Submitted:
11 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
Literature Search Strategy
Eligibility Criteria
Study Selection and Data Extraction
Quality Appraisal and Synthesis
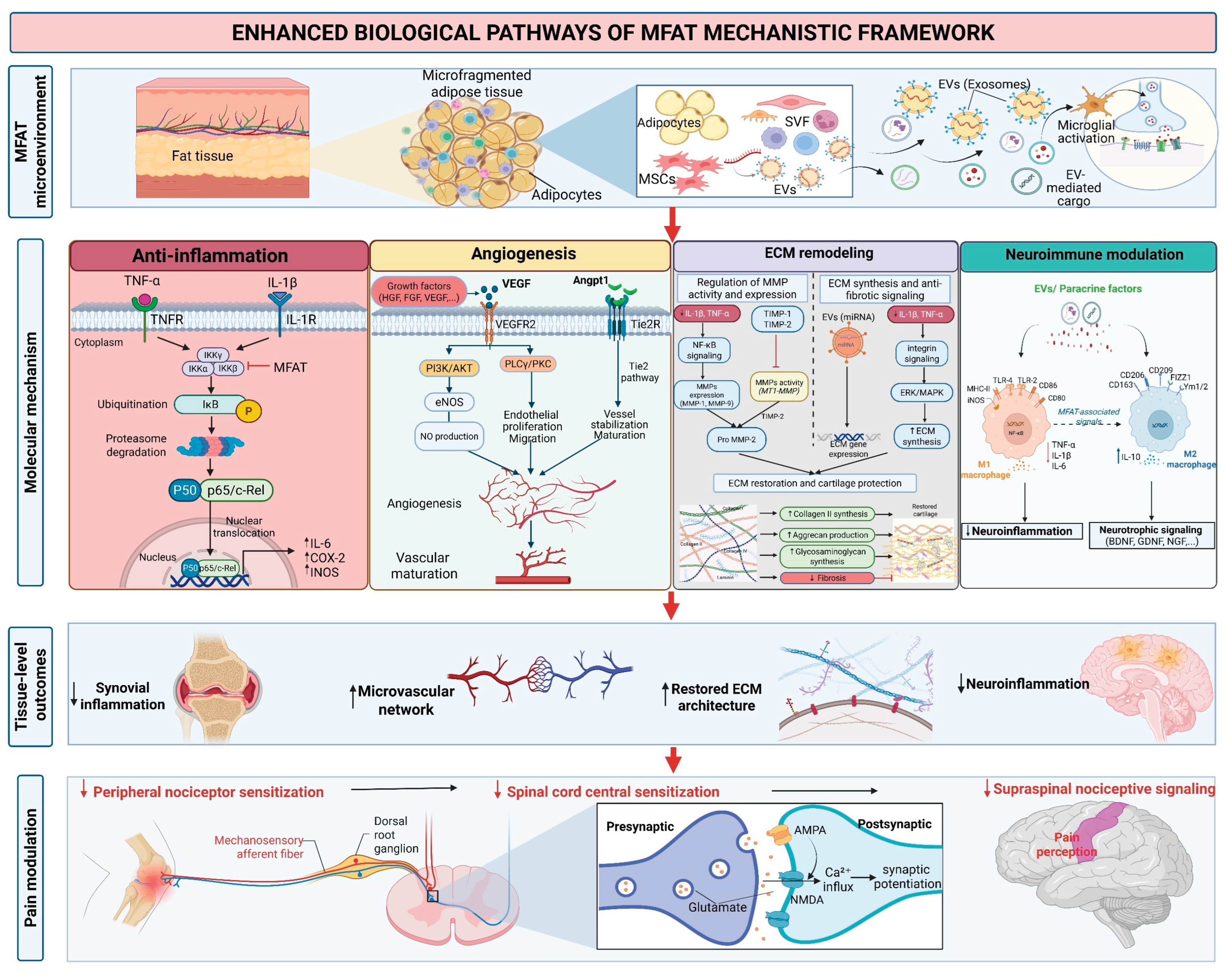
3. Biological Rationale of MFAT
Cellular Composition and Structural Preservation
Paracrine Signaling and Secretome
Immunomodulatory Effects
4. Mechanisms of Action in Tissue Repair and Pain Modulation
Anti-Inflammatory Effects
Angiogenesis and Tissue Regeneration
Extracellular Matrix Remodeling
Neuro-Modulatory Effects
| Mechanism Domain | Study Type | Reported Biological Effects | Proposed Mechanism of Pain Modulation |
Ref |
|---|---|---|---|---|
|
Cellular Architecture & Perivascular Niche Preservation |
In vivo In vitro SR/MA |
MAT pericyte fraction 33.5% vs LPA 8.39%; enzymatic dissociation abolished secretory advantage. WBC –79%, RBC –76% after microfragmentation; stromal proportions preserved. CD146+CD34–CD45– pericytes 14.6%±1.02% of SVF; clonal multipotency confirmed†. SMF/Lipogems grafts enriched in pericytes + ↑VEGF vs centrifuged fat. Lipogems filtration bag retains intact microvascular clusters | Intact perivascular niche may sustain prolonged paracrine signalling; 3D scaffold may act as sustained-release depot, potentially attenuating peripheral nociceptor sensitisation. | [20,21,22,23,62] |
|
Immune modulation & Macrophage Polarization |
In vitro In vivo Mechanistic/ Review |
MFAT co-culture with OA synoviocytes: CCL2↓, CCL3↓, CCL5↓, MMP-9, IL-10↑, PGE₂↑; miR-92a-3p → KLHL29 suppression confirmed by luciferase assay. MFAT-CM fully attenuated LPS-induced IL-1β and IL-6 in U937 macrophages, IL-1Rα, TGF-β1/β3, MIF progressively ↑ D1→D5. MSC-mediated M1→M2 shift via CARD9–NF-κB pathway demonstrated in in vivo MSC models†. | M1→M2 shift and cytokine attenuation may reduce synovial pro-algesic signalling, potentially decreasing peripheral sensitisation and nociceptor activation in OA joints. | [32,33,34,35,54,67,81] |
|
Paracrine Secretome & Extracellular Vesicles (EVs) |
In vitro Mechanistic/ Review |
MAT secretes quantitatively more growth factors/cytokines than SVF; enzymatic dissociation abolishes secretory advantage. 376/381 EV-miRNAs detected; μFAT-enriched subset corresponds to ASC-EV-specific miRNAs (proteomics confirmed). MFAT-CM: IL-1Rα, TGF-β1/β3, MIF ↑ D1→D5; HGF stable. ASC-secretome proteomic analysis: 101 immune-regulatory factors identified†. | Intact 3D scaffold may function as sustained paracrine depot; secreted factors and EV-miRNA cargo may modulate immune, angiogenic, and neuroprotective pathways, potentially contributing to multimodal analgesic and regenerative effects. | [19,20,25,27,34,62,77,81] |
|
Angiogenesis & Microvascular Remodeling |
SR/MA In vitro Mechanistic Review |
MFAT-CM → tube-like structure formation in BAECs (3/5 wells at 24 h → 5/5 at 5 d); p-ERK1/2 ↑. AT-MSC secretome ↑ HUVEC branching ×2.24 normoxia and ×2.44 hypoxia; significant in both HUVEC and RAEC models†. SMF grafts: enriched pericyte fraction + ↑VEGF vs centrifuged fat; improved graft retention in animal models. | MFAT paracrine factors may activate VEGFR2/ p-ERK1/2 in resident endothelial cells, potentially facilitating neovascularisation; preserved pericytes may stabilise capillaries and restore microcirculation, consistent with attenuation of ischaemia-driven peripheral sensitisation. | [17,21,41,43,44,46,81] |
|
Extracellular Matrix (ECM) Remodeling |
In vitro In vivo Mechanistic/ Review Observational |
Intra-articular MFAT: dGEMRIC GAG ↑ in 53.6% of 224 joint facets at 12 mo; deterioration in only 11.2%; VAS rest 3.94→0.56, VAS movement 7.33→3.17; no chondrotoxicity (n=17, 32 knees). ASC paracrine → collagen I (PRO-C1) ↑; juxtacrine → collagen VI ↑; TGF-β upregulated 18 ECM transcripts (COL1A1/A2, COL3A1, FN1, LOX1, POSTN, XYLT1)†. MFAT-CM: MMP-9↓, TIMP-1↑ in OA synoviocytes. | ASC-secreted TGF-β1/TIMP-1 may shift MMP/TIMP balance toward matrix preservation, potentially attenuating aggrecan degradation; proteoglycan restoration is consistent with reduced subchondral mechanosensitisation and nociceptor sensitisation. | [35,48,51,52,53,55,56,57,60,63] |
|
Neuroprotection & Neuroinflammation Attenuation |
In vivo In vitro Mechanistic Review |
IV hASC (1×10⁶, CCI model): complete thermal hyperalgesia reversal at day 1; IL-1β ↓ to sham levels in sciatic nerve by day 3; IL-10 ↑ progressively; iNOS fully restored at L4–L6 spinal cord at 14 d†. hASC-CM (MIA-OA mice): rapid antihyperalgesic + antiallodynic effect via IV/IA/IPL; DRG/spinal cord neuroinflammatory markers ↓ by RT-qPCR; 101 immune-regulatory secretome factors (mass spectrometry)†. AdMSC co-culture with rat SDH: LPS-induced TNFα↓, IL-6↓; NFκB nuclear translocation↓ in microglia; IL-10↑, TGF-β, TSG-6 expressed†. ADSC-Exo: Schwann cell apoptosis↓; Bcl-2↑, Bax↓; proliferation ↑ in SNI model†. | ASC secretome/ EVs may suppress microglial/astrocyte activation in DRG and dorsal horn, potentially reducing NFκB-driven neuroinflammation and central sensitisation; EV-mediated Schwann cell survival may preserve peripheral nerve integrity, attenuating ongoing nociceptive input. | [24,67,68,69,70,71,72,73,74,75,77,82] |
|
Study Type categories:SR/MA = Systematic review or meta-analysis; RCT = Randomized controlled trial; Controlled clinical = Non-randomized study with comparator; Observational clinical = Case series, cohort, before-after without control; Preclinical (in vivo) = Animal models; Preclinical (in vitro) = Cell culture, explant, organoid † General MSC/ASC/SVF study; findings not specific to MFAT/Lipogems® Abbreviations: ASC: adipose-derived stromal/stem cell; AdMSC: adipose-derived mesenchymal stem cell; AT-MSC: adipose tissue–derived mesenchymal stromal cell; BAEC: bovine aortic endothelial cell; Bax: BCL2-associated X protein; Bcl-2: B-cell lymphoma 2; CARD9: caspase recruitment domain-containing protein 9; CCI: chronic constriction injury; CCL: C-C motif chemokine ligand; CD: cluster of differentiation; CM: conditioned medium; COL: collagen; dGEMRIC: delayed gadolinium-enhanced MRI of cartilage; DRG: dorsal root ganglion; ECM: extracellular matrix; ERK1/2: extracellular signal-regulated kinase 1/2; EV: extracellular vesicle; FN1: fibronectin 1; GAG: glycosaminoglycan; hASC: human adipose-derived stromal/stem cell; HGF: hepatocyte growth factor; HUVEC: human umbilical vein endothelial cell; IA: intra-articular; IL: interleukin; IL-1Rα: interleukin-1 receptor antagonist; iNOS: inducible nitric oxide synthase; IPL: intraplantar; IV: intravenous; KLHL29: kelch-like protein 29; LOX1: lysyl oxidase 1; LPA: lipoaspirate; LPS: lipopolysaccharide; MAT: microfragmented adipose tissue; MFAT: microfragmented adipose tissue; MFAT-CM: microfragmented adipose tissue–conditioned medium; MIF: macrophage migration inhibitory factor; MIA: monosodium iodoacetate; miRNA: microRNA; MMP: matrix metalloproteinase; MSC: mesenchymal stromal/stem cell; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; NG2: neural/glial antigen 2; OA: osteoarthritis; PDGF: platelet-derived growth factor; PDGFR-β: platelet-derived growth factor receptor beta; PGE₂: prostaglandin E₂; POSTN: periostin; RAEC: rat aortic endothelial cell; RANTES: regulated on activation, normal T cell expressed and secreted (CCL5); RBC: red blood cell; RCT: randomized controlled trial; RT-qPCR: reverse transcription quantitative polymerase chain reaction; SDH: spinal dorsal horn; SMF: stromal microfragmented fat; SNI: spared nerve injury; SR/MA: systematic review and meta-analysis; SVF: stromal vascular fraction; TGF-β: transforming growth factor beta; TIMP: tissue inhibitor of metalloproteinases; TNF-α: tumor necrosis factor alpha; TSG-6: tumor necrosis factor-stimulated gene 6 protein; VAS: Visual Analogue Scale; VEGF: vascular endothelial growth factor; VEGFR2: vascular endothelial growth factor receptor 2; WBC: white blood cell; XYLT1: xylosyltransferase 1. | ||||
Clinical Applications of MFAT
Osteoarthritis
Tendinopathies
Sports Medicine and Orthopedics
Chronic Pain Syndromes
e. Low Back Pain and Discogenic Pain
Other Emerging Indications
| Author, Year | Study design |
N |
MFAT preparation |
Comparator | Follow-up (months) |
Primary outcome tool |
Key pain / functional Result |
Safety/ AEs reported |
|---|---|---|---|---|---|---|---|---|
| A. Knee Osteoarthritis | ||||||||
| Panchal et al., 2018 [84] | Case series | 17 | Lipogems system; autologous MFAT; mechanical micro-fragmentation; closed system; no enzymatic processing; preserves perivascular niche (~500 µm clusters); same-day processing and injection | None |
12 (6 wk, 6 mo, 12 mo follow-up) | NPRS; KSS (1989); Functional Score (FXN); LEAS | Significant improvement in pain (NPRS ↓ from ~5.7 → ~3.0 at 6 mo → ~4.35 at 12 mo); KSS ↑ (74 → 81.6 at 12 mo); FXN ↑ (65.4 → 76.4 at 12 mo); LEAS improved up to 6 mo (not significant at 12 mo) | No serious AEs; minor AEs: transient pain & swelling resolving within 48–72 h |
| Russo et al., 2017 [86] | Retrospective observational study | 30 | Lipogems®; autologous MFAT; mechanical micro-fragmentation; closed system; no enzymatic processing; intra-operative (one-step); 10–15 mL injected intra-articular | None |
12 | KOOS; IKDC-subjective; Tegner Lysholm; VAS | Median improvement: KOOS_total +20; IKDC +20; VAS −24; Tegner +31; ≥10-point improvement: KOOS 67%, IKDC 70%, VAS 83% | No major AEs; 3 minor events (2 hematoma harvest site, 1 recurrent effusion); no infection |
| Russo et al., 2018 [87] | Retrospective observational | 30 | Lipogems®; autologous MFAT; mechanical micro-fragmentation; closed system; no enzymatic processing; intra-operative injection | None |
36 (12 & 36 mo assessed) | KOOS; IKDC-subjective; Tegner Lysholm; VAS | Results at 1 year maintained at 3 years ; further improvement vs 1-year in: KOOS (64%), IKDC (55%), VAS (55%), Tegner (41%); >50% patients improved ≥20 points vs baseline | No AEs, 7 patients required additional treatments |
| Russo et al., 2023 [11] | Case series |
49 (67 knees) | Lipogems®; autologous MFAT; mechanical micro-fragmentation; closed system; no enzymatic processing; intra-articular injection (10 mL/knee); one-step | None (single arm) |
Mean 34 months (range 11–59; assessed up to 36 months) | VAS, KOOS | Significant improvement in WOMAC & KOOS at all follow-ups; improvement begins at 3 months → peaks ~6 months → stable to 24–36 months; ~80% đạt MCID WOMAC function; effect influenced by KL grade & gender; worse baseline → better response | No severe AEs; mild AE: knee pain/ swelling (10.4%), abdominal ecchymosis (6%) |
| Ulivi et al., 2023 [10] | Prospective RCT, single centre | 78 (final n=67) | Lipogems®; autologous mFAT; mechanical microfragmentation; intra-articular injection 6–8 mL; one-step procedure during surgery | Arthroscopic debridement alone |
6 (primary); mean 26.1 ± 9.5 months (follow-up) | VAS; KOOS-PS (primary); WOMAC; KSS; SF-12 | At 6 months: significant improvement in KOOS-PS (p=0.024) & KSS (p=0.046) vs AD; MCID achieved (VAS, KOOS-PS, KSS); At final follow-up (~26 months): trend but no significant difference; intra-group improvement maintained | No procedure-related serious AEs; 1 mild hematoma; serious AE unrelated |
| Bruno et al., 2025 [12] | Prospective clinical study (case series) | 41 (38 at 48 months) | Lipogems®; autologous MFAT; intra-articular injection after arthroscopic debridement | None (single arm) |
48 | KOOS, VAS | Significant reduction in VAS from 5.8 ± 1.63 to 2.22 ± 1.4 at 3 months, maintained at ~2.89 at 48 months; all KOOS subscales significantly improved from 3 months and sustained to 48 months; Tegner score improved modestly; greater benefit observed in KL I–II vs KL III–IV |
No serious AEs; minor complications: abdominal hematoma (self-resolving); 3 patients required total knee arthroplasty |
| Stanciu et al., 2025 [40] | Retrospective observational study | 335 | Lipogems®; autologous MFAT; mechanical micro-fragmentation; closed system; no enzymatic processing; single intra-articular injection (6–8 mL); outpatient procedure | None (single arm) |
36 | VAS; OKS; WOMAC; KOOS | Significant improvement in all scores at 3 months, sustained up to 3 years (p < 0.001); early improvement at 3 months strongly predicts long-term outcomes (1–3 years); e.g., VAS ↓ from ~43.5 to ~25.9 at 3 months and ~29.0 at 3 years; KOOS, WOMAC, OKS all improved significantly | No major safety concerns reported |
| De Groote et al., 2025 [85] | Longitudinal observational cohort | 39 (initial 58) | Autologous MFAT via MYFILL®; abdominal liposuction; centrifugation; 5–10 mL intra-articular injection | None (single arm) |
12 | KOOS | Significant improvement in all KOOS domains (except Symptoms not reaching MCID); peak at 6 months, slight decline at 12 months but still > baseline; ΔKOOS ~ +14.1 points at 12 months (> MCID) | Synovitis 18% (self-limited), no severe AEs |
| Zaffagnini et al., 2022 [96] | RCT (single-blind) | 118 (final: 108; MFAT =53, PRP =55) | Lipogems® system; autologous adipose tissue; mechanical microfragmentation; no enzymatic digestion; ~5 mL intra-articular injection | PRP (single intra-articular injection; leukocyte-rich PRP ~5× platelet concentration) | 24 | IKDC (primary), KOOS pain, VAS, EQ-5D, EQ-VAS | Both MFAT and PRP showed significant improvement in IKDC and KOOS pain from baseline to 6 months and sustained up to 24 months (p < 0.0005); no significant difference between groups; MFAT showed higher MCID achievement in moderate–severe OA at 6 months | Comparable safety; AEs: MFAT 18.9% vs PRP 10.9% (mostly mild, self-limiting); no treatment-related serious AEs |
| Ye et al., 2024 [13] | Systematic review & meta-analysis (4 RCTs) |
266 (326 knees) | MFAT intra-articular injection | PRP intra-articular injection | 6–24 months | VAS, KOOS, Tegner | PRP superior in VAS at 12 months (MD=0.99, p=0.004); MFAT superior in Tegner at 6 months (MD=0.65, p=0.02); no difference in KOOS domains | No serious AEs |
| Hu et al., 2025 [83] | Systematic review (2 RCTs + 4 retrospective) | 440 | Autologous MFAT; processed via Lipogems® / VacLock®; delivered intra-articularly under knee arthroscopy (combined with debridement / lavage) | Variable (some controlled vs arthroscopy alone/ conventional care; some single -arm) | 12–48 | VAS, WOMAC, KOOS, Lysholm, IKDC | Pain reduction: 44.4–62.2%; significant improvement in all functional scores; consistent clinical benefit across studies | Mild AEs only (pain, swelling, hematoma); no serious complications |
| B. Focal Chondral Lesion & Rotator Cuff | ||||||||
| Bisicchia et al., 2020 [97] | RCT (single-blind) |
40 | Autologous micro-fragmented stromal vascular fraction (SVF) via Lipogems®; mechanical processing; ~10 mL intra-articular injection under arthroscopy (same procedure as microfracture) | Micro-fractures alone |
12 | WOMAC (primary), VAS, Oxford Knee Score, EQ-5D | No difference at 1 month; at 3 months ↓VAS significantly in MFAT group (p=0.04); at 6 and 12 months: MFAT group superior in all outcomes; WOMAC significantly better at 12 months (17.7 vs 25.5; p=0.03); medium effect size (Cohen’s d ~0.65–0.75) | No AEs related to MFAT; one knee effusion in control group |
| Randelli et al., 2022 [98] | RCT (single-blind) |
44 (22 MFAT + repair; 22 repair alone) | Autologous microfragmented adipose tissue (Lipogems®; enzyme-free; intraoperative processing; ~60–100 mL injected at repair site) | Standard arthroscopic rotator cuff repair |
24 | VAS | No difference in early postoperative pain (first 4 weeks); at 6 months: improved CMS, ASES, SST, strength in MFAT group; no difference at 24 months | No serious AEs |
| C. Tendinopathy | ||||||||
| Usuelli et al., 2018 [90] | RCT |
44 | Autologous SVF from adipose tissue; mechanical processing (FastKit); intratendinous + peritendinous ultrasound-guided injection | Leukocyte -poor PRP | 6 | VAS | Both groups improved significantly vs baseline; SVF showed faster pain reduction at 15 and 30 days (p<0.05); no significant difference between groups at later time points (≥60 days) | No serious AEs; mild harvest-site discomfort/ hematoma (~25%) |
| D. Intervertebral Disc Degeneration/ Low Back Pain | ||||||||
| Comella et al., 2017 [112] | Prospective open-label pilot study | 15 | Autologous SVF (collagenase digestion) + PRP; intradiscal injection under fluoroscopy | None (single arm) |
6 (safety follow-up 12 months) | VAS, ODI, SF-36 | Significant pain reduction: VAS 5.6 → 3.6 at 6 months (p=0.01); improvement in PPI, flexion, SF-36; ODI/BDI trend improvement (NS) | No serious AEs; mild soreness post-procedure |
| Noriega DC et al., 2021 [101] | RCT (long-term follow-up) | 23 (from original RCT n=24) | Allogeneic bone marrow–derived MSCs (expanded ex vivo, 25×10⁶ cells/disc; intradiscal injection under local anesthesia) | Sham control (paravertebral infiltration) | 42 (~3.5 years) | VAS; ODI; MRI (Pfirrmann grade) | Sustained improvement: early VAS and ODI improvement maintained at 3.5 years; therapeutic efficiency increased over time (pain: 0.60; disability: 0.71); clear responder subgroup identified | No serious AEs in either group |
| Kumar et al., 2017 [102] | Phase I single-arm clinical trial | 10 | Autologous AT-MSCs (2×10⁷–4×10⁷ cells/disc) + HA derivative (Tissuefill®); intradiscal injection under fluoroscopy | None (safety study) |
12 | VAS; ODI; SF-36 | Significant improvement: VAS reduced from 6.5 → 2.9 at 12 months (p=0.002); ODI reduced from 42.8% → 16.8% (p=0.002); 6/10 patients achieved ≥50% improvement; partial increase in disc hydration (ADC MRI) in 3 patients | No procedure- or cell-related adverse events; no serious AEs |
| Schol J et al., 2024 [113] | Systematic review | ≥1974 patients (68 studies) | Mixed: MSCs (BM, AD, UC), BMC, BMA, PRP (LP-PRP, LR-PRP), SVF, cell combinations | Variable (placebo, steroid, surgery, conservative care, or none) | Up to 24–72 months (heterogeneous across studies) | VAS/NRS/NPS; ODI; QoL (SF-12/36); MRI (Pfirrmann, ADC, disc height) | Median pain reduction ≈ 3.2–3.8 points; ODI reduction ≈ 27 points at 12 months; ~60–78% studies reached MCID; outcomes comparable to spinal fusion at 2 years | Severe AEs rate ≈ 1.8% overall; mainly disc herniation and infection; generally favorable safety |
| Lee et al., 2023 [114] | Phase I single-arm open-label clinical trial | 8 | Autologous adipose-derived stromal cells (ASC) → matrilin-3 priming (10 ng/mL, 5 days) → spheroid formation (125 cells/ well) → combined with hyaluronic acid (HA); intradiscal injection (6×10⁶ cells/disc) | None |
6 | VAS; ODI; MRI | 6/8 patients (75%) achieved clinical success (≥2-point VAS ↓ and ≥10-point ODI ↓); marked reduction in VAS & ODI over time; partial MRI improvement (↓ HIZ, ↓ disc protrusion); no change in Pfirrmann grade | No AEs, normal lab parameters throughout follow-up |
| E. Lumbar Facet Joint Arthropathy | ||||||||
| Rothoerl et al., 2023 [125] | Observational cohort (single-arm) | 37 | Autologous ADRCs from lipoaspirate (50–100 mL), enzymatic isolation (Transpose RT + Matrase), point-of-care (no expansion), injected periarticular facet joints | None (single arm) |
60 | VAS, ODI | VAS: 6.8 → 1.5 (1 year) → 1.4 (5 years); ODI: 71% → 17.5% (1 year) → 18.7% (5 years); improvement in 100% patients | 1 hematoma (anti-coagulated patient); no infection, no systemic AEs |
| Qu W et al., 2025 [126] | Phase I, prospective, single-arm, open-label | 10 (9 completed) | Allogeneic bone marrow–derived mesenchymal stromal cells (BM-MSCs), culture-expanded under cGMP; 10 million cells per facet joint (2 joints injected) | None | 24 | VAS; PROMIS CAT Physical Function; MRI | Pain responder rate (≥50% VAS reduction): 33.3% at 3 mo, 75% at 6 mo, 66.7% at 12–24 mo; mean VAS reduction ≈ −4.27 (6 months), −4.04 (12 months), −3.37 (24 months); functional responder rate (PROMIS CAT PF ≥2.3 improvement): 55.6% across follow-up; MRI: reduced facet degeneration in 5/9 patients, unchanged in 3, progression in 1 | No study-related serious AEs; transient procedure-related AEs (e.g., injection-site pain, discomfort); reported serious AEs were unrelated to treatment |
| F. Other Musculoskeletal/ Wound-Healing Indications | ||||||||
| Lonardi et al., 2019 [127] | RCT |
114 (57 MFAT vs 57 control) | Autologous micro-fragmented adipose tissue (Lipogems®, mechanical processing, no enzymatic digestion, 50–100 mL lipoaspirate → 10–30 mL injected locally) | Standard wound care | 6 | Healing rate/ time; VAS (secondary) | Healing rate: 80% vs 46% (p=0.0064); healing time: no difference (~2.8 months both groups); pain (VAS): no significant difference between groups | No treatment-related AEs; 2 hematomas at harvest site (anti-coagulated patients); no relapse |
| Stark et al., 2020 [128] | Observational case series |
10 | Autologous micro-fragmented adipose tissue (Lipogems®, mechanical processing, no enzymatic digestion; ~200 mL lipoaspirate → ~20 mL MFAT injected) | None (single arm) |
6–16 months (planned up to 24 months) | FSFI; ICIQ-UI SF; VSQ; SF-12 | All patients showed symptom improvement; FSFI↑, ICIQ-UI↓, VSQ↓, SF-12↑; resolution or marked reduction of dyspareunia, dryness, and SUI in multiple cases | No intra-operative or post-operative AEs; no infection, no pain, no worsening symptoms |
| Abbreviations:ADC: apparent diffusion coefficient; ADRCs: adipose-derived regenerative cells; AEs: adverse events; ASC: adipose-derived stromal cells; ASES: American Shoulder and Elbow Surgeons score; AT-MSCs: adipose tissue-derived mesenchymal stromal/stem cells; BM-MSCs: bone marrow-derived mesenchymal stromal/stem cells; CMS: Constant–Murley Score; EQ-5D: EuroQol 5-Dimension; EQ-VAS: EuroQol Visual Analogue Scale; EVs: extracellular vesicles; FSFI: Female Sexual Function Index; HA: hyaluronic acid; HIZ: high-intensity zone; ICIQ-UI SF: International Consultation on Incontinence Questionnaire–Urinary Incontinence Short Form; IKDC: International Knee Documentation Committee score; KL: Kellgren–Lawrence grading system; KOOS: Knee injury and Osteoarthritis Outcome Score; KOOS-PS: KOOS Physical Function Shortform; KSS: Knee Society Score; LEAS: Lower Extremity Activity Scale; LoE: level of evidence; MCID: minimal clinically important difference; MFAT: microfragmented adipose tissue; MRI: magnetic resonance imaging; MSCs: mesenchymal stromal/stem cells; NPRS: numerical pain rating scale; ODI: Oswestry Disability Index; OKS: Oxford Knee Score; PROMIS: Patient-Reported Outcomes Measurement Information System; PRP: platelet-rich plasma; SF-12: 12-item Short Form Health Survey; SF-36: 36-item Short Form Health Survey; SST: Simple Shoulder Test; SVF: stromal vascular fraction; VAS: visual analogue scale; VSQ: Vaginal Symptoms Questionnaire; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index. | ||||||||
Safety Profile and Regulatory Considerations
Limitations of Current Evidence and Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Ethical approval
Consent to participate
Consent to publication
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abbreviation | Full Expansion |
| ADC | apparent diffusion coefficient |
| ADRCs | adipose-derived regenerative cells |
| AdMSC | adipose-derived medicinal signalling cell |
| ADAMTS | a disintegrin and metalloproteinase with thrombospondin motifs |
| AEs | adverse events |
| AKT | protein kinase B |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| Angpt1 | angiopoietin-1 |
| ASES | American Shoulder and Elbow Surgeons score |
| ASC | adipose-derived stromal cell |
| AT-MSC | adipose tissue-derived mesenchymal stromal/stem cell |
| BAEC | bovine aortic endothelial cell |
| BDNF | brain-derived neurotrophic factor |
| BM-MSC | bone marrow-derived mesenchymal stromal/stem cell |
| BMA | bone marrow aspirate |
| BMC | bone marrow concentrate |
| CARD9 | caspase recruitment domain family member 9 |
| CCL2,3,5 | C-C motif chemokine ligand 2,3,5 |
| CD | cluster of differentiation |
| CFR | Code of Federal Regulations |
| cGMP | current good manufacturing practice |
| CMS | Constant–Murley Score |
| COX-2 | cyclooxygenase-2 |
| CXCL-9 | C-X-C motif chemokine ligand 9 |
| dGEMRIC | delayed gadolinium-enhanced MRI of cartilage |
| DRG | dorsal root ganglion |
| ECM | extracellular matrix |
| eNOS | endothelial nitric oxide synthase |
| EQ-5D | EuroQol 5-Dimension questionnaire |
| EQ-VAS | EuroQol Visual Analogue Scale |
| ERK | extracellular signal-regulated kinase |
| EV | extracellular vesicle |
| FDA | U.S. Food and Drug Administration |
| FGF | fibroblast growth factor |
| FSFI | Female Sexual Function Index |
| GAG | glycosaminoglycan |
| GDNF | glial cell line-derived neurotrophic factor |
| HA | hyaluronic acid |
| HCT/P | human cells, tissues, and cellular and tissue-based products |
| HGF | hepatocyte growth factor |
| HIF-1α | hypoxia-inducible factor-1α |
| HIZ | high-intensity zone |
| HUVEC | human umbilical vein endothelial cell |
| ICIQ-UI SF | International Consultation on Incontinence Questionnaire–Urinary Incontinence Short Form |
| IDD | intervertebral disc degeneration |
| κB | inhibitor of nuclear factor-κB |
| IKK | IκB kinase |
| IKDC | International Knee Documentation Committee score |
| IL | Interleukin |
| IL-1β, 6,10 | interleukin-1β,6,10 |
| IL-1R | interleukin-1 receptor |
| IL-1Rα | interleukin-1 receptor antagonist |
| IL-17RA | interleukin-17 receptor A |
| iNOS | inducible nitric oxide synthase |
| KLHL29 | kelch-like protein 29 |
| KL | Kellgren–Lawrence grading system |
| KOOS | Knee injury and Osteoarthritis Outcome Score |
| KOOS-PS | Knee injury and Osteoarthritis Outcome Score Physical Function Short Form |
| KSS | Knee Society Score |
| LBP | low back pain |
| LEAS | Lower Extremity Activity Scale |
| LoE | level of evidence |
| LOX1 | lysyl oxidase 1 |
| LP-PRP | leukocyte-poor platelet-rich plasma |
| LR-PRP | leukocyte-rich platelet-rich plasma |
| M1, M2 | classically activated macrophage |
| MAPK | mitogen-activated protein kinase |
| MCID | minimal clinically important difference |
| MeSH | Medical Subject Headings |
| MFAT | microfragmented adipose tissue |
| MIF | macrophage migration inhibitory factor |
| miRNA | microRNA |
| MMP | matrix metalloproteinase |
| MRI | magnetic resonance imaging |
| MSC | mesenchymal stromal/stem cell |
| NF-κB | nuclear factor-κB |
| NGF | nerve growth factor |
| NMDA | N-methyl-D-aspartate |
| NPRS | numerical pain rating scale |
| NSAIDs | nonsteroidal anti-inflammatory drugs |
| OA | Osteoarthritis |
| ODI | Oswestry Disability Index |
| OKS | Oxford Knee Score |
| PDGF | platelet-derived growth factor |
| PDGFR-β | platelet-derived growth factor receptor beta |
| PGE₂ | prostaglandin E₂ |
| PI3K | phosphoinositide 3-kinase |
| PKC | protein kinase C |
| PLCγ | phospholipase Cγ |
| POSTN | Periostin |
| PROMIS | Patient-Reported Outcomes Measurement Information System |
| PRP | platelet-rich plasma |
| RAEC | rat aortic endothelial cell |
| RBC | red blood cell |
| RCT | randomized controlled trial |
| ROS | reactive oxygen species |
| SANRA | Scale for the Assessment of Narrative Review Articles |
| SDH | spinal dorsal horn |
| SF-12 | 12-item Short Form Health Survey |
| SF-36 | 36-item Short Form Health Survey |
| SMAD | mothers against decapentaplegic homolog |
| SR/MA | systematic review and/or meta-analysis |
| SST | Simple Shoulder Test |
| SVF | stromal vascular fraction |
| TGF-β | transforming growth factor-β |
| Tie2 | TEK receptor tyrosine kinase |
| TIMP | tissue inhibitor of metalloproteinases |
| TLR | Toll-like receptor |
| TLR4 | Toll-like receptor 4 |
| TNF-α | tumor necrosis factor-α |
| TNFR | tumor necrosis factor receptor |
| TSG-6 | tumor necrosis factor-stimulated gene 6 |
| UC | umbilical cord |
| VAS | visual analogue scale |
| VEC | vascular endothelial cell |
| VEGF | vascular endothelial growth factor |
| VEGFR2 | vascular endothelial growth factor receptor 2 |
| VSQ | Vaginal Symptoms Questionnaire |
| WBC | white blood cell |
| WOMAC | Western Ontario and McMaster Universities Osteoarthritis Index |
References
- Ząbek, Z.; Wyczałkowska-Tomasik, A.; Poboży, K.; et al. Understanding the Microenvironment of Intervertebral Disc Degeneration: A Comprehensive Review of Pathophysiological Insights and Therapeutic Implications. IJMS 2025, 26, 9938. [Google Scholar] [CrossRef]
- Wang, X.; Martin, G.; Sadeghirad, B.; et al. Common interventional procedures for chronic non-cancer spine pain: a systematic review and network meta-analysis of randomised trials. BMJ 2025, 388, e079971. [Google Scholar] [CrossRef]
- Shtroblia, V.; Petakh, P.; Kamyshna, I.; et al. Recent advances in the management of knee osteoarthritis: a narrative review. Front Med. 2025, 12, 1523027. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Ge, G.; Yang, P.; et al. An Update on Adipose-Derived Stem Cells for Regenerative Medicine: Where Challenge Meets Opportunity. Advanced Science 2023, 10, 2207334. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; et al. Multilineage Cells from Human Adipose Tissue: Implications for Cell-Based Therapies. Tissue Engineering 2001, 7, 211–28. [Google Scholar] [CrossRef]
- Wang, L.; Jiang, X.; Zhao, F.; et al. A review of adipose-derived mesenchymal stem cells‘ impacts and challenges: metabolic regulation, tumor modulation, immunomodulation, regenerative medicine and genetic engineering therapies. Front Endocrinol. 2025, 16, 1606847. [Google Scholar] [CrossRef]
- Tremolada, C.; Colombo, V.; Ventura, C. Adipose Tissue and Mesenchymal Stem Cells: State of the Art and Lipogems® Technology Development. Curr Stem Cell Rep. 2016, 2, 304–12. [Google Scholar] [CrossRef]
- Rodeo, S.A.; De La Huerta Meza, D.; Kirschner, J.; et al. The Hospital for Special Surgery Center for Regenerative Medicine: Clinical Registries, Basic and Translational Research, and Education Programs. HSS Journal®: The Musculoskeletal Journal of Hospital for Special Surgery 2025, 21, 361–5. [Google Scholar] [CrossRef] [PubMed]
- Parmar, T.; Lingam, S.; AlSamhori, J.; et al. Microfragmented adipose tissue in orthopedic regeneration: mechanisms, clinical evidence, and regulatory perspectives. Regenerative Medicine 2026, 1–13. [Google Scholar] [CrossRef]
- Ulivi, M.; Meroni, V.; Viganò, M.; et al. Micro-fragmented adipose tissue (mFAT) associated with arthroscopic debridement provides functional improvement in knee osteoarthritis: a randomized controlled trial. Knee surg sports traumatol arthrosc. 2023, 31, 3079–90. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Cortina, G.; Condello, V.; et al. Autologous micro-fragmented adipose tissue injection provides significant and prolonged clinical improvement in patients with knee osteoarthritis: a case-series study. J exp orthop. 2023, 10, 116. [Google Scholar] [CrossRef]
- Bruno, A.A.M.; Anzillotti, G.; De Donato, M.; et al. Arthroscopic debridement followed by intra-articular injection of micro-fragmented adipose tissue in patients affected by knee osteoarthritis: Clinical results up to 48 months from a prospective clinical study. J exp orthop. 2025, 12, e70144. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Shen, Z.; Li, X.; et al. Microfragmented adipose tissue versus platelet-rich plasma in the treatment of knee osteoarthritis: a systematic review and meta-analysis. Acta Orthop Belg. 2024, 90, 549–58. [Google Scholar] [CrossRef]
- Wang, C.; Yao, B. Efficacy and safety of platelet-rich plasma injections for the treatment of knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Eur J Med Res. 2025, 30, 992. [Google Scholar] [CrossRef]
- Alad, M.; Yousef, F.; Epure, L.M.; et al. Unraveling Osteoarthritis: Mechanistic Insights and Emerging Therapies Targeting Pain and Inflammation. Biomolecules 2025, 15, 874. [Google Scholar] [CrossRef]
- Baethge, C.; Goldbeck-Wood, S.; Mertens, S. SANRA—a scale for the quality assessment of narrative review articles. Res Integr Peer Rev. 2019, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Krawczenko, A.; Klimczak, A. Adipose Tissue-Derived Mesenchymal Stem/Stromal Cells and Their Contribution to Angiogenic Processes in Tissue Regeneration. IJMS 2022, 23, 2425. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, Y.; Zhang, Y.; et al. Engineered stromal vascular fraction for tissue regeneration. Front Pharmacol. 2025, 16, 1510508. [Google Scholar] [CrossRef]
- Da Silva, K.; Kumar, P.; Choonara, Y.E. The paradigm of stem cell secretome in tissue repair and regeneration: Present and future perspectives. Wound Repair Regeneration 2025, 33, e13251. [Google Scholar] [CrossRef] [PubMed]
- Vezzani, B.; Shaw, I.; Lesme, H.; et al. Higher Pericyte Content and Secretory Activity of Microfragmented Human Adipose Tissue Compared to Enzymatically Derived Stromal Vascular Fraction. Stem Cells Translational Medicine 2018, 7, 876–86. [Google Scholar] [CrossRef]
- McSweeney, J.E.; Yong, L.Y.; Goddard, N.V.; et al. Does Secondary Mechanical Manipulation of Lipoaspirate Enhance the Vasculogenic Potential of Fat Grafts? A Systematic Review. Ann Plast Surg. 2024, 93, 389–96. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Li, Z.; Zhang, Y.; et al. A Scoping Review on Mechanically Micronized Adipose-Derived Products: Technological Innovations and Regenerative Potential. Aesthetic Surgery Journal 2026, sjag025. [Google Scholar] [CrossRef]
- Crisan, M.; Yap, S.; Casteilla, L.; et al. A Perivascular Origin for Mesenchymal Stem Cells in Multiple Human Organs. Cell Stem Cell. 2008, 3, 301–13. [Google Scholar] [CrossRef] [PubMed]
- Dabravolski, S.A.; Andreeva, E.R.; Eremin, I.I.; et al. The Role of Pericytes in Regulation of Innate and Adaptive Immunity. Biomedicines 2023, 11, 600. [Google Scholar] [CrossRef]
- Liu, W.; Liu, T.; Zhao, Q.; et al. Adipose Tissue-Derived Extracellular Vesicles: A Promising Biomarker and Therapeutic Strategy for Metabolic Disorders. Yuan S, editor. Stem Cells International 2023, 2023, 1–16. [Google Scholar] [CrossRef]
- Giannasi, C.; Cadelano, F.; Della Morte, E.; et al. Unlocking the Therapeutic Potential of Adipose-Derived Stem Cell Secretome in Oral and Maxillofacial Medicine: A Composition-Based Perspective. Biology 2024, 13, 1016. [Google Scholar] [CrossRef]
- Wu, T.; Liu, Y.; Wang, S.; et al. MSC-Derived Extracellular Vesicles: Roles and Molecular Mechanisms for Tissue Repair. IJN 2025, Volume 20, 7953–74. [Google Scholar] [CrossRef]
- Mo, W.; Peng, Y.; Zheng, Y.; et al. Extracellular vesicle-mediated bidirectional communication between the liver and other organs: mechanistic exploration and prospects for clinical applications. J Nanobiotechnol 2025, 23, 190. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Liao, Y.; Li, Y. Effects of microenvironment and biological behavior on the paracrine function of stem cells. Genes & Diseases 2024, 11, 135–47. [Google Scholar] [CrossRef] [PubMed]
- Caplan, A.I.; Correa, D. PDGF in bone formation and regeneration: New insights into a novel mechanism involving MSCs. Journal Orthopaedic Research 2011, 29, 1795–803. [Google Scholar] [CrossRef]
- Medina-Urrutia, A.X.; Torre-Villalvazo, I.; Juárez-Rojas, J.G. Adipose Tissue Immunometabolism: Unveiling the Intersection of Metabolic and Immune Regulation. Revista de Investigación Clínica 2024, 76, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tian, J.; Wang, L.; et al. Mesenchymal stem cells regulate M1 polarization of peritoneal macrophages through the CARD9-NF-κB signaling pathway in severe acute pancreatitis. J Hepato Biliary Pancreat. 2023, 30, 338–50. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.; Sun, X.; Su, C.; et al. Macrophages—bone marrow mesenchymal stem cells crosstalk in bone healing. Front Cell Dev Biol. 2023, 11, 1193765. [Google Scholar] [CrossRef]
- Wong, C.; Stoilova, I.; Gazeau, F.; et al. Mesenchymal stromal cell derived extracellular vesicles as a therapeutic tool: immune regulation, MSC priming, and applications to SLE. Front Immunol. 2024, 15, 1355845. [Google Scholar] [CrossRef]
- Shi, Z.; He, J.; He, J.; et al. Micro-fragmented adipose tissue regulated the biological functions of osteoarthritis synoviocytes by upregulating MiR-92a-3p expression. Tissue and Cell. 2022, 74, 101716. [Google Scholar] [CrossRef]
- Moncada, M.A.A.; Tamayo, M.A.N.; Encinas, M.A.N.; et al. Immuno-Inflammatory Mechanisms in the Chronification of Pain. Pain Ther. 2026, 15, 443–64. [Google Scholar] [CrossRef]
- Puntillo, F.; Giglio, M.; Corriero, A.; et al. Unraveling the joints: a narrative review of osteoarthritis. European Review for Medical and Pharmacological Sciences 2024, 28, 4080–104. [Google Scholar] [CrossRef]
- Sethi, V.; Anand, C.; Della Pasqua, O. Clinical Assessment of Osteoarthritis Pain: Contemporary Scenario, Challenges, and Future Perspectives. Pain Ther. 2024, 13, 391–408. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Chen, Q.; Zhang, Q.; et al. Therapeutic potential of adipose-derived stem cell extracellular vesicles: from inflammation regulation to tissue repair. Stem Cell Res Ther. 2024, 15, 249. [Google Scholar] [CrossRef]
- Stanciu, N.; Heidari, N.; Slevin, M.; et al. Predicting Long-Term Benefits of Micro-Fragmented Adipose Tissue Therapy in Knee Osteoarthritis: Three-Year Follow-Up on Pain Relief and Mobility. JCM 2025, 14, 4549. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, M.; Zhang, W.; et al. Mechanical strategies to promote vascularization for tissue engineering and regenerative medicine. Burns & Trauma 2024, 12, tkae039. [Google Scholar] [CrossRef]
- Sheikh, AMd; Yano, S.; Tabassum, S.; et al. The Role of the Vascular System in Degenerative Diseases: Mechanisms and Implications. IJMS 2024, 25, 2169. [Google Scholar] [CrossRef]
- Akbarian, M.; Bertassoni, L.E.; Tayebi, L. Biological aspects in controlling angiogenesis: current progress. Cell Mol Life Sci. 2022, 79, 349. [Google Scholar] [CrossRef]
- Pinheiro-Machado, E.; Koster, C.; Smink, A. Modulating adipose-derived stromal cells’ secretomes by culture conditions: effects on angiogenesis, collagen deposition, and immunomodulation. Bioscience Reports 2025, BSR20241389. [Google Scholar] [CrossRef]
- Zhai, K.; Deng, L.; Wu, Y.; et al. Extracellular vesicle-derived miR-146a as a novel crosstalk mechanism for high-fat induced atherosclerosis by targeting SMAD4. Journal of Advanced Research 2025, 73, 729–41. [Google Scholar] [CrossRef]
- Xining, Z.; Sai, L. The Evolving Function of Vasculature and Pro-angiogenic Therapy in Fat Grafting. Cell Transplant. 2024, 33, 09636897241264976. [Google Scholar] [CrossRef] [PubMed]
- Chaliha, D.R.; Lam, V.; Sharif, A.; et al. Microvascular hypoxia and inflammation in chronic pain syndromes. Trends in Molecular Medicine 2025, S1471491425002862. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Xu, M.; Lu, F.; et al. Development of Matrix Metalloproteinases-Mediated Extracellular Matrix Remodeling in Regenerative Medicine: A Mini Review. Tissue Eng Regen Med. 2023, 20, 661–70. [Google Scholar] [CrossRef]
- Brizio, M.; Mancini, M.; Lora, M.; et al. Cytokine priming enhances the antifibrotic effects of human adipose derived mesenchymal stromal cells conditioned medium. Stem Cell Res Ther. 2024, 15, 329. [Google Scholar] [CrossRef] [PubMed]
- AlAdwan, S.; Abosaoda, M.K.; Hassoon, O.A.; et al. Cell-free therapeutics for non-healing wounds: role of MSC-derived exosomes in macrophage polarization, angiogenesis, and fibroblast-mediated ECM remodeling-bridging preclinical insights to clinical translation. Inflammopharmacol 2026, 34, 1343–60. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef]
- Troeberg, L.; Nagase, H. Proteases involved in cartilage matrix degradation in osteoarthritis. Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics 2012, 1824, 133–45. [Google Scholar] [CrossRef]
- Hu, Q.; Ecker, M. Overview of MMP-13 as a Promising Target for the Treatment of Osteoarthritis. IJMS 2021, 22, 1742. [Google Scholar] [CrossRef]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; et al. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Dalal, K. ADAMTS-4 and ADAMTS-5: Key enzymes in osteoarthritis. J Cell Biochem. 2011, 112, 3507–14. [Google Scholar] [CrossRef] [PubMed]
- Brew, K.; Nagase, H. The tissue inhibitors of metalloproteinases (TIMPs): An ancient family with structural and functional diversity. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 2010, 1803, 55–71. [Google Scholar] [CrossRef] [PubMed]
- Søndergaard, R.H.; Højgaard, L.D.; Reese-Petersen, A.L.; et al. Adipose-derived stromal cells increase the formation of collagens through paracrine and juxtacrine mechanisms in a fibroblast co-culture model utilizing macromolecular crowding. Stem Cell Res Ther. 2022, 13, 250. [Google Scholar] [CrossRef]
- Paolella, F.; Manferdini, C.; Gabusi, E.; et al. Effect of microfragmented adipose tissue on osteoarthritic synovial macrophage factors. Journal of Cellular Physiology 2019, 234, 5044–55. [Google Scholar] [CrossRef]
- Joo, H.S.; Suh, J.H.; Lee, H.J.; et al. Current Knowledge and Future Perspectives on Mesenchymal Stem Cell-Derived Exosomes as a New Therapeutic Agent. IJMS 2020, 21, 727. [Google Scholar] [CrossRef]
- Wang, L.; Hu, L.; Zhou, X.; et al. Exosomes secreted by human adipose mesenchymal stem cells promote scarless cutaneous repair by regulating extracellular matrix remodelling. Sci Rep. 2017, 7, 13321. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Shi, J.; et al. Exosomes derived from human adipose mesenchymal stem cells attenuate hypertrophic scar fibrosis by miR-192-5p/IL-17RA/Smad axis. Stem Cell Res Ther. 2021, 12, 221. [Google Scholar] [CrossRef]
- Ragni, E.; Viganò, M.; Torretta, E. Characterization of Microfragmented Adipose Tissue Architecture, Mesenchymal Stromal Cell Content and Release of Paracrine Mediators Located at: 35456324. J Clin Med. 2022, 11, 2231. [Google Scholar] [CrossRef] [PubMed Central]
- Hudetz, D.; Borić, I.; Rod, E.; et al. The Effect of Intra-articular Injection of Autologous Microfragmented Fat Tissue on Proteoglycan Synthesis in Patients with Knee Osteoarthritis. Genes. 2017, 8, 270. [Google Scholar] [CrossRef]
- Corriero, A.; Giglio, M.; Soloperto, R.; et al. Microbial Symphony: Exploring the Role of the Gut in Osteoarthritis-Related Pain. A Narrative Review. Pain Ther. 2024, 13, 409–33. [Google Scholar] [CrossRef]
- Kataria, S.; Patel, U.; Yabut, K.; et al. Recent Advances in Management of Neuropathic, Nociceptive, and Chronic Pain: A Narrative Review with Focus on Nanomedicine, Gene Therapy, Stem Cell Therapy, and Newer Therapeutic Options. Curr Pain Headache Rep. 2024, 28, 321–33. [Google Scholar] [CrossRef]
- Varrassi, G.; Farì, G.; Narvaez Tamayo, M.A.; et al. Mixed pain: clinical practice recommendations. Front Med. 2025, 12, 1659490. [Google Scholar] [CrossRef]
- Ji, R.R.; Chamessian, A.; Zhang, Y.Q. Pain regulation by non-neuronal cells and inflammation. Science 2016, 354, 572–7. [Google Scholar] [CrossRef] [PubMed]
- Huh, Y.; Ji, R.R.; Chen, G. Neuroinflammation, Bone Marrow Stem Cells, and Chronic Pain. Front Immunol. 2017, 8, 1014. [Google Scholar] [CrossRef] [PubMed]
- Latremoliere, A.; Woolf, C.J. Central Sensitization: A Generator of Pain Hypersensitivity by Central Neural Plasticity. The Journal of Pain 2009, 10, 895–926. [Google Scholar] [CrossRef]
- Sacerdote, P.; Niada, S.; Franchi, S.; et al. Systemic Administration of Human Adipose-Derived Stem Cells Reverts Nociceptive Hypersensitivity in an Experimental Model of Neuropathy. Stem Cells and Development 2013, 22, 1252–63. [Google Scholar] [CrossRef] [PubMed]
- Leisengang, S.; Heilen, L.B.; Klymiuk, M.C.; et al. Neuroinflammation in Primary Cultures of the Rat Spinal Dorsal Horn Is Attenuated in the Presence of Adipose Tissue–Derived Medicinal Signalling Cells (AdMSCs) in a Co-cultivation Model. Mol Neurobiol. 2022, 59, 475–94. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.J.; Pi, X.W.; Hu, D.X.; et al. Advances and challenges in cell therapy for neuropathic pain based on mesenchymal stem cells. Front Cell Dev Biol. 2025, 13, 1536566. [Google Scholar] [CrossRef] [PubMed]
- Shiue, S.J.; Rau, R.H.; Shiue, H.S.; et al. Mesenchymal stem cell exosomes as a cell-free therapy for nerve injury–induced pain in rats. Pain 2019, 160, 210–23. [Google Scholar] [CrossRef]
- Fan, B.; Chopp, M.; Zhang, Z.G.; et al. Treatment of diabetic peripheral neuropathy with engineered mesenchymal stromal cell-derived exosomes enriched with microRNA-146a provide amplified therapeutic efficacy. Experimental Neurology 2021, 341, 113694. [Google Scholar] [CrossRef]
- Liu, C.Y.; Yin, G.; Sun, Y.D.; et al. Effect of exosomes from adipose-derived stem cells on the apoptosis of Schwann cells in peripheral nerve injury. CNS Neuroscience & Therapeutics 2020, 26, 189–96. [Google Scholar] [CrossRef]
- Wei, S.; Dong, J.; Hu, Q.; et al. Advances in mesenchymal stem cells and their derivatives for promoting peripheral nerve regeneration. BURNS TRAUMA 2025, 13, tkaf027. [Google Scholar] [CrossRef]
- Amodeo, G.; Niada, S.; Moschetti, G.; et al. Secretome of human adipose-derived mesenchymal stem cell relieves pain and neuroinflammation independently of the route of administration in experimental osteoarthritis. Brain Behav Immun.;PubMed Located at: 33737173. 2021, 94, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Shahrezaei, A.; Sohani, M.; Nasirinezhad, F. Mesenchymal stem cells as a therapeutic strategy to combat oxidative stress-mediated neuropathic pain. Bioimpacts 2025, 15, 30648. [Google Scholar] [CrossRef]
- Salvemini, D.; Little, J.W.; Doyle, T.; et al. Roles of reactive oxygen and nitrogen species in pain. Free Radical Biology and Medicine 2011, 51, 951–66. [Google Scholar] [CrossRef] [PubMed]
- Di Cesare Mannelli, L.; Tenci, B.; Zanardelli, M.; et al. α 7 Nicotinic Receptor Promotes the Neuroprotective Functions of Astrocytes against Oxaliplatin Neurotoxicity. Neural Plasticity 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Guo, B.; Sawkulycz, X.; Heidari, N.; et al. Characterisation of Novel Angiogenic and Potent Anti-Inflammatory Effects of Micro-Fragmented Adipose Tissue. IJMS 2021, 22, 3271. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Chen, J.; Yang, H.; et al. microRNA-26a shuttled by extracellular vesicles secreted from adipose-derived mesenchymal stem cells reduce neuronal damage through KLF9-mediated regulation of TRAF2/KLF2 axis. Adipocyte 2021, 10, 378–93. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zhang, Z.; Zhang, W.; et al. Efficacy and safety of micro-fragmented adipose tissue combined with knee arthroscopy in the treatment of knee osteoarthritis: a systematic review. J Orthop Surg Res. 2025, 20, 646. [Google Scholar] [CrossRef] [PubMed]
- Panchal, J.; Malanga, G.; Sheinkop, M. Safety and Efficacy of Percutaneous Injection of Lipogems Micro-Fractured Adipose Tissue for Osteoarthritic Knees. Am J Orthop (Belle Mead NJ) 2018, 47. [Google Scholar] [PubMed]
- De Groote, J.; Roten, C.; Fomenko, E.; et al. Autologous Micro-Fragmented Adipose Tissue (MFAT) Injections May Be an Effective Treatment for Advanced Knee Osteoarthritis: A Longitudinal Study. JCM 2025, 14, 6571. [Google Scholar] [CrossRef]
- Russo, A.; Condello, V.; Madonna, V.; et al. Autologous and micro-fragmented adipose tissue for the treatment of diffuse degenerative knee osteoarthritis. J exp orthop. 2017, 4, 33. [Google Scholar] [CrossRef]
- Russo, A.; Screpis, D.; Di Donato, S.L.; et al. Autologous micro-fragmented adipose tissue for the treatment of diffuse degenerative knee osteoarthritis: an update at 3 year follow-up. J exp orthop. 2018, 5, 52. [Google Scholar] [CrossRef]
- Agarwal, N.; Mak, C.; Bojanic, C.; et al. Meta-Analysis of Adipose Tissue Derived Cell-Based Therapy for the Treatment of Knee Osteoarthritis. Cells 2021, 10, 1365. [Google Scholar] [CrossRef]
- Veronesi, F.; Andriolo, L.; Salerno, M.; et al. Adipose Tissue-Derived Minimally Manipulated Products versus Platelet-Rich Plasma for the Treatment of Knee Osteoarthritis: A Systematic Review of Clinical Evidence and Meta-Analysis. JCM 2023, 13, 67. [Google Scholar] [CrossRef]
- Usuelli, F.G.; Grassi, M.; Maccario, C.; et al. Intratendinous adipose-derived stromal vascular fraction (SVF) injection provides a safe, efficacious treatment for Achilles tendinopathy: results of a randomized controlled clinical trial at a 6-month follow-up. Knee Surg Sports Traumatol Arthrosc. 2018, 26, 2000–10. [Google Scholar] [CrossRef]
- Senesi, L.; De Francesco, F.; Marchesini, A.; et al. Efficacy of Adipose-Derived Mesenchymal Stem Cells and Stromal Vascular Fraction Alone and Combined to Biomaterials in Tendinopathy or Tendon Injury: Systematic Review of Current Concepts. Medicina 2023, 59, 273. [Google Scholar] [CrossRef] [PubMed]
- Andia, I.; Maffulli, N. Muscle and Tendon Injuries: The Role of Biological Interventions to Promote and Assist Healing and Recovery. Arthroscopy 2015, 31, 999–1015. [Google Scholar] [CrossRef]
- Chen, S.H.; Chen, Z.Y.; Lin, Y.H.; et al. Extracellular Vesicles of Adipose-Derived Stem Cells Promote the Healing of Traumatized Achilles Tendons. IJMS 2021, 22, 12373. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, M.; Shi, M.; et al. Adipose-derived mesenchymal stromal cell-derived exosomes promote tendon healing by activating both SMAD1/5/9 and SMAD2/3. Stem Cell Res Ther. 2021, 12, 338. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, X.; Peng, H.; et al. Effects of injecting micro-fragmented adipose tissue (MFAT) in the tendon-bone junction region during anterior cruciate ligament reconstruction on postoperative tendon-bone healing: a protocol for a randomised controlled trial in China. BMJ Open 2025, 15, e104249. [Google Scholar] [CrossRef] [PubMed]
- Zaffagnini, S.; Andriolo, L.; Boffa, A.; et al. Microfragmented Adipose Tissue Versus Platelet-Rich Plasma for the Treatment of Knee Osteoarthritis: A Prospective Randomized Controlled Trial at 2-Year Follow-up. Am J Sports Med. 2022, 50, 2881–92. [Google Scholar] [CrossRef]
- Bisicchia, S.; Bernardi, G.; Pagnotta, S.M.; et al. Micro-fragmented stromal-vascular fraction plus microfractures provides better clinical results than microfractures alone in symptomatic focal chondral lesions of the knee. Knee Surg Sports Traumatol Arthrosc. 2020, 28, 1876–84. [Google Scholar] [CrossRef]
- Randelli, P.S.; Cucchi, D.; Fossati, C.; et al. Arthroscopic Rotator Cuff Repair Augmentation With Autologous Microfragmented Lipoaspirate Tissue Is Safe and Effectively Improves Short-term Clinical and Functional Results: A Prospective Randomized Controlled Trial With 24-Month Follow-up. Am J Sports Med. 2022, 50, 1344–57. [Google Scholar] [CrossRef]
- Yuan, C.; Song, W.; Jiang, X.; et al. Adipose-derived stem cell-based optimization strategies for musculoskeletal regeneration: recent advances and perspectives. Stem Cell Res Ther. 2024, 15, 91. [Google Scholar] [CrossRef]
- Grossi, P.; Giarratana, S.; Cernei, S.; et al. Low back pain treated with disc decompression and autologous micro-fragmented adipose tissue: a case report. CellR4 2016, 4, e1770. [Google Scholar]
- Noriega, D.C.; Ardura, F.; Hernández-Ramajo, R.; et al. Treatment of Degenerative Disc Disease With Allogeneic Mesenchymal Stem Cells: Long-term Follow-up Results. Transplantation 2021, 105, e25–7. [Google Scholar] [CrossRef]
- Kumar, H.; Ha, D.H.; Lee, E.J.; et al. Safety and tolerability of intradiscal implantation of combined autologous adipose-derived mesenchymal stem cells and hyaluronic acid in patients with chronic discogenic low back pain: 1-year follow-up of a phase I study. Stem Cell Res Ther. 2017, 8, 262. [Google Scholar] [CrossRef]
- Ceccarelli, S.; Pontecorvi, P.; Anastasiadou, E.; et al. Immunomodulatory Effect of Adipose-Derived Stem Cells: The Cutting Edge of Clinical Application. Front Cell Dev Biol. 2020, 8, 236. [Google Scholar] [CrossRef]
- Lin, Y.; Mu, D. Immunomodulatory effect of human dedifferentiated fat cells: comparison with adipose-derived stem cells. Cytotechnology 2023, 75, 231–42. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, L.; Zhang, Z.; et al. Extracellular vesicles mediate the communication of adipose tissue with brain and promote cognitive impairment associated with insulin resistance. Cell Metabolism 2022, 34, 1264–1279.e8. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.L.; De Luca, K.; Haile, L.M.; et al. Global, regional, and national burden of low back pain, 1990–2020, its attributable risk factors, and projections to 2050: a systematic analysis of the Global Burden of Disease Study 2021. The Lancet Rheumatology 2023, 5, e316–29. [Google Scholar] [CrossRef] [PubMed]
- Vadalà, G.; Ambrosio, L.; Russo, F.; et al. Interaction between Mesenchymal Stem Cells and Intervertebral Disc Microenvironment: From Cell Therapy to Tissue Engineering. Stem Cells International 2019, 2019, 1–15. [Google Scholar] [CrossRef]
- Risbud, M.V.; Shapiro, I.M. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol. 2014, 10, 44–56. [Google Scholar] [CrossRef]
- Johnson, Z.; Schoepflin, Z.; Choi, H.; et al. Disc in flames: Roles of TNF-α and IL-1β in intervertebral disc degeneration. eCM 2015, 30, 104–17. [Google Scholar] [CrossRef]
- Li, Z.P.; Li, H.; Ruan, Y.H.; et al. Stem cell therapy for intervertebral disc degeneration: Clinical progress with exosomes and gene vectors. World J Stem Cells 2025, 17, 102945. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ishiguro, H.; Kaito, T.; Yarimitsu, S.; et al. Intervertebral disc regeneration with an adipose mesenchymal stem cell-derived tissue-engineered construct in a rat nucleotomy model. Acta Biomaterialia 2019, 87, 118–29. [Google Scholar] [CrossRef]
- Comella, K.; Silbert, R.; Parlo, M. Effects of the intradiscal implantation of stromal vascular fraction plus platelet rich plasma in patients with degenerative disc disease. J Transl Med. 2017, 15, 12. [Google Scholar] [CrossRef]
- Schol, J.; Tamagawa, S.; Volleman, T.N.E.; et al. A comprehensive review of cell transplantation and platelet-rich plasma therapy for the treatment of disc degeneration-related back and neck pain: A systematic evidence-based analysis. JOR Spine 2024, 7, e1348. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Park, K.S.; Shin, H.E.; et al. Safety and Feasibility of Intradiscal Administration of Matrilin-3-Primed Adipose-Derived Mesenchymal Stromal Cell Spheroids for Chronic Discogenic Low Back Pain: Phase 1 Clinical Trial. IJMS 2023, 24, 16827. [Google Scholar] [CrossRef] [PubMed]
- Pers, Y.M.; Soler-Rich, R.; Vadalà, G.; et al. Allogenic bone marrow–derived mesenchymal stromal cell–based therapy for patients with chronic low back pain: a prospective, multicentre, randomised placebo controlled trial (RESPINE study). Annals of the Rheumatic Diseases 2024, 83, 1572–83. [Google Scholar] [CrossRef] [PubMed]
- Vadalà, G.; Russo, F.; Lavazza, C.; et al. Intradiscal Mesenchymal Stromal Cell Therapy for the Treatment of Low Back Pain Due to Moderate-to-Advanced Multilevel Disc Degeneration: A Preliminary Report of a Double-Blind, Phase IIB Randomized Clinical Trial ( DREAM Study). JOR Spine 2025, 8, e70086. [Google Scholar] [CrossRef]
- Conti, M.; Giovannini, M.; Bianchini, A.; et al. Microdiscectomy Combined with Autologous Microfragmented Adipose Tissue Injection for the Treatment of Low Back Pain due to Intervertebral Lumbar Disc Degeneration [Internet]. Research Square; 2024. Available online: https://www.researchsquare.com/article/rs-4837039/v1 (accessed on 4 April 2026). [CrossRef]
- Tilotta, V.; Vadalà, G.; Ambrosio, L.; et al. Wharton’s Jelly mesenchymal stromal cell-derived extracellular vesicles promote nucleus pulposus cell anabolism in an in vitro 3D alginate-bead culture model. JOR Spine 2024, 7, e1274. [Google Scholar] [CrossRef]
- Munda, M.; Velnar, T. Stem cell therapy for degenerative disc disease: Bridging the gap between preclinical promise and clinical potential. Biomolecules and Biomedicine 2024, 24, 210–8. [Google Scholar] [CrossRef] [PubMed]
- Soufi, K.H.; Castillo, J.A.; Rogdriguez, F.Y.; et al. Potential Role for Stem Cell Regenerative Therapy as a Treatment for Degenerative Disc Disease and Low Back Pain: A Systematic Review. IJMS 2023, 24, 8893. [Google Scholar] [CrossRef] [PubMed]
- Manchikanti, L.; Kaye, A.D.; Soin, A.; et al. Comprehensive Evidence-Based Guidelines for Facet Joint Interventions in the Management of Chronic Spinal Pain: American Society of Interventional Pain Physicians (ASIPP) Guidelines Facet Joint Interventions 2020 Guidelines. Pain Physician 2020, 23, S1–127. [Google Scholar] [PubMed]
- Perolat, R.; Kastler, A.; Nicot, B.; et al. Facet joint syndrome: from diagnosis to interventional management. Insights Imaging 2018, 9, 773–89. [Google Scholar] [CrossRef]
- Cohen, S.P.; Huang, J.H.Y.; Brummett, C. Facet joint pain—advances in patient selection and treatment. Nat Rev Rheumatol. 2013, 9, 101–16. [Google Scholar] [CrossRef]
- Occhigrossi, F.; Carpenedo, R.; Leoni, M.L.G.; et al. Delphi-Based Expert Consensus Statements for the Management of Percutaneous Radiofrequency Neurotomy in the Treatment of Lumbar Facet Joint Syndrome. Pain Ther. 2023, 12, 863–77. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rothoerl, R.; Tomelden, J.; Alt, E.U. Safety and Efficacy of Autologous Stem Cell Treatment for Facetogenic Chronic Back Pain. J Pers Med. 2023, 13, 436. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Qu, W.; Yan, D.; Durand, N.C.; et al. Intra-articular delivery of allogeneic bone marrow derived mesenchymal stromal cells (BM-MSCs) for painful lumbar facet arthropathy: a phase I clinical trial. Stem Cell Res Ther. 2025, 16, 596. [Google Scholar] [CrossRef]
- Lonardi, R.; Leone, N.; Gennai, S.; et al. Autologous micro-fragmented adipose tissue for the treatment of diabetic foot minor amputations: a randomized controlled single-center clinical trial (MiFrAADiF). Stem Cell Res Ther. 2019, 10, 223. [Google Scholar] [CrossRef] [PubMed]
- Stark, L.; Razzaque, M.; Yoon, J.; et al. Safety and Feasibility of Autologous Micro-Fragmented Adipose Tissue Injections for the Treatment of Vaginal Atrophy, Vulvovaginal Dystrophy, and Stress Urinary Incontinence: An Observational Case Series. EMJ. 2020, 8, 29–37. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. 510(k) Premarket Notification K161636: Lipogems System [Internet]. Silver Spring, MD: U.S. Food and Drug Administration, Center for Devices and Radiological Health; 2016. Available online: https://www.accessdata.fda.gov/cdrh_docs/pdf16/k161636.pdf (accessed on 4 April 2026).
- Lipogems International S.p.A. Lipogems [Internet]. Milan, Italy: Lipogems International; 2026. Available online: https://www.lipogems.com/en/documentation/ (accessed on 4 April 2026).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




