Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
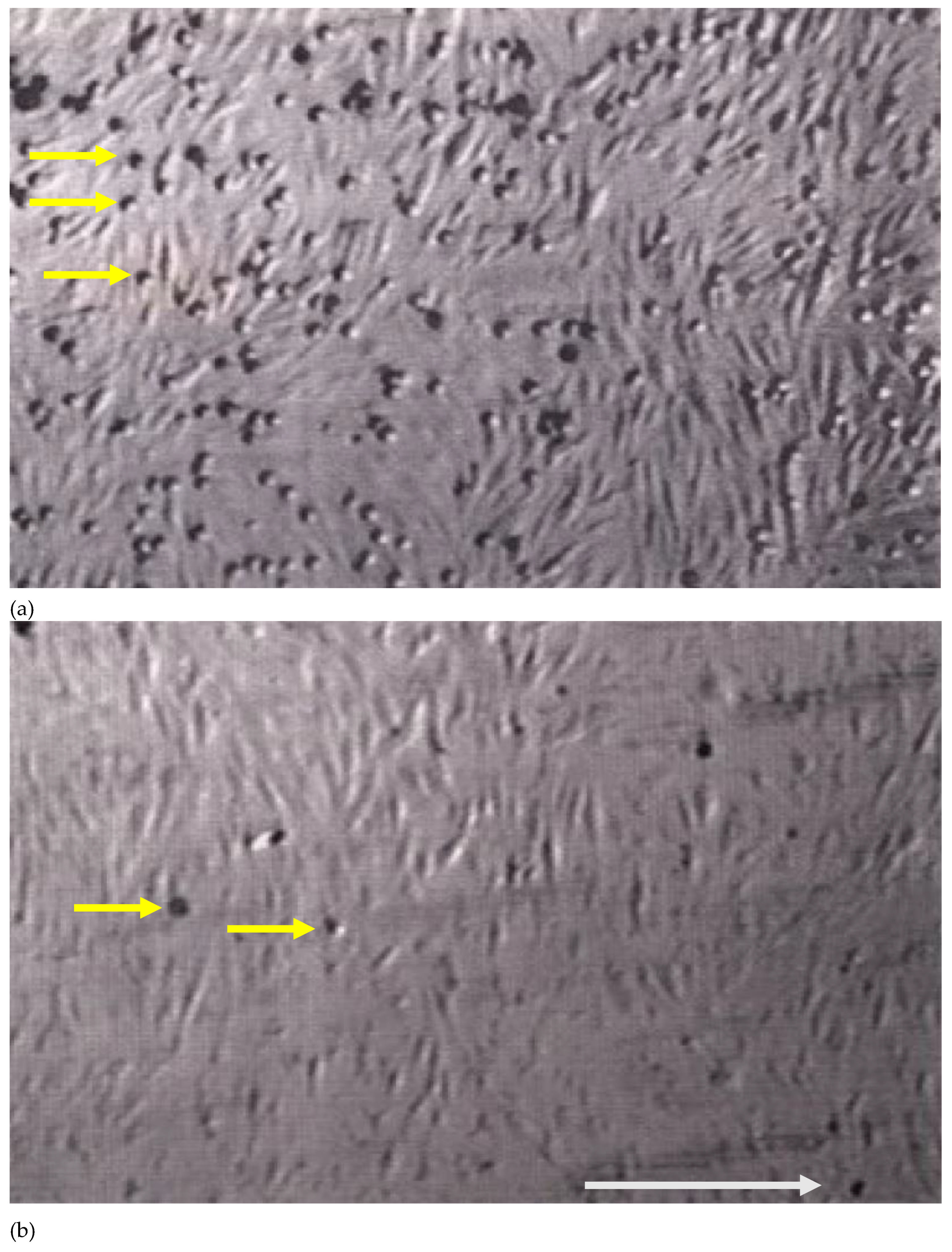
2.1. Rolling and Arrest of Metastatic Tumor Cells Under Flow Conditions on Activated Endothelial Monolayers
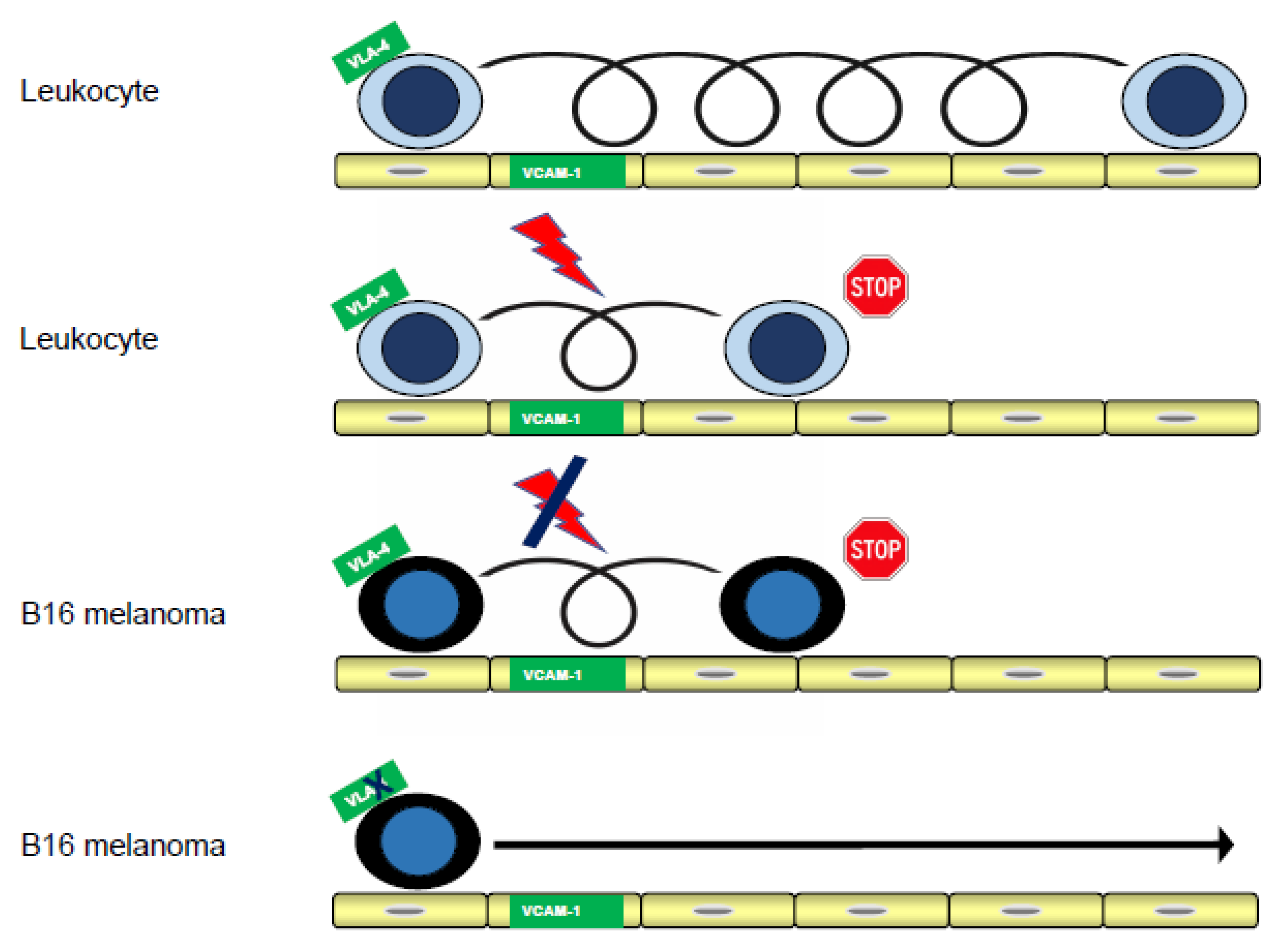
2.2. Blocking VLA-4 or VCAM-1 Abolishes Rolling and Arrest of B16 Melanoma Cells
2.3. Immobilized VCAM-1 Supports Rolling Sufficiency
2.4. Further Controls: No Effect of Chemokine, PTX, or Mg²⁺
2.5. CD44-HA Axis
3. Discussion
4. Materials and Methods
4.1. Cells
4.2. Antibodies and Reagents
4.3. Purified Proteins
4.4. Endothelial Monolayer Activation
4.5. Cell Preparation
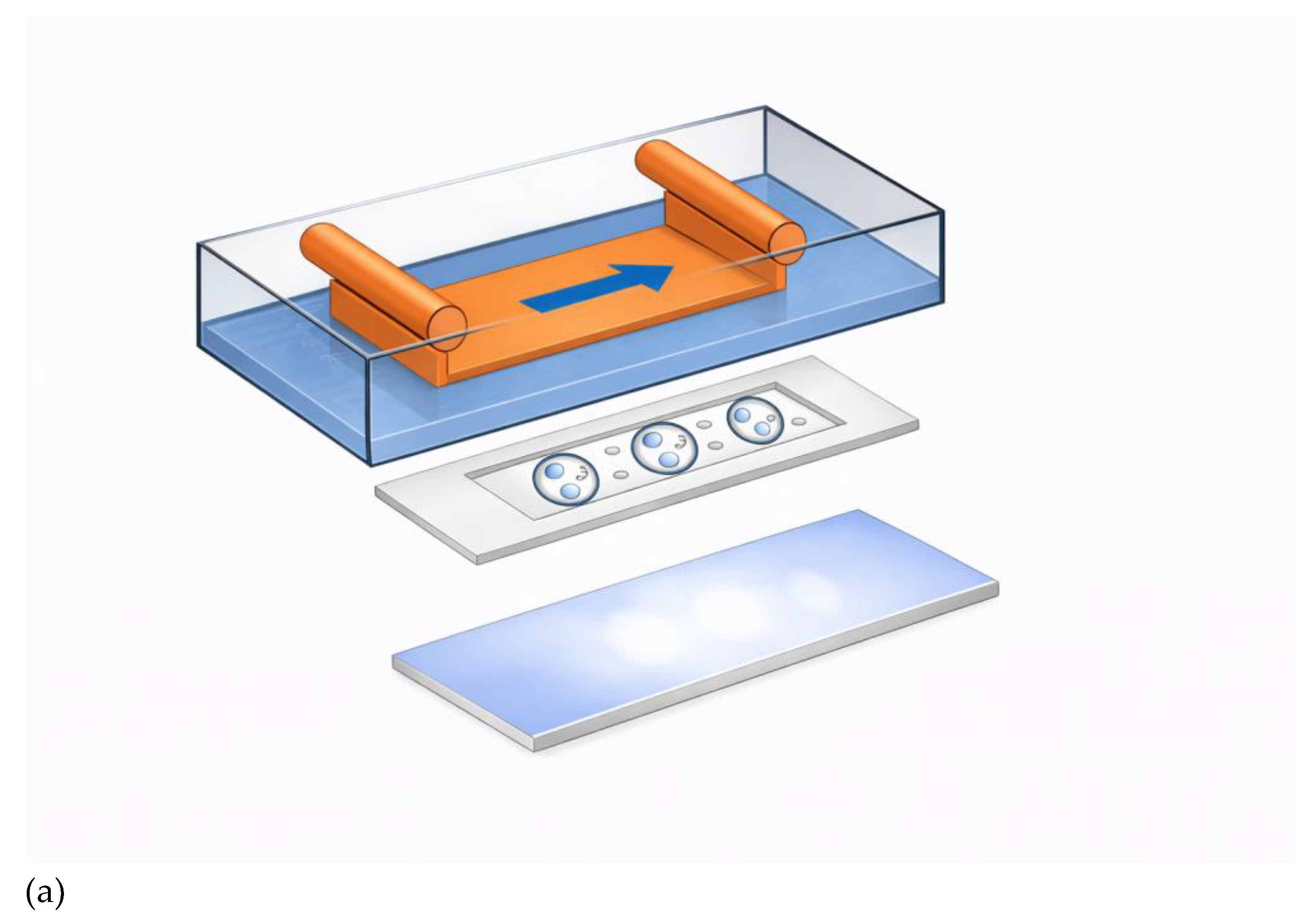
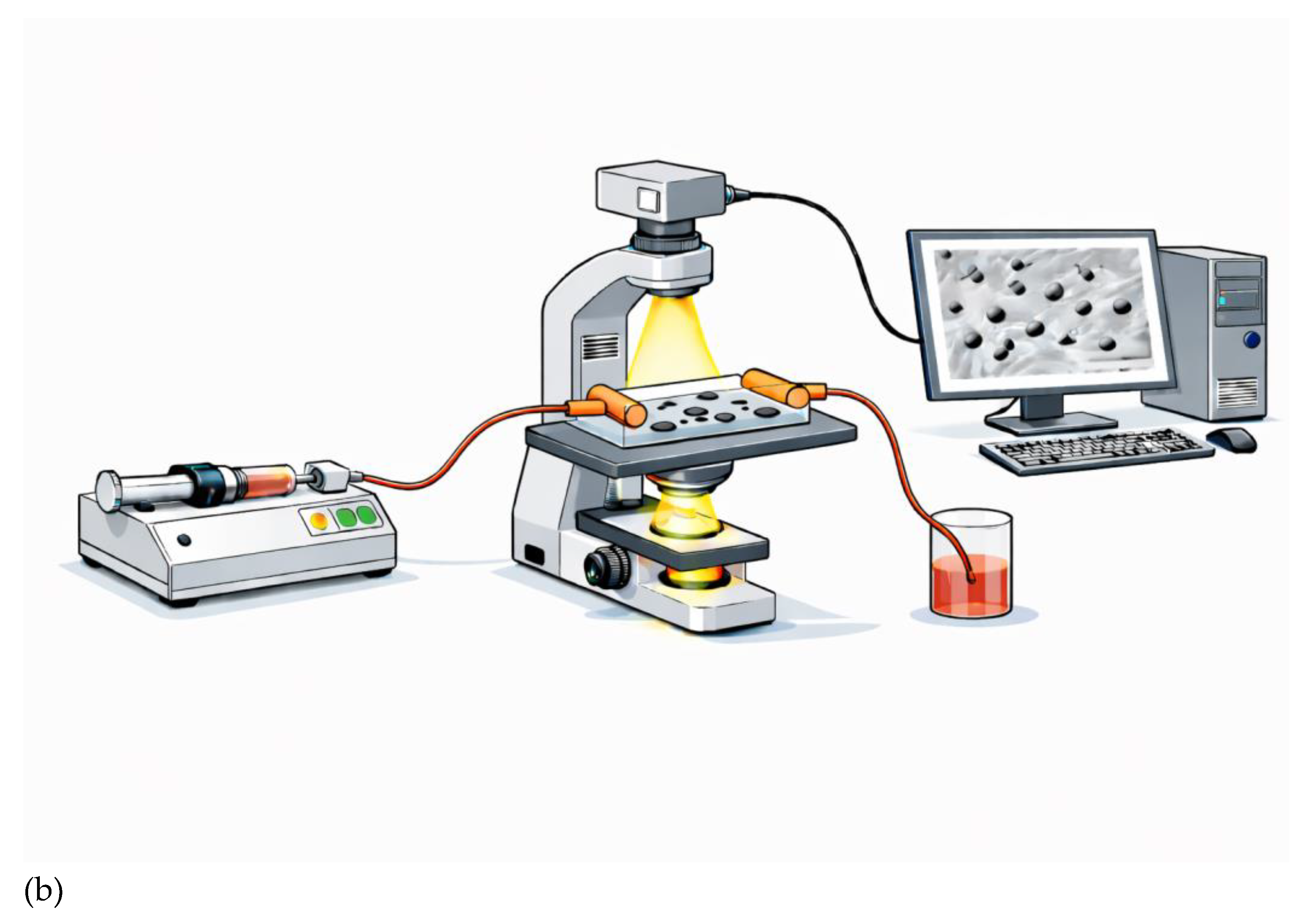
4.6. Flow Chamber Assay
4.7. Quantification and Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFM | Atomic force microscopy |
| CD44 | Cluster of differentiation 44, hyaluronan receptor |
| CXCR4 | C-X-C chemokine receptor type 4 |
| CXCL12 | A chemokine, also known as stromal cell-derived factor 1, SDF-1 |
| DMEM | Dulbecco’s modified Eagle’s medium |
| EDTA | Ethylenediaminetetraacetic acid |
| FCS | Fetal calf serum |
| Gai | Inhibitory G protein alpha subunit |
| HA | Hyaluronan |
| HUVEC | Human umbilical vein endothelial cells |
| ICAM-1 | Intercellular cell adhesion molecule 1 |
| IL-1b | Interleukin-1 beta |
| LPS | Lipopolysaccharide |
| MAdCAM-1 | Mucosal addressin cell adhesion molecule 1 |
| MAb | Monoclonal antibody |
| PTX | Pertussis toxin |
| TNF-α | Tumor necrosis factor alpha |
| VCAM-1 | Vascular cell adhesion molecule 1 |
| VLA-4 | Very late antigen, α4β1 integrin |
References
- Liotta, L.A.; Steeg, P.S.; Stetler-Stevenson, W.G. Cancer metastasis and angiogenesis: an imbalance of positive and negative regulation. Cell 1991, 64, 327–336. [Google Scholar] [CrossRef]
- Berlin, C.; Berg, E.L.; Briskin, M.J.; Andrew, D.P.; Kilshaw, P.J.; Holzmann, B.; Weissman, I.L.; Hamann, A.; Butcher, E.C. Alpha 4 beta 7 integrin mediates lymphocyte binding to the mucosal vascular addressin MAdCAM-1. Cell 1993, 74, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Springer, T.A. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell 1994, 76, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Ley, K.; Laudanna, C.; Cybulsky, M.I.; Nourshargh, S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 2007, 7, 678–689. [Google Scholar] [CrossRef]
- DeGrendele, H.C.; Estess, P.; Siegelman, M.H. Requirement for CD44 in activated T cell extravasation into an inflammatory site. Science 1997, 278, 672–675. [Google Scholar] [CrossRef]
- Gunthert, U.; Hofmann, M.; Rudy, W.; Reber, S.; Zoller, M.; Haussmann, I.; Matzku, S.; Wenzel, A.; Ponta, H.; Herrlich, P. A new variant of glycoprotein CD44 confers metastatic potential to rat carcinoma cells. Cell 1991, 65, 13–24. [Google Scholar] [CrossRef]
- Eibl, R.H.; Pietsch, T.; Moll, J.; Skroch-Angel, P.; Heider, K.H.; von Ammon, K.; Wiestler, O.D.; Ponta, H.; Kleihues, P.; Herrlich, P. Expression of variant CD44 epitopes in human astrocytic brain tumors. Journal of Neuro-Oncology 1995, 26, 165–170. [Google Scholar] [CrossRef]
- Eibl, R.H.; Schneemann, M. Circulating Tumor Cells in Glioblastoma. Cancers 2025, 18. [Google Scholar] [CrossRef]
- Eibl, R.H.; Schneemann, M. Medulloblastoma: From TP53 Mutations to Molecular Classification and Liquid Biopsy. Biology 2023, 12, 267. [Google Scholar] [CrossRef]
- Campbell, J.J.; Hedrick, J.; Zlotnik, A.; Siani, M.A.; Thompson, D.A.; Butcher, E.C. Chemokines and the arrest of lymphocytes rolling under flow conditions. Science 1998, 279, 381–384. [Google Scholar] [CrossRef]
- Alon, R.; Kassner, P.D.; Carr, M.W.; Finger, E.B.; Hemler, M.E.; Springer, T.A. The integrin VLA-4 supports tethering and rolling in flow on VCAM-1. J Cell Biol 1995, 128, 1243–1253. [Google Scholar] [CrossRef] [PubMed]
- Qian, F.; Vaux, D.L.; Weissman, I.L. Expression of the integrin alpha 4 beta 1 on melanoma cells can inhibit the invasive stage of metastasis formation. Cell 1994, 77, 335–347. [Google Scholar] [CrossRef]
- Biancone, L.; Araki, M.; Araki, K.; Vassalli, P.; Stamenkovic, I. Redirection of tumor metastasis by expression of E-selectin in vivo. J Exp Med 1996, 183, 581–587. [Google Scholar] [CrossRef]
- Liang, S.; Dong, C. Integrin VLA-4 enhances sialyl-Lewisx/a-negative melanoma adhesion to and extravasation through the endothelium under low flow conditions. Am J Physiol Cell Physiol 2008, 295, C701–707. [Google Scholar] [CrossRef]
- Okahara, H.; Yagita, H.; Miyake, K.; Okumura, K. Involvement of very late activation antigen 4 (VLA-4) and vascular cell adhesion molecule 1 (VCAM-1) in tumor necrosis factor alpha enhancement of experimental metastasis. Cancer Res 1994, 54, 3233–3236. [Google Scholar]
- Schadendorf, D.; Gawlik, C.; Haney, U.; Ostmeier, H.; Suter, L.; Czarnetzki, B.M. Tumour progression and metastatic behaviour in vivo correlates with integrin expression on melanocytic tumours. J Pathol 1993, 170, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, N.; Puzon-McLaughlin, W.; Irie, A.; Morikawa, Y.; Kakudo, K.; Takada, Y. Induction of experimental bone metastasis in mice by transfection of integrin alpha 4 beta 1 into tumor cells. Am J Pathol 1996, 148, 55–61. [Google Scholar]
- Chambers, A.F.; Groom, A.C.; MacDonald, I.C. Dissemination and growth of cancer cells in metastatic sites. Nat Rev Cancer 2002, 2, 563–572. [Google Scholar] [CrossRef]
- Lawrence, M.B.; Springer, T.A. Leukocytes roll on a selectin at physiologic flow rates: Distinction from and prerequisite for adhesion through integrins. Cell 1991, 65, 859–873. [Google Scholar] [CrossRef]
- Berlin, C.; Bargatze, R.F.; Campbell, J.J.; von Andrian, U.H.; Szabo, M.C.; Hasslen, S.R.; Nelson, R.D.; Berg, E.L.; Erlandsen, S.L.; Butcher, E.C. alpha 4 integrins mediate lymphocyte attachment and rolling under physiologic flow. Cell 1995, 80, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Poste, G.; Doll, J.; Hart, I.R.; Fidler, I.J. In vitro selection of murine B16 melanoma variants with enhanced tissue-invasive properties. Cancer Res 1980, 40, 1636–1644. [Google Scholar]
- Vajkoczy, P.; Laschinger, M.; Engelhardt, B. Alpha4-integrin-VCAM-1 binding mediates G protein-independent capture of encephalitogenic T cell blasts to CNS white matter microvessels. J Clin Invest 2001, 108, 557–565. [Google Scholar] [CrossRef]
- Eibl, R.H.; Benoit, M. Molecular resolution of cell adhesion forces. IEE Proc Nanobiotechnol 2004, 151, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Eibl, R.H.; Moy, V.T. Atomic force microscopy measurements of protein-ligand interactions on living cells. Methods Mol Biol 2005, 305, 439–450. [Google Scholar] [CrossRef]
- Eibl, R.H. Single-Molecule Studies of Integrins by AFM-Based Force Spectroscopy on Living Cells. In Scanning Probe Microscopy in Nanoscience and Nanotechnology 3; Bhushan, B., Ed.; NanoScience and Technology; Springer: Berlin, Heidelberg, 2013; pp. 137–169. [Google Scholar]
- Friederichs, J.; Zeller, Y.; Hafezi-Moghadam, A.; Grone, H.J.; Ley, K.; Altevogt, P. The CD24/P-selectin binding pathway initiates lung arrest of human A125 adenocarcinoma cells. Cancer Res 2000, 60, 6714–6722. [Google Scholar]
- Aigner, S.; Ramos, C.L.; Hafezi-Moghadam, A.; Lawrence, M.B.; Friederichs, J.; Altevogt, P.; Ley, K. CD24 mediates rolling of breast carcinoma cells on P-selectin. FASEB J 1998, 12, 1241–1251. [Google Scholar] [CrossRef]
- Fidler, I.J. The pathogenesis of cancer metastasis: the 'seed and soil' hypothesis revisited. Nat Rev Cancer 2003, 3, 453–458. [Google Scholar] [CrossRef]
- Montesano, R.; Pepper, M.S.; Möhle-Steinlein, U.; Risau, W.; Wagner, E.F.; Orci, L. Increased proteolytic activity is responsible for the aberrant morphogenetic behavior of endothelial cells expressing the middle T oncogene. Cell 1990, 62, 435–445. [Google Scholar] [CrossRef]
- Miyake, K.; Weissman, I.L.; Greenberger, J.S.; Kincade, P.W. Evidence for a role of the integrin VLA-4 in lympho-hemopoiesis. Journal of Experimental Medicine 1991, 173, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Jalkanen, S.; Bargatze, R.F.; de los Toyos, J.; Butcher, E.C. Lymphocyte recognition of high endothelium: antibodies to distinct epitopes of an 85-95-kD glycoprotein antigen differentially inhibit lymphocyte binding to lymph node, mucosal, or synovial endothelial cells. The Journal of Cell Biology 1987, 105, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, T.K.; Warnock, R.A.; Jutila, M.A.; Butcher, E.C.; Lane, C.; Anderson, D.C.; Smith, C.W. Antibodies Against Human Neutrophil LECAM-1 (LAM-l/Leu-8/DREG-56 antigen) and Endothelial Cell ELAM-1 Inhibit a Common CD18-Independent Adhesion Pathway In Vitro. Blood 1991, 78, 805–811. [Google Scholar] [CrossRef]
- Gallatin, W.M.; Weissman, I.L.; Butcher, E.C. A cell-surface molecule involved in organ-specific homing of lymphocytes. Nature 1983, 304, 30–34. [Google Scholar] [CrossRef]
- Streeter, P.R.; Berg, E.L.; Rouse, B.T.N.; Bargatze, R.F.; Butcher, E.C. A tissue-specific endothelial cell molecule involved in lymphocyte homing. Nature 1988, 331, 41–46. [Google Scholar] [CrossRef]
- Ledbetter, J.A.; Herzenberg, L.A. Xenogeneic Monoclonal Antibodies to Mouse Lymphoid Differentiation Antigens*. Immunological Reviews 1979, 47, 63–90. [Google Scholar] [CrossRef] [PubMed]
- Honda, S.; Campbell, J.J.; Andrew, D.P.; Engelhardt, B.; Butcher, B.A.; Warnock, R.A.; Ye, R.D.; Butcher, E.C. Ligand-induced adhesion to activated endothelium and to vascular cell adhesion molecule-1 in lymphocytes transfected with the N-formyl peptide receptor. The Journal of Immunology 1994, 152, 4026–4035. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.B.; McIntire, L.V.; Eskin, S.G. Effect of Flow on Polymorphonuclear Leukocyte/Endothelial Cell Adhesion. Blood 1987, 70, 1284–1290. [Google Scholar] [CrossRef]
- Lawrence, M.B.; Smith, C.W.; Eskin, S.G.; McIntire, L.V. Effect of Venous Shear Stress on CD18-Mediated Neutrophil Adhesion to Cultured Endothelium. Blood 1990, 75, 227–237. [Google Scholar] [CrossRef] [PubMed]
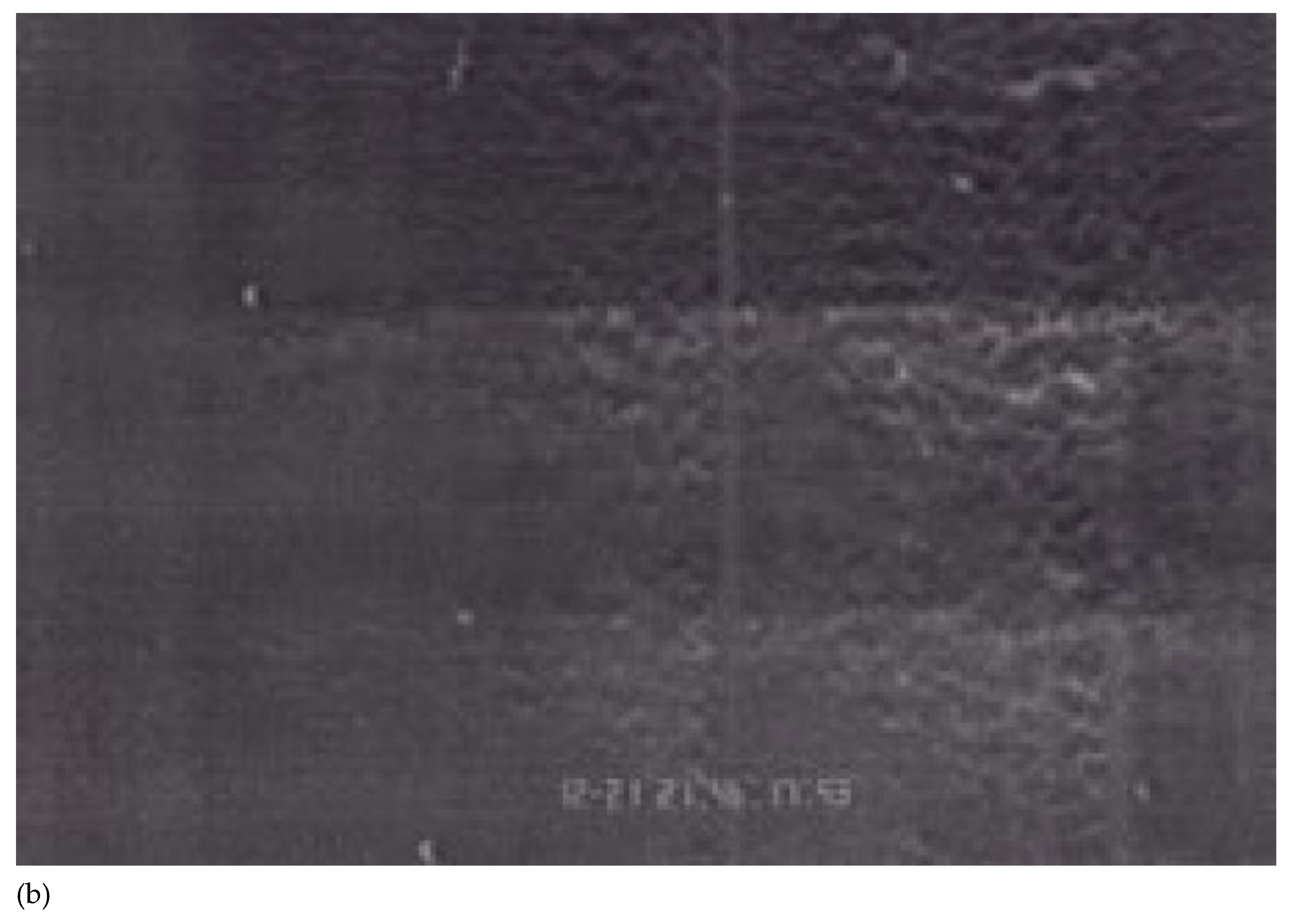
| Cell line | Species | Tumor type | Rolling score | Arrest score | Comment |
|---|---|---|---|---|---|
| B16-BL6 | Mouse | Melanoma | +++ | +++ | Rolling and arrest; leukocyte-like behavior |
| LS174T | Human | Colon carcinoma | + | − | Rolling |
| COLO205 | Human | Colon carcinoma | + | − | Rolling |
| HT-29 | Human | Colon carcinoma | − | − | No rolling or arrest |
| Caco-2 | Human | Colon carcinoma | − | − | No rolling or arrest |
| OVCAR-3 | Human | Ovarian carcinoma | − | − | No rolling or arrest |
| MDA-MB-231 | Human | Breast carcinoma | + | − | Rolling |
| MCF7 | Human | Breast carcinoma | + | − | Rolling |
| LNCaP | Human | Prostate carcinoma | − | − | No rolling or arrest |
| PC-3 | Human | Prostate carcinoma | − | − | No rolling or arrest |
| DU145 | Human | Prostate carcinoma | − | − | No rolling or arrest |
| BW5147 | Mouse | T-lymphoma | +++ | − | CD44-HA dependent rolling; positive control |
| Condition | Established role in leukocyte adhesion (refs) | Expected effect on adhesion | Observed effect on B16 |
|---|---|---|---|
| Mg²⁺ supplementation | Promotes high-affinity conformation of VLA-4 and other integrins in leukocytes[4,11] | Enhanced rolling and/or arrest | No visible increase of rolling/arrest; no detectable change |
| Pertussis toxin (PTX) | Blocks Gαi-dependent chemokine receptor signaling[10] | Reduced adhesion, if VLA-4 was chemokine-activated | Rolling/arrest preserved; no detectable change |
| CXCL12 | Activation of VLA-4, if CXCR4 is expressed, rapid arrest[10] | Activation of VLA-4, but not really expected here (control) | Rolling/arrest preserved; no detectable change |
| CXCL12+PTX | Blocked activation of CXCL12 and any other Gαi-dependent VLA-4 activation pathways[10] | Reduced adhesion, if any chemokines were involved (control, if CXCL12 alone had any effect) | Rolling/arrest preserved; no detectable change |
| 4°C cold room | Suppresses active signaling and energy-dependent processes | Reduced adhesion | No rolling/adhesion |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




