Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
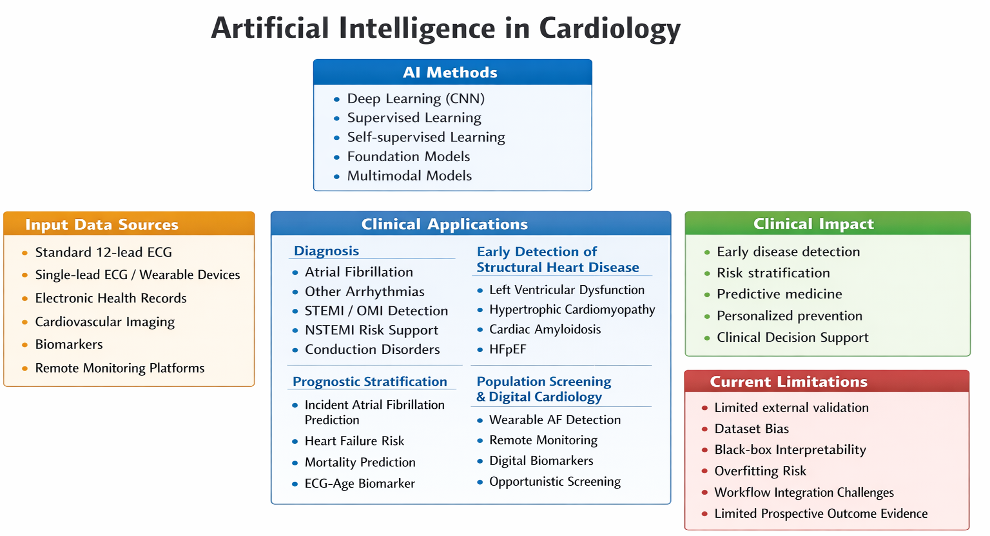
1. Introduction
2. Materials and Methods
3. Fundamentals of AI Applied to the ECG
4. Applications of AI-ECG in Arrhythmia Diagnosis
5. Identification of Structural Heart Disease Using AI-ECG
| Study | Population | AI model | ECG type | Task | Main results | Clinical implication |
|---|---|---|---|---|---|---|
| Desai et al., 2025 [25] | 103,492 ECGs from 45,873 consecutive adult patients (real-world tertiary-care cohort) | Convolutional neural network (Viz-HCM AI-ECG algorithm) | Standard 12-lead ECG | Detection of hypertrophic cardiomyopathy | At probability threshold ≥0.85: sensitivity 95–100%, specificity >98%, accuracy >98%; AI-ECG identified 5% previously undiagnosed HCM cases among flagged patients | Demonstrates feasibility of large-scale real-world AI-ECG screening for HCM and supports its role as an early detection tool to trigger downstream imaging and family screening |
| Siontis et al., 2021 [26] | 300 pediatric HCM patients (≤18 years) vs 18,439 matched controls | Deep learning convolutional neural network (AI-ECG) | Standard 12-lead ECG | Detection of HCM in children and adolescents | AUC 0.98; sensitivity 92%; specificity 95%; NPV 99%; performance remained high even in patients with normal ECG by conventional interpretation | Supports feasibility of AI-ECG as a non-invasive screening tool for early detection of pediatric HCM and surveillance of at-risk individuals such as athletes and genotype-positive relatives |
| Ko et al., 2020 [27] | 3,060 HCM patients vs 63,941 controls (Mayo Clinic dataset; separate training/validation/testing cohorts) | Convolutional neural network (deep learning) | Standard 12-lead ECG | Identification of hypertrophic cardiomyopathy | AUC 0.96; sensitivity 87%; specificity 90%; NPV 99%; high performance even with normal ECG and in patients meeting ECG-LVH criteria; accuracy particularly high in subjects <40 years | Supports feasibility of ECG-based AI screening for HCM, especially in young individuals and populations with familial risk |
| Bos et al., 2023 [28] | Multicenter real-world cohort of patients with HCM vs controls | Deep learning AI-ECG algorithm | Standard 12-lead ECG | Identification of hypertrophic cardiomyopathy | AUC 0.92; performance maintained in subgroups with apparently normal ECG or nonspecific LVH; predicted probability correlated with LV wall thickness and myocardial mass; performance independent of age, sex, and genotype | Supports use of AI-ECG as a non-invasive screening tool for HCM and for selecting patients for advanced imaging or genetic evaluation |
| Grogan et al., 2021 [30] | 2,541 patients with cardiac amyloidosis (AL + ATTR) vs 2,454 age- and sex-matched controls (Mayo Clinic cohort) | Convolutional neural network (deep learning AI-ECG) | Standard 12-lead ECG (with additional analysis of 6-lead and single-lead configurations) | Detection of cardiac amyloidosis | AUC 0.91 (95% CI 0.90–0.93); sensitivity 84%; specificity 85%; PPV 0.86; NPV 0.84; disease identified >6 months before clinical diagnosis in 59% of patients with prior ECGs; maintained performance with 6-lead (AUC 0.90) and single-lead ECG (AUC 0.86) | Supports use of AI-ECG as a non-invasive early screening tool for cardiac amyloidosis, including point-of-care and reduced-lead device settings |
| Goto et al., 2021 [31] | International multicenter cohorts with cardiac amyloidosis vs phenotypically overlapping conditions (HCM, hypertensive LVH, ESRD) | Multimodal CNN (2D CNN for ECG; 3D CNN for echocardiography) | Standard 12-lead ECG + apical four-chamber echocardiographic view | Detection of cardiac amyloidosis | ECG model C-statistic 0.85–0.91 in external validation; echocardiographic model C-statistic 0.89–1.00; sequential ECG→echo strategy increased PPV from ~33% to 74–77%; disease identified up to 1 year before clinical diagnosis; echocardiographic model outperformed cardiologist interpretation of images alone | Demonstrates effectiveness of multimodal ECG-first AI screening strategies for early identification of cardiac amyloidosis and improved diagnostic discrimination from phenotypically similar hypertrophic conditions |
| Liu et al., 2022 [32] | 34,103 patients from two centers (32,671 ECG training; 13,934 ECG internal/external validation) | Deep learning neural network (AI-ECG) | Standard 12-lead ECG (with additional analysis of 6-lead and single-lead configurations) | Detection of HFpEF | AUC 0.866 (internal) and 0.869 (external); maintained performance with 6-lead ECG (AUC 0.808–0.858) and single-lead ECG (AUC 0.784–0.845); among patients initially without HFpEF, predicted high-risk subgroup showed 24-month incidence 33.6% vs 8.4% (p<0.001); diastolic dysfunction detection AUC 0.837 | Supports use of AI-ECG as a non-invasive early screening tool for HFpEF, including simplified-lead and ambulatory/wearable-compatible ECG configurations |
6. AI-ECG in Acute Coronary Syndromes
| Study | Population | AI model | ECG type | Task | Main results | Clinical implication |
|---|---|---|---|---|---|---|
| Lee et al., 2025 (ROMIAE multicentre study) [33] | 8,493 adult patients presenting to 18 emergency departments within 24 h of symptom onset suspicious for AMI; AMI prevalence 18.6% (1,586/8,493) | Residual neural network–based AI-ECG model (AiTiAMI v1.00.00) | Standard 12-lead ECG (initial ED ECG only; raw signal 500 Hz) | Early detection and risk stratification of AMI and prediction of 30-day MACE | AMI detection: AUC 0.878, sensitivity 76.7%, specificity 84.8%, PPV 53.6%, NPV 94.1%; low-risk threshold sensitivity 99.6% and NPV 99.1%; high-risk threshold PPV 60.4%; STEMI AUC 0.971; NSTEMI AUC 0.814; 30-day MACE AUC 0.866; performance comparable to HEART score and superior to GRACE score, physician assessment, and initial hs-troponin; integration with HEART improved discrimination (C-index 0.926; NRI 19.6%) | Supports AI-ECG as a rapid ECG-only triage tool for early rule-out and rule-in of AMI in the emergency department, enabling accelerated chest-pain pathways and improved pre-biomarker risk stratification |
| Herman et al., 2023 [34] | International multicenter cohorts of patients with suspected acute coronary syndrome undergoing 12-lead ECG | Convolutional neural network (AI-ECG) | Standard 12-lead ECG | Detection of acute coronary occlusion myocardial infarction (OMI) | External validation AUC ≈ 0.93–0.95; improved identification of acute coronary occlusion compared with traditional STEMI criteria, including cases without ST elevation | Supports early AI-ECG–based decision support for identifying OMI missed by conventional STEMI criteria and prioritizing urgent reperfusion strategies |
| Herman et al., 2024 [35] | 1,032 patients with suspected STEMI undergoing urgent catheterization laboratory activation across 3 PCI centers in the United States | Convolutional neural network AI-ECG (Queen of Hearts, PMcardio) | Standard 12-lead ECG | Improvement of diagnostic accuracy in STEMI triage and detection of STEMI mimics | STEMI confirmed in 601/1,032 patients (58.2%); AI-ECG showed higher sensitivity than conventional triage and significantly reduced false catheterization laboratory activations while maintaining diagnostic performance | Supports early AI-assisted decision-making in STEMI triage, optimizing resource utilization and reducing unnecessary cath-lab activation without delaying reperfusion |
7. Prognostic Stratification and Prediction of Cardiovascular Outcomes Using AI-ECG
8. Population Screening, Wearable Devices, and Digital Biomarkers
9. Multimodal Integration and Future Perspectives of AI-ECG
10. Current Limitations of AI-ECG and Barriers to Clinical Implementation
11. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AF | Atrial Fibrillation |
| AI | Artificial Intelligence |
| AL | Amyloid Light-chain |
| AMI | Acute Myocardial Infarction |
| ATTRwt | Transthyretin Wild-Type amyloidosis |
| AUC | Area Under the Curve |
| CI | Confidence Interval |
| CNN | Convolutional Neural Network |
| CODE | Clinical Outcomes in Digital Electrocardiography |
| COVID-19 | Coronavirus Disease 2019 |
| ECG | Electrocardiogram |
| ED | Emergency Department |
| ESRD | End-Stage Renal Disease |
| GRACE | Global Registry of Acute Coronary Events (risk score) |
| HCM | Hypertrophic Cardiomyopathy |
| HEART | History, ECG, Age, Risk factors, Troponin (risk score) |
| HFpEF | Heart Failure with preserved Ejection Fraction |
| HFrEF | Heart Failure with reduced Ejection Fraction |
| HR | Hazard Ratio |
| LV | Left Ventricle |
| LVEF | Left Ventricular Ejection Fraction |
| LVH | Left Ventricular Hypertrophy |
| MACE | Major Adverse Cardiovascular Events |
| NPV | Negative Predictive Value |
| NRI | Net Reclassification Improvement |
| NSTEMI | Non–ST-segment Elevation Myocardial Infarction |
| OMI | Occlusion Myocardial Infarction |
| OR | Odds Ratio |
| PCI | Percutaneous Coronary Intervention |
| PMcardio | Powerful Medical Cardiology (AI-ECG software platform) |
| PPG | Photoplethysmography |
| PPV | Positive Predictive Value |
| ROMIAE | Risk-stratification Of Myocardial Infarction using Artificial intelligence in the Emergency department |
| STEMI | ST-Elevation Myocardial Infarction |
| TTE | Transthoracic Echocardiography |
| Viz-HCM | Visualization Hypertrophic Cardiomyopathy algorithm |
References
- Krittanawong, C.; Virk, H.U.H.; Bangalore, S.; Wang, Z.; Johnson, K.W.; Pinotti, R.; Zhang, H.; Kaplin, S.; Narasimhan, B.; Kitai, T.; Baber, U.; Halperin, J.L.; Tang, W.H.W. Machine learning prediction in cardiovascular diseases: a meta-analysis. Sci Rep. 2020, 10(1), 16057. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Attia, Z.I.; Harmon, D.M.; Behr, E.R.; Friedman, P.A. Application of AI to the ECG. Eur Heart J 2021, 42(46), 4717–4730. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Somani, S.; Russak, A.J.; Richter, F.; Zhao, S.; Vaid, A.; Chaudhry, F.; De Freitas, J.K.; Naik, N.; Miotto, R.; Nadkarni, G.N.; Narula, J.; Argulian, E.; Glicksberg, B.S. Deep learning and the ECG: review of the current state-of-the-art. Europace 2021, 23(8), 1179–1191. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ose, B.; Sattar, Z.; Gupta, A.; Toquica, C.; Harvey, C.; Noheria, A. AI Interpretation of the ECG: A State-of-the-Art Review. Curr Cardiol Rep. 2024, 26(6), 561–580. [Google Scholar] [CrossRef] [PubMed]
- Siontis, K.C.; Noseworthy, P.A.; Attia, Z.I.; Friedman, P.A. AI-enhanced electrocardiography in cardiovascular disease management. Nat Rev Cardiol 2021, 18(7), 465–478. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kashou, A.H.; Adedinsewo, D.A.; Siontis, K.C.; Noseworthy, P.A. AI-Enabled ECG: Physiologic and Pathophysiologic Insights and Implications. Compr Physiol. 2022, 12(3), 3417–3424. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jain, S.S.; Elias, P.; Poterucha, T.; Randazzo, M.; Lopez Jimenez, F.; Khera, R.; Perez, M.; Ouyang, D.; Pirruccello, J.; Salerno, M.; Einstein, A.J.; Avram, R.; Tison, G.H.; Nadkarni, G.; Natarajan, V.; Pierson, E.; Beecy, A.; Kumaraiah, D.; Haggerty, C.; Avari Silva, J.N.; Maddox, T.M. AI in Cardiovascular Care-Part 2: Applications: JACC Review Topic of the Week. J Am Coll Cardiol. 2024, 83(24), 2487–2496. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rjoob, K.; Bond, R.; Finlay, D.; McGilligan, V.; Leslie, S.J.; Rababah, A.; Iftikhar, A.; Guldenring, D.; Knoery, C.; McShane, A.; Peace, A.; Macfarlane, P.W. Machine learning and the ECG over two decades: Time series and meta-analysis of the algorithms, evaluation metrics and applications. Artif Intell Med 2022, 132, 102381. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.W.; Olgin, J.E.; Avram, R.; Abreau, S.A.; Sittler, T.; Radia, K.; Hsia, H.; Walters, T.; Lee, B.; Gonzalez, J.E.; Tison, G.H. Performance of a Convolutional Neural Network and Explainability Technique for 12-Lead ECG Interpretation. JAMA Cardiol. 2021, 6(11), 1285–1295. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nolin-Lapalme, A.; Sowa, A.; Delfrate, J.; Tastet, O.; Corbin, D.; Kulbay, M.; Ozdemir, D.; Noël, M.J.; Marois-Blanchet, F.C.; Harvey, F.; Sharma, S.; Ansari, M.; Chiu, I.M.; D’souza, V.; Friedman, S.F.; Chassé, M.; Potter, B.J.; Afilalo, J.; Elias, P.A.; Jabbour, G.; Bahani, M.; Dubé, M.P.; Boyle, P.M.; Chatterjee, N.A.; Barrios, J.; Tison, G.H.; Ouyang, D.; Maddah, M.; Khurshid, S.; Cadrin-Tourigny, J.; Tadros, R.; Hussin, J.; Avram, R. Foundation models for ECG interpretation: clinical implications Epub ahead of print. Eur Heart J Erratum in: Eur Heart J. 2026 Mar 13:ehag139. doi: 10.1093/eurheartj/ehag139. PMID: 41568699. 2026, ehaf1119. [Google Scholar] [CrossRef] [PubMed]
- Ansari, Y.; Mourad, O.; Qaraqe, K.; Serpedin, E. Deep learning for ECG Arrhythmia detection and classification: an overview of progress for period 2017-2023. Front Physiol. 2023, 14, 1246746. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Attia, Z.; Noseworthy, P.; Lopez-Jimenez Fet, a.l. An AI-enabled ECG algorithm for the identification of patients with AF during sinus rhythm: a retrospective analysis of outcome prediction. The Lancet 2019, 394, 861–867. [Google Scholar] [CrossRef]
- Christopoulos, Georgios; Attia, Zachi I; Van Houten, Holly K; Yao, Xiaoxi; Carter, Rickey E; Lopez-Jimenez, Francisco; Kapa, Suraj; Noseworthy, Peter A; Friedman, Paul A. AI—electrocardiography to detect AF: trend of probability before and after the first episode. European Heart Journal - Digital Health 2022, 3(Issue 2), 228–235. [Google Scholar] [CrossRef]
- Raghunath, S.; Pfeifer, J.M.; Ulloa-Cerna, A.E.; Nemani, A.; Carbonati, T.; Jing, L.; vanMaanen, D.P.; Hartzel, D.N.; Ruhl, J.A.; Lagerman, B.F.; Rocha, D.B.; Stoudt, N.J.; Schneider, G.; Johnson, K.W.; Zimmerman, N.; Leader, J.B.; Kirchner, H.L.; Griessenauer, C.J.; Hafez, A.; Good, C.W.; Fornwalt, B.K.; Haggerty, C.M. Deep Neural Networks Can Predict New-Onset AF From the 12-Lead ECG and Help Identify Those at Risk of AF-Related Stroke. Circulation 2021, 143(13), 1287–1298. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Elias, P.; Jain, S.S.; Poterucha, T.; Randazzo, M.; Lopez Jimenez, F.; Khera, R.; Perez, M.; Ouyang, D.; Pirruccello, J.; Salerno, M.; Einstein, A.J.; Avram, R.; Tison, G.H.; Nadkarni, G.; Natarajan, V.; Pierson, E.; Beecy, A.; Kumaraiah, D.; Haggerty, C.; Avari Silva, J.N.; Maddox, T.M. AI for Cardiovascular Care-Part 1: Advances: JACC Review Topic of the Week. J Am Coll Cardiol. 2024, 83(24), 2472–2486. [Google Scholar] [CrossRef] [PubMed]
- Tison, G.H.; Sanchez, J.M.; Ballinger, B.; Singh, A.; Olgin, J.E.; Pletcher, M.J.; Vittinghoff, E.; Lee, E.S.; Fan, S.M.; Gladstone, R.A.; Mikell, C.; Sohoni, N.; Hsieh, J.; Marcus, G.M. Passive Detection of AF Using a Commercially Available Smartwatch. JAMA Cardiol 2018, 3(5), 409–416. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Perez, MV; Mahaffey, KW; Hedlin, H; Rumsfeld, JS; Garcia, A; Ferris, T; Balasubramanian, V; Russo, AM; Rajmane, A; Cheung, L; Hung, G; Lee, J; Kowey, P; Talati, N; Nag, D; Gummidipundi, SE; Beatty, A; Hills, MT; Desai, S; Granger, CB; Desai, M; Turakhia, M.P. Apple Heart Study Investigators Large-Scale Assessment of a Smartwatch to Identify, A.F. N Engl J Med 2019, 381(20), 1909–1917. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Trayanova, N.A.; Popescu, D.M.; Shade, J.K. Machine Learning in Arrhythmia and Electrophysiology. Circ Res. 2021, 128(4), 544–566. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hannun, A.Y.; Rajpurkar, P.; Haghpanahi, M.; Tison, G.H.; Bourn, C.; Turakhia, M.P.; Ng, A.Y. Cardiologist-level arrhythmia detection and classification in ambulatory ECGs using a deep neural network. Nat Med. Erratum in: Nat Med. 2019 Mar;25(3):530. doi: 10.1038/s41591-019-0359-9. PMID: 30617320; PMCID: PMC6784839. 2019, 25(1), 65–69. [Google Scholar] [CrossRef]
- Yao, X.; Rushlow, D.R.; Inselman, J.W.; et al. AI–enabled ECGs for identification of patients with low ejection fraction: a pragmatic, randomized clinical trial. Nat Med 2021, 27, 815–819. [Google Scholar] [CrossRef]
- Attia, Z.I.; Kapa, S.; Noseworthy, P.A.; Lopez-Jimenez, F.; Friedman, P.A. AI ECG to Detect LV Dysfunction in COVID-19: A Case Series. Mayo Clin Proc. 2020, 95(11), 2464–2466. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Attia, Z.I.; Kapa, S.; Lopez-Jimenez, F.; McKie, P.M.; Ladewig, D.J.; Satam, G.; Pellikka, P.A.; Enriquez-Sarano, M.; Noseworthy, P.A.; Munger, T.M.; Asirvatham, S.J.; Scott, C.G.; Carter, R.E.; Friedman, P.A. Screening for cardiac contractile dysfunction using an AI-enabled ECG. Nat Med. 2019, 25(1), 70–74. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.M.; Kim, K.H.; Jeon, K.H.; Kim, H.M.; Kim, M.J.; Lim, S.M.; Song, P.S.; Park, J.; Choi, R.K.; Oh, B.H. Development and Validation of Deep-Learning Algorithm for Electrocardiography-Based Heart Failure Identification. Korean Circ J 2019, 49(7), 629–639. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Demolder, A; Herman, R; Vavrik, B; Martonak, M; Boza, V; Herman, M; Palus, T; Kresnakova, V; Bahyl, J; Iring, A; Hatala, R; Bartunek, J; Vanderheyden, M; Heggermont, W; Penicka, M. A smartphone based AI model to detect LV systolic dysfunction on 12-lead ECG. European Heart Journal 2024, 45, ehae666.3477. [Google Scholar] [CrossRef]
- Desai, M.Y.; Jadam, S.; Abusafia, M.; Rutkowski, K.; Ospina, S.; Gaballa, A.; Sultana, S.; Thamilarasan, M.; Xu, B.; Popovic, Z.B. Real-World AI-Based Electrocardiographic Analysis to Diagnose Hypertrophic Cardiomyopathy. JACC Clin Electrophysiol Epub 2025 Mar 17. 2025, 11(6), 1324–1333. [Google Scholar] [CrossRef] [PubMed]
- Siontis, K.C.; Liu, K.; Bos, J.M.; Attia, Z.I.; Cohen-Shelly, M.; Arruda-Olson, A.M.; Zanjirani Farahani, N.; Friedman, P.A.; Noseworthy, P.A.; Ackerman, M.J. Detection of HCMby an AI ECG in children and adolescents. Int J Cardiol. 2021, 340, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Ko, W.Y.; Siontis, K.C.; Attia, Z.I.; Carter, R.E.; Kapa, S.; Ommen, S.R.; Demuth, S.J.; Ackerman, M.J.; Gersh, B.J.; Arruda-Olson, A.M.; Geske, J.B.; Asirvatham, S.J.; Lopez-Jimenez, F.; Nishimura, R.A.; Friedman, P.A.; Noseworthy, P.A. Detection of HCMUsing a Convolutional Neural Network-Enabled ECG. J Am Coll Cardiol. 2020, 75(7), 722–733. [Google Scholar] [CrossRef] [PubMed]
- Babur Guler, G.; Guler, A.; Surgit, O.; Turkmen, I.; Atmaca, S.; Sahin, H.; Pay, D.; Almasri, M.; Coskun, G.; Yartasi, U.; Salduz, D.; Kuru Gorgulu, B.; Aydin, S.; Serbest, N.G.; Turkvatan Cansever, A.; Tanboga, I.H. Evaluation of AI-based ECG analysis tools in patients with hypertrophic cardiomyopathy. Eur Heart J Digit Health 2026, 7(2), ztag026. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kittleson, M.; Ambardekar, A.; Cheng, R.; et al. Transthyretin Cardiac Amyloidosis Evaluation and Management: 2025 ACC Concise Clinical Guidance. JACC 2026, 87(5), 549–565. [Google Scholar] [CrossRef] [PubMed]
- Grogan, M; Lopez-Jimenez, F; Cohen-Shelly, M; Dispenzieri, A; Attia, ZI; Abou Ezzedine, OF; Lin, G; Kapa, S; Borgeson, DD; Friedman, PA; Murphree, DH, Jr. AI-Enhanced ECG for the Early Detection of Cardiac Amyloidosis. Mayo Clin Proc. Erratum in: Mayo Clin Proc. 2023 Jan;98(1):211. doi: 10.1016/j.mayocp.2022.10.002. PMID: 34218880. 2021, 96(11), 2768–2778. [Google Scholar] [CrossRef]
- Goto, S.; Mahara, K.; Beussink-Nelson, L.; et al. AI-enabled fully automated detection of cardiac amyloidosis using ECGs and echocardiograms. Nat Commun 2021, 12, 2726. [Google Scholar] [CrossRef]
- Kwon, J.M.; Kim, K.H.; Eisen, H.J.; Cho, Y.; Jeon, K.H.; Lee, S.Y.; Park, J.; Oh, B.H. AI assessment for early detection of HFpEFbased on electrocardiographic features. Eur Heart J Digit Health Erratum in: Eur Heart J Digit Health. 2021 Nov 21;3(1):115-116. doi: 10.1093/ehjdh/ztab098. PMID: 36711179; PMCID: PMC9707919. 2020, 2(1), 106–116. [Google Scholar] [CrossRef]
- Lee, MS; Shin, TG; Lee, Y; Kim, DH; Choi, SH; Cho, H; Lee, MJ; Jeong, KY; Kim, WY; Min, YG; Han, C; Yoon, JC; Jung, E; Kim, WJ; Ahn, C; Seo, JY; Lim, TH; Kim, JS; Choi, J; Kwon, JM; Kim, K. ROMIAEstudy group AIapplied to ECGto rule out acute myocardial infarction: the ROMIAEmulticentre study Eur Heart, J. 2025, 46(20), 1917–1929. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Herman, R.; Meyers, H.P.; Smith, S.W.; Bertolone, D.T.; Leone, A.; Bermpeis, K.; Viscusi, M.M.; Belmonte, M.; Demolder, A.; Boza, V.; Vavrik, B.; Kresnakova, V.; Iring, A.; Martonak, M.; Bahyl, J.; Kisova, T.; Schelfaut, D.; Vanderheyden, M.; Perl, L.; Aslanger, E.K.; Hatala, R.; Wojakowski, W.; Bartunek, J.; Barbato, E. International evaluation of an AI-powered ECG model detecting acute coronary occlusion myocardial infarction. Eur Heart J Digit Health 2023, 5(2), 123–133. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Herman, R.; Mumma, B.E.; Hoyne, J.D.; Cooper, B.L.; Johnson, N.P.; Kisova, T.; Demolder, A.; Rafajdus, A.; Iring, A.; Palus, T.; Belmonte, M.; Barbato, E.; Baron, S.J.; Hatala, R.; Smith, S.W.; Meyers, H.P.; Sharkey, S.W.; Bartunek, J.; Henry, T.D. AI-Enabled ECG Analysis Improves Diagnostic Accuracy and Reduces False STEMI Activations: A Multicenter U.S. Registry. JACC Cardiovasc Interv. 2026, 19(2), 145–156. [Google Scholar] [CrossRef] [PubMed]
- Khera, R.; Oikonomou, E.K.; Nadkarni, G.N.; Morley, J.R.; Wiens, J.; Butte, A.J.; Topol, E.J. Transforming Cardiovascular Care With AI: From Discovery to Practice: JACC State-of-the-Art Review. J Am Coll Cardiol. 2024, 84(1), 97–114. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kwon, J.M.; Kim, K.H.; Jeon, K.H.; Lee, S.E.; Lee, H.Y.; Cho, H.J.; Choi, J.O.; Jeon, E.S.; Kim, M.S.; Kim, J.J.; Hwang, K.K.; Chae, S.C.; Baek, S.H.; Kang, S.M.; Choi, D.J.; Yoo, B.S.; Kim, K.H.; Park, H.Y.; Cho, M.C.; Oh, B.H. AI algorithm for predicting mortality of patients with acute heart failure. PLoS One 2019, 14(7), e0219302. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lima, E.M.; Ribeiro, A.H.; Paixão, G.M.M.; Ribeiro, M.H.; Pinto-Filho, M.M.; Gomes, P.R.; Oliveira, D.M.; Sabino, E.C.; Duncan, B.B.; Giatti, L.; Barreto, S.M.; Meira WJr Schön, T.B.; Ribeiro, A.L.P. Deep neural network-estimated electrocardiographic age as a mortality predictor. Nat Commun 2021, 12(1), 5117. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Raghunath, S.; Ulloa Cerna, A.E.; Jing, L.; vanMaanen, D.P.; Stough, J.; Hartzel, D.N.; Leader, J.B.; Kirchner, H.L.; Stumpe, M.C.; Hafez, A.; Nemani, A.; Carbonati, T.; Johnson, K.W.; Young, K.; Good, C.W.; Pfeifer, J.M.; Patel, A.A.; Delisle, B.P.; Alsaid, A.; Beer, D.; Haggerty, C.M.; Fornwalt, B.K. Prediction of mortality from 12-lead ECG voltage data using a deep neural network. Nat Med. 2020, 26(6), 886–891. [Google Scholar] [CrossRef] [PubMed]
- Spatz, E.S.; Ginsburg, G.S.; Rumsfeld, J.S.; Turakhia, M.P. Wearable Digital Health Technologies for Monitoring in Cardiovascular Medicine. N Engl J Med. 2024, 390(4), 346–356. [Google Scholar] [CrossRef] [PubMed]
- Barrera, N.; Solorzano, M.; Jimenez, Y.; Kushnir, Y.; Gallegos-Koyner, F.; Dagostin de Carvalho, G. Accuracy of Smartwatches in the Detection of AF: A Systematic Review and Diagnostic Meta-Analysis. JACC Adv. 2025, 4 11 Pt 1, 102133. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Attia, Z.I.; Harmon, D.M.; Behr, E.R.; Friedman, P.A. Application of AI to the ECG. Eur Heart J 2021, 42(46), 4717–4730. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Krittanawong, C.; Zhang, H.; Wang, Z.; Aydar, M.; Kitai, T. AI in Precision Cardiovascular Medicine. J Am Coll Cardiol. 2017, 69(21), 2657–2664. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.; Slomka, P.J.; Leeson, P.; Comaniciu, D.; Shrestha, S.; Sengupta, P.P.; Marwick, T.H. AI in Cardiovascular Imaging: JACC State-of-the-Art Review. J Am Coll Cardiol. 2019, 73(11), 1317–1335. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Moosavi, A.; Huang, S.; Vahabi, M.; Motamedivafa, B.; Tian, N.; Mahmood, R.; Liu, P.; Sun, C.L.F. Prospective Human Validation of AI Interventions in Cardiology: A Scoping Review. JACC Adv. 2024, 3(9), 101202. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Quer, G.; Arnaout, R.; Henne, M.; Arnaout, R. Machine Learning and the Future of Cardiovascular Care: JACC State-of-the-Art Review. J Am Coll Cardiol. 2021, 77(3), 300–313. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Karatzia, L.; Aung, N.; Aksentijevic, D. AI in cardiology: Hope for the future and power for the present. Front Cardiovasc Med. 2022, 9, 945726. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Armoundas, AA; Narayan, SM; Arnett, DK; Spector-Bagdady, K; Bennett, DA; Celi, LA; Friedman, PA; Gollob, MH; Hall, JL; Kwitek, AE; Lett, E; Menon, BK; Sheehan, KA; Al-Zaiti, SS. American Heart Association Institute for Precision Cardiovascular Medicine; Council on Cardiovascular and Stroke Nursing; Council on Lifelong Congenital Heart Disease and Heart Health in the Young; Council on Cardiovascular Radiology and Intervention; Council on Hypertension; Council on the Kidney in Cardiovascular Disease; and Stroke Council. Use of AI in Improving Outcomes in Heart Disease: A Scientific Statement From the American Heart Association. Circulation 2024, 149(14), e1028–e1050. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| Study | Population | AI model | ECG type | Task | Main results | Clinical implication |
|---|---|---|---|---|---|---|
| Attia et al., 2019 [12] | 180,922 patients; 649,931 sinus-rhythm ECGs | Convolutional neural network (CNN) | Standard 10-s 12-lead ECG | Detection of AF during sinus rhythm | AUC 0.87 (single ECG); sensitivity 79.0%; specificity 79.5%; accuracy 79.4%. Using multiple ECGs: AUC 0.90, sensitivity 82.3%, specificity 83.4% | Enables identification of occult or future AF from a single routine ECG, supporting opportunistic screening and selection of patients for prolonged rhythm monitoring |
| Christopoulos et al., 2022 [13] | 59,212 patients; 957,816 ECGs before and after first AF episode | Convolutional neural network (CNN) | Standard 10-s 12-lead ECG | Temporal prediction of AF onset | AI-ECG probability progressively increased up to years before first AF episode (19.8% at 2–5 years prior → 34.0% within 3 months prior); mean interval between first positive AI-ECG and AF diagnosis: 5.4 years | Serial AI-ECG assessment may identify patients at increasing risk of AF years before clinical onset and guide targeted rhythm monitoring strategies |
| Raghunath et al., 2021 [14] | 1.6 million resting 12-lead ECGs from ~430,000 adult patients without prior atrial fibrillation (Geisinger health system cohort) | Deep neural network (convolutional deep learning model) | Standard 10-s 12-lead ECG | Prediction of new-onset AF within 1 year and identification of patients at risk of AF-related stroke | AUC 0.85 for prediction of incident AF within 1 year; sensitivity 69%, specificity 81% in simulated deployment scenario; hazard ratio 7.2 for long-term AF risk in high- vs low-risk groups; model identified 62% of patients who developed AF-related stroke within 3 years before stroke occurrence | Supports use of AI-ECG for early identification of patients at risk for incident AF and AF-related stroke, enabling targeted rhythm monitoring and preventive anticoagulation strategies in high-risk populations |
| Tison et al., 2018 [16] | 9,750 participants (Health eHeart cohort); external validation in 51 cardioversion patients; exploratory ambulatory cohort 1,617 subjects | Deep neural network | Smartwatch photoplethysmography (heart rate + step count data) | Passive detection of AF from wearable data | External validation vs 12-lead ECG: AUC 0.97, sensitivity 98.0%, specificity 90.2%; ambulatory detection: AUC 0.72, sensitivity 67.7%, specificity 67.6% | Demonstrates feasibility of passive large-scale AF detection using commercially available smartwatches, supporting wearable-based population screening strategies |
| Perez et al., 2019 (Apple Heart Study) [17] | 419,297 smartwatch users monitored for median 117 days | Irregular pulse detection algorithm (photoplethysmography-based) | Smartwatch PPG signal with confirmatory ECG patch | Large-scale population screening for AF | Irregular pulse notifications in 0.52% overall (3.14% ≥65 years); AF confirmed in 34% of participants returning ECG patch; notification PPV 0.84; tachogram PPV 0.71; 43% of notified participants reported new AF diagnosis vs 1.0% in non-notified | Demonstrates feasibility of large-scale digital AF screening using consumer wearable devices and supports integration of smartwatch-based alerts into diagnostic pathways |
| Study | Population | AI model | ECG type | Task | Main results | Clinical implication |
|---|---|---|---|---|---|---|
| Yao et al., 2021 (EAGLE trial) [20] | 22,641 adult patients from 120 primary care teams | Convolutional neural network (AI-ECG) | Standard 12-lead ECG | Detection of reduced left LVEF ≤50% in routine clinical practice | New diagnosis of low LVEF within 90 days: 2.1% vs 1.6% (AI vs usual care; OR 1.32); in AI-positive patients: 19.5% vs 14.5% (OR 1.43); echocardiography use higher in AI-positive group (49.6% vs 38.1%) | Demonstrates that integration of AI-ECG Demonstrates that integration of AI-ECG into clinical workflows improves early detection of asymptomatic LV dysfunction in real-world primary care settings |
| Attia et al., 2020 [21] | 27 COVID-19 patients with ECG and echocardiography within 14 days | Convolutional neural network (AI-ECG) | Standard 12-lead ECG | Detection of LV dysfunction (LVEF ≤40%) in COVID-19 patients | AUC 0.95 for identification of LVEF ≤40%; LV dysfunction detected in 3 patients (11.1%); algorithm identified myocarditis-related LVEF reduction with predicted probability 90.2% corresponding to echocardiographic LVEF 35% | Supports feasibility of AI-ECG as rapid bedside screening tool for ventricular dysfunction in COVID-19, potentially reducing need for immediate echocardiography in high-risk infectious settings |
| Attia et al., 2019 [22] | 97,829 patients with paired ECG–echocardiogram within 2 weeks (test set: 52,870) | Convolutional neural network (CNN) | Standard 12-lead ECG | Detection of asymptomatic LV systolic dysfunction (LVEF ≤35%) | AUC 0.93; sensitivity 86.3%; specificity 85.7%; accuracy 85.7%. Patients with positive AI-ECG but normal LVEF had 4-fold higher risk of developing future LV dysfunction (HR 4.1) | Demonstrates feasibility of AI-ECG as a population-level screening tool for asymptomatic LV dysfunction and early identification of subjects at risk for future heart failure |
| Kwon et al., 2019 [23] | 55,163 ECGs from 22,765 patients across 2 hospitals | Deep neural network | Standard 12-lead ECG + demographic features | Detection of HFrEF (LVEF ≤40%) and LVEF ≤50% | AUC 0.843 (internal) and 0.889 (external) for LVEF ≤40%; AUC 0.821 and 0.850 for LVEF ≤50%; performance superior to logistic regression and random forest (p<0.001) | Demonstrates feasibility of ECG-based deep-learning screening for heart failure using routine clinical parameters, enabling early identification of patients requiring echocardiographic evaluation |
| Demolder et al., 2024 [24] | 109,809 paired ECG–echocardiogram datasets (56,236 patients); validation cohort: 25,510 ECG–TTE pairs | Deep learning AI-ECG integrated with smartphone ECG digitization | Standard 12-lead ECG (smartphone-based acquisition/digitization) | Detection of reduced LVEF (≤40% and <50%) | For LVEF ≤40%: AUC 0.963, sensitivity 0.924, specificity 0.887, NPV 0.995; for LVEF <50%: AUC 0.952, sensitivity 0.899, specificity 0.875, NPV 0.99 | Demonstrates feasibility of smartphone-based AI-ECG screening for LV systolic dysfunction, enabling scalable point-of-care identification of patients requiring echocardiographic evaluation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




