Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Simulation Details
3. Results and Discussion
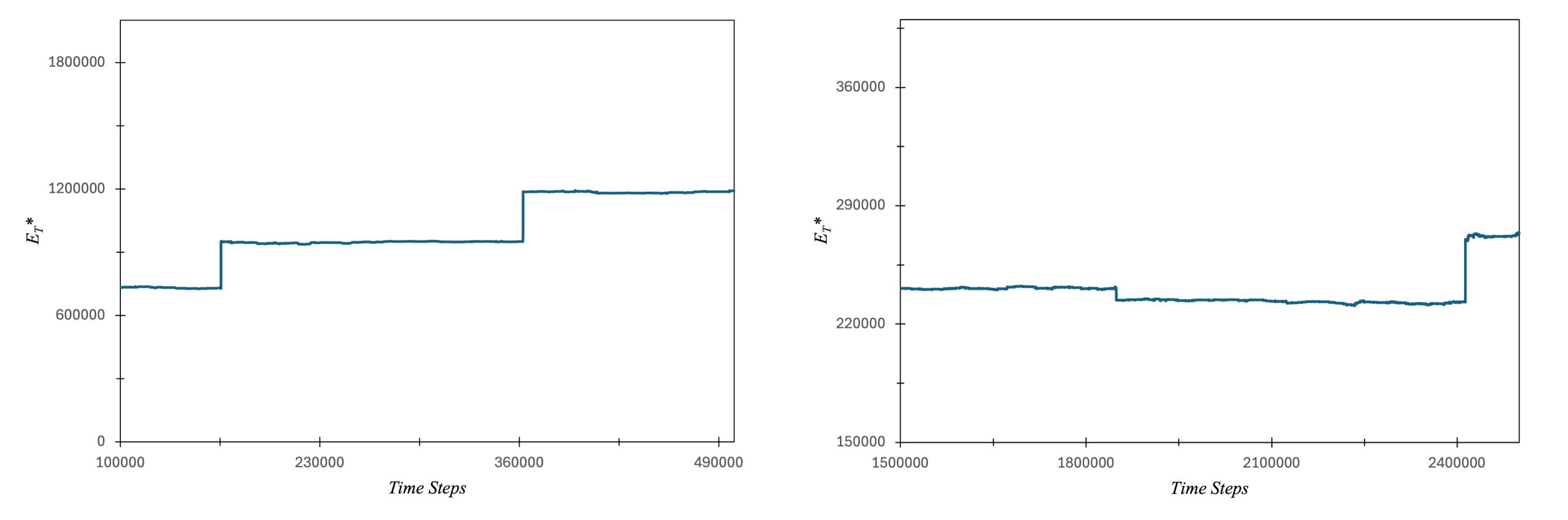
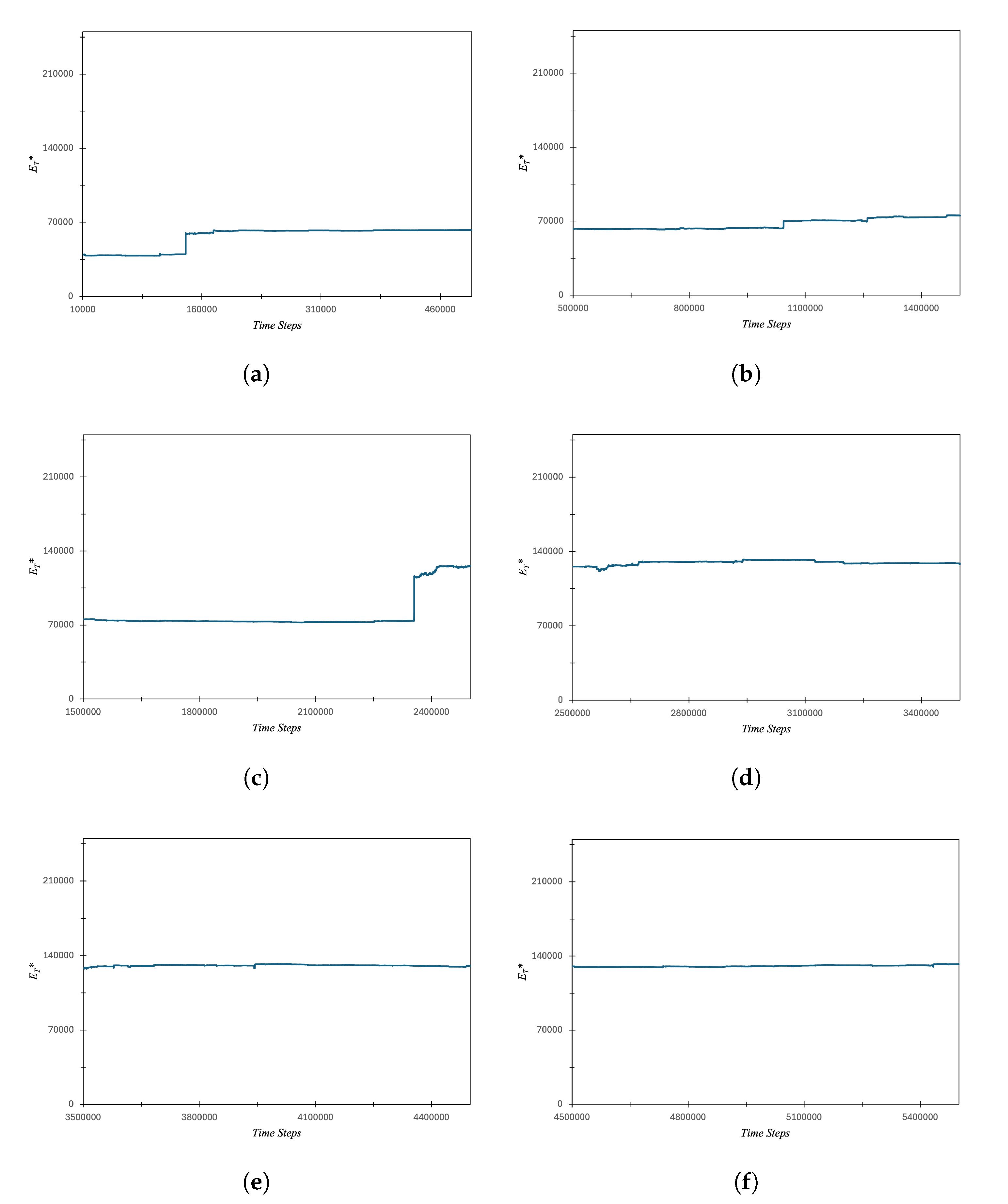
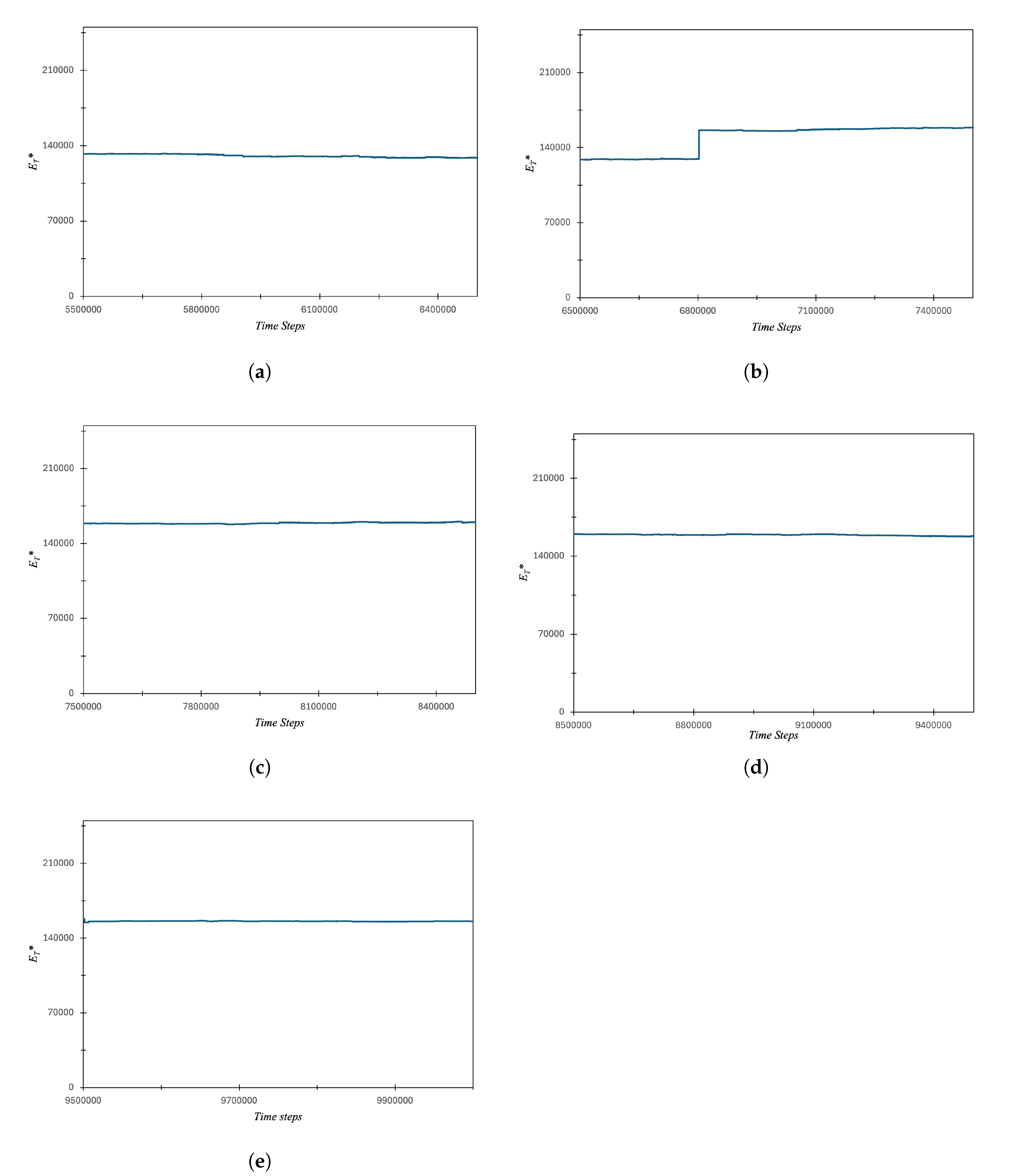
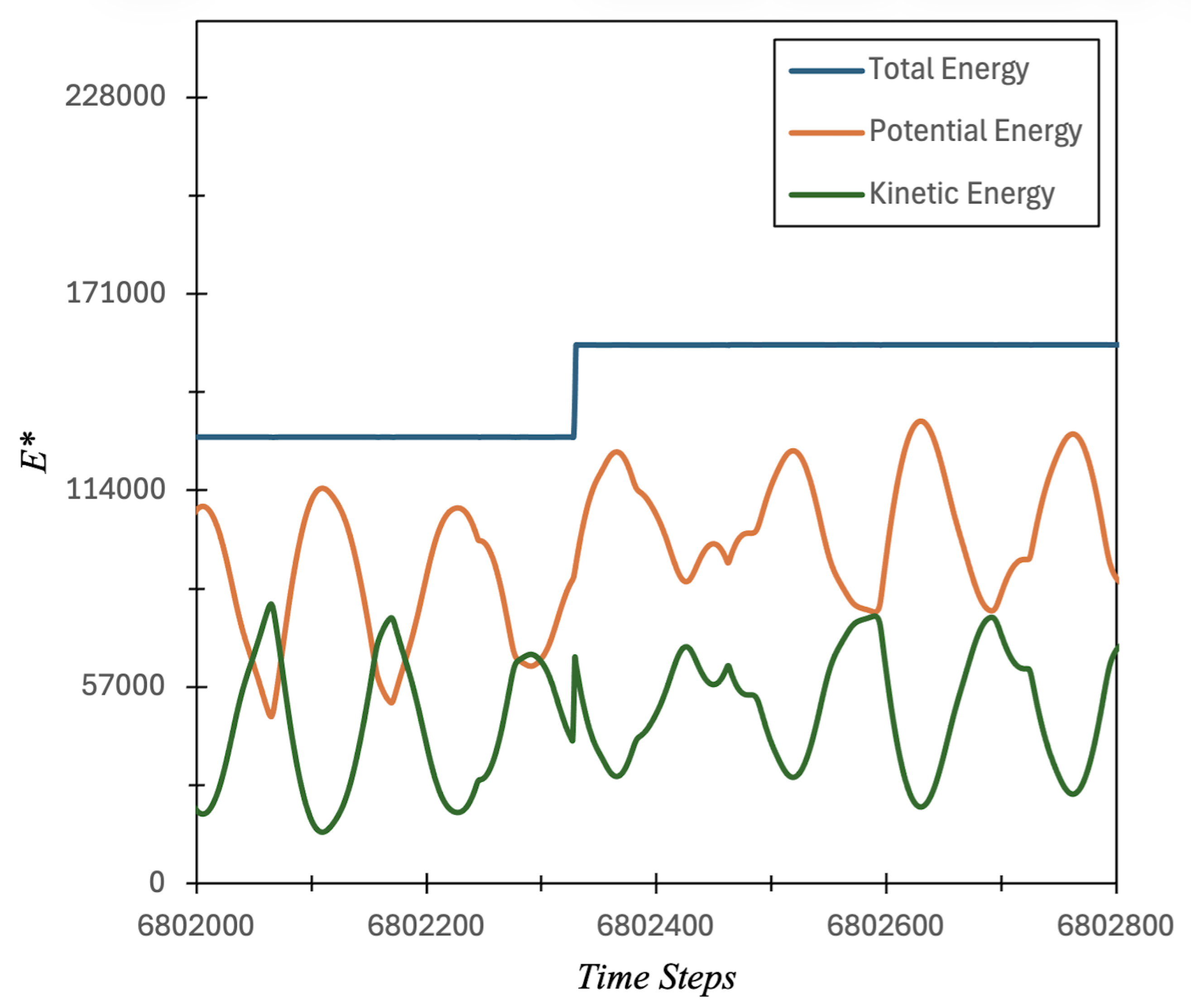
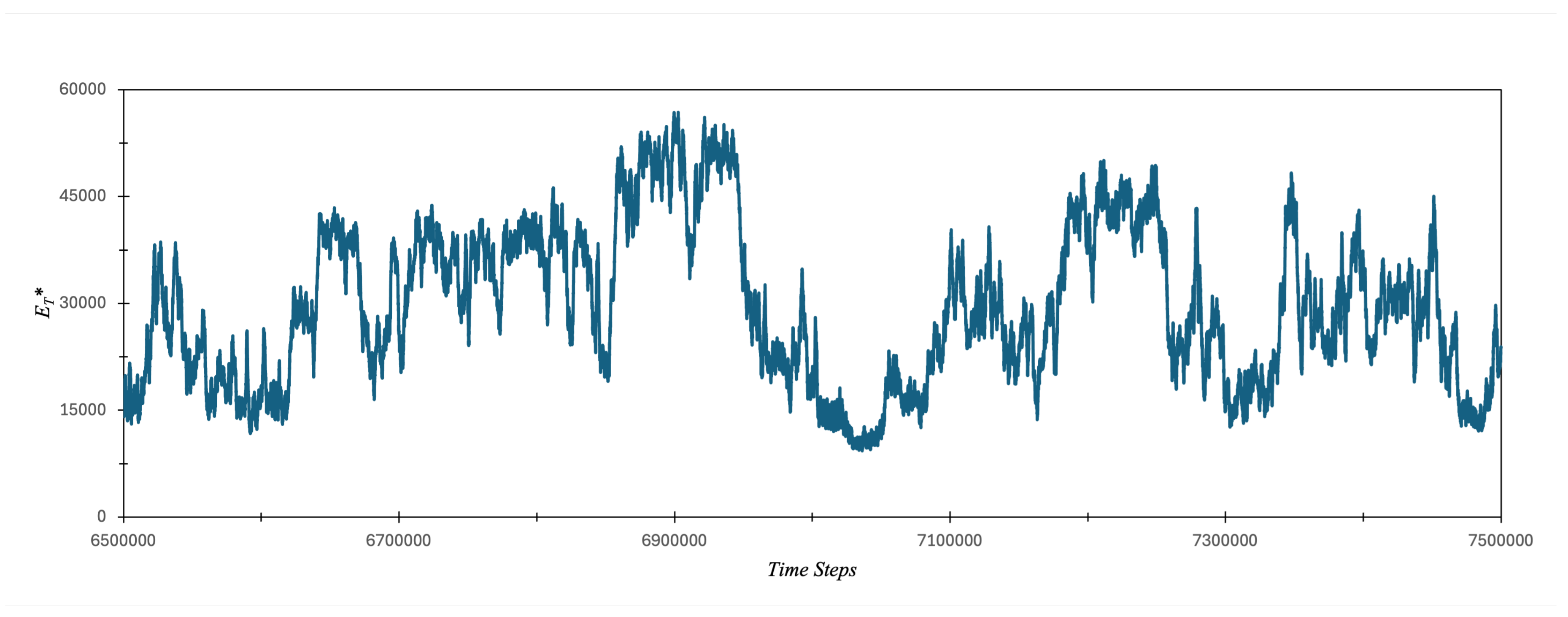
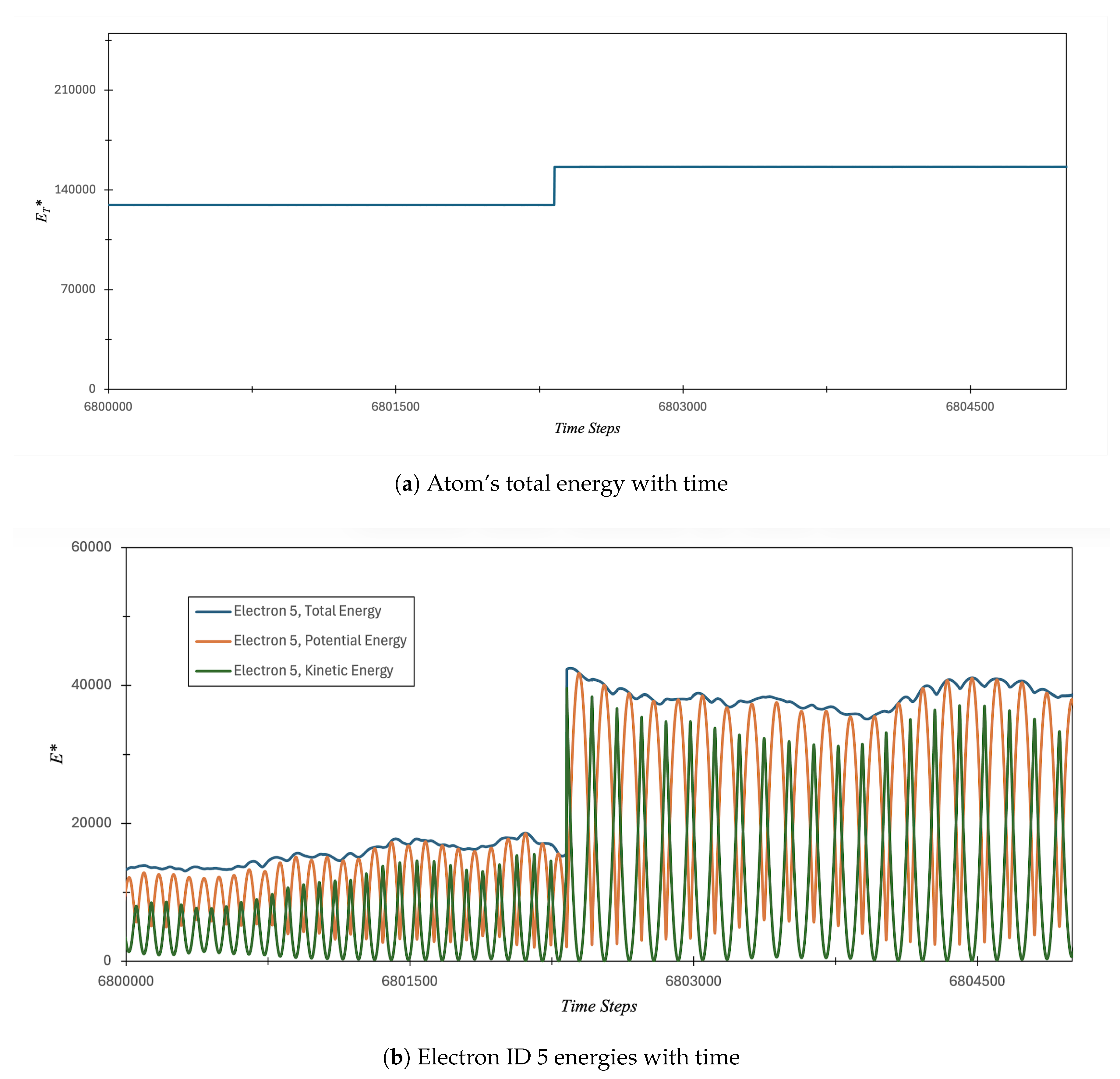
3.1. Carbon Atom Energy Analysis
3.1.1. Study of Electrons Energy Patterns during Atom’s Energy Quantization Shift
3.2. Quantum Effects of Fluorine, Magnesium and Chlorine Atom in Subatomic Particle Simulations
4. Conclusions
Acknowledgments
References
- Wollaston, W.H. XII. A Method of Examining the Refractive and Dispersive Powers of Different Substances, by Prismatic Reflection. Philosophical Transactions of the Royal Society of London 1802, 92, 365–380. [CrossRef]
- Fraunhofer, J.v. Bestimmung des Brechungs- und Farbenzerstreuungs-Vermögens verschiedener Glasarten, in Bezug auf die Vervollkommnung achromatischer Fernröhre. Denkschriften der Königlichen Akademie der Wissenschaften zu München 1817, 5, 193–226.
- Kirchhoff, G. Über die Fraunhofer’schen Linien. Annalen der Physik 1860, 185, 148–150.
- Kirchhoff, G.; Bunsen, R. Chemische Analyse durch Spectralbeobachtungen. Annalen der Physik und Chemie 1860, 110, 161–189. [CrossRef]
- Ångström, A.J. Optiska Undersökningar. Kongl. Svenska Vetenskaps-Akademiens Handlingar 1853.
- Thomsen, V. A Timeline of Atomic Spectroscopy. Spectroscopy 2006, 21, 32–38.
- Bohr, N. I. On the Constitution of Atoms and Molecules. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 1913, 26, 1–25. [CrossRef]
- Heisenberg, W. Über den anschaulichen Inhalt der quantentheoretischen Kinematik und Mechanik. Zeitschrift für Physik 1927, 43, 172–198. [CrossRef]
- Schödinger, E. Quantisierung als eigenwertproblem.(erste mitteilung). Annalen der Physik 1926, 79, 361–376.
- Heisenberg, W.; Pauli, W. Zur Quantentheorie der Wellenfelder. II. Zeitschrift für Physik 1930, 59, 168–190. [CrossRef]
- Heisenberg, W. Mehrkörperproblem und Resonanz in der Quantenmechanik. Zeitschrift für Physik 1926, 38, 411–426. [CrossRef]
- Bloch, F. Über die Quantenmechanik der Elektronen in Kristallgittern. Zeitschrift für Physik 1928, 52, 555–600. [CrossRef]
- Deutsch, D. Quantum Theory, the Church–Turing Principle and the Universal Quantum Computer. Proceedings of the Royal Society of London. A. Mathematical and Physical Sciences 1985, 400, 97–117. [CrossRef]
- Bennett, C.H.; Brassard, G. Quantum Cryptography: Public Key Distribution and Coin Tossing. In Proceedings of the Proceedings of the IEEE International Conference on Computers, Systems and Signal Processing, Bangalore, India, December 1984; pp. 175–179.
- Alder, B.J.; Wainwright, T.E. Phase Transition for a Hard Sphere System. The Journal of Chemical Physics 1957, 27, 1208. [CrossRef]
- Alder, B.J.; Wainwright, T.E. Studies in Molecular Dynamics. I. General Method. The Journal of Chemical Physics 1959, 31, 459–466. [CrossRef]
- Rahman, A. Correlations in the Motion of Atoms in Liquid Argon. Physical Review 1964, 136, A405–A411. [CrossRef]
- Kramida, A.; Ralchenko, Y.; Reader, J.; Team, N. A. NIST Atomic Spectra Database (ver. 5.12). https://physics.nist.gov/asd,2024; National Institute of Standards and Technology, Gaithersburg, MD.
- Coulomb, C.A. Premier mémoire sur l’électricité et le magnétisme. Histoire de l’Academie royale des sciences 1785, 569.
- Kane, C.; Balents, L.; Fisher, M.P. Coulomb interactions and mesoscopic effects in carbon nanotubes. Physical review letters 1997, 79, 5086.
- Cea, T.; Guinea, F. Coulomb interaction, phonons, and superconductivity in twisted bilayer graphene. Proceedings of the National Academy of Sciences 2021, 118, e2107874118.
- Venkatesan, S. Subatomic Particle Simulations using Monte Carlo and Molecular Dynamics Algorithms to Simulate Stable Atom and Model Electronic Structures. Preprints 2025. [CrossRef]
- Subatomic Particle Simulations using Monte Carlo and Molecular Dynamics Algorithms to Simulate Stable Atom and Model Electronic Structures. Innovative Journal of Applied Science 2025, 2, 20. [CrossRef]
- Swope, W.C.; Andersen, H.C.; Berens, P.H.; Wilson, K.R. A computer simulation method for the calculation of equilibrium constants for the formation of physical clusters of molecules: Application to small water clusters. The Journal of chemical physics 1982, 76, 637–649.
- Humphrey, W.; Dalke, A.; Schulten, K. VMD – Visual Molecular Dynamics. Journal of Molecular Graphics 1996, 14, 33–38.

| (Joules) | (picometers) | |
|---|---|---|
| Parameterized based on Carbon atom | 7.837 * | 0.7777 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




