978 | 4.983 | 0.772 (0.047, 12.689) | 0.8562 |
| Color | 5.000 | 5.000 | Not estimable | — |
| Aroma | 4.711 | 4.776 | 0.739 (0.240, 2.278) | 0.5985 |
| Texture | 4.911 | 4.914 | 0.587 (0.125, 2.765) | 0.5007 |
| Flavor | 4.800 | 4.776 | 0.810 (0.293, 2.234) | 0.6833 |
References
- Feng, G.; Targher, G.; Byrne, C.D.; Yilmaz, Y.; Wai-Sun Wong, V.; Adithya Lesmana, C.R.; Adams, L.A.; Boursier, J.; Papatheodoridis, G.; El-Kassas, M., et al. Global burden of metabolic dysfunction-associated steatotic liver disease, 2010 to 2021. JHEP Reports 2025, 7, 101271. [CrossRef]
- Golabi, P.; Isakov, V.; Younossi, Z.M. Nonalcoholic Fatty Liver Disease: Disease Burden and Disease Awareness. Clinics in liver disease 2023, 27, 173–186. [CrossRef]
- Kanwal, F.; Neuschwander-Tetri, B.A.; Loomba, R.; Rinella, M.E. Metabolic dysfunction–associated steatotic liver disease: Update and impact of new nomenclature on the American Association for the Study of Liver Diseases practice guidance on nonalcoholic fatty liver disease. Hepatology 2024, 79, 1212–1219. [CrossRef]
- Tacke, F.; Horn, P.; Wai-Sun Wong, V.; Ratziu, V.; Bugianesi, E.; Francque, S.; Zelber-Sagi, S.; Valenti, L.; Roden, M.; Schick, F., et al. EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). Journal of Hepatology 2024, 81, 492–542. [CrossRef]
- Kokkorakis, M.; Boutari, C.; Hill, M.A.; Kotsis, V.; Loomba, R.; Sanyal, A.J.; Mantzoros, C.S. Resmetirom, the first approved drug for the management of metabolic dysfunction-associated steatohepatitis: Trials, opportunities, and challenges. Metabolism: clinical and experimental 2024, 154, 155835. [CrossRef]
- Petta, S.; Kim, K.; Targher, G.; Romeo, S.; Sookoian, S.; Zheng, M.H.; Aghemo, A.; Valenti, L. Focus on Semaglutide 2.4 mg/week for the Treatment of Metabolic Dysfunction-Associated Steatohepatitis. Liver international : official journal of the International Association for the Study of the Liver 2025, 45, e70407. [CrossRef]
- Dobbie, L.J.; Burgess, J.; Hamid, A.; Nevitt, S.J.; Hydes, T.J.; Alam, U.; Cuthbertson, D.J. Effect of a Low-Calorie Dietary Intervention on Liver Health and Body Weight in Adults with Metabolic-Dysfunction Associated Steatotic Liver Disease (MASLD) and Overweight/Obesity: A Systematic Review and Meta-Analysis. Nutrients 2024, 16, 1030. [CrossRef]
- Pi, S.; Zhang, S.; Zhang, J.; Guo, Y.; Li, Y.; Deng, J.; Du, H. Low-carbohydrate diets reduce cardiovascular risk factor levels in patients with metabolic dysfunction-associated steatotic liver disease: a systematic review and meta-analysis of randomized controlled trials. Front Nutr 2025, 12, 1626352. [CrossRef]
- Lee, Y.C.; Lee, H.S.; Jeon, S.; Lee, Y.J.; Kwon, Y.J.; Lee, J.W. Assessing Nutritional Factors for Metabolic Dysfunction-Associated Steatotic Liver Disease via Diverse Statistical Tools. Diabetes Metab J 2025, 50, 178–189. [CrossRef]
- Hartmann, P.; Mouzaki, M.; Hassan, S.; Kehar, M.; Mysore, K.R.; Mauney, E.; Nonga, D.; Karjoo, S.; Sood, S.; Tou, A., et al. Call to action-Pediatric MASLD requires immediate attention to curb health crisis. Hepatology 2025, 82, 1341–1351. [CrossRef]
- Malespin, M.H.; Barritt, A.S.t.; Watkins, S.E.; Schoen, C.; Tincopa, M.A.; Corbin, K.D.; Mospan, A.R.; Munoz, B.; Trinh, H.N.; Weiss, L.M., et al. Weight Loss and Weight Regain in Usual Clinical Practice: Results From the TARGET-NASH Observational Cohort. Clin Gastroenterol Hepatol 2022, 20, 2393–2395 e2394. [CrossRef]
- Funuyet-Salas, J.; Martin-Rodriguez, A.; Perez-San-Gregorio, M.A.; Romero-Gomez, M. Influence of Psychological Biomarkers on Therapeutic Adherence by Patients with Non-Alcoholic Fatty Liver Disease: A Moderated Mediation Model. J Clin Med 2021, 10, 2208. [CrossRef]
- Goff, H.D.; Hartel, R.W. Ice Cream, 7 ed.; Springer: New York, NY, 2013; pp. 462.
- Ming, D.; Hellekant, G. Brazzein, a new high-potency thermostable sweet protein from Pentadiplandra brazzeana B. FEBS Lett. 1994, 355, 106–108. [CrossRef]
- Kant, R. Sweet proteins--potential replacement for artificial low calorie sweeteners. Nutrition journal 2005, 4, 5. [CrossRef]
- Novik, T.S.; Koveshnikova, E.I.; Kotlobay, A.A.; Sycheva, L.P.; Kurochkina, K.G.; Averina, O.A.; Belopolskaya, M.V.; Sergiev, P.V.; Dontsova, O.A.; Lazarev, V.N., et al. Sweet-Tasting Natural Proteins Brazzein and Monellin: Safe Sugar Substitutes for the Food Industry. Foods 2023, 12, 4065. [CrossRef]
- FDA, U.S. GRN No.1142 Brazzein produced by Komagataella phaffii expressing a gene encoding for brazzein from Pentadiplandra brazzeana. 2024.
- FDA, U.S. GRN No.1207 Brazzein preparation produced by Aspergillus oryzae 90402 expressing a gene encoding for brazzein from Pentadiplandra brazzeana. 2025.
- Jiang, P.; Ji, Q.; Liu, Z.; Snyder, L.A.; Benard, L.M.; Margolskee, R.F.; Max, M. The cysteine-rich region of T1R3 determines responses to intensely sweet proteins. The Journal of biological chemistry 2004, 279, 45068–45075. [CrossRef]
- Ghanavatian, P.; Khalifeh, K.; Jafarian, V. Structural features and activity of Brazzein and its mutants upon substitution of a surfaced exposed alanine. Biochimie 2016, 131, 20–28. [CrossRef]
- Zeece, M. Flavors. In Introduction to the Chemistry of Food, Elsevier: 2020; 10.1016/b978-0-12-809434-1.00006-2pp. 213–250.
- Izawa, K.; Amino, Y.; Kohmura, M.; Ueda, Y.; Kuroda, M. 4.16 - Human–Environment Interactions – Taste. In Comprehensive Natural Products II, Liu, H.-W., Mander, L., Eds. Elsevier: Oxford, 2010; pp. 631–671. [CrossRef]
- Rajan, V.; Howard, J.A. Brazzein: A Natural Sweetener. In Sweeteners: Pharmacology, Biotechnology, and Applications, Mérillon, J.-M., Ramawat, K.G., Eds. Springer International Publishing: Cham, 2018; 10.1007/978-3-319-27027-2_3pp. 17–33.
- Kim, H.; Kang, J.; Hong, S.; Jo, S.; Noh, H.; Kang, B.H.; Park, S.; Seo, Y.J.; Kong, K.H.; Hong, S. 3M-Brazzein as a Natural Sugar Substitute Attenuates Obesity, Metabolic Disorder, and Inflammation. Journal of agricultural and food chemistry 2020, 68, 2183–2192. [CrossRef]
- Lynch, B.; Wang, T.; Vo, T.; Tafazoli, S.; Ryder, J. Safety evaluation of oubli fruit sweet protein (brazzein) derived from Komagataella phaffii, intended for use as a sweetener in food and beverages. Toxicology Research and Application 2023, 7, 23978473231151258. [CrossRef]
- Veselovsky, V.A.; Boldyreva, D.I.; Olekhnovich, E.I.; Klimina, K.M.; Babenko, V.V.; Zakharevich, N.V.; Larin, A.K.; Morozov, M.D.; Zoruk, P.Y.; Sergiev, P.V., et al. Effect of the consumption of brazzein and monellin, two recombinant sweet-tasting proteins, on rat gut microbiota. Front Nutr 2024, 11, 1362529. [CrossRef]
- Schiatti-Siso, I.P.; Quintana, S.E.; Garcia-Zapateiro, L.A. Stevia (Stevia rebaudiana) as a common sugar substitute and its application in food matrices: an updated review. J Food Sci Technol 2023, 60, 1483–1492. [CrossRef]
- Abril, J.R.; Stull, J.W.; Taylor, R.R.; Angus, R.C.; Daniel, T.C. Characteristics of Frozen Desserts Sweetened with Xylitol and Fructose. Journal of Food Science 1982, 47, 472–475. [CrossRef]
- Kalicka, D.; Znamirowska, A.; Pawlos, M.; Buniowska, M.; Szajnar, K. Physical and sensory characteristics and probiotic survival in ice cream sweetened with various polyols. International Journal of Dairy Technology 2019, 72, 456–465. [CrossRef]
- Lacerda, R.C.C.; Andersen, G.H.; Junge, J.Y.; Kidmose, U.; Bolini, H.M.A. Perception and acceptance of natural sweeteners in a plant-based cocoa-flavored ice cream: Difference between Danish and Brazilian consumers. Journal of Sensory Studies 2024, 39, e12890. [CrossRef]
- Karlas, T.; Petroff, D.; Sasso, M.; Fan, J.G.; Mi, Y.Q.; de Ledinghen, V.; Kumar, M.; Lupsor-Platon, M.; Han, K.H.; Cardoso, A.C., et al. Individual patient data meta-analysis of controlled attenuation parameter (CAP) technology for assessing steatosis. J Hepatol 2017, 66, 1022–1030. [CrossRef]
- Tsochatzis, E.A.; Gurusamy, K.S.; Ntaoula, S.; Cholongitas, E.; Davidson, B.R.; Burroughs, A.K. Elastography for the diagnosis of severity of fibrosis in chronic liver disease: a meta-analysis of diagnostic accuracy. J Hepatol 2011, 54, 650–659. [CrossRef]
- de Medeiros, A.C.; Filho, E.R.T.; Bolini, H.M.A. Impact of Natural and Artificial Sweeteners Compounds in the Sensory Profile and Preference Drivers Applied to Traditional, Lactose-Free, and Vegan Frozen Desserts of Chocolate Flavor. Journal of Food Science 2019, 84, 2973–2982. [CrossRef]
- Long, M.; Wei, Y.; Tao, S.; Wu, Y.; Wang, J.; Zhou, D.; Cai, H.; Zhan, G. Ice cream with sucralose, stevioside, and erythritol as sugar substitutes: Sensory profile and customer preference. Food Science and Technology International 2023, 30, 273–281. [CrossRef]
- Muenprasitivej, N.; Tao, R.; Nardone, S.J.; Cho, S. The Effect of Steviol Glycosides on Sensory Properties and Acceptability of Ice Cream. Foods 2022, 11, 1745. [CrossRef]
- Moriano, M.E.; Alamprese, C. Honey, trehalose and erythritol as sucrose-alternative sweeteners for artisanal ice cream. A pilot study. LWT 2017, 75, 329–334. [CrossRef]
- Peres, J.; Esmerino, E.; da Silva, A.L.; Racowski, I.; Bolini, H. Sensory Profile, Drivers of Liking, and Influence of Information on the Acceptance of Low-Calorie Synbiotic and Probiotic Chocolate Ice Cream. Journal of Food Science 2018, 83, 1350–1359. [CrossRef]
- Palazzo, A.B.; Bolini, H.M.A. Sweeteners in diet chocolate ice cream: Penalty analysis and acceptance evaluation. J. Food Stud. 2017, 6, 1. [CrossRef]
- Whelan, A.P.; Vega, C.; Kerry, J.P.; Goff, H.D. Physicochemical and sensory optimisation of a low glycemic index ice cream formulation. International Journal of Food Science & Technology 2008, 43, 1520–1527. [CrossRef]
- Joseph, J.A.; Akkermans, S.; Nimmegeers, P.; Van Impe, J.F.M. Bioproduction of the Recombinant Sweet Protein Thaumatin: Current State of the Art and Perspectives. Front Microbiol 2019, 10, 695. [CrossRef]
- Saraiva, A.; Carrascosa, C.; Ramos, F.; Raheem, D.; Pedreiro, S.; Vega, A.; Raposo, A. Brazzein and Monellin: Chemical Analysis, Food Industry Applications, Safety and Quality Control, Nutritional Profile and Health Impacts. Foods 2023, 12, 1943. [CrossRef]
- Pekarska, K.; Brennan, P.N.; Ivancovsky Wajcman, D.; Towey, J.; Cox, L.; Weatherby, K.; McPherson, S.; Stine, J.; Willemse, J.; Li, W., et al. Nutrition in MASLD: a patient focused, evidence-based clinician’s guide. Frontline Gastroenterology 2025, 10.1136/flgastro-2025-103183, flgastro–2025–103183. [CrossRef]
- Simancas-Racines, D.; Annunziata, G.; Verde, L.; Fasci-Spurio, F.; Reytor-Gonzalez, C.; Muscogiuri, G.; Frias-Toral, E.; Barrea, L. Nutritional Strategies for Battling Obesity-Linked Liver Disease: the Role of Medical Nutritional Therapy in Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) Management. Curr Obes Rep 2025, 14, 7. [CrossRef]
- London, A.; Richter, M.M.; Sjoberg, K.A.; Wewer Albrechtsen, N.J.; Povazan, M.; Drici, L.; Schaufuss, A.; Madsen, L.; Oyen, J.; Madsbad, S., et al. The impact of short-term eucaloric low- and high-carbohydrate diets on liver triacylglycerol content in males with overweight and obesity: a randomized crossover study. The American journal of clinical nutrition 2024, 120, 283–293. [CrossRef]
- Rinaldi, R.; De Nucci, S.; Donghia, R.; Donvito, R.; Cerabino, N.; Di Chito, M.; Penza, A.; Mongelli, F.P.; Shahini, E.; Zappimbulso, M., et al. Gender Differences in Liver Steatosis and Fibrosis in Overweight and Obese Patients with Metabolic Dysfunction-Associated Steatotic Liver Disease before and after 8 Weeks of Very Low-Calorie Ketogenic Diet. Nutrients 2024, 16, 1408. [CrossRef]
- Hutchings, S.C.; Low, J.Y.Q.; Keast, R.S.J. Sugar reduction without compromising sensory perception. An impossible dream? Critical reviews in food science and nutrition 2019, 59, 2287–2307. [CrossRef]
- Markey, O.; Lovegrove, J.A.; Methven, L. Sensory profiles and consumer acceptability of a range of sugar-reduced products on the UK market. Food Research International 2015, 72, 133–139. [CrossRef]
- Musialik, J.; Suchecka, W.; Klimacka-Nawrot, E.; Petelenz, M.; Hartman, M.; Blonska-Fajfrowska, B. Taste and appetite disorders of chronic hepatitis C patients. Eur J Gastroenterol Hepatol 2012, 24, 1400–1405. [CrossRef]
- Deems, R.O.; Friedman, M.I.; Friedman, L.S.; Munoz, S.J.; Maddrey, W.C. Chemosensory function, food preferences and appetite in human liver disease. Appetite 1993, 20, 209–216. [CrossRef]
- Dallio, M.; Romeo, M.; Di Nardo, F.; Napolitano, C.; Vaia, P.; Iadanza, G.; Olivieri, S.; Coppola, A.; Niosi, M.; Federico, A. Dysgeusia in MASLD-related advanced chronic liver disease (ACLD): a silent driver towards the "Bermuda" triangle of malnutrition-sarcopenia-frailty severely affecting prognosis. Nutrition journal 2025, 24, 10. [CrossRef]
- Juárez-Hernández, E.; López-Méndez, I.; Uribe, M.; Chávez-Tapia, N.; Meneses-Mayo, M. Zinc supplementation in patients with cirrhosis and dysgeusia: Randomized Clinical Trial. Proceedings of Scientific Research Universidad Anáhuac. Multidisciplinary Journal of Healthcare 2024, 4, 13–22. [CrossRef]
- Waimaleongora-ek, P.; Prinyawiwatkul, W. Comparison of discriminability of common food acceptance scales for the elderly. International Journal of Food Science and Technology 2020, 56, 148–157. [CrossRef]
- Addo-Preko, E.; Amissah, J.G.N.; Adjei, M.Y.B. The relevance of the number of categories in the hedonic scale to the Ghanaian consumer in acceptance testing. Frontiers in Food Science and Technology 2023, 3, 1071216. [CrossRef]
- Ennis, J.M.; Rousseau, B.; Ennis, D.M. Sensory Difference Tests as Measurement Instruments: a Review of Recent Advances. Journal of Sensory Studies 2014, 29, 89–102. [CrossRef]
- Meyners, M.; Carr, B.T.; Kunert, J. Equivalence and non-inferiority tests using replicated discrimination and preference data. Journal of Sensory Studies 2023, 38, e12882. [CrossRef]
- Bi, J. Similarity (equivalence) testing. In Sensory Discrimination Tests and Measurements, John Wiley&Sons: 2015. pp. 98–138. [CrossRef]
- Meyners, M. Least equivalent allowable differences in equivalence testing. Food Quality and Preference 2007, 18, 541–547. [CrossRef]
- Wichchukit, S.; O'Mahony, M. The 9-point hedonic scale and hedonic ranking in food science: some reappraisals and alternatives. J Sci Food Agric 2015, 95, 2167–2178. [CrossRef]
| Characteristic | Overall (n = 103) |
Sequence A (n = 45) | Sequence B (n = 58) | p-value* |
|---|---|---|---|---|
| Age, years (mean ± SD) | 54.7 ± 9.7 | 54.6 ± 10.7 | 54.8 ± 9.0 | 0.918 |
| Sex, n (%) Male Female |
24 (23.3%) 79 (76.7%) |
11 (24.4%) 34 (75.6%) |
13 (22.4%) 45 (77.6%) |
0.995 |
| BMI, kg/m2 (mean ± SD) | 38.5 ± 6.2 | 38.8 ± 5.4 | 38.3 ± 6.7 | 0.678 |
| BMI category, n (%) | 0.582 | |||
| Overweight (25-29.9) Obese Class I (30-34.9) Obese Class II+ (≥35) |
2 (1.9%) 31 (30.1%) 69 (67.0%) |
0 (0.0%) 13 (28.9%) 32 (71.1%) |
2 (3.4%) 18 (31.0%) 37 (63.8%) |
|
| Diabetes, n (%) | 45 (43.7%) | 16 (35.6%) | 29 (50.0%) | 0.206 |
| Liver steatosis S3 n (%) | 83 (80.6%) | 36 (80.0%) | 47 (81.0%) | 1.000 |
| Liver fibrosis F3/F4 n (%) | 14 (13.6%) | 4 (8.9%) | 10 (17.2%) | 0.349 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




