Submitted:
11 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction: The Paradox of BCG and SCID
2. The Molecular Architecture of Trained Immunity
2.1. Receptor Engagement and Metabolic Reprogramming
2.2. Epigenetic Modifications and Cytokine Profiling
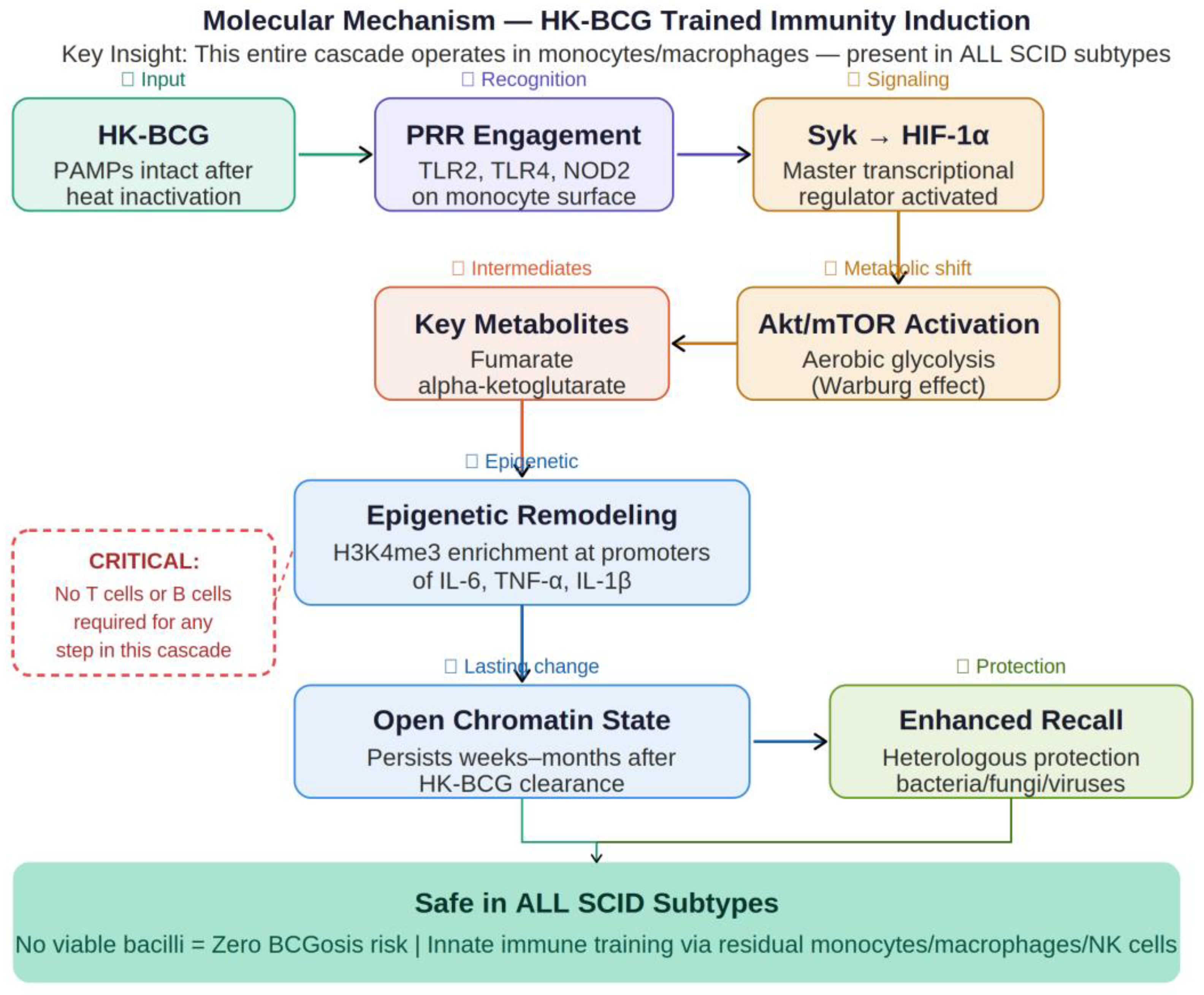
2.3. The Cytokine Paradox: Amplified Recall vs. Systemic Dampening
3. The Paradigm-Shifting Application: HK-BCG for SCID
3.1. Understanding SCID: The Immunological Landscape
| SCID Subtype | T cells | B cells | NK cells | HK-BCG Innate Target | Predicted HK-BCG Benefit |
|---|---|---|---|---|---|
| X-linked SCID (IL2RG) | Absent | Present (non-functional) | Absent | Monocytes, Macrophages, DCs | Moderate — monocyte trained immunity; bridge to HSCT |
| RAG1/RAG2 Deficiency | Absent | Absent | Present | NK cells + Monocytes + Macrophages | HIGH — NK cell training + monocyte epigenetic priming |
| ADA Deficiency | Absent | Absent | Absent | Monocytes (variable function) | Low-Moderate — dependent on ERT restoration |
| Jak3 Deficiency | Absent | Present | Absent | Monocytes, Macrophages | Moderate — innate arm partially trainable |
| Post-HSCT Reconstitution | Recons. | Recons. | Recons. | All innate + emerging adaptive | OPTIMAL — HK-BCG as post-HSCT immune priming platform |
3.2. Mechanistic Rationale: Why HK-BCG Can Work in SCID
3.2.1. Monocyte and Macrophage Training
3.2.2. NK Cell Training in T⁻B⁻NK⁺ SCID Subtypes
3.2.3. Dendritic Cell Activation and Post-HSCT Priming
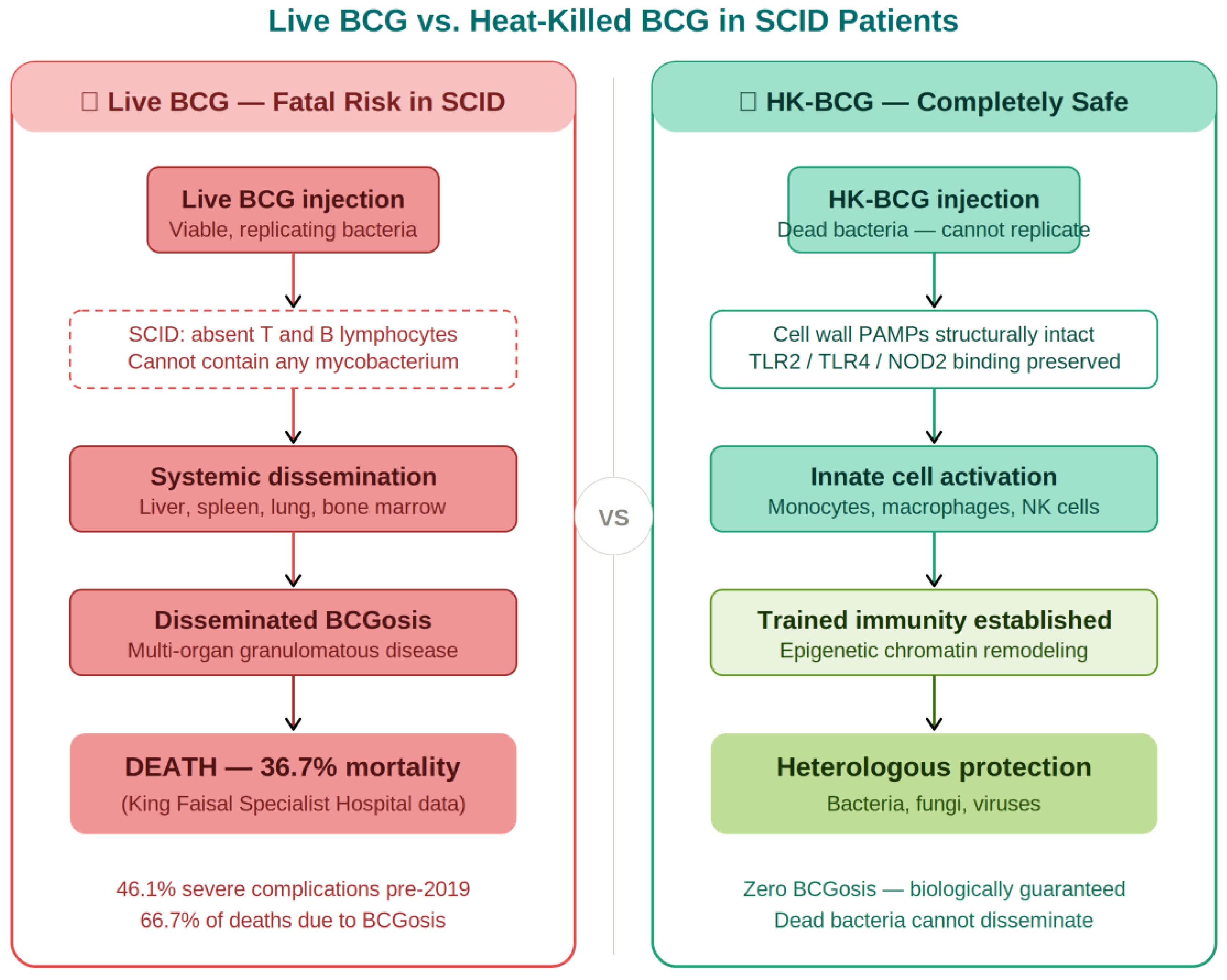
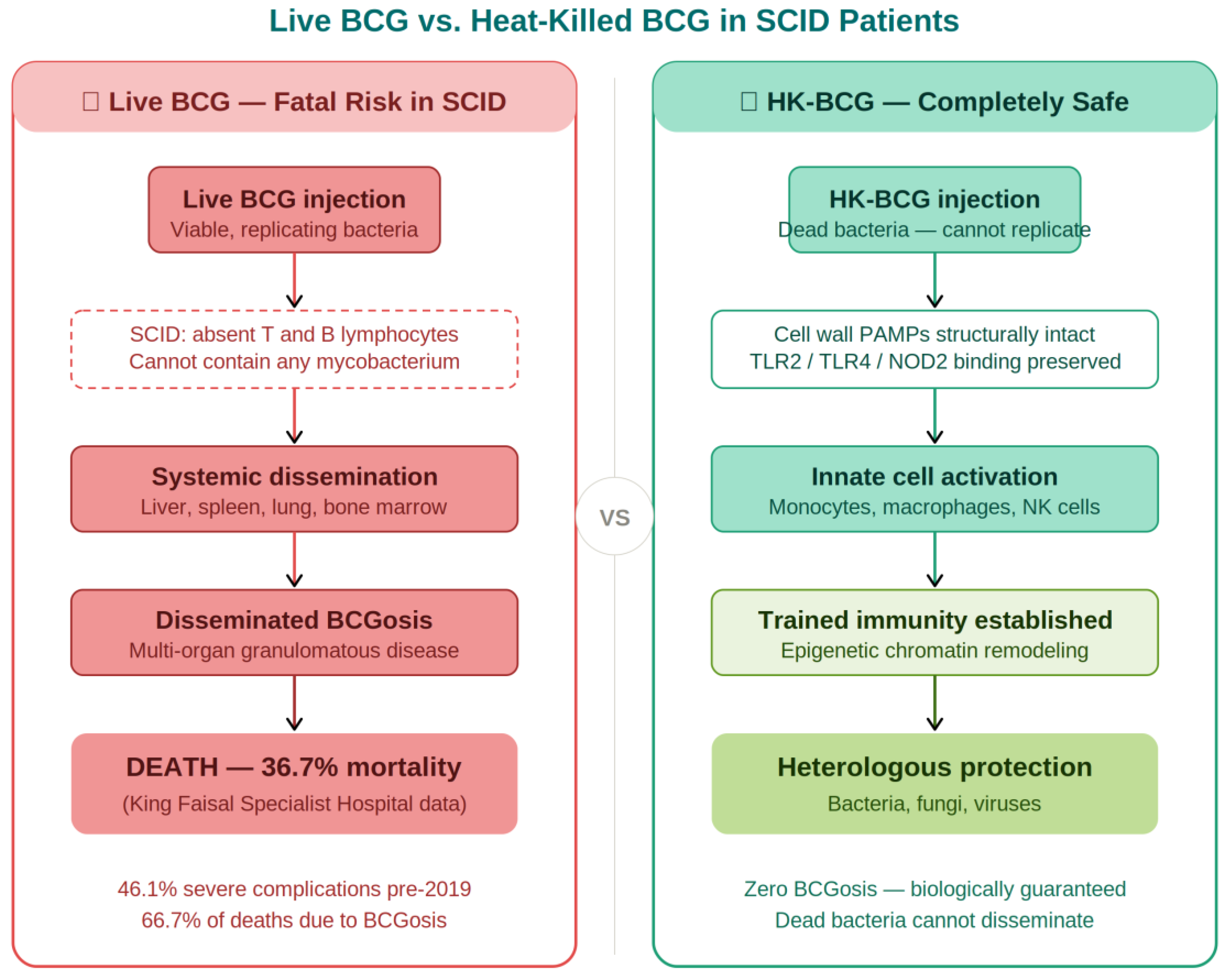
3.3. Safety Profile in SCID: The Definitive Case
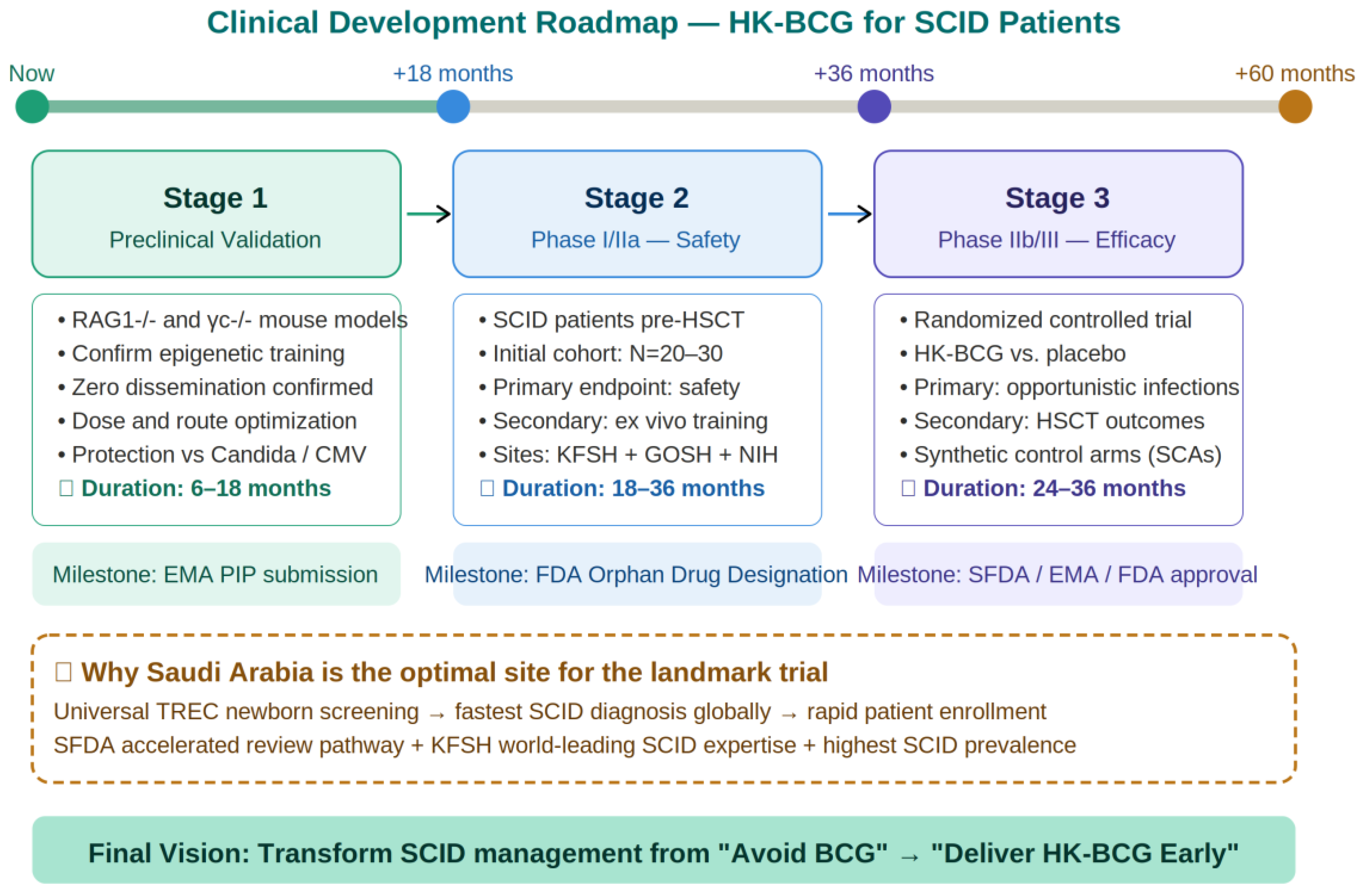
3.4. Clinical Implementation: A Proposed Framework
3.5. Theoretical Limitations and Counterarguments
4. The Myeloid-Derived Suppressor Cell (MDSC) Evasion Mechanism
5. Fractionation and Formulation Engineering: The BCG Cell Wall Skeleton
5.1. Nanoparticle Encapsulation — Enabling Systemic Delivery
6. Clinical Applications in Urologic Oncology: NMIBC
| Agent | Status | Advantages | Limitations | Safe in SCID? |
|---|---|---|---|---|
| Live BCG | Live attenuated | Highest immunogenicity; proven clinical record | Fatal BCGosis in SCID; cold chain dependency | CONTRAINDICATED — Fatal risk |
| Mitomycin C | Chemotherapy | No infection risk; usable post-op | No immunological memory; higher recurrence vs BCG | Tolerable, but no immune benefit |
| HK-BCG | Non-viable whole | Zero infection risk; trains innate immunity; thermal stability | Slightly reduced cytotoxicity; MDSC induction (addressable) | SAFE — Cannot cause BCGosis |
| Liposomal BCG-CWS | Purified fraction | Activates AMPK; IV delivery possible; highly dispersible | Requires LEEL nanomanufacturing; limited PID data | Potentially SAFE — requires PID studies |
7. Next-Generation Combinatorial Therapeutics
8. Dermatological and Mucosal Applications
9. Safety Profiles and the Immunocompromised Host
10. Global Supply Chain, Thermal Stability, and Regulatory Modernization
11. Conclusions
Acknowledgments
Conflict of Interest Statement
References
- Aksungur, N.; Odabaşı, Ş; Çetin, İ; et al. Unleashing the power of the BCG vaccine in modulating viral immunity through heterologous protection: a scoping review. Hum Vaccin Immunother. 2025, 21, 2521190. [Google Scholar] [CrossRef]
- Chen, J.; Gao, L.; Wu, X.; et al. BCG-induced trained immunity: history, mechanisms and potential applications. J Transl Med. 2023, 21, 106. [Google Scholar] [CrossRef]
- Covián, C.; Fernández-Fierro, A.; Retamal-Díaz, A.; et al. BCG-induced cross-protection and development of trained immunity: implication for vaccine design. Front Immunol. 2019, 10, 2806. [Google Scholar] [CrossRef] [PubMed]
- Ciarlo, E.; Heinonen, T.; Théroude, C.; et al. Heat-killed Mycobacterium tuberculosis induces trained immunity in vitro and in vivo administered systemically or intranasally. iScience 2024, 27, 108563. [Google Scholar] [CrossRef]
- Al-Herz, W.; Essa, S.; Rezk, A. BCGitis and BCGosis: clinical spectrum, immunological mechanisms, and risk management. Vaccines (Basel) 2025, 13, 1179. [Google Scholar] [CrossRef]
- Aldhaheri, A.; Alyabes, O.; Aljumaah, S.; et al. The effects of postponing BCG vaccination on the risk of BCG-related complications among patients with severe combined immunodeficiency disease in Saudi Arabia. Front Immunol. 2025, 16, 1596963. [Google Scholar] [CrossRef]
- Marciano, B.E.; Huang, C.Y.; Joshi, G.; et al. BCG vaccination in patients with severe combined immunodeficiency: complications, risks, and vaccination policies. J Allergy Clin Immunol. 2014, 133, 1134–1141. [Google Scholar] [CrossRef]
- Netea, M.G.; Giamarellos-Bourboulis, E.J.; Domínguez-Andrés, J.; et al. Trained immunity: a tool for reducing susceptibility to and the severity of SARS-CoV-2 infection. Cell. 2020, 181, 969–977. [Google Scholar] [CrossRef]
- Mourits, V.P.; Koeken, V.A.C.M.; de Bree, L.C.J.; et al. BCG-induced trained immunity in healthy individuals: the effect of plasma muramyl dipeptide concentrations. J Immunol Res. 2020, 2020, 5812743. [Google Scholar] [CrossRef]
- Jensen, K.J.; Larsen, N.; Biering-Sørensen, S.; et al. Neonatal BCG vaccination influences cytokine responses to toll-like receptor ligands and heterologous antigens. J Allergy Clin Immunol. 2015, 135, 483–487. [Google Scholar] [CrossRef]
- Nigou, J.; Zelle-Rieser, C.; Gilleron, M.; et al. Maturation of human dendritic cells by cell wall skeleton of Mycobacterium bovis Bacillus Calmette-Guérin: involvement of toll-like receptors. Infect Immun. 2001, 69, 1775–1783. [Google Scholar]
- Uchida, M.; Maeda, H.; Makino, M.; et al. Application of BCG-CWS as a systemic adjuvant by using nanoparticulation technology. Mol Pharm. 2019, 16, 1543–1553. [Google Scholar] [CrossRef]
- Soukaina, B.; Khalid, E.K.; Hicham, E.E.; et al. Efficacy of different Bacillus Calmette-Guérin (BCG) strains on recurrence rates among intermediate/high-risk non-muscle invasive bladder cancers (NMIBCs): single-arm study, systematic review, cumulative and network meta-analysis. Cancers (Basel) 2023, 15, 1937. [Google Scholar] [CrossRef]
- Cheng, C.W.; Liu, H.W.; Lin, P.H.; et al. The comparison of different BCG strains in the intravesical treatment of non-muscle invasive urothelial carcinoma of urinary bladder: a real-world practice. J Chin Med Assoc. 2021, 84, 387–393. [Google Scholar] [CrossRef]
- Morales, A.; Cohen, Z. Loss of BCG viability adversely affects the direct response of urothelial carcinoma to BCG. Can Urol Assoc J 2018, 12, E424–E430. [Google Scholar]
- Ito, M.; Hiramatsu, H.; Kabashima, K.; et al. Bladder cancer medication Bacillus Calmette-Guérin-cell wall skeleton: focusing on alternatives and developments to limitations. Cancer Sci. 2025, 116, 10–20. [Google Scholar] [CrossRef]
- Camacho-Sandoval, R.; Nieto-Patlán, A.; Herrera, M.T.; et al. Bladder cancer immunotherapy: parallel advances in BCG optimization and next-generation vaccine platforms. Vaccines (Basel) 2025, 13, 306. [Google Scholar] [CrossRef]
- González-Aparicio, M.; Viales-Bastida, R.R.; Vázquez, J.A.; et al. Revitalizing Bacillus Calmette-Guérin immunotherapy for bladder cancer: nanotechnology and bioengineering approaches. Nanomaterials (Basel) 2025, 15, 33. [Google Scholar] [CrossRef]
- Cossio, M.L.; Rubio, R.; Oliva, C.; et al. Comparative analysis of very reduced vs full dose BCG treatment for high-risk non-muscle invasive bladder cancer: a contemporary experience from Chile. Bladder Cancer 2024, 10, 51–60. [Google Scholar] [CrossRef]
- Subiela, J.D.; Palou, J.; Gaya, J.M.; et al. Outcomes of intravesical Bacillus Calmette-Guérin in patients with non-muscle invasive bladder cancer: a retrospective study in Australia. Front Urol. 2024, 4, 1309532. [Google Scholar] [CrossRef]
- Gonzalez-Aparicio, M.; Alfaro, C. MDSC depletion during immunization with heat-killed Mycobacterium tuberculosis increases protection against BCG infection. Front Immunol. 2025, 16, 1646526. [Google Scholar] [CrossRef]
- Gonzalez-Aparicio, M.; Alfaro, C.; Lozano, T.; et al. Heat-killed Mycobacterium tuberculosis prime-boost vaccination induces myeloid-derived suppressor cells with spleen dendritic cell-killing capability. JCI Insight 2020, 5, e137018. [Google Scholar]
- Kleinnijenhuis, J.; Quintin, J.; Preijers, F.; et al. Bacille Calmette-Guérin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc Natl Acad Sci U S A 2012, 109, 17537–17542. [Google Scholar] [CrossRef]
- Arts, R.J.W.; Moorlag, S.J.C.F.M.; Novakovic, B.; et al. BCG vaccination protects against experimental viral infection in humans through the induction of cytokines associated with trained immunity. Cell Host Microbe 2018, 23, 89–100.e5. [Google Scholar] [CrossRef]
- de Bree, L.C.J.; Koeken, V.A.C.M.; Joosten, L.A.B.; et al. Whole blood profiling of Bacillus Calmette-Guérin-induced trained innate immunity in infants identifies epidermal growth factor, IL-6, platelet-derived growth factor-AB/BB, and natural killer cell activation. Front Immunol. 2019, 10, 1251. [Google Scholar] [CrossRef]
- Escribà-Garcia, L.; Alvarez-Fernández, C.; Mancebo-Sierra, E.; et al. BCG activation of trained immunity is associated with induction of cross-reactive COVID-19 antibodies in a BCG vaccinated population. medRxiv 2024. [Google Scholar] [CrossRef]
- Kleinnijenhuis, J.; Quintin, J.; Preijers, F.; et al. BCG vaccination in humans inhibits systemic inflammation in a sex-dependent manner. J Clin Invest. 2012, 122, 4692–4694. [Google Scholar] [CrossRef]
- Ristori, G.; Buzzi, M.G.; Sabatini, U.; et al. Research progress on specific and non-specific immune effects of BCG and the possibility of BCG protection against COVID-19. Front Immunol. 2023, 14, 1118378. [Google Scholar] [CrossRef]
- Yamamoto, K.; Torigoe, T.; Saito, M.; et al. Culture filtrate proteins from BCG act as adjuvants for cytotoxic T lymphocyte induction. Front Immunol. 2023, 14, 1271228. [Google Scholar] [CrossRef]
- Freudenstein, O.; Tak, T.; Biesma, D.; et al. Beyond tuberculosis: the surprising immunological benefits of the Bacillus Calmette-Guérin vaccine in infectious, auto-immune, and inflammatory diseases. Pathogens 2025, 14, 196. [Google Scholar] [CrossRef]
- Brewer, T.F.; Colditz, G.A. Relationship between Bacille Calmette-Guérin (BCG) strains and the efficacy of BCG vaccine in the prevention of tuberculosis. Clin Infect Dis. 1995, 20, 126–135. [Google Scholar] [CrossRef]
- Notarangelo, L.D. Primary immunodeficiencies. J Allergy Clin Immunol. 2010, 125, S182–S194. [Google Scholar] [CrossRef]
- Fischer, A.; Notarangelo, L.D.; Neven, B.; et al. Severe combined immunodeficiencies and related disorders. Nat Rev Dis Primers 2015, 1, 15061. [Google Scholar] [CrossRef] [PubMed]
- Immune adjuvant therapy using Bacillus Calmette-Guérin cell wall skeleton (BCG-CWS) in advanced malignancies: a phase 1 study of safety and immunogenicity assessments. Medicine (Baltimore) 2022, 101, e29810. [CrossRef]
- Intralesional immunotherapy for non-genital viral warts: a review of current evidence and future perspectives. Int J Mol Sci. 2025, 26, 5644. [CrossRef]
- Sharma, V.K.; Sahni, K.; Wadhwani, A.R. Intralesional immunotherapy with killed Mycobacterium indicus pranii vaccine for the treatment of extensive cutaneous warts. Indian J Dermatol Venereol Leprol. 2015, 81, 419–422. [Google Scholar] [CrossRef]
- Gupta, S.; Malhotra, A.K.; Verma, K.K.; et al. Mycobacterium indicus pranii and Mycobacterium bovis BCG lead to differential macrophage activation in toll-like receptor-dependent manner. Immunology 2012, 143, 258–268. [Google Scholar] [CrossRef]
- Khan, T.; Ali, M.; Khan, A.N. Managing recalcitrant warts: facts about Bacillus Calmette-Guérin (BCG), Mycobacterium indicus pranii (Mw vaccine), and purified protein derivative (PPD) as immunotherapy. J Cosmet Dermatol Sci Appl. 2018, 8, 230–247. [Google Scholar] [CrossRef]
- Suresh, R.; Padmavathi, Y. An intention-to-treat-analysis of the efficacy of immunotherapy using Mycobacterium w vaccine and purified protein derivative of tuberculin for warts with assessment of improvement in quality of life. Dermatol Pract Concept. 2022, 12, e2022068. [Google Scholar] [CrossRef]
- Berendsen, M.L.T.; Øland, C.B.; Bles, P.; et al. Maternal priming: Bacillus Calmette-Guérin (BCG) vaccine scarring in mothers enhances the survival of their child with a BCG vaccine scar. J Pediatr Infect Dis Soc. 2020, 9, 166–172. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




