Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
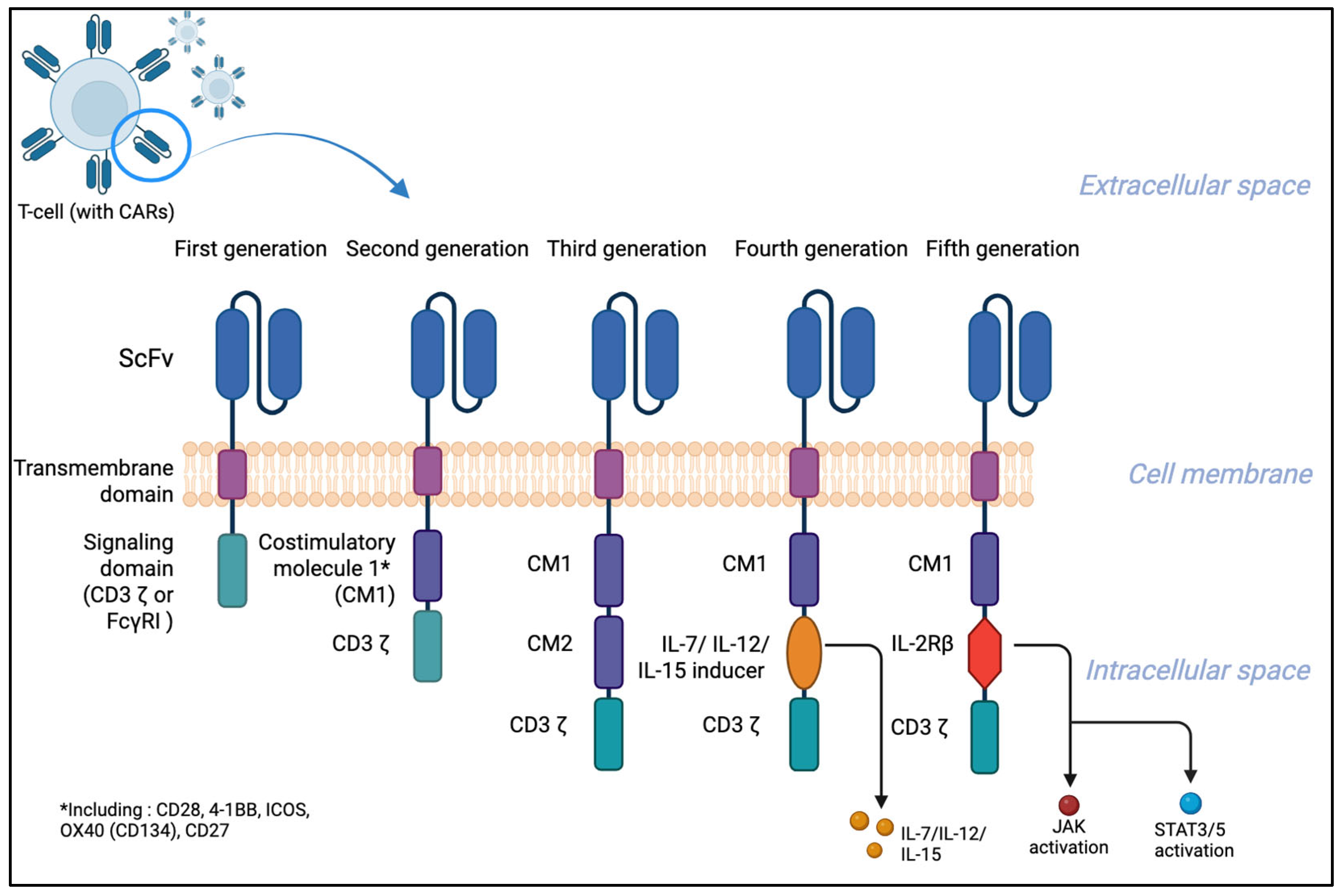
2. Systemic Lupus Erythematosus (SLE)
| Author | Year | Number of Patients | Age (years) | Sex | SLEDAI | Organ involvements | Previous treatment* | CAR T-Cell protocol | Response to CAR T-Cell therapy | Complications |
|---|---|---|---|---|---|---|---|---|---|---|
| Jinhui Shu, Wei Xie et al. [74] |
2025 | 12 (4 excluded, finally 8 patients proceeded to receive relma-cel infusions) | 18-70 years | 8 F | NR | Kidney, blood | GCS, AZA MMF, MTX, CYC, CS, TAC, LEF |
Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3); autologous CD19 CAR T ~1.1×10⁶/kg on day 0 a single dose of relma-cel infusion at four dose levels (DLs): 25 × 106 cells (n = 3), 50 × 106 cells (n = 2), 75 × 106 cells (n = 2), and 100 × 106 cells (n = 1) |
Mean SELENA-SLEDAI decreased from 11.75 at baseline to 1.63 at 6 months, PGA from 1.82 to 0.52, and BILAG total score from 6.50 to 0.88. Six patients achieved SELENA-SLEDAI ≤4 within 3 months, including three who reached 0 (meeting MFR criteria); one patient (P7) achieved MFR within 1 month. All patients achieved an SRI response. |
1 serologic relapse (month 3; withdrew month 6). All pts had AEs, mostly G1–2. Most common: cytopenia (100%), CRS (88%), hypogammaglobulinemia (88%); no ICANS or hepatic/renal toxicity. ICAHT ≤G1 (7 pts). |
| Müller et al. [64] |
2024 | 8 SLE patients | Mean 26.6 (18–38) |
7 F 1 M |
Varied by disease |
Skin, kidneys, lungs, heart, joints, Bone marrow |
GCS, HCQ, MMF, MTX, RTX, NIN, TOC, CYC |
Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3), Patient 14: 50% dose; autologous CD19 CAR T ~1.1×10⁶/kg on day 0 | Complete B-cell aplasia; durable remission SLE patients achieved SLEDAI scores of 0 |
CRS in all SLE patients (one grade 2); Patient 8 hospitalized for pneumonia 7 weeks post-CAR T, resolved with antibiotics; other infections mild (mostly URTIs); New hypogammaglobulinemia rare. |
| Wang et al. [72] |
2024 | 12 | NR | NR | NR | Kidney, joints, skin, heart, lungs | HCQ, GCS, CYC, MMF, TAC, RTX, |
3×10⁶/kg cCAR T | Plasma cells eradicated <1 mo; C3/C4 normalized ≤21 days; several pts with >1 year drug-free remission; renal improvement <6 mo | No CRS; no ICANS; normal immune recovery by ~150 days |
| Krickau et al. [71] |
2024 | 1 | 15 | 1 F | SLEDAI 23 | severe kidney disease requiring dialysis | HCQ, AZA, MMF, BEL | Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3); autologous CD19 CAR T ~1.1×10⁶/kg on day 0 | dialysis-free after 3 weeks; remission of symptoms (eGFR improved from 8 to 42 mL/min/1.73 m²) | CRS grade 1 – managed with tocilizumab |
| Friedberg et al. [73] |
2024 | 1 | 65 years old | F | NR | Blood, bone marrow | VKA, HCQ, GCS |
Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3); autologous CD19 CAR T ~1.1×10⁶/kg on day 0 |
Complete B-cell aplasia; sustained disappearance of antiphospholipid antibodies; serological remission of APS; effective lymphoma control |
CRS grade 1, which was treated with tocilizumab with dexamethasone, and ICANS, grade 4, for which patient received methylprednisolone. |
| Taubmann et al. [69] |
2023 | 7 | 19-39 | 6 F, 1 M | NR | kidney, heart, lungs, pleura, joints, skin, muscles and bone marrow |
HCQ, AZA, MMF, BEL | Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3); autologous CD19 CAR T ~1.1×10⁶/kg on day 0 |
100% DORIS remission, median B-cell aplasia 120 days | CRS (mostly grade 1); no severe ICANS |
| Huang et al. [70] | 2023 | 12 | NR | NR | 18.3 (mean) | Kidney, lungs, joints, skin, bone marrow, muscles |
GCS, MMF, MTX, HCQ, CYC, TCZ, RTX, NIN BEL, TET |
3 patients received 1x10^6/kg CD19 CAR T cells and BCMA CAR T cells, and 9 patients received 2x10^6/kg CD19 CAR T cells and BCMA CAR T cells 2x10^6/kg |
SLEDAI-2K score decreased in all patients, from a mean of 18.3 to 1.5 |
All patients had grade 1 CRS (fever) with no ICANS. Hematologic toxicity occurred in 12 patients (11 grade 4, 1 grade 3), and four infections (COVID-19 n=2, GI n=1, pulmonary n=1) were fully resolved within 6 months. Mean SLEDAI-2K dropped from 18.3 to 1.5 in all patients. |
3. Systemic Sclerosis (SSc)
| Author | Year | Number of Patients | Age (years) | Sex | mRSS at baseline | Organ involvements | Previous treatment | CAR T Cell protocol | Response to CAR T-Cell therapy | Complications |
|---|---|---|---|---|---|---|---|---|---|---|
| Pecher et al. [87] | 2025 | 5 | 42-68 | M:1F:4 | 7-32 | Skin, lungs, heart, gastrointestinal tract, kidneys |
MTX, MMF, HCQ, CSA, CYC, RTX, NIN, TCZ, HSCT | Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3); autologous CD19 CAR T ~1.1×10⁶/kg on day 0 |
mRSS reduction in all patients (5/5); fewer digital ulcers in 1/5 patients and improved FVC and DLCO in 4/5 patients; weight gain after severe GI weight loss and stabilization of cardiac disease without new events during follow-up; complete B-cell depletion by day +7 in all patients, with reappearance in 3/5 by 3 months; Scl-70 autoantibodies became negative in 2/5 patients around month 5 and later reappeared without clinical SSc activity, while RNA polymerase III levels declined in 2/5 patients but subsequently returned | CRS grade 1 in 4/5 cases, HLH in 1/5 patient resulting in death |
| Merkt et al. [90] | 2025 | 1 | 38 | F:1 | 22 | Skin, lungs, heart | CYC, MMF, NIN | Fludarabine 30 mg/m2 i.v. (on days −4, –3, −2) and cyclophosphamide 500 mg/m2 i.v. (on days −4, –3, −2) CD19.CAR- T- cells 400×10^6 (5×106/kg of body weight) i.v. on day 0 |
Over 24 months, mRSS remained stable with a 59% reduction from baseline; dyspnea improved to NYHA I–II, and lung function increased (FVC +38%, DLCO-SB +38%, DLCO +14%); CT scans showed a 72% reduction in ground-glass opacities and 55% decrease in fibrosis; cardiac and inflammatory markers normalized; CAR T cells persisted >24 months, and circulating B cells and anti-Scl-70 autoantibodies remained absent |
CRS grade 1 |
| Auth et al. [88] | 2024 | 6 | 36-53 | M:4F:2 | 17-35 | Skin, lungs, heart, kidneys | GCS, MMF, MTX, HCQ, CYC, TCZ, RTX, NIN |
Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3); MB-CART19.1 1X10^6 per kg bodyweight i.v. on day 0 |
Median mRSS reduction of 31% (~8 points) within 100 days and a fourfold decrease in digital ulcers in half of the cases; hand function improved in all patients, with increased grip strength and faster Moberg test completion; lung function remained stable with a trend toward improvement, and the ILD extent decreased by a median of 4%, mainly due to reduced ground-glass opacities; myocardial fibrosis was stable, NT-proBNP decreased in 3/6 patients, and renal function improved in one case; peripheral B cells were depleted within a week and recovered in 2–6 months, while ANA titres (anti-RNAPIII and anti-Scl-70) declined tenfold within 3 months |
CRS grade 1 in 3/6 patients, and grade 2 in 2/6 patients, Hypogammaglobulinemia in 6/6 cases, 4/6 patients required IVIG replacement therapy |
| Müller et al. [64] | 2024 | 4 | 36-60 | M:3 F:1 |
18.8-30.8 | Skin, kidneys, lungs, heart, joints | GCS, HCQ, MMF, MTX, RTX, NIN, TOC, CYC | Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 1 g/m² i.v. (day −3); patient 14 received 50% reduced dose, MB-CART19.1 1X10^6 per kg bodyweight i.v. on day 0 |
The EUSTAR activity index decreased by a median of −4.2 points, and mRSS declined by a median of −9 points after ≥6 months; disease-associated autoantibodies decreased or disappeared, and reconstituted B-cell populations showed a predominantly naïve phenotype with persistent depletion of memory and pathogenic B cells |
CRS grade 1 in 3/4 patients with SSc, hypogammaglobulinemia, no information about IVIG replacement therapy |
| Wang et al. [89] | 2024 | 2 | 45- 56 | M:2 | 26-39 | Skin, lungs, heart, gastrointestinal tract | GCS, CYC, HCQ, MMF, TAC, TZC, BLM, RAPA, MSC |
Fludarabine 25 mg/m² i.v. (days −5 to −3), cyclophosphamide 300 mg/m2/day i.v. (days –5 and –4); CAR-positive TyU19 cells 1x10^6 per kg i.v. on day 0 |
ACR-CRISS scores ≥0.996 were achieved within 1–2 months and maintained through 6 months, with marked mRSS reduction and improved skin elasticity; both patients showed improvement in lung fibrosis and reduced cardiac fibrosis; Anti-Scl-70 autoantibody levels decreased significantly, including near-complete elimination in one case |
None |
| Claus et al. [92] |
2024 | 1 | NR | NR | NR | Lungs | RTX, TOC, NIN, MMF, GCS |
NR | mRSS decreased by 31–59% over 3–24 months, hand function and Raynaud’s symptoms improved; lung function remained stable or increased (FVC +38%, DLCO-SB +38%, DLCO +14–20%); CT showed reduced ground-glass opacities (−72%) and fibrosis (−55%), while heart function stabilized (EF 55–60%, PASP −26%, RA area −45.2%) and NT-proBNP/troponin normalized; pathogenic B cells were depleted within 7 days, autoantibodies decreased or disappeared, and CAR-T cells persisted >24 months, leading to durable clinical and immunological remission |
None |
| Bergmann et al. [91] | 2023 | 1 | 60 | M:1 | 20 | Skin, lungs, heart | MTX, MMF |
Fludarabine 12.5 mg/m2 (on days −5 to −3) and cyclophosphamide 500 mg/m2 (on day −3), 1× 10⁶ CAR T cells/kg on day 0 |
By 3 months, skin fibrosis improved and remained stable, with patients reporting milder and less frequent Raynaud’s attacks; lung function was preserved, with DLCO increasing 20.4% at 6 months, and cardiac function showed stable EF, improved PASP (−26%), and reduced RA area (−45.2%); immune cells reconstituted rapidly, ANA reactivity was abolished, and RP11 autoantibodies became undetectable at 3–6 months follow-up. |
CRS grade 1 |
4. Melanoma
| Author | Year | Clinical trial ID | Number of patients | Sex | Targeted antigen | CAR T cell protocol | Response to CAR T cell therapy | Complications |
|---|---|---|---|---|---|---|---|---|
| Gargett et al. [107] | 2024 | ACTRN12613000198729 | 9 | NR | GD2 | 1 × 10⁶ GD2-CAR T cells/kg on day 0, combined with BRAF and MEK inhibitors (dabrafenib and trametinib) starting seven days prior and continuing for 28 days | CAR T cells were detected in tumor biopsies, but tumor response was limited; most patients had disease progression or transient stabilization | Mild AEs included rash, fever, diarrhea, and anorexia; no neurotoxicity observed |
| Aleksandrova et al. [108] | 2024 | NCT03893019 | 3 | F:2 M:1 |
CD20 | Patients were pre-treated with 60 mg/kg body weight cyclophosphamide (day −7 and day −6) and 25 mg/m2 body surface area fludarabine (day −5 to day −1); MB-CART20.1 on day 0 | All CAR T products demonstrated the ability to activate T cells upon contact with target cells marked by an increase in the secretion of pro-inflammatory cytokines and an increase in CAR T cell proliferation; there were differences in the levels of secreted cytokines and the degree of CAR T cell amplification depending on the patient, T-cell activation by CAR depended on the level of target antigen expression |
NR |
| Shah et al. [109] | 2023 | NCT03060356 | 3 | F:2 M:1 |
cMET | Patients received up to six infusions (1 × 10e8 T cells/dose) of CAR T cells without lymphodepleting chemotherapy. | Disease stability was achieved in 4 cases, disease progression was observed in 3 subjects, mRNA signals corresponding to CART cells were detected by RT-PCR in all patients’ blood, five subjects underwent post infusion biopsy with no CART-cell signals seen in tumor, three subjects had paired tumor tissue; IHC showed increases in CD8 and CD3 and decreases in pS6 and Ki67 |
All patients experienced some grade 1 or 2 toxicity including: anemia, fatigue, malaise, one patient experienced grade 1 CRS |
| Rosenberg et al. [110] | 2010 | NCT01218867 | 24 | NR | VEGFR2 | Non-myeloablative conditioning chemotherapy (e.g., cyclophosphamide + fludarabine) followed by infusion of CAR T cells plus IL-2 (aldesleukin) support |
In the trial, no objective clinical responses—neither complete nor partial remission—were observed, and 23 of 24 patients experienced disease progression, with only one patient achieving stable disease; due to the lack of sustained objective responses, the study was terminated. | About 21% of patients (5/24) experienced serious adverse events, including elevated liver enzymes, bilirubin, and hypoxia; CRS occurred rarely and was generally mild, presenting as fever, mild hypotension, or transient fatigue, with no grade 3–4 CRS events reported; no cases of ICANS were observed. |
5. Cutaneous Lymphomas
5.1. Cutaneous T Cell Lymphoma (CTCL)
| Author | Year | Targeted antigen | Study type | CAR T cell protocol | Response to CAR T cell therapy |
Complications |
|---|---|---|---|---|---|---|
| To et al. [117] | 2025 | TAG-72 & CD30 | Preclinical (in vitro; cell lines) | NA | Three CAR-T lines showed potent, specific cytotoxicity against CTCL cells, reduced tumor burden, improved mouse survival, and caused no serious side effects. |
NA |
| Evtimov et al. [116] | 2024 | TAG-72 | Preclinical (in vitro and in vivo, mice) | Mice were randomized into experimental groups (3–6 mice/group); two injections of 5 × 106 CAR T cells i.v. at 5-day intervals; control mice received T cells (no CAR) at comparable dosages and intervals. | CTCL patients’ circulating CD3+ and CD4+ T cells showed higher TAG-72 expression than healthy donors, and anti-TAG-72 CAR-T cells specifically and effectively eliminated these TAG-72+ cells in vitro. | NA |
| Watanabe et al. [119] | 2024 | CCR4 (mogamulizumab-based CAR T) | Preclinical (in vitro and in vivo, mice) | 0.5 or 2 × 106 CAR-positive T cells or untransduced (UTD) T cells | CCR4-CAR-T cells exhibited strong cytotoxicity against CTCL, proliferated robustly, eliminated CCR4+ T cells, suppressed Th2/Th17/Treg functions while sparing CD8+ and Th1 cells, and mogamulizumab-based CCR4-CAR-T showed superior antitumor efficacy and long-term remission in mice. | NA |
| Iyer et al. [121] | 2024 | CD70 (CTX130) | Single-arm phase I clinical trial | Fludarabine 30 mg/m2 and cyclophosphamide 500 mg/m2 (i.v. daily for 3 days), followed by intravenous CTX130 infusion at dose from 3 × 107 CAR+ T cells (dose level 1) to 9 × 108 CAR+ T cells (dose level 4). | 41 patients; 39 (95%) received CTX130; Objective response rate (ORR): 18 of 39 patients (46.2%); Complete response (CR): 6 patients (19.4%) Partial response (PR): 10 patients (32.3%) |
The most common AE was CRS, occurring in 67% of patients (mostly grade 1–2; one grade 4 at the highest dose). Neurotoxicity was mild (grade 1–2) in 10%. Grade 3–4 events included neutropenia (36%), anemia (28%), and thrombocytopenia (15%). Overall, 64% experienced serious adverse events, 36% were CTX130-related, mainly CRS. There were 21 deaths, 16 from disease progression and 5 unrelated to CTX130 |
| Reef et al. [120] | 2024 | CCR4.CD30. CART | Phase I clinical trial | Patients received lymphodepletion with fludarabine ± bendamustine or cyclophosphamide before CAR-T infusion. CCR4.CD30 CAR-T doses escalated from 2×10⁷ to 1×10⁸ cells/m², with alternating dose levels also receiving 1×10⁸ CD30 CAR-T cells/m² in a 3+3 design | CCR4.CD30 CAR-T cells expanded in blood and infiltrated tumors, with a median skin tumor reduction of 42.2%; 50% of patients achieved stable disease, none progressed, but all required further therapy, and median overall survival was 23.9 months. | No CRS or ICANS occurred. Grade 3–4 adverse events were hematologic (neutropenia, lymphopenia, anemia, thrombocytopenia), with two severe events: grade 3 diverticulitis in one patient and grade 3 neutropenia with infection in another. |
5.2. Cutaneous B Cell Lymphoma (CBCL)
6. Safety Concerns of CAR T Cell Therapy
6.1. Cytokine Release Syndrome (CRS)
6.2. Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS)
6.3. Immune Effector Cell–Associated Hematotoxicity (ICAHT)
6.4. On-Target Off-Tumor Toxicity (OTOT)
6.5. B-Cell Depletion
6.6. Malignances Secondary to Theraphy
6.7. Fertility
7. Future Perspectives
7.1. Future Perspectives in Autoimmune Diseases
7.1.1. Future Perspectives in Systemic Lupus Erythematosus (SLE)
7.1.2. Future Perspectives in Systemic Sclerosis (SSc)
7.1.3. Future Perspectives in Psoriasis
7.1.4. Future Perspectives in Dermatomyositis
7.1.5. Future Perspectives in Sjögren’s Syndrome (SS)
7.1.6. Future Perspectives in Pemphigus Vulgaris (PV)
7.2. Future Perspectives in Oncology
7.2.1. Melanoma
7.2.2. Cutaneous T-Cell Lymphoma (CTCL)
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sterner, R. C.; Sterner, R. M. CAR T Cell Therapy: Current Limitations and Potential Strategies. Blood Cancer J. 2021, 11(4), 69. [Google Scholar] [CrossRef]
- Syed, F.; El Fakih, R.; Alahmari, A. D.; Ali, A. S. O.; Aljurf, M. Chimeric Antigen Receptor Structure and Manufacturing of Clinical Grade CAR Engineered Cells Using Different Bioreactors. Hematology/Oncology and Stem Cell Therapy 2022, 15(3), 137–152. [Google Scholar] [CrossRef]
- Guedan, S.; Calderon, H.; Posey, A. D.; Maus, M. V. Engineering and Design of Chimeric Antigen Receptors. Molecular Therapy - Methods & Clinical Development 2019, 12, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Chen, R.; Huang, Y.; Meng, X.; Chen, J.; Liao, C.; Tang, Y.; Zhou, C.; Gao, X.; Sun, J. Tuning the Ignition of CAR: Optimizing the Affinity of scFv to Improve CAR T Therapy. Cell Mol Life Sci 2021, 79(1), 14. [Google Scholar] [CrossRef] [PubMed]
- Barden, M.; Elsenbroich, P. R.; Haas, V.; Ertelt, M.; Pervan, P.; Velas, L.; Gergely, B.; Szöőr, Á; Harrer, D. C.; Bezler, V.; Holzinger, A.; Friis, R. U. W.; Vereb, G.; Schütz, G. J.; Schoeder, C. T.; Hombach, A. A.; Abken, H. Integrating Binding Affinity and Tonic Signaling Enables a Rational CAR Design for Augmented T Cell Function. J Immunother Cancer 2024, 12(12), e010208. [Google Scholar] [CrossRef] [PubMed]
- Roselli, E.; Faramand, R.; Davila, M. L. Insight into Next-Generation CAR Therapeutics: Designing CAR T Cells to Improve Clinical Outcomes. Journal of Clinical Investigation 2021, 131(2), e142030. [Google Scholar] [CrossRef]
- Taheri, F. H.; Hassani, M.; Sharifzadeh, Z.; Behdani, M.; Abdoli, S.; Sayadi, M.; Bagherzadeh, K.; Arashkia, A.; Abolhassani, M. Tuning Spacer Length Improves the Functionality of the Nanobody-Based VEGFR2 CAR T Cell. BMC Biotechnol 2024, 24(1), 1. [Google Scholar] [CrossRef]
- Xiong, Y.; Libby, K. A.; Su, X. The Physical Landscape of CAR T Synapse. Biophys J 2024, 123(15), 2199–2210. [Google Scholar] [CrossRef]
- Kong, Y.; Li, J.; Zhao, X.; Wu, Y.; Chen, L. CAR T Cell Therapy: Developments, Challenges and Expanded Applications from Cancer to Autoimmunity. Front Immunol 2024, 15, 1519671. [Google Scholar] [CrossRef]
- Elazar, A.; Chandler, N. J.; Davey, A. S.; Weinstein, J. Y.; Nguyen, J. V.; Trenker, R.; Cross, R. S.; Jenkins, M. R.; Call, M. J.; Call, M. E.; Fleishman, S. J. De Novo-Designed Transmembrane Domains Tune Engineered Receptor Functions. Elife 2022, 11, e75660. [Google Scholar] [CrossRef]
- Patel, K. K.; Tariveranmoshabad, M.; Kadu, S.; Shobaki, N.; June, C. From Concept to Cure: The Evolution of CAR T Cell Therapy. Mol Ther 2025, 33(5), 2123–2140. [Google Scholar] [CrossRef]
- Khan, S. H.; Choi, Y.; Veena, M.; Lee, J. K.; Shin, D. S. Advances in CAR T Cell Therapy: Antigen Selection, Modifications, and Current Trials for Solid Tumors. Front Immunol 2024, 15, 1489827. [Google Scholar] [CrossRef] [PubMed]
- Sadelain, M.; Brentjens, R.; Rivière, I. The Promise and Potential Pitfalls of Chimeric Antigen Receptors. Current Opinion in Immunology 2009, 21(2), 215–223. [Google Scholar] [CrossRef] [PubMed]
- Brocker, T. Chimeric Fv-ζ or Fv-ε Receptors Are Not Sufficient to Induce Activation or Cytokine Production in Peripheral T Cells. Blood 2000, 96(5), 1999–2001. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Liu, J.; Zhong, J. F.; Zhang, X. Engineering CAR T Cells. Biomark Res 2017, 5(1), 22. [Google Scholar] [CrossRef]
- Van Der Stegen, S. J. C.; Hamieh, M.; Sadelain, M. The Pharmacology of Second-Generation Chimeric Antigen Receptors. Nat Rev Drug Discov 2015, 14(7), 499–509. [Google Scholar] [CrossRef]
- Tomasik, J.; Jasiński, M.; Basak, G. W. Next Generations of CAR T Cells - New Therapeutic Opportunities in Hematology? Front. Immunol. 2022, 13, 1034707. [Google Scholar] [CrossRef]
- Chmielewski, M.; Abken, H. TRUCKs: The Fourth Generation of CARs. Expert Opinion on Biological Therapy 2015, 15(8), 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Li, A.; Liu, Q.; Li, T.; Yuan, X.; Han, X.; Wu, K. Chimeric Antigen Receptor T Cells: A Novel Therapy for Solid Tumors. J Hematol Oncol 2017, 10(1), 78. [Google Scholar] [CrossRef]
- Malaer, J. D.; Marrufo, A. M.; Mathew, P. A. 2B4 (CD244, SLAMF4) and CS1 (CD319, SLAMF7) in Systemic Lupus Erythematosus and Cancer. Clinical Immunology 2019, 204, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Abreu, T. R.; Fonseca, N. A.; Gonçalves, N.; Moreira, J. N. Current Challenges and Emerging Opportunities of CAR T Cell Therapies. Journal of Controlled Release 2020, 319, 246–261. [Google Scholar] [CrossRef] [PubMed]
- Vormittag, P.; Gunn, R.; Ghorashian, S.; Veraitch, F. S. A Guide to Manufacturing CAR T Cell Therapies. Current Opinion in Biotechnology 2018, 53, 164–181. [Google Scholar] [CrossRef]
- Asmamaw Dejenie, T.; Tiruneh G/Medhin, M.; Dessie Terefe, G.; Tadele Admasu, F.; Wale Tesega, W.; Chekol Abebe, E. Current Updates on Generations, Approvals, and Clinical Trials of CAR T-Cell Therapy. Human Vaccines & Immunotherapeutics 2022, 18(6), 2114254. [Google Scholar] [CrossRef]
- Lopatnikova, J. A.; Sennikov, S. V. Bispecific Immunotherapy Based on Antibodies, T-Cell Receptors, and Aptamers: Mechanisms of Action, Adverse Effects, and Future Perspectives. Front. Immunol. 2025, 16, 1679092. [Google Scholar] [CrossRef] [PubMed]
- Lee, D. W.; Gardner, R.; Porter, D. L.; Louis, C. U.; Ahmed, N.; Jensen, M.; Grupp, S. A.; Mackall, C. L. Current Concepts in the Diagnosis and Management of Cytokine Release Syndrome. Blood 2014, 124(2), 188–195. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Rivière, I. Clinical Manufacturing of CAR T Cells: Foundation of a Promising Therapy. Molecular Therapy - Oncolytics 2016, 3, 16015. [Google Scholar] [CrossRef]
- Ayala Ceja, M.; Khericha, M.; Harris, C. M.; Puig-Saus, C.; Chen, Y. Y. CAR T Cell Manufacturing: Major Process Parameters and next-Generation Strategies. J Exp Med 2024, 221(2), e20230903. [Google Scholar] [CrossRef] [PubMed]
- Levine, B. L.; Miskin, J.; Wonnacott, K.; Keir, C. Global Manufacturing of CAR T Cell Therapy. Molecular Therapy - Methods & Clinical Development 2017, 4, 92–101. [Google Scholar] [CrossRef]
- Short, L.; Holt, R. A.; Cullis, P. R.; Evgin, L. Direct in Vivo CAR T Cell Engineering. Trends in Pharmacological Sciences 2024, 45(5), 406–418. [Google Scholar] [CrossRef]
- Akhavan, D.; Alizadeh, D.; Wang, D.; Weist, M. R.; Shepphird, J. K.; Brown, C. E. CAR T Cells for Brain Tumors: Lessons Learned and Road Ahead. Immunol Rev 2019, 290(1), 60–84. [Google Scholar] [CrossRef]
- Brudno, J. N.; Maus, M. V.; Hinrichs, C. S. CAR T Cells and T-Cell Therapies for Cancer: A Translational Science Review. JAMA 2024, 332(22), 1924. [Google Scholar] [CrossRef]
- Abdalhadi, H. M.; Chatham, W. W.; Alduraibi, F. K. CAR T-Cell Therapy for Systemic Lupus Erythematosus: A Comprehensive Overview. IJMS 2024, 25(19), 10511. [Google Scholar] [CrossRef]
- Del Galdo, F.; Lescoat, A.; Conaghan, P. G.; Bertoldo, E.; Čolić, J.; Santiago, T.; Suliman, Y. A.; Matucci-Cerinic, M.; Gabrielli, A.; Distler, O.; Hoffmann-Vold, A.-M.; Castellví, I.; Balbir-Gurman, A.; Vonk, M.; Ananyeva, L.; Rednic, S.; Tarasova, A.; Ostojic, P.; Boyadzhieva, V.; El Aoufy, K.; Farrington, S.; Galetti, I.; Denton, C. P.; Kowal-Bielecka, O.; Mueller-Ladner, U.; Allanore, Y. EULAR Recommendations for the Treatment of Systemic Sclerosis: 2023 Update. Annals of the Rheumatic Diseases 2025, 84(1), 29–40. [Google Scholar] [CrossRef]
- Soltantoyeh, T.; Akbari, B.; Karimi, A.; Mahmoodi Chalbatani, G.; Ghahri-Saremi, N.; Hadjati, J.; Hamblin, M. R.; Mirzaei, H. R. Chimeric Antigen Receptor (CAR) T Cell Therapy for Metastatic Melanoma: Challenges and Road Ahead. Cells 2021, 10(6), 1450. [Google Scholar] [CrossRef] [PubMed]
- Araghi, F.; Dadkhahfar, S.; Robati, R. M.; Tabary, M.; Shahidi-Dadras, M. The Emerging Role of T Cells in Pemphigus Vulgaris: A Systematic Review. Clin Exp Med 2022, 23(4), 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- To, V.; Evtimov, V. J.; Jenkin, G.; Pupovac, A.; Trounson, A. O.; Boyd, R. L. CAR T Cell Development for Cutaneous T Cell Lymphoma: Current Limitations and Potential Treatment Strategies. Front. Immunol. 2022, 13, 968395. [Google Scholar] [CrossRef] [PubMed]
- Fava, Andrea, i Michelle Petri. „Systemic Lupus Erythematosus: Diagnosis and Clinical Management”. Journal of Autoimmunity, t. 96, stycznia 2019, s. 1–13. DOI.org. [CrossRef]
- Bernatsky, S., et al. „Mortality in Systemic Lupus Erythematosus”. Arthritis & Rheumatism, t. 54, nr 8, sierpnia 2006, s. 2550–57. DOI.org. [CrossRef]
- SRahman, Anisur, i David A. Isenberg. „Systemic Lupus Erythematosus”. New England Journal of Medicine, t. 358, nr 9, lutego 2008, s. 929–39. DOI.org. [CrossRef]
- Mak, Anselm, i Nien Yee Kow. „The Pathology of T Cells in Systemic Lupus Erythematosus”. Journal of Immunology Research, t. 2014, 2014, s. 1–8. DOI.org. [CrossRef]
- Al-Mayouf; Sulaiman, M.; et al. Loss-of-Function Variant in DNASE1L3 Causes a Familial Form of Systemic Lupus Erythematosus”. Nature Genetics 2011, t. 43(nr 12), s. 1186–88. [Google Scholar] [CrossRef]
- Ameer, Muhammad Atif; et al. „An Overview of Systemic Lupus Erythematosus (SLE) Pathogenesis, Classification, and Management”. Cureus 2022. [Google Scholar] [CrossRef]
- Karrar, Sarah, i Deborah S. Cunninghame Graham. „Review: Abnormal B Cell Development in Systemic Lupus Erythematosus: What the Genetics Tell Us”. Arthritis & Rheumatology 2018, t. 70(nr 4), s. 496–507. [CrossRef]
- Nemazee, David. Mechanisms of Central Tolerance for B Cells”. Nature Reviews Immunology 2017, t. 17(nr 5), s. 281–94. [Google Scholar] [CrossRef]
- Kaminski, Denise A.; et al. Advances in Human B Cell Phenotypic Profiling”. Frontiers in Immunology 2012, t. 3 DOI.org (Crossref). [Google Scholar] [CrossRef] [PubMed]
- Nashi, Emil; et al. The Role of B Cells in Lupus Pathogenesis”. The International Journal of Biochemistry & Cell Biology 2010, t. 42(nr 4), s. 543–50. [Google Scholar] [CrossRef]
- Xu, Xiao, i Su‐Hua Su. „CAR T Cell Therapies in Autoimmune Rheumatic Diseases: A Brief Report on the Clinical Trial Landscape, Current Status, and Future Perspectives”. Frontiers in Immunology, t. 16, października 2025. Frontiers. [CrossRef]
- Tur, Carlo; et al. „CD19-CAR T-Cell Therapy Induces Deep Tissue Depletion of B Cells”. Annals of the Rheumatic Diseases 2025, t. 84(nr 1), s. 106–14. [Google Scholar] [CrossRef]
- Su, Ding-Lei; et al. Roles of Pro- and Anti-Inflammatory Cytokines in the Pathogenesis of SLE”. Journal of Biomedicine and Biotechnology t. 2012(2012), s. 1–15. [CrossRef] [PubMed]
- Fanouriakis, Antonis; et al. 2019 Update of the EULAR Recommendations for the Management of Systemic Lupus Erythematosus”. Annals of the Rheumatic Diseases 2019, t. 78(nr 6), s. 736–45. [Google Scholar] [CrossRef]
- Doria, Andrea; et al. Optimizing Outcome in SLE: Treating-to-Target and Definition of Treatment Goals”. Autoimmunity Reviews 2014, t. 13(nr 7), s. 770–77. [Google Scholar] [CrossRef]
- Ponticelli, C., i G. Moroni. „Hydroxychloroquine in Systemic Lupus Erythematosus (SLE)”. Expert Opinion on Drug Safety 2017, t. 16(nr 3), s. 411–19. [CrossRef] [PubMed]
- Ruiz-Irastorza, G.; et al. Glucocorticoid Use and Abuse in SLE”. Rheumatology 2012, t. 51(nr 7), s. 1145–53. [Google Scholar] [CrossRef]
- Porta, Sabrina; et al. Glucocorticoids in Systemic Lupus Erythematosus. Ten Questions and Some Issues”. Journal of Clinical Medicine 2020, t. 9(nr 9), s. 2709. [Google Scholar] [CrossRef]
- Luijten, Remco K.M.A.C.; et al. „The Use of Glucocorticoids in Systemic Lupus Erythematosus. After 60years Still More an Art than Science”. Autoimmunity Reviews 2013, t. 12(nr 5), s. 617–28. [Google Scholar] [CrossRef] [PubMed]
- Pego-Reigosa, José M.; et al. Efficacy and Safety of Nonbiologic Immunosuppressants in the Treatment of Nonrenal Systemic Lupus Erythematosus: A Systematic Review”. Arthritis Care & Research 2013, t. 65(nr 11), s. 1775–85. [Google Scholar] [CrossRef]
- Xiong, Wen, i Robert G. Lahita. „Pragmatic Approaches to Therapy for Systemic Lupus Erythematosus”. Nature Reviews Rheumatology 2014, t. 10(nr 2), s. 97–107. [CrossRef]
- Bruce, Ian N. Re-Evaluation of Biologic Therapies in Systemic Lupus Erythematosus”. Current Opinion in Rheumatology 2010, t. 22(nr 3), s. 273–77. [Google Scholar] [CrossRef]
- Merrill, Joan T.; et al. Efficacy and Safety of Rituximab in Moderately-to-severely Active Systemic Lupus Erythematosus: The Randomized, Double-blind, Phase Ii/Iii Systemic Lupus Erythematosus Evaluation of Rituximab Trial”. Arthritis & Rheumatism 2010, t. 62(nr 1), s. 222–33. [Google Scholar] [CrossRef]
- Rovin, Brad H.; et al. Efficacy and Safety of Rituximab in Patients with Active Proliferative Lupus Nephritis: The Lupus Nephritis Assessment with Rituximab Study”. Arthritis & Rheumatism 2012, t. 64(nr 4), s. 1215–26. [Google Scholar] [CrossRef]
- Mysler, Eduardo F.; et al. Efficacy and Safety of Ocrelizumab in Active Proliferative Lupus Nephritis: Results From a Randomized, Double-Blind, Phase III Study”. Arthritis & Rheumatism 2013, t. 65(nr 9), s. 2368–79. [Google Scholar] [CrossRef]
- Arnold, J.; et al. AB0434 EFFICACY AND SAFETY OF OBINUTUZUMAB IN SYSTEMIC LUPUS ERYTHEMATOSUS PATIENTS WITH SECONDARY NON-RESPONSE TO RITUXIMAB. Annals of the Rheumatic Diseases 2022, t. 81 DOI.org (Crossref), s. 1345. [Google Scholar] [CrossRef]
- Furie, Richard A; et al. B-Cell Depletion with Obinutuzumab for the Treatment of Proliferative Lupus Nephritis: A Randomised, Double-Blind, Placebo-Controlled Trial”. Annals of the Rheumatic Diseases 2022, t. 81(nr 1), s. 100–07. [Google Scholar] [CrossRef] [PubMed]
- Müller, Fabian; et al. „CD19 CAR T-Cell Therapy in Autoimmune Disease — A Case Series with Follow-Up”. New England Journal of Medicine 2024, t. 390(nr 8), s. 687–700. [Google Scholar] [CrossRef]
- Duan, Meixue; et al. Understanding Heterogeneity of Human Bone Marrow Plasma Cell Maturation and Survival Pathways by Single-Cell Analyses”. Cell Reports 2023, t. 42(nr 7), s. 112682. [Google Scholar] [CrossRef]
- Mougiakakos, Dimitrios; et al. „CD19-Targeted CAR T Cells in Refractory Systemic Lupus Erythematosus”. New England Journal of Medicine 2021, t. 385(nr 6), s. 567–69. [Google Scholar] [CrossRef]
- Zhang, Wenli; et al. „Treatment of Systemic Lupus Erythematosus Using BCMA-CD19 Compound CAR”. Stem Cell Reviews and Reports 2021, t. 17(nr 6), s. 2120–23. [Google Scholar] [CrossRef]
- Mackensen, Andreas; et al. Author Correction: Anti-CD19 CAR T Cell Therapy for Refractory Systemic Lupus Erythematosus”. Nature Medicine 2023, t. 29(nr 11), s. 2956–2956. [Google Scholar] [CrossRef] [PubMed]
- Taubmann, J.; et al. OP0141 LONG TERM SAFETY AND EFFICACY OF CAR T CELL TREATMENT IN REFRACTORY SYSTEMIC LUPUS ERYTHEMATOSUS - DATA FROM THE FIRST SEVEN PATIENTS”. Annals of the Rheumatic Diseases 2023, t. 82, s. 93–94. [Google Scholar] [CrossRef]
- Feng, Jingjing; et al. „CD19/BCMA CAR T Cell Therapy for Refractory Systemic Lupus Erythematosus - Safety and Preliminary Efficacy Data from a Phase I Clinical Study”. Blood 2023, t. 142, nr Supplement 1, s. 4835–4835. [Google Scholar] [CrossRef]
- Krickau, Tobias; et al. „CAR T-Cell Therapy Rescues Adolescent with Rapidly Progressive Lupus Nephritis from Haemodialysis”. The Lancet 2024, t. 403(nr 10437), s. 1627–30. [Google Scholar] [CrossRef]
- Wang, Weijia; et al. BCMA-CD19 Compound CAR T Cells for Systemic Lupus Erythematosus: A Phase 1 Open-Label Clinical Trial”. Annals of the Rheumatic Diseases 2024, t. 83(nr 10), s. 1304–14. [Google Scholar] [CrossRef]
- Friedberg, Eleonora; et al. „Disappearance of Antiphospholipid Antibodies after Anti-CD19 Chimeric Antigen Receptor T-Cell Therapy of B-Cell Lymphoma in a Patient with Systemic Lupus Erythematosus and Antiphospholipid Syndrome”. Journal of Thrombosis and Haemostasis 2025, t. 23(nr 1), s. 262–66. [Google Scholar] [CrossRef]
- Shu, Jinhui; et al. „Safety and Clinical Efficacy of Relmacabtagene Autoleucel (Relma-Cel) for Systemic Lupus Erythematosus: A Phase 1 Open-Label Clinical Trial”. eClinicalMedicine 2025, t. 83, s. 103229. [Google Scholar] [CrossRef]
- Sieiro Santos, C.; Del Galdo, F. New Horizons in Systemic Sclerosis Treatment: Advances and Emerging Therapies in 2025. RMD Open 2025, 11(3), e005776. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L. I.; Katsiari, C.; Syrmou, V.; Chikanza, I. C. CAR T Cells in Systemic Sclerosis. Clin Rheumatol 2025. [Google Scholar] [CrossRef]
- Misra, D. P.; Ahmed, S.; Agarwal, V. Is Biological Therapy in Systemic Sclerosis the Answer? Rheumatol Int 2020, 40(5), 679–694. [Google Scholar] [CrossRef]
- Beesley, C. F.; Goldman, N. R.; Taher, T. E.; Denton, C. P.; Abraham, D. J.; Mageed, R. A.; Ong, V. H. Dysregulated B Cell Function and Disease Pathogenesis in Systemic Sclerosis. Front. Immunol. 2023, 13, 999008. [Google Scholar] [CrossRef]
- Thoreau, B.; Chaigne, B.; Mouthon, L. Role of B-Cell in the Pathogenesis of Systemic Sclerosis. Front. Immunol. 2022, 13, 933468. [Google Scholar] [CrossRef] [PubMed]
- Cavazzana, I.; Vojinovic, T.; Airo’, P.; Fredi, M.; Ceribelli, A.; Pedretti, E.; Lazzaroni, M. G.; Garrafa, E.; Franceschini, F. Systemic Sclerosis-Specific Antibodies: Novel and Classical Biomarkers. Clinic Rev Allerg Immunol 2022, 64(3), 412–430. [Google Scholar] [CrossRef]
- Silva, I. S.; Ferreira, B. H.; Almeida, C. R. Molecular Mechanisms Behind the Role of Plasmacytoid Dendritic Cells in Systemic Sclerosis. Biology 2023, 12(2), 285. [Google Scholar] [CrossRef] [PubMed]
- Numajiri, H.; Kuzumi, A.; Fukasawa, T.; Ebata, S.; Yoshizaki-Ogawa, A.; Asano, Y.; Kazoe, Y.; Mawatari, K.; Kitamori, T.; Yoshizaki, A.; Sato, S. B Cell Depletion Inhibits Fibrosis via Suppression of Profibrotic Macrophage Differentiation in a Mouse Model of Systemic Sclerosis. Arthritis & Rheumatology 2021, 73(11), 2086–2095. [Google Scholar] [CrossRef] [PubMed]
- Horii, M.; Fushida, N.; Ikeda, T.; Oishi, K.; Hamaguchi, Y.; Ikawa, Y.; Komuro, A.; Matsushita, T. Cytokine-producing B-cell Balance Associates with Skin Fibrosis in Patients with Systemic Sclerosis. The Journal of Dermatology 2022, 49(10), 1012–1019. [Google Scholar] [CrossRef]
- Moradzadeh, M.; Aghaei, M.; Mehrbakhsh, Z.; Arab-Bafrani, Z.; Abdollahi, N. Efficacy and Safety of Rituximab Therapy in Patients with Systemic Sclerosis Disease (SSc): Systematic Review and Meta-Analysis. Clin Rheumatol 2021, 40(10), 3897–3918. [Google Scholar] [CrossRef]
- Shouval, R.; Furie, N.; Raanani, P.; Nagler, A.; Gafter-Gvili, A. Autologous Hematopoietic Stem Cell Transplantation for Systemic Sclerosis: A Systematic Review and Meta-Analysis. Biology of Blood and Marrow Transplantation 2018, 24(5), 937–944. [Google Scholar] [CrossRef]
- Matsushita, T.; Kobayashi, T.; Mizumaki, K.; Kano, M.; Sawada, T.; Tennichi, M.; Okamura, A.; Hamaguchi, Y.; Iwakura, Y.; Hasegawa, M.; Fujimoto, M.; Takehara, K. BAFF Inhibition Attenuates Fibrosis in Scleroderma by Modulating the Regulatory and Effector B Cell Balance. Sci. Adv. 2018, 4(7), eaas9944. [Google Scholar] [CrossRef]
- Pecher, A.-C.; Hensen, L.; Schairer, R.; Klein, R.; Bethge, W.; Lengerke, C.; Henes, J. CD19-Targeting CAR T Cell Therapy in Five Patients with Systemic Sclerosis Unsuitable for Autologous Stem Cell Transplantation. EULAR Rheumatology Open 2025, S3050708125000084. [Google Scholar] [CrossRef]
- Auth, J.; Müller, F.; Völkl, S.; Bayerl, N.; Distler, J. H. W.; Tur, C.; Raimondo, M. G.; Chenguiti Fakhouri, S.; Atzinger, A.; Coppers, B.; Eckstein, M.; Liphardt, A.-M.; Bäuerle, T.; Tascilar, K.; Aigner, M.; Kretschmann, S.; Wirsching, A.; Taubmann, J.; Hagen, M.; Györfi, A.-H.; Kharboutli, S.; Krickau, T.; Dees, C.; Spörl, S.; Rothe, T.; Harrer, T.; Bozec, A.; Grieshaber-Bouyer, R.; Fuchs, F.; Kuwert, T.; Berking, C.; Horch, R. E.; Uder, M.; Mackensen, A.; Schett, G.; Bergmann, C. CD19-Targeting CAR T-Cell Therapy in Patients with Diffuse Systemic Sclerosis: A Case Series. The Lancet Rheumatology 2025, 7(2), e83–e93. [Google Scholar] [CrossRef]
- Wang, X.; Wu, X.; Tan, B.; Zhu, L.; Zhang, Y.; Lin, L.; Xiao, Y.; Sun, A.; Wan, X.; Liu, S.; Liu, Y.; Ta, N.; Zhang, H.; Song, J.; Li, T.; Zhou, L.; Yin, J.; Ye, L.; Lu, H.; Hong, J.; Cheng, H.; Wang, P.; Li, W.; Chen, J.; Zhang, J.; Luo, J.; Huang, M.; Guo, L.; Pan, X.; Jin, Y.; Ye, W.; Dai, L.; Zhu, J.; Sun, L.; Zheng, B.; Li, D.; He, Y.; Liu, M.; Wu, H.; Du, B.; Xu, H. Allogeneic CD19-Targeted CAR T Therapy in Patients with Severe Myositis and Systemic Sclerosis. Cell 2024, 187(18), 4890–4904.e9. [Google Scholar] [CrossRef]
- Merkt, W.; Röhrich, M.; Mavriopoulou, E.; Stütz, A. N.; Distler, J. H. W.; Schmitt, A.; Polke, M.; Heußel, C. P.; Schmitt, M.; Lorenz, H.-M. Persisting CD19.CAR T Cells in Combination with Nintedanib: Clinical Response in a Patient with Systemic Sclerosis-Associated Pulmonary Fibrosis after 2 Years. The Lancet Respiratory Medicine 2025, 13(7), 651–654. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, C.; Müller, F.; Distler, J. H. W.; Györfi, A.-H.; Völkl, S.; Aigner, M.; Kretschmann, S.; Reimann, H.; Harrer, T.; Bayerl, N.; Boeltz, S.; Wirsching, A.; Taubmann, J.; Rösler, W.; Spriewald, B.; Wacker, J.; Atzinger, A.; Uder, M.; Kuwert, T.; Mackensen, A.; Schett, G. Treatment of a Patient with Severe Systemic Sclerosis (SSc) Using CD19-Targeted CAR T Cells. Annals of the Rheumatic Diseases 2023, 82(8), 1117–1120. [Google Scholar] [CrossRef] [PubMed]
- Claus, M.; Freitag, M.; Ewald, M.; Rodon, L.; Deicher, F.; Watzl, C.; Kolb, P.; Lorenz, H.-M.; Schmitt, M.; Merkt, W. Immunological Effects of CD19.CAR T Cell Therapy in Systemic Sclerosis: An Extended Case Study. Arthritis Res Ther 2024, 26(1), 211. [Google Scholar] [CrossRef]
- Schett, G.; Bohr, D.; Mueller, F.; Hagen, M.; Bergmann, C.; Tur, C.; Völkl, S.; Aigner, M.; Krestchmann, S.; Spörl, S.; Vasova, I.; Aletaha, D.; Kiener, H.; Natalello, G.; Grieshaber-Bouyer, R.; Bozec, A.; Locatelli, F.; D`agostino, M. A.; Mackensen, A. POS0030 SAFETY AND PRELIMINARY EFFICACY OF CD19 CAR T CELL TREATMENT IN RHEUMATIC DISEASE- DATA FROM THE FIRST PART OF THE PHASE I/II CASTLE BASKET STUDY. Annals of the Rheumatic Diseases 2024, 83, 327. [Google Scholar] [CrossRef]
- Bristol Myers Squibb. Bristol Myers Squibb Presents Encouraging Data from Phase 1 Breakfree‐1 Study of CD19 NEX‐T CAR‐T Cell Therapy in Three Chronic Autoimmune Diseases at ACR Convergence 2025. Bristol Myers Squibb News, Nov 8, 2025. https://news.bms.com/news/details/2025/Bristol‐Myers‐Squibb‐Presents‐ Encouraging‐Data‐from‐Phase‐1‐Breakfree‐1‐Study‐of‐CD19‐NEX‐T‐CAR T‐Cell‐Therapy‐in‐Three‐ Chronic‐Autoimmune‐Diseases‐at‐ACR‐Convergence‐2025/.
- Lopes, J.; Rodrigues, C. M. P.; Gaspar, M. M.; Reis, C. P. Melanoma Management: From Epidemiology to Treatment and Latest Advances. Cancers (Basel) 2022, 14(19), 4652. [Google Scholar] [CrossRef]
- Lu, C.-T.; Lin, T.-L.; Mukundan, A.; Karmakar, R.; Chandrasekar, A.; Chang, W.-Y.; Wang, H.-C. Skin Cancer: Epidemiology, Screening and Clinical Features of Acral Lentiginous Melanoma (ALM), Melanoma In Situ (MIS), Nodular Melanoma (NM) and Superficial Spreading Melanoma (SSM). J. Cancer 2025, 16(13), 3972–3990. [Google Scholar] [CrossRef] [PubMed]
- Guy, G. P.; Thomas, C. C.; Thompson, T.; Watson, M.; Massetti, G. M.; Richardson, L. C.; Centers for Disease Control and Prevention (CDC). Vital Signs: Melanoma Incidence and Mortality Trends and Projections - United States, 1982-2030. MMWR Morb Mortal Wkly Rep 2015, 64(21), 591–596. [Google Scholar] [PubMed]
- Saginala, K.; Barsouk, A.; Aluru, J. S.; Rawla, P.; Barsouk, A. Epidemiology of Melanoma. Medical Sciences 2021, 9(4), 63. [Google Scholar] [CrossRef] [PubMed]
- Conforti, C.; Zalaudek, I. Epidemiology and Risk Factors of Melanoma: A Review. Dermatol Pract Concept 2021, 11 (Suppl 1), e2021161S. [Google Scholar] [CrossRef]
- Kalal, B. S.; Upadhya, D.; Pai, V. R. Chemotherapy Resistance Mechanisms in Advanced Skin Cancer. Oncol Rev 2017, 11(1), 326. [Google Scholar] [CrossRef]
- Carlino, M. S.; Larkin, J.; Long, G. V. Immune Checkpoint Inhibitors in Melanoma. Lancet 2021, 398(10304), 1002–1014. [Google Scholar] [CrossRef]
- Vanella, V.; Festino, L.; Trojaniello, C.; Vitale, M. G.; Sorrentino, A.; Paone, M.; Ascierto, P. A. The Role of BRAF-Targeted Therapy for Advanced Melanoma in the Immunotherapy Era. Curr Oncol Rep 2019, 21(9), 76. [Google Scholar] [CrossRef]
- Bagley, S. J.; O’Rourke, D. M. Clinical Investigation of CAR T Cells for Solid Tumors: Lessons Learned and Future Directions. Pharmacol Ther 2020, 205, 107419. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Qin, J.; Lin, B.; Zhang, J.; Li, D.; Liu, M. CAR T Therapy in Solid Tumors. Cancer Cell 2025, 43(4), 665–679. [Google Scholar] [CrossRef]
- Balch, C. M.; Gershenwald, J. E.; Soong, S.-J.; Thompson, J. F.; Atkins, M. B.; Byrd, D. R.; Buzaid, A. C.; Cochran, A. J.; Coit, D. G.; Ding, S.; Eggermont, A. M.; Flaherty, K. T.; Gimotty, P. A.; Kirkwood, J. M.; McMasters, K. M.; Mihm, M. C.; Morton, D. L.; Ross, M. I.; Sober, A. J.; Sondak, V. K. Final Version of 2009 AJCC Melanoma Staging and Classification. J Clin Oncol 2009, 27(36), 6199–6206. [Google Scholar] [CrossRef] [PubMed]
- Kasumova, G. G.; Haynes, A. B.; Boland, G. M. Lymphatic versus Hematogenous Melanoma Metastases: Support for Biological Heterogeneity without Clear Clinical Application. J Invest Dermatol 2017, 137(12), 2466–2468. [Google Scholar] [CrossRef] [PubMed]
- Gargett, T.; Truong, N. T. H.; Gardam, B.; Yu, W.; Ebert, L. M.; Johnson, A.; Yeo, E. C. F.; Wittwer, N. L.; Tapia Rico, G.; Logan, J.; Sivaloganathan, P.; Collis, M.; Ruszkiewicz, A.; Brown, M. P. Safety and Biological Outcomes Following a Phase 1 Trial of GD2-Specific CAR T Cells in Patients with GD2-Positive Metastatic Melanoma and Other Solid Cancers. J Immunother Cancer 2024, 12(5), e008659. [Google Scholar] [CrossRef]
- Aleksandrova, K.; Leise, J.; Priesner, C.; Aktas, M.; Apel, M.; Assenmacher, M.; Bürger, I.; Richter, A.; Altefrohne, P.; Schubert, C.; Holzinger, A.; Barden, M.; Bezler, V.; Von Bergwelt-Baildon, M.; Borchmann, P.; Goudeva, L.; Glienke, W.; Arseniev, L.; Esser, R.; Abken, H.; Koehl, U. Automated Manufacturing and Characterization of Clinical Grade Autologous CD20 CAR T Cells for the Treatment of Patients with Stage III/IV Melanoma. Front. Immunol. 2024, 15, 1328368. [Google Scholar] [CrossRef]
- Shah, P. D.; Huang, A. C.; Xu, X.; Orlowski, R.; Amaravadi, R. K.; Schuchter, L. M.; Zhang, P.; Tchou, J.; Matlawski, T.; Cervini, A.; Shea, J.; Gilmore, J.; Lledo, L.; Dengel, K.; Marshall, A.; Wherry, E. J.; Linette, G. P.; Brennan, A.; Gonzalez, V.; Kulikovskaya, I.; Lacey, S. F.; Plesa, G.; June, C. H.; Vonderheide, R. H.; Mitchell, T. C. Phase I Trial of Autologous RNA-Electroporated cMET-Directed CAR T Cells Administered Intravenously in Patients with Melanoma and Breast Carcinoma. Cancer Research Communications 2023, 3(5), 821–829. [Google Scholar] [CrossRef]
- Rosenberg, S.A.; Somerville, R.; Feldman, S.A.; Restifo, N.P.; Yang, J.C.; Sherry, R.; Kammula, U.S.; Goff, S.; Klebanoff, C. Protocol: Phase I/II study of anti-VEGFR2 CAR-T cell therapy in metastatic melanoma. National Cancer Institute; ClinicalTrials.gov Identifier: NCT01218867. 2010. Available online: https://cdn.clinicaltrials.gov/large-docs/67/NCT01218867/Prot_SAP_000.pdf.
- Jilani, S.; Saco, J. D.; Mugarza, E.; Pujol-Morcillo, A.; Chokry, J.; Ng, C.; Abril-Rodriguez, G.; Berger-Manerio, D.; Pant, A.; Hu, J.; Gupta, R.; Vega-Crespo, A.; Baselga-Carretero, I.; Chen, J. M.; Shin, D. S.; Scumpia, P.; Radu, R. A.; Chen, Y.; Ribas, A.; Puig-Saus, C. CAR T Cell Therapy Targeting Surface Expression of TYRP1 to Treat Cutaneous and Rare Melanoma Subtypes. Nat Commun 2024, 15(1), 1244. [Google Scholar] [CrossRef]
- Bagherani, N.; Smoller, B. R. An Overview of Cutaneous T Cell Lymphomas. F1000Res 2016, 5, F1000 Faculty Rev–1882. [Google Scholar] [CrossRef]
- Brunner, P. M.; Jonak, C.; Knobler, R. Recent Advances in Understanding and Managing Cutaneous T-Cell Lymphomas. F1000Res 2020, 9, F1000 Faculty Rev–331. [Google Scholar] [CrossRef] [PubMed]
- Lovgren, M.-L.; Scarisbrick, J. J. Update on Skin Directed Therapies in Mycosis Fungoides. Chin Clin Oncol 2019, 8(1), 7. [Google Scholar] [CrossRef] [PubMed]
- Case, K. B.; Allen, P. B. Advances in Novel Systemic Therapies for the Management of Cutaneous T Cell Lymphoma (CTCL). Curr Hematol Malig Rep 2025, 20(1), 5. [Google Scholar] [CrossRef]
- Evtimov, V. J.; Hammett, M. V.; Pupovac, A.; Nguyen, N.-Y. N.; Shu, R.; Van Der Weyden, C.; Twigger, R.; Nisbet, I. T.; Trounson, A. O.; Boyd, R. L.; Prince, H. M. Targeting TAG-72 in Cutaneous T Cell Lymphoma. Heliyon 2024, 10(17), e36298. [Google Scholar] [CrossRef]
- To, V.; Evtimov, V. J.; Shu, R.; Pupovac, A.; Boyd, R. L.; Trounson, A. O. Engineering TAG-72 and CD30 CAR T Cells for T Cell Malignancies. Immunol Invest 2025, 54(8), 1399–1420. [Google Scholar] [CrossRef]
- Nicolay, J. P.; Albrecht, J. D.; Alberti-Violetti, S.; Berti, E. CCR4 in Cutaneous T-Cell Lymphoma: Therapeutic Targeting of a Pathogenic Driver. Eur J Immunol 2021, 51(7), 1660–1671. [Google Scholar] [CrossRef]
- Watanabe, K.; Gomez, A. M.; Kuramitsu, S.; Siurala, M.; Da, T.; Agarwal, S.; Song, D.; Scholler, J.; Rotolo, A.; Posey, A. D.; Rook, A. H.; Haun, P. L.; Ruella, M.; Young, R. M.; June, C. H. Identifying Highly Active Anti-CCR4 CAR T Cells for the Treatment of T-Cell Lymphoma. Blood Adv 2023, 7(14), 3416–3430. [Google Scholar] [CrossRef]
- Reef, D. K.; Cheng, C. J. A.; Babinec, C.; Hoye, K.; West, J.; Morrison, J. K.; Buchanan, F. B.; Zanter, A.; Spruill, A. D.; Kasow, K. A.; Ivanova, A.; Bowers, E.; Serody, J.; Savoldo, B.; Dotti, G.; Grover, N.; Beaven, A. W. Phase 1 Trial of CD30-Directed, CCR4 Co-Expressing Chimeric Antigen Receptor T-Cells in Patients with CD30+ Cutaneous T-Cell Lymphomas Refractory to Brentuximab Vedotin. Blood 2024, 144 (Supplement 1), 3468–3468. [Google Scholar] [CrossRef]
- Iyer, S. P.; Sica, R. A.; Ho, P. J.; Prica, A.; Zain, J.; Foss, F. M.; Hu, B.; Beitinjaneh, A.; Weng, W.-K.; Kim, Y. H.; Khodadoust, M. S.; Huen, A. O.; Williams, L. M.; Ma, A.; Huang, E.; Ganpule, A.; Nagar, S. D.; Sripakdeevong, P.; Cullingford, E. L.; Karnik, S.; Dequeant, M.-L.; Patel, J. N.; He, X. S.; Li, Z.; He, Q. A.; Mendonez, J. H.; Keegan, A.; Horwitz, S. M. Safety and Activity of CTX130, a CD70-Targeted Allogeneic CRISPR-Cas9-Engineered CAR T-Cell Therapy, in Patients with Relapsed or Refractory T-Cell Malignancies (COBALT-LYM): A Single-Arm, Open-Label, Phase 1, Dose-Escalation Study. Lancet Oncol 2025, 26(1), 110–122. [Google Scholar] [CrossRef]
- Weber, T.; Burchert, A.; Subklewe, M. [CAR T-cell therapy of mature B-cell neoplasms-Current use and practical considerations]. Inn Med (Heidelb) 2025, 66(8), 793–802. [Google Scholar] [CrossRef] [PubMed]
- Dumont, M.; Battistella, M.; Ram-Wolff, C.; Bagot, M.; de Masson, A. Diagnosis and Treatment of Primary Cutaneous B-Cell Lymphomas: State of the Art and Perspectives. Cancers (Basel) 2020, 12(6), 1497. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, J.; Qiao, L.; Chen, L.; Chen, C.; Zhang, H.; Luo, R.; Xiao, Y. Case Report: Cryoablation as a Novel Bridging Strategy Prior to CAR T Cell Therapy for B Cell Malignancies with Bulky Disease. Front Oncol 2023, 13, 1008828. [Google Scholar] [CrossRef] [PubMed]
- Bouchkouj, N.; Kasamon, Y. L.; de Claro, R. A.; George, B.; Lin, X.; Lee, S.; Blumenthal, G. M.; Bryan, W.; McKee, A. E.; Pazdur, R. FDA Approval Summary: Axicabtagene Ciloleucel for Relapsed or Refractory Large B-Cell Lymphoma. Clin Cancer Res 2019, 25(6), 1702–1708. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. FDA Approves Tisagenlecleucel for Adults with Relapsed or Refractory Large B-Cell Lymphoma. U.S. Food and Drug Administration, 1 May 2018. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-tisagenlecleucel-adults-relapsed-or-refractory-large-b-cell-lymphoma (accessed on 31 December 2025).
- Barbati, Z. R.; Charli-Joseph, Y. Unveiling Primary Cutaneous B-Cell Lymphomas: New Insights into Diagnosis and Treatment Strategies. Cancers (Basel) 2025, 17(7), 1202. [Google Scholar] [CrossRef] [PubMed]
- Mohty, R.; Moustafa, M. A.; Aljurf, M.; Murthy, H.; Kharfan-Dabaja, M. A. Emerging Role of Autologous CD19 CAR T-Cell Therapies in the Second-Line Setting for Large B-Cell Lymphoma: A Game Changer? Hematol Oncol Stem Cell Ther 2022, 15(3), 73–80. [Google Scholar] [CrossRef] [PubMed]
- Locke, F.L.; Miklos, D.B.; Jacobson, C.A.; Perales, M.A.; Kersten, M.J.; Oluwole, O.O.; Ghobadi, A.; Rapoport, A.P.; McGuirk, J.; Pagel, J.M.; et al. All ZUMA-7 Investigators and Contributing Kite Members. Axicabtagene Ciloleucel as Second-Line Therapy for Large B-Cell Lymphoma. N. Engl. J. Med. 2022, 386, 640–654. [Google Scholar] [CrossRef]
- Bishop, M.R.; Dickinson, M.; Purtill, D.; Barba, P.; Santoro, A.; Hamad, N.; Kato, K.; Sureda, A.; Greil, R.; Thieblemont, C.; et al. Second-Line Tisagenlecleucel or Standard Care in Aggressive B-Cell Lymphoma. N. Engl. J. Med. Google Scholar] [CrossRef. 2022, 386, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues Dos Santos, Andressa; et al. Cytokine Release Syndrome after Chimeric Antigen Receptor T Cell Therapy in Patients with Diffuse Large B-Cell Lymphoma: A Systematic Review”. Hematology, Transfusion and Cell Therapy 2024, t. 46, s. S306–15. [Google Scholar] [CrossRef]
- Nasiri, Fatemeh; et al. Updates on CAR T Cell Therapy in Multiple Myeloma”. Biomarker Research 2024, t. 12(nr 1), s. 102. [Google Scholar] [CrossRef] [PubMed]
- Choy, Ernest H.; et al. Translating IL-6 Biology into Effective Treatments”. Nature Reviews Rheumatology 2020, t. 16(nr 6), s. 335–45. [Google Scholar] [CrossRef] [PubMed]
- Giavridis, Theodoros; et al. „CAR T Cell–Induced Cytokine Release Syndrome Is Mediated by Macrophages and Abated by IL-1 Blockade”. Nature Medicine 2018, t. 24(nr 6), s. 731–38. [Google Scholar] [CrossRef]
- Strati, Paolo; et al. Prognostic Impact of Dose, Duration, and Timing of Corticosteroid Therapy in Patients with Large B-Cell Lymphoma Treated with Standard of Care Axicabtagene Ciloleucel (Axi-Cel). Journal of Clinical Oncology 2020, t. 38(nr 15_) suppl, s. 8011–8011. [Google Scholar] [CrossRef]
- Frigault, Matthew J.; et al. „Single-Cell Dynamics of Breakthrough Toxicities after Anakinra Prophylaxis for Axicabtagene Ciloleucel in Lymphoma”. Blood Advances 2025, t. 9(nr 9), s. 2122–35. [Google Scholar] [CrossRef] [PubMed]
- Dubovsky, Jason A.; et al. Ibrutinib Is an Irreversible Molecular Inhibitor of ITK Driving a Th1-Selective Pressure in T Lymphocytes”. Blood 2013, t. 122(nr 15), s. 2539–49. [Google Scholar] [CrossRef]
- Hernani, Rafael; et al. Toxicities Following CAR T Therapy for Hematological Malignancies”. Cancer Treatment Reviews 2022, t. 111, s. 102479. [Google Scholar] [CrossRef]
- Santomasso, Bianca D.; et al. Clinical and Biological Correlates of Neurotoxicity Associated with CAR T-Cell Therapy in Patients with B-Cell Acute Lymphoblastic Leukemia”. Cancer Discovery 2018, t. 8(nr 8), s. 958–71. [Google Scholar] [CrossRef]
- Mahadeo, Kris M.; et al. Management Guidelines for Paediatric Patients Receiving Chimeric Antigen Receptor T Cell Therapy”. Nature Reviews Clinical Oncology 2019, t. 16(nr 1), s. 45–63. [Google Scholar] [CrossRef]
- Santomasso, Bianca D.; et al. Management of Immune-Related Adverse Events in Patients Treated With Chimeric Antigen Receptor T-Cell Therapy: ASCO Guideline”. Journal of Clinical Oncology 2021, t. 39(nr 35), s. 3978–92. [Google Scholar] [CrossRef] [PubMed]
- Diorio, Caroline; et al. „Comprehensive Serum Proteome Profiling of Cytokine Release Syndrome and Immune Effector Cell–Associated Neurotoxicity Syndrome Patients with B-Cell ALL Receiving CAR T19”. Clinical Cancer Research 2022, t. 28(nr 17), s. 3804–13. [Google Scholar] [CrossRef]
- Rejeski, Kai; et al. Immune Effector Cell-Associated Haematotoxicity after CAR T-Cell Therapy: From Mechanism to Management”. The Lancet Haematology 2024, t. 11(nr 6), s. e459–70. [Google Scholar] [CrossRef]
- Rejeski, Kai; et al. Immune Effector Cell–Associated Hematotoxicity: EHA/EBMT Consensus Grading and Best Practice Recommendations”. Blood 2023, t. 142(nr 10), s. 865–77. [Google Scholar] [CrossRef]
- Rejeski, Kai; et al. „CAR-HEMATOTOX: A Model for CAR T-Cell–Related Hematologic Toxicity in Relapsed/Refractory Large B-Cell Lymphoma”. Blood 2021, t. 138(nr 24), s. 2499–513. [Google Scholar] [CrossRef]
- Sterner, Rosalie M.; et al. GM-CSF Inhibition Reduces Cytokine Release Syndrome and Neuroinflammation but Enhances CAR T Cell Function in Xenografts”. Blood 2019, t. 133(nr 7), s. 697–709. [Google Scholar] [CrossRef] [PubMed]
- Rejeski, Kai; et al. Applying the EHA/EBMT Grading for ICAHT after CAR T: Comparative Incidence and Association with Infections and Mortality”. Blood Advances 2024, t. 8(nr 8), s. 1857–68. [Google Scholar] [CrossRef]
- Deyà-Martínez, A.; et al. „Kinetics of Humoral Deficiency in CART19-Treated Children and Young Adults with Acute Lymphoblastic Leukaemia”. Bone Marrow Transplantation 2021, t. 56(nr 2), s. 376–86. [Google Scholar] [CrossRef]
- Parker, Kevin R.; et al. Single-Cell Analyses Identify Brain Mural Cells Expressing CD19 as Potential Off-Tumor Targets for CAR T Immunotherapies”. Cell 2020, t. 183(nr 1), s. 126–142.e17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Yugu; et al. Exploring CAR T Cell Therapy Side Effects: Mechanisms and Management Strategies”. Journal of Clinical Medicine 2023, t. 12(nr 19), s. 6124. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, Ton N., i Robert D. Schreiber. Neoantigens in Cancer Immunotherapy”. Science 2015, t. 348(nr 6230), s. 69–74. [CrossRef]
- Flugel, Christian L.; et al. Overcoming On-Target, off-Tumour Toxicity of CAR T Cell Therapy for Solid Tumours”. Nature Reviews Clinical Oncology 2023, t. 20(nr 1), s. 49–62. [Google Scholar] [CrossRef]
- Peters, Daniel T.; et al. Building Safety into CAR T Therapy”. Human Vaccines & Immunotherapeutics 2023, t. 19(nr 3), s. 2275457. [Google Scholar] [CrossRef]
- Abou-el-Enein, Mohamed; et al. „Scalable Manufacturing of CAR T Cells for Cancer Immunotherapy”. Blood Cancer Discovery 2021, t. 2(nr 5), s. 408–22. [Google Scholar] [CrossRef] [PubMed]
- June, Carl H., i Michel Sadelain. Chimeric Antigen Receptor Therapy”. New England Journal of Medicine 2018, t. 379(nr 1), s. 64–73. [CrossRef]
- Bot, Adrian; et al. In Vivo Chimeric Antigen Receptor (CAR)-T Cell Therapy”. Nature Reviews Drug Discovery 2026, t. 25(nr 2), s. 116–37. [Google Scholar] [CrossRef] [PubMed]
- Yang, Chunmei; et al. „Allogeneic Anti-CD19 CAR T Cells Induce Remission in Refractory Systemic Lupus Erythematosus”. Cell Research 2025, t. 35(nr 8), s. 607–09. [Google Scholar] [CrossRef]
- Sayed, Omima Ahmed; et al. CAR T Cell Therapy Efficacy and Safety in SLE: A Systematic Review and Pooled Analysis of 47 Patients across 10 Studies”. Naunyn-Schmiedeberg’s Archives of Pharmacology 2026, t. 399(nr 2), s. 1565–88. [Google Scholar] [CrossRef]
- Wu, Yuanhao; et al. „CAR T therapy: pioneering a new era in the treatment of autoimmune diseases”. Frontiers in Immunology 2025, t. 16, s. 1625166. [Google Scholar] [CrossRef] [PubMed]
- Matarneh, Ahmad; et al. „Chimeric Antigen Receptor T Cell Therapy in Systemic Lupus Erythematosus: Mechanisms, Clinical Advances, and Future Directions a Comprehensive Review”. Clinical Reviews in Allergy & Immunology 2025, t. 68(nr 1), s. 103. [Google Scholar] [CrossRef]
- Schett, Georg, i Carl H. June. „CAR T Cells in Autoimmune Disease: On the Road to Remission”. Immunity 2024, t. 57(nr 12), s. 2705–09. [CrossRef]
- Wang, Qian; et al. „In Vivo CD19 CAR T-Cell Therapy for Refractory Systemic Lupus Erythematosus”. New England Journal of Medicine 2025, t. 393(nr 15), s. 1542–44. [Google Scholar] [CrossRef]
- Cappell, Kathryn M., i James N. Kochenderfer. Long-Term Outcomes Following CAR T Cell Therapy: What We Know so Far”. Nature Reviews Clinical Oncology 2023, t. 20(nr 6), s. 359–71. [CrossRef] [PubMed]
- Pecher, A.C.; Hensen, L.; Klein, R.; et al. CD19-Targeting CAR T Cells for Myositis and Interstitial Lung Disease Associated With Antisynthetase Syndrome. JAMA 2023, 329(24), 2154–2162. [Google Scholar] [CrossRef] [PubMed]
- Schett, Georg; et al. Advancements and Challenges in CAR T Cell Therapy in Autoimmune Diseases”. Nature Reviews Rheumatology 2024, t. 20(nr 9), s. 531–44. [Google Scholar] [CrossRef]
- Avouac, Jérôme; et al. „Roads and Detours for CAR T Cell Therapy in Autoimmune Diseases”. Nature Reviews Drug Discovery 2026. [Google Scholar] [CrossRef]
- Mackensen, Andreas; et al. „Anti-CD19 CAR T Cell Therapy for Refractory Systemic Lupus Erythematosus”. Nature Medicine 2022, t. 28(nr 10), s. 2124–32. [Google Scholar] [CrossRef]
- Auth, Janina; et al. „CD19-Targeting CAR T-Cell Therapy in Patients with Diffuse Systemic Sclerosis: A Case Series”. The Lancet Rheumatology 2025, t. 7(nr 2), s. e83–93. [Google Scholar] [CrossRef]
- Wang, X.; Wu, X.; Tan, B.; et al. Allogeneic CD19-targeted CAR T therapy in patients with severe myositis and systemic sclerosis. Cell. 2024, 187(18), 4890–4904.e9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Song-yun; et al. Incidentally cured psoriasis in a patient with refractory/relapsed diffuse large B-cell lymphoma receiving CD19 CAR T cell therapy: a case report”. Frontiers in Immunology 2024, t. 15, s. 1418768. [Google Scholar] [CrossRef] [PubMed]
- Rendon, Adriana, i Knut Schäkel. Psoriasis Pathogenesis and Treatment”. International Journal of Molecular Sciences 2019, t. 20(nr 6), s. 1475. [CrossRef] [PubMed]
- Petit; Robert Gironés; et al. Psoriasis: From Pathogenesis to Pharmacological and Nano-Technological-Based Therapeutics”. International Journal of Molecular Sciences 2021, t. 22(nr 9), s. 4983. [Google Scholar] [CrossRef]
- Bottai, Matteo; et al. EULAR/ACR Classification Criteria for Adult and Juvenile Idiopathic Inflammatory Myopathies and Their Major Subgroups: A Methodology Report”. RMD Open 2017, t. 3(nr 2), s. e000507. [Google Scholar] [CrossRef]
- Groener, Marwin, i Julie J. Paik. „Emerging B and plasma cell-targeting immune therapies in idiopathic inflammatory myopathies”. Frontiers in Immunology 2025, t. 16, s. 1581323. [CrossRef]
- Wang, Guochun; i Neil, John; McHugh. An Update on Myositis Autoantibodies and Insights into Pathogenesis”. Clinical and Experimental Rheumatology 2024. [Google Scholar] [CrossRef] [PubMed]
- Patil, Harshwardhan; et al. CAR T Cell Therapy in Rheumatic Diseases: A Review Article”. Clinical Rheumatology 2026, t. 45(nr 2), s. 637–50. [Google Scholar] [CrossRef] [PubMed]
- alTrials.gov. Phase I Dose Escalation Study of Systemically Administered IL13Ra2 Chimeric Antigen Receptor (CAR) T Cells After a Nonmyeloablative Conditioning Regimen in Patients With Metastatic Melanoma and Other Solid Tumors; Identifier NCT04119024; U.S. National Library of Medicine: Bethesda, MD; Available online: https://clinicaltrials.gov/study/NCT04119024 (accessed on 2026-03-04).
- Kwon, N.; Chen, Y. Y. Overcoming Solid-Tumor Barriers: Armored CAR T Cell Therapy. Trends in Cancer 2025, 11(10), 1019–1029. [Google Scholar] [CrossRef]
- Marotte, L.; Simon, S.; Vignard, V.; Dupre, E.; Gantier, M.; Cruard, J.; Alberge, J.-B.; Hussong, M.; Deleine, C.; Heslan, J.-M.; Shaffer, J.; Beauvais, T.; Gaschet, J.; Scotet, E.; Fradin, D.; Jarry, A.; Nguyen, T.; Labarriere, N. Increased Antitumor Efficacy of PD-1-Deficient Melanoma-Specific Human Lymphocytes. J Immunother Cancer 2020, 8(1), e000311. [Google Scholar] [CrossRef]
- Masoumi, E.; Jafarzadeh, L.; Mirzaei, H. R.; Alishah, K.; Fallah-Mehrjardi, K.; Rostamian, H.; Khakpoor-Koosheh, M.; Meshkani, R.; Noorbakhsh, F.; Hadjati, J. Genetic and Pharmacological Targeting of A2a Receptor Improves Function of Anti-Mesothelin CAR T Cells. J Exp Clin Cancer Res 2020, 39(1), 49. [Google Scholar] [CrossRef] [PubMed]
- Beavis, P. A.; Henderson, M. A.; Giuffrida, L.; Mills, J. K.; Sek, K.; Cross, R. S.; Davenport, A. J.; John, L. B.; Mardiana, S.; Slaney, C. Y.; Johnstone, R. W.; Trapani, J. A.; Stagg, J.; Loi, S.; Kats, L.; Gyorki, D.; Kershaw, M. H.; Darcy, P. K. Targeting the Adenosine 2A Receptor Enhances Chimeric Antigen Receptor T Cell Efficacy. Journal of Clinical Investigation 2017, 127(3), 929–941. [Google Scholar] [CrossRef]
- Chinnasamy, D.; Yu, Z.; Kerkar, S. P.; Zhang, L.; Morgan, R. A.; Restifo, N. P.; Rosenberg, S. A. Local Delivery of Lnterleukin-12 Using T Cells Targeting VEGF Receptor-2 Eradicates Multiple Vascularized Tumors in Mice. Clinical Cancer Research 2012, 18(6), 1672–1683. [Google Scholar] [CrossRef]
- Sikorski, H.; Żmijewski, M. A.; Piotrowska, A. Tumor Microenvironment in Melanoma—Characteristic and Clinical Implications. IJMS 2025, 26(14), 6778. [Google Scholar] [CrossRef]
- Mollica Poeta, V.; Massara, M.; Capucetti, A.; Bonecchi, R. Chemokines and Chemokine Receptors: New Targets for Cancer Immunotherapy. Front. Immunol. 2019, 10, 379. [Google Scholar] [CrossRef]
- Harlin, H.; Meng, Y.; Peterson, A. C.; Zha, Y.; Tretiakova, M.; Slingluff, C.; McKee, M.; Gajewski, T. F. Chemokine Expression in Melanoma Metastases Associated with CD8+ T-Cell Recruitment. Cancer Research 2009, 69(7), 3077–3085. [Google Scholar] [CrossRef]
- Tan, K. W.; Evrard, M.; Tham, M.; Hong, M.; Huang, C.; Kato, M.; Prevost-Blondel, A.; Donnadieu, E.; Ng, L. G.; Abastado, J.-P. Tumor Stroma and Chemokines Control T-Cell Migration into Melanoma Following Temozolomide Treatment. OncoImmunology 2015, 4(2), e978709. [Google Scholar] [CrossRef]
- Caruana, I.; Savoldo, B.; Hoyos, V.; Weber, G.; Liu, H.; Kim, E. S.; Ittmann, M. M.; Marchetti, D.; Dotti, G. Heparanase Promotes Tumor Infiltration and Antitumor Activity of CAR-Redirected T Lymphocytes. Nat Med 2015, 21(5), 524–529. [Google Scholar] [CrossRef] [PubMed]
- Merimsky, O.; Shoenfeld, Y.; Chaitchik, S.; Yecheskel, G.; Fishman, P. Antigens and Antibodies in Malignant Melanoma. Tumor Biol 1994, 15(4), 188–202. [Google Scholar] [CrossRef] [PubMed]
- Grzywa, T. M.; Paskal, W.; Włodarski, P. K. Intratumor and Intertumor Heterogeneity in Melanoma. Transl Oncol 2017, 10(6), 956–975. [Google Scholar] [CrossRef]
- Kailayangiri, S.; Altvater, B.; Wiebel, M.; Jamitzky, S.; Rossig, C. Overcoming Heterogeneity of Antigen Expression for Effective CAR T Cell Targeting of Cancers. Cancers 2020, 12(5), 1075. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Melero, S.; Hassouneh, F.; Vallejo-Bermúdez, I. M.; Agüera-Morales, E.; Solana, R.; Caballero-Villarraso, J. Tandem CAR T Cell Therapy: Recent Advances and Current Challenges. Front. Immunol. 2025, 16, 1546172. [Google Scholar] [CrossRef]
- Chen, T.; Wang, M.; Chen, Y.; Liu, Y. Current Challenges and Therapeutic Advances of CAR T Cell Therapy for Solid Tumors. Cancer Cell Int 2024, 24(1), 133. [Google Scholar] [CrossRef]
- Flugel, C. L.; Majzner, R. G.; Krenciute, G.; Dotti, G.; Riddell, S. R.; Wagner, D. L.; Abou-el-Enein, M. Overcoming On-Target, off-Tumour Toxicity of CAR T Cell Therapy for Solid Tumours. Nat Rev Clin Oncol 2023, 20(1), 49–62. [Google Scholar] [CrossRef]
- Chawla-Sarkar, M.; Bae, S. I.; Reu, F. J.; Jacobs, B. S.; Lindner, D. J.; Borden, E. C. Downregulation of Bcl-2, FLIP or IAPs (XIAP and Survivin) by siRNAs Sensitizes Resistant Melanoma Cells to Apo2L/TRAIL-Induced Apoptosis. Cell Death Differ 2004, 11(8), 915–923. [Google Scholar] [CrossRef]
- Pourzia, A. L.; Olson, M. L.; Bailey, S. R.; Boroughs, A. C.; Aryal, A.; Ryan, J.; Maus, M. V.; Letai, A. Quantifying Requirements for Mitochondrial Apoptosis in CAR T Killing of Cancer Cells. Cell Death Dis 2023, 14(4), 267. [Google Scholar] [CrossRef]
- Zhang, L.; Fang, B. Mechanisms of Resistance to TRAIL-Induced Apoptosis in Cancer. Cancer Gene Ther 2005, 12(3), 228–237. [Google Scholar] [CrossRef]
- Diepstraten, S. T.; Anderson, M. A.; Czabotar, P. E.; Lessene, G.; Strasser, A.; Kelly, G. L. The Manipulation of Apoptosis for Cancer Therapy Using BH3-Mimetic Drugs. Nat Rev Cancer 2022, 22(1), 45–64. [Google Scholar] [CrossRef]
- Ozyerli-Goknar, E.; Bagci-Onder, T. Epigenetic Deregulation of Apoptosis in Cancers. Cancers (Basel) 2021, 13(13), 3210. [Google Scholar] [CrossRef]
- Refaat, A.; Abd-Rabou, A.; Reda, A. TRAIL Combinations: The New ‘Trail’ for Cancer Therapy (Review). Oncology Letters 2014, 7(5), 1327–1332. [Google Scholar] [CrossRef] [PubMed]
- Andtbacka, R. H. I.; Kaufman, H. L.; Collichio, F.; Amatruda, T.; Senzer, N.; Chesney, J.; Delman, K. A.; Spitler, L. E.; Puzanov, I.; Agarwala, S. S.; Milhem, M.; Cranmer, L.; Curti, B.; Lewis, K.; Ross, M.; Guthrie, T.; Linette, G. P.; Daniels, G. A.; Harrington, K.; Middleton, M. R.; Miller, W. H.; Zager, J. S.; Ye, Y.; Yao, B.; Li, A.; Doleman, S.; VanderWalde, A.; Gansert, J.; Coffin, R. S. Talimogene Laherparepvec Improves Durable Response Rate in Patients With Advanced Melanoma. JCO 2015, 33(25), 2780–2788. [Google Scholar] [CrossRef]
- Tanoue, K.; Rosewell Shaw, A.; Watanabe, N.; Porter, C.; Rana, B.; Gottschalk, S.; Brenner, M.; Suzuki, M. Armed Oncolytic Adenovirus–Expressing PD-L1 Mini-Body Enhances Antitumor Effects of Chimeric Antigen Receptor T Cells in Solid Tumors. Cancer Research 2017, 77(8), 2040–2051. [Google Scholar] [CrossRef]
- Guedan, S.; Alemany, R. CAR T Cells and Oncolytic Viruses: Joining Forces to Overcome the Solid Tumor Challenge. Front. Immunol. 2018, 9, 2460. [Google Scholar] [CrossRef]
- Evgin, L.; Huff, A. L.; Wongthida, P.; Thompson, J.; Kottke, T.; Tonne, J.; Schuelke, M.; Ayasoufi, K.; Driscoll, C. B.; Shim, K. G.; Reynolds, P.; Monie, D. D.; Johnson, A. J.; Coffey, M.; Young, S. L.; Archer, G.; Sampson, J.; Pulido, J.; Perez, L. S.; Vile, R. Oncolytic Virus-Derived Type I Interferon Restricts CAR T Cell Therapy. Nat Commun 2020, 11(1), 3187. [Google Scholar] [CrossRef]
- Ott, P. A.; Hodi, F. S.; Robert, C. CTLA-4 and PD-1/PD-L1 Blockade: New Immunotherapeutic Modalities with Durable Clinical Benefit in Melanoma Patients. Clin Cancer Res 2013, 19(19), 5300–5309. [Google Scholar] [CrossRef] [PubMed]
- Misawa, K.; Bhat, H.; Adusumilli, P. S.; Hou, Z. Combinational CAR T-Cell Therapy for Solid Tumors: Requisites, Rationales, and Trials. Pharmacology & Therapeutics 2025, 266, 108763. [Google Scholar] [CrossRef]
- Grosser, R.; Cherkassky, L.; Chintala, N.; Adusumilli, P. S. Combination Immunotherapy with CAR T Cells and Checkpoint Blockade for the Treatment of Solid Tumors. Cancer Cell 2019, 36(5), 471–482. [Google Scholar] [CrossRef] [PubMed]
- Szczygielski, O.; Dąbrowska, E.; Niemyjska, S.; Przylipiak, A.; Zajkowska, M. Targeting Matrix Metalloproteinases and Their Inhibitors in Melanoma. IJMS 2024, 25(24), 13558. [Google Scholar] [CrossRef] [PubMed]
- Faraj, J. A.; Al-Athari, A. J. H.; Mohie, S. E. D.; Kadhim, I. K.; Jawad, N. M.; Abbas, W. J.; Jalil, A. T. Reprogramming the Tumor Microenvironment to Improve the Efficacy of Cancer Immunotherapies. Med Oncol 2022, 39(12), 239. [Google Scholar] [CrossRef] [PubMed]
- Habib, S.; Osborn, G.; Willsmore, Z.; Chew, M. W.; Jakubow, S.; Fitzpatrick, A.; Wu, Y.; Sinha, K.; Lloyd-Hughes, H.; Geh, J. L. C.; MacKenzie-Ross, A. D.; Whittaker, S.; Sanz-Moreno, V.; Lacy, K. E.; Karagiannis, S. N.; Adams, R. Tumor Associated Macrophages as Key Contributors and Targets in Current and Future Therapies for Melanoma. Expert Review of Clinical Immunology 2024, 20(8), 895–911. [Google Scholar] [CrossRef]
- Scarfò, I.; Frigault, M. J.; Maus, M. V. CAR-Based Approaches to Cutaneous T-Cell Lymphoma. Front. Oncol. 2019, 9, 259. [Google Scholar] [CrossRef]
- Bupha-Intr, O.; Haeusler, G.; Chee, L.; Thursky, K.; Slavin, M.; Teh, B. CAR T Cell Therapy and Infection: A Review. Expert Review of Anti-infective Therapy 2021, 19(6), 749–758. [Google Scholar] [CrossRef]
- Gaydosik, A. M.; Tabib, T.; Geskin, L. J.; Bayan, C.-A.; Conway, J. F.; Lafyatis, R.; Fuschiotti, P. Single-Cell Lymphocyte Heterogeneity in Advanced Cutaneous T-Cell Lymphoma Skin Tumors. Clinical Cancer Research 2019, 25(14), 4443–4454. [Google Scholar] [CrossRef]
- Decroos, A.; Cheminant, M.; Bruneau, J.; Carras, S.; Parinet, V.; Pelletier, L.; Lacroix, L.; Martin, N.; Giustiniani, J.; Lhermitte, L.; Asnafi, V.; Battistella, M.; Lemonnier, F.; De Leval, L.; Sicard, H.; Bonnafous, C.; Gauthier, L.; Genestier, L.; Caruso, S.; Gaulard, P.; Hermine, O.; Ortonne, N. KIR3DL2 May Represent a Novel Therapeutic Target in Aggressive Systemic Peripheral T-Cell Lymphoma. haematol 2023, 108(10), 2830–2836. [Google Scholar] [CrossRef]
- Thonnart, N.; Caudron, A.; Legaz, I.; Bagot, M.; Bensussan, A.; Marie-Cardine, A. KIR3DL2 Is a Coinhibitory Receptor on Sézary Syndrome Malignant T Cells That Promotes Resistance to Activation-Induced Cell Death. Blood 2014, 124(22), 3330–3332. [Google Scholar] [CrossRef]
- Cwynarski, K.; Iacoboni, G.; Tholouli, E.; Menne, T.; Irvine, D. A.; Balasubramaniam, N.; Wood, L.; Shang, J.; Xue, E.; Zhang, Y.; Basilico, S.; Neves, M.; Raymond, M.; Scott, I.; El-Kholy, M.; Jha, R.; Dainton-Smith, H.; Hussain, R.; Day, W.; Ferrari, M.; Thomas, S.; Lilova, K.; Brugger, W.; Marafioti, T.; Lao-Sirieix, P.; Maciocia, P.; Pule, M. TRBC1-CAR T Cell Therapy in Peripheral T Cell Lymphoma: A Phase 1/2 Trial. Nat Med 2025, 31(1), 137–143. [Google Scholar] [CrossRef]
- Maciocia, P. M.; Wawrzyniecka, P. A.; Philip, B.; Ricciardelli, I.; Akarca, A. U.; Onuoha, S. C.; Legut, M.; Cole, D. K.; Sewell, A. K.; Gritti, G.; Somja, J.; Piris, M. A.; Peggs, K. S.; Linch, D. C.; Marafioti, T.; Pule, M. A. Targeting the T Cell Receptor β-Chain Constant Region for Immunotherapy of T Cell Malignancies. Nat Med 2017, 23(12), 1416–1423. [Google Scholar] [CrossRef]
- Ren, A.; Tong, X.; Xu, N.; Zhang, T.; Zhou, F.; Zhu, H. CAR T-Cell Immunotherapy Treating T-ALL: Challenges and Opportunities. Vaccines 2023, 11(1), 165. [Google Scholar] [CrossRef]
- To, V.; Evtimov, V. J.; Jenkin, G.; Pupovac, A.; Trounson, A. O.; Boyd, R. L. CAR T Cell Development for Cutaneous T Cell Lymphoma: Current Limitations and Potential Treatment Strategies. Front Immunol 2022, 13, 968395. [Google Scholar] [CrossRef]
- Han, X.; Wang, Y.; Wei, J.; Han, W. Multi-Antigen-Targeted Chimeric Antigen Receptor T Cells for Cancer Therapy. J Hematol Oncol 2019, 12(1), 128. [Google Scholar] [CrossRef] [PubMed]
- Breman, E.; Demoulin, B.; Agaugué, S.; Mauën, S.; Michaux, A.; Springuel, L.; Houssa, J.; Huberty, F.; Jacques-Hespel, C.; Marchand, C.; Marijsse, J.; Nguyen, T.; Ramelot, N.; Violle, B.; Daro, D.; De Waele, P.; Gilham, D. E.; Steenwinckel, V. Overcoming Target Driven Fratricide for T Cell Therapy. Front. Immunol. 2018, 9, 2940. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Zhao, H.; Han, S.; Kong, D.; Zhang, Q.; Zhang, M.; Chen, Y.; Zhang, M.; Hu, Y.; Huang, H. Chimeric Antigen Receptor-T Cell Therapy for T Cell-Derived Hematological Malignancies. Exp Hematol Oncol 2024, 13(1), 117. [Google Scholar] [CrossRef]
- Jiang, J.; Chen, J.; Liao, C.; Duan, Y.; Wang, Y.; Shang, K.; Huang, Y.; Tang, Y.; Gao, X.; Gu, Y.; Sun, J. Inserting EF1α-Driven CD7-Specific CAR at CD7 Locus Reduces Fratricide and Enhances Tumor Rejection. Leukemia 2023, 37(8), 1660–1670. [Google Scholar] [CrossRef]
- Safarzadeh Kozani, P.; Safarzadeh Kozani, P.; Rahbarizadeh, F. CAR T Cell Therapy in T-Cell Malignancies: Is Success a Low-Hanging Fruit? Stem Cell Res Ther 2021, 12(1), 527. [Google Scholar] [CrossRef]
- Angelos, M. G.; Patel, R. P.; Ruella, M.; Barta, S. K. Progress and Pitfalls of Chimeric Antigen Receptor T Cell Immunotherapy against T Cell Malignancies. Transplant Cell Ther 2024, 30(2), 171–186. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Niu, S.; Li, Y.-R.; Yang, L. Innovative Gene Engineering Strategies to Address Tumor Antigen Escape in Cell Therapy. J Transl Med 2025, 23(1), 1227. [Google Scholar] [CrossRef]
- Abou-El-Enein, M.; Elsallab, M.; Feldman, S. A.; Fesnak, A. D.; Heslop, H. E.; Marks, P.; Till, B. G.; Bauer, G.; Savoldo, B. Scalable Manufacturing of CAR T Cells for Cancer Immunotherapy. Blood Cancer Discov 2021, 2(5), 408–422. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Zeng, Y.; Song, Z.; Liu, Y.; Ou, K.; Wu, Y.; Huang, M.; Li, Y.; Tu, S. Genome-Edited Allogeneic CAR T Cells: The next Generation of Cancer Immunotherapies. J Hematol Oncol 2025, 18(1), 90. [Google Scholar] [CrossRef]
- Mukhametshin, S. A.; Gilyazova, E. M.; Davletshin, D. R.; Ganeeva, I. A.; Zmievskaya, E. A.; Chasov, V. V.; Petukhov, A. V.; Valiullina, A. Kh.; Spada, S.; Bulatov, E. R. Allogeneic NKG2D CAR T Cell Therapy: A Promising Approach for Treating Solid Tumors. Biomedicines 2025, 13(9), 2314. [Google Scholar] [CrossRef]
- Aparicio, C.; Acebal, C.; González-Vallinas, M. Current Approaches to Develop “off-the-Shelf” Chimeric Antigen Receptor (CAR)-T Cells for Cancer Treatment: A Systematic Review. Exp Hematol Oncol 2023, 12(1), 73. [Google Scholar] [CrossRef]
- Graham, C.; Jozwik, A.; Pepper, A.; Benjamin, R. Allogeneic CAR T Cells: More than Ease of Access? Cells 2018, 7(10), 155. [Google Scholar] [CrossRef]
- Evtimov, V. J.; Hammett, M. V.; Pupovac, A.; Nguyen, N.-Y. N.; Shu, R.; Van Der Weyden, C.; Twigger, R.; Nisbet, I. T.; Trounson, A. O.; Boyd, R. L.; Prince, H. M. Targeting TAG-72 in Cutaneous T Cell Lymphoma. Heliyon 2024, 10(17), e36298. [Google Scholar] [CrossRef]
- Kalay Yildizhan, I.; Sanli, H.; Akay, B. N.; Sürgün, E.; Heper, A. CD8+ Cytotoxic Mycosis Fungoides: A Retrospective Analysis of Clinical Features and Follow-up Results of 29 Patients. Int J Dermatology 2020, 59(1), 127–133. [Google Scholar] [CrossRef]
- Tao, Z.; Chyra, Z.; Kotulová, J.; Celichowski, P.; Mihályová, J.; Charvátová, S.; Hájek, R. Impact of T Cell Characteristics on CAR T Cell Therapy in Hematological Malignancies. Blood Cancer J. 2024, 14(1), 213. [Google Scholar] [CrossRef]
- Lee, S. Y.; Lee, D. H.; Sun, W.; Cervantes-Contreras, F.; Basom, R. S.; Wu, F.; Liu, S.; Rai, R.; Mirzaei, H. R.; O’Steen, S.; Green, D. J.; Shadman, M.; Till, B. G. CD8+ Chimeric Antigen Receptor T Cells Manufactured in Absence of CD4+ Cells Exhibit Hypofunctional Phenotype. J Immunother Cancer 2023, 11(11), e007803. [Google Scholar] [CrossRef] [PubMed]
- Janelle, V.; Delisle, J.-S. T-Cell Dysfunction as a Limitation of Adoptive Immunotherapy: Current Concepts and Mitigation Strategies. Cancers 2021, 13(4), 598. [Google Scholar] [CrossRef] [PubMed]
- Volkov, D. V.; Stepanova, V. M.; Yaroshevich, I. A.; Gabibov, A. G.; Rubtsov, Y. P. The Impact of the Intracellular Domains of Chimeric Antigenic Receptors on the Properties of CAR T-Cells. Acta Naturae 2025, 17(3), 4–17. [Google Scholar] [CrossRef]
- Malik, A. M.; Tupchong, S.; Huang, S.; Are, A.; Hsu, S.; Motaparthi, K. An Updated Review of Pemphigus Diseases. Medicina 2021, 57(10), 1080. [Google Scholar] [CrossRef]
- Morais, H. G. D. F.; Costa, C. S. D. O.; Medeiros, M. R. D. S.; Carlan, L. M.; Gonzaga, A. K. G.; Freitas, R. D. A.; Da Silveira, É J. D.; De Oliveira, P. T. Pemphigus Vulgaris and Mucous Membrane Pemphigoid: A Systematic Review of Clinical Manifestations, Diagnosis, and Treatment. Journal of Stomatology, Oral and Maxillofacial Surgery 2024, 125(5), 101960. [Google Scholar] [CrossRef]
- Morales-Hernández, O.; Méndez-Flores, S. [CAR T cells in Dermatology: Mechanisms of action and applications in autoimmune diseases]. Rev Med Inst Mex Seguro Soc 2025, 63(2), e6626. [Google Scholar] [CrossRef] [PubMed]
- Abulikemu, K.; Hu, F.; Liang, J.; Kang, X. Targeting Therapy in Pemphigus: Where Are We Now and Where Are We Going? Heliyon 2023, 9(6), e16679. [Google Scholar] [CrossRef] [PubMed]
- Ellebrecht, C. T.; Bhoj, V. G.; Nace, A.; Choi, E. J.; Mao, X.; Cho, M. J.; Di Zenzo, G.; Lanzavecchia, A.; Seykora, J. T.; Cotsarelis, G.; Milone, M. C.; Payne, A. S. Reengineering Chimeric Antigen Receptor T Cells for Targeted Therapy of Autoimmune Disease. Science 2016, 353(6295), 179–184. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lundgren, D. K.; Mao, X.; Manfredo-Vieira, S.; Nunez-Cruz, S.; Williams, E. F.; Assenmacher, C.-A.; Radaelli, E.; Oh, S.; Wang, B.; Ellebrecht, C. T.; Fraietta, J. A.; Milone, M. C.; Payne, A. S. Antigen-Specific B Cell Depletion for Precision Therapy of Mucosal Pemphigus Vulgaris. J Clin Invest 2020, 130(12), 6317–6324. [Google Scholar] [CrossRef]
- Abeles, I.; Palma, C.; Meednu, N.; Payne, A. S.; Looney, R. J.; Anolik, J. H. B Cell–Directed Therapy in Autoimmunity. Annual Review of Immunology 2024, 42(1), 103–126. [Google Scholar] [CrossRef]
- Ramos, C. A.; Rouce, R.; Robertson, C. S.; Reyna, A.; Narala, N.; Vyas, G.; Mehta, B.; Zhang, H.; Dakhova, O.; Carrum, G.; Kamble, R. T.; Gee, A. P.; Mei, Z.; Wu, M.-F.; Liu, H.; Grilley, B.; Rooney, C. M.; Heslop, H. E.; Brenner, M. K.; Savoldo, B.; Dotti, G. In Vivo Fate and Activity of Second- versus Third-Generation CD19-Specific CAR T Cells in B Cell Non-Hodgkin’s Lymphomas. Molecular Therapy 2018, 26(12), 2727–2737. [Google Scholar] [CrossRef]
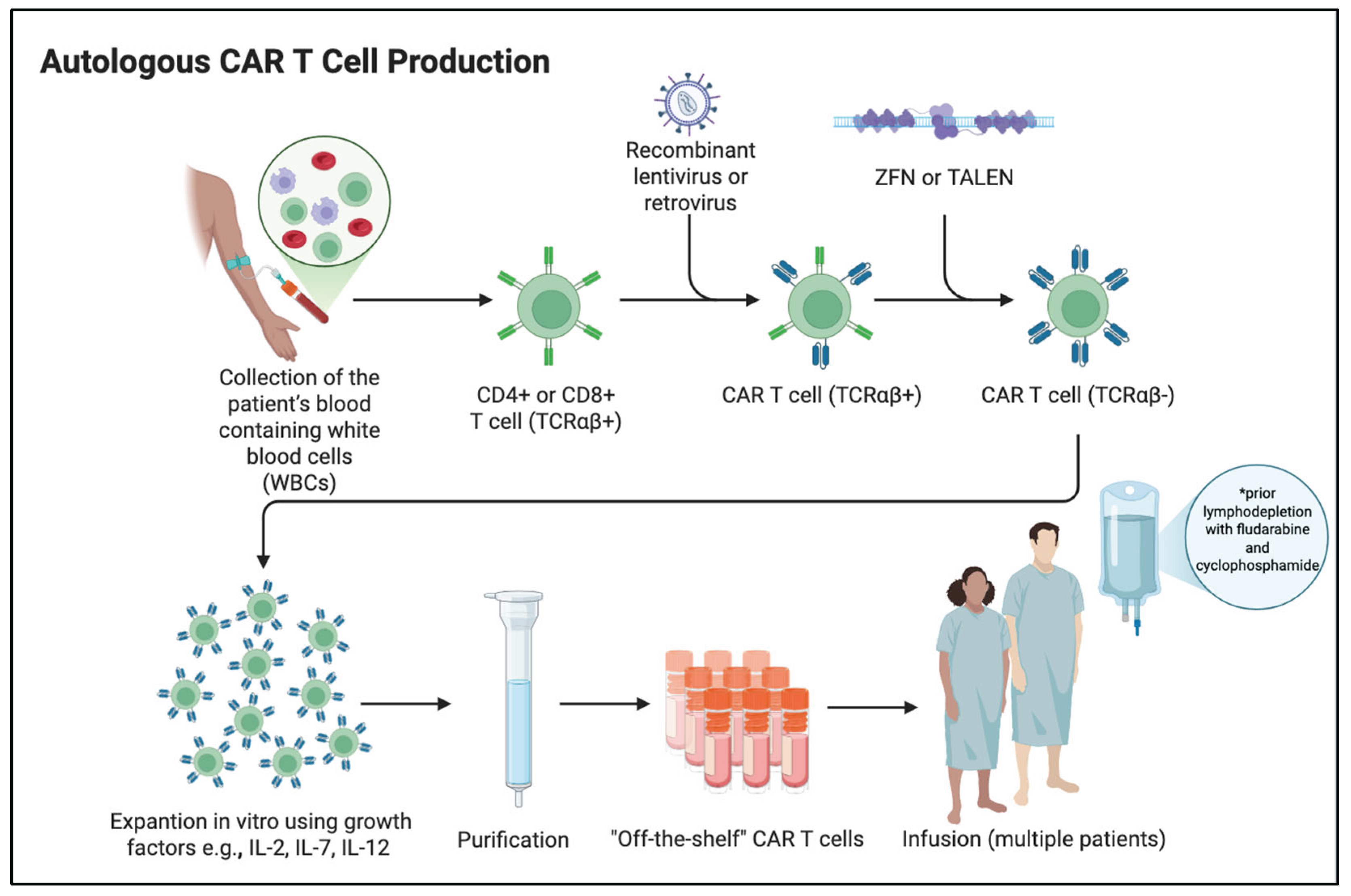
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




