Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
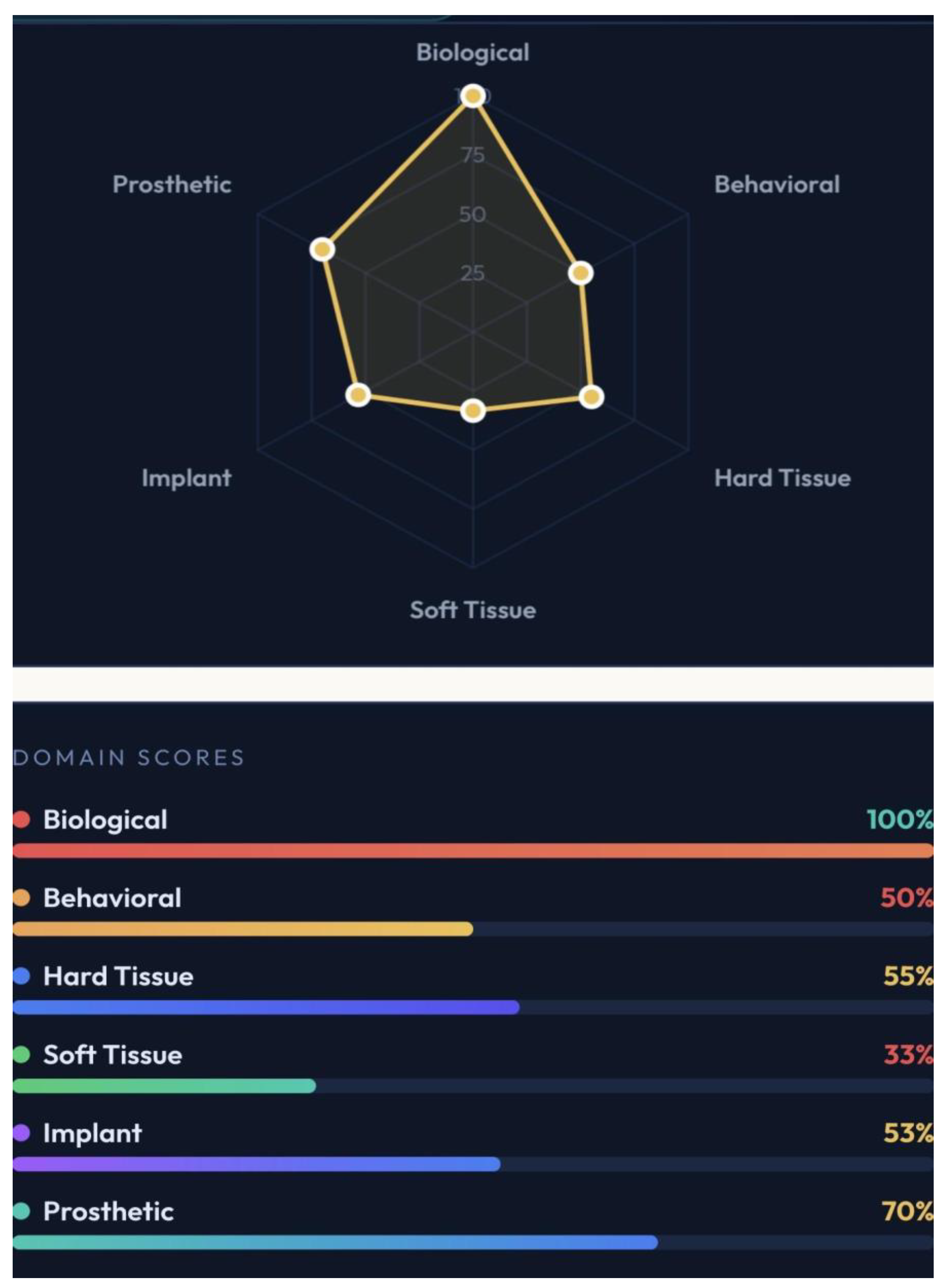
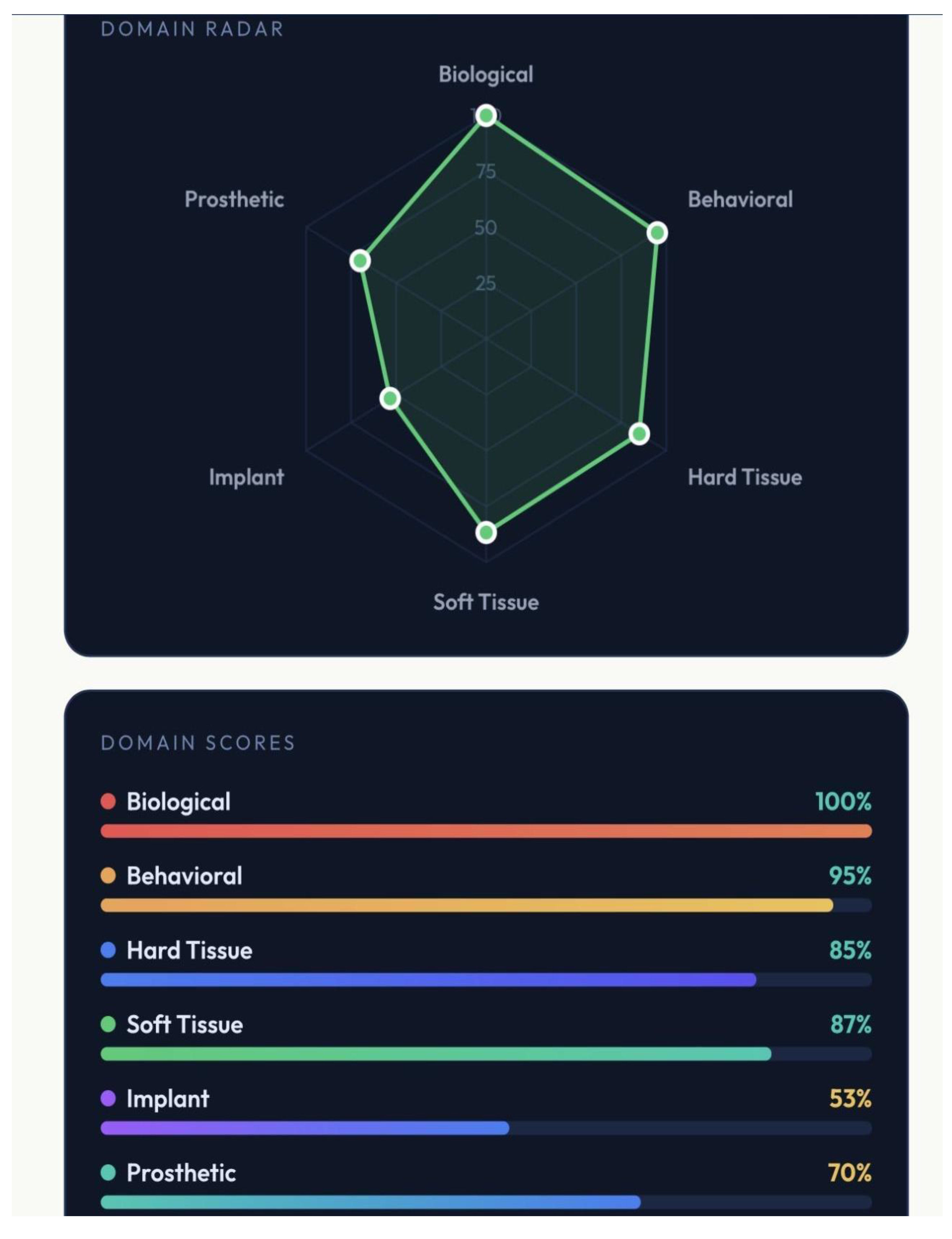
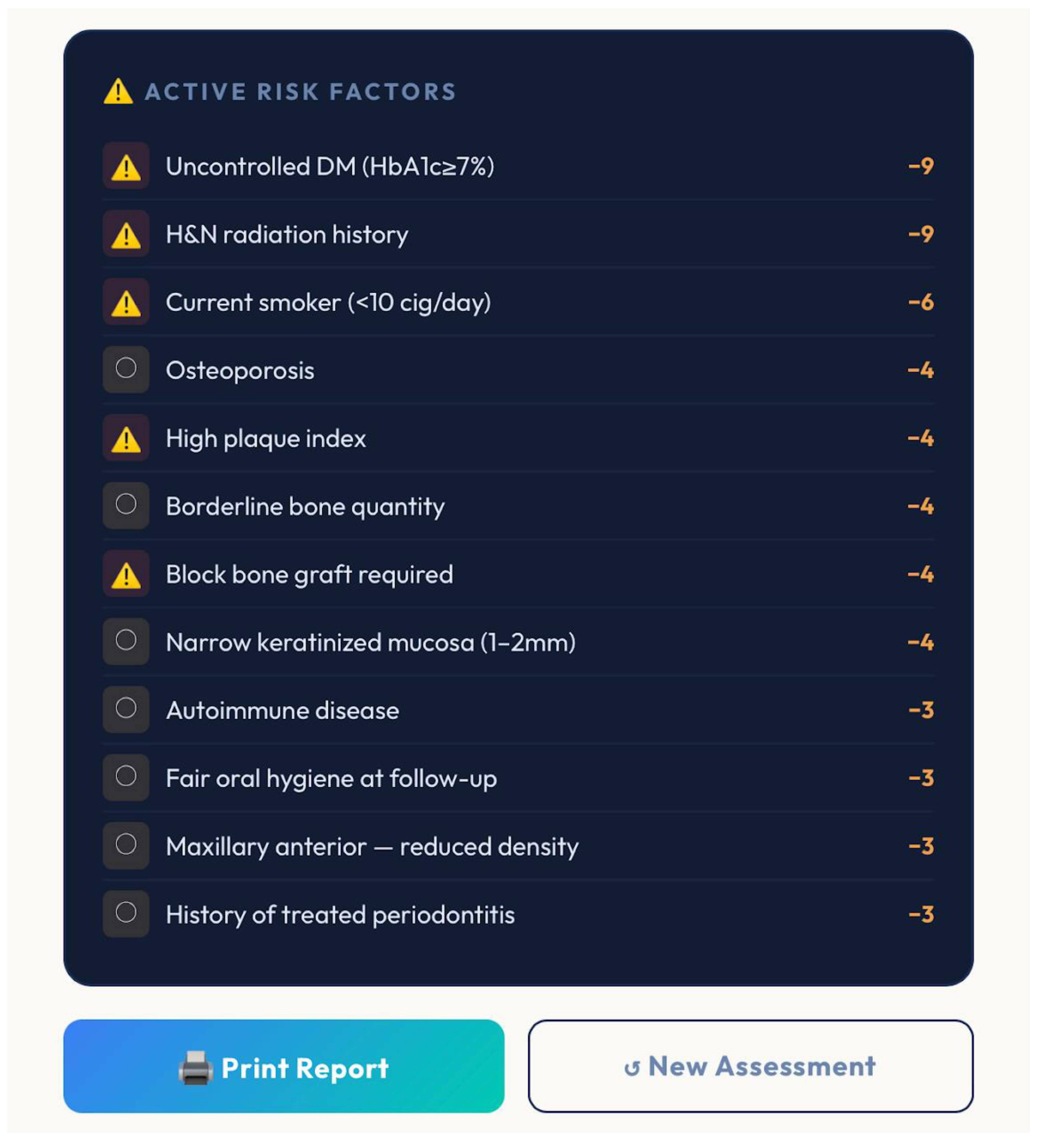
Dental implant therapy demonstrates high long-term survival; however, biological, behavioral, and technical complications remain prevalent. The objective of this study was to introduce GF-Predictability for Dental Implants (GF-PreDImp), the first multidomain predictive tool in the literature, designed to quantify implant success predictability through a structured, evidence-based scoring system. The model integrates six domains: Biological, Behavioral, Hard tissue, Soft tissue, Implant, and Prosthetic, approaching systemic, behavioral, anatomical, surgical, and prosthetic variables into a 100-point composite index. The Biological/Systemic point (20 points) involves diabetes (HbA1c), bisphosphonates, head and neck radiation, cardiovascular disease, osteoporosis, and immunosuppression; the Behavioral/External topic (20 points) approaches post-implant smoking, oral hygiene, plaque/calculus index, brushing performance, alcohol usage, and patient’s compliance; the Hard Tissue (20 points) analyzed bone quality (densities: D1–D4), bone quantity, arch position, guided-bone regeneration (GBR) need, sinus lift, cone beam computed tomography (CBCT) height/width; the Soft Tissue evolution (15 points) observes keratinized mucosa width (KMW), periodontal history, gingival phenotype, bleeding on probing (BoP), and probing depth (PD); the Implant Parameters topic (15 points) assessed tooth position, loading timing, primary stability (ISQ), length/diameter, and surface treatment; and the last point analyzed, Prosthetic/Surgical (10 points), appraisal bruxism characteristic, occlusal contacts, crown-to-implant ratio, cantilever, surgeon experience, and antibiotic protocol. The final GF-PreDImp score could be excellent (≥ 85), good (70 – 84), moderate to guarded (55-69), guarded to high risk (40-54), and poor (<40). Results: Predictors were derived from literature on implant failure, peri-implant disease, and risk assessment. The tool generates dynamic visual outputs, including radar charts and domain-specific scores, enabling real-time clinical interpretation. Each domain can achieve up to 100%, and the average results predict the predictability of dental implant therapy. The final screen of the GF-PreDImp outcome presents a summary of the worst areas to clarify possible risks for clinicians and patients. The graphic and result can be printed for electronic filing and/or shown and given to the patient. The GF-PreDImp system can provide a comprehensive framework for individualized risk stratification and treatment optimization. Its implementation can improve clinical decision-making and enhance long-term implant outcomes. Further clinical assessments must be done to confirm the findings in future studies.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Conceptual Development of GF-PreDImp
2.2. Structure of the GF-PreDImp
2.3. GF-PreDImp Score
3. Results
3.1. Visualization and Functional Interface
3.2. Clinical Interpretation and Application
4. Discussion
4.1. The Shift Toward Holistic Risk Assessment
4.2. Biological and Behavioral Interplay
4.3. The Critical Role of Local and Biomechanical Factors
4.4. Visual Analytics in Shared Decision-Making
4.5. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Morena, D.; Leitão-Almeida, B.; Pereira, M.; Resende, R.; Fernandes, J.C.H.; Fernandes, G.V.O.; Borges, T. Comparative Clinical Behavior of Zirconia versus Titanium Dental Implants: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Clin. Med. 2024, 13, 4488. [Google Scholar] [CrossRef] [PubMed]
- Remísio, M.J.S.; Borges, T.; Castro, F.M.C.; Gehrke, S.A.; Fernandes, J.C.H.; Fernandes, G.V.O. Histological osseointegration level comparing titanium and zirconia dental implants: Meta-analysis of pre-clinical studies. Int. J. Oral Maxillofac. Implants 2023, 38(4), 667–680. [Google Scholar] [CrossRef]
- Heitz-Mayfield, L.J.A. Peri-implant diseases: diagnosis and risk indicators. J. Clin. Periodontol. 2008, 35, 292–304. [Google Scholar] [CrossRef]
- Sánchez-Pérez, A.; Moya-Villaescusa, M.J.; Caffesse, R.G. Tobacco as a risk factor for survival of dental implants. J. Periodontol. 2007, 78(2), 351–359. [Google Scholar] [CrossRef]
- Şahin, T. Evaluation of implants in smoking and non-smoking patients with peri-implant disease risk analysis and esthetic scores: an observational study. BMC Oral Health 2023, 23(1), 925. [Google Scholar] [CrossRef]
- Fan, Y.-Y.; Li, S.; Cai, Y.-J.; Wei, T.; Ye, P. Smoking in relation to early dental implant failure: A systematic review and meta-analysis. J. Dent. 2024, 151, 105396. [Google Scholar] [CrossRef] [PubMed]
- Marcantonio, C.; Nicoli, L.G.; Junior, E.M.; Zandim-Barcelos, D.L. Prevalence and Possible Risk Factors of Peri-implantitis: A Concept Review. J. Contemp. Dent. Pract. 2015, 16(9), 750–757. [Google Scholar] [CrossRef]
- Baqain, Z.H.; Moqbel, W.Y.; Sawair, F.A. Early dental implant failure: risk factors. Br. J. Oral Maxillofac. Surg. 2012, 50(3), 239–243. [Google Scholar] [CrossRef]
- Mustapha, A.; Salame, Z.; Chrcanovic, B. Smoking and dental implants: A systematic review and meta-analysis. Medicina 2021, 58(1), 39. [Google Scholar] [CrossRef]
- Reddy, K.S.; Biswas, S.; Sarangi, S.; Chaurasia, A.; Reddy, M.P.; Jose, A.T.; Kashwani, R. Impact of smoking on dental implant: A review. Bioinformation 2024, 20(1), 1750–1753. [Google Scholar] [CrossRef] [PubMed]
- Meyle, J.; Casado, P.; Fourmousis, I.; Kumar, P.; Quirynen, M.; Salvi, G.E. General genetic and acquired risk factors, and prevalence of peri-implant diseases - Consensus report of working group 1. Int. Dent. J. 2019, 69(1), 3–6. [Google Scholar] [CrossRef]
- Polymeri, A.; Loos, B.G.; Aronovich, S.; Steigmann, L.; Inglehart, M.R. Risk factors, diagnosis, and treatment of peri-implantitis: A cross-cultural comparison of U.S. and European periodontists’ considerations. J. Periodontol. 2021, 93(4), 481–492. [Google Scholar] [CrossRef] [PubMed]
- Taide, P.D.; Faizan, M.; Salunkhe, G.; Sutradhar, W.; Yerra, S.; Gavara, S.G.; Sharma, M. Automated Prediction of Dental Implant Success Using a Mask Region-Based Convolutional Neural Network on Preoperative Cone-Beam Computed Tomography Scans. Cureus 2025, 17(9), e93378. [Google Scholar] [CrossRef]
- Fernandes, G.V.O.; Mosley, G.; Cañizares, A.C.; Fernandes, J.C.H.; Muller, R. Standardizing clinical evaluations of periodontal condition to guide assessments and diagnoses using the Periodontal Assessment Protocol (GF-PAPro). Adv. Clin. Exp. Med. 2025, 34(7). [Google Scholar] [CrossRef]
- Fernandes, G.V.O.; Fernandes, J.C.H. A New Algorithm/Tool Used to Achieve the Periodontal Risk Assessment, Diagnosis, and Prognosis (GF-PeDRA): A Clinical Study with 221 Patients. Dent Med Probl. 2025, 62(5), 801–813. [Google Scholar] [CrossRef]
- Degidi, M.; Daprile, G.; Battelli, F.; Caselli, E.; Cisternino, L.; Greco, A.; Palumbo, D.; Quasso, F.; Rossi, F.; Tavelli, C.; Zaccheroni, Z. The Use of an Artificial Intelligence-Driven Novel Tool for the Evaluation of Dental Implants Primary Stability and Immediate Loading Feasibility: A Multicenter Retrospective Study. J. Clin. Med. 2025, 14(6), 2011. [Google Scholar] [CrossRef]
- Singh, N.; Rajesh, N.; Ramesh, A. Assessment of implant stability with resonance frequency analysis and changes in the thickness of keratinized tissue and crestal bone level using cone-beam computed tomography in two-stage implants: A three-dimensional clinicoradiographic study. J. Indian Soc. Periodontol. 2024, 28(3), 368–375. [Google Scholar] [CrossRef]
- Dawood, E.A.; Abumaylih, R.; Elgarba, B.M.; Fontenele, R.C.; Jacobs, R. Advances in artificial intelligence enhanced robotics for dental implant placement: A scoping review. J. Dent. 2026, 169, 106628. [Google Scholar] [CrossRef]
- Qiang, Z.; Guo, S.; Li, Y.; Li, Z.; Wang, D.; Zhang, K. Analysis of risk indicators for implant failure in patients with chronic periodontitis. BMC Oral Health 2024, 24(1), 1051. [Google Scholar] [CrossRef] [PubMed]
- Sadek, H.S.; Anany, N.M.; El-Anwar, M.I.; Alhotan, A.; Diab, A.-H.; Fayad, B.R.; Heiba, I.M.; Shahin, I.G.; Bourauel, C.; Elshazly, T.M. Biomechanical Performance of Implant-Tooth-Supported Prostheses: A Numerical 3D Finite Element Analysis. Int. Dent. J. 2025, 75(6), 103882. [Google Scholar] [CrossRef] [PubMed]
- Di Fiore, A.; Montagner, M.; Sivolella, S.; Stellini, E.; Yilmaz, B.; Brunello, G. Peri-Implant Bone Loss and Overload: A Systematic Review Focusing on Occlusal Analysis through Digital and Analogic Methods. J. Clin.. Med. 2022, 11(16), 4812. [Google Scholar] [CrossRef]
- Lang, N.P.; Tonetti, M.S. Periodontal risk assessment (PRA) for patients in supportive periodontal therapy (SPT). Oral Health Prev Dent. 2003, 1(1), 7–16. [Google Scholar]
- Heitz-Mayfield, L.J.A.; Heitz, F.; Lang, N.P. Implant Disease Risk Assessment IDRA-a tool for preventing peri-implant disease. Clin Oral Implants Res. 2020, 31(4), 397–403. [Google Scholar] [CrossRef] [PubMed]
- Naujokat, H;.; Kunzendorf, B.; Wiltfang, J. Dental implants and diabetes mellitus - a systematic review. Int. J. Imp. Dent. 2016, 2(1), 5. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.H.; Chan, H.L.; Wang, H.-L. The significance of keratinized mucosa on implant health: a systematic review. J. Periodontol. 2013, 84(12), 1755–1767. [Google Scholar] [CrossRef] [PubMed]
- Chrcanovic, B.R.; Kisch, J.; Albrektsson, T.; Wennerberg, A. Bruxism and dental implant treatment complications: a retrospective comparative study of 98 bruxer patients and a matched group. Clin. Oral Implants Res. 2017, 28(7), e1–e9. [Google Scholar] [CrossRef]
- Revilla-León, M.; Gómez-Polo, M.; Vyas, S.; Barmak, B.A.; Gallucci, G.O.; Att, W.; Krishnamurthy, V.R. Artificial intelligence applications in implant dentistry: A systematic review. J. Prosth. Dent. 2023, 129(2), 293–300. [Google Scholar] [CrossRef]


| SCORE RANGE | VERDICT | CLINICAL MEANING |
|---|---|---|
| ≥ 85 | Excellent Predictability | 5-year survival >95% — Proceed with confidence |
| 70 – 84 | Good Predictability | High success likelihood — Manage identified risks |
| 55 – 69 | Moderate / Guarded | Guarded prognosis — Risk modification required |
| 40 – 54 | Guarded / High Risk | High risk — Address contraindications before proceeding |
| < 40 | Poor Predictability | Multiple major risk factors — Reconsider implant therapy |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




