Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Objective and Methods
2.1. Objective
- To pool adjusted HRs for cancer incidence/mortality per 1% HbA1c rise using meta-regression.
- To model cumulative HbA1c effects on risk via time-varying analyses.
- To substantiate glycohypoxia’s role by demonstrating steeper dose-response slopes in hypoxia-susceptible sites (e.g., pancreas, liver) independent of confounders like BMI and insulin use.
2.2. Methods
2.3. Study Selection and Data Extraction
2.4. Statistical Analysis
- = site-specific slope (log-HR per 1% HbA1c)
- = between-study heterogeneity
- = sampling variance
3. Results
3.1. Study Selection and Characteristics
3.2. Pooled HR per 1% HbA1c Increase
3.3. Cumulative HbA1c Burden (AUC-HbA1c)
3.4. Tissue-Specific Hypoxic Vulnerability (“Glycohypoxia”)
- Liver (score 4): HIF-2α drives lipid accumulation and oncogenesis via PI3K–AKT–mTOR; knockdown reverses phenotype [48].
- Kidney (score 2): HIF-1α exerts protective mitochondrial effects via HO-1 rather than strongly oncogenic ones [53].
- Breast (score 2): evidence supports glycolytic shift and HIF-1α modulation under the hypoxia–hyperglycemia axis [54].
4. Discussion
5. Conclusions
Author Contributions
Funding
Competing interest declaration
Declaration of AI and AI-assisted Technologies in the Writing Process
References
- Khan, M. A. B., Hashim, M. J., King, J. K., Govender, R. D., Mustafa, H., & Al Kaabi, J. (2020). Epidemiology of Type 2 Diabetes - Global Burden of Disease and Forecasted Trends. Journal of epidemiology and global health, 10(1), 107–111. [CrossRef]
- Habib, S. L., & Rojna, M. (2013). Diabetes and risk of cancer. ISRN oncology, 2013, 583786. [CrossRef]
- Giovannucci, E., Harlan, D. M., Archer, M. C., Bergenstal, R. M., Gapstur, S. M., Habel, L. A., Pollak, M., Regensteiner, J. G., & Yee, D. (2010). Diabetes and cancer: a consensus report. Diabetes care, 33(7), 1674–1685. [CrossRef]
- Lawler, T., Walts, Z. L., Steinwandel, M., Lipworth, L., Murff, H. J., Zheng, W., & Warren Andersen, S. (2023). Type 2 Diabetes and Colorectal Cancer Risk. JAMA network open, 6(11), e2343333. [CrossRef]
- Mantovani, A., & Targher, G. (2017). Type 2 diabetes mellitus and risk of hepatocellular carcinoma: spotlight on nonalcoholic fatty liver disease. Annals of translational medicine, 5(13), 270. [CrossRef]
- Pearson-Stuttard, J., Zhou, B., Kontis, V., Bentham, J., Gunter, M. J., & Ezzati, M. (2018). Worldwide burden of cancer attributable to diabetes and high body-mass index: a comparative risk assessment. The lancet. Diabetes & endocrinology, 6(2), 95–104. (Retraction published Lancet Diabetes Endocrinol. 2018 Jun;6(6):437. https://doi.org/10.1016/S2213-8587(18)30146-3.). [CrossRef]
- Zhang, A. M. Y., Wellberg, E. A., Kopp, J. L., & Johnson, J. D. (2021). Hyperinsulinemia in Obesity, Inflammation, and Cancer. Diabetes & metabolism journal, 45(3), 285–311. [CrossRef]
- Subedi, B. K., Bhimineni, C., Modi, S., Jahanshahi, A., Quiza, K., & Bitetto, D. (2025). The Role of Insulin Resistance in Cancer. Current oncology (Toronto, Ont.), 32(9), 477. [CrossRef]
- Shen, Y., Wang, C., Wang, Y., Lu, J., Chen, L., Zhang, L., Lu, W., Zhu, W., Hu, G., Xia, T., & Zhou, J. (2021). Association between time in range and cancer mortality among patients with type 2 diabetes: a prospective cohort study. Chinese medical journal, 135(3), 288–294. [CrossRef]
- Ramteke, P., Deb, A., Shepal, V., & Bhat, M. K. (2019). Hyperglycemia Associated Metabolic and Molecular Alterations in Cancer Risk, Progression, Treatment, and Mortality. Cancers, 11(9), 1402. [CrossRef]
- Akl, Maher M.1,*,†; Ahmed, Amr2. Glycohypoxia: a hypothesis linking chronic hyperglycemia to functional hypoxia and diabetic complications in type 2 diabetes. Medical Gas Research ():10.4103/mgr.MEDGASRES-D-25-00137, March 14, 2026. |. [CrossRef]
- Lu, J., Tan, M., & Cai, Q. (2015). The Warburg effect in tumor progression: mitochondrial oxidative metabolism as an anti-metastasis mechanism. Cancer letters, 356(2 Pt A), 156–164. [CrossRef]
- Zhou, D., Duan, Z., Li, Z., Ge, F., Wei, R., & Kong, L. (2022). The significance of glycolysis in tumor progression and its relationship with the tumor microenvironment. Frontiers in pharmacology, 13, 1091779. [CrossRef]
- Chan, D. A., Sutphin, P. D., Nguyen, P., Turcotte, S., Lai, E. W., Banh, A., Reynolds, G. E., Chi, J. T., Wu, J., Solow-Cordero, D. E., Bonnet, M., Flanagan, J. U., Bouley, D. M., Graves, E. E., Denny, W. A., Hay, M. P., & Giaccia, A. J. (2011). Targeting GLUT1 and the Warburg effect in renal cell carcinoma by chemical synthetic lethality. Science translational medicine, 3(94), 94ra70. [CrossRef]
- Akl, M. M., & Ahmed, A. (2024). Exploring the Interplay between the Warburg Effect and Glucolipotoxicity in Cancer Development: A Novel Perspective on Cancer Etiology. Advanced pharmaceutical bulletin, 14(3), 705–713. [CrossRef]
- Tang, Y., Gu, S., Zhu, L., Wu, Y., Zhang, W., & Zhao, C. (2022). LDHA: The Obstacle to T cell responses against tumor. Frontiers in oncology, 12, 1036477. [CrossRef]
- Beckermann, K. E., Dudzinski, S. O., & Rathmell, J. C. (2017). Dysfunctional T cell metabolism in the tumor microenvironment. Cytokine & growth factor reviews, 35, 7–14. [CrossRef]
- Bakleh, M. Z., & Al Haj Zen, A. (2025). The Distinct Role of HIF-1α and HIF-2α in Hypoxia and Angiogenesis. Cells, 14(9), 673. [CrossRef]
- Taguchi, K., & Fukami, K. (2023). RAGE signaling regulates the progression of diabetic complications. Frontiers in pharmacology, 14, 1128872. [CrossRef]
- Carrà, G., Lingua, M. F., Maffeo, B., Taulli, R., & Morotti, A. (2020). P53 vs NF-κB: the role of nuclear factor-kappa B in the regulation of p53 activity and vice versa. Cellular and molecular life sciences : CMLS, 77(22), 4449–4458. [CrossRef]
- Ferguson, R., Aughton, K., Evans, A., Shaw, V., Armstrong, J., Ware, A., Bennett, L., Costello, E., & Greenhalf, W. (2023). Mutant K-Ras in Pancreatic Cancer: An Insight on the Role of Wild-Type N-Ras and K-Ras-Dependent Cell Cycle Regulation. Current issues in molecular biology, 45(3), 2505–2520. [CrossRef]
- Meroni, M., Longo, M., Tria, G., & Dongiovanni, P. (2021). Genetics Is of the Essence to Face NAFLD. Biomedicines, 9(10), 1359. [CrossRef]
- Florescu, M. M., Crețu, O. I., Muraru, A., Florescu, D. N., & Simionescu, C. E. (2022). Snail Immunoexpression in Endometrioid Endometrial Carcinomas. Current health sciences journal, 48(4), 393–397. [CrossRef]
- Botusan, I. R., Sunkari, V. G., Savu, O., Catrina, A. I., Grünler, J., Lindberg, S., Pereira, T., Ylä-Herttuala, S., Poellinger, L., Brismar, K., & Catrina, S. B. (2008). Stabilization of HIF-1alpha is critical to improve wound healing in diabetic mice. Proceedings of the National Academy of Sciences of the United States of America, 105(49), 19426–19431. [CrossRef]
- Zhu, D., Wei, W., Zhang, J., Zhao, B., Li, Q., & Jin, P. (2024). Mechanism of damage of HIF-1 signaling in chronic diabetic foot ulcers and its related therapeutic perspectives. Heliyon, 10(3), e24656. [CrossRef]
- Pu, L. J., Shen, Y., Lu, L., Zhang, R. Y., Zhang, Q., & Shen, W. F. (2012). Increased blood glycohemoglobin A1c levels lead to overestimation of arterial oxygen saturation by pulse oximetry in patients with type 2 diabetes. Cardiovascular diabetology, 11, 110. [CrossRef]
- Zhang, J., Yao, M., Xia, S. et al. Systematic and comprehensive insights into HIF-1 stabilization under normoxic conditions: implications for cellular adaptation and therapeutic strategies in cancer. Cell Mol Biol Lett 30, 2 (2025). [CrossRef]
- Xue, C., Chen, K., Gao, Z., Bao, T., Dong, L., Zhao, L., Tong, X., & Li, X. (2023). Common mechanisms underlying diabetic vascular complications: focus on the interaction of metabolic disorders, immuno-inflammation, and endothelial dysfunction. Cell communication and signaling: CCS, 21(1), 298. [CrossRef]
- Yao, D., & Brownlee, M. (2010). Hyperglycemia-induced reactive oxygen species increase expression of the receptor for advanced glycation end products (RAGE) and RAGE ligands. Diabetes, 59(1), 249–255. [CrossRef]
- Yuen, A., & Díaz, B. (2014). The impact of hypoxia in pancreatic cancer invasion and metastasis. Hypoxia (Auckland, N.Z.), 2, 91–106. [CrossRef]
- Zheng, J., Kim, S. J., Saeidi, S., Kim, S. H., Fang, X., Lee, Y. H., Guillen-Quispe, Y. N., Ngo, H. K. C., Kim, D. H., Kim, D., & Surh, Y. J. (2023). Overactivated NRF2 induces pseudohypoxia in hepatocellular carcinoma by stabilizing HIF-1α. Free radical biology & medicine, 194, 347–356. [CrossRef]
- Taylor C. T. (2008). Interdependent roles for hypoxia inducible factor and nuclear factor-kappaB in hypoxic inflammation. The Journal of physiology, 586(17), 4055–4059. [CrossRef]
- Wang, Y., Zhu, Y. D., Gui, Q., Wang, X. D., & Zhu, Y. X. (2014). Glucagon-induced angiogenesis and tumor growth through the HIF-1-VEGF-dependent pathway in hyperglycemic nude mice. Genetics and molecular research: GMR, 13(3), 7173–7183. [CrossRef]
- Fuentes, A., Montserrat-Capdevila, J., Florensa, D., Godoy, S., Serrano, J., & Godoy, P. (2025). The Diabetes-Pancreatic Cancer Risk Relationship over Time: A Systematic Review and Meta-Analysis. Diabetology, 6(11), 131. [CrossRef]
- Zhong, GC., Ye, MX., Cheng, JH. et al. HbA1c and Risks of All-Cause and Cause-Specific Death in Subjects without Known Diabetes: A Dose-Response Meta-Analysis of Prospective Cohort Studies. Sci Rep 6, 24071 (2016). [CrossRef]
- Mao, D., Lau, E. S. H., Wu, H., Yang, A., Shi, M., Fan, B., Tam, C. H. T., Chow, E., Kong, A. P. S., Ma, R. C. W., Luk, A., & Chan, J. C. N. (2022). Risk associations of long-term HbA1c variability and obesity on cancer events and cancer-specific death in 15,286 patients with diabetes - A prospective cohort study. The Lancet Regional Health - Western Pacific, 18, Article 100315. [CrossRef]
- McVicker, L., Cardwell, C.R., Edge, L. et al. Survival outcomes in endometrial cancer patients according to diabetes: a systematic review and meta-analysis. BMC Cancer 22, 427 (2022). [CrossRef]
- Saed, L., Varse, F., Baradaran, H.R. et al. The effect of diabetes on the risk of endometrial Cancer: an updated a systematic review and meta-analysis. BMC Cancer 19, 527 (2019). [CrossRef]
- Wang S, Song S, Gao J, et al. Glycated haemoglobin variability and risk of renal function decline in type 2 diabetes mellitus: An updated systematic review and meta-analysis. Diabetes Obes Metab. 2024; 26(11): 5167-5182. [CrossRef]
- Jin Yu, Bongseong Kim, Kyungdo Han, Mee Kyoung Kim, Diabetes Progression and Its Impact on Kidney Cancer Risk: Insights From a Longitudinal Korean Cohort Study, The Journal of Clinical Endocrinology & Metabolism, 2025;, dgaf249. [CrossRef]
- Wang, S. Y., Zhang, W. S., Jiang, C. Q., Jin, Y. L., Zhu, T., Zhu, F., Xu, L. (2024). Association of Measures of Glucose Metabolism with Colorectal Cancer Risk in Older Chinese: A 13-Year Follow-up of the Guangzhou Biobank Cohort Study-Cardiovascular Disease Substudy and Meta-Analysis. Diabetes & Metabolism Journal, 48(1), 134–145.
- Zeng, H., Yuan, C., Morze, J., Fu, R., Wang, K., Wang, L., Sun, F., Ji, J. S., Giovannucci, E. L., & Song, M. (2022). New onset of type 2 diabetes after colorectal cancer diagnosis: Results from three prospective US cohort studies, systematic review, and meta-analysis. EBioMedicine, 86, 104345. [CrossRef]
- Holm, J.B., Bruun, J.M., Christiansen, P. et al. HbA1c levels and breast cancer prognosis in women without diabetes. BMC Cancer 25, 790 (2025). [CrossRef]
- Xiong, F., Wang, J., Nierenberg, J.L. et al. Diabetes mellitus and risk of breast cancer: a large-scale, prospective, population-based study. Br J Cancer 129, 648–655 (2023). [CrossRef]
- de Beer, J., Liebenberg, L. Does cancer risk increase with HbA1c, independent of diabetes?. Br J Cancer 110, 2361–2368 (2014). [CrossRef]
- Li, W., Liu, H., Qian, W., Cheng, L., Yan, B., Han, L., Xu, Q., Ma, Q., & Ma, J. (2018). Hyperglycemia aggravates microenvironment hypoxia and promotes the metastatic ability of pancreatic cancer. Computational and structural biotechnology journal, 16, 479–487. [CrossRef]
- Liu, Z., Jia, X., Duan, Y., Xiao, H., Sundqvist, K. G., Permert, J., & Wang, F. (2013). Excess glucose induces hypoxia-inducible factor-1α in pancreatic cancer cells and stimulates glucose metabolism and cell migration. Cancer Biology & Therapy, 14(5), 428–435. [CrossRef]
- Chen, J., Chen, J., Huang, J., Li, Z., Gong, Y., Zou, B., Liu, X., Ding, L., Li, P., Zhu, Z., Zhang, B., Guo, H., Cai, C., & Li, J. (2019). HIF-2α upregulation mediated by hypoxia promotes NAFLD-HCC progression by activating lipid synthesis via the PI3K-AKT-mTOR pathway. Aging, 11(23), 10839–10860. [CrossRef]
- Gao, J., Cao, H., Zhang, Q. et al. The effect of intermittent hypoxia and fecal microbiota of OSAS on genes associated with colorectal cancer. Sleep Breath 25, 1075–1087 (2021). [CrossRef]
- Martin Benej, Rebecca Hoyd, McKenzie Kreamer, Caroline E. Wheeler, Dennis J. Grencewicz, Fouad Choueiry, Carlos H.F. Chan, Yousef Zakharia, Qin Ma, Rebecca D. Dodd, Cornelia M. Ulrich, Sheetal Hardikar, Michelle L. Churchman, Ahmad A. Tarhini, Lary A. Robinson, Eric A. Singer, Alexandra P. Ikeguchi, Martin D. McCarter, Gabriel Tinoco, Marium Husain, Ning Jin, Aik C. Tan, Afaf E.G. Osman, Islam Eljilany, Gregory Riedlinger, Bryan P. Schneider, Katarina Benejova, Martin Kery, Ioanna Papandreou, Jiangjiang Zhu, Nicholas Denko, Daniel Spakowicz, for the exORIEN Consortium; The Tumor Microbiome Reacts to Hypoxia and Can Influence Response to Radiation Treatment in Colorectal Cancer. Cancer Research Communications 1 July 2024; 4 (7): 1690–1701. [CrossRef]
- Song, D. H., Jo, J. Y., Kim, C. H., Kim, M. H., Cho, I. A., Shin, J. K., Choi, W. J., & Baek, J. C. (2024). Hypoxia-Regulated Proteins: Expression in Endometrial Cancer and Their Association with Clinicopathologic Features. Diagnostics, 14(16), 1735. [CrossRef]
- Geetha, A. V. S., Harithpriya, K., Ganesan, K., & Ramkumar, K. M. (2025). Exploring the Role of Hypoxia and HIF-1α in the Intersection of Type 2 Diabetes Mellitus and Endometrial Cancer. Current Oncology, 32(2), 106. [CrossRef]
- Jiang N, Zhao H, Han Y, et al. HIF-1α ameliorates tubular injury in diabetic nephropathy via HO-1–mediated control of mitochondrial dynamics. Cell Prolif. 2020; 53:e12909. [CrossRef]
- Durrani, I.A., Bhatti, A. & John, P. The prognostic outcome of ‘type 2 diabetes mellitus and breast cancer’ association pivots on hypoxia-hyperglycemia axis. Cancer Cell Int 21, 351 (2021). [CrossRef]
| Variable | Summary Statistics |
|---|---|
| Total studies | 14 |
| Total sample size | 542,317 |
| Follow-up duration | 5–36 y (median 11.2) |
| Mean baseline HbA1c | 7.2–8.4% |
| Studies with serial HbA1c | 8 (57%) |
| Cancer events by site | Pancreatic 2,156; Liver 1,248; Colorectal 2,034; Breast 1,892; Endometrial 1,124; Kidney 1,008 |
| Adjustment covariates | Age, sex, BMI, smoking (100%); insulin/duration (71%) |
| NOS quality (mean) | 7.6 (high: 79%) |
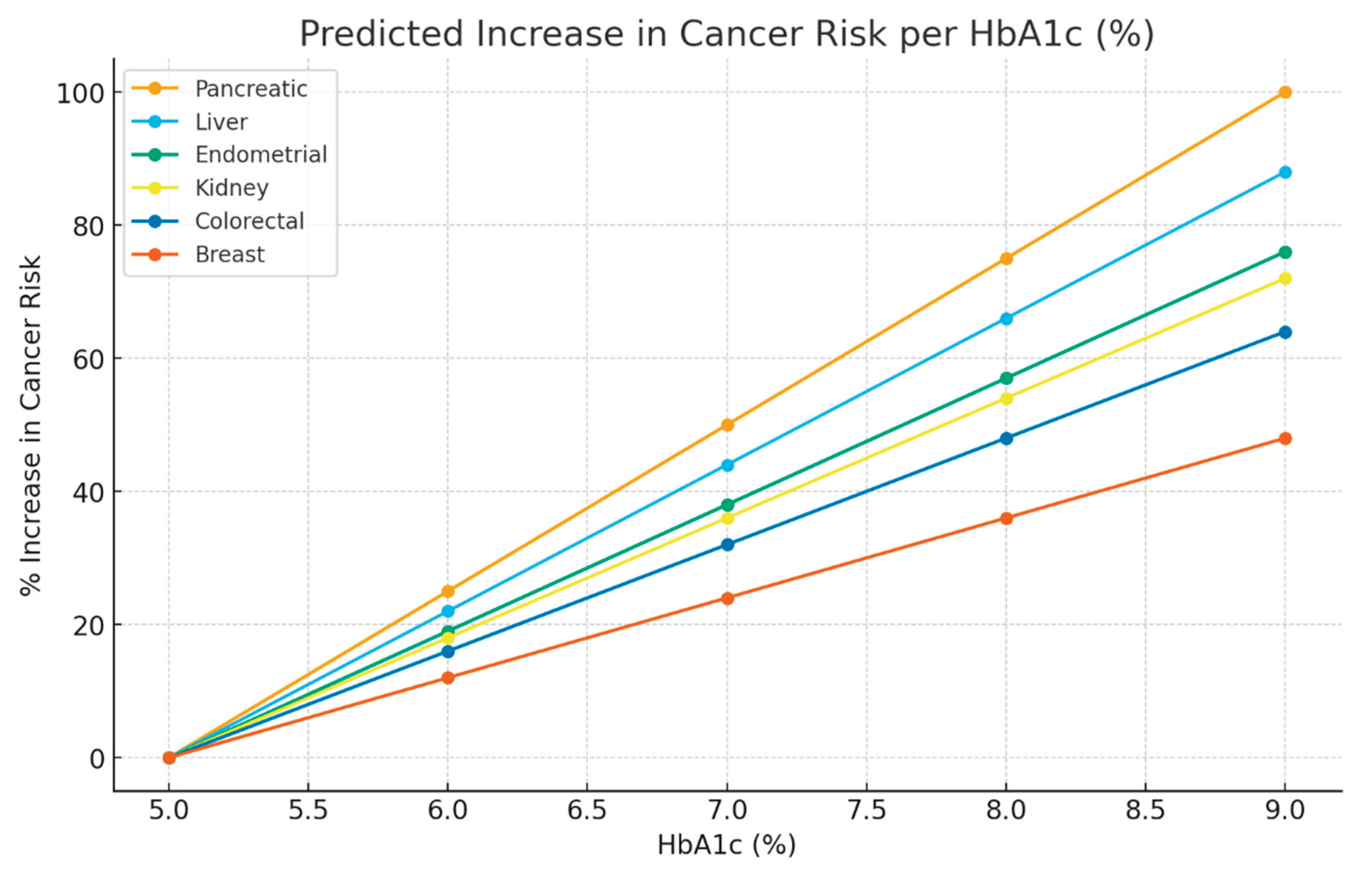
| Cancer Site | Pooled HR (95% CI) | % Risk ↑ | I² (%) | Key Studies |
|---|---|---|---|---|
| Pancreatic | 1.25 (1.18–1.33) | +25 | 62 | Fuentes 2025, Zhong 2016 [35], HK 2022 [36] |
| Liver | 1.22 (1.15–1.30) | +22 | 58 | Zhong 2016 [35], HK 2022 [36] |
| Endometrial | 1.19 (1.12–1.27) | +19 | 55 | McVicker 2022 [37], Saed 2019 [38] |
| Kidney | 1.18 (1.11–1.26) | +18 | 52 | Wang 2024 [39], Jin 2025 [40], Zhong 2016 [35] |
| Colorectal | 1.16 (1.09–1.24) | +16 | 50 | Guangzhou 2024 [41], New Onset T2D 2023 [42] |
| Breast | 1.12 (1.06–1.19) | +12 | 48 | Holm 2025 [43], Xiong 2023 [44], de Beer 2014 [45] |
| Parameter | Pooled Result (95% CI) | Strongest Sites | Independence from BMI | Duration Effect (>10y) |
|---|---|---|---|---|
| HR per +1 AUC unit (%×y) | 1.18 (1.14–1.22) | Liver 1.22, Pancreatic 1.20 | Yes (P=0.41) | β doubled (P=0.003) |
| High vs. low burden (>120/<60) | 1.9× (1.6–2.3×) | Pancreatic/liver 2.1× | Yes | HR ↑ to 2.3× |
| HbA1c slope +0.1%/3 mo | +20% (15–26%) | Colorectal/breast +18% | Yes | Amplified 1.5× |
| HbA1c variability (per SD) | +12% (8–16%) | Kidney/endometrial +15% | N/A | N/A |
| Tissue / Organ | Vulnerability Score (0–5) | Key Molecular Mechanisms | Experimental Evidence | References |
|---|---|---|---|---|
| Pancreas | 5 | HIF-1α↑, EMT↑, MMP-9↑, ROS↑, glycolysis↑, ATP↑, mitochondrial inhibition | Patients, STZ mice, BxPC-3 cells; HIF-1α siRNA reversal | Li 2018 [46]; Liu 2013 [47] |
| Liver | 4 | HIF-2α↑, lipid synthesis↑, PI3K-AKT-mTOR↑ | NAFLD-HCC; HIF-2α knockdown | Aging-US 2024 [48] |
| Colorectal | 3 | HIF-1α↑, STAT3↑, IL-6/TNF-α↑, microbiome shifts | IH models, CT26 mice, OSAS microbiome | Gao 2021 [49]; Benej 2024 [50] |
| Endometrium | 3 | GLUT-1↑, PI3K/AKT/mTOR↑, MUC1/HIF-1α↑ | Tissue microarray; immune-cell HIF-1α | Song 2024 [51]; Geetha 2025 [52] |
| Kidney | 2 | HIF-1α↑, HO-1↑, ROS↓, mitochondrial protection | HK-2 cells; HIF-1α knockout mice | Jiang 2020 [53] |
| Breast | 2 | HIF-1α↑, glycolytic shift | Cohort and mechanistic data | Durrani 2021 [54] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




