Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Study Design and Participants
2.2. Data Collection
2.2.1. Blood Sample Collection and Processing
2.2.2. Determination of Common Inflammatory Blood Indices
2.2.3. Determination of Immune Cell-Based Blood Inflammatory Indices
2.3. Statistical Analysis
3. Results
3.1. Baseline Demographic and Clinical Characteristics of COVID-19 Subjects on Admission
3.2. Sex-Related Differences in Inflammatory-Immune Blood Indices During COVID-19 Development
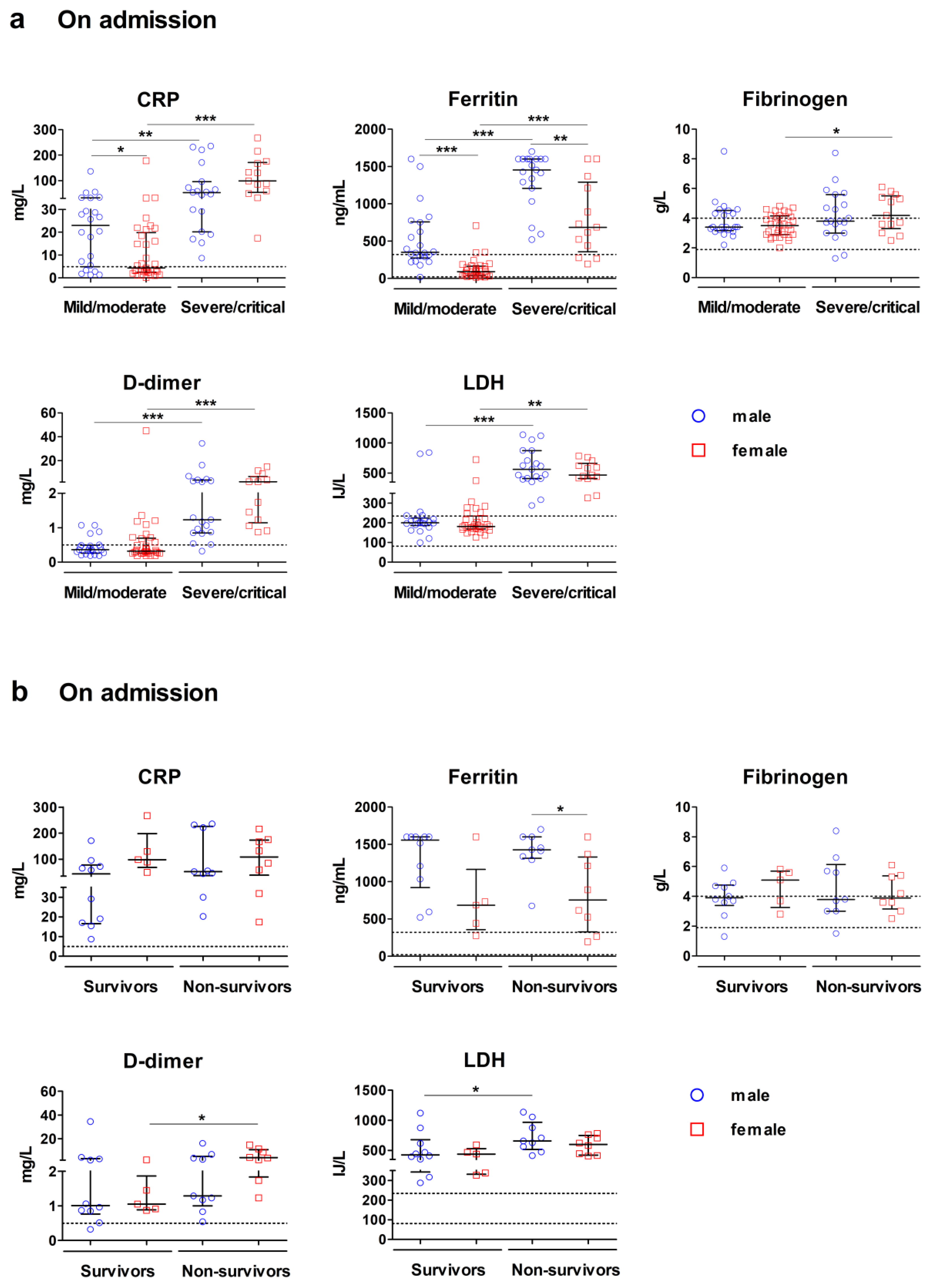
3.2.1. Sex-Related Differences in Blood Levels of Acute-Phase Proteins in Covid-19 on Admission
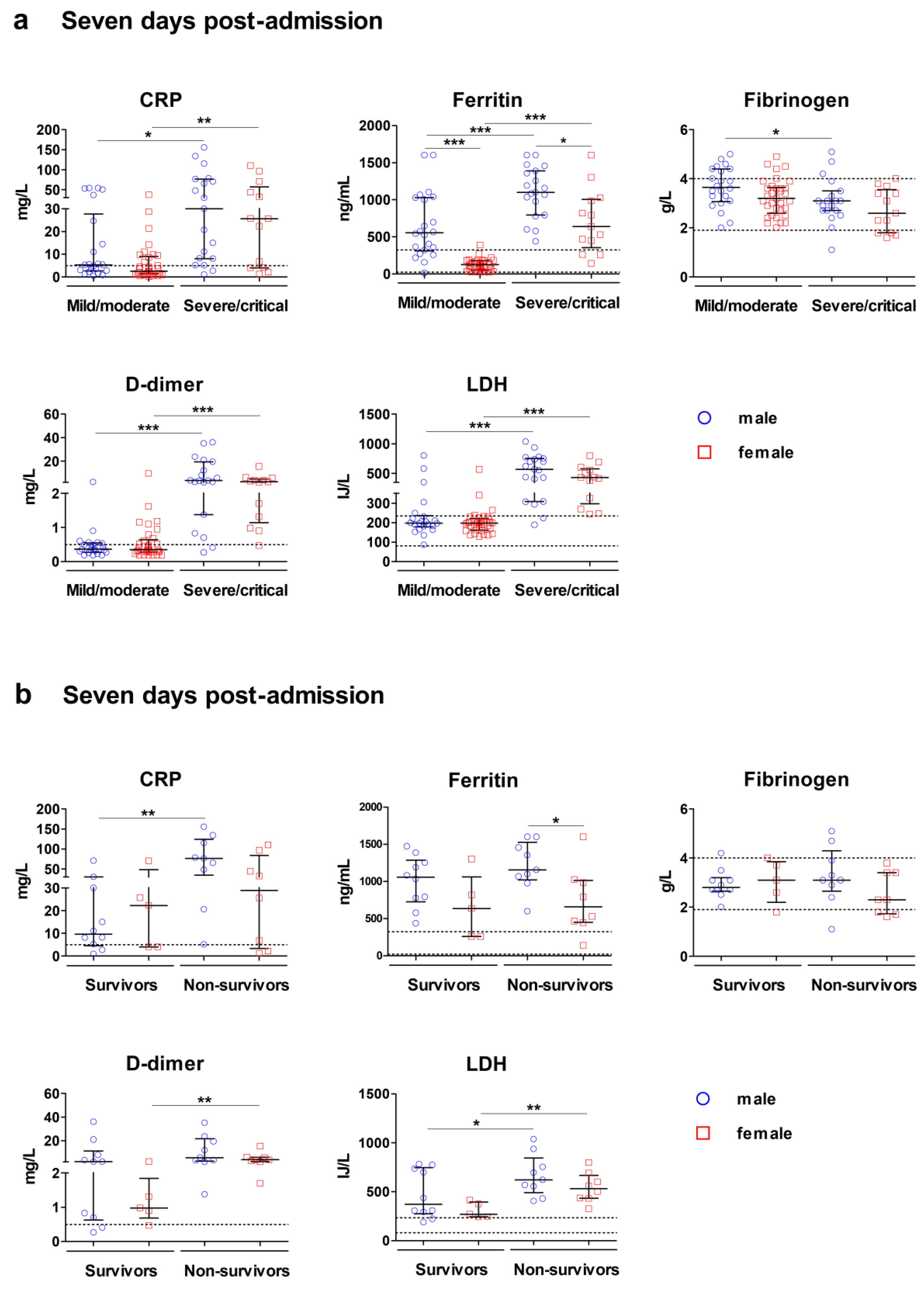
Seven Days Post-Admission
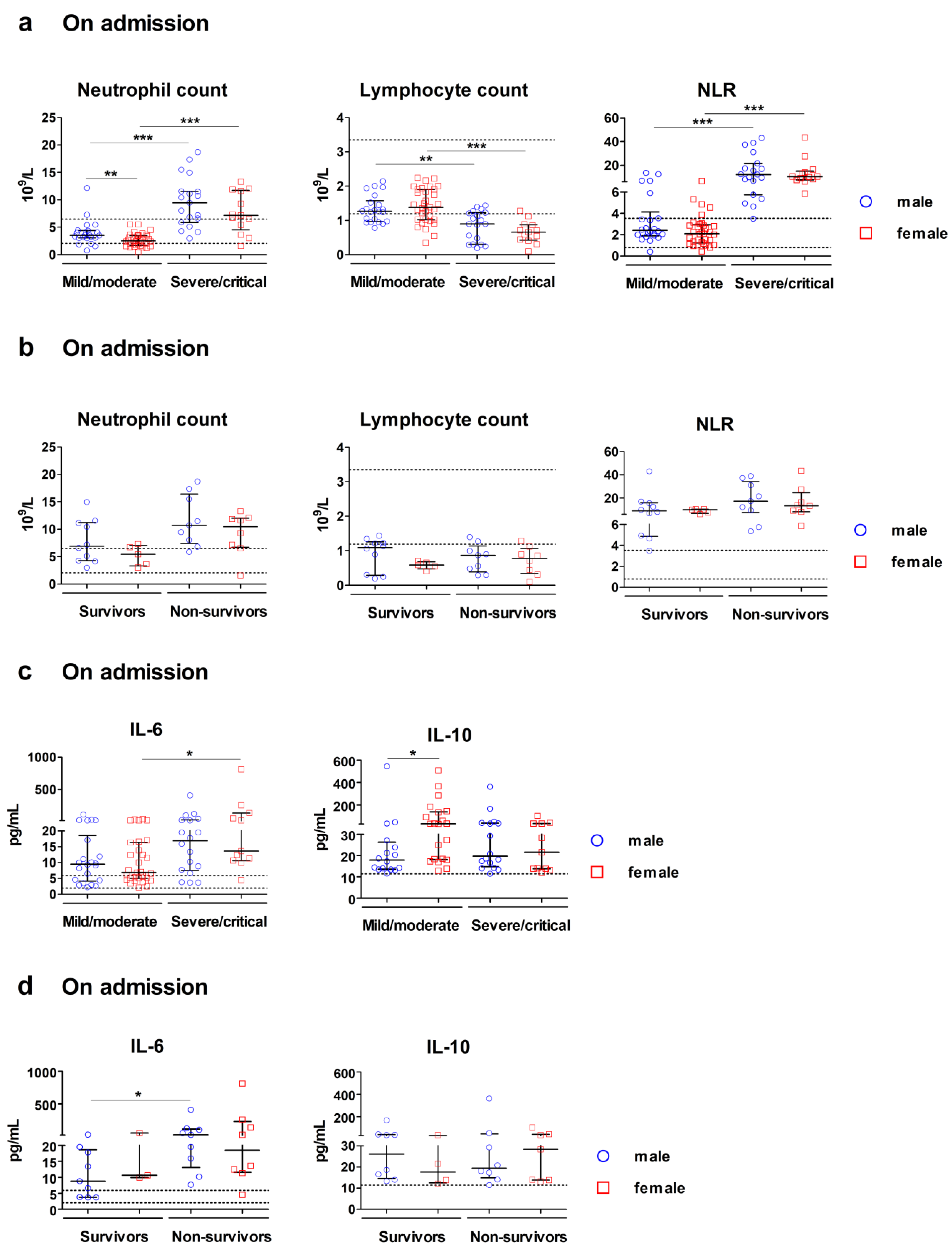
3.2.2. Sex-Related Differences in Blood Levels of Immune Cell-Related Indices in COVID-19 on Admission
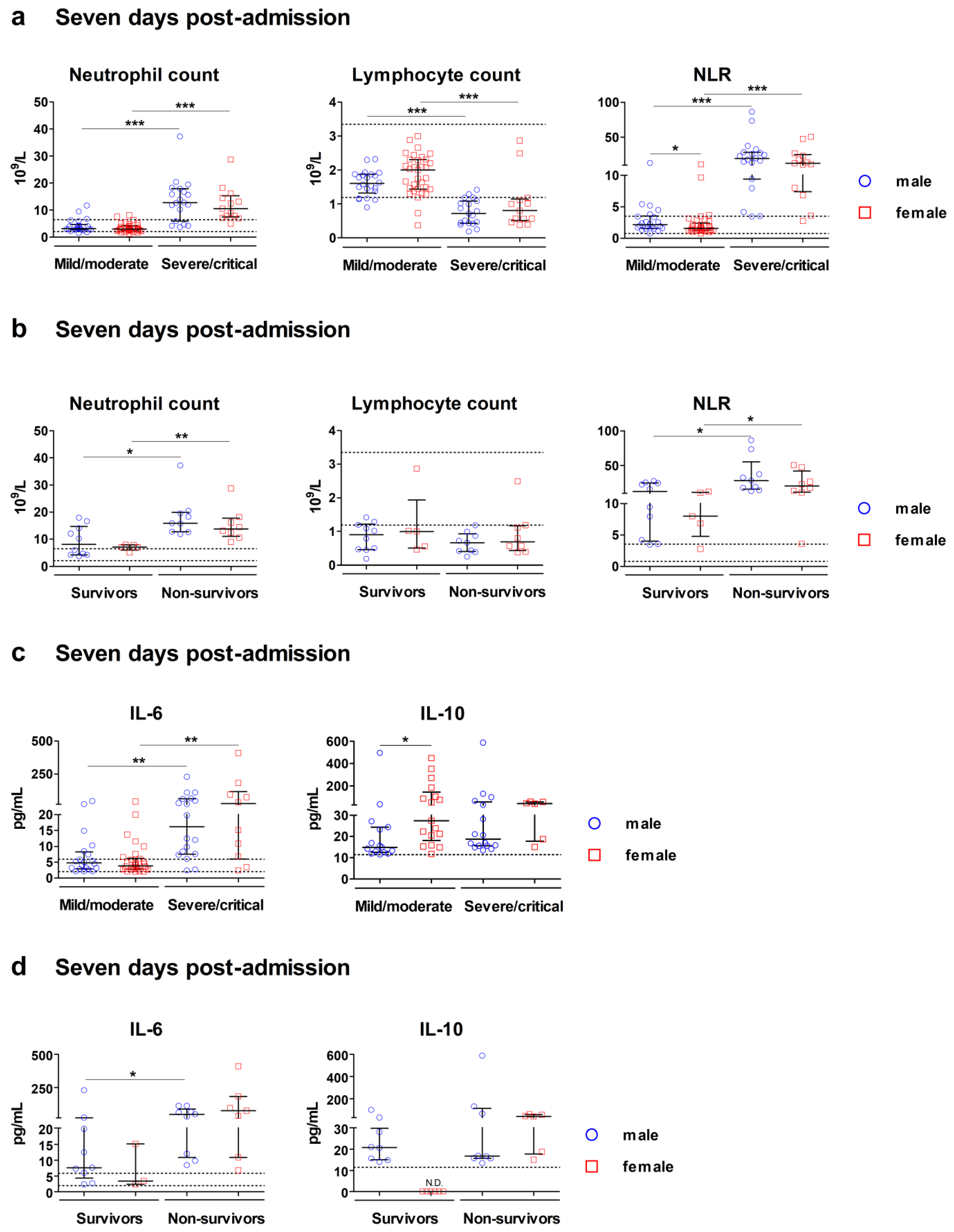
Seven Days Post-Admission
3.3. Evaluation of Inflammatory-Immune Blood Indices for Their Capacity to Predict Severe/Critical Disease During COVID-19 Early Development in Males and Females
3.3.1. Evaluation of Acute-Phase Proteins as Predictors of the Severe/Critical Disease
Seven Days Post-Admission
3.3.2. Evaluation of Immune Cell-Related Blood Indices as Predictors on Admission
Seven Days Post-Admission
3.4. Evaluation of Inflammatory-Immune Blood Indices for Their Capacity to Predict COVID-19 Death Outcome During Early Disease Development in Males and Females
3.4.1. On Admission
3.4.2. Seven Days Post-Admission
Acute-Phase Proteins as Predictors
Immune Cell-Related Blood Indices as Predictors
4. Disscussion
Sex Specificity in Predictive Capacity of Acute-Phase Proteins
Sex Specificity in Predictive Capacity of Immune Cell-Related Blood Indices
Strengths and Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thevarajan I, Buising KL, Cowie BC. Clinical presentation and management of COVID-19. Med J Aust. 2020;213(3):134-139. [CrossRef]
- Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130(5):2620-2629. [CrossRef]
- World Health Organization. Listing of WHO’s response to COVID-19 [Internet]. Geneva: World Health Organization; 2020 [updated 2020 Jun 29; cited 2026 Apr 4]. Available from: https://www.who.int/news/item/29-06-2020-covidtimeline.
- UN News. WHO chief declares end to COVID-19 as a global health emergency [Internet]. New York: United Nations; 2023 [released 2023 May 5; cited 2026 Apr 4]. Available from: https://news.un.org/en/story/2023/05/1136367.
- Roknuzzaman A, Sarker R, Nazmunnahar, Shahriar M, Al Mosharrafa R, Islam MR. The WHO has Declared COVID-19 is No Longer a Pandemic-Level Threat: A Perspective Evaluating Potential Public Health Impacts. Clin Pathol. 2024;17:2632010X241228053. [CrossRef]
- KFF. Global COVID-19 Tracker [Internet]. San Francisco: KFF; 2024 [updated 2024 Mar 20; cited 2026 Apr 4]. Available from: https://www.kff.org/covid-19/global-covid-19-tracker/.
- Markov PV, Katzourakis A, Stilianakis NI. Antigenic evolution will lead to new SARS-CoV-2 variants with unpredictable severity. Nat Rev Microbiol. 2022;20(5):251-252. [CrossRef]
- Lu J, Hu S, Fan R, Liu Z, Yin X, Wang Q, et al. ACP risk grade: a simple mortality index for patients with confirmed or suspected severe acute respiratory syndrome coronavirus 2 disease (COVID-19) during the early stage of outbreak in Wuhan in China. medRxiv [Preprint]. 2020. [CrossRef]
- Liu J, Liu Y, Xiang P, Pu L, Xiong H, Li C, et al. Neutrophil-to-lymphocyte ratio predicts critical illness patients with 2019 coronavirus disease in the early stage. J Transl Med. 2020;18(1):206. [CrossRef]
- Asghar MS, Kazmi SJH, Khan NA, Akram M, Hassan M, Rasheed U, et al. Poor prognostic biochemical markers predicting fatalities caused by COVID-19: A retrospective observational study from a developing country. Cureus. 2020;12(8):e9575. [CrossRef]
- Kumar A, Yendamuri S, Ahmad F, Mukherjee PB, Kumar R, Manrai M, et al. Inflammatory biomarkers and adverse outcome in COVID-19: Prelude for future viral pandemics. J Family Med Prim Care. 2025;14(2):720-728. [CrossRef]
- Wang D, Li R, Wang J, Jiang Q, Gao C, Yang J, et al. Correlation analysis between disease severity and clinical and biochemical characteristics of 143 cases of COVID-19 in Wuhan, China: a descriptive study. BMC Infect Dis. 2020;20(1):519. [CrossRef]
- Li Y, Hu Y, Yu J, Ma T. Retrospective analysis of laboratory testing in 54 patients with severe-or critical-type 2019 novel coronavirus pneumonia. Lab Invest. 2020;100:1-7. [CrossRef]
- Kadhim AS, Abdullah YJ. Serum levels of interleukin-6, ferritin, C-reactive protein, lactate dehydrogenase, D-dimer, and count of lymphocytes and neutrophils in COVID-19 patients: Its correlation to the disease severity. Biomed Biotechnol Res J. 2021;5(1):69-73. [CrossRef]
- Pal S, Sengupta S, Lahiri S, Ghosh A, Bhowmick K. Role of biomarkers in prognostication of moderate and severe COVID-19 cases. J Family Med Prim Care. 2023;12(12):3186-3193. [CrossRef]
- Taha SI, Fouad SH, El-Sehsah EM, Mohamed MM, Omran A, Hamdy M, et al. Role of immune-inflammatory biomarkers and their derived ratio in predicting COVID-19 severity and mortality. Sci Rep. 2025;15(1):39003. [CrossRef]
- Shenoy MT, Mohanty PK, Suganthy K, Manavalan JK, Hariharan AL. Utility of Biochemical Markers in Predicting Severe COVID-19: Experience from a Tertiary Hospital in South India. EJIFCC. 2022;33(2):131-144.NEMA DOI.
- Grasselli G, Greco M, Zanella A, Albano G, Antonelli M, Bellani G, et al. Risk factors associated with mortality among patients with COVID-19 in intensive care units in Lombardy, Italy. JAMA Intern Med. 2020;180(10):1345-1355. [CrossRef]
- Petrilli CM, Jones SA, Yang J, Rajagopalan H, O'Donnell L, Chernyak Y, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. [CrossRef]
- Gupta S, Hayek SS, Wang W, Chan L, Mathews KS, Melamed ML, et al. Factors associated with death in critically ill patients with coronavirus disease 2019 in the US. JAMA Intern Med. 2020;180(11):1436-1447. [CrossRef]
- World Health Organization. Diagnostic testing for SARS-CoV-2: interim guidance [Internet]. Geneva: World Health Organization; 2020 [released 2020 Sep 11, cited 2026 Apr 4]. Available from: https://iris.who.int/server/api/core/bitstreams/7caa3b48-f99b-4cc2-b0f3-a954da4060d7/content.
- National Institutes of Health. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines [Internet]. Bethesda (MD): National Institutes of Health; 2021 [updated 2024 Feb 29; cited 2026 Apr 4]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK570371/pdf/Bookshelf_NBK570371.pdfz.
- Mendoza-Hernandez MA, Hernandez-Fuentes GA, Sanchez-Ramirez CA, Rojas-Larios F, Guzman-Esquivel J, Rodriguez-Sanchez IP, et al. Time-dependent ROC curve analysis to determine the predictive capacity of seven clinical scales for mortality in patients with COVID-19: Study of a hospital cohort with very high mortality. Biomed Rep. 2024;20(6):100. [CrossRef]
- Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062. [CrossRef]
- World Health Organization. Clinical management of COVID-19 [Internet]. Geneva: World Health Organization; 2020 [updated 2025 Jun; cited 2026 Apr 4]. Available from: https://www.who.int/teams/health-care-readiness/covid-19.
- Cruciata A, Volpicelli L, Di Bari S, Iaiani G, Cirillo B, Pugliese F et al. Risk of Seven-Day Worsening and Death: A New Clinically Derived COVID-19 Score. Viruses. 2022;14(3):642. [CrossRef]
- Han LL, Wang YX, Li J, Zhang XL, Bian C, Wang H, et al. Gender differences in associations of serum ferritin and diabetes, metabolic syndrome, and obesity in the China Health and Nutrition Survey. Mol Nutr Food Res. 2014;58(11):2189-2195. [CrossRef]
- Hastak P, Cromer D, Malycha J, Andersen CR, Raith EM, Davenport MP, et al. Defining the correlates of lymphopenia and independent predictors of poor clinical outcome in adults hospitalized with COVID-19 in Australia. Sci Rep. 2024;14(1):11102. [CrossRef]
- Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, et al. Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis. 2020;71(15):762-768. [CrossRef]
- Sun S, Cai X, Wang H, He G, Lin Y, Lu B, et al. Abnormalities of Peripheral Blood System in Patients with COVID-19 in Wenzhou, China. Clin Chim Acta. 2020;507:174-180. [CrossRef]
- Ulloque-Badaracco JR, Salas-Tello WI, Al-kassab-Cordova A, Alarcon-Braga EA, Benites-Zapata VA, Maguina JL, et al. Prognostic value of neutrophil-to-lymphocyte ratio in COVID-19 patients: A systematic review and meta-analysis. Int J Clin Pract. 2021;75(11):e14596. [CrossRef]
- El Azhary K, Ghazi B, Kouhen F, El Bakkouri J, Chamlal H, El Ghanmi A, et al. Clinical Impact of Neutrophil Variation on COVID-19 Complications. Diagnostics (Basel). 2025;15(4):457. [CrossRef]
- Han H, Ma Q, Li C, Liu R, Zhao L, Wang W, et al. Profiling serum cytokines in COVID-19 patients reveals IL-6 and IL-10 are disease severity predictors. Emerg Microbes Infect. 2020;9(1):1123-1130. [CrossRef]
- Azaiz MB, Jemaa AB, Sellami W, Romdhani C, Ouslati R, Gharsallah H, et al. Deciphering the balance of IL-6/IL-10 cytokines in severe to critical COVID-19 patients. Immunobiology. 2022;227(4):152236. [CrossRef]
- Krivosova M, Hanusrichterova JU, Lucansky V, Samec M, Bobcakova A, Baranovicova E, et al. Comparative Study of Cytokine Profiles in SARS-CoV-2 Delta and Omicron Variants. Bratisl Med J. 2025;126:286-298. [CrossRef]
- Maaß H, Ynga-Durand M, Milosevic M, Krstanovic F, Matešić MP, Zuža I, et al. Serum cytokine dysregulation signatures associated with COVID-19 outcomes in high mortality intensive care unit cohorts across pandemic waves and variants. Sci Rep. 2024;14(1):13605. [CrossRef]
- Nguyen CV, Luong CQ, Dao CX, Nguyen MH, Pham DT, Khuat NH, et al. Predictive validity of interleukin 6 (IL-6) for the mortality in critically ill COVID-19 patients with the B.1.617.2 (Delta) variant in Vietnam: a single-centre, cross-sectional study. BMJ Open. 2024;14(12):e085971. [CrossRef]
- Smail SW, Babaei E, Amin K, Abdulahad WH. Serum IL-23, IL-10, and TNF-α predict in-hospital mortality in COVID-19 patients. Front Immunol. 2023;14:1145840. [CrossRef]
- Corbacioglu SK, Aksel G. Receiver operating characteristic curve analysis in diagnostic accuracy studies: A guide to interpreting the area under the curve value. Turk J Emerg Med. 2023;23(4):195-198. [CrossRef]
- Shultz EK. Multivariate Receiver-Operating Characteristic Curve Analysis: Prostate cancer screening as example. Clin Chem. 1995;41(8 Pt 2):1248-1255.
- Pederson JR. Multiple Regression. In: Allen M, editor. The SAGE Encyclopedia of Communication Research Methods. Thousand Oaks: SAGE Publications, Inc. 2017; p. 1041-1052. [CrossRef]
- Canon-Estrada JA, Munoz-Ordonez MA, Escalante-Forero M, Rodas Y, Arteaga-Tobar AA, Azcarate-Rodriguez V, et al. Biochemical differences based on sex and clusters of biomarkers in patients with COVID-19: analysis from the CARDIO COVID 19–20 registry. BMC Cardiovasc Disord. 2025;25:147. [CrossRef]
- Gandini O, Criniti A, Ballesio L, Giglio S, Galardo G, Gianni W, et al. Serum Ferritin is an independent risk factor for Acute Respiratory Distress Syndrome in COVID-19. J Infect. 2020;81(6):979-997. [CrossRef]
- Gandini O, Criniti A, Gagliardi MCG, Ballesio L, Giglio S, Balena A, et al. Sex-disaggregated data confirm serum ferritin as an independent predictor of disease severity both in male and female COVID-19 patients. J Infect. 2021;82(3):414-451. [CrossRef]
- Liu T, Zhang J, Yang Y, Ma H, Li Z, Zhang J, et al. The role of interleukin-6 in monitoring severe case of coronavirus disease 2019. EMBO Mol Med. 2020;12(7):e12421. [CrossRef]
- Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033-1034. [CrossRef]
- Garcia-Casal MN, Pasricha SR, Martinez RX, Lopez-Perez L, Pena-Rosas JP. Serum or plasma ferritin concentration as an index of iron deficiency and overload. Cochrane Database Syst Rev. 2021;5(5):CD010876. [CrossRef]
- Badenhorst CE, Forsyth AK, Govus AD. A contemporary understanding of iron metabolism in active premenopausal females. Front Sports Act Living. 2022;4:903937. [CrossRef]
- Beggs LA, Yarrow JF, Conover CF, Meuleman JR, Beck DT, Morrow M, et al. Testosterone alters iron metabolism and stimulates red blood cell production independently of dihydrotestosterone. Am J Physiol Endocrinol Metab. 2014;307(5):E456-E461. [CrossRef]
- Qi S, Ngwa C, Morales Scheihing DA, Al Mamun A, Ahnstedt HW, Finger CE, et al. Sex differences in the immune response to acute COVID-19 respiratory tract infection. Biol Sex Differ. 2021;12(1):66. [CrossRef]
- Vadakedath S, Kandi V, Mohapatra RK, Pinnelli VBK, Yegurla RR, Shahapur PR, et al. Immunological aspects and gender bias during respiratory viral infections including novel Coronavirus disease-19 (COVID-19): A scoping review. J Med Virol. 2021;93(9):5295-5309. [CrossRef]
- Koyou HL, Salleh MN, Jelemie CS, Badrin MJQ, Prastiyanto ME, Ramachandran V. TMPRSS2: A Key Host Factor in SARS-CoV-2 Infection and Potential Therapeutic Target. Medeni Med J. 2025;40(2):101-109. [CrossRef]
- Tremellen K, McPhee N, Pearce K, Benson S, Schedlowski M, Engler H. Endotoxin-initiated inflammation reduces testosterone production in men of reproductive age. Am J Physiol Endocrinol Metab. 2018;314(3):E206-E213. [CrossRef]
- Herrington DM, Brosnihan KB, Pusser BE, Seely EW, Ridker PM, Rifai N, et al. Differential effects of E and raloxifene on C-reactive protein and other markers of inflammation in healthy postmenopausal women. J Clin Endocrinol Metab. 2001;86(9):4216-4222. [CrossRef]
- Son DS, Roby KF. Interleukin-1alpha-induced chemokines in mouse granulosa cells: impact on keratinocyte chemo-attractant chemokine, a CXC subfamily. Mol Endocrinol. 2006;20(11):2999-3013. [CrossRef]
- Liu Z, Ye F, Zhang H, Gao Y, Tan A, Zhang S, et al. The Association between the Levels of Serum Ferritin and Sex Hormones in a Large Scale of Chinese Male Population. PLoS One. 2013;8(10):e75908. [CrossRef]
- Chao KC, Chang CC, Chiou HY, Chang JS. Serum Ferritin Is Inversely Correlated with Testosterone in Boys and Young Male Adolescents: A Cross-Sectional Study in Taiwan. PLoS One. 2015;10(12):e0144238. [CrossRef]
- Farhan SS, Mahgoub SS, Hussain SA. Effects of progesterone and estradiol on the inflammatory and apoptotic markers of ovariectomized rats challenged with acute septic systemic inflammation. J Appl Pharm Sci. 2019;9(12):103-107. [CrossRef]
- Fabiao J, Sassi B, Pedrollo EF, Gerchman F, Kramer CK, Leitao CB, et al. Why do men have worse COVID-19-related outcomes? Braz J Med Biol Res. 2022;55:e11711. [CrossRef]
- Alam S, Pinkhasov O, Seckin S, Muneyyirci-Delale O. Why are men more severely affected by COVID-19? J Allergy Infect Dis. 2022;3(1):10-16. [CrossRef]
- Kernan KF, Carcillo JA. Hyperferritinemia and inflammation. Int Immunol. 2017;29(9):401-409. [CrossRef]
- Jahan H, Yeasmin T, Roy M. Elevated D-dimers associated with severity of COVID-19: A systematic review and meta-analysis. Med Res Arch. 2022;10(7). [CrossRef]
- Minutti-Zanella C, Gallardo-Perez MM, Ruiz-Arguelles GJ. D-dimer in Coronavirus 2019: An Acute Phase Reactant? Semin Thromb Hemost. 2024;50(2):295-297. [CrossRef]
- Stephen IR, Suman FR, Balasubramanian J, Shanmugam SG, Man R. D-dimer as a Predictor of ICU Admission and Mortality in COVID-19 Patients: Insights From a Two-Year Retrospective Study From a Tertiary Care Center in South India. Cureus. 2024;16(10):e70682. [CrossRef]
- Lowe GD. Fibrin D-dimer and cardiovascular risk. Semin Vasc Med. 2005;5(4):387-398. [CrossRef]
- Hunt BJ, Levi M. Re: The source of elevated plasma D-dimer levels in COVID-19 infection. Br J Haematol. 2020;190(3):e133-e134. [CrossRef]
- Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135(23):2033-2040. [CrossRef]
- Haase C, Joergensen M, Ellervik C, Joergensen MK, Bathum L. Age and sex-dependent reference intervals for D-dimer: evidence for a marked increase by age. Thromb Res. 2013;132(6):676-680. [CrossRef]
- Legnani C, Cini M, Cosmi B, Carraro P, Tripodi A, Erba N, et al. Age and gender specific cut-off values to improve the performance of D-dimer assays to predict the risk of venous thromboembolism recurrence. Intern Emerg Med. 2013;8(3):229-236. [CrossRef]
- Selcuk M, Cinar T, Gunay N, Keskin M, Cicek V, Kilic S, et al. Comparison of D-dimer level measured on the third day of hospitalization with admission D-dimer level in predicting in-hospital mortality in COVID-19 patients. Medeni Med J. 2021;36(1):1-6. [CrossRef]
- Mukhopadhyay A, Talmor N, Xia Y, Berger JS, Iturrate E, Adhikari S, et al. Sex differences in the prognostic value of troponin and D-dimer in COVID-19 illness. Heart Lung. 2023;58:1-5. [CrossRef]
- Mitra P, Guha D, Nag SS, Mondal BC, Dasgupta S. Role of Plasma Fibrinogen in Diagnosis and Prediction of Short Term Outcome in Neonatal Sepsis. Indian J Hematol Blood Transfus. 2017;33(2):195-199. [CrossRef]
- Mori K, Tsujita Y, Yamane T, Eguchi Y. Decreasing Plasma Fibrinogen Levels in the Intensive Care Unit Are Associated with High Mortality Rates In Patients With Sepsis-Induced Coagulopathy. Clin Appl Thromb Hemost. 2022;28:10760296221101386. [CrossRef]
- Feng Y, Xiong Y, Qiao T, Li X, Jia L, Han Y. Lactate dehydrogenase A: A key player in carcinogenesis and potential target in cancer therapy. Cancer Med. 2018;7(12):6124-6136. [CrossRef]
- Qin L, Li X, Shi J, Yu M, Wang K, Tao Y, et al. Gendered effects on inflammation reaction and outcome of COVID-19 patients in Wuhan. J Med Virol. 2020;92(11):2684-2692. [CrossRef]
- Sheriff A, Kayser S, Brunner P, Vogt B. C-Reactive Protein Triggers Cell Death in Ischemic Cells. Front Immunol. 2021;12:630430. [CrossRef]
- Zhao Q, Meng M, Kumar R, Wu Y, Huang J, Deng Y, et al. Lymphopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A systemic review and meta-analysis. Int J Infect Dis. 2020;96:131-135. [CrossRef]
- Fei J, Fu L, Li Y, Xiang HX, Xiang Y, Li MD, et al. Reduction of lymphocyte count at early stage elevates severity and death risk of COVID-19 patients: a hospital-based case-cohort study. Arch Med Sci. 2023;19(5):1303-1313. [CrossRef]
- Chan AS, Rout A. Use of neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios in COVID-19. J Clin Med Res. 2020;12(7):448-453. [CrossRef]
- McKenna E, Wubben R, Isaza-Correa JM, Melo AM, Mhaonaigh AU, Conlon N, et al. Neutrophils in COVID-19: Not innocent bystanders. Front Immunol. 2022;13:864387. [CrossRef]
- Hammad MO, Alseoudy MM. The sex-related discrepancy in laboratory parameters of severe COVID-19 patients with diabetes: A retrospective cohort study. Prim Care Diabetes. 2021;15(4):713-718. [CrossRef]
- Pan Q, Zhang W, Li X, Chen Z, Yang Y, Wang G. Sex Difference in the Association Between Neutrophil to Lymphocyte Ratio and Severity of Coronary Artery Disease. Angiology. 2022;73(5):470-477. [CrossRef]
- Lombardi CM, Specchia C, Conforti F, La Rovere MT, Carubelli V, Agostoni P, et al. Sex-related differences in patients with coronavirus disease 2019: results of the Cardio-COVID-Italy multicentre study. J Cardiovasc Med (Hagerstown). 2022;23(4):254-263. [CrossRef]
- Forsblom E, Helanne H, Kortela E, Silén S, Meretoja A, Järvinen A. Inflammation parameters predict fatal outcome in male COVID-19 patients in a low case-fatality area - a population-based registry study. Infect Dis (Lond). 2022;54(8):558-571. [CrossRef]
- Takahashi T, Ellingson MK, Wong P, Israelow B, Lucas C, Klein J, et al. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature. 2020;588(7837):315-320. [CrossRef]
- Montano L, Donato F, Bianco PM, Lettieri G, Guglielmino A, Motta O, et al. Air pollution and COVID-19: a possible dangerous synergy for male fertility. Int J Environ Res Public Health. 2021;18(13):6846. [CrossRef]
- Montano L, Donato F, Bianco PM, Motta O, Bonapace IM, Piscopo M. Semen quality as a potential susceptibility indicator to SARS-CoV-2 insults in polluted areas. Environ Sci Pollut Res Int. 2021;28(28):37031-37040. [CrossRef]
- Takahashi T, Iwasaki A. Sex differences in immune responses. Science. 2021;371(6527):347-348. [CrossRef]
- Ceolin C, Liberati V, Acunto V, Vergadoro M, Simonato C, Cazzavillan S, et al. Neutrophil-to-lymphocyte ratio as a sex-specific predictor of short-term mortality in hospitalised older adults with COVID-19: a pragmatic biomarker of inflammaging in acute vulnerability. Immun Ageing. 2025;22(1):55. [CrossRef]
- Calabrese F, Pezzuto F, Fortarezza F, Hofman P, Kern I, Panizo A, et al. Pulmonary Pathology and COVID-19: Lessons From Autopsy. The Experience of European Pulmonary Pathologists. Virchows Arch. 2020;477(3):359-372. [CrossRef]
- Zuo Y, Zuo M, Yalavarthi S, Gockman K, Madison J, Shi H, et al. Neutrophil Extracellular Traps and Thrombosis in COVID-19. J Thromb Thrombolysis. 2021;51(2):446-453. [CrossRef]
| Male | Female | |||
| Univariate Logistic Regression | Multivariable Logistic Regression |
Univariate Logistic Regression |
Multivariable Logistic Regression |
|
| Inflammatory blood indices | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) |
| On admission | ||||
| CRP | 1.284 (1.010 - 1.634) p ≤ 0.05 |
- | 1.482 (1.153 - 1.905) p ≤ 0.01 |
- |
| Ferritin | 1.038 (1.018 - 1.030) p ≤ 0.001 |
1.030 (1.007 - 1.053) p ≤ 0.05 |
1.093 (1.034 - 1.155) p ≤ 0.01 |
1.100 (1.023 - 1.255) p ≤ 0.05 |
| Fibrinogen | - | - | 2.626 (1.224 - 5.633) p ≤ 0.05 |
- |
| D-dimer | 1.537 (1.158 - 2.038) p ≤ 0.01 |
1.392 (1.025 - 1.891) p ≤ 0.05 |
1.153 (1.030 - 1.292) p ≤ 0.05 |
1.136 (1.027 - 1.255) p ≤ 0.05 |
| LDH | 1.078 (1.028 - 1.130) p ≤ 0.01 |
- | 1.163 (1.063 - 1.272) p ≤ 0.001 |
- |
| Seven days post-admission | ||||
| CRP | 1.330 (1.039 - 1.704) p ≤ 0.05 |
- | 2.246 (1.216 - 4.146) p ≤ 0.01 |
- |
| Ferritin | 1.027 (1.009 - 1.046) p ≤ 0.01 |
1.047 (1.004 - 1.091) p ≤ 0.05 |
1.171 (1.048 - 1.309) p ≤ 0.01 |
1.154 (1.003 - 1.326) p ≤ 0.05 |
| Fibrinogen | 0.464 (0.210 - 1.024) p > 0.05 |
0.095 (0.010 - 0.867) p ≤ 0.05 |
- | - |
| D-dimer | 1.217 (1.052 - 1.409) p ≤ 0.01 |
1.354 (1.058 - 1.733) p ≤ 0.05 | 1.067 (1.010 - 1.126) p ≤ 0.05 |
- |
| LDH | 1.077 (1.030 - 1.127) p ≤ 0.001 |
- | 1.176 (1.066 - 1.297) p ≤ 0.001 |
1.121 (0.995 - 1.263) p > 0.05 |
| Male | Female | |||||||||
| Inflammatory blood indices | AUC (95% CI) |
Sensitivity (%) | Specificity (%) | Cut-off | J* | AUC (95% CI) |
Sensitivity (%) | Specificity (%) | Cut-off | J* |
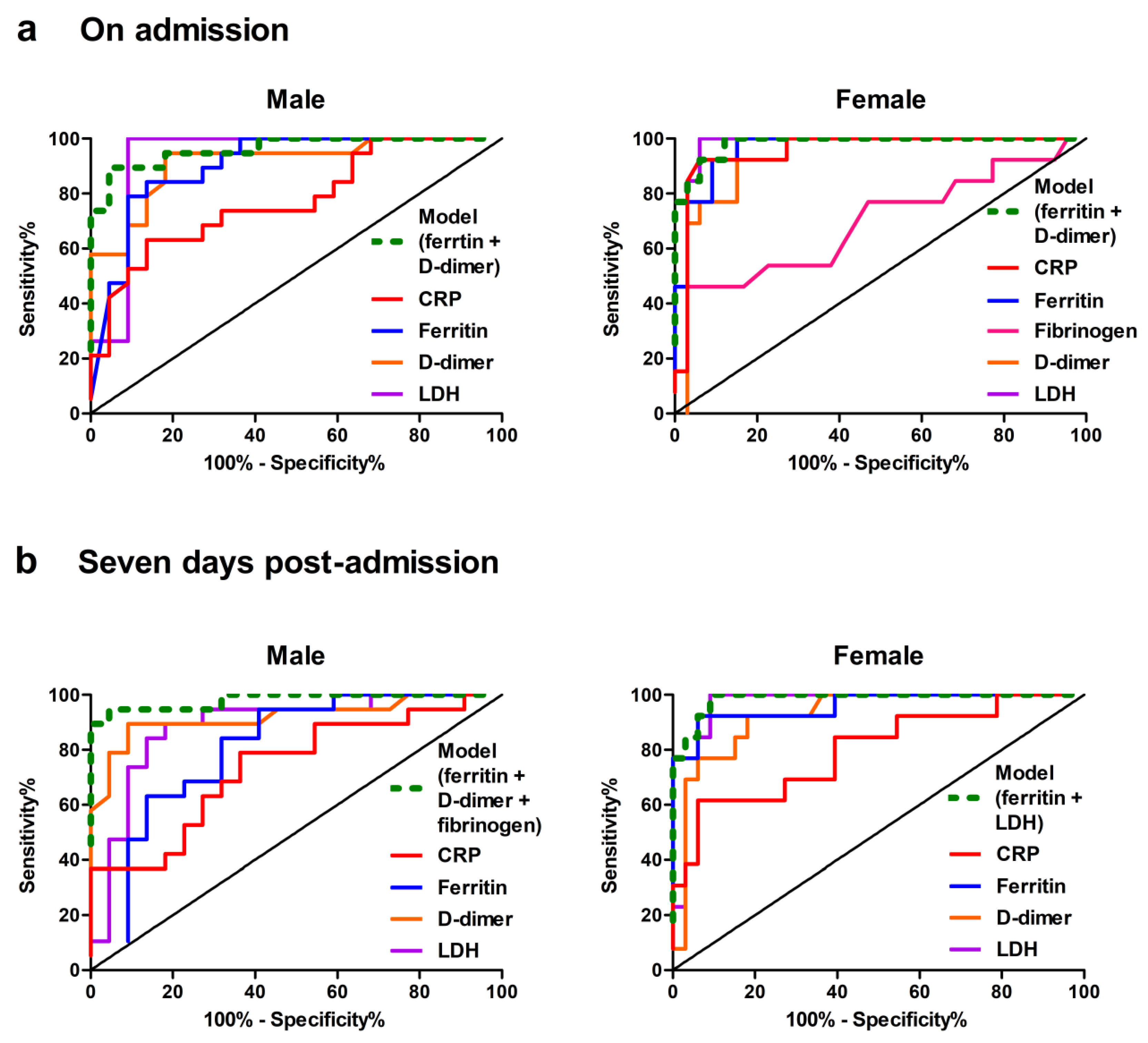
| On admission | ||||||||||
| Model (ferritin + D-dimer) | 0.962 (0.909 - 1) p ≤ 0.001 |
89.5 | 95.5 | - | 0.849 | 0.984 (0.956 - 1) p ≤ 0.001 |
100 | 87.9 | - | 0.879 |
| CRP (mg/L) | 0.774 (0.630 - 0.918) p ≤ 0.01 |
63.2 | 86.4 | 38.7 | 0.495 | 0.955 (0.892 - 1) p ≤ 0.001 |
92.3 | 93.9 | 31.3 | 0.862 |
| Ferritin (ng/mL) | 0.903 (0.809 - 0.997) p ≤ 0.001 |
84.2 | 86.4 | 929 | 0.706 | 0.965 (0.919 - 1) p ≤ 0.001 |
100 | 84.8 | 191 | 0.848 |
| Fibrinogen (g/L) | - | - | - | - | - | 0.696 (0.504 - 0.887) p ≤ 0.05 |
46.2 | 100 | 4.95 | 0.462 |
| D-dimer (mg/L) | 0.914 (0.825 - 1) p ≤ 0.001 |
94.7 | 81.8 | 0.505 | 0.766 | 0.946 (0.885 - 1) p ≤ 0.001 |
100 | 84.8 | 0.84 | 0.848 |
| LDH (IJ/L) | 0.933 (0.842 - 1) p ≤ 0.001 |
100 | 90.9 | 271.5 | 0.909 | 0.970 (0.918 - 1) p ≤ 0.001 |
100 | 93.9 | 316 | 0.939 |
| Seven days post-admission | ||||||||||
| Model (ferritin + D-dimer + fibrinogen) |
0.981 (0.945 - 1) p ≤ 0.001 |
94.7 | 95.5 | - | 0.902 | - | - | - | - | - |
| Model (ferritin + LDH) |
- | - | - | - | - | 0.986 (0.962 - 1) p ≤ 0.001 |
100 | 90.9 | - | 0.909 |
| CRP (mg/L) | 0.737 (0.582 - 0.891) p ≤ 0.01 |
78.9 | 63.6 | 6.95 | 0.426 | 0.809 (0.664 - 0.953) p ≤ 0.001 |
61.5 | 93.9 | 21.8 | 0.555 |
| Ferritin (ng/mL) | 0.804 (0.666 - 0.942) p ≤ 0.001 |
94.7 | 59.1 | 575 | 0.538 | 0.960 (0.898 - 1) p ≤ 0.001 |
92.3 | 93.9 | 237.5 | 0.862 |
| D-dimer (mg/L) | 0.920 (0.828 - 1) p ≤ 0.001 |
89.5 | 90.9 | 0.65 | 0.804 | 0.924 (0.845 - 1) p ≤ 0.001 |
92.3 | 81.8 | 0.845 | 0.741 |
| LDH (IJ/L) | 0.885 (0.775 - 0.995) p ≤ 0.001 |
89.5 | 81.8 | 270.5 | 0.713 | 0.963 (0.910 - 1) p ≤ 0.001 |
100 | 90.9 | 241 | 0.909 |
| Male | Female | |||
| Univariate Logistic Regression | Multivariable Logistic Regression |
Univariate Logistic Regression |
Multivariable Logistic Regression |
|
| Immune cell-based blood indices | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) |
| On admission | ||||
| Neutrophil count | 1.054 (1.019 - 1.091) p ≤ 0.01 |
1.051 (1.013 - 1.089) p ≤ 0.01 |
1.097 (1.036 - 1.162) p ≤ 0.01 |
- |
| Lymphocyte count | 0.736 (0.592 - 0.916) p ≤ 0.01 |
0.766 (0.594 - 0.988) p ≤ 0.05 |
0.658 (0.507 - 0.854) p ≤ 0.01 |
- |
| NLR | 1.033 (1.011 - 1.056) p ≤ 0.01 |
- | 1.217 (0.988 - 1.500) p > 0.05 |
1.206 (0.987 - 1.473) p > 0.05 |
| IL-6 | - |
- | 1.530 (0.885-2.643) p > 0.05 |
- |
| Seven days post-admission | ||||
| Neutrophil count | 1.041 (1.017 - 1.066) p ≤ 0.001 |
- | 1.117 (1.037 - 1.203) p ≤ 0.01 |
- |
| Lymphocyte count | 0.529 (0.350 - 0.799) p ≤ 0.01 |
0.456 (0.256 - 0.815) p ≤ 0.01 | 0.823 (0.726 - 0.934) p ≤ 0.01 |
- |
| NLR | 1.040 (1.011 - 1.068) p ≤ 0.01 |
- | 1.047 (1.019 - 1.076) p ≤ 0.001 |
1.061 (1.016 - 1.107) p ≤ 0.01 |
| IL-6 | 1.895 (1.003 - 3.580) p ≤ 0.05 | - | 1.972 (1.009 - 3.853) p ≤ 0.05 |
- |
| Male | Female | |||||||||
|
Immune cell-based blood indices |
AUC (95% CI) |
Sensitivity (%) | Specificity (%) | Cut-off | J* | AUC (95% CI) |
Sensitivity (%) | Specificity (%) | Cut-off | J* |
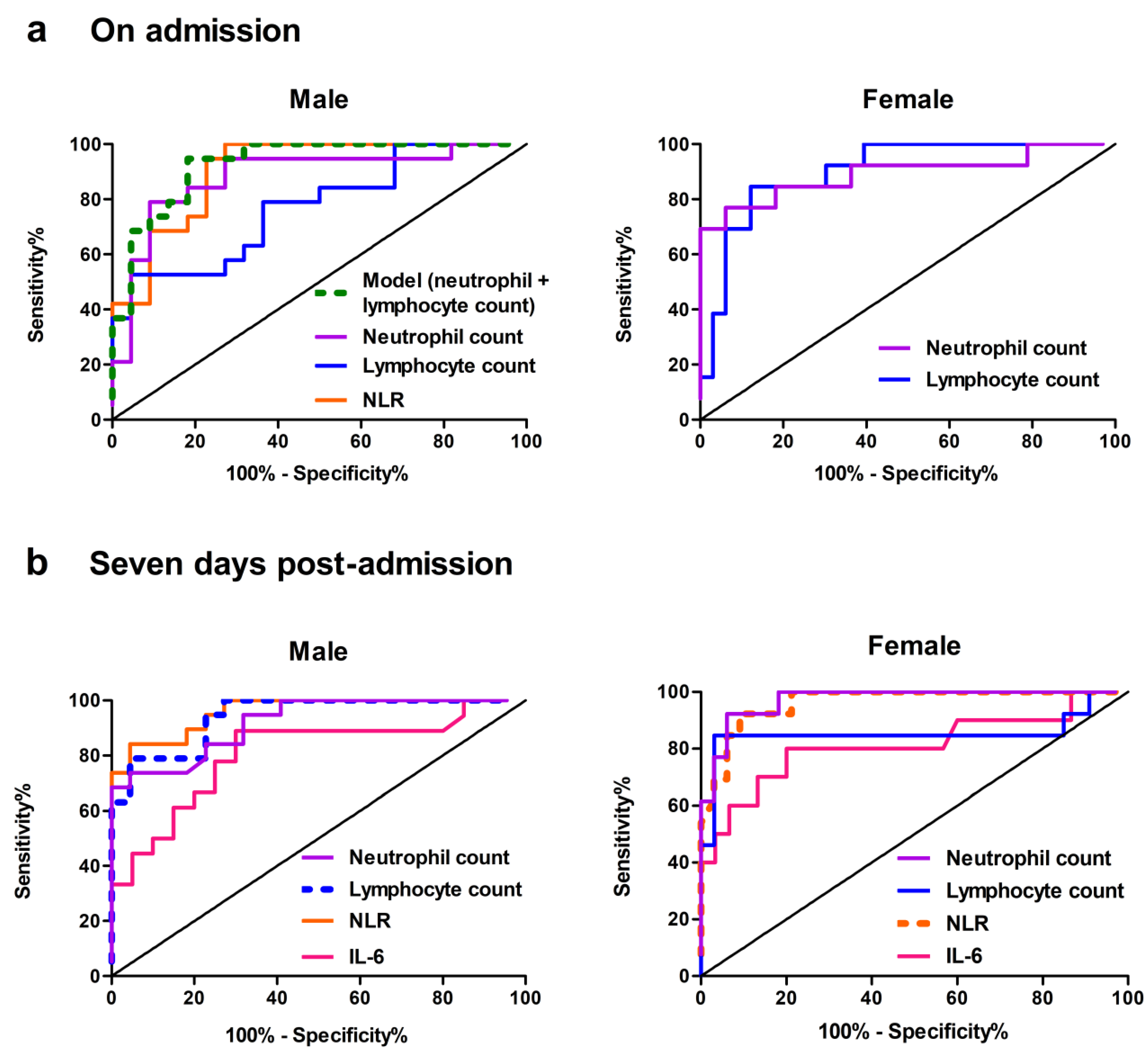
| On admission | ||||||||||
| Model (neutrophil count + lymphocyte count) |
0.928 (0.852 - 1) p ≤ 0.001 |
94.7 | 81.8 | - | 0.766 | - | - | - | - | - |
| Neutrophil count (109/L) | 0.883 (0.770 - 0.994) p ≤ 0.001 |
78.9 | 90.9 | 5.67 | 0.699 | 0.893 (0.767 - 1) p ≤ 0.001 |
76.9 | 93.9 | 4.96 | 0.709 |
| Lymphocyte count (109/L) | 0.770 (0.626 - 0.915) p ≤ 0.01 |
52.6 | 95.5 | 0.9 | 0.481 | 0.902 (0.812 - 0.993) p ≤ 0.001 |
84.6 | 87.9 | 0.9 | 0.725 |
| NLR | 0.904 (0.814 - 0.995) p ≤ 0.001 |
100 | 72.7 | 3.463 | 0.727 | - | - | - | - | - |
| Seven days post-admission | ||||||||||
| Neutrophil count (109/L) | 0.920 (0.841 - 0.999) p ≤ 0.001 |
73.7 | 95.5 | 9.85 | 0.691 | 0.972 (0.932 - 1) p ≤ 0.001 |
92.3 | 93.9 | 6.62 | 0.862 |
| Lymphocyte count (109/L) a | 0.943 (0.879 - 1) p ≤ 0.001 |
78.9 | 95.5 | 1.1 | 0.744 | 0.823 (0.643 - 1) p ≤ 0.001 |
84.6 | 93.9 | 1.18 | 0.786 |
| NLR b | 0.959 (0.908 - 1) p ≤ 0.001 |
84.2 | 95.5 | 6.661 | 0.797 | 0.965 (0.919 - 1) p ≤ 0.001 |
92.3 | 90.9 | 3.52 | 0.832 |
| IL-6 (pg/mL) | 0.807 (0.663 - 0.951) p ≤ 0.001 |
88.9 | 70.0 | 5.9 | 0.589 | 0.812 (0.627 - 0.996) p ≤ 0.01 |
80 | 80 | 6.7 | 0.600 |
| Male | Female | |||
| Univariate Logistic Regression | Multivariable Logistic Regression |
Univariate Logistic Regression |
Multivariable Logistic Regression |
|
| Seven days post-admission | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) |
| Inflammatory blood indices | ||||
| CRP | 1.612 (1.054 - 2.465) p ≤ 0.05 |
1.612 (1.054 - 2.465) p ≤ 0.05 |
- | - |
| D-dimer | - | - | 1.285 (0.953 - 1.733) p > 0.05 |
- |
| LDH | 1.039 (0.993 - 1.087) p > 0.05 |
- | 1.245 (0.975 - 1.589) p > 0.05 |
- |
|
Immune cell-based blood indices |
||||
| Neutrophil count | 1.364 (1.017 - 1.829) p ≤ 0.05 |
1.364 (1.017 - 1.829) p ≤ 0.05 |
1.003 (0.523 - 1.925) p > 0.05 |
- |
| NLR | 1.106 (0.987 - 1.239) p > 0.05 |
- | 1.242 (0.937 - 1.646) p > 0.05 |
- |
| IL-6 | 1.054 (0.883 - 1.257) p > 0.05 |
- | - | - |
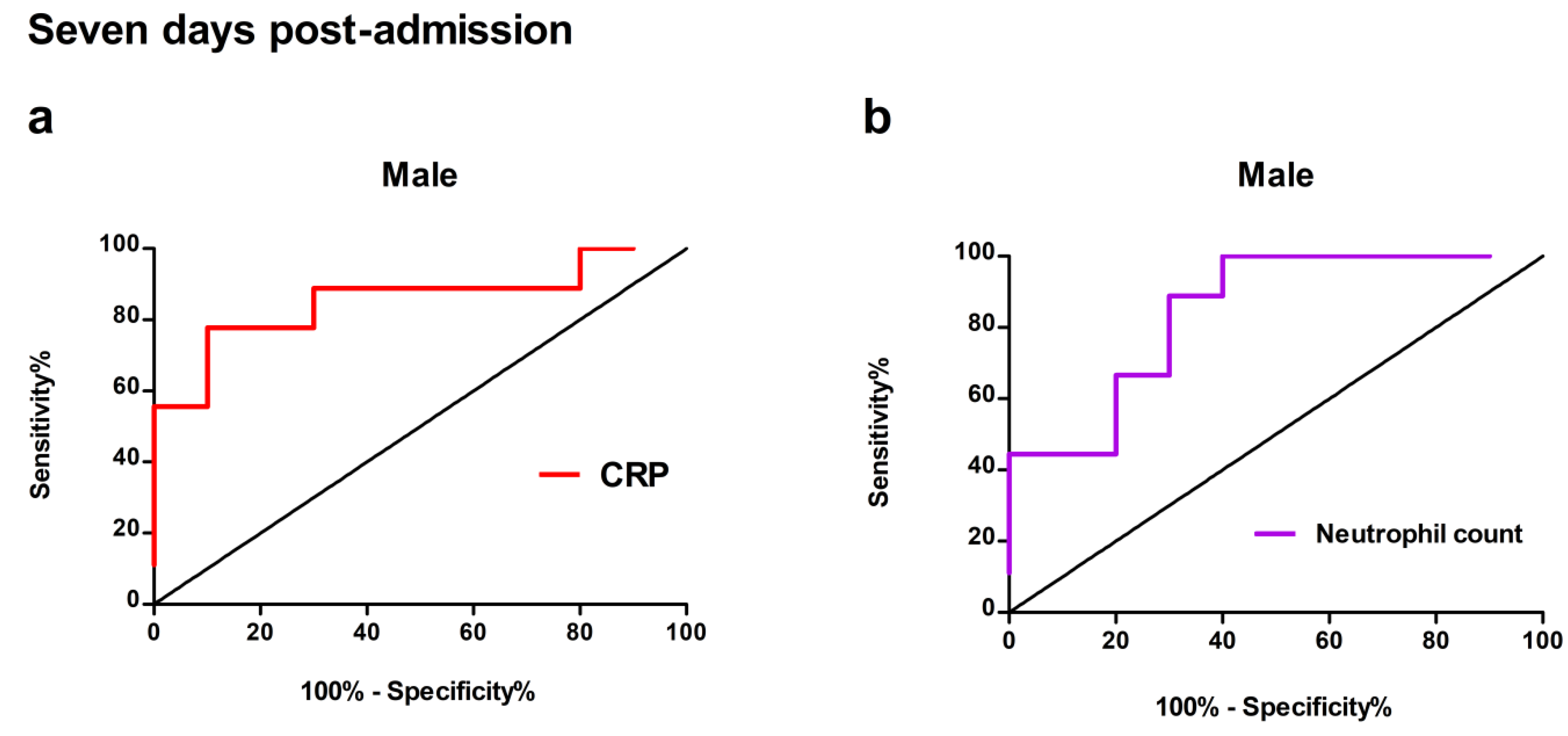
| Male | |||||
|
Seven days post-admission |
AUC (95% CI) |
Sensitivity (%) | Specificity (%) | Cut-off | J* |
| Inflammatory blood indices | |||||
| CRP (mg/L) |
0.856 (0.670 - 1) p ≤ 0.01 |
77.8 | 90 | 39.25 | 0.678 |
|
Immune cell-based blood indices |
|||||
| Neutrophil count (109/L) |
0.844 (0.668 - 1) p ≤ 0.05 |
100 | 60 | 11.03 | 0.600 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




