Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
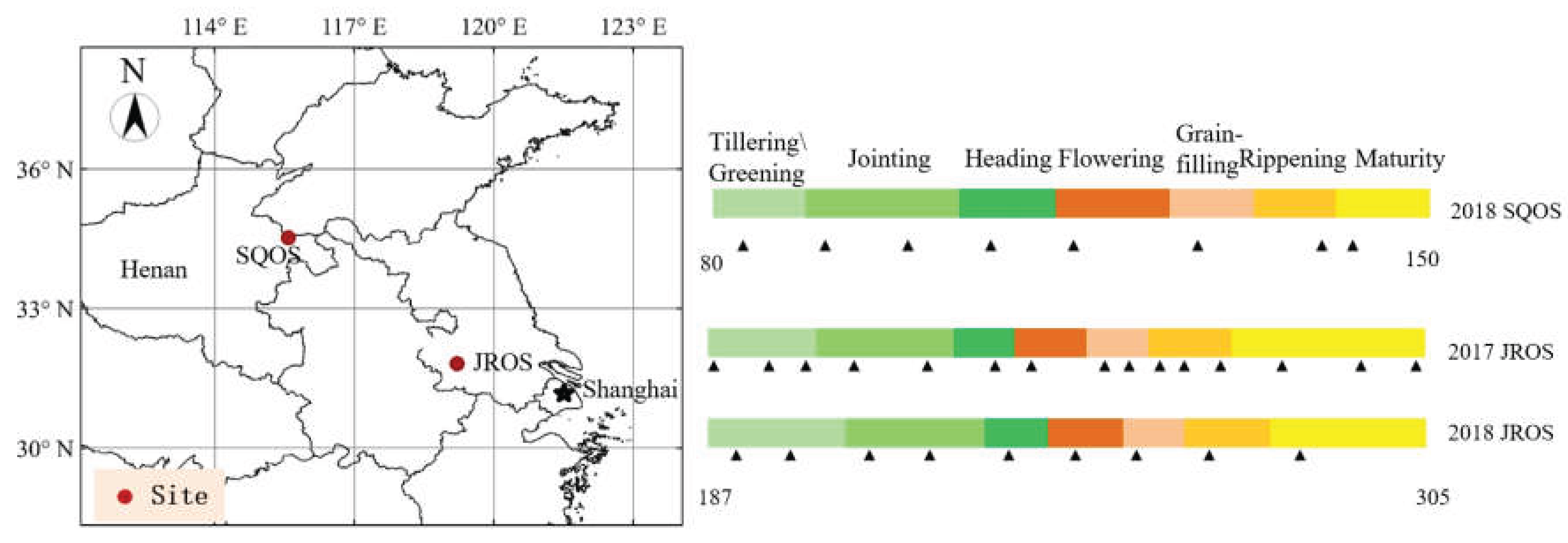
2.1. Field Sites
2.2. Measurements of Leaf Gas Exchange
2.3. Measurements of Leaf Biochemical Parameters
2.4. Analysis
3. Results
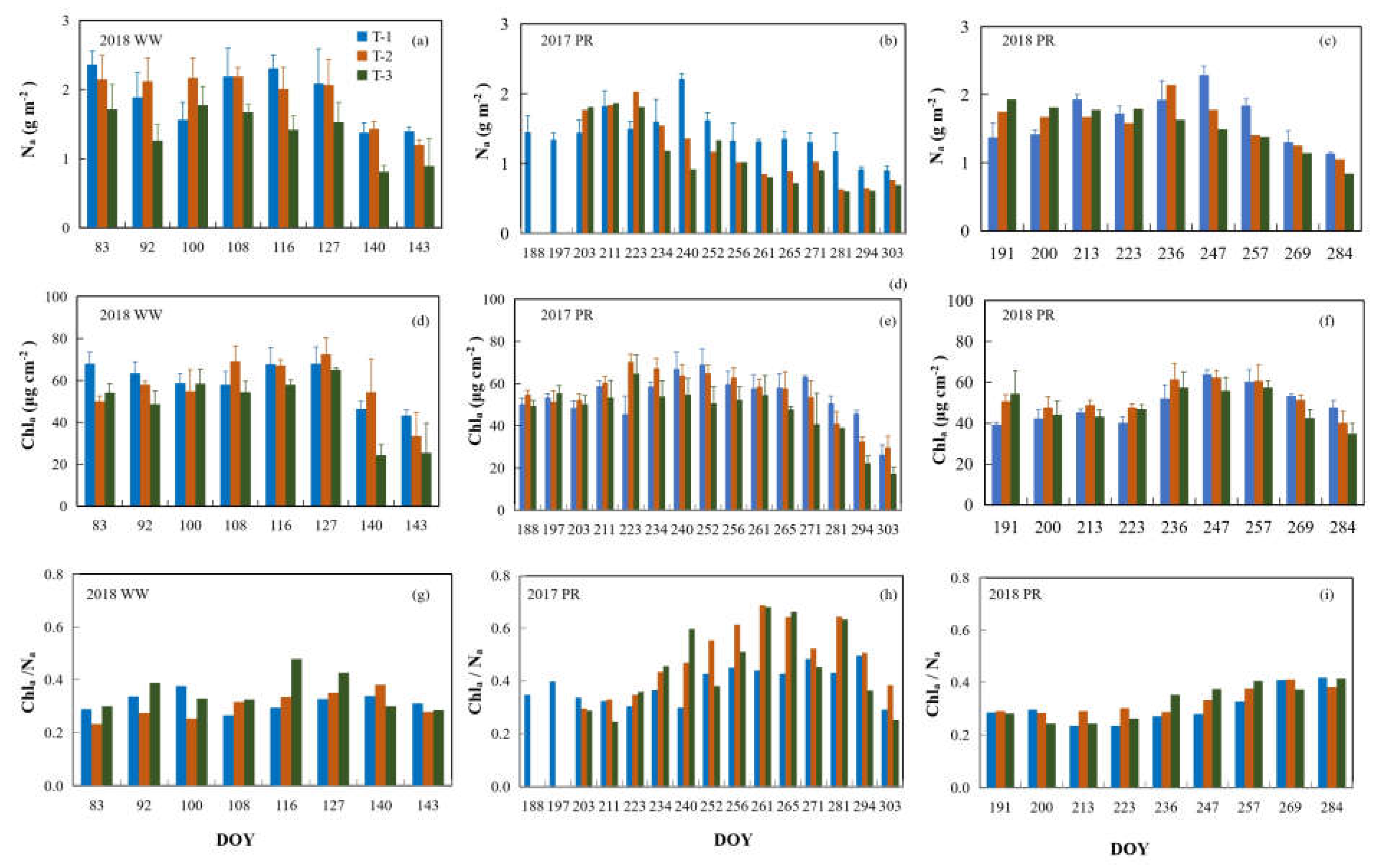
3.1. Seasonal Variations of Leaf Na and Chla in Crop Canopies
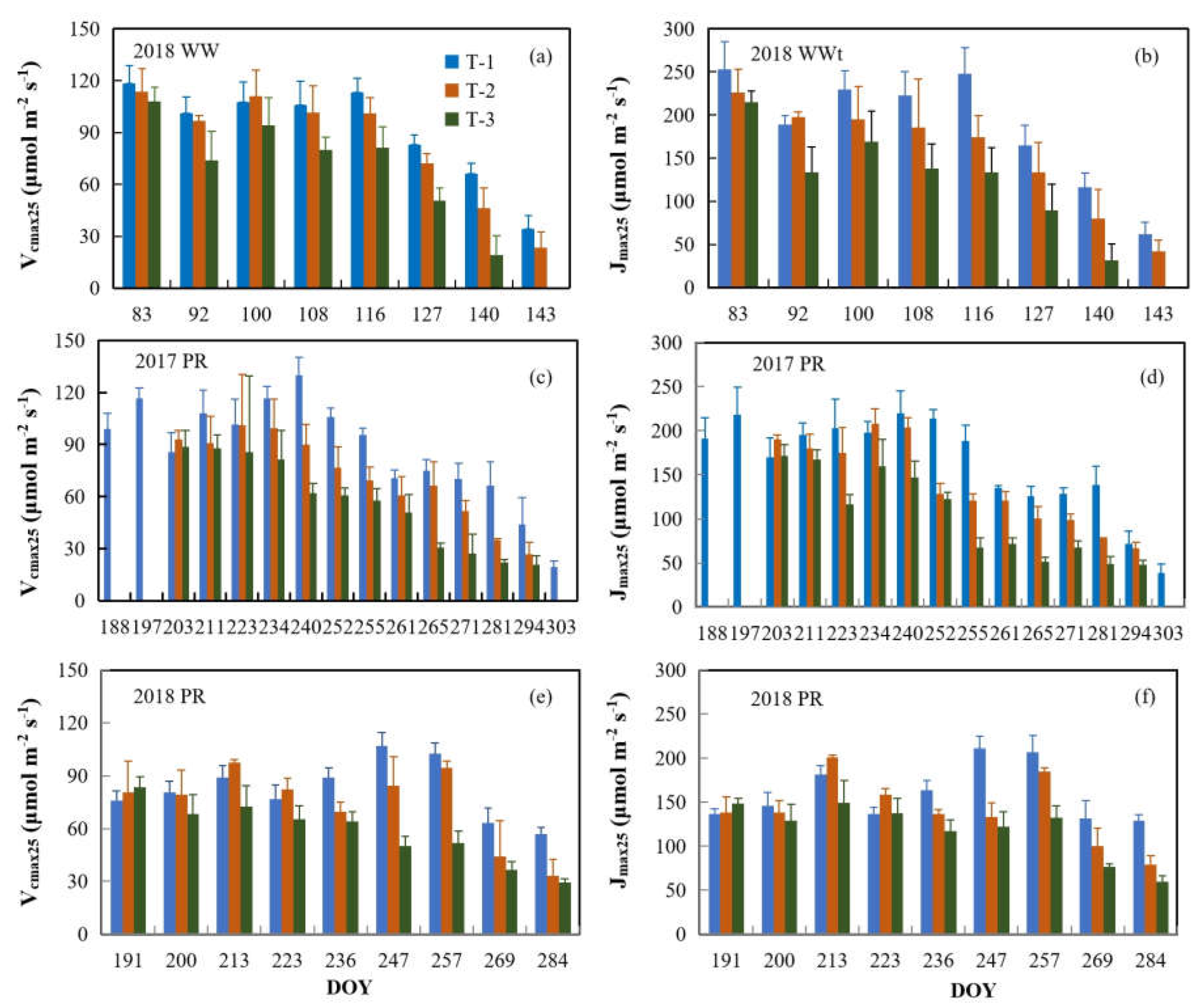
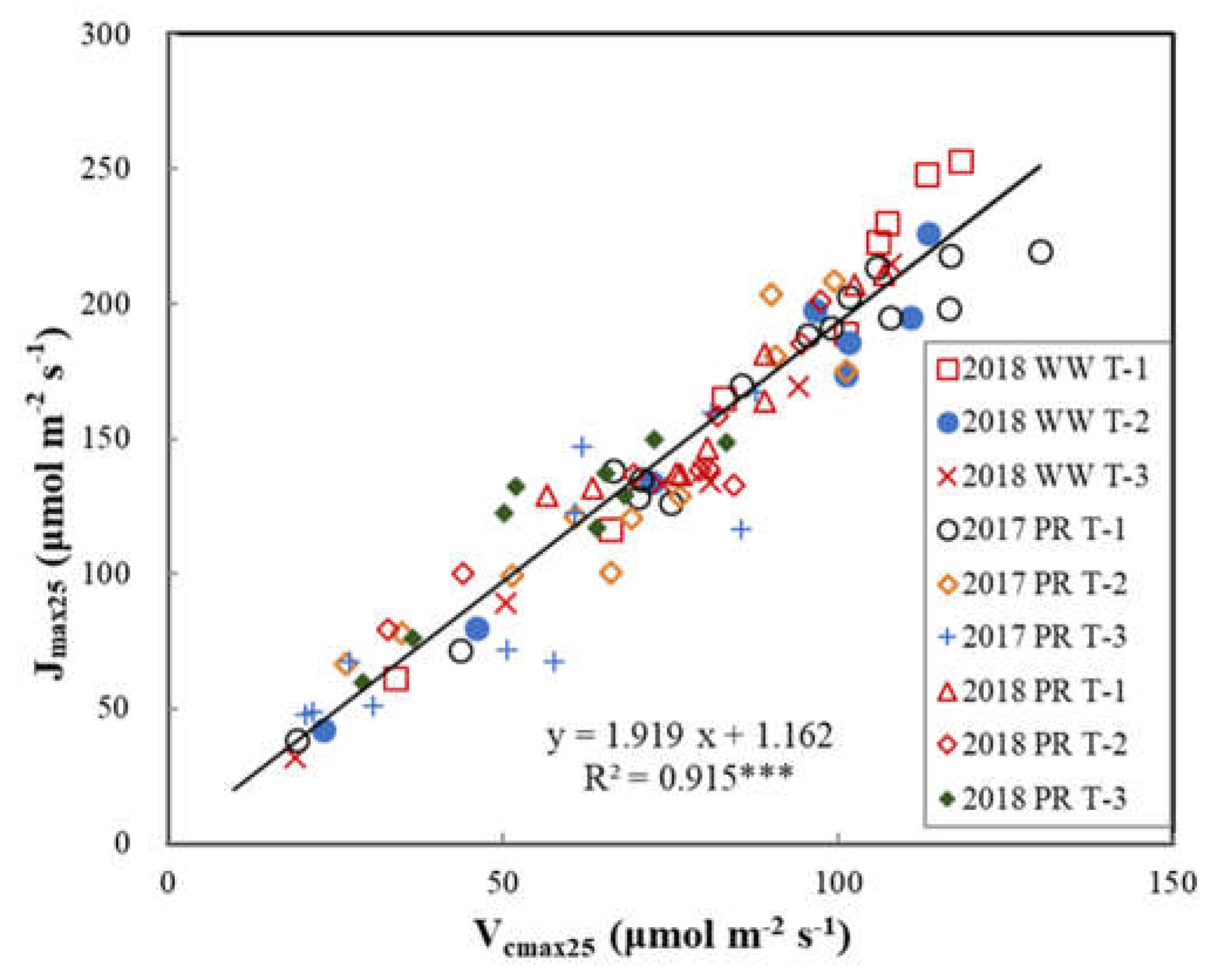
3.2. Seasonal Variations of Leaf Vcmax25 and Jmax25 in Crop Canopies
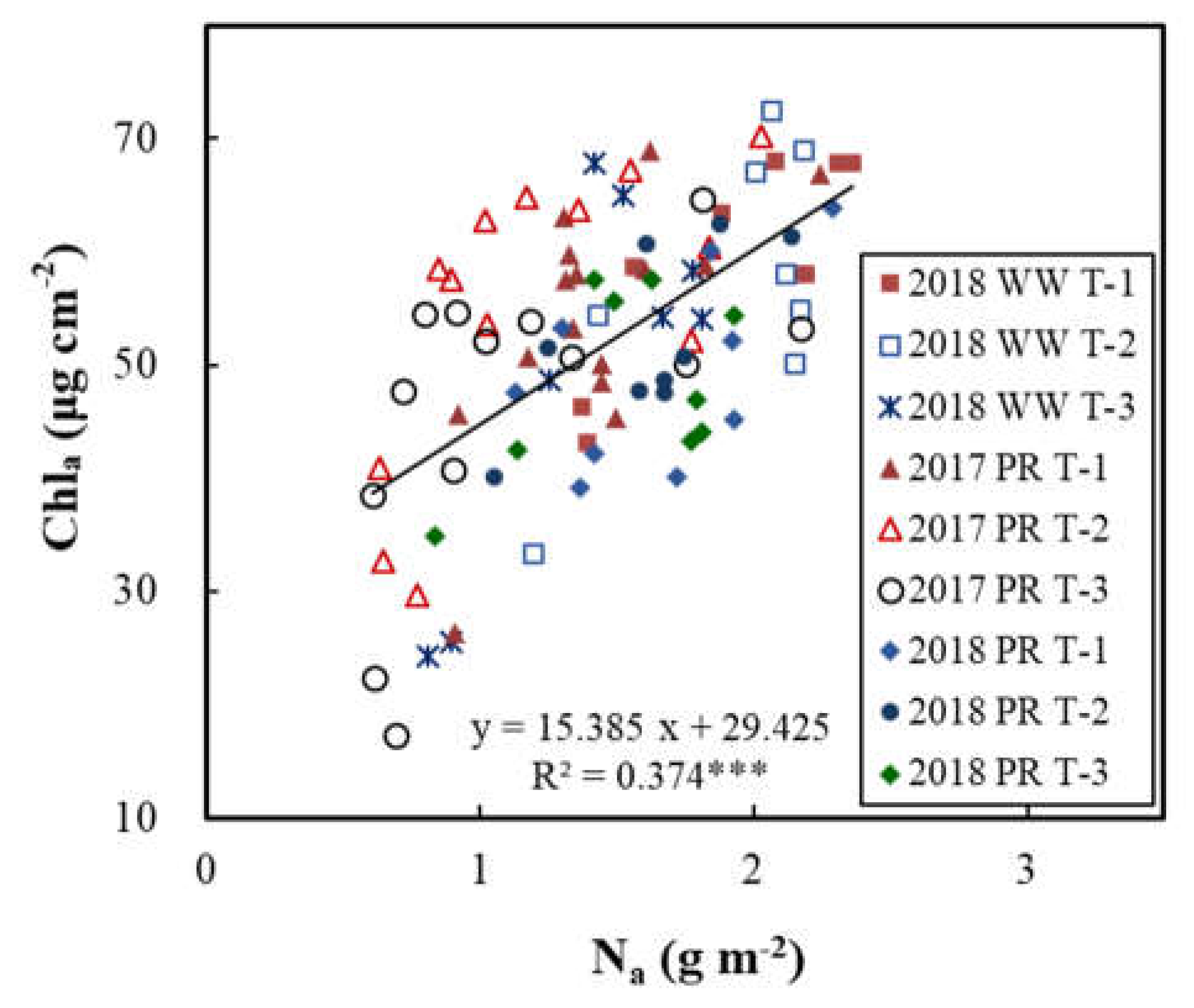
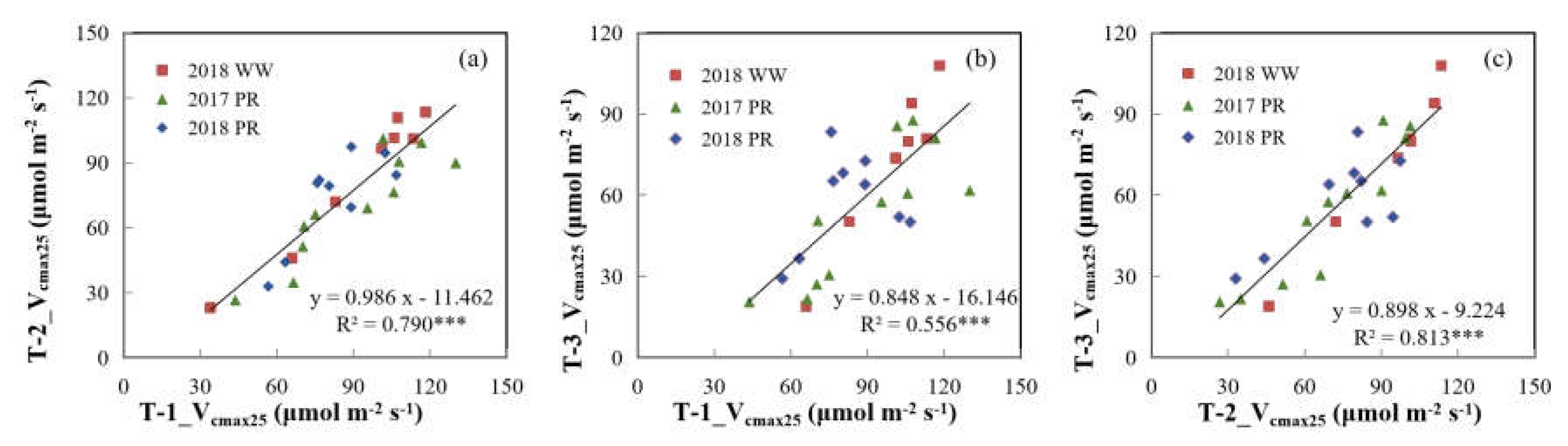
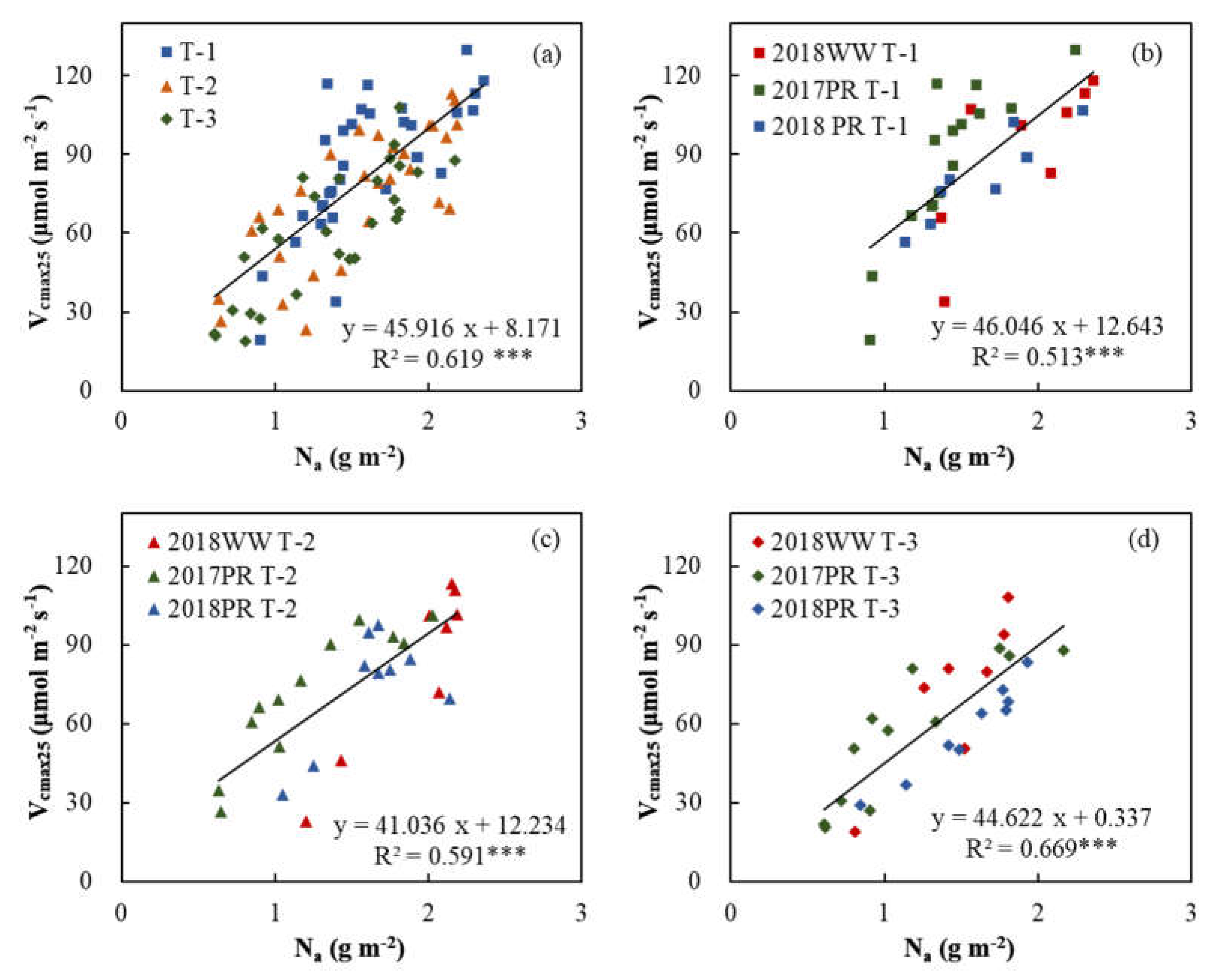
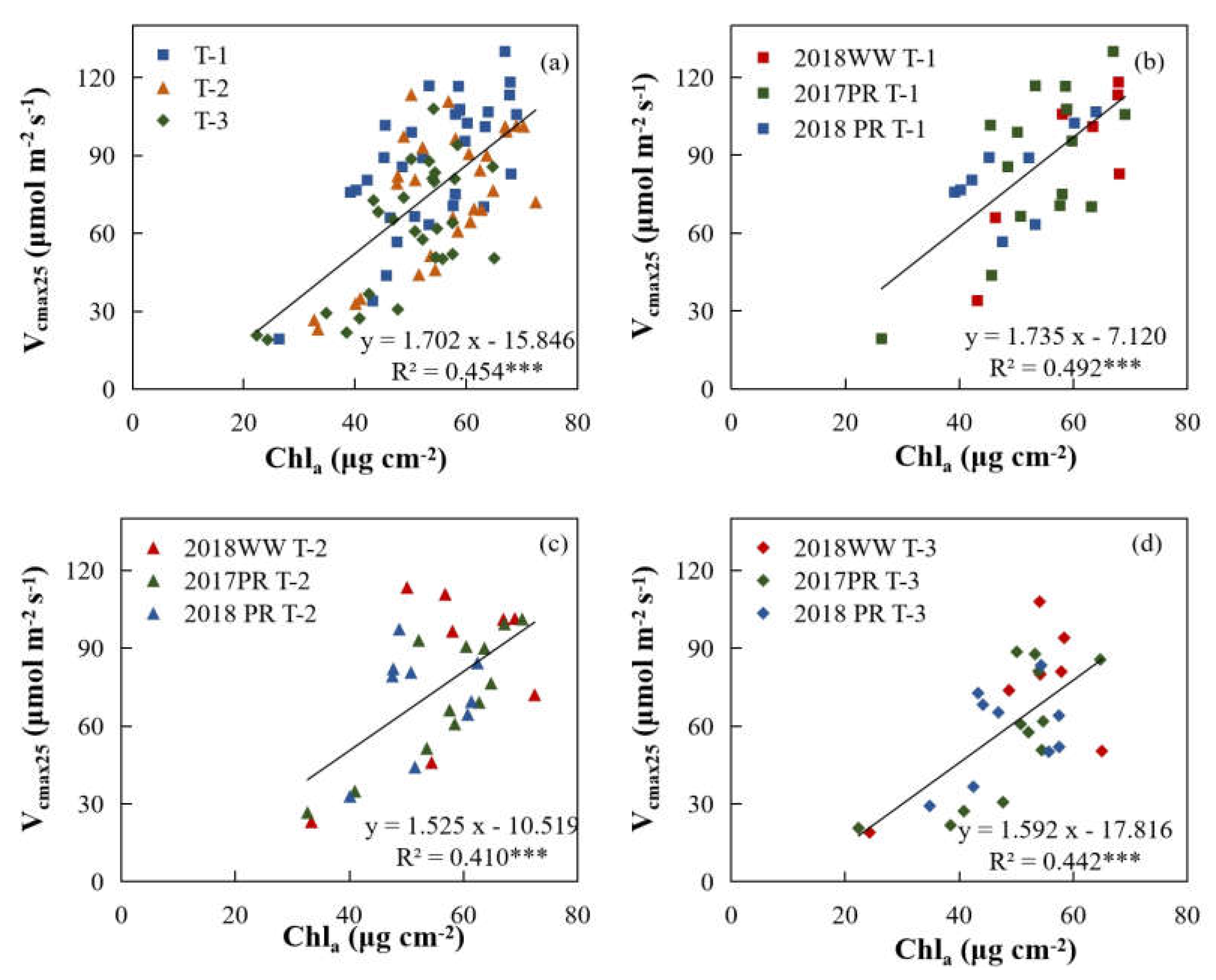
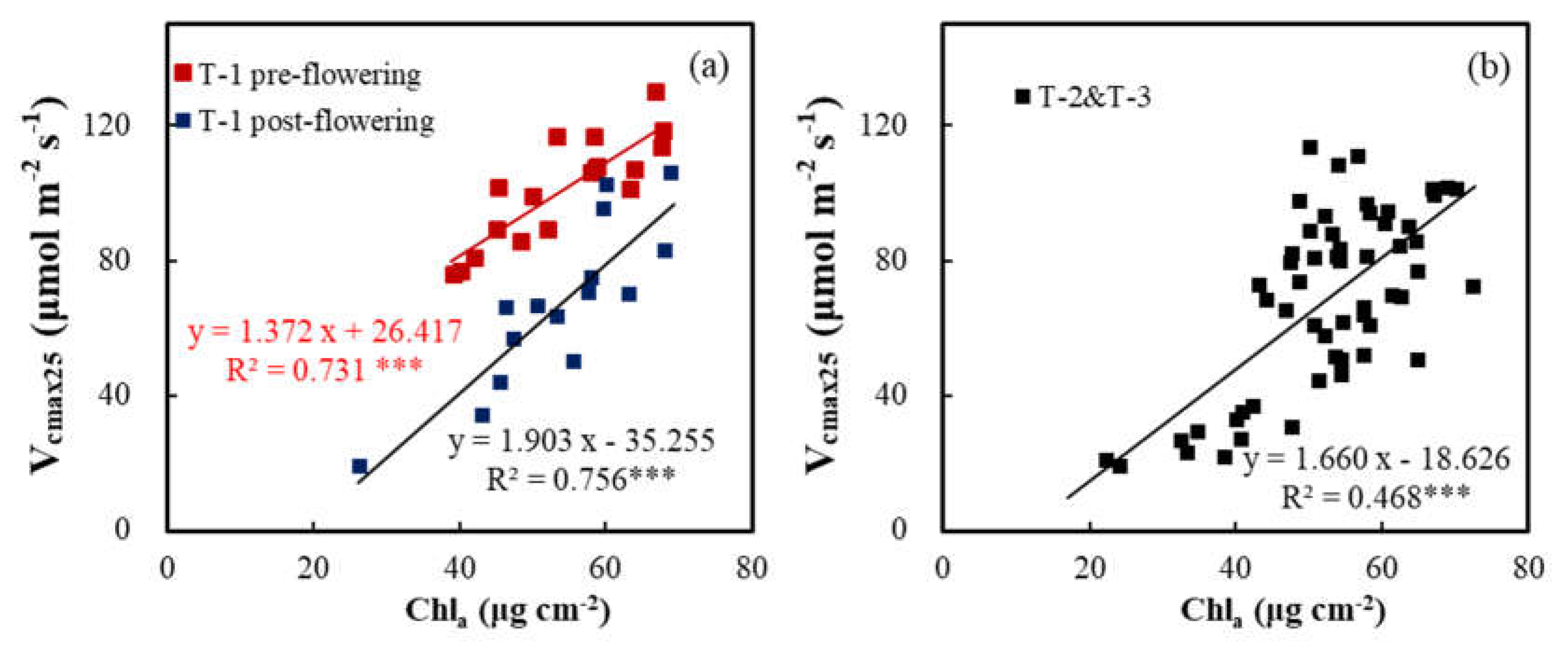
3.3. Relationships of Vcmax25 with Na and Chla
| Group Description | Independent Variable | Slope | Intercept | R² | Significance |
|---|---|---|---|---|---|
| T-1+T-2+T-3 | Nₐ | 45.916 | 8.171 | 0.619 | *** |
| T-1 | Nₐ | 46.046 | 12.643 | 0.513 | *** |
| T-2 | Nₐ | 41.036 | 12.234 | 0.591 | *** |
| T-3 | Nₐ | 44.622 | 0.337 | 0.669 | *** |
| T-1+T-2+T-3 | Chlₐ | 1.702 | -15.846 | 0.454 | *** |
| T-1 | Chlₐ | 1.735 | -7.120 | 0.492 | *** |
| T-2 | Chlₐ | 1.525 | -10.519 | 0.410 | *** |
| T-3 | Chlₐ | 1.592 | -17.816 | 0.442 | *** |
| T-1 pre-flowering | Chlₐ | 1.372 | 26.417 | 0.731 | *** |
| T-1 post-flowering | Chlₐ | 1.903 | -35.255 | 0.756 | *** |
| T-2 + T-3 | Chlₐ | 1.660 | -18.626 | 0.468 | *** |
4. Discussion
4.1. Difference in Vertical Patterns of Photosynthetic and Biochemical Parameters
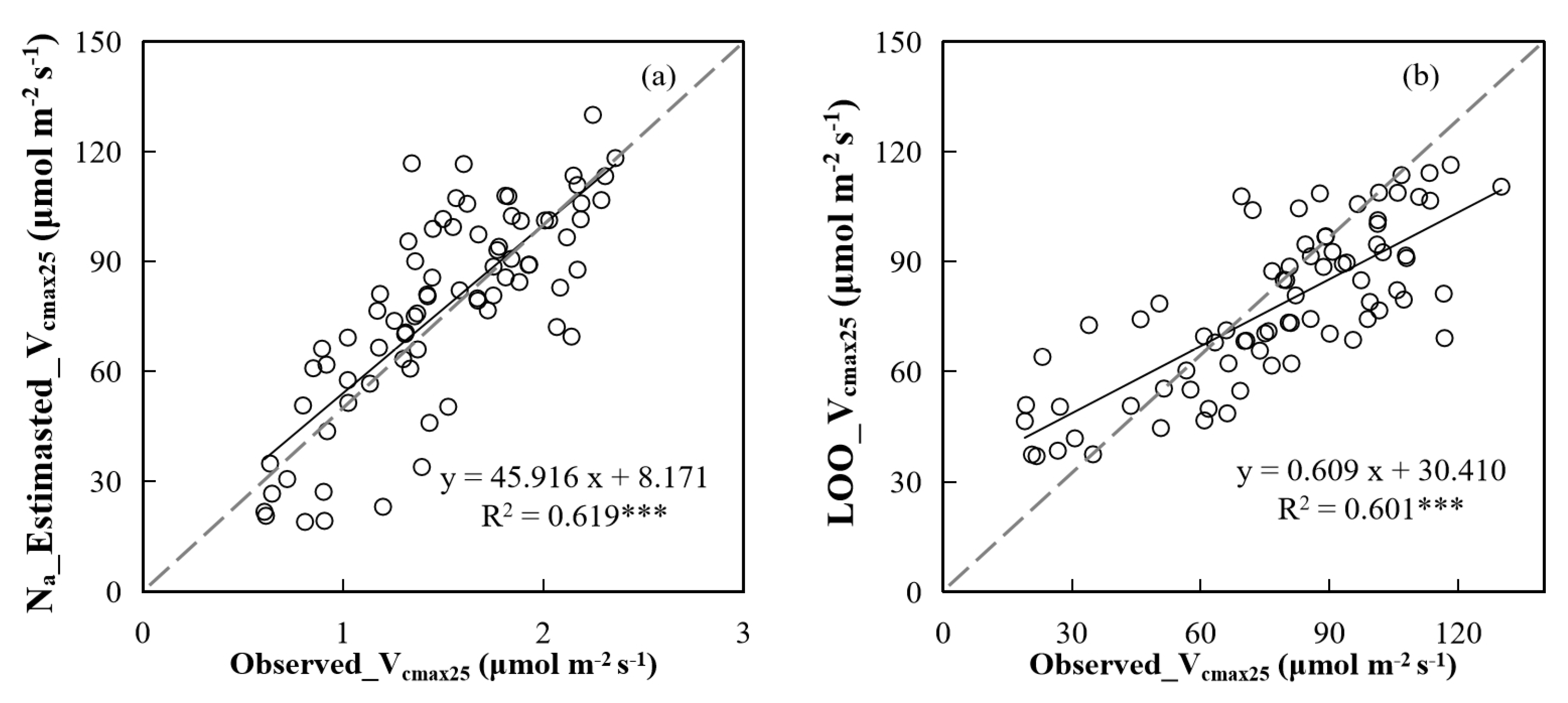
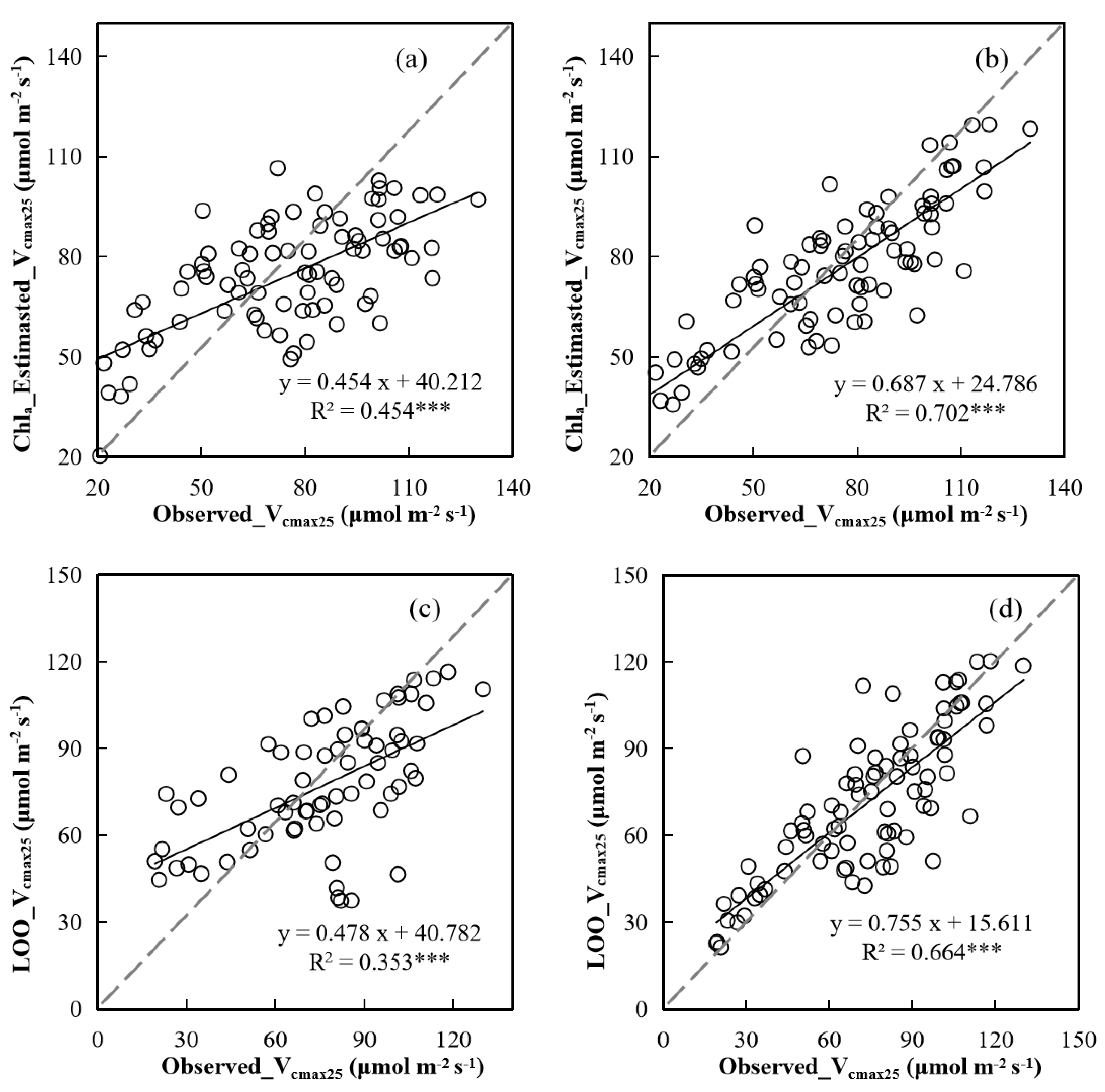
4.2. The Ability of Chla and Na to Characterize Vcmax25 of Leaves at Different Positions in the Canopy
4.3. Implications and Uncertainties in Remote Sensing of Canopy Photosynthetic Capacity
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kang, M.; Zhang, L.; Qin, T.; An, J.; Wang, C.; Wang, S.; Ali, I.; Liu, B.; Liu, L.; Tang, L.; et al. Bridging Chlorophyll Content and Vertical Nitrogen Distribution for Accurate Canopy Photosynthesis Simulation. Computers and Electronics in Agriculture 2025, 239, 110885. [CrossRef]
- Kaneko, T.; Nomura, K.; Yasutake, D.; Iwao, T.; Okayasu, T.; Ozaki, Y.; Mori, M.; Hirota, T.; Kitano, M. A Canopy Photosynthesis Model Based on a Highly Generalizable Artificial Neural Network Incorporated with a Mechanistic Understanding of Single-Leaf Photosynthesis. Agricultural and Forest Meteorology 2022, 323, 109036. [CrossRef]
- Kull, O.; Jarvis, P.G. The Role of Nitrogen in a Simple Scheme to Scale up Photosynthesis from Leaf to Canopy. Plant, Cell & Environment 1995, 18, 1174–1182. [CrossRef]
- Kull, O. Acclimation of Photosynthesis in Canopies: Models and Limitations. Oecologia 2002, 133, 267–279. [CrossRef]
- Kenzo, T.; Ichie, T.; Watanabe, Y.; Yoneda, R.; Ninomiya, I.; Koike, T. Changes in Photosynthesis and Leaf Characteristics with Tree Height in Five Dipterocarp Species in a Tropical Rain Forest. Tree Physiol 2006, 26, 865–873. [CrossRef]
- Legner, N.; Fleck, S.; Leuschner, C. Within-Canopy Variation in Photosynthetic Capacity, SLA and Foliar N in Temperate Broad-Leaved Trees with Contrasting Shade Tolerance. Trees 2014, 28, 263–280. [CrossRef]
- D’Odorico, P.; Emmel, C.; Revill, A.; Liebisch, F.; Eugster, W.; Buchmann, N. Vertical Patterns of Photosynthesis and Related Leaf Traits in Two Contrasting Agricultural Crops. Functional Plant Biology 2019, 46, 213–227. [CrossRef]
- Niinemets, Ü. Variation in Leaf Photosynthetic Capacity within Plant Canopies: Optimization, Structural, and Physiological Constraints and Inefficiencies. Photosynthesis research 2023, 158. [CrossRef]
- Ma, X.R.; Song, X.M.; Zhang, E.Z.; Du, J.B.; Sun, X. Role of Nitrogen Utilization in Facilitating Photosynthetic Compensation of Soybean under Vertically Heterogeneous Light. Photosynt. 2025, 63, 291–295. [CrossRef]
- Archontoulis, S.V.; Vos, J.; Yin, X.; Bastiaans, L.; Danalatos, N.G.; Struik, P.C. Temporal Dynamics of Light and Nitrogen Vertical Distributions in Canopies of Sunflower, Kenaf and Cynara. Field Crops Research 2011, 122, 186–198. [CrossRef]
- Lemaire, G.; Onillon, B.; Gosse, G.; Chartier, M.; Allirand, J.M. Nitrogen Distribution within a Lucerne Canopy during Regrowth: Relation with Light Distribution. Annals of Botany 1991, 68, 483–488.
- Sultana, F.; Dev, W.; Xin, M.; Han, Y.; Feng, L.; Lei, Y.; Yang, B.; Wang, G.; Li, X.; Wang, Z.; et al. Competition for Light Interception in Different Plant Canopy Characteristics of Diverse Cotton Cultivars. Genes (Basel) 2023, 14, 364. [CrossRef]
- Meir, P.; Kruijt, B.; Broadmeadow, M.; Kull, O.; Carswell, F.; Nobre, A. Acclimation of Photosynthetic Capacity to Irradiance in Tree Canopies in Relation to Leaf Nitrogen Concentration and Leaf Mass per Unit Area. Plant, Cell and Environment 2002, 25, 343–357. [CrossRef]
- Farquhar, G.D.; von Caemmerer, S.; Berry, J.A. A Biochemical Model of Photosynthetic CO 2 Assimilation in Leaves of C 3 Species. Planta 1980, 149, 78–90.
- Wullschleger, S.D. Biochemical Limitations to Carbon Assimilation in C 3 Plants—a Retrospective Analysis of the a/C i Curves from 109 Species. Journal of Experimental Botany 1993, 44, 907–920.
- Walker, A.P.; Beckerman, A.P.; Gu, L.; Kattge, J.; Cernusak, L.A.; Domingues, T.F.; Scales, J.C.; Wohlfahrt, G.; Wullschleger, S.D.; Woodward, F.I. The Relationship of Leaf Photosynthetic Traits - V Cmax and J Max - to Leaf Nitrogen, Leaf Phosphorus, and Specific Leaf Area: A Meta-Analysis and Modeling Study. Ecol Evol 2014, 4, 3218–3235. [CrossRef]
- Wang, X.; Shi, J. Leaf Chlorophyll Content Is the Crucial Factor for the Temporal and Spatial Variation of Global Plants Leaf Maximum Carboxylation Rate. Science of The Total Environment 2024, 927, 172280. [CrossRef]
- Ali, A.A.; Xu, C.; Rogers, A.; McDowell, N.G.; Medlyn, B.E.; Fisher, R.A.; Wullschleger, S.D.; Reich, P.B.; Vrugt, J.A.; Bauerle, W.L.; et al. Global-Scale Environmental Control of Plant Photosynthetic Capacity. Ecological Applications 2015, 25, 2349–2365. [CrossRef]
- Wu, J.; Rogers, A.; Albert, L.P.; Ely, K.; Prohaska, N.; Wolfe, B.T.; Oliveira Jr, R.C.; Saleska, S.R.; Serbin, S.P. Leaf Reflectance Spectroscopy Captures Variation in Carboxylation Capacity across Species, Canopy Environment and Leaf Age in Lowland Moist Tropical Forests. New Phytologist 2019, 224, 663–674. [CrossRef]
- Rogers, A.; Serbin, S.P.; Ely, K.S.; Sloan, V.L.; Wullschleger, S.D. Terrestrial Biosphere Models Underestimate Photosynthetic Capacity and CO2 Assimilation in the Arctic. New Phytol 2017, 216, 1090–1103. [CrossRef]
- Li, J.; Lu, X.; Ju, W.; Li, J.; Zhu, S.; Zhou, Y. Seasonal Changes of Leaf Chlorophyll Content as a Proxy of Photosynthetic Capacity in Winter Wheat and Paddy Rice. Ecological Indicators 2022, 140, 109018. [CrossRef]
- Qian, X.; Zhang, Y.; Liu, L. Growth-Stage-Dependent Relationship between Photosynthetic Capacity and Leaf Biochemical Traits in Cotton. Industrial Crops and Products 2025, 235, 121650. [CrossRef]
- Chen, J.M.; Wang, R.; Liu, Y.; He, L.; Croft, H.; Luo, X.; Wang, H.; Smith, N.G.; Keenan, T.F.; Prentice, I.C.; et al. Global Datasets of Leaf Photosynthetic Capacity for Ecological and Earth System Research.
- Croft, H.; Chen, J.M.; Luo, X.; Bartlett, P.; Chen, B.; Staebler, R.M. Leaf Chlorophyll Content as a Proxy for Leaf Photosynthetic Capacity. Glob Chang Biol 2017, 23, 3513–3524. [CrossRef]
- Niinemets, Ü.; Keenan, T.F.; Hallik, L. A Worldwide Analysis of Within-Canopy Variations in Leaf Structural, Chemical and Physiological Traits across Plant Functional Types. New Phytologist 2015, 205, 973–993. [CrossRef]
- Niinemets, U. Photosynthesis and Resource Distribution through Plant Canopies. Plant Cell Environ 2007, 30, 1052–1071. [CrossRef]
- Chen, J.M.; Liu, J.; Cihlar, J.; Goulden, M.L. Daily Canopy Photosynthesis Model through Temporal and Spatial Scaling for Remote Sensing Applications. Ecological Modelling 1999, 124, 99–119. [CrossRef]
- Luo, X.; Croft, H.; Chen, J.M.; He, L.; Keenan, T.F. Improved Estimates of Global Terrestrial Photosynthesis Using Information on Leaf Chlorophyll Content. Global Change Biology 2019, 25, 2499–2514. [CrossRef]
- Ye, M.; Wu, M.; Zhang, Y.; Wang, Z.; Zhang, H.; Zhang, Z. Physiological Factors Limiting Leaf Net Photosynthetic Rate in C3 Crops like Rice and Approaches for Improving It. Agronomy 2022, 12, 1830. [CrossRef]
- Albert, L.P.; Wu, J.; Prohaska, N.; de Camargo, P.B.; Huxman, T.E.; Tribuzy, E.S.; Ivanov, V.Y.; Oliveira, R.S.; Garcia, S.; Smith, M.N.; et al. Age-Dependent Leaf Physiology and Consequences for Crown-Scale Carbon Uptake during the Dry Season in an Amazon Evergreen Forest. New Phytologist 2018, 219, 870–884. [CrossRef]
- Dai, S.; Ju, W.; Zhang, Y.; He, Q.; Song, L.; Li, J. Variations and Drivers of Methane Fluxes from a Rice-Wheat Rotation Agroecosystem in Eastern China at Seasonal and Diurnal Scales. Science of The Total Environment 2019, 690, 973–990. [CrossRef]
- Li, J.; Zhang, Y.; Gu, L.; Li, Z.; Li, J.; Zhang, Q.; Zhang, Z.; Song, L. Seasonal Variations in the Relationship between Sun-Induced Chlorophyll Fluorescence and Photosynthetic Capacity from the Leaf to Canopy Level in a Rice Crop. J Exp Bot 2020, 71, 7179–7197. [CrossRef]
- Lu, X.; Ju, W.; Li, J.; Croft, H.; Chen, J.M.; Luo, Y.; Yu, H.; Hu, H. Maximum Carboxylation Rate Estimation with Chlorophyll Content as a Proxy of Rubisco Content. Journal of Geophysical Research: Biogeosciences 2020, 125, e2020JG005748. [CrossRef]
- Li, T.; Angeles, O.; Marcaida, M.; Manalo, E.; Manalili, M.P.; Radanielson, A.; Mohanty, S. From ORYZA2000 to ORYZA (v3): An Improved Simulation Model for Rice in Drought and Nitrogen-Deficient Environments. Agricultural and Forest Meteorology 2017, 237–238, 246–256. [CrossRef]
- Duursma, R.A. Plantecophys - an R Package for Analysing and Modelling Leaf Gas Exchange Data. PLoS ONE 2015, 10, e0143346. [CrossRef]
- Long, S.P.; Bernacchi, C.J. Gas Exchange Measurements, What Can They Tell Us about the Underlying Limitations to Photosynthesis? Procedures and Sources of Error. Journal of Experimental Botany 2003, 54, 2393–2401.
- Gujarati, D. Use of Dummy Variables in Testing for Equality between Sets of Coefficients in Linear Regressions: A Generalization. The American Statistician 1970, 24, 18–22. [CrossRef]
- Zhang, C.; Yi, Y.; Zhang, S.; Li, P. Quantitative Analysis of Vertical and Temporal Variations in the Chlorophyll Content of Winter Wheat Leaves via Proximal Multispectral Remote Sensing and Deep Transfer Learning. Agriculture 2024, 14, 1685. [CrossRef]
- Lambers, H.; Chapin III, F.S.; Pons, T. Plant Physiological Ecology: Second Edition; 2008; p. 604; ISBN 978-0-387-78340-6.
- Wang, Z.; Wang, J.; Zhao, C.; Zhao, M.; Huang, W.; Wang, C. Vertical Distribution of Nitrogen in Different Layers of Leaf and Stem and Their Relationship with Grain Quality of Winter Wheat. Journal of Plant Nutrition 2005, 28, 73–91. [CrossRef]
- Šimpraga, M.; Verbeeck, H.; Bloemen, J.; Vanhaecke, L.; Demarcke, M.; Joó, E.; Pokorska, O.; Amelynck, C.; Schoon, N.; Dewulf, J.; et al. Vertical Canopy Gradient in Photosynthesis and Monoterpenoid Emissions: An Insight into the Chemistry and Physiology Behind. Atmospheric Environment 2013, 80, 85–95. [CrossRef]
- Yu, W.; Ji, R.; Jia, Q.; Feng, R.; Wu, J.; Zhang, Y. Vertical Distribution Characteristics of Photosynthetic Parameters for Phragmites Australis in Liaohe River Delta Wetland, China. Journal of Freshwater Ecology 2017, 32, 557–573. [CrossRef]
- Song, G.; Wang, Q.; Jin, J. Exploring the Instability of the Relationship between Maximum Potential Electron Transport Rate and Maximum Carboxylation Rate in Cool-Temperate Deciduous Forests. Agricultural and Forest Meteorology 2021, 308–309, 108614. [CrossRef]
- Zhuang, J.; Zhou, L.; Wang, Y.; Chi, Y. Nitrogen Allocation Regulates the Relationship between Maximum Carboxylation Rate and Chlorophyll Content along the Vertical Gradient of Subtropical Forest Canopy. Agricultural and Forest Meteorology 2021, 307, 108512. [CrossRef]
- Wu, J.; Serbin, S.P.; Xu, X.; Albert, L.P.; Chen, M.; Meng, R.; Saleska, S.R.; Rogers, A. The Phenology of Leaf Quality and Its Within-Canopy Variation Is Essential for Accurate Modeling of Photosynthesis in Tropical Evergreen Forests. Global Change Biology 2017, 23, 4814–4827. [CrossRef]
- Li, H.W.; Zhang, J.; Zheng, Q.; Li, B.; Li, Z.S. Comparative Study of Photosynthetic Capacity in Lower Leaves in the Canopy of Dwarf and Semidwarf Wheat. Photosynthetica 2022, 60, 445–456. [CrossRef]
- Davidson, K.J.; Lamour, J.; McPherran, A.; Rogers, A.; Serbin, S.P. Seasonal Trends in Leaf-Level Photosynthetic Capacity and Water Use Efficiency in a North American Eastern Deciduous Forest and Their Impact on Canopy-Scale Gas Exchange. New Phytol 2023, 240, 138–156. [CrossRef]
- Ding, H.; Wang, Z.; Zhang, Y.; Li, J.; Jia, L.; Chen, Q.; Ding, Y.; Wang, S. A Mechanistic Model for Estimating Rice Photosynthetic Capacity and Stomatal Conductance from Sun-Induced Chlorophyll Fluorescence. Plant Phenomics 2023, 5. [CrossRef]
- Yan, Z.; Guo, Z.; Serbin, S.P.; Song, G.; Zhao, Y.; Chen, Y.; Wu, S.; Wang, J.; Wang, X.; Li, J.; et al. Spectroscopy Outperforms Leaf Trait Relationships for Predicting Photosynthetic Capacity across Different Forest Types. New Phytologist 2021, 232, 134–147. [CrossRef]
- Beck, P.S.A.; Atzberger, C.G.; Høgda, K.A.; Johansen, B.; Skidmore, A.K. Improved Monitoring of Vegetation Dynamics at Very High Latitudes: A New Method Using MODIS NDVI. REMOTE SENS ENVIRON 2006, 100, 321–334. [CrossRef]
- Bin Wu; Fan, L.; Xu, B.; Yang, J.; Zhao, R.; Wang, Q.; Ai, X.; Zhao, H.; Yang, Z. UAV-Based LiDAR and Multispectral Sensors Fusion for Cotton Yield Estimation: Plant Height and Leaf Chlorophyll Content as a Bridge Linking Remote Sensing Data to Yield. Industrial Crops and Products 2025, 230, 121110. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




