Submitted:
11 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction: What Determines What a Body Can Do?
1.1. The ARCH × Φ Framework
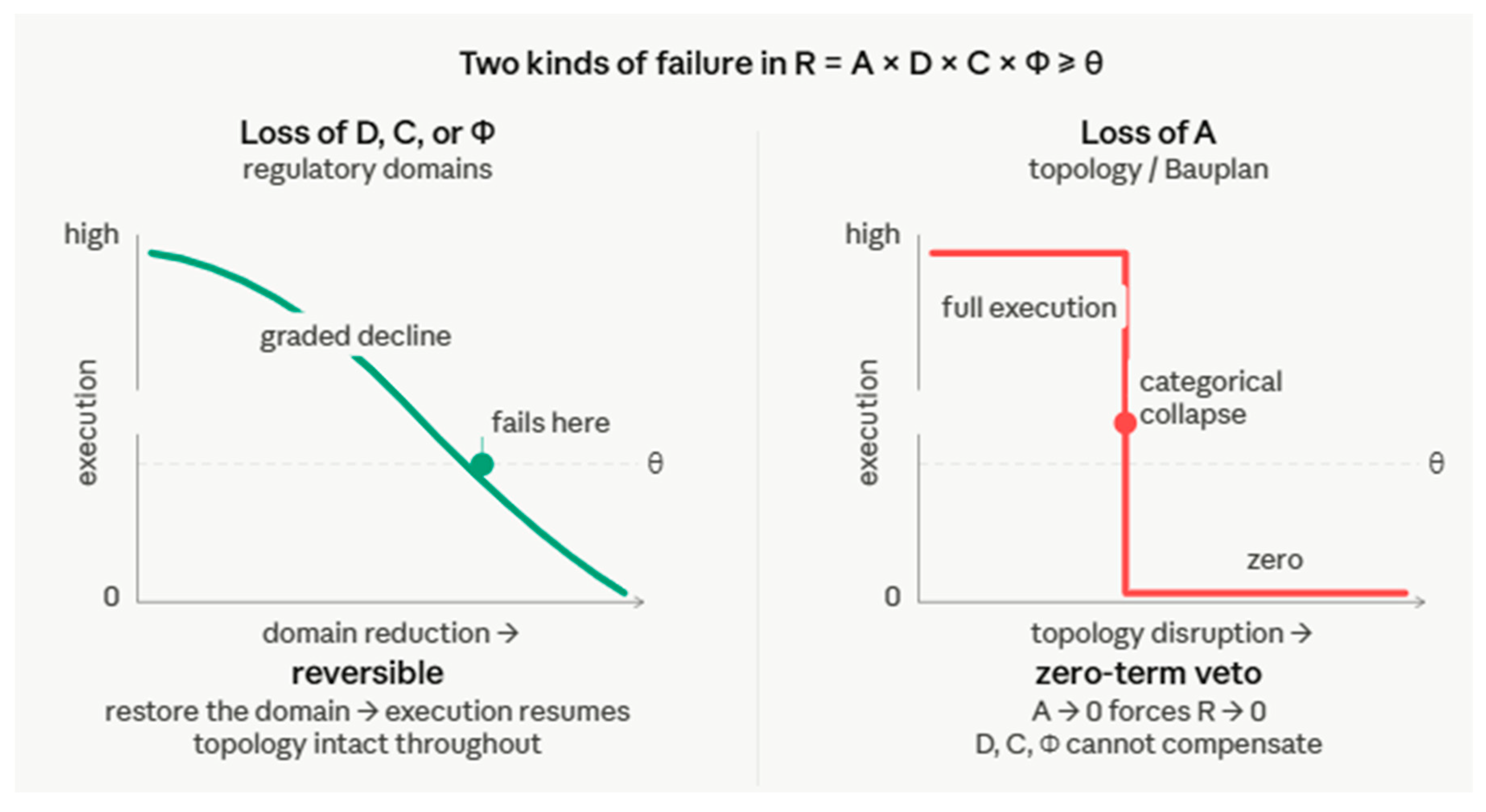
1.2. The A-Domain Gap
1.3. Three Empirical Anchors and a Formal Claim

2. What the ARCH × Φ Framework Is: Three Instantiations
2.1. DNA Replication: A Molecular Zero-Term Veto
2.2. Clownfish Sex Change: Hysteresis and Irreversibility
2.3. Venus Flytrap: Zero-Term Veto Without a Nervous System
3. Redefining A, D, C, and Φ for Organ Morphology
3.1. Archetype and Topology
3.2. A — ECM Scaffold Topology and Circuit Geometry
3.3. D — Metabolic-Hemodynamic Demand
3.4. C — The Releasing Trigger
3.5. Φ — YAP/TAZ, Chromatin State, and the Sterol Connection
3.6. Are Sterols Involved in Epigenetic Φ?
3.7. Cellular Bauplan: Intracellular Topology as the A-Domain
4. Empirical Proof Line 1: Deep Homology and the Appendage Bauplan
4.1. The ZRS Enhancer: Bauplan Encoded in 770 Base Pairs
4.2. Paddlefish: Bauplan Predates the Structure It Specifies
4.3. The Snake Series: A → 0 from Bauplan Erosion Across Evolutionary Time
5. Empirical Proof Line 2: Organ Topology as Bauplan
5.1. The Heart: ECM Scaffold and the Conduction System Bauplan
5.2. The Liver: Hepatostat, Lobular Topology, and Zonation
5.3. The Kidney Nephron: Geometry as Mechanistic Prerequisite
6. Empirical Proof Line 3: Minimal Systems
6.1. C. elegans: The Connectome as Literal Bauplan
6.2. Zebrafish: Vertebrate Escape Circuit Bauplan
6.3. Smith-Lemli-Opitz Syndrome: One Molecule, Two Roles
7. The Four-Tier A-Domain Hierarchy: From Phylotypic to State
7.1. The Tier Structure
| Tier | Content | Set by | Timescale | Reversibility |
|---|---|---|---|---|
| I — Phylotypic | Body axis, organ topology, Hox patterning, ZRS-Shh circuit | Gastrulation and early organogenesis | Once per lifetime, irreversible | None on behavioral timescales |
| II — Ontogenetic | Adult size, organ proportions, allometry | Growth and metabolic rate | Developmental time | Limited; constrained by Tier I |
| III — Functional | ECM collagen topology, circuit wiring, receptor density | Development plus experience | Years; plastic | Partial; can be driven by Φ lock |
| IV — State | Posture, arousal, muscular pre-loading | Moment-to-moment signals | Seconds to minutes | Complete; what ARCH × Φ gates |
7.2. YAP/TAZ Is Φ, Not A
8. Φ Lock Driving Tier III A Remodeling: Cardiac Hypertrophy and Fibrosis
8.1. Two Simultaneous Processes
8.2. The LVAD Dissociation
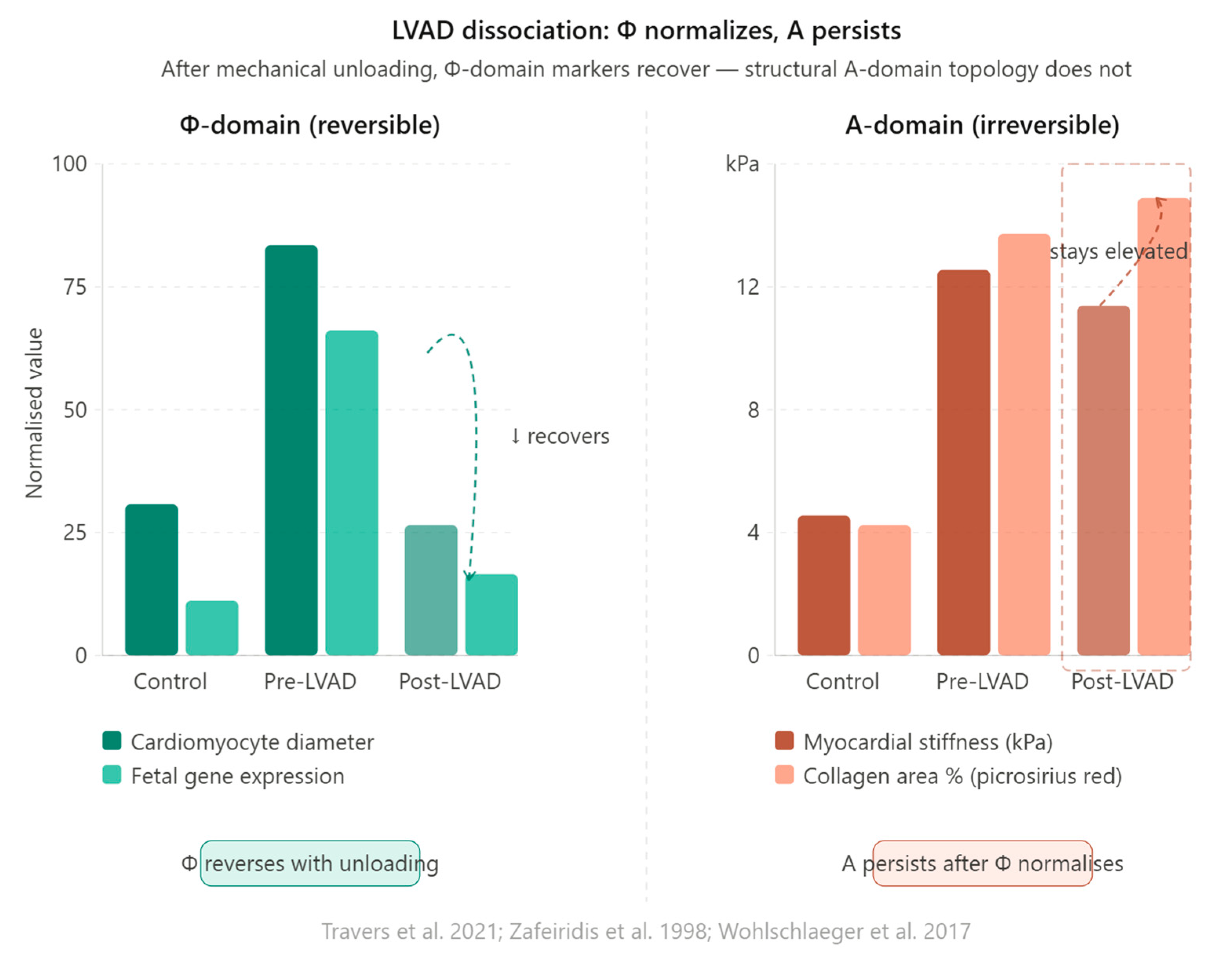
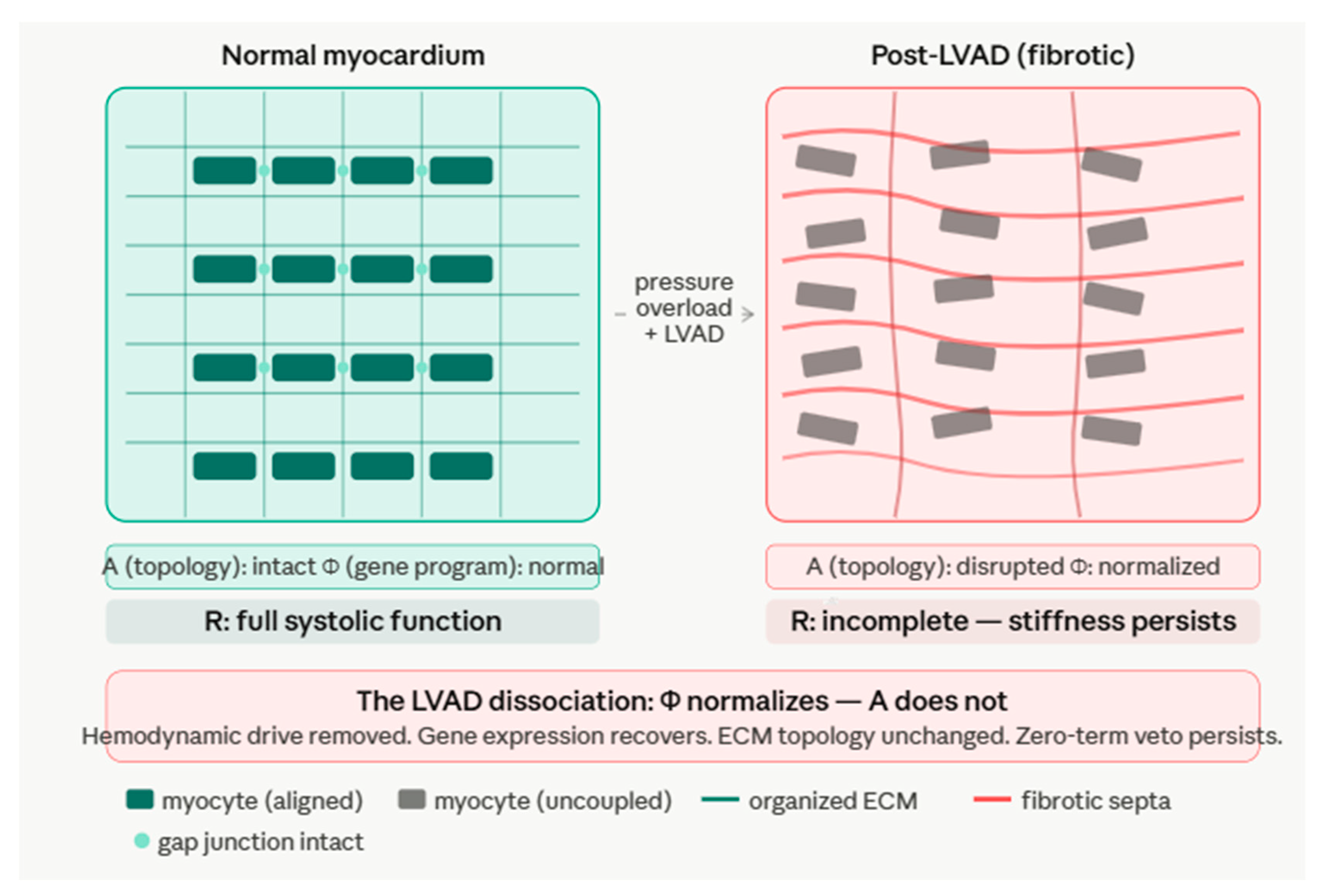
8.3. The General Rule
9. Quantification: Topological Connectivity Density
9.1. The Problem
9.2. Fractal Dimension and TCD
9.3. Validation and Properties
| Failure Mode | Mechanism | Clinical Example | ARCH Prediction | Φ-Therapy Expected Effective? |
|---|---|---|---|---|
| A → 0 (congenital) | Bauplan topology never formed; ZRS/Hox/ECM morphogen disruption | Congenital heart defects (ISL1 mutation); polydactyly (ZRS mutation); SLO syndrome | Categorical execution failure irreversible by D, C, or Φ manipulation | No — A must be rebuilt; not addressable by Φ modulation |
| A erosion (acquired) | Progressive Bauplan destruction; cirrhosis, DCM, MS plaques | Cirrhosis → metabolic zonation loss; DCM → systolic failure; MS → conduction block | Execution failure proportional to A erosion; no compensation threshold | Only if A template survives; ineffective once template destroyed |
| Φ lock → A remodeling | Sustained Φ-active state drives Tier III ECM remodeling beyond reversibility threshold | Pathological cardiac hypertrophy → fibrosis; pressure-overload DCM | Partial irreversibility; Φ-tier reversal possible but collagen topology persists | Effective if applied before fibroblast mechano-sensing uncoupling; insufficient after |
Disclosures
References
- Antos, CL; McKinsey, TA; Dreitz, M; Hollingsworth, LM; Zhang, CL; Schreiber, K; et al. Dose-dependent blockade to cardiomyocyte hypertrophy by histone deacetylase inhibitors. J Biol Chem. 2003, 278(31), 28930–7. [Google Scholar] [CrossRef] [PubMed]
- Avery, L; Horvitz, HR. Pharyngeal pumping continues after laser killing of the pharyngeal nervous system of C. elegans. Neuron 1989, 3(4), 473–85. [Google Scholar] [CrossRef]
- Baicu, CF; Stroud, JD; Livesay, VA; Hapke, E; Holder, J; Spinale, FG; et al. Changes in extracellular collagen matrix alter myocardial systolic performance. Am J Physiol Heart Circ Physiol. 2003, 284(1), H122–32. [Google Scholar] [CrossRef]
- Baptista, PM; Siddiqui, MM; Lozier, G; Rodriguez, SR; Atala, A; Soker, S. The use of whole organ decellularization for the generation of a vascularized liver organoid. Hepatology 2011, 53(2), 604–17. [Google Scholar] [CrossRef]
- Barr, AR; Heldt, FS; Zhang, T; Bakal, C; Novak, B. A dynamical framework for the all-or-none G1/S transition. Cell Syst. 2016, 2(1), 27–37. [Google Scholar] [CrossRef]
- Bell, SP; Dutta, A. DNA replication in eukaryotic cells. Annu Rev Biochem. 2002, 71, 333–74. [Google Scholar] [CrossRef] [PubMed]
- Birks, EJ; Tansley, PD; Hardy, J; George, RS; Bowles, CT; Burke, M; et al. Left ventricular assist device and drug therapy for the reversal of heart failure. N Engl J Med. 2006, 355(18), 1873–84. [Google Scholar] [CrossRef] [PubMed]
- Böhm, J; Scherzer, S; Krol, E; Kreuzer, I; von Meyer, K; Lorey, C; et al. The Venus flytrap Dionaea muscipula counts prey-induced action potentials to induce sodium uptake. Curr Biol. 2016, 26(3), 286–95. [Google Scholar] [CrossRef]
- Bruckner, BA; Stetson, SJ; Perez-Verdia, A; Youker, KA; Radovancevic, B; Connelly, JH; et al. Regression of fibrosis and hypertrophy in failing myocardium following mechanical circulatory support. J Heart Lung Transplant. 2001, 20(4), 457–64. [Google Scholar] [CrossRef]
- Camargo, FD; Gokhale, S; Johnnidis, JB; Fu, D; Bell, GW; Jaenisch, R; et al. YAP1 increases organ size and expands undifferentiated progenitor cells. Curr Biol. 2007, 17(23), 2054–60. [Google Scholar] [CrossRef]
- Campagnola, PJ; Loew, LM. Second-harmonic imaging microscopy for visualizing biomolecular arrays in cells, tissues and organisms. Nat Biotechnol. 2003, 21(11), 1356–60. [Google Scholar] [CrossRef] [PubMed]
- Casas, L; Saborido-Rey, F; Ryu, T; Michell, C; Ravasi, T; Irigoien, X. Sex change in clownfish: molecular insights from transcriptome analysis. Sci Rep. 2016, 6, 35461. [Google Scholar] [CrossRef] [PubMed]
- Casey, DT; Lahue, KG; Mori, V; Herrmann, J; Hall, JK; Suki, B; et al. Local fractal dimension of collagen detects increased spatial complexity in fibrosis. Histochem Cell Biol. 2024, 161(1), 29–42. [Google Scholar] [CrossRef] [PubMed]
- Christoffels, VM; Moorman, AF. Development of the cardiac conduction system: why are some regions of the heart more arrhythmogenic than others? Circ Arrhythm Electrophysiol. 2009, 2(2), 195–207. [Google Scholar] [CrossRef]
- Cobham, AE; Mirth, CK. The development of body and organ shape. BMC Zool 2020, 5, 14. [Google Scholar] [CrossRef]
- Davis, MC; Dahn, RD; Shubin, NH. An autopodial-like pattern of Hox expression in the fins of a basal actinopterygian fish. Nature 2007, 447(7143), 473–6. [Google Scholar] [CrossRef]
- Derrick, CJ; Sanchez-Posada, J; Hussein, F; Tessadori, F; Pollitt, EJ; Savage, AM; et al. Asymmetric Hapln1a drives regionalized cardiac ECM expansion and promotes heart morphogenesis in zebrafish development. Cardiovasc Res. 2022, 118(1), 226–40. [Google Scholar] [CrossRef]
- Dong, J; Feldmann, G; Huang, J; Wu, S; Zhang, N; Comerford, SA; et al. Elucidation of a universal size-control mechanism in Drosophila and mammals. Cell. 2007, 130(6), 1120–33. [Google Scholar] [CrossRef]
- Eftimie, LG; Padrez, Y; Golubewa, L; Rutkauskas, D; Hristu, R. Widefield polarization-resolved second harmonic generation imaging of entire thyroid nodule sections for the detection of capsular invasion. Biomed Opt Express 2024, 15(8), 4705–18. [Google Scholar] [CrossRef]
- Farris, SD; Don, C; Helterline, D; Costa, C; Plummer, T; Steffes, S; Mahr, C; Mokadam, NA; Stempien-Otero, A. Cell-Specific Pathways Supporting Persistent Fibrosis in Heart Failure J Am Coll Cardiol. 2017, 70(3), 344–354. [Google Scholar] [CrossRef]
- Fausto, N; Campbell, JS; Riehle, KJ. Liver regeneration. J Hepatol. 2012, 57(3), 692–4. [Google Scholar] [CrossRef] [PubMed]
- Gilham, D; Wasiak, S; Daze, E; Boulet, L; Laliberte, S; Chami, M; et al. BET inhibition reduces hepatic steatosis and liver fibrosis through a BET-bromodomain- and cholesterol-dependent mechanism in HFD-fed mice. Eur J Pharmacol. 2022, 924, 174964. [Google Scholar] [CrossRef]
- Hafen, E; Stocker, H. How are the sizes of cells, organs, and bodies controlled? PLoS Biol. 2003, 1(3), e86. [Google Scholar] [CrossRef]
- Hall, JK; Mori, V; Herrmann, J; Casey, DT; Suki, B; Bates, JH; et al. Imaging the extracellular matrix structure and remodeling in healthy, fibrotic, and emphysematous human precision-cut lung slices. Respir Res. 2025, 26. [Google Scholar] [CrossRef]
- Hang, CT; Yang, J; Han, P; Cheng, HL; Shang, C; Ashley, E; et al. Chromatin regulation by Brg1 underlies heart muscle development and disease. Nature 2010, 466(7302), 62–7. [Google Scholar] [CrossRef]
- Hedrich, R; Salvador-Recatala, V; Dreyer, I. Electrical wiring and long-distance plant communication. Trends Plant Sci. 2016, 21(5), 376–87. [Google Scholar] [CrossRef]
- Hecker, A; Schulze, W; Oster, J; Richter, DO; Schuster, S. Removing a single neuron in a vertebrate brain forever abolishes an essential behavior. Proc Natl Acad Sci USA 2020, 117(6), 3254–60. [Google Scholar] [CrossRef]
- Huch, M; Gehart, H; van Boxtel, R; Hamer, K; Blokzijl, F; Verstegen, MM; et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell. 2015, 160(1-2), 299–312. [Google Scholar] [CrossRef]
- Jiang, QX. Cholesterol-dependent gating effects on ion channels. Adv Exp Med Biol. 2019, 1115, 167–90. [Google Scholar] [CrossRef] [PubMed]
- Kee, HJ; Sohn, IS; Nam, KI; Park, JE; Qian, YR; Yin, Z; et al. Inhibition of histone deacetylation blocks cardiac hypertrophy induced by angiotensin II infusion and aortic banding. Circulation 2006, 113(1), 51–9. [Google Scholar] [CrossRef]
- Kemper, JK; Xiao, Z; Ponugoti, B; Miao, J; Fang, S; Kanamaluru, D; et al. FXR acetylation is normally dynamically regulated by p300 and SIRT1 but constitutively elevated in metabolic disease states. Cell Metab. 2009, 10(5), 392–404. [Google Scholar] [CrossRef]
- Kimmel, CB; Sessions, SK; Kimmel, RJ. Morphogenesis and synaptogenesis of the zebrafish Mauthner neuron. J Comp Neurol. 1981, 198(1), 101–20. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, CB; Eaton, RC; Powell, SL. Decreased fast-start performance of zebrafish larvae lacking Mauthner neurons. J Comp Physiol. 1980, 140, 343–50. [Google Scholar] [CrossRef]
- Kohashi, T; Oda, Y. Initiation of Mauthner- or non-Mauthner-mediated fast escape evoked by different modes of sensory input. J Neurosci. 2008, 28(42), 10641–53. [Google Scholar] [CrossRef]
- Koshman, YE; Patel, N; Chu, M; Iyengar, R; Kim, T; Ersahin, C; Lewis, W; Heroux, A; Samarel, AM. Regulation of connective tissue growth factor gene expression and fibrosis in human heart failure. J Card Fail. 2013, 19(4), 283–94. [Google Scholar] [CrossRef]
- Kvon, EZ; Kamneva, OK; Melo, US; Barozzi, I; Osterwalder, M; Mannion, BJ; et al. Progressive loss of function in a limb enhancer during snake evolution. Cell. 2016, 167(3), 633–642.e11. [Google Scholar] [CrossRef]
- Leal, F; Cohn, MJ. Loss and re-emergence of legs in snakes by modular evolution of Sonic hedgehog and HOXD enhancers. Curr Biol. 2016, 26(21), 2966–73. [Google Scholar] [CrossRef]
- Lettice, LA; Heaney, SJ; Purdie, LA; Li, L; de Beer, P; Oostra, BA; et al. A long-range Shh enhancer regulates expression in the developing limb and fin and is associated with preaxial polydactyly. Hum Mol Genet. 2003, 12(14), 1725–35. [Google Scholar] [CrossRef]
- Lettice, LA; Hill, AE; Devenney, PS; Hill, RE. Point mutations in a distant Sonic hedgehog cis-regulator generate a variable regulatory output responsible for preaxial polydactyly. Hum Mol Genet. 2008, 17(7), 978–85. [Google Scholar] [CrossRef] [PubMed]
- Lettice, LA; Devenney, P; De Angelis, C; Hill, RE. The conserved Sonic hedgehog limb enhancer consists of discrete functional elements that regulate precise spatial expression. Cell Rep. 2017, 20(6), 1396–408. [Google Scholar] [CrossRef] [PubMed]
- Mazza, G; Rombouts, K; Hall, AR; Urbani, L; Luong, TV; Al-Akkad, W; et al. Decellularized human liver as a natural 3D-scaffold for liver bioengineering and transplantation. Sci Rep. 2015, 5, 13079. [Google Scholar] [CrossRef]
- Meaney, S. Epigenetic regulation of cholesterol homeostasis. Front Genet. 2014, 5, 311. [Google Scholar] [CrossRef]
- Michalopoulos, GK; DeFrances, MC. Liver regeneration. Science 1997, 276(5309), 60–6. [Google Scholar] [CrossRef] [PubMed]
- Miyaoka, Y; Ebato, K; Kato, H; Arakawa, S; Shimizu, S; Miyajima, A. Hypertrophy and unconventional cell division of hepatocytes underlie liver regeneration. Curr Biol. 2012, 22(13), 1166–75. [Google Scholar] [CrossRef]
- Motola, DL; Cummins, CL; Rottiers, V; Sharma, KK; Li, T; Li, Y; et al. Identification of ligands for DAF-12 that govern dauer formation and reproduction in C. elegans. Cell. 2006, 124(6), 1209–23. [Google Scholar] [CrossRef]
- Mwinyi, J; Lindberg, J; Hamsten, A; Hellenius, ML; Lagercrantz, J; Orho-Melander, M; et al. Zebrafish models of skeletal dysplasia induced by cholesterol biosynthesis deficiency. Dis Model Mech. 2020, 13(6), dmm042549. [Google Scholar] [CrossRef]
- Naoumov, NV; Brees, D; Locker, J; et al. Digital pathology with artificial intelligence analyses provides greater insights into treatment-induced fibrosis regression in NASH. J Hepatol. 2022, 77(5), 1399–409. [Google Scholar] [CrossRef] [PubMed]
- Parker, CG; Gruenhagen, GW; Hegarty, BE; Histed, AR; Streelman, JT; Rhodes, JS; et al. Adult sex change leads to extensive forebrain reorganization in clownfish. Biol Sex Differ. 2024, 15(1), 58. [Google Scholar] [CrossRef]
- Parker, CG; Lee, JS; Histed, AR; Craig, SE; Rhodes, JS. Stable and persistent male-like behavior during male-to-female sex change in the common clownfish Amphiprion ocellaris. Horm Behav. 2022, 145, 105239. [Google Scholar] [CrossRef]
- Piccolo, S; Dupont, S; Cordenonsi, M. The biology of YAP/TAZ: Hippo signaling and beyond. Physiol Rev. 2014, 94(4), 1287–312. [Google Scholar] [CrossRef]
- Porter, FD. Smith-Lemli-Opitz syndrome: pathogenesis, diagnosis and management. Eur J Hum Genet. 2008, 16(5), 535–41. [Google Scholar] [CrossRef] [PubMed]
- Porter, JA; Young, KE; Beachy, PA. Cholesterol modification of hedgehog signaling proteins in animal development. Science 1996, 274(5285), 255–9. [Google Scholar] [CrossRef] [PubMed]
- Rahman, T; Zorumski, CF; Meloy, JR. The ARCH model: a neuroevolutionary framework for behavioral execution. Front Psychiatry 2025, 16, 1669530. [Google Scholar] [CrossRef] [PubMed]
- Rahman, T. A multiplicative behavioral model of DNA replication initiation in cells. Open Life Sci. 2025, 20(1), 20251229. [Google Scholar] [CrossRef]
- Rahman, T. Sex change in clownfish as an ARCH-governed biological decision. Horm Behav. 2026, 180, 105907. [Google Scholar] [CrossRef]
- Rahman, T. Threshold decision-making in the Venus flytrap. Plant Sci. 2026, 365, 113010. [Google Scholar] [CrossRef]
- Sagai, T; Hosoya, M; Mizushina, Y; Tamura, M; Shiroishi, T. Elimination of a long-range cis-regulatory module causes complete loss of limb-specific Shh expression and truncation of the mouse limb. Development 2005, 132(4), 797–803. [Google Scholar] [CrossRef]
- Saito, S; Matsumiya, G; Sakaguchi, T; Miyagawa, S; Yamauchi, T; Kuratani, T; et al. Cardiac fibrosis and cellular hypertrophy decrease the degree of reverse remodeling and improvement in cardiac function during left ventricular assist device support. J Heart Lung Transplant. 2010, 29, 672–9. [Google Scholar] [CrossRef]
- Shubin, N; Tabin, C; Carroll, S. Deep homology and the origins of evolutionary novelty. Nature 2009, 457(7231), 818–23. [Google Scholar] [CrossRef]
- Sun, W; Luo, Y; Lee, AM; Bhargava, A; Sato, M; Bhate, S; et al. Nonlinear optical microscopy: use of second harmonic generation and two-photon microscopy for automated quantitative liver fibrosis studies. J Biomed Opt. 2008, 13(6), 064010. [Google Scholar] [CrossRef]
- Travers, JG; Wennersten, SA; Pena, B; Bagchi, RA; Smith, HE; Hirsch, RA; et al. HDAC inhibition reverses preexisting diastolic dysfunction and blocks covert extracellular matrix remodeling. Circulation 2021, 143(19), 1874–90. [Google Scholar] [CrossRef]
- Wang, Y; Chen, L; Pandak, WM; Heuman, D; Hylemon, PB; Ren, S. High glucose induces lipid accumulation via 25-hydroxycholesterol DNA-CpG methylation. iScience 2020, 23(5), 101102. [Google Scholar] [CrossRef]
- Weber, MC; Schmidt, K; Buck, A; Kasajima, A; Becker, S; Li, C; et al. Fractal analysis of extracellular matrix for observer-independent quantification of intestinal fibrosis in Crohn's disease. Sci Rep. 2024, 14(1), 3988. [Google Scholar] [CrossRef]
- Weber, KT; Sun, Y; Katwa, LC. Myofibroblasts and local angiotensin II in rat cardiac tissue repair. Int J Biochem Cell Biol. 1994, 26(3), 323–42. [Google Scholar] [CrossRef]
- Weatherbee, SD; Halder, G; Kim, J; Hudson, A; Carroll, S. Ultrabithorax regulates genes at several levels of the wing-patterning hierarchy to shape the development of the Drosophila haltere. Genes Dev. 1998, 12(10), 1474–82. [Google Scholar] [CrossRef]
- White, JG; Southgate, E; Thomson, JN; Brenner, S. The structure of the nervous system of the nematode Caenorhabditis elegans. Philos Trans R Soc Lond B Biol Sci. 1986, 314(1165), 1–340. [Google Scholar] [CrossRef] [PubMed]
- Yang, X; Xu, T. Molecular mechanism of size control in development and human diseases. Cell Res. 2011, 21(5), 715–29. [Google Scholar] [CrossRef]
- Zanconato, F; Cordenonsi, M; Piccolo, S. YAP/TAZ at the roots of cancer. Cancer Cell. 2016, 29(6), 783–803. [Google Scholar] [CrossRef] [PubMed]
- Zafeiridis, A; Jeevanandam, V; Houser, SR; Margulies, KB. Regression of cellular hypertrophy after left ventricular assist device support. Circulation 1998, 98(7), 656–62. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




