Submitted:
12 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
2.1. Instrumentation and Chemicals Used in the Present Work
2.2. Selection of Detection Wavelength and Preparation of Solutions
2.3. Method Validation
3. Results and Discussion

4. Conclusion
References
- Gioumouxouzis, C.I.; Eleftheriadis, G.K.; Kyriakidis, A.S.; Karavasili, C. Translation of pharmaceutical 3D printing to clinical point-of-care and industrial manufacturing. In Drug Delivery and Translational Research; 2026; pp. 1–14. [Google Scholar]
- Carou-Senra, P.; Rodríguez-Pombo, L.; Alvarez-Lorenzo, C.; Goyanes, A. Accelerating dataset generation for machine learning using large language models: a pharmaceutical additive manufacturing case. International Journal of Pharmaceutics 2026, 126587. [Google Scholar] [CrossRef] [PubMed]
- Mascareñas-García, M.; Rivero-de-Aguilar, A.; Candal-Pedreira, C.; García, G.; Guerra-Tort, C.; Martín-Gisbert, L.; Rey-Brandariz, J.; Ríos, M.P.R.; Casal-Acción, B.; Santiago-Pérez, M.I.; Varela-Lema, L. Distortion in the communication of nonsignificant primary outcomes: the spin strategy in multiple sclerosis trials. Annals of Neurology 2026. [Google Scholar] [CrossRef] [PubMed]
- Zhavoronkov, A.; Gennert, D.; Shi, J. From Prompt to Drug: Toward Pharmaceutical Superintelligence. In ACS Central Science; 2026. [Google Scholar]
- Tanga, S.; Ramburrun, P.; Aucamp, M. From Liquid SNEDDS to Solid SNEDDS: A Comprehensive Review of Their Development and Pharmaceutical Applications. The AAPS Journal 2026, 28(1), 1–18. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ramírez, V.I.; Varela-Rodríguez, H.; Varela-Rodríguez, L.; Sierra-López, F.; San Juan-Mora, D.E.; Morales-Mora, J.D.; Falcón-Navarrete, D.; Osorio-Trujillo, C.; Ríos-López, J.; Rodríguez-Mera, I.B.; Carrasco-Yépez, M.M. Acanthamoeba castellanii: Non-Steroidal Anti-Inflammatory Drugs Affect Adhesion, Motility, and Encystment, Suggesting a Link with a gp63-like Protein Candidate. Pathogens 2026, 15(3), 263. [Google Scholar] [CrossRef] [PubMed]
- Hayat, A.; Khanem, A.; Ullah, I.; Riaz, M.; Younas, F. Reversion of multidrug resistance in clinical isolates of Escherichia coli using non-steroidal anti-inflammatory drugs (NSAIDs). The Journal of Antibiotics 2026, 79(2), 110–121. [Google Scholar] [CrossRef] [PubMed]
- Hesham, K.; Aboulthana, W.M.; Ragab, A. Development of novel salicylic acid derivatives with dual anti-inflammatory and anti-arthritic potentials: synthesis, in vitro bio-evaluation, and in silico toxicity prediction with molecular modeling simulations. RSC advances 2026, 16(4), 3509–3526. [Google Scholar] [CrossRef] [PubMed]
- Peng, K.P.; Chang, K.H. Long-term non-steroidal anti-inflammatory drug (NSAID) use and dementia or cognitive decline risk in adults over 50 based on epidemiological studies: A narrative review. In Tungs’ Medical Journal; pp. 10–4103.
- Shaikh, F.; Uzgare, A. HIGH-PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC): PRINCIPLE, APPLICATIONS, AND THE ROLE OF ARTIFICIAL INTELLIGENCE–A REVIEW. Journal of microbiology, biotechnology and food sciences 2026, 15(4), e13541–e13541. [Google Scholar] [CrossRef]
- Halko, R.; Pavelek, D.; Kaykhaii, M. High performance liquid chromatography-fourier transform infrared spectroscopy coupling: a comprehensive review. Critical Reviews in Analytical Chemistry 2026, 56(1), 167–178. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Zhang, L.; Wang, L. Quantification of Protochlorophyllide (Pchlide) Content in Arabidopsis Seedlings Using a High-Performance Liquid Chromatography (HPLC) System. Bio-protocol 2026, 16(1). [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Cen, G.; Zhou, H.; Zhang, C.; Wang, L.; Su, L.; Gu, W.; Pan, H.; Li, Y. Locked Nucleic Acid-Modified Parallel Entropy-Driven Circuit Integrated with High-Performance Liquid Chromatography for Sensitive, Automated, and Multiplexed Detection of Breast Cancer-Related MicroRNAs. Sensors and Actuators B: Chemical 2026, 139545. [Google Scholar] [CrossRef]
- Xu, D.; Yan, D.; Zhang, C.; Sun, Y.; Wang, R.; Yang, L.; Xing, R.; Hu, S.; Chen, X. A novel biomimetic nanovesicle-based microextraction combined with high performance liquid chromatography for the determination of flavonoids in Huangqisan. Microchemical Journal 2026, 117151. [Google Scholar] [CrossRef]
- Machireddy, N.R.; Katam, K. Pharmaceutical Ingredients (API) Industry. Sustainable Advanced Technologies for Industrial Pollution Control: Proceedings of ATIPC 2026, 277. [Google Scholar]
- Tareq, A.Z.; Hyder, M.; Chippindale, A.M.; Spencer, N.; Mohan, S.; Kaur, A.; Hallett, J.; Zinn, T.; Shankland, K.; Hayes, W. Low-molecular weight organogel matrices as crystallisation media for active pharmaceutical ingredients. Journal of Materials Chemistry B 2026. [Google Scholar] [CrossRef] [PubMed]
- Romano, S.; Damian, M.; Nardi, M.; Procopio, A.; Strähler, S.; Preschel, D.; Oliverio, M.; Mutti, F.G. Stereoselective Bio-Organocatalytic Cascade to Chiral Amides as Active Pharmaceutical Ingredient Intermediates Using ω-Transaminase and Choline Chloride Under Microwave Irradiation. ChemSusChem 2026, 19(6), e202501501. [Google Scholar] [CrossRef] [PubMed]




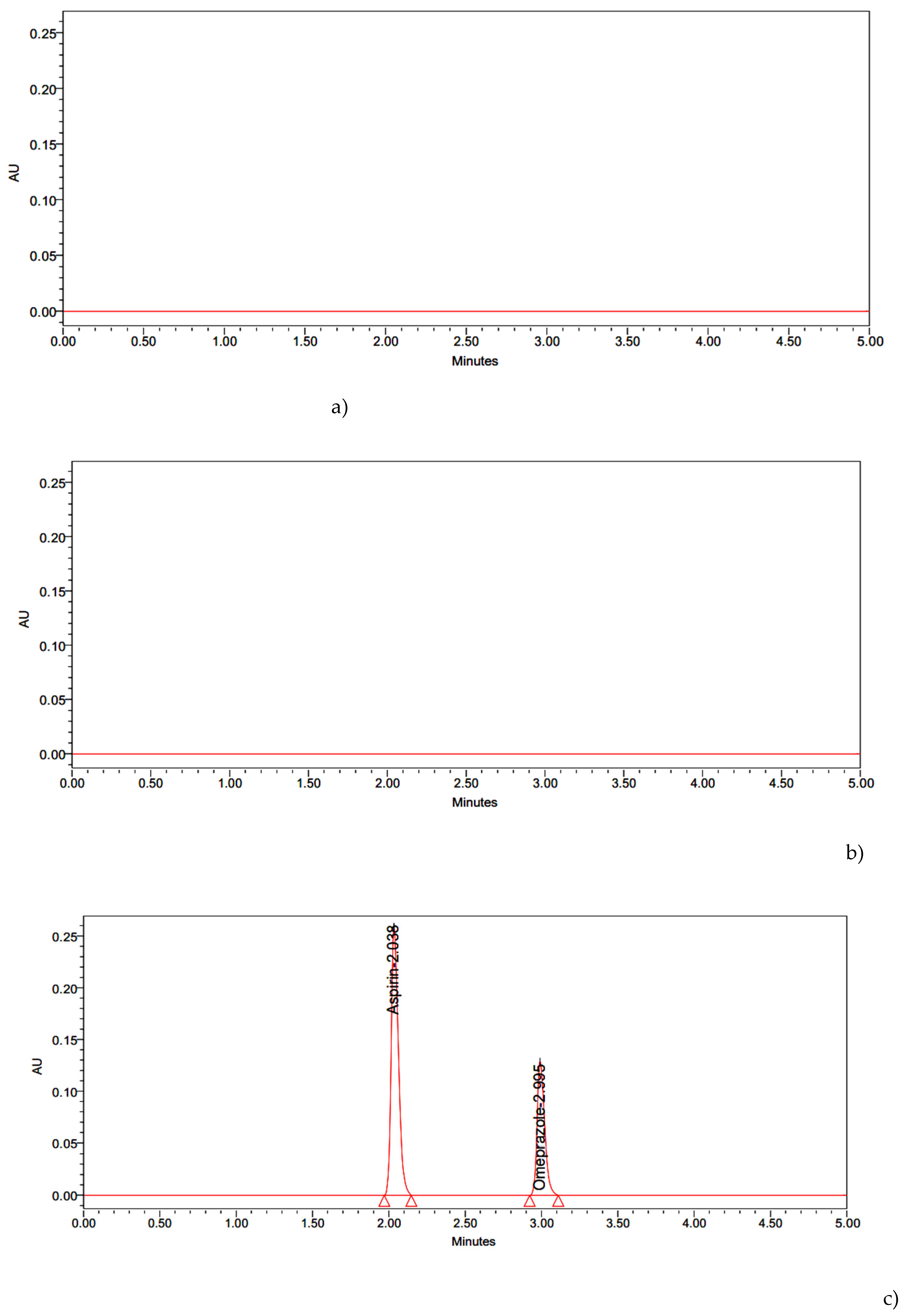
| S.No | Instrument | Model / Specification | Manufacturer |
| 1 | HPLC System | Waters Alliance e2695 | Waters |
| 2 | pH Meter | pH700 | Eutech |
| 3 | Weighing balance | BSA224S-CW | Sartorius |
| 4 | Ultrasonicator | UCA 701 | Unichrome |
| 5 | Glassware | Class A | Borosil |
| S.No | Chemical | Grade | Manufacturer |
| 1 | Acetonitrile | HPLC | Merck |
| 2 | Water (Milli-Q) | HPLC | In-house |
| 3 | Perchloric Acid | AR | Merck |
| Parameter | Condition |
| Column | Luna Phenyl Hexyl (250 × 4.6 mm, 5 µm) |
| Mobile Phase | Acetonitrile : 0.1% Perchloric acid (20:80) |
| Flow rate | 1.0 mL/min |
| Detection Wavelength | 249 nm |
| Injection Volume | 10 µL |
| Run Time | 5 min |
| Parameters | Observation |
| Instrument used | Waters HPLC with auto sampler and PDA detector. |
| Injection volume | 10µl |
| Mobile Phase | Acetonitrile: 0.1% Perchloric acid (20:80) |
| Column | Luna Phenyl Hexyl (250x4.6mm, 5µm) |
| Detection Wave Length | 249nm |
| Flow Rate | 1 mL/min |
| Runtime | 5min |
| Temperature | Ambient(25° C) |
| Mode of separation | Isocratic mode |
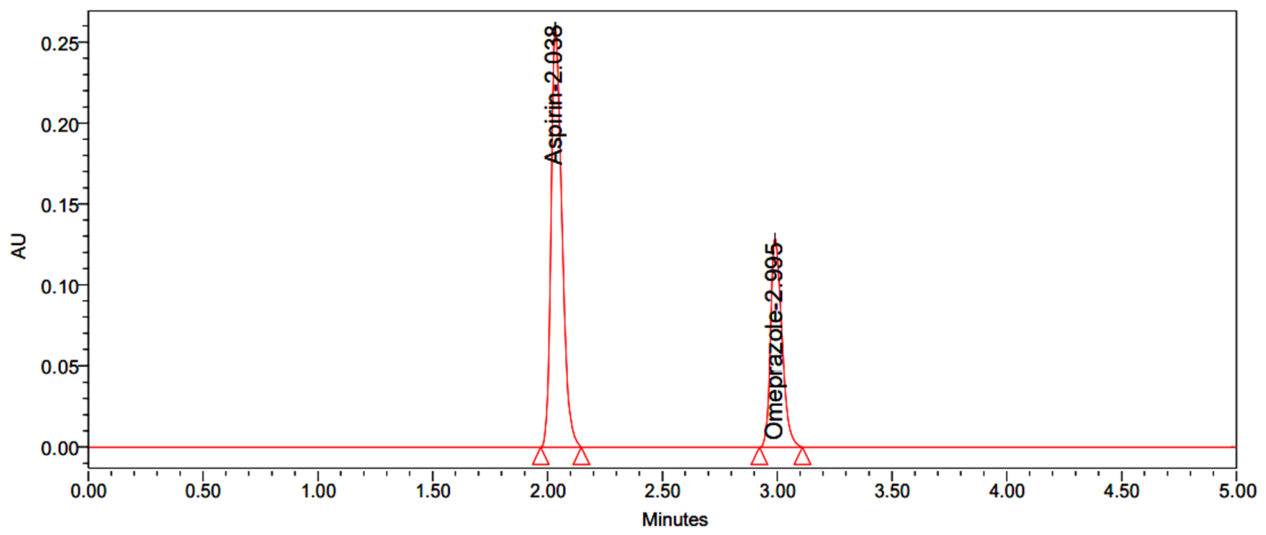
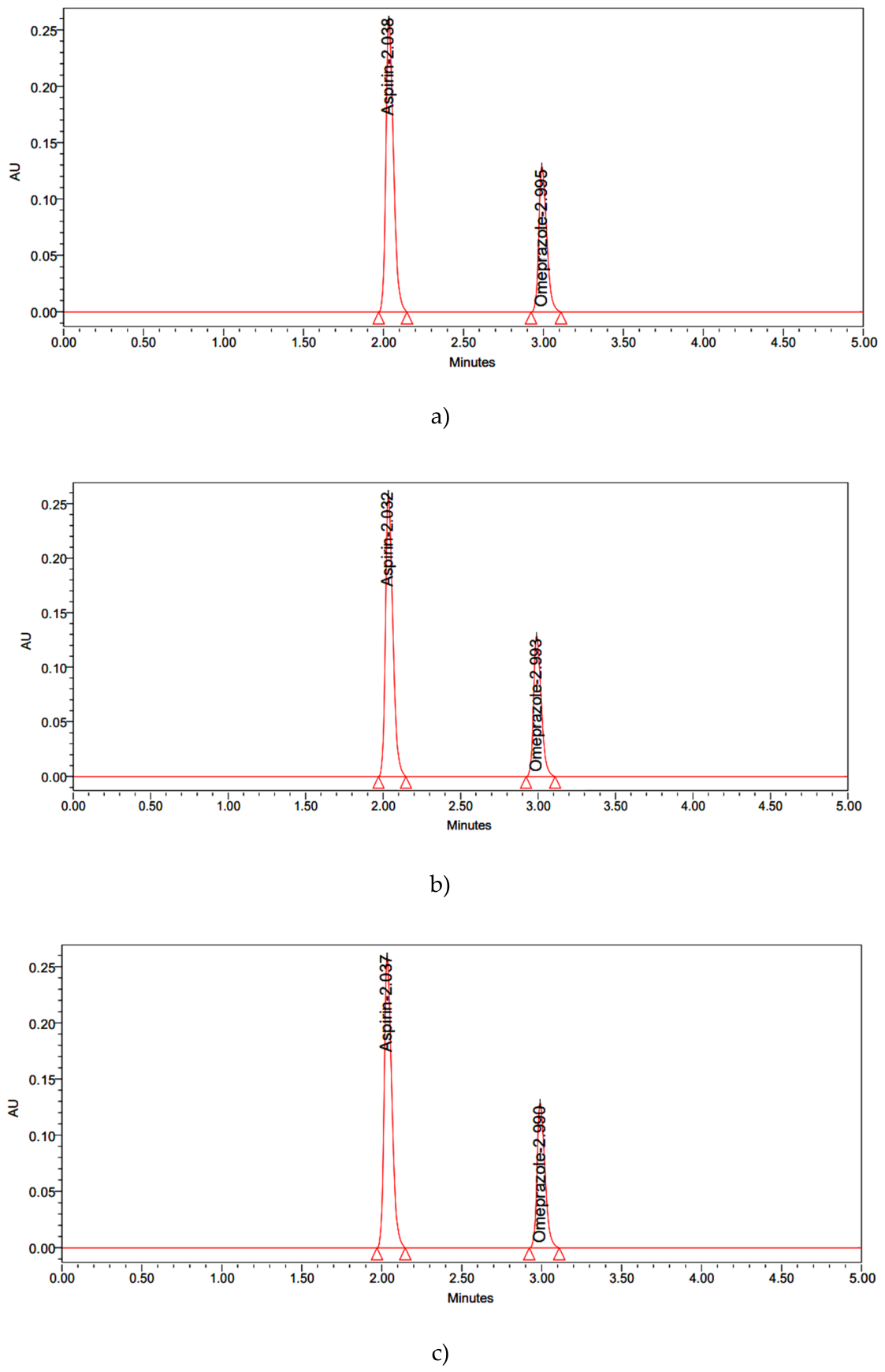
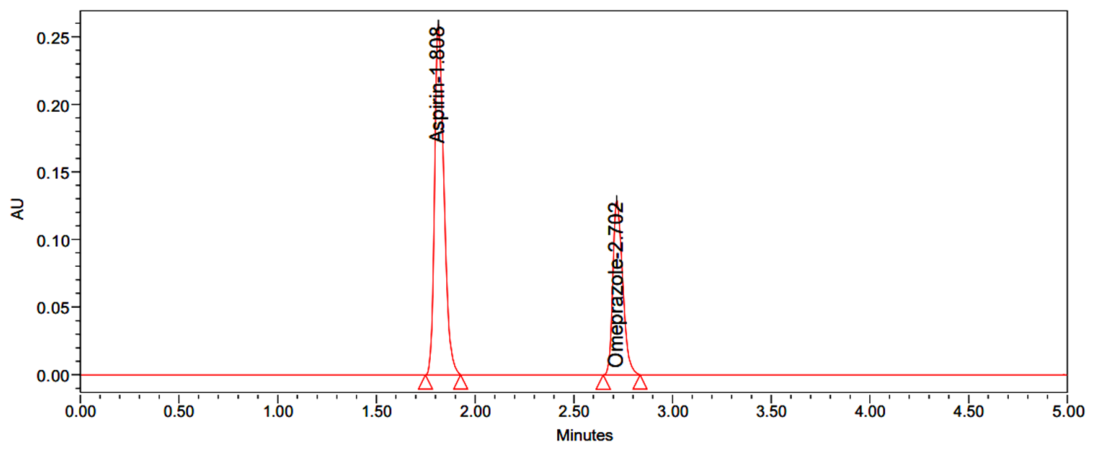
| Name | RT | Area | USP Resolution | USP Tailing | USP Plate Count |
| Aspirin | 2.038 | 2726427 | - | 0.97 | 11485 |
| Omeprazole | 2.995 | 1282031 | 4.63 | 1.03 | 7693 |
| S.no | Parameter | Aspirin | Omeprazole |
| 1 | Retention time | 2.038 | 2.995 |
| 2 | Plate count | 11485 | 7693 |
| 3 | Tailing factor | 0.97 | 1.03 |
| 4 | Resolution | ---- | 4.63 |
| 5 | %RSD | 0.17 | 0.42 |
| ConcentrationAspirin(µg/ml) |
Area of Aspirin |
Concentration of Omeprazole (µg/ml) |
Area of Omeprazole |
|
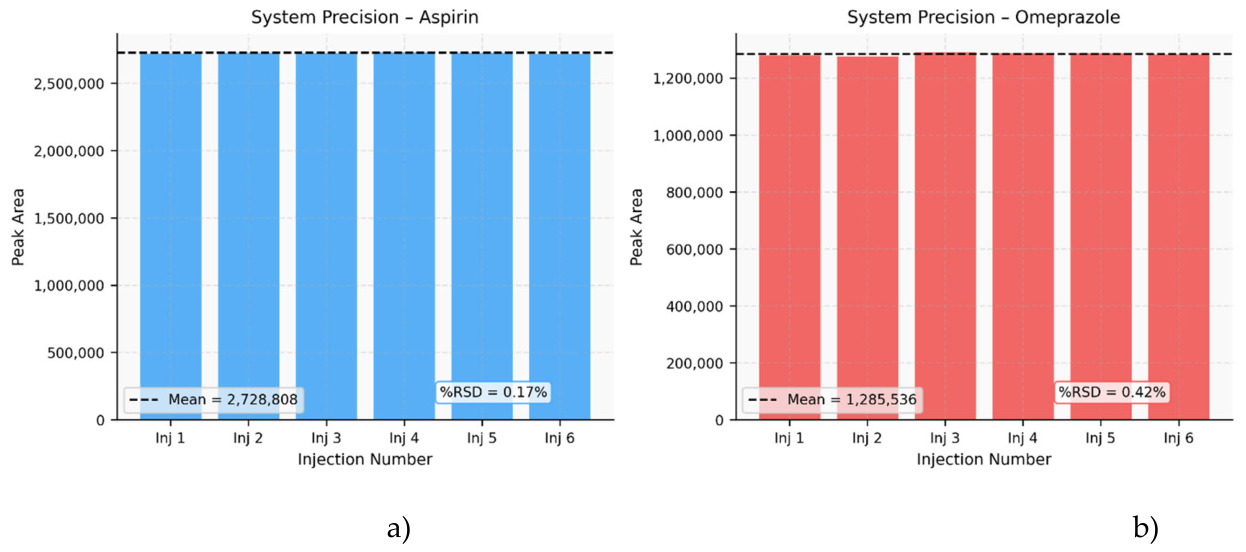
| 1. | 81 | 2726427 | 40 | 1282031 |
| 2. | 81 | 2732136 | 40 | 1277094 |
| 3. | 81 | 2728974 | 40 | 1291557 |
| 4. | 81 | 2733127 | 40 | 1289461 |
| 5. | 81 | 2731329 | 40 | 1288950 |
| 6. | 81 | 2720852 | 40 | 1284123 |
| Mean | 2728808 | 1285536 | ||
| S.D | 4584.31 | 5456.42 | ||
| %RSD | 0.17 | 0.42 | ||
| S. No. | Area for Aspirin | Area for Omeprazole |
| 1 | 2746531 | 1272322 |
| 2 | 2721402 | 1291542 |
| 3 | 2715620 | 1285690 |
| 4 | 2703102 | 1290972 |
| 5 | 2725876 | 1286541 |
| 6 | 2711172 | 1269003 |
| Average | 2720617 | 1282678 |
| STD Dev | 14971.720 | 9649.955 |
| %RSD | 0.55 | 0.75 |
| S. No. | Area | |
| Aspirin | Omeprazole | |
| 1 | 2734860 | 1278683 |
| 2 | 2714532 | 1280152 |
| 3 | 2706130 | 1269887 |
| 4 | 2715613 | 1293543 |
| 5 | 2721584 | 1286791 |
| 6 | 2727625 | 1290956 |
| Average | 2720057 | 1283335 |
| Standard Deviation | 10227.133 | 8796.001 |
| %RSD | 0.38 | 0.69 |
| S.NO | Aspirin | Omeprazole | ||
| Conc.(µg/ml) | Peak area | Conc.(µg/ml) | Peak area | |
| 1 | 20.25 | 724355 | 10.00 | 331897 |
| 2 | 40.50 | 1389604 | 20.00 | 663072 |
| 3 | 60.75 | 2013896 | 30.00 | 962431 |
| 4 | 81.00 | 2728462 | 40.00 | 1286541 |
| 5 | 101.25 | 3425137 | 50.00 | 1602253 |
| 6 | 121.50 | 4141720 | 60.00 | 1889428 |
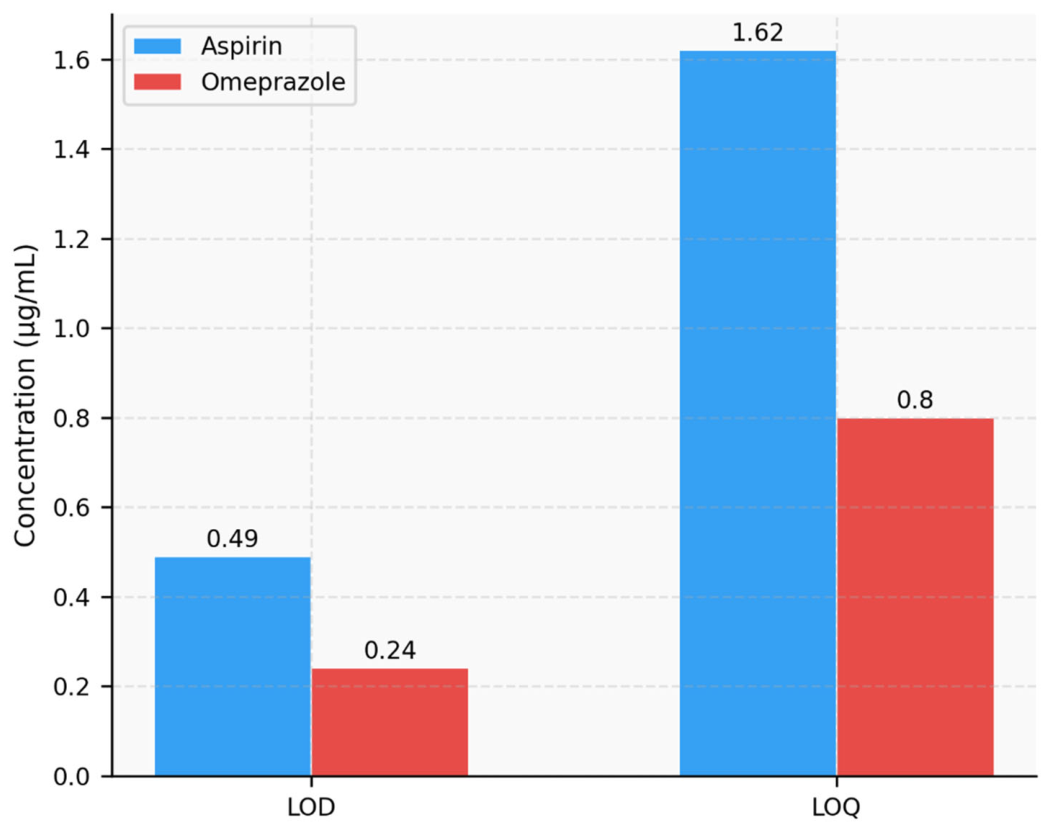
| Regression equation | y = 33801.73x +6998.21 | y =12908.70x + 16825.71 | ||
| Slope | 33801.73 | 31544.52 | ||
| Intercept | 6998.21 | 15896.18 | ||
| R2 | 0.99983 | 0.99981 | ||
| %Concentration(at specification Level) | Area |
Amount Added (mg) |
Amount Found (mg) |
% Recovery | Mean Recovery |
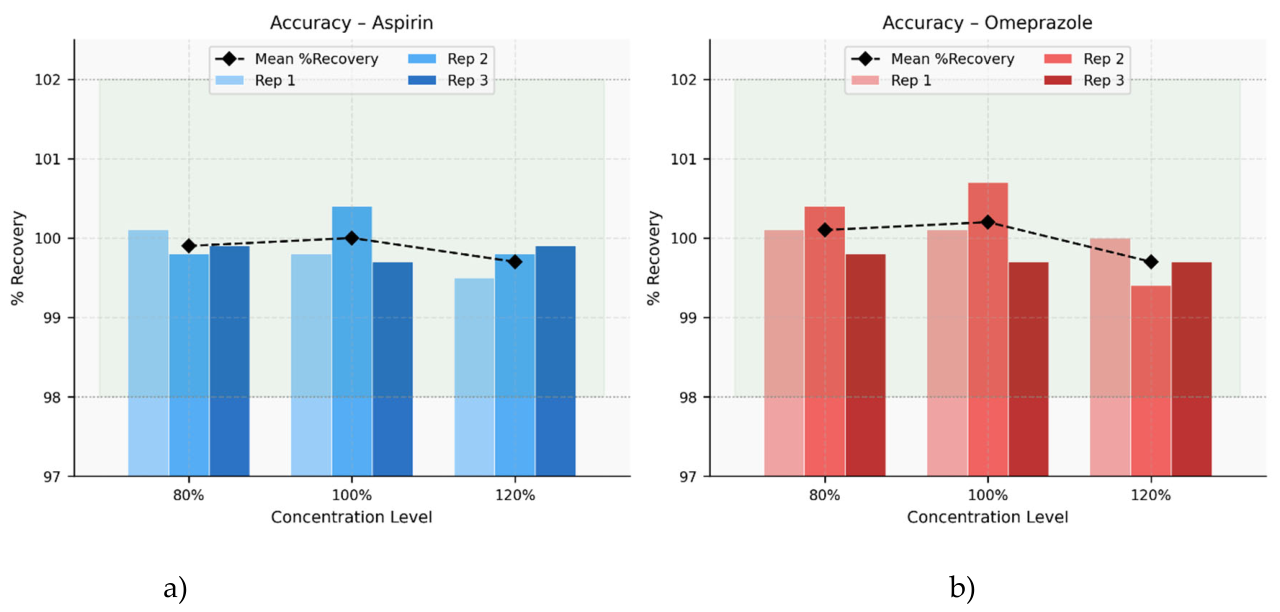
| 80% | 4915263 | 14.58 | 14.59 | 100.1 | 99.9 |
| 4903187 | 14.58 | 14.55 | 99.8 | ||
| 4909866 | 14.58 | 14.57 | 99.9 | ||
| 100% | 5446318 | 16.20 | 16.17 | 99.8 | 100.0 |
| 5478749 | 16.20 | 16.26 | 100.4 | ||
| 5440612 | 16.20 | 16.15 | 99.7 | ||
| 120% | 5972161 | 17.82 | 17.73 | 99.5 | 99.7 |
| 5991320 | 17.82 | 17.78 | 99.8 | ||
| 5996224 | 17.82 | 17.80 | 99.9 |
|
%Concentration (at specification Level) |
Area |
Amount Added (mg) |
Amount Found (mg) |
% Recovery | Mean Recovery |
| 80% | 2316850 | 7.2 | 7.21 | 100.1 | 100.1 |
| 2323568 | 7.2 | 7.23 | 100.4 | ||
| 2308191 | 7.2 | 7.18 | 99.8 | ||
| 100% | 2574813 | 8.0 | 8.01 | 100.1 | 100.2 |
| 2590255 | 8.0 | 8.06 | 100.7 | ||
| 2562161 | 8.0 | 7.97 | 99.7 | ||
| 120% | 2828913 | 8.8 | 8.80 | 100.0 | 99.7 |
| 2812106 | 8.8 | 8.75 | 99.4 | ||
| 2820465 | 8.8 | 8.78 | 99.7 |
| Parameter | Aspirin | ||||||
| Condition | Retention time(min) | Peak area | Resolution | Tailing | Plate count | % RSD | |
| Flow rate Change (mL/min) |
Less flow (0.9ml) | 2.207 | 2624513 | - | 1.01 | 11142 | 0.57 |
| Actual (1.0ml) | 2.038 | 2726427 | - | 0.97 | 11485 | 0.17 | |
| More flow (1.1ml) | 1.914 | 2916432 | - | 0.93 | 11644 | 0.53 | |
| Organic Phase change | Less Org (18:82) | 2.375 | 2481793 | - | 1.10 | 10813 | 0.31 |
| Actual (20:80) | 2.032 | 2732136 | - | 0.92 | 11413 | 0.17 | |
| More Org (22:78) | 1.808 | 3088792 | - | 0.88 | 11814 | 0.41 | |
| Parameter | Omeprazole | ||||||
| Condition | Retention time(min) | Peak area | Resolution | Tailing | Plate count | %RSD | |
| Flow rate Change (mL/min) |
Less flow (0.9ml) | 3.135 | 1126354 | 4.32 | 1.12 | 7429 | 0.50 |
| Actual (1.0ml) | 2.995 | 1282031 | 4.63 | 1.03 | 7693 | 0.42 | |
| More flow (1.1ml) | 2.847 | 1436211 | 4.38 | 0.97 | 7978 | 0.73 | |
| Organic Phase change | Less Org (18:82) | 3.263 | 1028562 | 3.91 | 1.18 | 7107 | 0.99 |
| Actual (20:80) | 2.993 | 1277094 | 4.65 | 1.05 | 7677 | 0.42 | |
| More Org (22:78) | 2.702 | 1589456 | 4.09 | 0.94 | 8276 | 1.12 | |
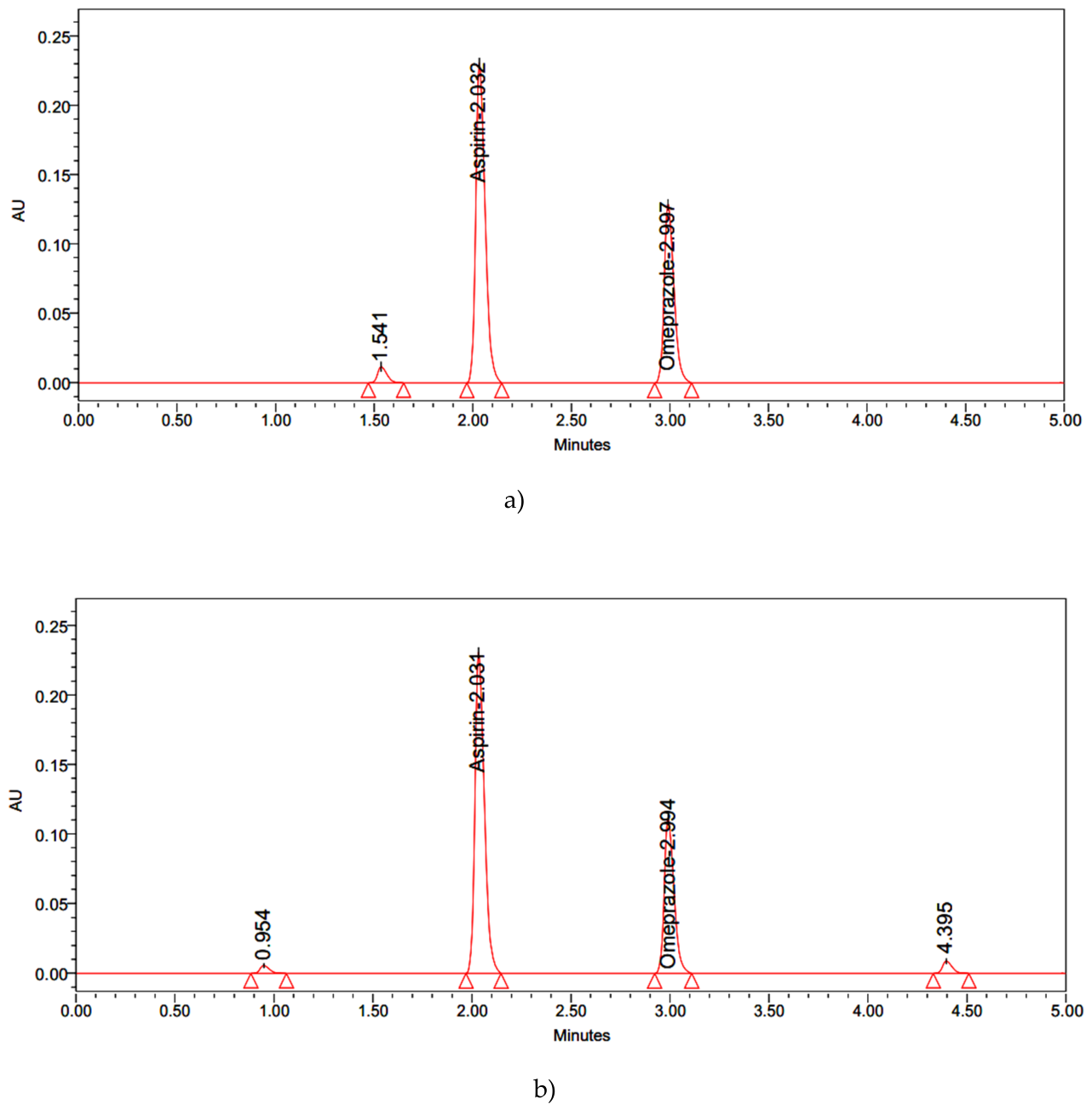
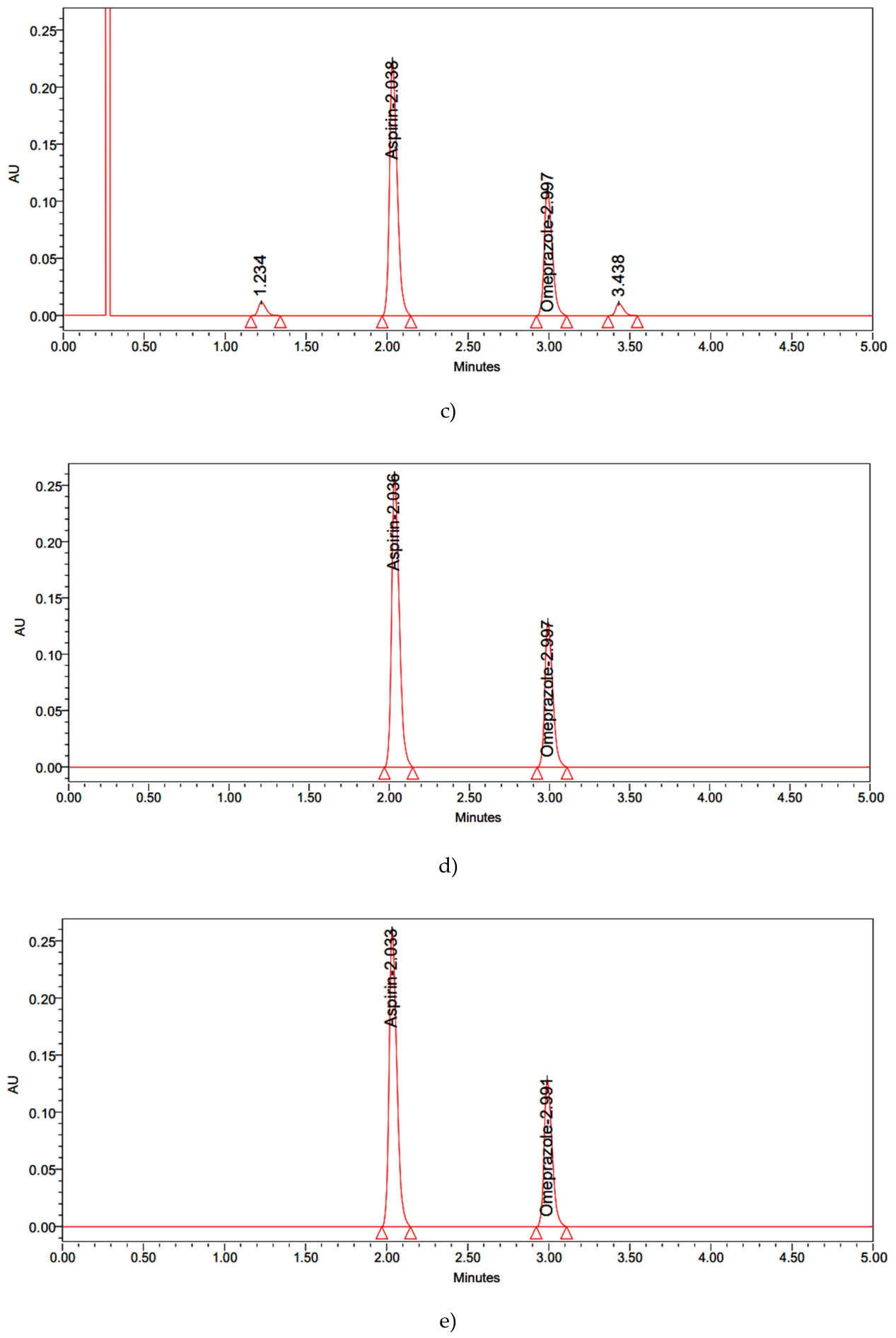
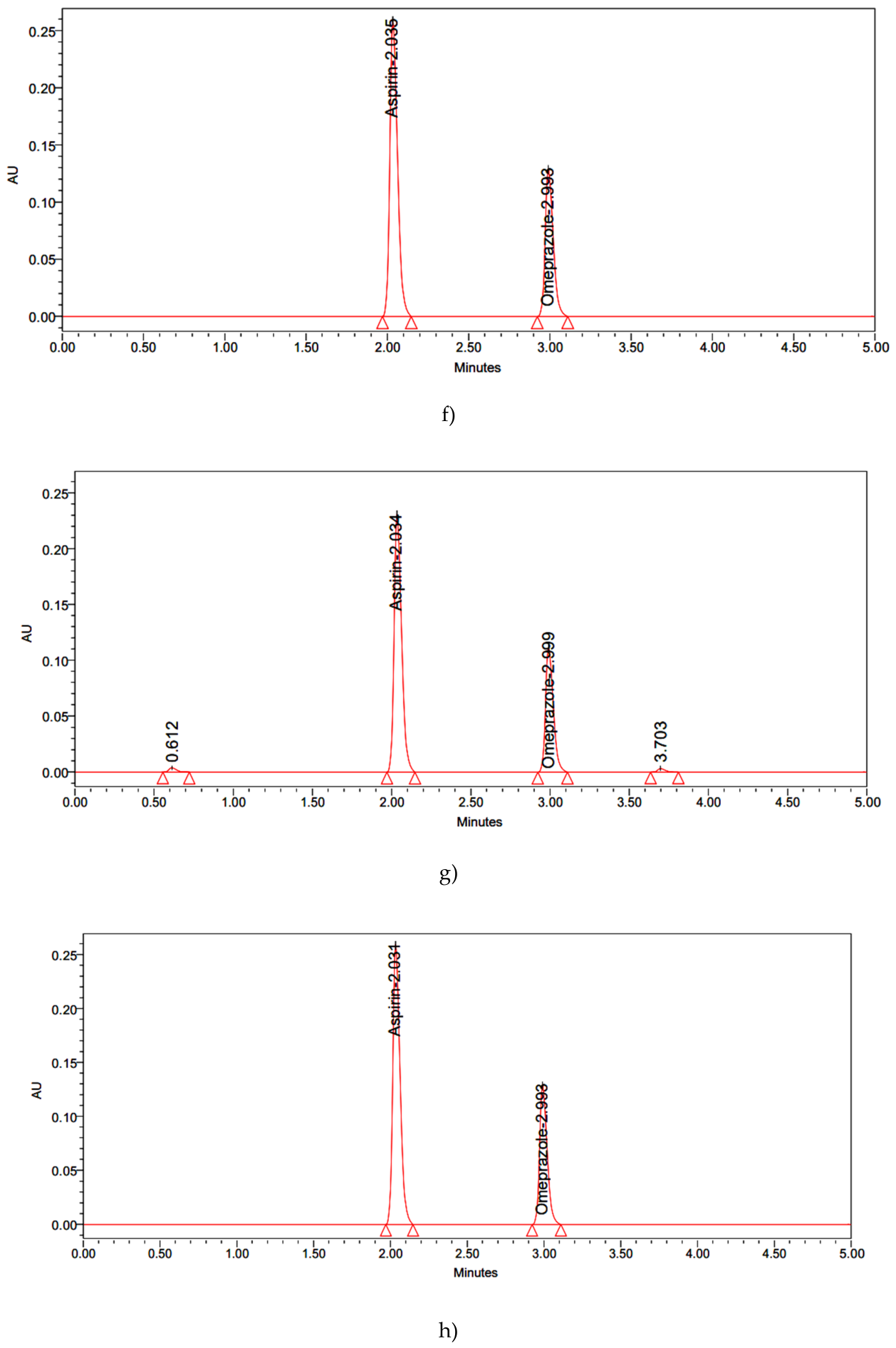
| Results: % Degradation results | Aspirin | Omeprazole | ||||||||
| Area | % Assay | % Deg | Purity Angle | Purity Threshold | Area | % Assay | % Deg | Purity Angle | Purity Threshold | |
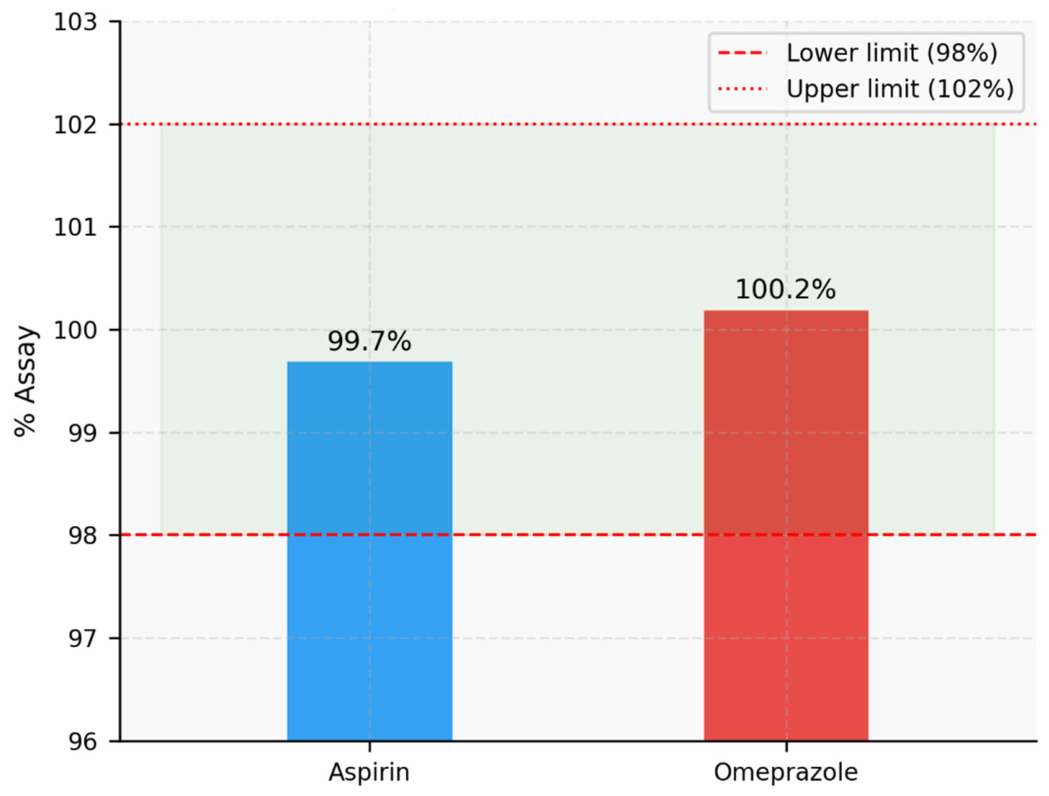
| Control | 2717859 | 100 | 0 | 1.632 | 3.011 | 1279657 | 100 | 0 | 2.145 | 3.716 |
| Acid | 2393654 | 88.1 | 11.9 | 1.628 | 3.034 | 1255360 | 98.1 | 1.9 | 2.173 | 3.742 |
| Alkali | 2412970 | 88.8 | 11.2 | 1.667 | 3.068 | 1121798 | 87.7 | 12.3 | 2.121 | 3.738 |
| Peroxide | 2334286 | 85.9 | 14.1 | 1.605 | 3.017 | 1108724 | 86.7 | 13.3 | 2.169 | 3.719 |
| Reduction | 2662463 | 98.0 | 2.0 | 1.689 | 3.024 | 1243516 | 97.2 | 2.8 | 2.105 | 3.765 |
| Thermal | 2667091 | 98.2 | 1.8 | 1.624 | 3.033 | 1260427 | 98.5 | 1.5 | 2.127 | 3.744 |
| Photolytic | 2680728 | 98.7 | 1.3 | 1.688 | 3.076 | 1234238 | 96.5 | 3.5 | 2.143 | 3.720 |
| Hydrolysis | 2431300 | 89.5 | 10.5 | 1.673 | 3.091 | 1149677 | 89.9 | 10.1 | 2.119 | 3.718 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




