Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction

2. Materials and Methods
3. Results and Discussion
3.1. Synthesis of 5-Arylidenerhodanines I-III
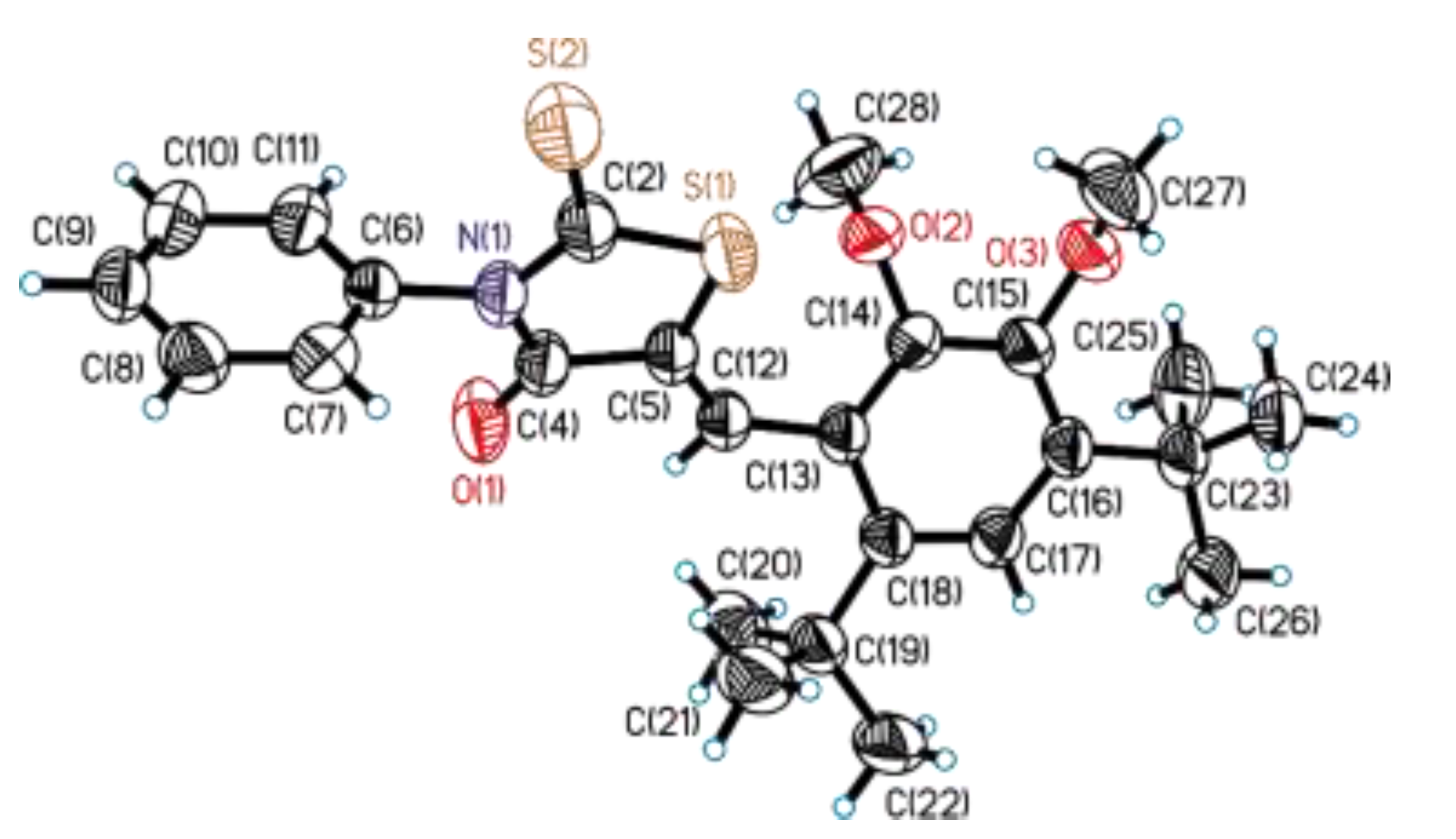
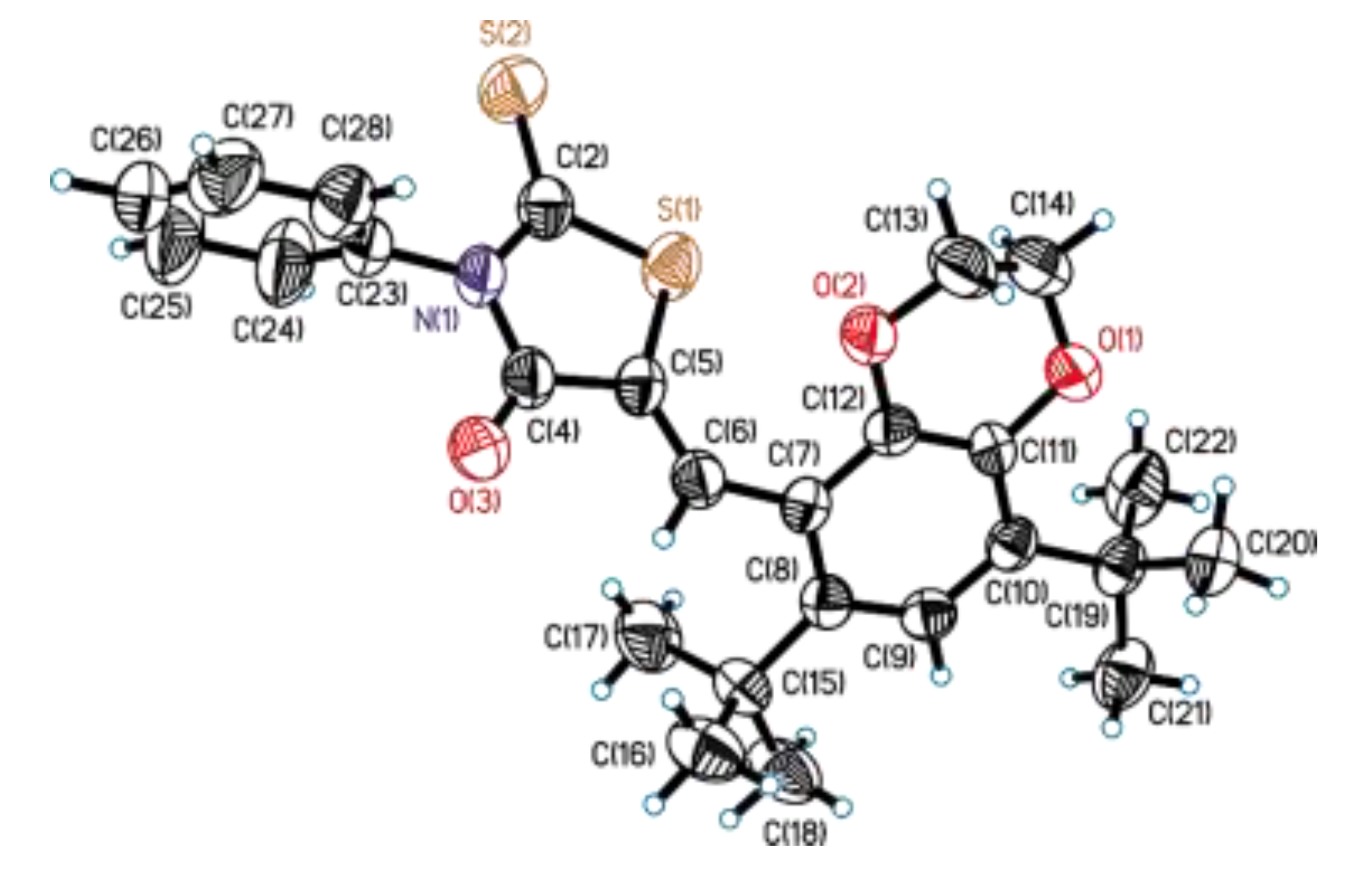
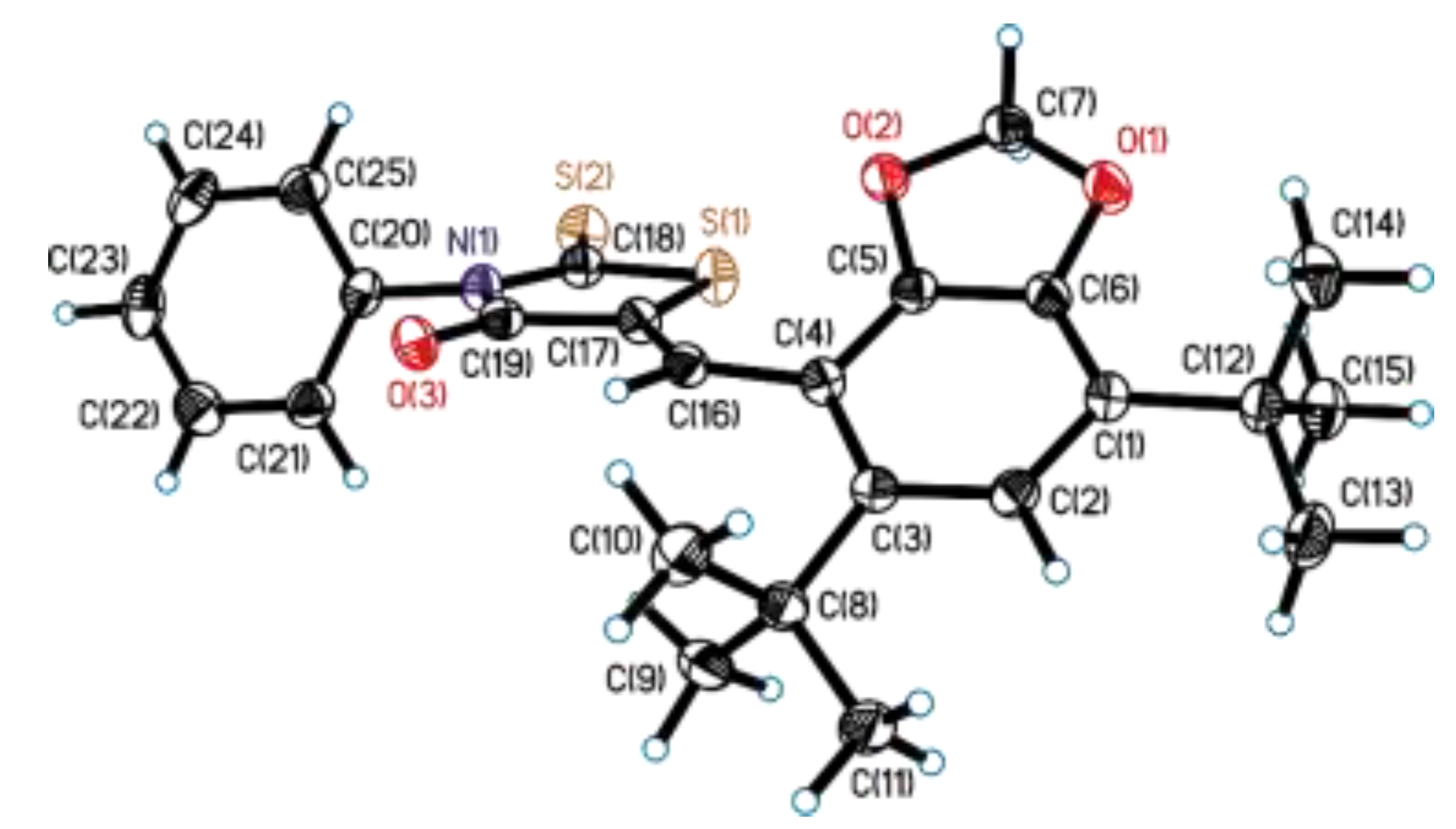
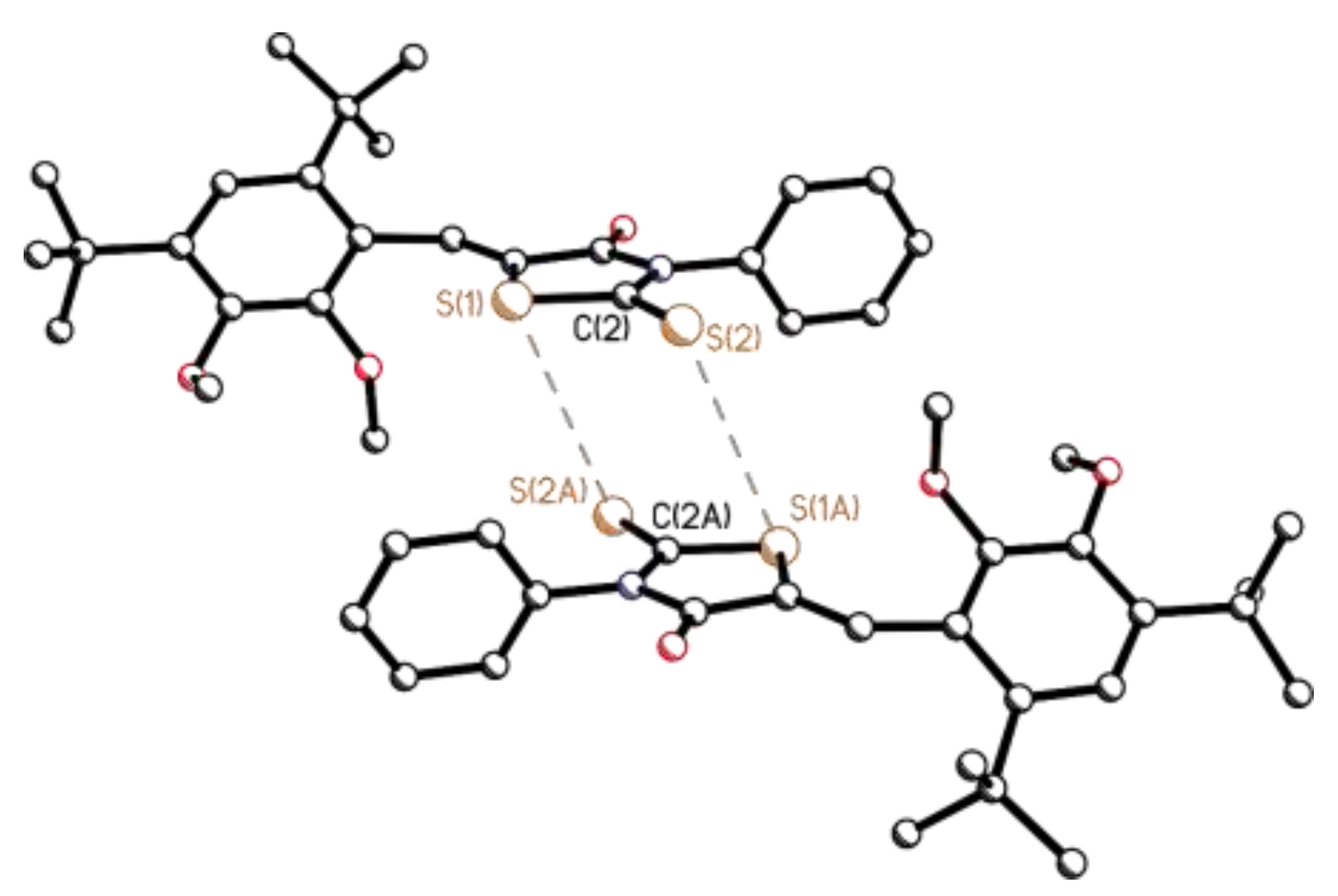
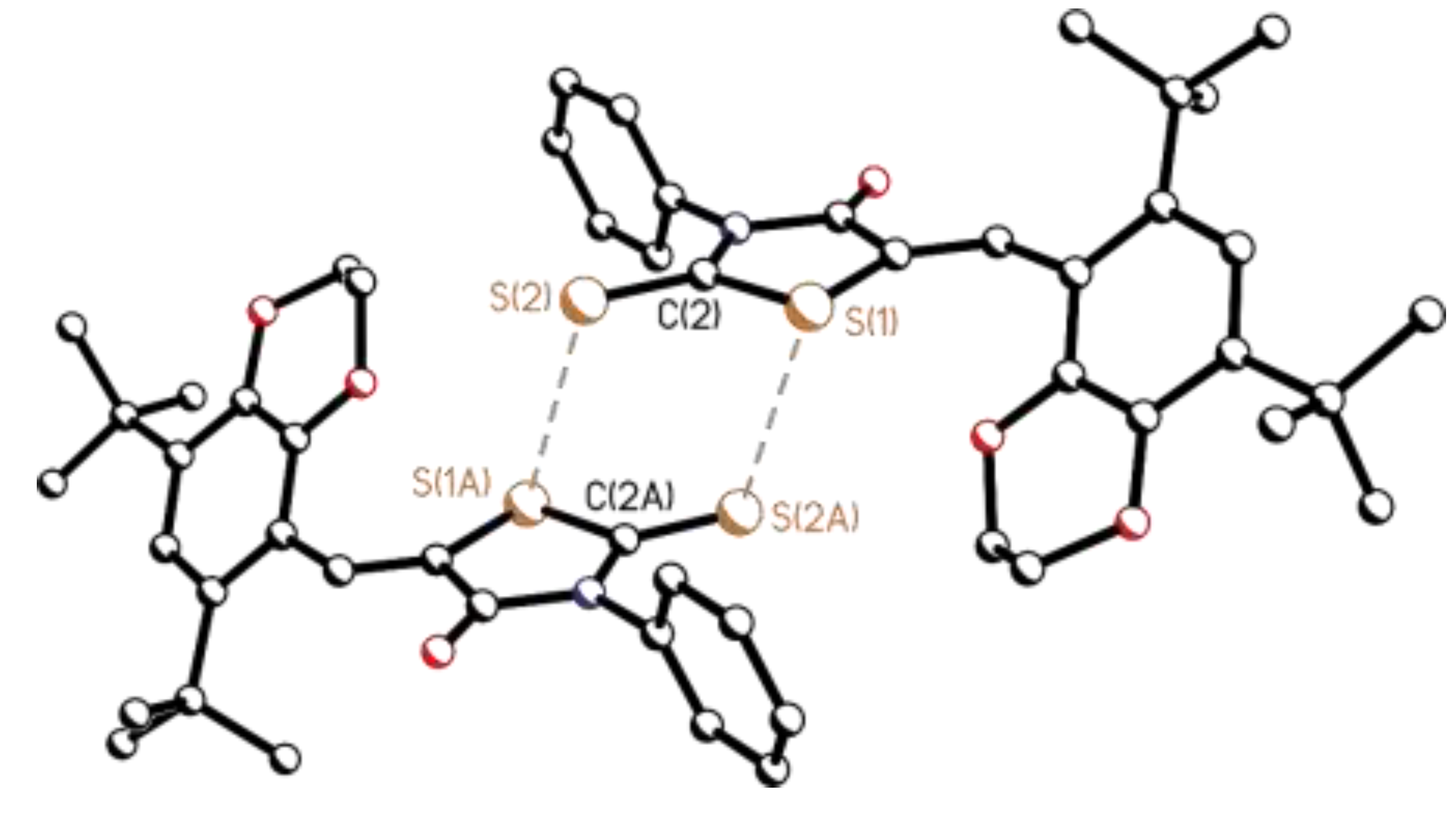
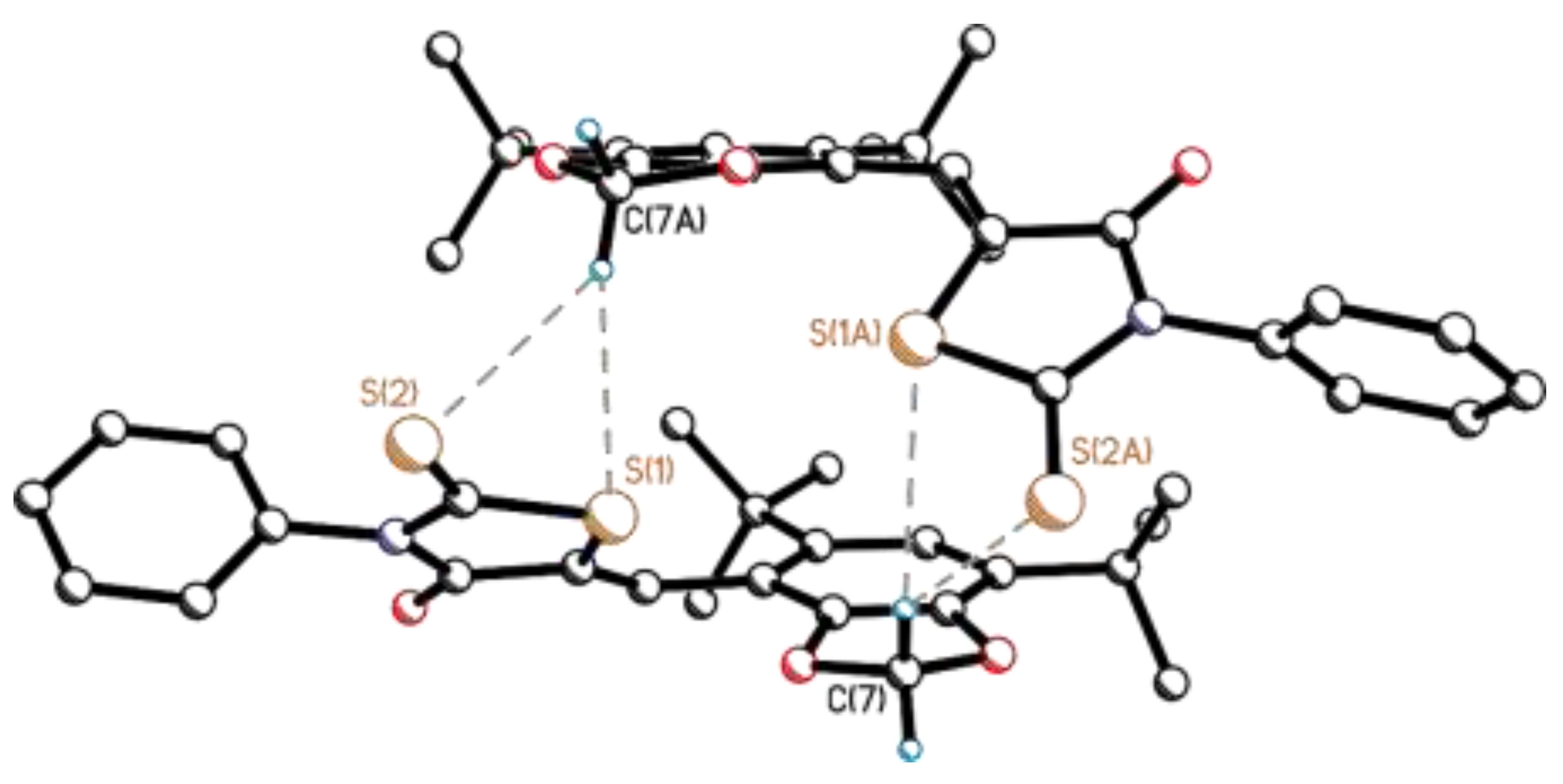
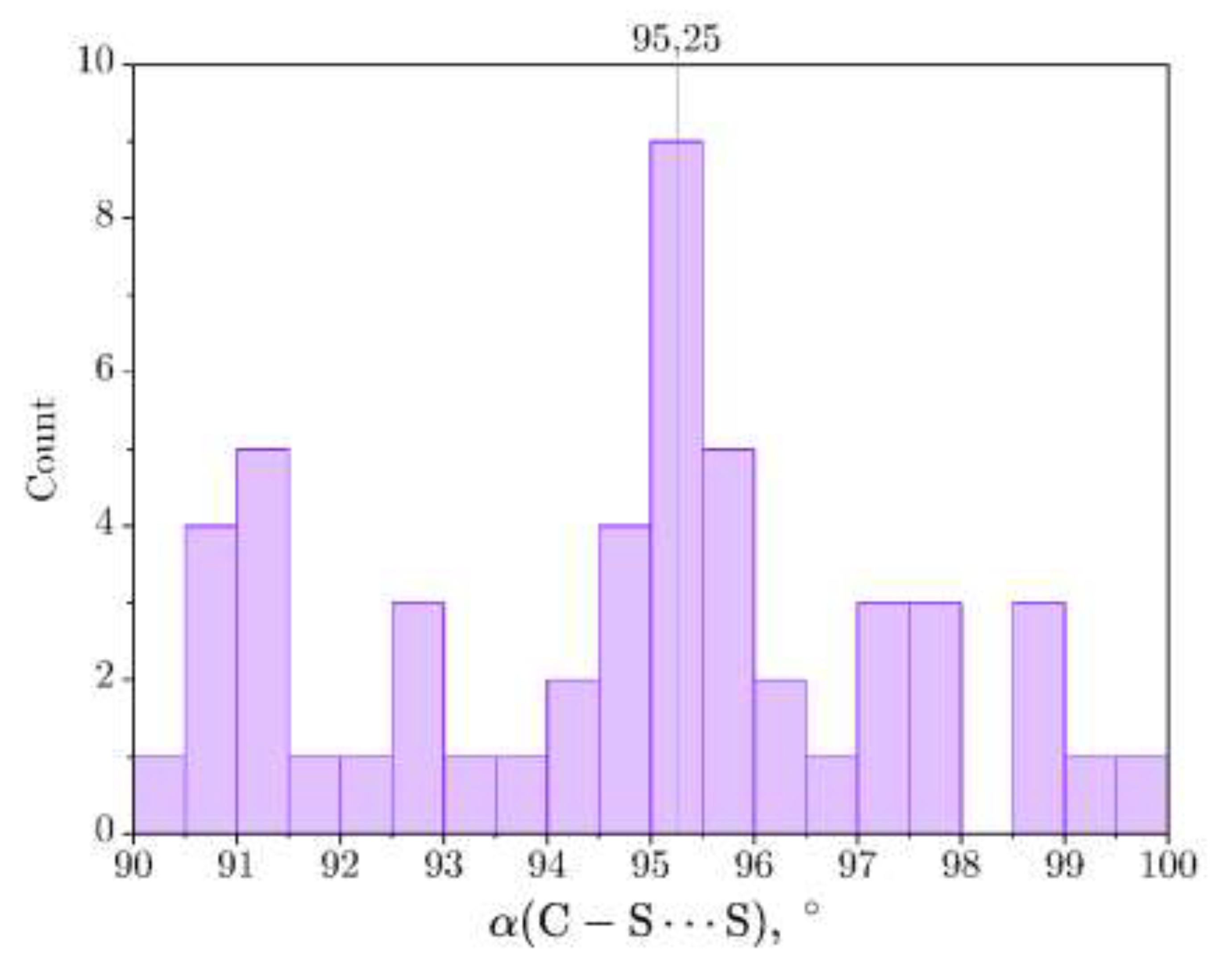
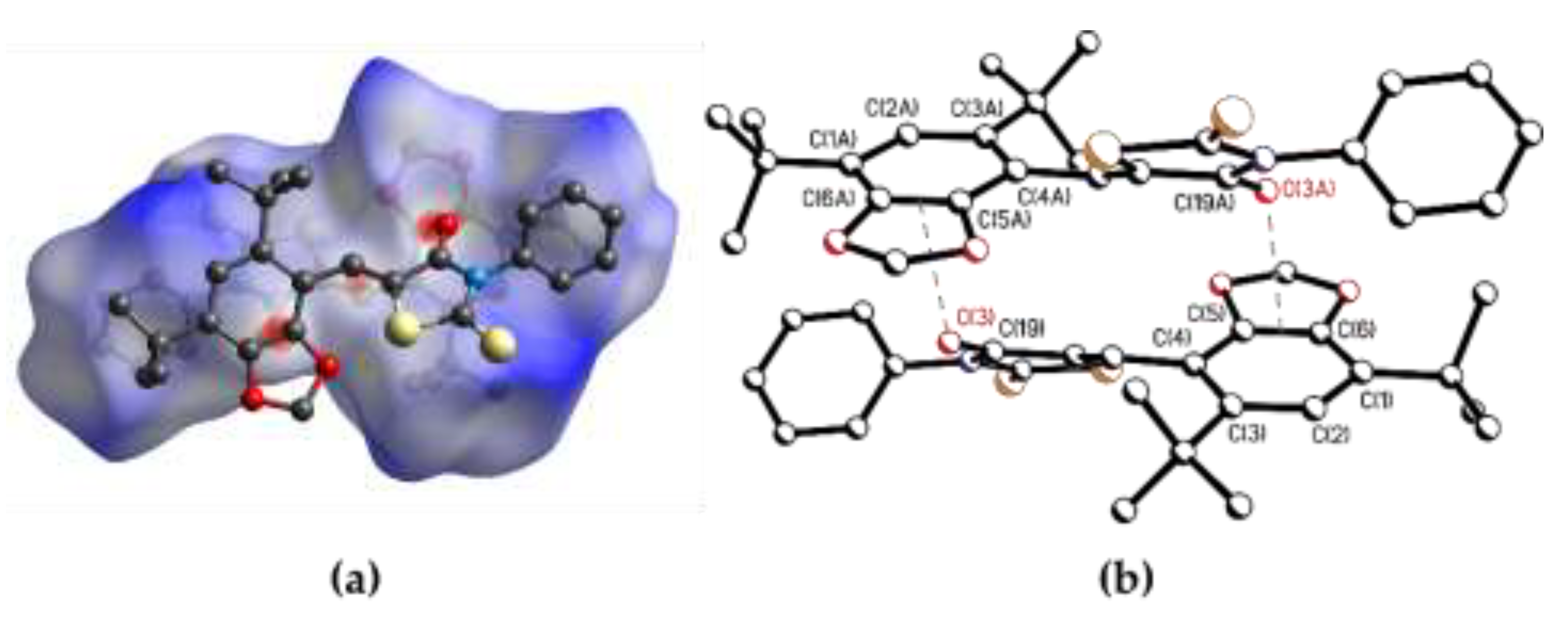
3.2. Single-Crystal X-Ray Diffraction
3.3. Hirshfield Surface Analysis
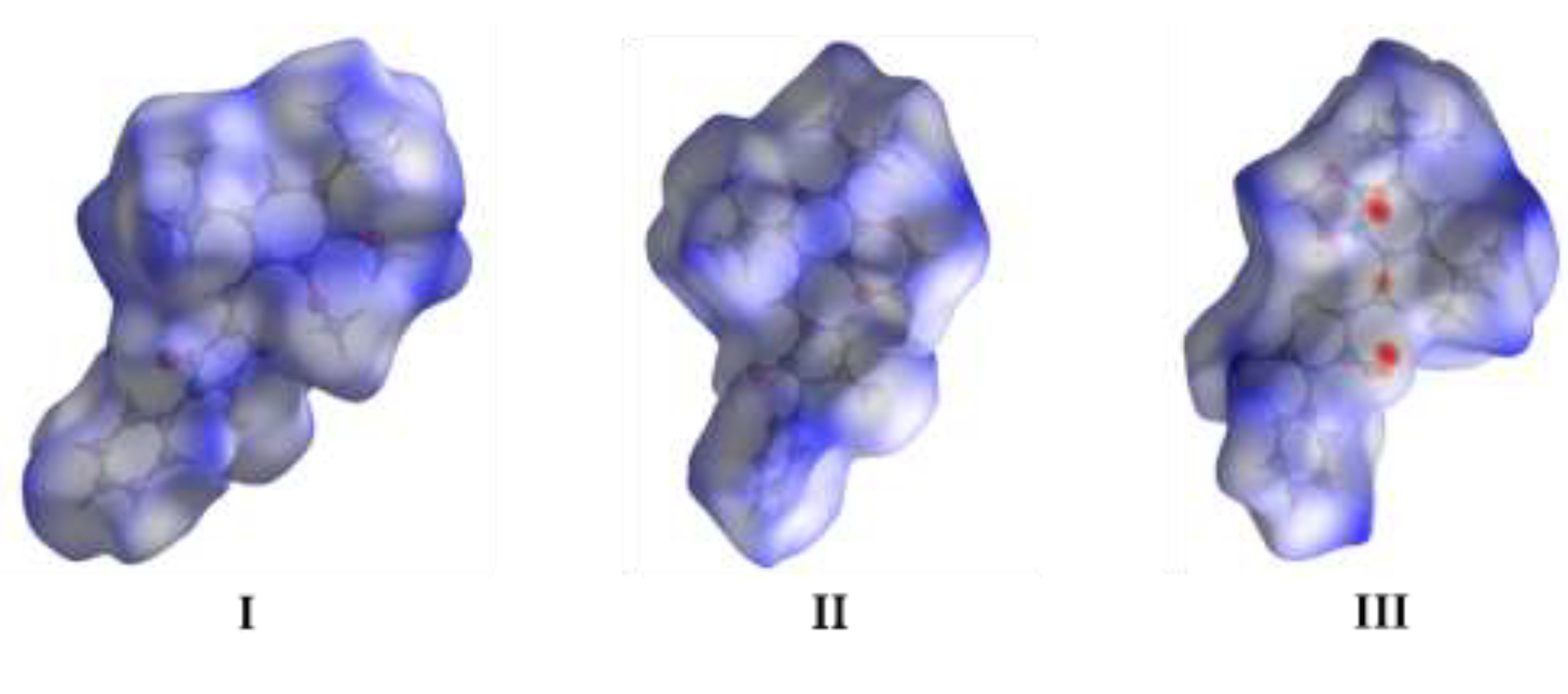
4. Conclusions
Data Availability Statement\
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Jabeen, S.; Palmer, R.A.; Potter, B.S.; Helliwell, M.; Dines, T.J.; Chowdhry, B.Z. Low Temperature Crystal Structures of Two Rhodanine Derivatives, 3-Amino Rhodanine and 3-Methyl Rhodanine: Geometry of the Rhodanine Ring. J Chem Crystallogr 2009, 39, 151–156. [Google Scholar] [CrossRef]
- Guerraoui, A.; Goudjil, M.; Direm, A.; Guerraoui, A.; Şengün, İY.; Parlak, C.; Djedouani, A.; Chelazzi, L.; Monti, F.; Lunedei, E.; Boumaza, A. A rhodanine derivative as a potential antibacterial and anticancer agent: Crystal structure, spectral characterization, DFT calculations, Hirshfeld surface analysis, in silico molecular docking and ADMET studies. Journal of Molecular Structure 2023, 1280, 135025. [Google Scholar] [CrossRef]
- Tejchman, W.; Zborowski, K.; Nitek, W.; Żesławska, E. Structural insights into 3-[5-(chloro benzyl idene)rhodanine]propionic acid isomers with anti bacterial activity. Acta Cryst. 2025, C81, 420–429. [Google Scholar] [CrossRef]
- Ramkumar, K.; Yarovenko, V.N.; Nikitina, A.S.; Zavarzin, I.V.; Krayushkin, M.M.; Kovalenko, L.V.; Esqueda, A.; Odde, S.; Neamati, N. Design, Synthesis and Structure-activity Studies of Rhodanine Derivatives as HIV-1 Integrase Inhibitors. Molecules 2010, 15, 3958–3992. [Google Scholar] [CrossRef] [PubMed]
- Chinchilli, K.K.; Akunuri, R.; Ghouse, S.M.; Soujanya, D.; Angeli, A.; Parupalli, R.; Arifuddin, M.; Yaddanapudi, V.M.; Supuran, C.T.; Nanduri, S. Design, synthesis, and structure–activity studies of new rhodanine derivatives as carbonic anhydrase II, IX inhibitors. Arch. Pharm. 2023, 356, e2300205. [Google Scholar] [CrossRef]
- Casas, J.S.; Castaño, M.V.; Couce, M.D.; Sánchez, A.; Sordo, J.; Torres, M.D.; Vázquez, S.A.; Vázquez-López, E.M. Relevance of weak intermolecular forces on the supramolecular structure of free or DMSO solvated 5-(4-X-benzylidene)rhodanines (X=F, Cl, Br, I). Journal of Molecular Structure 2016, 1120, 100–114. [Google Scholar] [CrossRef]
- Żesławska, E.; Zakrzewski, R.; Nowicki, A.; Korona-Głowniak, I.; Lyčka, A.; Kania, A.; Zborowski, K.K.; Suder, P.; Skórska-Stania, A.; Tejchman, W. Synthesis, Crystal Structures, Lipophilic Properties and Antimicrobial Activity of 5-Pyridylmethylidene-3-rhodanine-carboxyalkyl Acids Derivatives. Molecules 2022, 27, 3975. [Google Scholar] [CrossRef]
- Baryshnikov, G.V.; Minaev, B.F.; Minaeva, V.A.; Podgornaya, A.T. Theoretical Study OfThe Dimerization Of Rhodanine In Various Tautomeric Forms. Chemistry of Heterocyclic Compounds 2012, 47(10), 1268–1279. [Google Scholar] [CrossRef]
- Loncharich, R.J.; Nissen, J.S.; Boyd, D.B. Molecular orbital study of the structure and interactions of ylidene rhodanines. Struct Chem 1996, 7, 37–49. [Google Scholar] [CrossRef]
- Van der Helm, D.; Lessor, Jnr A.E.; Merritt Jnr, L.L. Acta Cryst 1962, 15, 1227–1232. [CrossRef]
- Merritt, L.L.; Lessor, A.E. Crystallographic data for rhodanine, C3H3S2NO. Acta Cryst. 1955, 8, 848. [Google Scholar] [CrossRef]
- ArmaregoW, L. F.; Chai, C. Purification of Laboratory Chemicals, 7th edition; 2013. [Google Scholar] [CrossRef]
- Janiak, C. A critical account on π–π stacking in metal complexes with aromatic nitrogen-containing ligands. In J. Chem. Soc.; Dalton, Translator; 2000; pp. 3885–3896. [Google Scholar] [CrossRef]
- X-Area Integrate. STOE Cie GmbH: Darmstadt, Germany, 2016.
- X-Area Recipe. STOE Cie GmbH: Darmstadt, Germany, 2015.
- X-Area Pilatus3_SV. STOE Cie GmbH: Darmstadt, Germany, 2016.
- X-Area LANA. STOE Cie GmbH: Darmstadt, Germany, 2016.
- APEX3 Crystallography Software Suite, version v2016.9-0.
- Bruker AXS Inc.; Madison, WI, USA, 2016.
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of silver and molybdenum microfocus X-Ray sources for single-crystal structure determination. J. Appl. Cryst. 2015, 48, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELXT – Integrated space-group and crystal-structure determination. Acta Crystallogr., Sect. A: Found. Adv. 2015, A71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr., Sect. A: Found. Adv. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- JonesG. The Knoevenagel Condensation. In Organic Reactions; 2011. [Google Scholar] [CrossRef]
- Antonijević, I.S.; Janjić, G.V.; Milčić, M.K.; Zarić, S.D. Preferred Geometries and Energies of Sulfur–Sulfur Interactions in Crystal Structures. Cryst. Growth Des. 2016, 16(2), 632–639. [Google Scholar] [CrossRef]
- Pascoe, D. J.; Ling, K. B.; Cockroft, S. L. The Origin of Chalcogen-Bonding Interactions. J. Am. Chem. Soc. 2017, 139(42), 15160–15167. [Google Scholar] [CrossRef] [PubMed]
- Mahmudov, K. T.; Kopylovich, M. N.; Guedes da Silva, M. F. C.; Pombeiro, A. J. L. Chalcogen bonding in synthesis, catalysis and design of materials. Dalton Trans. 2017, 46, 10121–10138. [Google Scholar] [CrossRef]
- Arca, M.; Lippolis, V.; Aragoni, M. C.; Podda, E.; Ciancaleoni, G.; Pintus, A. Computational approaches to the study of chalcogen bonding interactions. Coord. Chem. Rev. 2026, 556, 217514. [Google Scholar] [CrossRef]
- Spackman, P.R.; Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer: a program for Hirshfeld surface analysis, visualization and quantitative analysis of molecular crystals. J Appl Crystallogr 2021, 27(54), 1006–1011. [Google Scholar] [CrossRef]
- Churakov, A.V.; Medvedev, A.G.; Shishkina, A.V.; Frolov, N.E.; Vener, M.V. Tuning Supramolecular Structure in Trimethylglycine Cocrystals: Competition Between Hydrogen and Halogen Bonding upon Cl/Br Substitution. Molecules 2026, 31, 1047. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Vik, E.C.; Maier, J.M.; Karki, I.; Strickland, S.M.S.; Umana, J.M.; Smith, M.D.; Pellechia, P.J.; Shimizu, K.D. Electrostatically Driven CO−π Aromatic Interactions. J. Am. Chem. Soc. 2019, 141(32), 12513–12517. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Das, A. The π→π* interaction: a rapidly emerging non-covalent interaction. Phys. Chem. Chem. Phys. 2015, 17, 9596–9612. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



