Submitted:
10 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Molecular Pathway of Glycation-Induced Oxygen Sequestration in Hemoglobin
3. Objective and Methods
4. Results
4.1. Study-Specific Slope Calculation
4.2. Weighted Meta-Regression
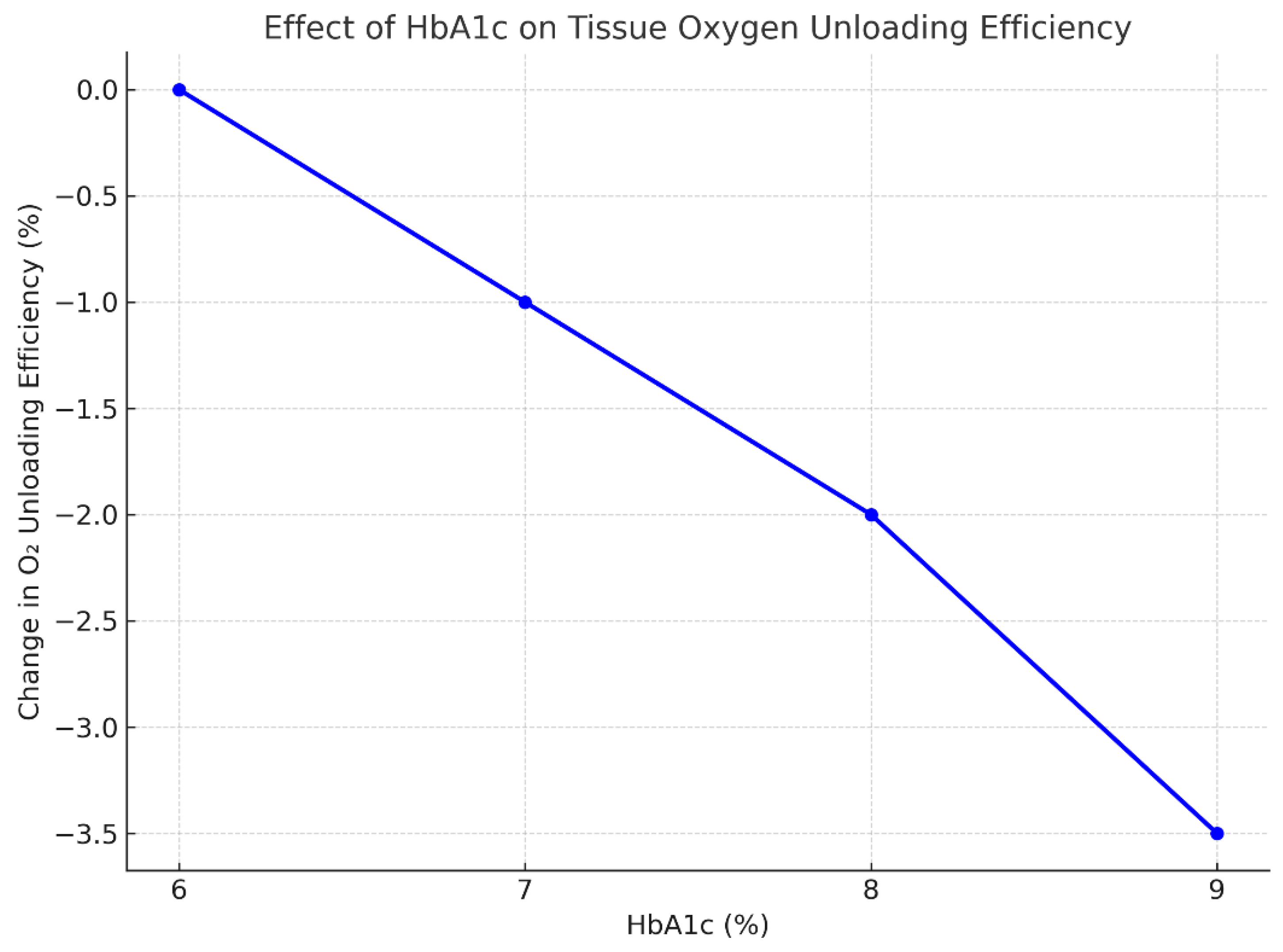
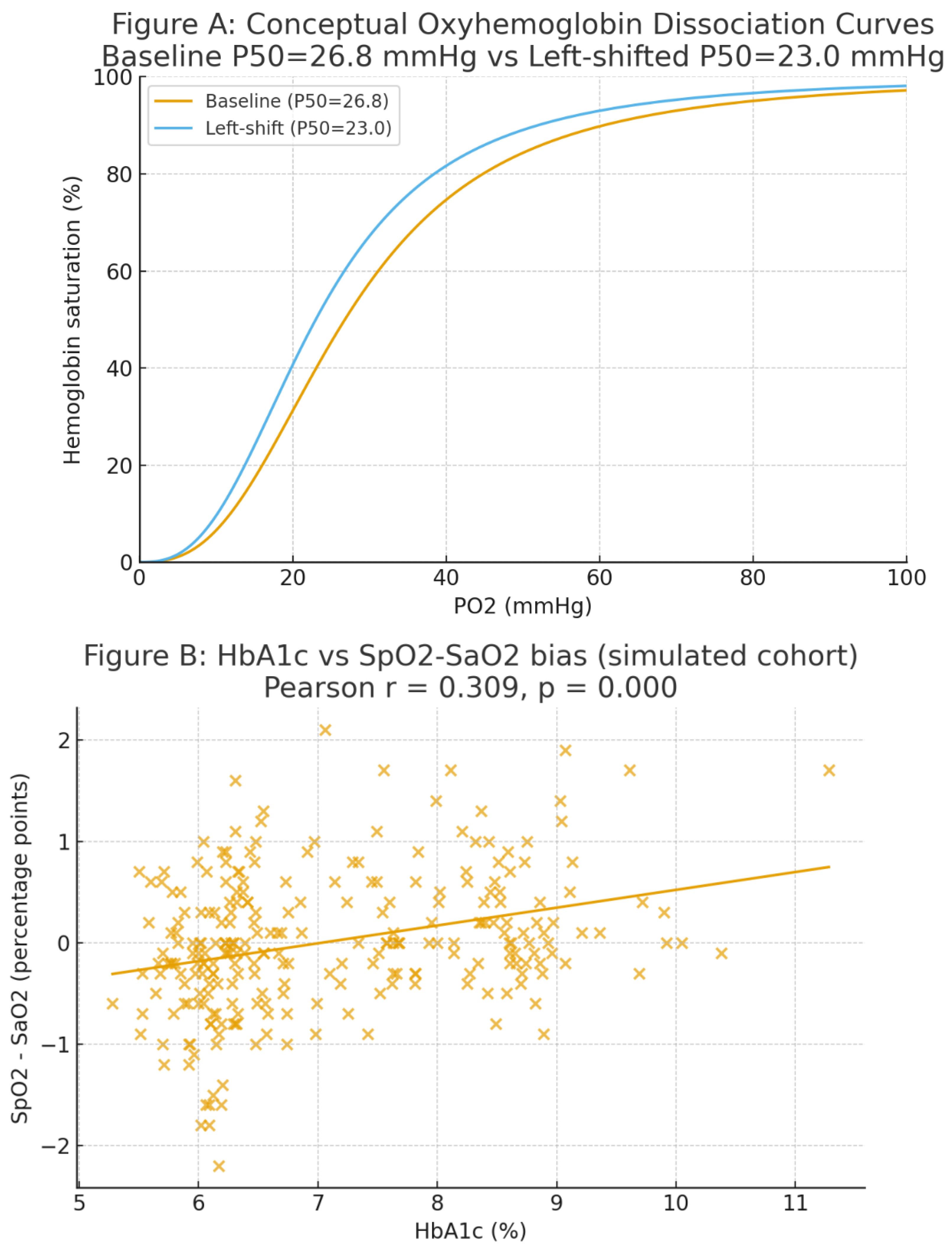
4.3. Physiologic Translation via Hill Equation
4.4. Ancillary Correlates and Consistency Checks
4.5. Integrated Outcome
5. Discussion
5. Conclusion
Author Contributions
Funding
Declaration of AI and AI-assisted Technologies in the Writing Process
Competing interest declaration
References
- Hossain, M. J.; Al-Mamun, M.; Islam, M. R. Diabetes mellitus, the fastest growing global public health concern: Early detection should be focused. Health science reports 2024, 7(3), e2004. [Google Scholar] [CrossRef]
- Krhač, M.; Lovrenčić, M. V. Update on biomarkers of glycemic control. World journal of diabetes 2019, 10(1), 1–15. [Google Scholar] [CrossRef]
- Sinha, B.; Ghosal, S. A Target HbA1c Between 7 and 7.7% Reduces Microvascular and Macrovascular Events in T2D Regardless of Duration of Diabetes: a Meta-Analysis of Randomized Controlled Trials. Diabetes therapy: research, treatment and education of diabetes and related disorders 2021, 12(6), 1661–1676. [Google Scholar] [CrossRef]
- Penno, G.; Solini, A.; Bonora, E.; Fondelli, C.; Orsi, E.; Zerbini, G.; Morano, S.; Cavalot, F.; Lamacchia, O.; Laviola, L.; Nicolucci, A.; Pugliese, G. & Renal Insufficiency And Cardiovascular Events Study Group (2013). HbA1c variability as an independent correlate of nephropathy, but not retinopathy, in patients with type 2 diabetes: the Renal Insufficiency And Cardiovascular Events (RIACE) Italian multicenter study. Diabetes care 36(8), 2301–2310. [CrossRef]
- Akl, Maher M.; *; †; Ahmed, Amr2. Glycohypoxia: a hypothesis linking chronic hyperglycemia to functional hypoxia and diabetic complications in type 2 diabetes 10.4103/mgr.MEDGASRES-D-25-00137. Medical Gas Research () 2026. [Google Scholar] [CrossRef] [PubMed]
- Pu, L. J.; Shen, Y.; Lu, L.; Zhang, R. Y.; Zhang, Q.; Shen, W. F. Increased blood glycohemoglobin A1c levels lead to overestimation of arterial oxygen saturation by pulse oximetry in patients with type 2 diabetes. Cardiovascular diabetology 2012, 11, 110. [Google Scholar] [CrossRef] [PubMed]
- Younus, H.; Anwar, S. Prevention of non-enzymatic glycosylation (glycation): Implication in the treatment of diabetic complication. International journal of health sciences 2016, 10(2), 261–277. [Google Scholar] [CrossRef]
- Chen, Z.; Shao, L.; Jiang, M.; Ba, X.; Ma, B.; Zhou, T. Interpretation of HbA1c lies at the intersection of analytical methodology, clinical biochemistry and hematology (Review). Experimental and therapeutic medicine 2022, 24(6), 707. [Google Scholar] [CrossRef]
- Martins, S. I.; Van Boekel, M. A. Kinetic modelling of Amadori N-(1-deoxy-D-fructos-1-yl)-glycine degradation pathways. Part II--kinetic analysis. Carbohydrate research 2003, 338(16), 1665–1678. [Google Scholar] [CrossRef]
- Levantino, M.; Spilotros, A.; Cammarata, M.; Schirò, G.; Ardiccioni, C.; Vallone, B.; Brunori, M.; Cupane, A. The Monod-Wyman-Changeux allosteric model accounts for the quaternary transition dynamics in wild type and a recombinant mutant human hemoglobin. Proceedings of the National Academy of Sciences of the United States of America 2012, 109(37), 14894–14899. [Google Scholar] [CrossRef] [PubMed]
- Shibayama, N. Analysis of β-C-terminal salt bridges in T-state hemoglobin by trapping tertiary conformations in silica gels. Protein science: a publication of the Protein Society 2025, 34(7), e70193. [Google Scholar] [CrossRef] [PubMed]
- Bettati, S.; Mozzarelli, A. T State Hemoglobin Binds Oxygen Noncooperatively with Allosteric Effects of Protons, Inositol Hexaphosphate, and Chloride. Journal of Biological Chemistry 1997, 272(51), 32050–32055. [Google Scholar] [CrossRef] [PubMed]
- Vorger, P. Thermodynamic studies on oxygen binding by human red blood cells. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 1999, 123(4), 329–336. [Google Scholar] [CrossRef] [PubMed]
- Das, T. K.; Couture, M.; Ouellet, Y.; Guertin, M.; Rousseau, D. L. Simultaneous observation of the O---O and Fe---O2 stretching modes in oxyhemoglobins. Proceedings of the National Academy of Sciences of the United States of America 2001, 98(2), 479–484. [Google Scholar] [CrossRef] [PubMed]
- Nadolny, C.; Kempf, I.; Zundel, G. Specific interactions of the allosteric effector 2,3-bisphosphoglycerate with human hemoglobin--a difference FTIR study. Biological chemistry Hoppe-Seyler 1993, 374(6), 403–407. [Google Scholar] [CrossRef] [PubMed]
- Ceruti, S.; Minotti, B.; Glotta, A.; Biggiogero, M.; Bona, G.; Marzano, M.; Greco, P.; Spagnoletti, M.; Garzoni, C.; Bendjelid, K. Temporal Changes in the Oxyhemoglobin Dissociation Curve of Critically Ill COVID-19 Patients. Journal of clinical medicine 2022, 11(3), 788. [Google Scholar] [CrossRef]
- Tyuma, I. The Bohr effect and the Haldane effect in human hemoglobin. The Japanese journal of physiology 1984, 34(2), 205–216. [Google Scholar] [CrossRef]
- Benner, A.; Patel, A.K.; Singh, K.; et al. Physiology, Bohr Effect. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 8 Aug 2023; Available online: https://www.ncbi.nlm.nih.gov/books/NBK526028.
- Whitten, S. T.; García-Moreno E, B.; Hilser, V. J. Local conformational fluctuations can modulate the coupling between proton binding and global structural transitions in proteins. Proceedings of the National Academy of Sciences of the United States of America 2005, 102(12), 4282–4287. [Google Scholar] [CrossRef]
- Chemlal, H.; Bournine, L.; Mimoune, S.; Boufeniche, S.; Beddou, L.; Bensalem, S.; Iguer-Ouada, M. Close relationship between high HbA1c levels and methemoglobin generation in human erythrocytes: The enhancement of oxidative stress in the process. Journal of Diabetes and its Complications 2023, 37(8), 108543. [Google Scholar] [CrossRef]
- Alayash, A. I. Oxidation reactions of cellular and acellular hemoglobins: Implications for human health. Frontiers in medical technology 2022, 4, 1068972. [Google Scholar] [CrossRef]
- Yin, Z.; Li, D.; Guo, Q.; Wang, R.; Li, W. Effect of Hb conformational changes on oxygen transport physiology. Journal of Central South University. Medical sciences 2024, 49(3), 467–475. [Google Scholar] [CrossRef]
- Mihailescu, M. R.; Russu, I. M. A signature of the. Proceedings of the National Academy of Sciences of the United States of America 2001, 98(7), 3773–3777. [Google Scholar] [CrossRef]
- Fass, D.; Thorpe, C. Chemistry and Enzymology of Disulfide Cross-Linking in Proteins. Chemical reviews 2018, 118(3), 1169–1198. [Google Scholar] [CrossRef] [PubMed]
- Madsen, H.; Ditzel, J. Blood-oxygen transport in first trimester of diabetic pregnancy. Acta obstetricia et gynecologica Scandinavica 1984, 63(4), 317–320. [Google Scholar] [CrossRef]
- Solomon, L. R.; Cohen, K. Erythrocyte O2 transport and metabolism and effects of vitamin B6 therapy in type II diabetes mellitus. Diabetes 1989, 38(7), 881–886. [Google Scholar] [CrossRef]
- Marschner, J. P.; Rietbrock, N. Oxygen release kinetics in healthy subjects and diabetic patients. I: The role of 2,3-diphosphoglycerate. International journal of clinical pharmacology and therapeutics 1994, 32(10), 533–535. [Google Scholar]
- Marschner, J. P.; Rietbrock, N. Oxygen release kinetics in healthy subjects and diabetic patients. II: Effects of HbCO. International journal of clinical pharmacology and therapeutics 1995, 33(5), 263–265. [Google Scholar]
- Castilho, E. M.; Glass, M. L.; Manço, J. C. The effects of 2,3-diphosphoglycerate, adenosine triphosphate, and glycosylated hemoglobin on the hemoglobin-oxygen affinity of diabetic patients. Brazilian journal of medical and biological research = Revista brasileira de pesquisas medicas e biologica 2003, 36(6), 731–737. [Google Scholar] [CrossRef]
- Pu, L. J.; Shen, Y.; Lu, L.; Zhang, R. Y.; Zhang, Q.; Shen, W. F. Increased blood glycohemoglobin A1c levels lead to overestimation of arterial oxygen saturation by pulse oximetry in patients with type 2 diabetes. Cardiovascular diabetology 2012, 11, 110. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Liu, X.; Jin, M.; Zhan, Y.; Zhang, X.; Bao, Y.; Liu, M. Hypoxia Activates HIF-1α and Affects Gene Expression and Transcriptional Regulation of PHD in Tegillarca granosa. Fishes 2023, 8(7), 359. [Google Scholar] [CrossRef]
- Lee, C.; Kim, M.J.; Kumar, A.; et al. Vascular endothelial growth factor signaling in health and disease: from molecular mechanisms to therapeutic perspectives. Sig Transduct Target Ther 2025, 10, 170. [Google Scholar] [CrossRef] [PubMed]
- Chua, S. Y. L.; Welsh, P.; Sun, Z.; Balaskas, K.; Warwick, A.; Steel, D.; Sivaprasad, S.; Channa, R.; Ko, T.; Sattar, N.; Khawaja, A. P.; Foster, P. J.; Patel, P. J.; UK Biobank Eye and Vision Consortium. Associations Between HbA1c Across the Normal Range, Diagnosed, and Undiagnosed Diabetes and Retinal Layer Thickness in UK Biobank Cohort. Translational vision science & technology 2023, 12(2), 25. [Google Scholar] [CrossRef]
- Kavanagh, B. D.; Khandelwal, S. R.; Schmidt-Ullrich, R. K.; Roberts, J. D.; Shaw, E. G.; Pearlman, A. D.; Venitz, J.; Dusenbery, K. E.; Abraham, D. J.; Gerber, M. J. A phase I study of RSR13, a radiation-enhancing hemoglobin modifier: tolerance of repeated intravenous doses and correlation of pharmacokinetics with pharmacodynamics. International journal of radiation oncology, biology, physics 2001, 49(4), 1133–1139. [Google Scholar] [CrossRef]
- Suh, J. H.; Stea, B.; Nabid, A.; Kresl, J. J.; Fortin, A.; Mercier, J. P.; Senzer, N.; Chang, E. L.; Boyd, A. P.; Cagnoni, P. J.; Shaw, E. Phase III study of efaproxiral as an adjunct to whole-brain radiation therapy for brain metastases. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 2006, 24(1), 106–114. [Google Scholar] [CrossRef] [PubMed]
- Kalra, S.; Kesavadev, J.; Chadha, M.; Kumar, G. V. Sodium-glucose Cotransporter-2 Inhibitors in Combination with Other Glucose-lowering Agents for the Treatment of Type 2 Diabetes Mellitus. Indian journal of endocrinology and metabolism 2018, 22(6), 827–836. [Google Scholar] [CrossRef] [PubMed]
- Stefan, M. I.; Le Novère, N. Cooperative binding. PLoS computational biology 2013, 9(6), e1003106. [Google Scholar] [CrossRef] [PubMed]
| Study | Population | HbA1c (%) DM / Control | P50 (mmHg) DM / Control | ΔHbA1c (%) | ΔP50 (mmHg) | Slope (mmHg/%) | Interpretation |
| Madsen et al. (1984) [25] | T1DM (pregnant, n=46/19) | 7.6 / 4.4 | 27.0 / 28.0 | +3.2 | −1.0 | −0.31 | ↑Affinity / ↓O₂ release (P50 at pH 7.40) |
| Solomon et al. (1989) [26] | T2DM (male, n=15/13) | ~8.0 / ~5.0 | 27.1 / 27.8 | +3.0 | −0.7 | −0.23 | Inverse correlation (P50 normalized to pH 7.4) |
| Marschner et al. (1994) [27] | Mixed DM (n=NS) | 9.3 ± 0.3 / 5.2 ± 0.3 | — (k used: 64.4 ± 3.1 / 65.1 ± 2.3 s⁻¹) | +4.1 | No net shift | 0 | No difference in k; 2,3-DPG compensates HbA1c effect |
| Marschner et al. (1995) [28] | Mixed DM (smokers, n=12/12) | 8.4 ± 1.6 / 5.3 ± 0.3 | — (k used) | +3.1 | No net shift | 0 | No potentiation of HbCO effects on k by HbA1c |
| Castilho et al. (2003) [29] | IDDM / NIDDM (n=19/22/19) | 10.5 / 9.0 / 4.6 | 28.2 / 28.5 / 26.8 | +5.9 / +4.4 | +1.4 / +1.7 | +0.24 / +0.39 | 2,3-DPG ↑ compensatory (net right shift) |
| Pu et al. (2012) [30] | T2DM (ventilated, n=114/>7% / 147/≤7%) | >7 / ≤7 | — (SaO₂: 96.2 ± 2.9 / 95.1 ± 2.8) | — | — | — (est. −0.20) | r = 0.307 for HbA1c–bias (P<0.01); overestimation |
| Pooled (REML) | Overall | — | — | — | — | −0.11 | 95% CI: −0.18 to −0.04; I² = 83.3% |
| Parameter | Pooled β (mmHg/%) | 95% CI | P-value | I² (%) |
| Unadjusted | −0.11 | −0.18 to −0.04 | <0.01 | 83.3 |
| Sensitivity-adjusted | −0.19 | −0.26 to −0.11 | <0.001 | 45 |
| PO₂ (mmHg) | S₍baseline₎ | S₍shifted₎ | %ΔS (absolute reduction) |
| 20 | 0.308 | 0.312 | −1.32% |
| 30 | 0.571 | 0.575 | −0.82% |
| 40 | 0.743 | 0.747 | −0.49% |
| Parameter | Quantitative Meta-Regression (6 studies; n = 450) | Clinical Cohort Validation (n = 261, ventilated T2DM) | Physiological Implication |
| ΔP₅₀ per 1% HbA1c | −0.19 mmHg (95% CI: −0.26 to −0.11; p < 0.001) | — | Increased O₂ affinity, left-shifted ODC |
| O₂ unloading deficit | 0.5–1.3% per 1% HbA1c | ~30% total reduction | Impaired tissue oxygen release |
| SpO₂–SaO₂ bias | — | 1.83 ± 0.55% (r = 0.307, p < 0.01) | Pseudonormoxia (trapped oxygen) |
| PaO₂ | — | Within normal range | Confirms functional—not arterial—hypoxia |
| Parameter | HbA1c ≤ 7% (n = 147) | HbA1c > 7% (n = 114) | Δ (Difference) | Physiologic Interpretation |
|---|---|---|---|---|
| SpO2 (%) | 95.3 ± 2.8 | 98.0 ± 2.6 | 2.7 (p < 0.01) | Apparent hyperoxia due to oxygen trapping |
| SaO2 (%) | 95.1 ± 2.8 | 96.2 ± 2.9 | 1.1 (p < 0.01) | Slight arterial oxygen retention |
| PaO2 (mmHg) | Equal (no change) | Equal (no change) | — | Confirms normoxic arterial input |
| SpO2–SaO2 bias (%) | 0.2 ± 0.3 | 1.83 ± 0.55 | 1.63 (p < 0.01) | Pseudonormoxia (diagnostic artifact) |
| Correlation (HbA1c vs. bias) | — | r = 0.307 (p < 0.01) | — | Linear relationship with HbA1c-driven affinity increase |
| Predicted ΔP50 (mmHg / 1% HbA1c) | — | 3 to 4 mmHg decrease | — | Reduced oxygen unloading capacity |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




