Submitted:
11 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
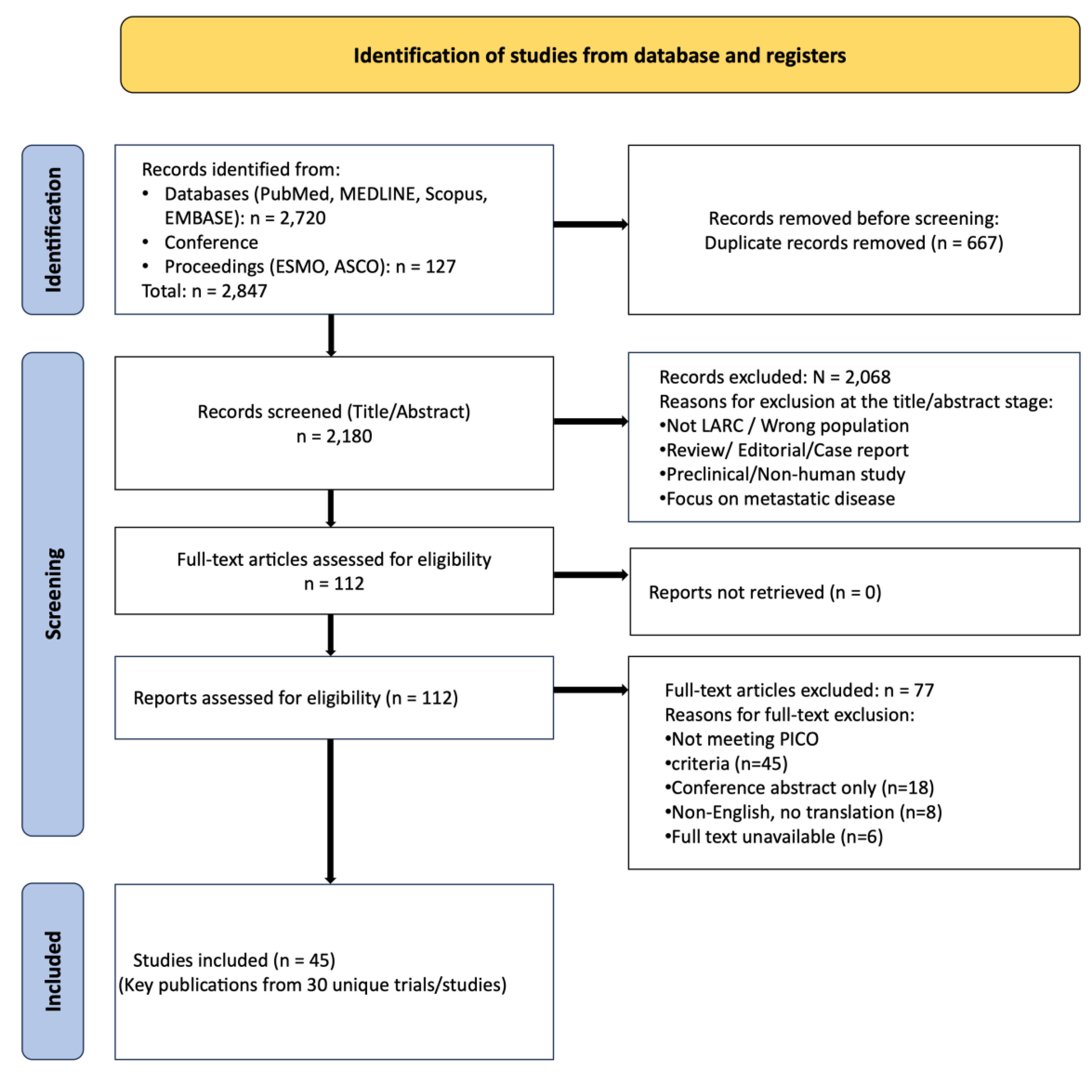
2.1. Search Strategy and Study Selection
2.2. Study Selection and Analytical Approach
2.3. Protocol Registration
2.4. Data Synthesis
3. Results
3.1. Study Selection and Characteristics
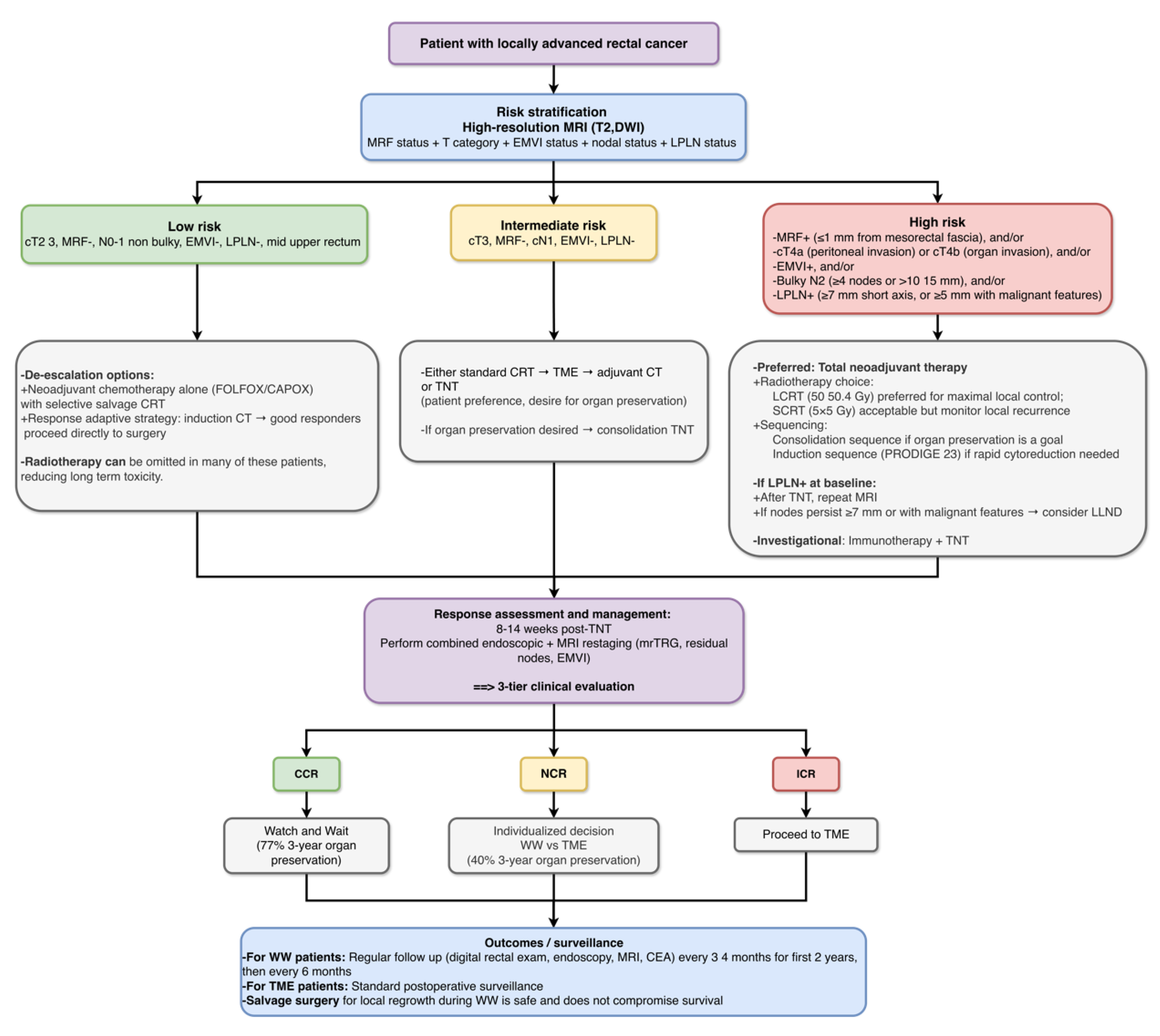
3.1.1. MRI-Based Risk Stratification: Defining Three Clinical Risk Groups
High-Risk Features (Indicate Need for TNT Intensification)
- •
- Threatened or involved mesorectal fascia (MRF+): Tumour or lymph node within ≤1 mm of the mesorectal fascia. Associated with local failure rates >20% after standard CRT alone.
- •
- cT4 category:
- -
- T4a: tumour invades the visceral peritoneum (peritoneal reflection). High risk of peritoneal dissemination.
- -
- T4b: tumour directly invades adjacent organs (bladder, prostate, seminal vesicles, sacrum, vagina, pelvic sidewall). Very high risk of involved margins and local relapse.
- •
- Extramural vascular invasion (EMVI+): Presence of tumour within veins outside the muscularis propria (serpentine appearance, expansion of vessel). Strongly associated with synchronous and metachronous distant metastases.
- •
- Bulky N2 disease: Multiple regional lymph nodes (≥4) or large nodes (>10-15 mm) in the mesorectum. Indicates high tumour burden and risk of residual disease after CRT.
- •
- Lateral pelvic lymph node involvement (LPLN+): Nodes in the internal iliac, obturator, or external iliac chains. Size ≥7 mm short axis, or ≥5 mm with malignant features (irregular border, mixed signal intensity, loss of fatty hilum). These nodes are not adequately treated by standard TME and may require lateral lymph node dissection (LLND) after TNT.
Intermediate-Risk Features (Standard CRT or TNT May Be Considered)
- cT3 with clear MRF (>1 mm)
- cN1 (1-3 small nodes)
- No EMVI
- No LPLN enlargement
Low-Risk Features (De-Escalation Possible)
- cT2-3, MRF-, N0-1 (non-bulky), EMVI-, LPLN-
- Mid-upper rectal location (≥5-10 cm from anal verge)
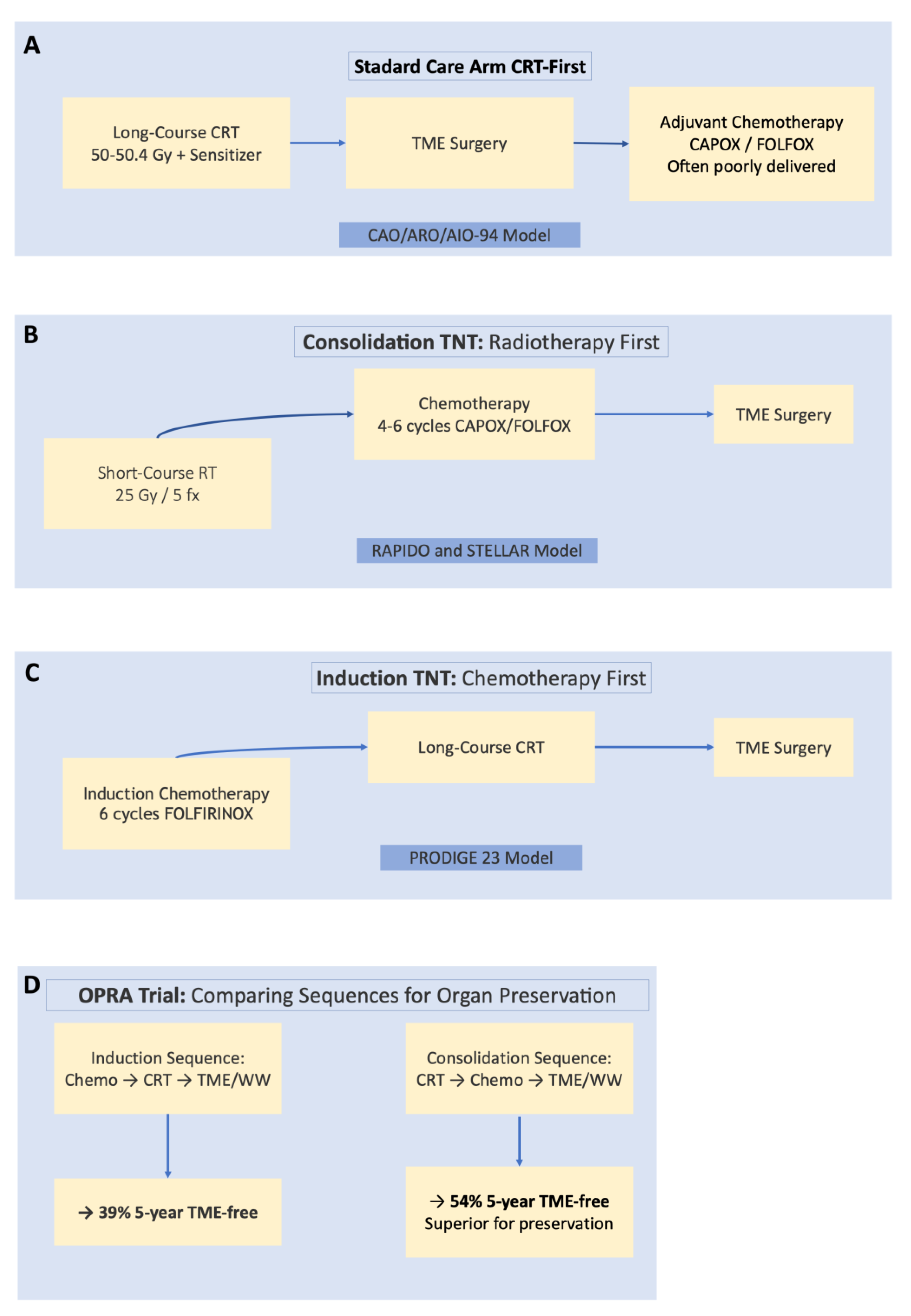
3.2. The Efficacy of Total Neoadjuvant Therapy: A Confirmed Benefit with a Critical Caveat
3.2.1. Baseline Heterogeneity Among TNT Trials: A Critical Comparison
- The absolute pCR rate (28% in RAPIDO vs 27.5% in PRODIGE 23) appears similar, but the RAPIDO population was much higher risk. Therefore, the relative benefit of TNT over standard CRT may be greater in higher-risk patients.
- The increased locoregional recurrence seen in RAPIDO (10% vs 6%) may be partly explained by the high-risk features (cT4, EMVI+), which are known to predispose to local failure, and by the use of SCRT instead of LCRT.
- De-escalation trials (PROSPECT, CONVERT, FOWARC) intentionally excluded many high-risk patients (e.g., cT4, MRF+), so their results cannot be generalized to the high-risk LARC population.
3.3. De-Escalation: The PROSPECT Trial and Beyond
3.4. Immunotherapy in MSS Disease: A Paradigm Shift in Waiting, Not Yet Realized
3.5. Organ Preservation: Proven Efficacy in Expert Centers, Questions of Generalizability
3.6. Predictive Factors and Risk Stratification: Informing Personalization and the Role of ctDNA
3.7. Toxicity, Compliance, and Patient-Reported Quality of Life
3.8. Technological Advances in Radiotherapy: MRI-LINAC and Adaptive Planning
3.9. Health Economics and Real-World Feasibility
3.10. Synthesis of Key Findings
4. Discussion
4.1. Implementation of Total Neoadjuvant Therapy
4.2. Organ Preservation Paradigm and Precision Patient Selection
4.2.1. Integrating MRI into Clinical Decision-Making
4.3. Immunotherapy Integration in MSS Tumors: Promise Requiring Confirmation
4.4. Predictive Biomarkers and ctDNA-Guided Therapy: The Future of Personalization
4.5. Technological Advances in Radiotherapy: MRI-LINAC
4.6. Management of Specific Subgroups
4.7. Health Economics and Real-World Feasibility
4.8. Clinical Implications and Future Directions
- ctDNA-guided therapy: The final results of the CINTS-R trial (2-year DrTF rate) will determine whether ctDNA-guided risk stratification should become standard practice.
- Biomarker integration: Validation and clinical implementation of Immunoscore, ctDNA dynamics, and radiomic signatures are paramount.
- Understanding and mitigating trade-offs: Further research is required to elucidate the mechanisms behind increased locoregional recurrence risk and optimize radiotherapy techniques.
- Health economic analysis: Rigorous cost-effectiveness analyses of TNT, OP strategies, and novel technologies are essential.
- Trials for specific subgroups: Dedicated prospective trials are required for elderly/frail patients and those with LPLN+.
- Radiotherapy advancement: Continued optimization of techniques, including proton therapy [23].
5. Conclusions
Abbreviations
| Abbreviation | Full Form |
| Adj CT | Adjuvant Chemotherapy |
| AE | Adverse Event |
| APC | Article Processing Charge (cover letter only) |
| APR | Abdominoperineal Resection |
| ASCO | American Society of Clinical Oncology |
| AV | Anal Verge |
| CAP | College of American Pathologists (tumour regression grading) |
| CapeOX / CAPOX | Capecitabine + Oxaliplatin (chemotherapy regimen) |
| cCR | Clinical Complete Response |
| CEA | Carcinoembryonic Antigen |
| CGA | Comprehensive Geriatric Assessment |
| CI | Confidence Interval |
| CINTS-R | ctDNA-guided neoadjuvant treatment strategy for locally advanced rectal cancer (trial) |
| CNA | Copy Number Alteration |
| CR | Complete Response |
| CRM | Circumferential Resection Margin |
| CRT | Chemoradiotherapy / Chemoradiation |
| CT | Chemotherapy or Computed Tomography |
| ctDNA | Circulating Tumour DNA |
| CTV | Clinical Target Volume |
| CXB | Contact X-ray Brachytherapy |
| DFS | Disease-Free Survival |
| dMMR | Deficient Mismatch Repair |
| DrTF | Disease-related Treatment Failure |
| DWI | Diffusion-Weighted Imaging |
| EBRT | External Beam Radiotherapy |
| ECOG | Eastern Cooperative Oncology Group |
| eCRF | Electronic Case Report Form |
| EDC | Electronic Data Capture |
| ELAPE | Extralevator Abdominoperineal Excision |
| EMBASE | Excerpta Medica dataBASE |
| EMVI | Extramural Vascular Invasion |
| ERUS | Endorectal Ultrasound |
| ESMO | European Society for Medical Oncology |
| EUS | Endoscopic Ultrasound |
| FOLFIRINOX | Folinic acid, Fluorouracil, Irinotecan, Oxaliplatin |
| FOLFOX | Folinic acid, Fluorouracil, Oxaliplatin |
| G2/G3/G4 | Grade 2 / Grade 3 / Grade 4 (toxicity grading) |
| GI | Gastrointestinal |
| GTV | Gross Tumour Volume |
| Gy | Gray (unit of radiation dose) |
| HR | Hazard Ratio |
| ICI / ICIs | Immune Checkpoint Inhibitor(s) |
| ICR | Incomplete Response |
| IMRT | Intensity-Modulated Radiotherapy |
| irAE | Immune-related Adverse Event |
| ISR | Intersphincteric Resection |
| ITT | Intention-To-Treat |
| LARC | Locally Advanced Rectal Cancer |
| LARS | Low Anterior Resection Syndrome |
| LCRT | Long-Course Radiotherapy / Long-Course Chemoradiotherapy |
| LE | Local Excision |
| LLND | Lateral Lymph Node Dissection |
| LMIC | Low- and Middle-Income Countries |
| LoRP | Library of Reference Plans |
| LPLN | Lateral Pelvic Lymph Node(s) |
| LPLN+ | Lateral Pelvic Lymph Node-Positive |
| LPLN- | Lateral Pelvic Lymph Node-Negative |
| LR | Locoregional Recurrence |
| LRRFS | Locoregional Recurrence-Free Survival |
| MeSH | Medical Subject Headings |
| mFOLFOX6 | Modified FOLFOX regimen (6-drug variant) |
| MFS | Metastasis-Free Survival |
| MMR | Mismatch Repair |
| MPR | Major Pathological Response |
| MR | Magnetic Resonance |
| MRF | Mesorectal Fascia |
| MRF+ | Mesorectal Fascia Involvement / Positive |
| MRF- | Mesorectal Fascia Uninvolved / Negative |
| MRI | Magnetic Resonance Imaging |
| MRI-LINAC | Magnetic Resonance Imaging-guided Linear Accelerator |
| MSI-H | Microsatellite Instability-High |
| MSS | Microsatellite Stable |
| mITT | Modified Intention-To-Treat |
| N+ | Node-Positive |
| N0 | Node-Negative |
| NACRT | Neoadjuvant Chemoradiotherapy |
| NAR | Neoadjuvant Rectal (score) |
| NCCN | National Comprehensive Cancer Network |
| nCRT | Neoadjuvant Chemoradiotherapy |
| NCR | Near-Complete Response |
| nCT | Neoadjuvant Chemotherapy |
| NGS | Next-Generation Sequencing |
| NI | Non-Inferiority |
| NK | Natural Killer (cells) |
| NOM | Non-Operative Management |
| NS | Not Significant |
| OAR | Organ at Risk |
| OP | Organ Preservation |
| OPS | Organ Preservation Strategies |
| OR | Odds Ratio |
| OS | Overall Survival |
| pCR | Pathological Complete Response |
| PD-1 | Programmed Death-1 |
| PD-L1 | Programmed Death-Ligand 1 |
| PET/CT | Positron Emission Tomography / Computed Tomography |
| pMMR | Proficient Mismatch Repair |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| PRO | Patient-Reported Outcome |
| PROSPERO | International Prospective Register of Systematic Reviews |
| PTV | Planning Target Volume |
| QALY | Quality-Adjusted Life Year |
| QLQ-C30 | Quality of Life Questionnaire – Core 30 (EORTC) |
| QoL | Quality of Life |
| RCT | Randomised Controlled Trial |
| RNA-seq | RNA Sequencing |
| RoB 2 | Revised Cochrane Risk of Bias tool for randomised trials (version 2) |
| RT | Radiotherapy |
| SAE | Serious Adverse Event |
| SBRT | Stereotactic Body Radiotherapy |
| SCRT | Short-Course Radiotherapy |
| scRNA-seq | Single-Cell RNA Sequencing |
| SIB | Simultaneous Integrated Boost |
| SNI | Selective Nodal Irradiation |
| SNV | Single-Nucleotide Variant |
| TaTME | Transanal Total Mesorectal Excision |
| TCGA | The Cancer Genome Atlas |
| TEM | Transanal Endoscopic Microsurgery |
| TIL | Tumour-Infiltrating Lymphocyte |
| TMB | Tumour Mutational Burden |
| TME | Total Mesorectal Excision |
| TNT | Total Neoadjuvant Therapy |
| TPS | Tumour Proportion Score |
| TRG | Tumour Regression Grade |
| VAF | Variant Allele Frequency |
| W&W | Watch-and-Wait |
| XELOX | Capecitabine + Oxaliplatin (same as CAPOX) |
| yp | Pathological stage after neoadjuvant therapy |
References
- Bray, F.; Laversanne, M.; Sung, H.; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024, *74*, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Heald, R.J.; Ryall, R.D. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1986, 1, 1479–1482. [Google Scholar] [CrossRef] [PubMed]
- Kapiteijn, E.; Marijnen, C.A.; Nagtegaal, I.D.; et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N. Engl. J. Med. 2001, 345, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Rödel, C.; Graeven, U.; Fietkau, R.; et al. Oxaliplatin added to fluorouracil-based preoperative chemoradiotherapy and postoperative chemotherapy of locally advanced rectal cancer (the German CAO/ARO/AIO-04 study): Final results of the multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2015, 16, 979–989. [Google Scholar] [CrossRef]
- Sauer, R.; Becker, H.; Hohenberger, W.; et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 2004, 351(17), 1731–40. [Google Scholar] [CrossRef] [PubMed]
- Sauer, R.; Liersch, T.; Merkel, S.; et al. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: Results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J. Clin. Oncol. 2012, 30, 1926–1933. [Google Scholar] [CrossRef]
- Tarazona, N.; Gimeno-Valiente, F.; Gambardella, V.; et al. Targeted next-generation sequencing of circulating-tumor DNA for tracking minimal residual disease in localized colon cancer. Ann Oncol. 2019, 30(11), 1804–1812. [Google Scholar] [CrossRef] [PubMed]
- Cercek, A.; Roxburgh, C.S.D.; Strombom, P.; et al. Adoption of Total Neoadjuvant Therapy for Locally Advanced Rectal Cancer. JAMA Oncol 2018, 4(6), e180071. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rutten, H.J.; den Dulk, M.; Lemmens, V.E.; et al. Controversies of total mesorectal excision for rectal cancer in elderly patients. Lancet Oncol 2008, 9(5), 494–501. [Google Scholar] [CrossRef] [PubMed]
- Maas, M.; Beets-Tan, R.G.; Lambregts, D.M.; et al. Wait-and-see policy for clinical complete responders after chemoradiation for rectal cancer. J Clin Oncol. 2011, 29(35), 4633–40. [Google Scholar] [CrossRef] [PubMed]
- Bahadoer RR, Dijkstra EA, van Etten B, et al. Short-course radiotherapy followed by chemotherapy before total mesorectal excision (TME) versus preoperative chemoradiotherapy, TME, and optional adjuvant chemotherapy in locally advanced rectal cancer (RAPIDO): a randomised, open-label, phase 3 trial. Lancet Oncol. 2021 Jan;22(1):29-42. Epub 2020 Dec 7. Erratum in: Lancet Oncol. 2021 Feb;22(2):e42. [CrossRef] [PubMed]
- Conroy, T.; Bosset, J.F.; Etienne, P.L.; et al. Neoadjuvant chemotherapy with FOLFIRINOX and preoperative chemoradiotherapy for patients with locally advanced rectal cancer (UNICANCER-PRODIGE 23): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2021, 22(5), 702–715. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Tang, Y.; Hu, C.; et al. Multicenter, Randomized, Phase III Trial of Short-Term Radiotherapy Plus Chemotherapy Versus Long-Term Chemoradiotherapy in Locally Advanced Rectal Cancer (STELLAR). J Clin Oncol. 2022, 40(15), 1681–1692. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, X.; Liu, P.; Xiao, Y.; Meng, W.; Tang, Y.; Zhou, J.; Ding, P.R.; Ding, K.F.; Wang, B.; Guo, Q.; Sun, H.; Qiu, J.; Yu, Y.; Wu, B.; Zeng, H.; Deng, X.; Jiang, D.; Shen, Y.; Zhou, Z.; Wang, Z. Total neoadjuvant treatment with long-course radiotherapy versus concurrent chemoradiotherapy in local advanced rectal cancer with high risk factors (TNTCRT): a multicenter, randomized, open-label, phase 3 trial. J Clin Oncol. 2024, 42((17_) suppl, LBA3511. [Google Scholar] [CrossRef]
- Garcia-Aguilar, J.; Patil, S.; Gollub, M.J.; et al. Organ Preservation in Patients With Rectal Adenocarcinoma Treated With Total Neoadjuvant Therapy. J Clin Oncol. 2022, 40(23), 2546–2556. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gerard, J.P.; Barbet, N.; Schiappa, R.; et al. Neoadjuvant chemoradiotherapy with radiation dose escalation with contact x-ray brachytherapy boost or external beam radiotherapy boost for organ preservation in early cT2-cT3 rectal adenocarcinoma (OPERA): a phase 3, randomised controlled trial. Lancet Gastroenterol Hepatol 2023, 8(4), 356–367. [Google Scholar] [CrossRef] [PubMed]
- Glimelius, B.; Khan, T.; Adolfsson, K.; et al. Total neoadjuvant treatment using short-course radiotherapy and four CAPOX cycles in locally advanced rectal cancer with high-risk criteria for recurrence: a Swedish nationwide cohort study (LARCT-US). EClinicalMedicine 2024, 75, 102771. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lin, Z.Y.; Zhang, P.; Chi, P.; et al. Neoadjuvant short-course radiotherapy followed by camrelizumab and chemotherapy in locally advanced rectal cancer (UNION): early outcomes of a multicenter randomized phase III trial. Ann Oncol. 2024, 35(10), 882–891. [Google Scholar] [CrossRef] [PubMed]
- Shamseddine, A.; Turfa, R.; Chehade, L.; et al. Short-course radiation followed by mFOLFOX-6 plus avelumab for locally-advanced microsatellite stable rectal adenocarcinoma: The Averectal study. Eur J Cancer 2025, 222, 115428. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Li, H.Y.; Wei, L.C.; et al. Short-course-based TNT with or without PD-1 inhibitor for pMMR locally advanced rectal cancer: Phase 2 results of a randomized trial (STELLAR II). Med. 2025, 6(11), 100807. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.; Dai, H.; Sha, D.; et al. Total neoadjuvant treatment with short-course radiotherapy followed by sintilimab plus capecitabine-oxaliplatin versus short-course radiotherapy followed by capecitabine-oxaliplatin in patients with locally advanced rectal cancer (SPRING-01): a single-centre, open-label, phase 2, randomised controlled trial. Lancet 2025, (in press).
- Wang, F.; Lai, C.; Lv, Y.; et al. Efficacy and safety of combining short-course neoadjuvant chemoradiotherapy with enzalofilimab in locally advanced rectal cancer patients with microsatellite stability: a phase II PRECAM experimental study. 2024; (published).
- Pedone, C.; Sorcini, B.; Staff, C.; et al. Preoperative short-course radiation therapy with PROtons compared to photons in high-risk RECTal cancer (PRORECT): Initial dosimetric experience. Clin Transl Radiat Oncol 2022, 39, 100562. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Meyer, J.; Koessler, T. Comment on: A phase III randomised trial on the addition of a contact X-ray brachytherapy boost to standard neoadjuvant chemo-radiotherapy for organ preservation in early rectal adenocarcinoma: 5 year results of the OPERA trial. Ann Oncol. Epub. 2025, 36(5), 601. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhang, X.; Liu, Q.; et al. Rationale and design of a multicentre randomised controlled trial on circulating tumour DNA-guided neoadjuvant treatment strategy for locally advanced rectal cancer (CINTS-R). BMJ Open 2025, 15, e090765. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhou, J.; Geng, J.; et al. Feasibility of ctDNA-guided precision neoadjuvant therapy in locally advanced rectal cancer: Insights from the ongoing CINTS-R trial. Interim analysis 2025. [Google Scholar] [CrossRef]
- Benson, A.B.; Venook, A.P.; Al-Hawary, M.M.; et al. Rectal Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2022, 20(10), 1139–1167. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.N.; Jiang, J.; Jiang, L.M.; et al. Post-hoc analysis of clinicopathological factors affecting lateral lymph node metastasis based on STELLAR study for rectal cancer. Radiother Oncol. 2024, 200, 110512. [Google Scholar] [CrossRef] [PubMed]
- Manceau, G.; Karoui, M.; Werner, A.; Mortensen, N.J.; Hannoun, L. Comparative outcomes of rectal cancer surgery between elderly and non-elderly patients: a systematic review. Lancet Oncol 2012, 13(12), e525-36. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Zhou, J.; Qiu, H.; et al. A new treatment strategy for mid-low rectal cancer patients exhibiting a clinical complete or near-complete response to neoadjuvant chemoradiotherapy: Transanal endoscopic microsurgery – A multicenter prospective case-control clinical trial by MONT-R. Eur J Cancer 2025, 216, 115156. [Google Scholar] [CrossRef]
- Franke, A.J.; Parekh, H.; Starr, J.S.; et al. Total Neoadjuvant Therapy: A Shifting Paradigm in Locally Advanced Rectal Cancer Management. Clin Colorectal Cancer 2018, 17(1), 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hofheinz, R.D.; Wenz, F.; Post, S.; et al. Chemoradiotherapy with capecitabine versus fluorouracil for locally advanced rectal cancer: a randomised, multicentre, non-inferiority, phase 3 trial. Lancet Oncol 2012, 13(6), 579–88. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Pedersen, K.; Olsen, J.R.; et al. Nonoperative Rectal Cancer Management With Short-Course Radiation Followed by Chemotherapy: A Nonrandomized Control Trial. Clin. Colorectal. Cancer 2021, 20, e185–e193. [Google Scholar] [CrossRef] [PubMed]
- Markovina, S.; Youssef, F.; Roy, A.; et al. Improved Metastasis- and Disease-Free Survival With Preoperative Sequential Short-Course Radiation Therapy and FOLFOX Chemotherapy for Rectal Cancer Compared With Neoadjuvant Long-Course Chemoradiotherapy: Results of a Matched Pair Analysis. Int J Radiat Oncol Biol Phys Epub. 2017, 99(2), 417–426. [Google Scholar] [CrossRef] [PubMed]
- Ngan SY, Burmeister B, Fisher RJ, et al. Randomized trial of short-course radiotherapy versus long-course chemoradiation comparing rates of local recurrence in patients with T3 rectal cancer: Trans-Tasman Radiation Oncology Group trial 01.04. J Clin Oncol. 2012 Nov 1;30(31):3827-33. Epub 2012 Sep 24. Erratum in: J Clin Oncol. 2013 Jan 20;31(3):399. [CrossRef]
- Sauer, R.; Liersch, T.; Merkel, S.; et al. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: Results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J. Clin. Oncol. 2012, 30, 1926–1933. [Google Scholar] [CrossRef]
- Conroy, T.; Castan, F.; Etienne, P.L.; et al. Total neoadjuvant therapy with mFOLFIRINOX versus preoperative chemoradiotherapy in patients with locally advanced rectal cancer: long-term results of the UNICANCER-PRODIGE 23 trial. Ann Oncol. 2024, 35(10), 873–881. [Google Scholar] [CrossRef] [PubMed]
- Zwart, W.H.; Temmink, S.J.D.; Hospers, G.A.P.; et al. Oncological outcomes after a pathological complete response following total neoadjuvant therapy or chemoradiotherapy for high-risk locally advanced rectal cancer in the RAPIDO trial. Eur J Cancer 2024, 204, 114044. [Google Scholar] [CrossRef] [PubMed]
- Cisel, B.; Pietrzak, L.; Michalski, W.; et al. 5 x 5 Gy and consolidation chemotherapy for clinical T4 and fixed clinical T3 rectal cancer: long-term results of the randomized Polish II study. Ann Oncol (long-term follow-up). 2019. [Google Scholar]
- Schrag, D.; Shi, Q.; Weiser, M.R.; Gollub, M.J.; Saltz, L.B.; Musher, B.L.; Goldberg, J.; Al Baghdadi, T.; Goodman, K.A.; McWilliams, R.R.; Farma, J.M.; George, T.J.; Kennecke, H.F.; Shergill, A.; Montemurro, M.; Nelson, G.D.; Colgrove, B.; Gordon, V.; Venook, A.P.; O’Reilly, E.M.; Meyerhardt, J.A.; Dueck, A.C.; Basch, E.; Chang, G.J.; Mamon, H.J. Preoperative Treatment of Locally Advanced Rectal Cancer. N Engl J Med 2023, 389(4), 322–334. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mei, W.J.; Wang, X.Z.; Zhang, X.; et al. Neoadjuvant Chemotherapy With CAPOX Versus Chemoradiation for Locally Advanced Rectal Cancer With Uninvolved Mesorectal Fascia (CONVERT): Final Results of a Phase III Trial. J Clin Oncol. 2026. [Google Scholar] [CrossRef]
- Rouanet, P.; Rullier, E.; Lelong, B.; et al. Tailored Strategy for Locally Advanced Rectal Carcinoma (GRECCAR 4): Long-term Results From a Multicenter, Randomized, Open-Label, Phase II Trial. Dis Colon Rectum 2022, 65(8), 986–995. [Google Scholar] [CrossRef]
- Lin, Z.; Zhang, P.; Cai, M.; Li, G.; Liu, T.; Cai, K.; Wang, J.; Liu, J.; Liu, H.; Zhang, W.; Gao, J.; Wu, C.; Wang, L.; Wang, Z.; Hou, Z.; Kou, H.; Tao, K.; Zhang, T. Neoadjuvant short-course radiotherapy followed by camrelizumab and chemotherapy for locally advanced rectal cancer: 3-year survival from a phase 2 study. BMC Med. 2025, 23(1), 273. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, L.; Meng, X.; Wu, Q.; et al. Comparative efficacy of neoadjuvant short-course versus long-course radiotherapy-based regimens with or without immunotherapy for locally advanced pMMR rectal cancer: a systematic review and network meta-analysis. BMC Med 2026. [Google Scholar] [CrossRef]
- Shamseddine, A.; Turfa, R.; Chehade, L.; et al. Short-course radiation followed by mFOLFOX-6 plus avelumab for locally-advanced microsatellite stable rectal adenocarcinoma: The Averectal study. Eur J Cancer 2025, 222, 115428. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.; Dai, H.; Sha, D.; et al. Total neoadjuvant treatment with short-course radiotherapy followed by sintilimab plus capecitabine-oxaliplatin versus short-course radiotherapy followed by capecitabine-oxaliplatin in patients with locally advanced rectal cancer (SPRING-01): a single-centre, open-label, phase 2, randomised controlled trial. Lancet 2025, (in press).
- Yang, L.; Cui, X.; Wu, F.; Chi, Z.; Xiao, L.; Wang, X.; Liang, Z.; Li, X.; Yu, Q.; Lin, X.; Gao, C.; 47. The efficacy and safety of neoadjuvant chemoradiotherapy combined with immunotherapy for locally advanced rectal cancer patients: a systematic review. Front Immunol 2024, 15, 1392499. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- George, T.J.; Yothers, G.; Rahma, O.E.; et al. Long-term results from NRG-GI002: A phase II clinical trial platform using total neoadjuvant therapy (TNT) in locally advanced rectal cancer (LARC). ASCO Annual Meeting. (long-term results).
- Verheij, F.S.; Omer, D.M.; Williams, H.; et al. Long-Term Results of Organ Preservation in Patients With Rectal Adenocarcinoma Treated With Total Neoadjuvant Therapy: The Randomized Phase II OPRA Trial. J Clin Oncol. 2024, 42(5), 500–506. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gani, C.; Fokas, E.; Polat, B.; et al. Organ preservation after total neoadjuvant therapy for locally advanced rectal cancer (CAO/ARO/AIO-16): an open-label, multicentre, single-arm, phase 2 trial. Lancet Gastroenterol Hepatol 2025, 50 10(6), 562–572. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.N.; Jiang, J.; Jiang, L.M.; et al. Post-hoc analysis of clinicopathological factors affecting lateral lymph node metastasis based on STELLAR study for rectal cancer. Radiother Oncol. 2024, 200, 110512. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, E.A.; Hospers, G.A.P.; Kranenbarg, E.M.; et al. Quality of life and late toxicity after short-course radiotherapy followed by chemotherapy or chemoradiotherapy for locally advanced rectal cancer - The RAPIDO trial. Radiother Oncol. 2022, 171, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Bascoul-Mollevi, C.; Gourgou, S.; Borg, C.; et al. Neoadjuvant chemotherapy with FOLFIRINOX and preoperative chemoradiotherapy for patients with locally advanced rectal cancer (UNICANCER PRODIGE 23): Health-related quality of life longitudinal analysis. Eur J Cancer 2023, 186, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Li, H.; Xu, T.; et al. Quality of Life and Functional Outcomes in Patients With Locally Advanced Rectal Cancer Receiving Total Neoadjuvant Therapy Versus Concurrent Chemoradiation Therapy: An Analysis of the STELLAR Trial. Int J Radiat Oncol Biol Phys 2025, 122(1), 43–51. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Yang, X.; Qin, S.; et al. Efficient strategy for magnetic resonance image-guided adaptive radiotherapy of rectal cancer using a library of reference plans. Phys Imaging Radiat Oncol. 2025, 33, 100747. [Google Scholar] [CrossRef] [PubMed]
- Fokas, E.; Allgäuer, M.; Polat, B.; et al. Randomized Phase II Trial of Chemoradiotherapy Plus Induction or Consolidation Chemotherapy as Total Neoadjuvant Therapy for Locally Advanced Rectal Cancer: CAO/ARO/AIO-12. J Clin Oncol. Epub. 2019, 37(34), 3212–3222. [Google Scholar] [CrossRef] [PubMed]
- Bujko, K.; Wyrwicz, L.; Rutkowski, A.; et al. Long-course oxaliplatin-based preoperative chemoradiation versus 5 × 5 Gy and consolidation chemotherapy for cT4 or fixed cT3 rectal cancer: results of a randomized phase III study. Ann Oncol. 2016, 27(5), 834–42. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.; Thompson, H.M.; Lin, S.T.; et al. Endoscopic Predictors of Residual Tumor After Total Neoadjuvant Therapy: A Post Hoc Analysis From the Organ Preservation in Rectal Adenocarcinoma Trial. Dis Colon Rectum 2024, 67(3), 369–376. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- El Sissy, C.; Kirilovsky, A.; Van den Eynde, M.; et al. A Diagnostic Biopsy-Adapted Immunoscore Predicts Response to Neoadjuvant Treatment and Selects Patients with Rectal Cancer Eligible for a Watch-and-Wait Strategy. Clin Cancer Res. Epub. 2020, 26(19), 5198–5207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chi, P.; Shi, L.; et al. Neoadjuvant Modified Infusional Fluorouracil, Leucovorin, and Oxaliplatin With or Without Radiation Versus Fluorouracil Plus Radiation for Locally Advanced Rectal Cancer: Updated Results of the FOWARC Study After a Median Follow-Up of 10 Years. J Clin Oncol. 2024, 43(6), 633–640. [Google Scholar] [CrossRef]
| Trial/Analysis (Primary Publication) | Phase | Sample Size | Control Arm | Experimental Arm | Primary Endpoint(s) |
|---|---|---|---|---|---|
| Landmark Phase III TNT Trials | |||||
| RAPIDO | III | 920 | LCRT → TME (± Adj CT) | SCRT → CT (CAPOX/FOLFOX) → TME | 3-year DrTF |
| PRODIGE 23 | III | 461 | LCRT → TME → Adj CT | 6x FOLFIRINOX → LCRT → TME → 6x mFOLFOX6 | 3-year DFS |
| STELLAR | III | 599 | LCRT → TME → 6x CAPOX | SCRT → 4x CAPOX → TME → 2x CAPOX | 3-year DFS |
| TNTCRT | III | 458 | LCRT + capecitabine → TME → Adj CAPOX | LCRT + 6x CAPOX (1 ind, 2 conc, 3 cons) → TME | 3-year DFS |
| Neoadjuvant Chemotherapy Without Routine RT (De-escalation) | |||||
| PROSPECT [40] | III | 1194 | LCRT (5.5 weeks) → TME | mFOLFOX6 (6 cycles) → TME (± selective LCRT) | 5-year DFS (NI) |
| CONVERT | III | 663 | LCRT + capecitabine → TME | 4x CAPOX → TME | 3-year LRRFS (NI) |
| FOWARC 10-year | III | 495 | 5-FU + LCRT | mFOLFOX6 ± LCRT | 10-year DFS |
| GRECCAR 4 | II | 148 | Response-adaptive (induction CT → good responders → surgery; poor responders → CRT) | — | pCR, OP rate |
| Immunotherapy-TNT Trials | |||||
| UNION | III | 231 | LCRT → 2x CAPOX → TME → 6x CAPOX | SCRT → 2x CAPOX + Camrelizumab → TME → 6x CAPOX + Camrelizumab | pCR |
| STELLAR II | II | 218 | SCRT → 4x CAPOX → TME/WW → 2x CAPOX | SCRT → 4x CAPOX + Sintilimab → TME/WW → 2x CAPOX + Sintilimab | CR rate |
| SPRING-01 | II | 98 | SCRT → 6x CAPOX → TME | SCRT → 6x CAPOX + Sintilimab → TME | pCR |
| PRECAM | II | 34 | — (single-arm) | SCRT → 2x CAPEOX + 6x Enzalofilimab → TME | pCR |
| NRG-GI002 | II platform | 178 (EA1), 185 (EA2) | FOLFOX → CRT → TME | EA2: + Pembrolizumab during CRT | NAR score reduction |
| Averectal [45] | II | 44 | Single-arm: SCRT → 6x mFOLFOX-6 + Avelumab → TME | — | pCR vs historical |
| Organ Preservation and Sequencing | |||||
| OPRA [21] | II | 324 | Induction: CRT → TME/WW | Consolidation: CRT → TME/WW | 3-yr DFS; OP |
| CAO/ARO/AIO-16 [20] | II | 91 | Single-arm: CRT → 3x FOLFOX → Response → TME/WW | — | CCR rate |
| OPERA [22] | III | 148 | LCRT + EBRT Boost → TME/LE/WW | LCRT + CXB Boost → TME/LE/WW | 3-year OP rate |
| MONT-R TEM [30] | Prospective case-control | 80 | Radical surgery (TME) | TEM local excision (cCR/near-cCR) | 5-year DFS |
| Predictive Factor and Subgroup Analyses | |||||
| RAPIDO pCR Analysis [54] | Post-hoc | 920 | Analysis of factors associated with pCR | — | Factors for pCR; prognosis of pCR |
| OPRA Response Grade [51] | Post-hoc | 324 | Analysis correlating 3-tier response with outcomes | — | OP and DFS by CCR/NCR/ICR |
| STELLAR LPLN Analysis [10] | Post-hoc | 599 | Subgroup analysis of LPLN+ patients | — | Outcomes in LPLN+ |
| CINTS-R | III RCT | 470 (planned) | Conventional nCRT | ctDNA-guided: high-risk → TNT; low-risk → nCRT; dMMR/TMB-H → immunotherapy | 2-year DrTF |
| Radiotherapy Modality | |||||
| PRORECT | Dosimetric | 128 (plan) | Photon CRT | Proton CRT | Dosimetric comparison; predicted acute ≥G2 GI toxicity |
| MRI-LINAC LoRP | Retrospective | 10 pts (50 fractions) | Conventional couch shift / fully adaptive | Library of reference plans (LoRP) | Target coverage, treatment time |
| Other Key Trials | |||||
| Polish II long-term | III | 515 | LCRT + oxaliplatin | SCRT + 3x FOLFOX4 → TME | OS, DFS, local failure |
| MONT-R chemo [30] | III | 505 | nCRT + capecitabine | nCRT + CapeOX | 3-year DFS |
| CAO/ARO/AIO-12 [56] | II | 306 | Induction FOLFOX → CRT → TME | CRT → Consolidation FOLFOX → TME | pCR rate |
| NOMINATE | II | 66 (planned) | CRT → 6x CapeOx | 3x CapeOx+Bev → CRT → 3x CapeOx | pCR/CCR ≥2 years |
| Risk Group | MRI Features | Recommended Treatment Approach | Supporting Evidence |
|---|---|---|---|
| High risk | -MRF+ (≤1 mm) -cT4a (peritoneal invasion) or cT4b (organ invasion) -EMVI+ -Bulky N2 (≥4 nodes or >10-15 mm) -LPLN+ (≥7 mm or ≥5 mm with malignant features) |
TNT (preferred) -Consider LCRT-based TNT (PRODIGE 23, TNTCRT) for highest local risk -SCRT-based TNT (RAPIDO, STELLAR) is an option but monitor local control -For LPLN+: consider LLND after TNT if nodes persist -Immunotherapy + TNT (investigational, e.g., UNION, SPRING-01, PRECAM) |
RAPIDO, PRODIGE 23, STELLAR, TNTCRT, UNION, SPRING-01, STELLAR LPLN analysis, Ogura criteria |
| Intermediate risk | -cT3, MRF- (>1 mm) -cN1 (1-3 small nodes) -EMVI- -LPLN- |
-Standard CRT → TME → adjuvant CT or -TNT (depending on patient age, comorbidities, desire for organ preservation) |
CAO/ARO/AIO-04, German CRT trials, OPRA (consolidation TNT for OP goal) |
| Low risk | -cT2-3, MRF-, N0-1 (non-bulky) -EMVI-, LPLN- -Mid-upper rectum (≥5-10 cm from AV) |
De-escalation options: -Neoadjuvant chemotherapy alone (FOLFOX/CAPOX) with selective CRT (PROSPECT, CONVERT, FOWARC) -Short-course RT alone (Stockholm III) -Response-adaptive strategies (GRECCAR 4) |
PROSPECT, CONVERT, FOWARC 10-year, GRECCAR 4 |
| Trial / Analysis | Primary Efficacy Endpoint | pCR / cCR Rate | Key Efficacy Insights |
|---|---|---|---|
| RAPIDO [11] | 3-year DrTF: 23.7% vs 30.4%* | 28% vs 14%* | TNT reduced distant metastases (20.0% vs 26.8%) and improved DrTF. |
| PRODIGE 23 [12] | 7-year DFS: 67.6% vs 62.5%; 7-year OS: 81.9% vs 76.1% | 28% vs 12%* | TNT improved DFS, reduced distant metastases (17.8% vs 25.5%), and showed OS benefit. |
| STELLAR [28] | 3-year DFS: 64.5% vs 62.3% (NI) | 21.8% vs 12.3%* | SCRT-based TNT was non-inferior to LCRT-based therapy. |
| TNTCRT [14] | 3-year DFS: 77.0% vs 67.9%* (HR 0.623); 3-year MFS: 83.0% vs 74.2%* | 27.5% vs 9.9%* | LCRT-based TNT with CAPOX significantly improved DFS, MFS, and pCR. |
| PROSPECT [40] | 5-year DFS: 80.8% vs 81.7% (NI) | 21.4% vs 24.0% (NS) | FOLFOX with selective LCRT non-inferior to routine LCRT; 9.1% required salvage LCRT. |
| CONVERT [41] | 3-year LRRFS: 96.3% vs 97.4% (NI not formally met) | ~20% vs ~22% | nCT offered comparable DFS/OS with significantly less long-term toxicity. |
| FOWARC 10-year | 10-year DFS: 60.5-62.6% vs 52.5% (NS); 10-year LR: 9.6% vs 10.8% (NS) | ~28% vs ~14%* | mFOLFOX6 alone non-inferior to CRT at 10 years; pCR predicts excellent survival. |
| GRECCAR 4 [42] | 5-year DFS (response-adaptive) | Not reported | Good responders to induction CT can avoid CRT without compromising outcomes. |
| UNION [18] | - | 39.8% vs 15.3%* | Adding camrelizumab to SCRT+CAPOX nearly tripled pCR rate in MSS. |
| STELLAR II [20] | - | CR: 44.0% vs 22.9%* | Adding sintilimab increased cCR rate in MSS. |
| SPRING-01 [21] | - | pCR: 59.2% vs 32.7%; CR: 61.2% vs 32.7% | Adding sintilimab to SCRT+CAPOX significantly increased pCR and CR rates (p=0.015). |
| PRECAM [22] | - | pCR: 62.5%; MPR (TRG 0-1): 75% | Short-course nCRT + enzalofilimab achieved highest pCR in MSS LARC to date. |
| NRG-GI002 [48] | NAR score diff: 2.9 (p=0.21); 3-year OS: 95% vs 87%* | Not reported | Pembrolizumab added to TNT improved 3-year OS but not DFS; NAR reduction not significant. |
| Averectal [45] | - | pCR: 36% | SCRT + mFOLFOX-6 + avelumab achieved 36% pCR. |
| OPRA [15] | 5-year DFS: ~70% (both arms) | CCR/NCR: 76% vs 72% | 5-year TME-free survival: 54% (Consolidation) vs 39% (Induction)*; Consolidation favored for OP. |
| OPERA [16] | - | CCR/NCR: 92% vs 64%* | 5-year OP: 79% (CXB) vs 56% (EBRT)*; CXB boost superior. |
| MONT-R TEM [30] | 5-year DFS: 75.6% vs 80.9% (NS); 5-year OS: 93.2% vs 88.2% (NS) | pCR (TEM): 57.9% ypT0 | Local excision (TEM) after cCR/near-cCR provides comparable survival with better function. |
| RAPIDO pCR | 5-year OS after pCR: >90% (both arms) | - | Predictors of pCR: TNT (OR 2.70), CEA <5 µg/L, tumor <40 mm. |
| OPRA Grade | 3-year DFS: 88% (CCR) vs 69% (NCR) vs 56% (ICR) | - | 3-year OP: 77% (CCR) vs 40% (NCR); three-tier response highly prognostic. |
| STELLAR LPLN [51] | 3-year DFS in LPLN+: 51.7% vs 66.2% (LPLN-) | - | LPLN+ remains a negative prognostic factor despite TNT. |
| CINTS-R (interim) [25,26] | Primary endpoint (2-year DrTF) not yet reported | - | ctDNA-guided stratification feasible; substantial discordance with clinical risk. |
| Polish II long-term [39] | 8-year OS: 49% vs 49% (NS); 8-year DFS: 43% vs 41% (NS) | 16% vs 12% (NS) | Early OS benefit of SCRT-based TNT lost with longer follow-up; no difference in late complications. |
| MONT-R chemo [30] | 3-year DFS: comparable (NS) | 25.5% vs 25.3% (NS) | Adding oxaliplatin to nCRT improved tumor regression (CAP 0-1: 58.6% vs 46.8%*) but not survival. |
| Trial | cT4 (%) | cN2 (%) | EMVI+ (%) | MRF+ (%) | Other high-risk features | Radiotherapy backbone | pCR rate (TNT arm) | 3-year DFS / OS |
|---|---|---|---|---|---|---|---|---|
| High-risk LARC trials | ||||||||
| RAPIDO | 74% | 86% | 53% | ~35-40% | - | SCRT (5x5 Gy) | 28% | 3-yr DrTF 23.7% (improved) |
| PRODIGE 23 | 26% | ~70% | Not reported | Not reported | - | LCRT (50.4 Gy) | 27.5% | 7-yr OS 81.9% (improved) |
| STELLAR | 37% | 74% | Not reported | Not reported | - | SCRT (5x5 Gy) | 21.8% | 3-yr DFS 64.5% (NI) |
| TNTCRT | Included (cT4a-b) | Included | Included | Included | Enlarged lateral nodes | LCRT (50-50.4 Gy) | 27.5% | 3-yr DFS 77.0% (improved) |
| Polish II [39] | Majority cT4 or fixed cT3 | Not specified | Not specified | Not specified | Locally recurrent (3%) | SCRT (5x5 Gy) | 16% | 8-yr OS 49% (NS) |
| MONT-R chemo [30] | High-risk (cT4, cN2, EMVI+, MRF+) | Included | Included | Included | - | LCRT | 25.5% (CapeOX) | 3-yr DFS comparable (NS) |
| Lower-risk / de-escalation trials | ||||||||
| PROSPECT [40] | 0% (excluded cT4) | ~75% (cN1-2) | Not specified | 0% (CRM-negative) | Mid-upper rectal tumors | LCRT (selective) | 21.4% (FOLFOX alone) | 5-yr DFS 80.8% (NI) |
| CONVERT | 0% (excluded cT4b, MRF+) | Included (cN2 allowed) | 17-22% | 0% (uninvolved MRF) | Distance 5-12 cm from AV | None (nCT alone) | ~20% | 3-yr DFS 89.2% (comparable) |
| FOWARC | ~25-27% (cT4a-b) | ~60-70% | Not reported | ~31-35% | - | LCRT (in RT arms) | 27.5% (mFOLFOX+RT) | 10-yr DFS 60-62% (NS) |
| Trial | Acute grade ≥3 toxicity (Pre-operative) | Completion of planned pre-operative therapy | Notable Toxicity and Clinical Safety Observations |
|---|---|---|---|
| RAPIDO [52] | 47.6% vs 24.7%* (TNT vs CRT) | 84.6% vs 90.0%* | No significant difference in global QoL, bowel function (LARS), or chronic toxicity at 3 years. |
| PRODIGE 23 [53] | 46.9% vs 35.6%* (TNT vs CRT) | 89.6% vs 98.7%* | TNT transiently reduced QoL during CT; long-term QoL converged. Baseline physical function prognostic. |
| STELLAR [54] | 26.5% vs 12.6%* (TNT vs CRT) | 82.6% vs 95.2%* | At 6-year follow-up, no clinically significant difference in global QoL or anal function (Wexner). |
| TNTCRT [14] | Thrombocytopenia (10.3% grade 3-4 in TNT arm) | High in both arms | TNT well tolerated; no significant difference in severe post-op morbidity. |
| PROSPECT [40] | 41.0% vs 22.6%* (FOLFOX vs CRT) | 89.5% vs 84.3% | FOLFOX: higher neuropathy, fatigue, nausea; CRT: higher diarrhea. Long-term: CRT worse sexual function. |
| CONVERT [41] | Grade 2-4 long-term AEs: 16.0% vs 26.3%* | ~90% both arms | nCT significantly reduced proctitis (33.6% vs 41.7%, p=0.049) and long-term toxicity. |
| FOWARC 10-year [60] | Not reported in long-term update | Not reported | Long-term survival comparable; pCR associated with excellent outcomes (10-year OS 92.4%). |
| GRECCAR 4 | Not reported | High | Response-adaptive strategy feasible; good responders avoided CRT toxicity. |
| UNION | ~45% vs ~35% | ~88% both arms | Adding camrelizumab increased irAEs (rash, thyroiditis) typically grade 1-2. |
| STELLAR II | 34.5% vs 19.4%* (iTNT vs TNT) | High both arms | Grade 3-4 irAEs: 5.5%; manageable. |
| SPRING-01 | Grade 3-4: 33% vs 35% (NS) | 82% vs 84% | Most common grade 3-4: thrombocytopenia (12% vs 22%). No treatment-related deaths in iTNT arm. |
| PRECAM | Grade 3: 2/32 (6.25%) | 32/34 completed | Adverse events: tenesmus (78.1%), diarrhea (62.5%), leukopenia (40.6%); manageable. |
| NRG-GI002 | Not reported in long-term | Not reported | No unexpected safety signals with pembrolizumab. |
| OPRA | ~38% (induction) vs ~41% (consolidation) | ~85% | No significant difference in late toxicity between sequences. |
| CAO/ARO/AIO-16 | 36% (during TNT) | 90/91 RT; 82/88 CT | Sustained CCR patients had better bowel function (lower LARS/Wexner) at 18/36 months vs immediate TME. |
| OPERA | Not reported | High | CXB boost well tolerated; no increase in severe late toxicity. |
| MONT-R TEM | Not applicable (post-nCRT) | - | TEM: significantly shorter operation time, less blood loss, shorter hospital stay, better sphincter function (Wexner 1 vs 4, LARS 0 vs 17). |
| PRORECT | Acute ≥G2 Diarrhea: 10% vs 27%* (Proton vs Photon) | Similar | Dosimetric comparison suggested proton therapy reduces acute GI toxicity. |
| MRI-LINAC LoRP | Not reported | - | LoRP reduced treatment session duration by >20 min vs fully adaptive; 92% of LoRP plans acceptable vs 74% for couch shift. |
| Polish II long-term | Acute toxicity lower in SCRT-based TNT | ~85% | Late grade 3+ complications: 11% vs 9% (NS); no difference in late toxicity. |
| MONT-R chemo | Grade 3-4: 14.1% vs 9.3% (NS) | ~91% | CapeOX increased tumor regression (CAP 0-1) without significant increase in severe AEs. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




