Submitted:
11 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Public Transcriptomic Datasets
2.2. Data Retrieval and Preprocessing
2.3. Processing of Single-Cell RNA-seq Data
2.4. Quality Control Filtering
2.5. Single-Cell RNA-seq Analysis
2.6. Leydig Cell RNA-seq Analysis
2.7. Statistical Analysis
2.8. Data Visualization
2.9. Software Environment
3. Results
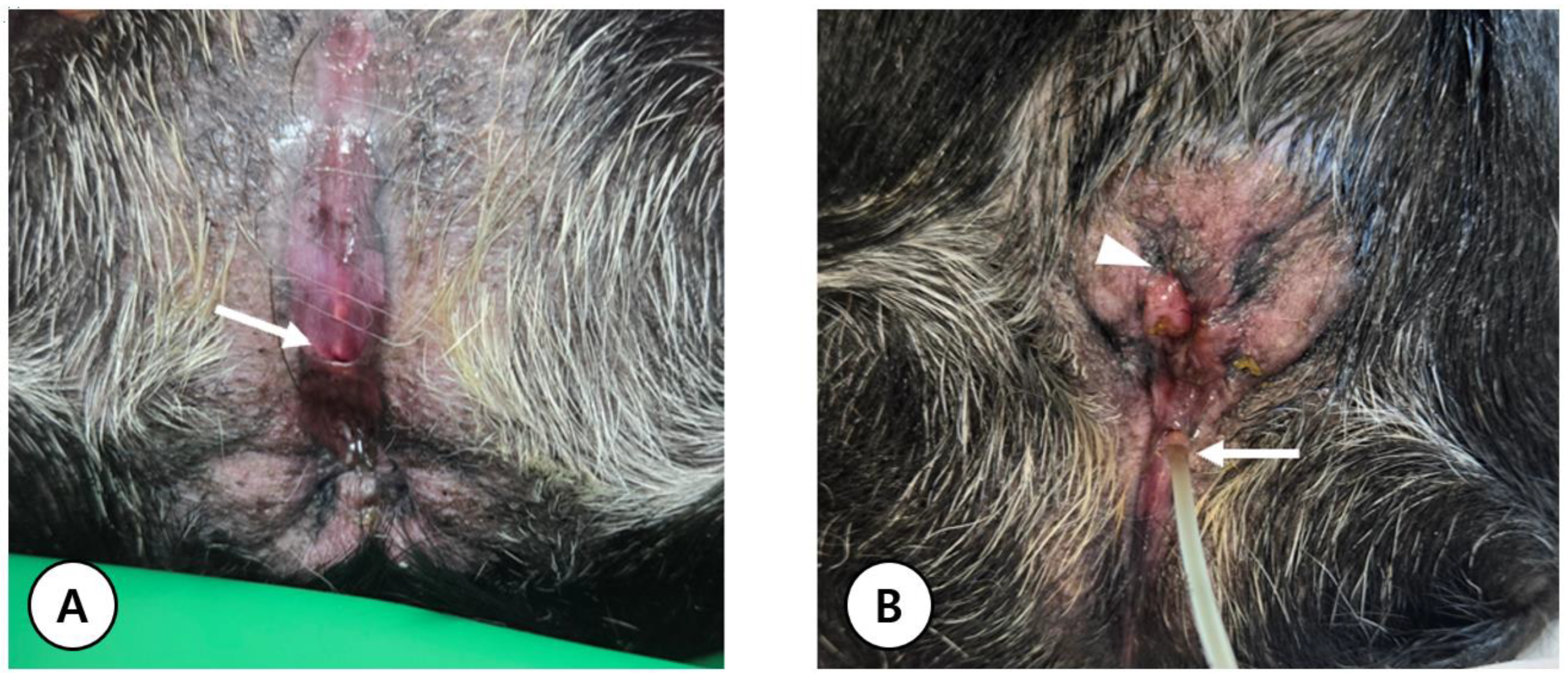
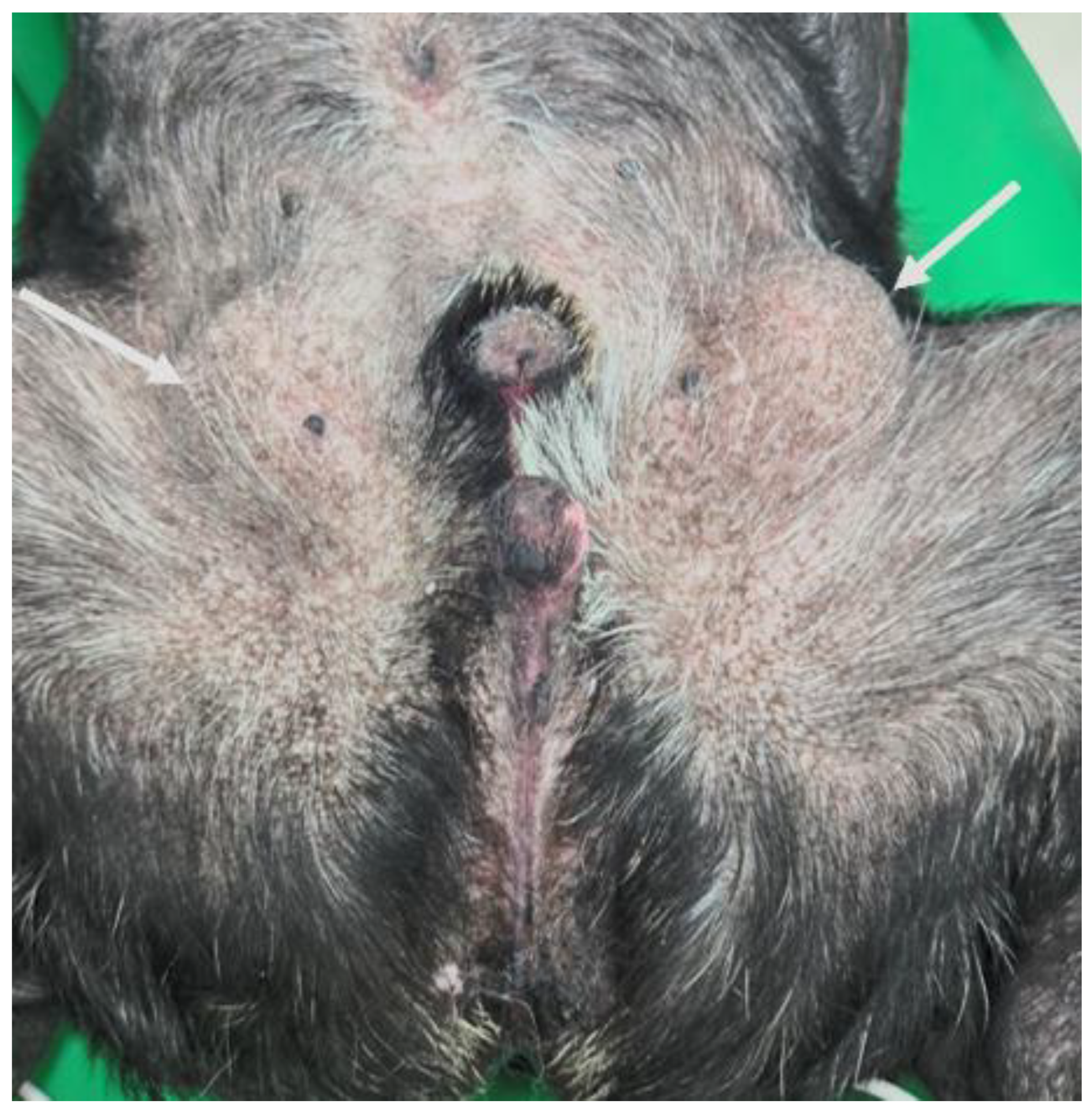
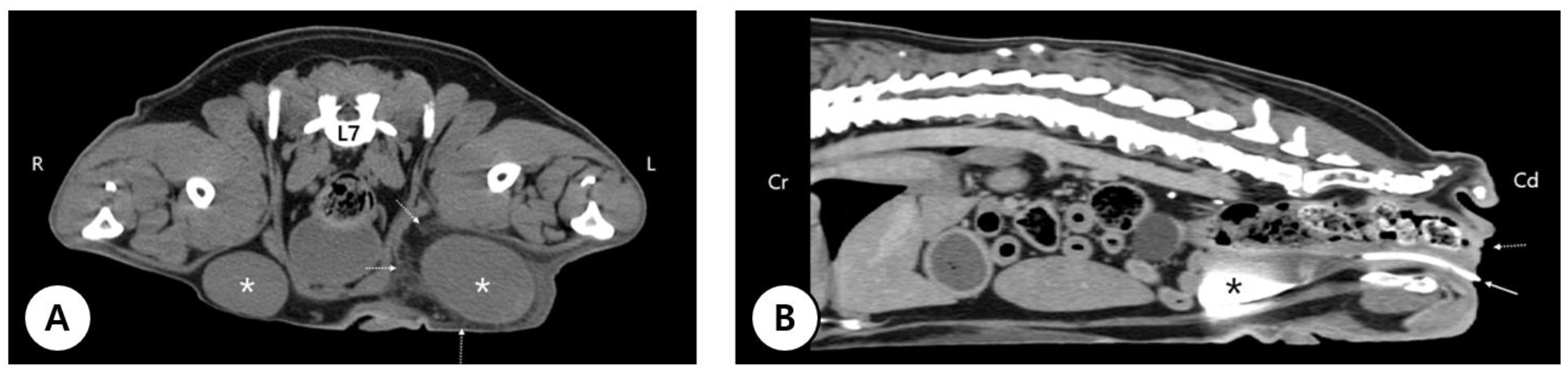
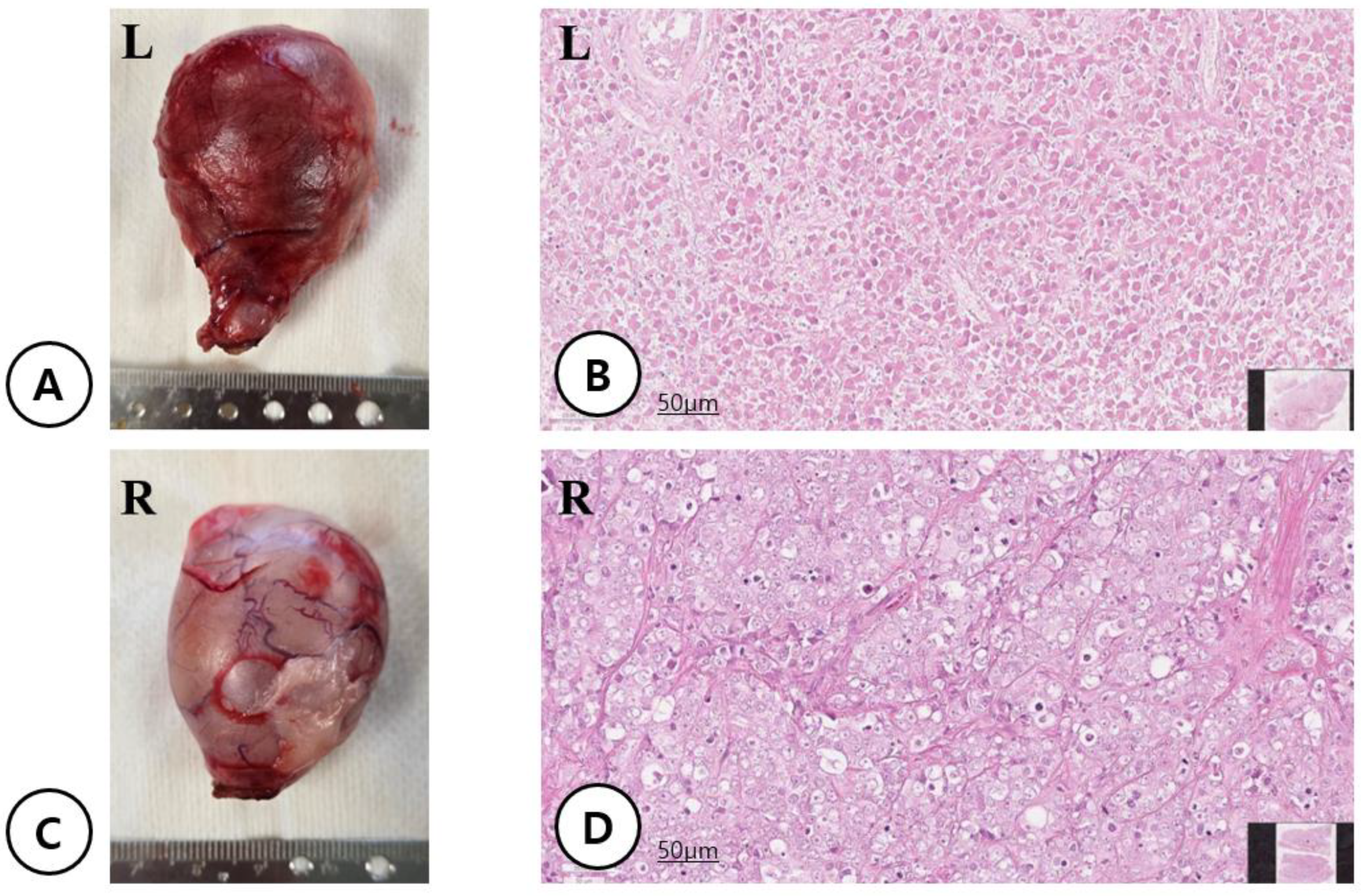
3.1. Clinical Presentation and Long-Term Conservative Management of Severe Perineal Hypospadias
3.2. Contextual Reference to Developmental Pathways in Public Mouse Genital Tubercle Transcriptomic Data
3.3. Contextual Reference to Steroidogenic Pathways in a Public Mouse Leydig Cell Dataset
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mouriquand, P., D.B. Gorduza, and P.-Y. Mure, Hypospadias, in Pediatric surgery: Pediatric urology. 2023, Springer. p. 527–553.
- Cunha, G.R.; Sinclair, A.; Risbridger, G.; Hutson, J.; Baskin, L.S. Current understanding of hypospadias: relevance of animal models. Nat. Rev. Urol. 2015, 12, 271–280. [CrossRef]
- Lavoie, C.; Chun, B.; Au, M.; Do, C.; Sparks, S.S.; Chang, A.Y. Prevalence of disorders of sex development in patients with hypospadias and cryptorchidism. J. Pediatr. Urol. 2024, 21, 71–77. [CrossRef]
- Jurka, P.; Galanty, M.; Zielinska, P.; Max, A.; Sysa, P. Hypospadias in six dogs. Veter- Rec. 2009, 164, 331–333. [CrossRef]
- Switonski, M.; Dzimira, S.; Aleksiewicz, R.; Szczerbal, I.; Nowacka-Woszuk, J.; Krzeminska, P.; Deska, T.; Nizanski, W. Hypospadias Is Not Rare in Dogs: Five New Cases, a Retrospective Study, and a Review of the Literature. Sex. Dev. 2018, 12, 244–250. [CrossRef]
- Cassata, R.; Iannuzzi, A.; Parma, P.; De Lorenzi, L.; Peretti, V.; Perucatti, A.; Iannuzzi, L.; Di Meo, G. Clinical, cytogenetic and molecular evaluation in a dog with bilateral cryptorchidism and hypospadias. Cytogenet. Genome Res. 2008, 120, 140–143. [CrossRef]
- Galanty, M.; Jurka, P.; Zielińska, P. Surgical treatment of hypospadias. Techniques and results in six dogs. Polish Journal of Veterinary Sciences 2008, 11, 235–43.
- Cashmore, R.G.; Ladlow, J.F. Creation of a Urethral Conduit from a Preputial Indirect Flap in a Dog with Perineal Hypospadias. Veter- Surg. 2010, 39, 14–20. [CrossRef]
- Perriton, C.L.; Powles, N.; Chiang, C.; Maconochie, M.K.; Cohn, M.J. Sonic hedgehog Signaling from the Urethral Epithelium Controls External Genital Development. Dev. Biol. 2002, 247, 26–46. [CrossRef]
- Zhang, L.-F.; Qin, C.; Wei, Y.-F.; Wang, Y.; Chang, J.-K.; Mi, Y.-Y.; Ma, L.; Jiang, J.-T.; Feng, N.-H.; Wang, Z.-J.; et al. Differential expression of the Wnt/β-catenin pathway in the genital tubercle (GT) of fetal male rat following maternal exposure to di-n-butyl phthalate (DBP). Syst. Biol. Reprod. Med. 2011, 57, 244–250. [CrossRef]
- Haraguchi, R.; Suzuki, K.; Murakami, R.; Sakai, M.; Kamikawa, M.; Kengaku, M.; Sekine, K.; Kawano, H.; Kato, S.; Ueno, N.; et al. Molecular analysis of external genitalia formation: the role of fibroblast growth factor (Fgf) genes during genital tubercle formation. Development 2000, 127, 2471–2479. [CrossRef]
- Kurzrock, E.A.; Baskin, L.S.; Li, Y.; Cunha, G.R. Epithelial-Mesenchymal Interactions in Development of the Mouse Fetal Genital Tubercle. Cells Tissues Organs 1999, 164, 125–130. [CrossRef]
- Carmichael, S.L.; Ma, C.; Choudhry, S.; Lammer, E.J.; Witte, J.S.; Shaw, G.M. Hypospadias and Genes Related to Genital Tubercle and Early Urethral Development. J. Urol. 2013, 190, 1884–1892. [CrossRef]
- Ivell, R.; Mamsen, L.S.; Andersen, C.Y.; Anand-Ivell, R. Expression and Role of INSL3 in the Fetal Testis. Front. Endocrinol. 2022, 13, 868313. [CrossRef]
- Martin, L.J. Cell interactions and genetic regulation that contribute to testicular Leydig cell development and differentiation. Mol. Reprod. Dev. 2016, 83, 470–487. [CrossRef]
- Ivell, R.; Heng, K.; Anand-Ivell, R. Insulin-Like Factor 3 and the HPG Axis in the Male. Front. Endocrinol. 2014, 5, 6. [CrossRef]
- Fénichel, P.; Chevalier, N.; Lahlou, N.; Coquillard, P.; Wagner-Mahler, K.; Pugeat, M.; Panaïa-Ferrari, P.; Brucker-Davis, F. Endocrine Disrupting Chemicals Interfere With Leydig Cell Hormone Pathways During Testicular Descent in Idiopathic Cryptorchidism. Front. Endocrinol. 2019, 9, 786. [CrossRef]
- Ciarloni, A.; Muti, N.D.; Sacco, S.; Ambo, N.; Di Giacomi, V.; Perrone, M.; Rossi, S.; Balercia, M.; Salvio, G.; Balercia, G. Genetic, Epigenetic, and Non-Genetic Factors in Testicular Dysgenesis Syndrome: A Narrative Review. Genes 2025, 17, 40. [CrossRef]
- McInnes, L., J. Healy, and J. Melville, Umap: Uniform manifold approximation and projection for dimension reduction. arXiv preprint arXiv:1802.03426, 2018.
- E Adelsberger, M.; Smeak, D.D. Repair of extensive perineal hypospadias in a Boston terrier using tubularized incised plate urethroplasty. The Canadian Veterinary Journal 2009, 50, 937–42.
- De Win, G., et al., Long-term outcomes of pediatric hypospadias and surgical intervention. Pediatric Health, Medicine and Therapeutics, 2012: p. 69–77.
- Lin, C.; Yin, Y.; Long, F.; Ma, L. Tissue-specific requirements of β-catenin in external genitalia development. Development 2008, 135, 2815–2825. [CrossRef]
- Amato, C.M.; Yao, H.H.-C.; Zhao, F. One Tool for Many Jobs: Divergent and Conserved Actions of Androgen Signaling in Male Internal Reproductive Tract and External Genitalia. Front. Endocrinol. 2022, 13, 910964. [CrossRef]
- E Virtanen, H.; Cortes, D.; Meyts, E.R.; Ritzén, E.M.; Nordenskjöld, A.; E Skakkebaek, N.; Toppari, J. Development and descent of the testis in relation to cryptorchidism. Acta Paediatr. 2007, 96, 622–627. [CrossRef]
- Soto-Heras, S.; Reinacher, L.; Wang, B.; Oh, J.E.; Bunnell, M.; Park, C.J.; A Hess, R.; Ko, C.J. Cryptorchidism and testicular cancer in the dog: unresolved questions and challenges in translating insights from human studies. Biol. Reprod. 2024, 111, 269–291. [CrossRef]
- Chen, Z.; Lei, Y.; Finnell, R.H.; Ding, Y.; Su, Z.; Wang, Y.; Xie, H.; Chen, F. Whole-exome sequencing study of hypospadias. iScience 2023, 26, 106663. [CrossRef]
- Gurney, J.K., et al., Risk factors for cryptorchidism. Nature Reviews Urology, 2017. 14(9): p. 534–548.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



