Chemistry
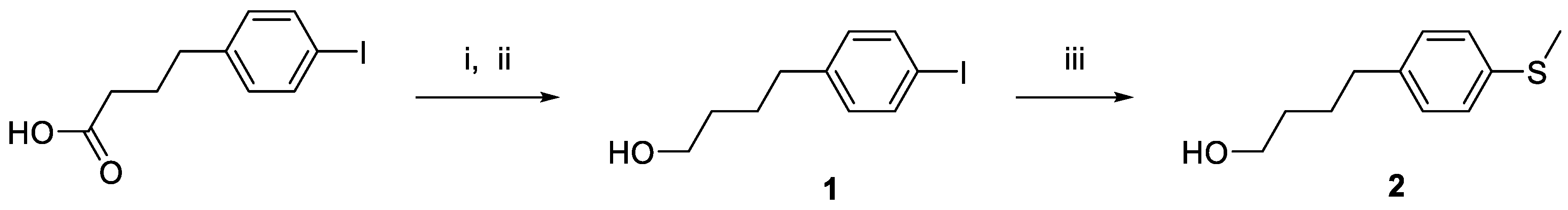
4. -(4-Iodophenyl)butan-1-ol (1).(48) 4-(p-Iodophenyl)butyric acid (11.87 g, 40.9 mmol) was dissolved in THF (100 mL) from a SealSure bottle (Sigma Aldrich, Burlington, MA). The solution was stirred while BH3·THF (1 M, 100 mL) was added slowly, resulting in gas release. The addition was controlled and the reaction temperature was maintained below 40 oC. A clear, colorless solution formed after 5 min. Reversed phase HPLC (general method A) showed complete conversion of the starting acid to the alcohol. Extending the reaction time to 72 h showed no observable change. Methanol (100 mL) was then added slowly to quench any remaining borane, resulting in hydrogen gas evolution. Once gas evolution had ceased, water (100 mL) was added gradually; no additional gas or heat was generated. The reaction mixture was then stirred at RT for 1 h. A saturated solution of ammonium chloride (100 mL) was then added. The product was extracted with DCM (3 × 100 mL) and the combined organic layers were dried (MgSO₄) and the solvent removed under vacuum. The resultant oil was redissolved in acetonitrile, transferred, and then dried with Centrifan to give 1 as a pale yellow oil (14.87 g, yield 86%). 1H-NMR (CDCl3): δ 7.59 (d, 2H, 3JHH = 12 Hz, Ar-H), 6.93 (d, 2H, 3JHH = 8.0 Hz, Ar-H), 3.64 (t, 2H, 3JHH = 6.0 Hz, CH2OH), 2.58 (t, 2H, 3JHH = 8.0 Hz, CH2OH), 1.68 (m, 2H, CH2), 1.59 (m, 2H, CH2). 13C-NMR (CDCl3): δ 142.07 (CAr), 137.44 (CAr), 130.69 (CAr), 90.90 (CAr-I), 62.71 (CH2OH), 36.24 (CH2), 32.26 (CH2), 27.49 (CH2).
4-(4-Methylthiophenyl)butan-1-ol (2).(49) 4-(p-Iodophenyl)butan-1-ol (1; 2.01 g, 7.3 mmol), NaSMe (0.60 g, 8.6 mmol), Me3SnCl (1.67 g, 8.4 mmol), and [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II)·CH2Cl2 (0.82 g, 1.1 mmol) were dissolved in DMSO (10 mL) in a 35-mL microwave tube. The reaction mixture was then heated in a microwave reactor (110 °C, 30 min, 50 W, 250 psi). Reversed phase HPLC (general method A) showed complete conversion. The reaction mixture was then dissolved in DCM (200 mL) and washed with water. The organic layer was then dried over MgSO4, filtered, and then added to silica gel (200 mesh, 50 mL). The solvent was removed from the silica gel slurry under vacuum and the reaction mixture was purified (Combi-Flash; silica, hexanes/ethyl acetate). LC-MS analysis confirmed product isolation. The product fractions were dried under vacuum. The resulting residue was then redissolved in acetonitrile, and passed through a 0.2 µm syringe filter, and then dried with Centrifan to give 2 as a brown oil (0.69 g, yield 48%). 1H-NMR (CDCl3): δ 7.20 (d, 2H, 3JHH = 8.0 Hz, Ar-H), 7.11 (d, 2H, 3JHH = 8.0 Hz, Ar-H), 3.66 (t, 2H, 3JHH = 6.0 Hz, CH2OH), 2.61 (t, 2H, 3JHH = 8.0 Hz, CH2OH), 2.47 (s, 3H, CH3), 1.68 (m, 2H, CH2), 1.59 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 139.69 (CAr), 135.41 (CAr), 129.17 (CAr), 127.39 (C Ar-I), 63.01 (CH2OH), 35.28 (CH2), 32.47 (CH2), 27.72 (CH2), 16.59 (SCH3). HRMS calcd for C11H17O2S [M + OH]+: m/z = 213.0952, found 213.0949; error: 1.4 ppm.
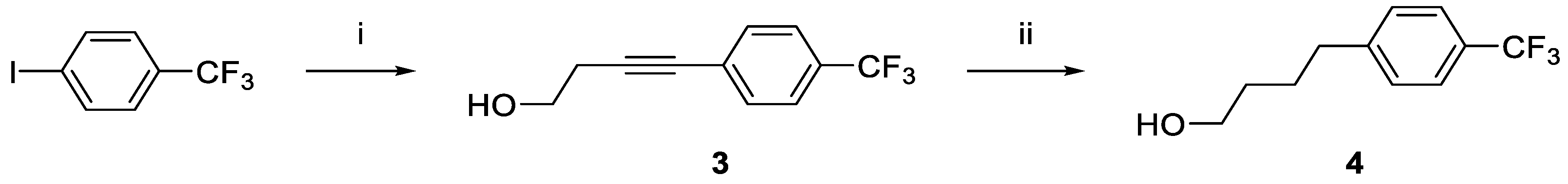
4-(4-(Trifluoromethyl)phenyl)but-3-yn-1-ol (3).(50) 1-Iodo-4-(trifluoromethyl)benzene (20.0 g, 73.5 mmol), CuI (2.1 g, 11.0 mmol), Pd(PPh3)2Cl2·CH2Cl2 (2.9 g, 3.7 mmol), and but-3-yn-1-ol (10.3 g, 147.1 mmol) were added to triethylamine (300 mL) under nitrogen. The mixture was heated at 60 °C for 16 h. Reversed phase HPLC (general method B) showed near complete consumption of the starting material. The reaction solution was cooled to RT, ethyl acetate (550 mL) was added, stirred for 20 min, and then filtered. The filter cake was then washed with ethyl acetate (200 mL). The combined ethyl acetate washings were then dried under vacuum to give a black oil, which was purified (Combi-Flash; silica gel, petroleum ether/ethyl acetate, from 50: 1 to 3: 1 v/v) to give 3 as a light-yellow liquid (9.7 g, yield 62.0%). 1H-NMR (CDCl3): δ 7.55 (d, 2H, 3JHH = 8.0 Hz, Ar-H), 7.50 (d, 2H, 3JHH = 8.0 Hz, Ar-H), 3.84 (t, 2H, 3JHH = 6.0 Hz, CH2OH), 2.72 (t, 2H, 3JHH = 8.0 Hz, CH2CH2OH). 13C{1H}-NMR (CDCl3): δ 132.12 (CH), 129.92 (q, 2JCF = 32.3 Hz), 127.41, 125.39 (q, 2JCF = 4.0 Hz, CH), 124.14 (q, 1JCF = 270 Hz, CF3), 89.41 (C2), 81.43 (C2), 61.22 (CH2O), 23.99. HRMS calcd for C11H10OF3 [M + H]+: m/z = 215.0684, found 215.0681; error: –0.4 ppm.
4-(4-(Trifluoromethyl)phenyl)butan-1-ol (4).(50) Compound 3 (9.7 g, 45.3 mmol) was dissolved in methanol (160 mL) and Pd/C (1.0 g, 10% wt) was added. The reaction flask was then evacuated and filled with hydrogen gas to 1 atmosphere. The reaction mixture was then stirred at 40 ℃ for 8 h. TLC showed consumption of the starting material. The reaction mixture was then cooled to RT and filtered. After solvent removal under vacuum, the reaction mixture was purified (Combi-Flash; silica gel, hexane/ethyl acetate) to give 4 as a pale yellow liquid (7.7 g, yield 78%). 1H-NMR (CDCl3): δ 7.53 (d, 2H, 3JHH = 8.0 Hz, Ar-H), 7.29 (d, 2H, 3JHH = 8.0 Hz, Ar-H), 3.67 (t, 2H, 3JHH = 6.0 Hz, CH2OH), 2.71 (t, 2H, 3JHH = 6.0 Hz, CH2CH2OH), 1.72 (quint, 2H, 3JHH = 6.0 Hz, CH2CH2OH), 1.62 (quint, 2H, 3JHH = 6.0 Hz, CH2CH2OH). 13C{1H}-NMR (CDCl3): δ 146.62, 128.90 (CH), 128.38 (q, 2JCF = 33.0 Hz, CCF), 127.41, 125.45 (q, 2JCF = 4.0 Hz, CHCF), 124.57 (q, 1JCF = 270 Hz, CF3), 62.88 (CH2O), 35.68, 32.37, 27.52.
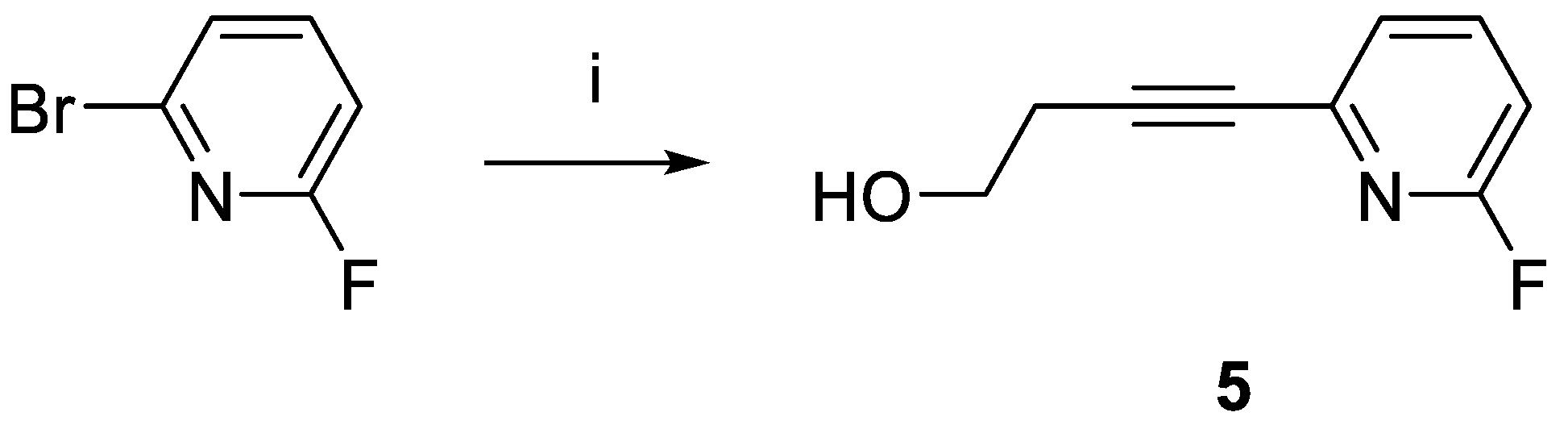
4-(6-Fluoropyridin-2-yl)but-3-yn-1-ol (5). 2-Bromo-6-fluoropyridine (6.11 g, 34.7 mmol), but-3-yn-1-ol (2.61g, 37.2 mmol), CuBr (509.6 mg, 3.6 mmol), Pd(PPh3)2Cl2 (1.2 g, 1.7 mmol), and PPh3 (0.96 g, 3.7 mmol) were dissolved in Et2NH (10 mL) resulting in a green solution and white precipitate. This mixture was stirred at RT for 1 h and then heated at 88 oC for 24 h. The solution turned brown after stirring at RT for 5 min, forming an off-white precipitate. HPLC showed complete conversion after heating for 3 h, however, heating continued for the full 24 h. The next day, solvent was removed under vacuum and the residue was redissolved in DCM (200 mL). Silica gel (80 mL) was added, the solvent was removed under vacuum, and the reaction mixture was purified (Combi-Flash; silica gel, hexane/ethyl acetate) to give 5 as a colorless oil (0.50 g, yield 88%). 1H-NMR (CDCl3): δ 7.73 (dd, 1H, 3JHH = 8.0 Hz, Ar-H), 7.27 (dd, 1H, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, Ar-H), 6.88 (dd, 1H, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, Ar-H), 3.86 (t, 2H, 3JHH = 6.0 Hz, CH2OH), 2.71 (t, 2H, 3JHH = 8.0 Hz, CH2OH), 3.20 (brs, 1H, OH). 13C{1H}-NMR (CDCl3): δ 163.03 (d, 1JCF = 241.5 Hz, Fpy, CF), 141.52 (d, 4JCF = 8.0 Hz, Fpy, CH), 141.37 (CH), 124.55 (d, 4JCF = 4.0 Hz, Fpy, CH), 89.31 (C2), 80.76 (C2), 60.85 (CH2O), 23.90. HRMS calcd for C9H9NOF [M + H]+: m/z = 166.0668, found 166.0665; error: –1.8 ppm.
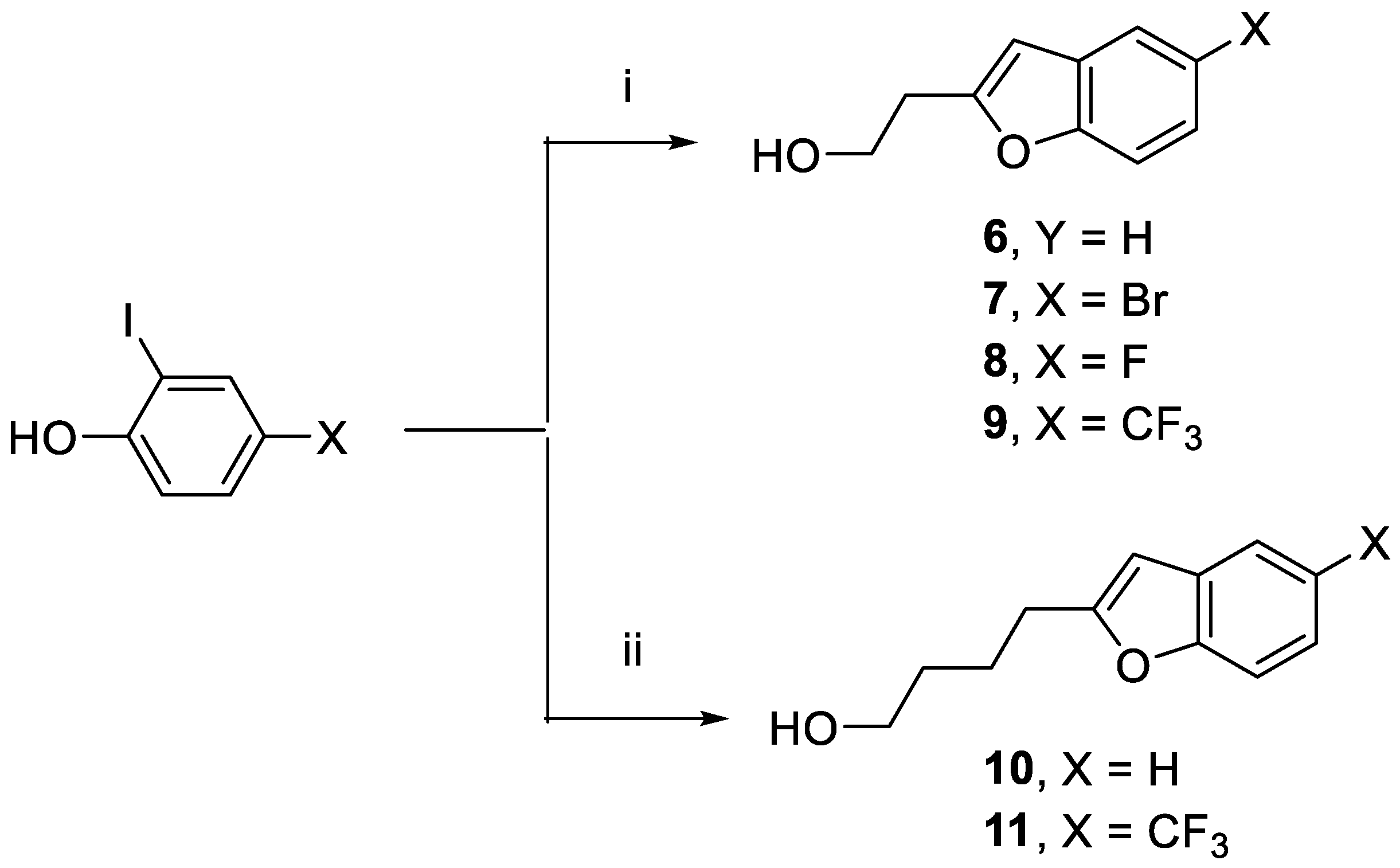
2-(Benzofuran-2-yl)ethan-1-ol (6).(51,52) 2-Iodophenol (1.069 g, 4.86 mmol), but-3-yn-1-ol (0.340 g, 4.85 mmol), CuBr (67.8 mg, 0.47 mmol), (Ph3P)2●PdCl2 (172.4 mg, 0.246 mmol), and Ph3P (152.7 mg, 0.582 mmol) were dissolved in Et2NH (10 mL). The reaction mixture was heated at 90 o C for 4 h. Reversed phase HPLC (general method B) showed starting material consumption. The reaction mixture was left to stir overnight. The following morning DCM (200 mL) was added and this was stirred for 5 min. Silica gel (20 mL) was then added. After removal of the solvent under vacuum, the reaction mixture was purified (Combi-Flash, silica gel, ethyl acetate/hexane) to give 6 as a colorless oil (0.531 g, yield 67%). 1H-NMR (CDCl3): δ 7.50 (dd, 3JHH = 8.0 Hz, 4JHH = 2.0 Hz, 1H, Ar-H), 7.42 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 7.24 (dt, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 7.19 (dt, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.50 (s, 1H, Ar-H), 3.98 (t, 3JHH = 6.0 Hz, 2H, CH2O), 3.04 (t, 3JHH = 6.0 Hz, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 156.14, 155.01, 128.88, 123.74 (CH), 122.84 (CH), 120.63 (CH), 111.06 (CH), 103.89 (CH), 60.95 (CH2O), 32.26 (CH2).
2-(5-Bromobenzofuran-2-yl)ethan-1-ol (7).(53) Use of the method for 6 in same molar proportions to 4-bromo-2-iodophenol (2.12 g, 7.10 mmol) gave 7 as a colorless oil (1.26 g, yield 76%). 1H-NMR (CDCl3): δ 7.61 (s, 1H, Ar-H), 7.32 (d, 3JHH = 8.9 Hz, 1H, Ar-H), 7.28 (d, 3JHH = 8.6 Hz, 1H, Ar-H), 6.45 (s, 1H, Ar-H), 3.98 (t, 3JHH = 6.2 Hz, 2H, CH2O), 3.03 (t, 3JHH = 6.2 Hz, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 157.74, 153.76, 130.92, 126.58 (CH), 123.29 (CH), 115.88, 112.49 (CH), 103.41 (CH), 60.76 (CH2O), 32.20 (CH2).
2-(5-Fluorobenzofuran-2-yl)ethan-1-ol (8). Use of the method for 6 in same molar proportions to 4-fluoro-2-iodophenol (2.29 g, 9.61 mmol) gave 8 as a colorless oil (1.41 g, yield 81%). 1H-NMR (CDCl3): δ 7.33 (dd, 3JHF = 8.0 Hz, 4JHF = 4.0 Hz, 1H, Ar-H), 7.14 (dd, 3JHH = 8.0 Hz, 4JHF = 2.0 Hz, 1H, Ar-H), 6.94 (dt, 3JHF = 3JHH = 8.0 Hz, 4JHF = 2.0 Hz, 1H, Ar-H), 6.47 (s, 1H, Ar-H), 3.98 (t, 3JHH = 6.0 Hz, 2H, CH2O), 3.02 (t, 3JHH = 6.0 Hz, 2H, CH2). 13C{1H}-NMR (CDCl3)3: δ 159.36 (d, 1JCF = 236.0 Hz, CF), 158.18, 151.21, 129.71 (d, C, JCF = 11.0 Hz), 111.55 (d, CH, JCF = 9.0 Hz), 111.23 (d, CH, JCF = 26 Hz), 106.20 (d, CH, JCF = 25.0 Hz), 104.11 (d, CH, JCF = 4.0 Hz), 60.78 (CH2O), 32.28 (CH2).
2-(5-(Trifluoromethyl)benzofuran-2-yl)ethan-1-ol (9). Use of method for 6 in same molar proportions to 2-iodo-4-(trifluoromethyl)phenol (1.06 g, 3.68 mmol) gave 9 as a colorless oil (661 mg, yield 78%). 1H-NMR (CDCl3): δ 7.79 (s, 1H, Ar-H), 7.50 (s, 2H, Ar-H), 6.59 (s, 1H, Ar-H), 4.02 (t, 3JHH = 6.2 Hz, 2H, CH2O), 3.08 (t, 3JHH = 6.2 Hz, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 158.34, 156.36, 129.02, 126.25, 125.58 (q, 2JCF = 31.9 Hz, CCF3), 123.55, 120.94 (q, 3JCF = 3.6 Hz, CHCCF3), 118.30 (q, 3JCF = 3.6 Hz, CHCCF3), 111.40 (CH), 104.10 (CH), 60.74 (CH2O), 32.22 (CH2).
4-(Benzofuran-2-yl)butan-1-ol (10).(54) 2-Iodophenol (1.363 g, 6.20 mmol), hex-5-yn-1-ol (0.646 g, 6.58 mmol), CuBr (94.5 mg, 0.66 mmol), (Ph3P)2PdCl2 (222.3 mg, 0.32 mmol), and Ph3P (161.6 mg, 0.62 mmol) were dissolved in Et2NH (10 mL). The reaction mixture was heated at 90 oC for 4 to 5 h, turning a deep red. HPLC (general method B) showed consumption of the starting material. The reaction mixture was stirred overnight. DCM (200 mL) was then added, and this was stirred for 5 min. Silica gel (20 mL) was then added and the solvent was removed under vacuum. The reaction mixture was purified (Combi-Flash, silica gel, ethyl acetate/hexane) to give 10 as a colorless oil (986 mg, yield 84%). 1H-NMR (CDCl3): δ 7.49 (d, 3JHH = 7.4 Hz, 1H, Ar-H), 7.42 (d, 3JHH = 7.6 Hz, 1H, Ar-H), 7.23–7.16 (m, 2H, Ar-H), 6.40 (s, 1H, Ar-H), 3.68 (t, 3JHH = 6.4 Hz, 2H, CH2O), 2.81 (t, 3JHH = 6.4 Hz, 2H, CH2), 1.86–1.82 (m, 2H, CH2), 1.69–1.59 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 159.31, 154.81, 129.10, 123.32 (CH), 122.60 (CH), 120.39 (CH), 110.89 (CH), 102.23 (CH), 62.70 (CH2O), 32.29, 28.33, 24.14.
4-(5-(Trifluoromethyl)benzofuran-2-yl)butan-1-ol (11). Use of the method for 10 in same molar proportions to 2-iodo-4-(trifluoromethyl)phenol (1.06 g, 3.68 mmol) gave 11 as a colorless oil (637 mg, yield 67%). 1H-NMR (CDCl3): δ 7.76 (vs, 1H, Ar-H), 7.47 (vs, 2H, Ar-H), 6.46 (s, 1H, Ar-H), 3.70 (t, 3JHH = 8.0 Hz, 2H, CH2O), 2.83 (t, 3JHH = 8.0 Hz, 2H, CH2), 1.89‒1.82 (m, 2H, CH2), 1.71–1.51 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 161.44, 156.22, 129.28, 129.20 (CH), 129.10, 125.34 (q, 2JCF = 31.7 Hz), 124.97 (q, 1JCF = 270 Hz, CF3), 120.56 (q, 2JCF = 3.7 Hz, CH), 110.05 (q, 2JCF = 4.0 Hz, CH), 62.66 (CH2O), 32.26, 28.37, 24.06.
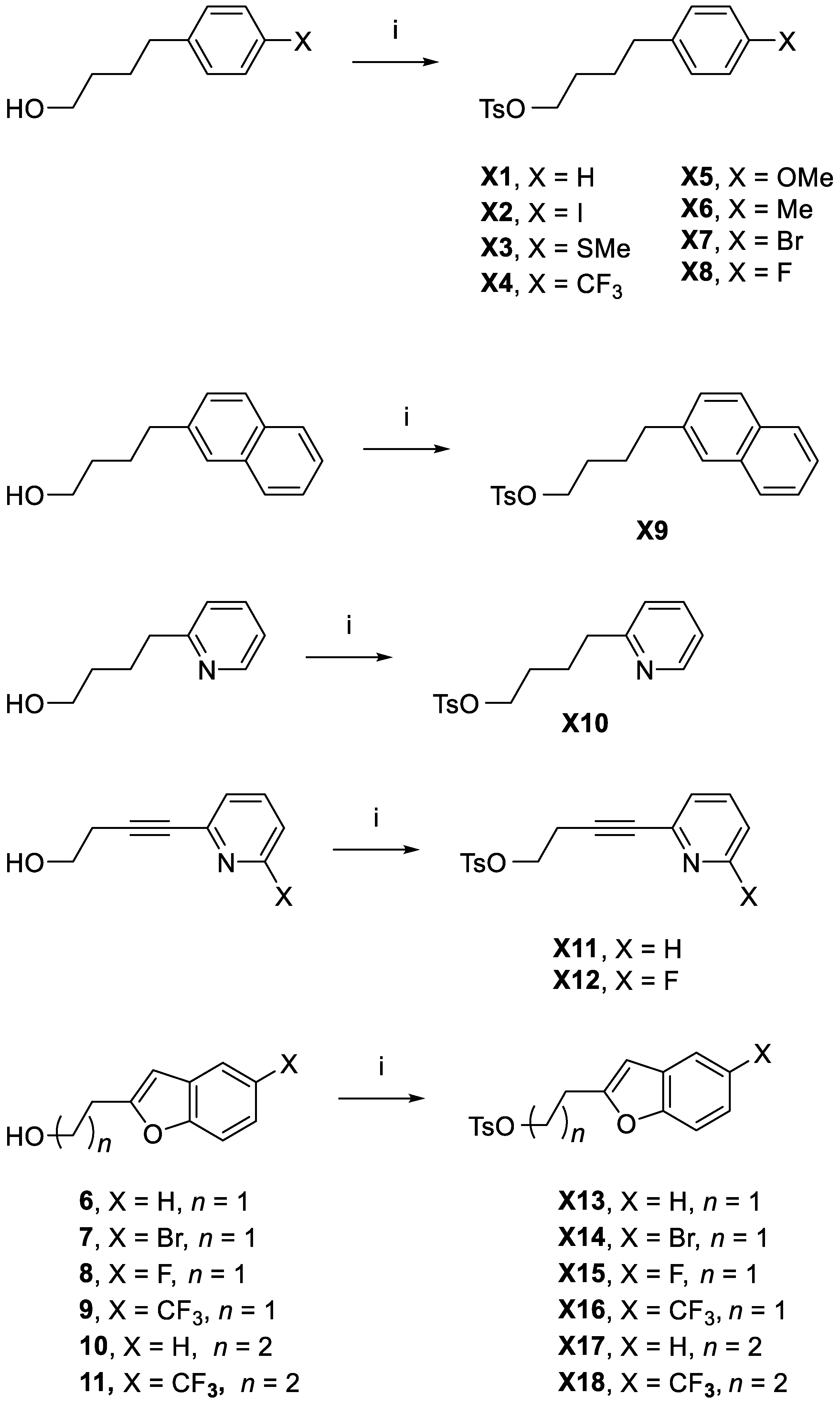
4-Phenylbutyl 4-methylbenzenesulfonate (X1). 4-Phenylbutan-1-ol (6.5 mL, d = 0.984 g/mL, 42.6 mmol), tosyl chloride (15.11 g, 79.3 mmol), and Et3N (21 mL, d = 0.726 g/mL, 15.2 g, 151 mmol) were dissolved in DCM (100 mL) and stirred at RT overnight. A saturated sodium solution (200 mL) was added the following day and this was extracted with DCM . The organic layer was dried (MgSO4), filtered, and then added to silica gel (60 mL). The solvent was removed under vacuum. The reaction mixture was purified (Combi-Flash, silica gel, hexane/ethyl acetate) to give XI as a colorless waxy material (10.0 g, yield 77%). 1H-NMR (CDCl3): δ 7.78 (d, 3JHH = 12 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.26 (vt, 3JHH = 8.0 Hz, 2H, Ar-H), 7.17 (t, 3JHH = 8.0 Hz, 1H, Ar-H), 7.10 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 4.04 (t, 3JHH = 6.0 Hz, 2H, CH2O), 2.56 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.45 (s, 3H, CH3), 1.66 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 144.88, 141.75, 133.35, 130.02 (CH), 128.54 (CH), 128.06 (CH), 126.10 (CH), 70.58 (CH2O), 35.26, 28.51, 27.26, 21.82 (CH3). HRMS calcd for C17H24NO3 [M + NH4]+: m/z = 322.1477, found 322.1476; error: –0.3 ppm.
4-(4-Iodophenyl)butyl 4-methylbenzenesulfonate (X2). Compound 1 (1.20 g, 4.35 mmol), tosyl chloride (1.07 g, 5.61 mmol), and Et3N (2.0 mL, d = 0.726 g/mL, 14.3 mmol) were dissolved in DCM (20 mL) and stirred at RT for 24 h. Reaction progress was monitored with reversed phase HPLC (general method B). The next day, silica gel (25 mL) was added and the solvent removed under vacuum. The reaction mixture was then purified (Combi-Flash, silica gel, hexane/ethyl acetate) to give X2 as a white crystalline solid (1.13 g. yield 61%). Mp: 58–60 °C. 1H-NMR (CDCl3): δ 7.77 (d, 3JHH = 8.2 Hz, 2H, Ar-H), 7.57 (d, 3JHH = 8.2 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.1 Hz, 2H, Ar-H), 6.86 (d, 3JHH = 8.2 Hz, 2H, Ar-H), 4.03 (t, 3JHH = 5.8 Hz, 2H, CH2O), 2.51 (t, 3JHH = 5.8 Hz, 2H, CH2O), 2.45 (s, 3H, CH3), 1.63–1.61 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 144.96, 141.38, 137.61 (CH), 133.32, 130.67 (CH), 130.05 (CH), 128.08 (CH), 91.15, 70.42 (CH2O), 34.78, 28.45, 27.11, 21.86 (CH3). HRMS calcd for C17H23NO3SI [M + NH4]+: m/z = 448.0443, found 448.0441; error: –0.4 ppm.
4-(4-(Methylthio)phenyl)butyl 4-methylbenzenesulfonate (X3). Compound 2 (0.51 g, 2.60 mmol), tosyl chloride (0.56 g, 2.94 mmol), and Et3N (2.1 mL, d = 0.726 g/mL, 15.0 mmol) were dissolved in DCM (20 mL). The reaction mixture was stirred at RT for 24 h. Reaction progress was monitored with reversed phase HPLC (general method B). The next day, silica gel (10 mL) then was added and the solvent removed under vacuum. The reaction mixture was then purified (Combi-Flash, silica gel, hexane/ethyl acetate) to give X3 as a colorless oil (0.43 g, yield 47%). 1H-NMR (CDCl3): δ 7.77 (d, 3JHH = 12 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.17 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.03 (d, 3JHH = 12 Hz, 2H, Ar-H), 4.03 (t, 3JHH = 6.0 Hz, 2H, CH2O), 2.52 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.46 (s, 3H, CH3), 2.44 (s, 3H, CH3), 1.64 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 144.91, 138.88, 135.70, 133.38, 130.04, 129.11, 128.09, 127.34, 70.54 (CH2O), 34.73, 28.49, 27.27, 21.86 (CH3), 16.51 (SCH3). HRMS calcd for C18H26NO3S2 [M + NH4]+: m/z = 368.1354, found 368.1348; error: –1.6 ppm.
4-(4-(Trifluoromethyl)phenyl)butyl 4-methylbenzenesulfonate (X4). Use of the method for X3 in same molar proportions to 5 (1.0 g, 4.58 mmol) gave X4 as a white crystalline solid (1.26 g, yield 74%). Mp: 52–54 °C. 1H-NMR (CDCl3): δ 7.78 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.51 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.22 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 4.04 (d, 3JHH = 6.0 Hz, 2H, CH2), 2.65 (d, 3JHH = 6.0 Hz, 2H, CH2), 1.68 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 145.87, 144.99, 133.31, 130.05, 128.85, 128.55 (2JCF = 32.2 Hz), 128.07, 125.50 (3JCF = 4.0 Hz), 124.51 (1JCF = 272.7 Hz), 70.33 (CH2O), 35.11, 28.49, 27.06, 21.82 (CH3). HRMS calcd for C18H23NO3SF3 [M + NH4]+: m/z = 390.1351, found 390.1347; error: –1.0 ppm.
4-(4-Methoxyphenyl)butyl 4-methylbenzenesulfonate (X5). Use of the method for X3 in the same molar proportions to 4-(4-methoxyphenyl)butan-1-ol (1.0 g, 5.5 mmol) gave X5 as a pale yellow oil (0.39 g, yield 21%). 1H-NMR (CDCl3): δ 7.77 (d, 3JHH = 8.1 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.01 (d, 3JHH = 8.4 Hz, 2H, Ar-H), 7.28 (d, 3JHH = 8.5 Hz, 2H, Ar-H), 4.03 (t, 3JHH = 6.0 Hz, 2H, CH2), 3.78 (s, 3H, OCH3), 2.50 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.44 (s, CH3), 1.65–1.58 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 158.03, 144.87, 133.82, 133.38, 130.01 (CH), 129.42 (CH), 128.07 (CH), 113.96 (CH), 70.63 (CH2O), 55.45 (OCH3), 34.35, 28.47, 27.49, 21.83 (CH3). HRMS calcd for C18H26NO4S [M + NH4]+: m/z = 352.1583, found 352.1581; error: –0.6 ppm.
4-(p-Tolyl)butyl 4-methylbenzenesulfonate (X6). Use of the method for X3 in the same molar proportions to 4-(p-tolyl)butan-1-ol (1.0 g, 6.1 mmol) gave X6 as a colorless waxy solid (0.46 g, yield 24%). 1H-NMR (CDCl3): δ 7.78 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.07 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 6.99 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 4.03 (t, 3JHH = 6.0 Hz, 2H, CH2O), 2.52 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.44 (s, 3H, CH3), 2.31 (s, 3H, CH3), 1.66‒1.61 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 144.87, 138.67, 135.57, 133.40, 130.02 (CH), 129.24 (CH), 128.43 (CH), 128.09 (CH), 70.63 (CH2O), 34.83, 28.53, 27.38, 21.85 (CH3), 21.19 (CH3). HRMS calcd for C18H26NO3S [M + NH4]+: m/z = 336.1633, found 366.1633; error: 0.0 ppm.
4-(4-Bromophenyl)butyl 4-methylbenzenesulfonate (X7). 4-(4-Bromophenyl)butan-1-ol (1.0 g, 4.4 mmol), tosyl chloride (0.99 g, 5.2 mmol) and Et3N (2 mL, d = 0.726 g/mL, 14.3 mmol) were dissolved in DCM (20 mL). The reaction mixture was stirred at RT for 5 d. Reaction progress was monitored with HPLC(general method A). Next, silica gel (60 mL) was added and the solvent removed under vacuum. The reaction mixture was then purified (Combi-Flash, silica gel, hexane/ethyl acetate) to give X7 as a pale yellow waxy solid (0.97 g, yield 58%). 1H-NMR (CDCl3): δ 7.77 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.36 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 6.97 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 4.02 (t, 3JHH = 6.0 Hz, 2H, CH2O), 2.51 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.44 (s, 3H, CH3), 1.64–1.62 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 144.93, 140.68, 133.27, 131.56 (CH), 130.28 (CH), 130.01 (Ts, CH), 128.02 (Ts, CH), 119.80, 70.40 (CH2O), 34.63, 28.40, 27.10, 21.81 (CH3). HRMS calcd for C17H23NO3SBr [M + NH4]+: m/z = 400.0582, found 400.0588; error: 1.5 ppm.
4-(4-Fluorophenyl)butyl 4-methylbenzenesulfonate (X8). 4-(4-Fluorophenyl)butan-1-ol (1.0 g, 4.4 mmol), tosyl chloride (1.22 g, 6.4 mmol), and Et3N (2.5 mL, d = 0.726 g/mL, 17.9 mmol) were dissolved in DCM (20 mL). The reaction mixture was stirred at RT for 5 d. Next, silica gel (20 mL) was added, and the solvent was removed under vacuum. The reaction mixture was then purified (Combi-Flash, silica gel, hexane/ethyl acetate) to give X8 as a colorless oil (0.99 g. yield 52%). 1H-NMR (CDCl3): δ 7.78 (d, 3JHH = 12.0 Hz, 2H, Ar-H), 7.33 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.06 (dd, 3JHH = 8.0 Hz, 3JHF = 4.0 Hz, 2H, Ar-H), 6.94 (t, 3JHH = 8.0 Hz, 2H, Ar-H), 4.03 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.54 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.44 (s, CH3), 1.64 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 161.49 (d, 1JCF = 243.5 Hz, Fph, C-F), 144.92, 137.6 (d, 4JCF = 3.0 Hz, Fph, CHCHCHCF), 133.35, 130.03 (CH), 129.85 (d, 3JCF = 8.0 Hz, Fph, CHCHCF), 128.07 (CH), 115.29 (d, 2JCF = 21.1 Hz, Fph, CHCF), 70.49 (CH2O), 34.47, 28.46, 27.42, 21.83 (CH3). HRMS calcd for C17H23NO3SF [M + NH4]+: m/z = 340.1383, found 340.1378; error: –1.5 ppm.
4-(Naphthalen-2-yl)butyl 4-methylbenzenesulfonate (X9). Use of the method for X8 in the same molar proportions to 4-(naphthalen-2-yl)butan-1-ol (1.0 g, 5.0 mmol) gave X9 was as a tan crystalline solid (0.39 g, yield 22%). Mp: 34–36 °C). 1H-NMR (CDCl3): δ 7.81–7.75 (m, 5H, Ar-H), 7.54 (s, 1H, Ar-H), 7.46 (t, 3JHH = 6.5 Hz, 1H, Ar-H), 7.42 (t, 3JHH = 6.5 Hz, 1H, Ar-H), 7.30 (d, 3JHH = 8.0 Hz, 2H, Ts, Ts-H), 7.26 (d, 3JHH = 8.3 Hz, 1H, Np-H), 4.06 (t, 3JHH = 5.8 Hz, 2H, CH2), 2.74 (t, 3JHH = 5.8 Hz, 2H, CH2), 2.42 (s, CH3), 1.75–1.70 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 144.87, 139.24, 133.75, 133.34, 132.22, 130.01 (Ts, CH), 128.14 (CH), 128.06 (Ts, CH), 127.80 (CH), 127.58 (CH), 127.32 (CH), 126.63 (CH), 126.16 (CH), 125.42 (CH), 70.59 (CH2O), 35.40, 28.52, 27.13, 21.80 (CH3). HRMS calcd for C21H26NO3S [M + NH4]+: m/z = 372.1633, found 372.1638; error: 1.3 ppm.
4-(Pyridin-2-yl)butyl 4-methylbenzenesulfonate (X10). 4-(Pyridin-2-yl)butan-1-ol (5.0 g, 33.1 mmol), tosyl chloride (7.66 g, 40.2 mmol), and Et3N (12 mL, d = 0.726 g/mL, 86.1 mmol) were dissolved in ethyl acetate (100 mL). The reaction mixture was stirred at RT for 24 h. Reaction progress was monitored with HPLC. The next day, silica gel (60 mL) was added and the solvent removed under vacuum. The reaction mixture was then purified (Combi-Flash, silica gel, hexane and ethyl acetate) to give X10 as a thick colorless oil (2.09 g, yield 21%). 1H-NMR (CDCl3): δ 8.42 (d, 3JHH = 4.0 Hz, 1H, py-H), 8.36 (s, 1H, py-H), 7.76 (d, 3JHH = 12.0 Hz, 2H, Ts-H), 7.42 (d, 3JHH = 8.0 Hz, 1H, py-H), 7.32 (d, 3JHH = 12.0 Hz, 2H, Ts-H), 7.18 (dd, 3JHH = 8.0 Hz, 3JHH = 4.0 Hz, 1H, py-H), 4.02 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.55 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.42 (s, 3H, CH3), 1.66–1.64 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 149.99 (CH), 147.70 (CH), 144.96, 136.88, 135.85 (CH), 133.18, 130.00 (Ts, CH), 127.99 (Ts, CH), 123.46, 70.22 (CH2O), 32.34, 28.40, 27.00, 21.77 (CH3). HRMS calcd for C7H7O3S-: m/z = 171.0116, found 171.0119. Error (ppm): 1.8. HRMS calcd for C9H12N+: m/z = 134.0970, found 134.0967; error: –2.2 ppm.
4-(Pyridin-2-yl)but-3-yn-1-yl 4-methylbenzenesulfonate (X11). 4-(Pyridin-2-yl)but-3-yn-1-ol (1.99 g, 13.5 mmol), tosyl chloride (3.11 g, 16.3 mmol), and Et3N (6.0 mL, d = 0.726 g/mL, 43.0 mmol) were dissolved in DCM (100 mL). The reaction mixture was stirred at RT for 24 h. Reaction progress was monitored with HPLC (general method B). The next day, silica gel (60 mL) was added and the solvent removed under vacuum. The reaction mixture was then purified (Combi-Flash, silica gel, hexane/ethyl acetate) to give X11 as a clear waxy solid (0.88 g, yield 22%). 1H-NMR (CDCl3): δ 8.54 (d, 3JHH = 8.0 Hz, 1H, py-H), 7.82 (d, 3JHH = 8.0 Hz, 2H, Ts-H), 7.63 (dt, 3JHH = 8.0 Hz, 4JHH = 2.0 Hz, 1H, py-H), 7.34 (d, 3JHH = 8.0 Hz, 1H, py-H), 7.32 (d, 3JHH = 8.0 Hz, 2H, Ts-H), 7.21 (dd, 3JHH = 8.0 Hz, 3JHH = 4.0 Hz, 1H, py-H), 4.20 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.82 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.43 (s, 3H, CH3). 13C{1H}-NMR (CDCl3): δ 150.13 (py, CH), 145.21, 143.20, 136.33 (py, CH), 132.98, 130.13 (Ts, CH), 128.22 (Ts, CH), 127.20 (py, CH), 123.08 (py, CH), 84.36 (C2), 82.48 (C2), 67.49 (CH2O), 21.86 (CH3), 20.50. HRMS calcd for C16H16NO3S [M + H]+: m/z = 302.0851, found 302.0854; error: 1.0 ppm.
4-(6-Fluoropyridin-2-yl)but-3-yn-1-yl 4-methylbenzenesulfonate (X12). 4-(6-Fluoropyridin-2-yl)but-3-yn-1-ol (3, 2.65 g, 16.0 mmol), tosyl chloride (3.18 g, 16.7 mmol), and Et3N (10 mL, d = 0.726 g/mL, 71.7 mmol) were dissolved in DCM (40 mL). The reaction mixture was stirred at RT for 24 h. Reaction progress was monitored with HPLC (general method B). The next day silica gel (60 mL) was added and the solvent removed under vacuum. The reaction mixture was then purified (Combi-Flash, (silica gel, hexane/ethyl acetate) to give X12 as a clear waxy solid (0.68 g, yield 13%). 1H-NMR (CDCl3): δ 7.82 (d, 3JHH = 8.1 Hz, 2H, Ar-H), 7.73 (pseudo-q, 3JHH = 3JHF = 7.9 Hz, 1H, Ar-H), 7.34 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.23 (dd, 3JHH = 7.4 Hz, 5JHF = 1.9 Hz, 1H, Ar-H), 6.88 (dd, 3JHH = 8.3 Hz, 3JHF = 2.7 Hz, 1H, Ar-H), 4.19 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.81 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.44 (s, CH3). 13C{1H}-NMR (CDCl3): δ 163.1 (d, 1JCF = 250 Hz), 145.28, 141.38 (d, JCF = 8.1 Hz), 141.25 (d, JCF = 37.5 Hz), 132.93, 130.18 (Ts, CH), 128.22 (Ts, CH), 124.69 (d, JCF = 4.0 Hz), 109.66 (d, 1JCF = 36.6 Hz), 85.87 (C2), 81.23 (C2), 67.28 (CH2O), 21.86, 20.50 (CH3). HRMS calcd for C16H15NO3FS [M + H]+: m/z = 320.0757, found 320.0763; error: 1.9 ppm.
2-(Benzofuran-2-yl)ethyl 4-methylbenzenesulfonate (X13). Use of the method for X12 in the same molar proportions to 6 (910 mg, 5.61 mmol) gave X13 as a white crystalline solid (799 mg, yield 45%). Mp: 76–77 °C. 1H-NMR (CDCl3): δ 7.67 (d, 3JHH = 8.0 Hz, 2H, Ts, Ar-H), 7.46 (d, 3JHH = 7.3 Hz, 1H, BFu, Ar-H), 7.30 (d, 3JHH = 7.7 Hz, 1H, BFu, Ar-H,), 7.24‒7.18 (m, 2H, BFu Ar-H), 7.17 (d, 3JHH = 8.0 Hz, 2H, Ts, Ar-H), 6.42 (s, 1H, BFu, Ar-H), 4.36 (t, 3JHH = 6.3 Hz, 2H, CH2), 3.12 (t, 3JHH = 6.3 Hz, 2H, CH2), 2.37 (s, 3H, CH3). 13C{1H}-NMR (CDCl3): δ 154.90, 153.29, 144.98, 132.83, 129.92 (Ts, CH), 128.64, 127.99 (Ts, CH), 123.92 (BFu, CH), 122.87 (BFu, CH), 120.77 (BFu, CH), 111.02 (BFu, CH), 104.54 (BFu, CH), 67.55 (CH2O), 28.75 (CH2), 21.81 (CH3). HRMS calcd for C17H20NO4S [M + NH4]+: m/z = 334.1113, found 334.1114; error: 0.3 ppm.
2-(5-Bromobenzofuran-2-yl)ethyl 4-methylbenzenesulfonate (X14). Use of the method for X12 in the same molar proportions to 7 (790 mg, 3.28 mmol) gave X14 as a white crystalline solid (699 mg, yield 54%). Mp: 88–90 °C. 1H-NMR (CDCl3): δ 7.67 (d, 3JHH = 8.0 Hz, 2H, Ts, Ar-H), 7.58 (s, 1H, BFu, Ar-H), 7.31 (d, 3JHH = 8.7 Hz, 1H, BFu, Ar-H), 7.18 (d, 3JHH = 8.0 Hz, 3JHH = 8.7 Hz, 3H, BFu-Ts, Ar-H), 6.36 (s, 1H, BFu, Ar-H), 4.36 (t, 3JHH = 6.3 Hz, 2H, CH2), 3.11 (t, 3JHH = 6.3 Hz, 2H, CH2), 2.38 (s, 3H, CH3). 13C{1H}-NMR (CDCl3): δ 154.91, 153.65, 145.06, 132.84, 130.64, 129.93 (Ts, CH), 127.97 (Ts, CH), 126.82 (BFu, CH), 123.44 (BFu, CH), 115.93, 112.48 (BFu, CH), 104.10 (BFu, CH), 67.32 (CH2O), 28.76 (CH2), 21.83 (CH3). HRMS calcd for C17H19NO4SBr [M + NH4]+: m/z = 412.0218, found 412.0225; error: 1.7 ppm.
2-(5-Fluorobenzofuran-2-yl)ethyl 4-methylbenzenesulfonate (X15). Use of the method for X12 in the same molar proportions to 8 (610 mg, 3.39 mmol) gave X15 as a white crystalline solid (815 mg, yield 72%). Mp: 76–78 °C. 1H-NMR (CDCl3): δ 7.68 (d, 3JHH = 8.1 Hz, 2H, Ts, Ar-H), 7.23 (dd, 3JHF = 4.1 Hz, 3JHH = 8.9 Hz, 1H, BFu), 7.20 (d, 3JHH = 8.1 Hz, 2H, Ts, Ar-H), 7.11 (dd, 4JHF = 2.5 Hz, 3JHH = 9.0 Hz, 1H, BFu, Ar-H), 6.93 (td, 3JHF = 4JHH = 8.6 Hz, 4JHH = 2.5 Hz, 1H, BFu, Ar-H), 6.40 (s, 1H), 4.36 (t, 3JHH = 6.4 Hz, 2H, CH2), 3.11 (t, 3JHH = 6.4 Hz, 2H, CH2), 2.38 (s, 3H, CH3). 13C{1H}-NMR (CDCl3): δ 154.35 (d, 1JCF = 236.4 Hz), 155.32, 151.13, 145.04, 132.87, 129.94 (Ts, CH), 129.45 (d, JCF = 10.8 Hz, CH), 128.01 (Ts, CH), 111.64 (d, JCF = 2.7 Hz, CH), 111.46 (d, JCF = 13.8 Hz, CH), 106.35 (d, JCF = 25.0 Hz, CH), 104.81 (d, JCF = 4.0 Hz, CH), 67.37 (CH2O), 28.84 (CH2), 21.82 (CH3). HRMS calcd for C17H19NO4FS [M + NH4]+: m/z = 352.1019, found 352.1017; error:–0.6 ppm.
2-(5-Trifluoromethylbenzofuran-2-yl)ethyl 4-methylbenzenesulfonate (X16). Use of the method for X12 in the same molar proportions to 9 (1.07 g, 4.65 mmol) gave X16 as a white crystalline solid (697 mg, yield 39%). Mp: 94–95 °C. 1H-NMR (CD3OD): δ 7.74 (s, 1H, Ar-H), 7.53 (dd, 3JHH = 6.6 Hz, 4JHH = 1.6 Hz, 2H, Ar-H), 7.44 (AB, 3JHH = 8.7 Hz, 4JHH = 1.7 Hz, 1H, Ar-H), 7.40 (AB, 4JHH = 8.7 Hz, 1H, Ar-H), 7.09 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 6.52 (d, 4JHH = 0.7 Hz, 1H, Ar-H), 4.32 (t, 3JHH = 5.7 Hz, 2H, CH2), 3.07 (t, 3JHH = 5.7 Hz, 2H, CH2), 2.24 (s, 3H, Ar-CH3). 13C{1H}-NMR (CD3OD): δ 157.93, 157.68, 146.47, 134.19, 130.96 (Ts, CH), 130.44, 128.95 (Ts, CH), 126.37 (q, 1JCF = 271.0 Hz, 1C, CF), 126.51 (q, 2JCF = 32.0 Hz, 1C), 122.06 (q, 3JCF = 3.6 Hz, 1C, BFu, CH), 119.38 (q, 3JCF = 4.3 Hz, 1C, BFu CH), 112.58 (BFu, CH), 105.70 (BFu, CH), 69.02, (CH2O), 29.40 (CH 2), 21.66 (CH3). HRMS calcd for C18H19NO4F3S [M + NH4]+: m/z = 402.0987, found 402.0992; error: 1.2 ppm.
4-(Benzofuran-2-yl)butyl 4-methylbenzenesulfonate (X17). Use of the method for X12 in the same molar proportions to 10 (710 mg, 3.73 mmol) gave X17 as a clear waxy solid (977 mg, yield 76%). 1H-NMR (CDCl3): δ 7.78 (d, 3JHH = 8.0 Hz, 2H, Ts, Ar-H), 7.47 (d, 3JHH = 7.4 Hz, 1H, BFu, Ar-H), 7.39 (d, 3JHH = 7.6 Hz, 1H, BFu, Ar-H), 7.32 (d, 3JHH = 8.0 Hz, 2H, Ts, Ar-H), 7.21–7.17 (m, 2H, BFu, Ar-H), 6.34 (s, 1H, BFu, Ar-H), 4.07 (t, 3JHH = 5.7 Hz, 2H, CH2), 2.72 (t, 3JHH = 5.7 Hz, 2H, CH2), 2.43 (s, 3H, CH3), 1.76 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 158.49, 154.83, 144.95, 132.83, 130.05 (Ts, CH), 128.98, 128.08 (Ts, CH), 123.48 (BFu, CH), 122.69 (BFu, CH), 120.47 (BFu, CH), 111.93 (BFu, CH), 102.53 (BFu, CH), 70.29 (CH2O), 28.41 (CH2), 27.84 (CH2), 23.82 (CH2), 21.82 (CH3). HRMS calcd for C19H24NO4S [M + NH4]+: m/z = 362.1426, found 362.1419; error: –1.9 ppm.
4-(5-(Trifluoromethyl)benzofuran-2-yl)butyl 4-methylbenzenesulfonate (X18). Use of the method for X12 in the same molar proportions to 11 (425 mg, 1.65 mmol) gave X18 as a clear waxy solid (380 mg, yield 56%). 1H-NMR (CDCl3): δ 7.78 (d, 3JHH = 8.0 Hz, 2H, Ts-H), 7.76 (s, 1H, Ar-H), 7.49-7.45 (AB, 3JHH = 8.0 Hz, 2H, Ar-H), 7.32 (d, 3JHH = 8.0 Hz, 2H, Ts-H), 6.42 (s, 1H, Ar-H), 4.07 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.77 (t, 3JHH = 6.0 Hz, 2H, CH2), 2.43 (s, 3H, CH3), 1.83–1.61 (m, 4H, CH2CH2). 13C{1H}-NMR (CDCl3): δ 160.62, 156.22, 145.02 (Ts), 133.26 (Ts), 130.06 (Ts, CH), 129.07, 128.08 (Ts, CH), 125.44 (q, 2JCF = 32 Hz), 124.93 (q, 1JCF = 271 Hz, CF3), 120.72 (q, 2JCF = 3.0 Hz, CHCF), 110.13 (q, 2JCF = 4.0 Hz, CHCF), 70.14 (CH2O), 28.43, 27.87, 23.73, 21.82. HRMS calcd for C20H23NO4F3S [M + NH4]+: m/z = 430.1300, found 430.1305; error: 1.2 ppm.
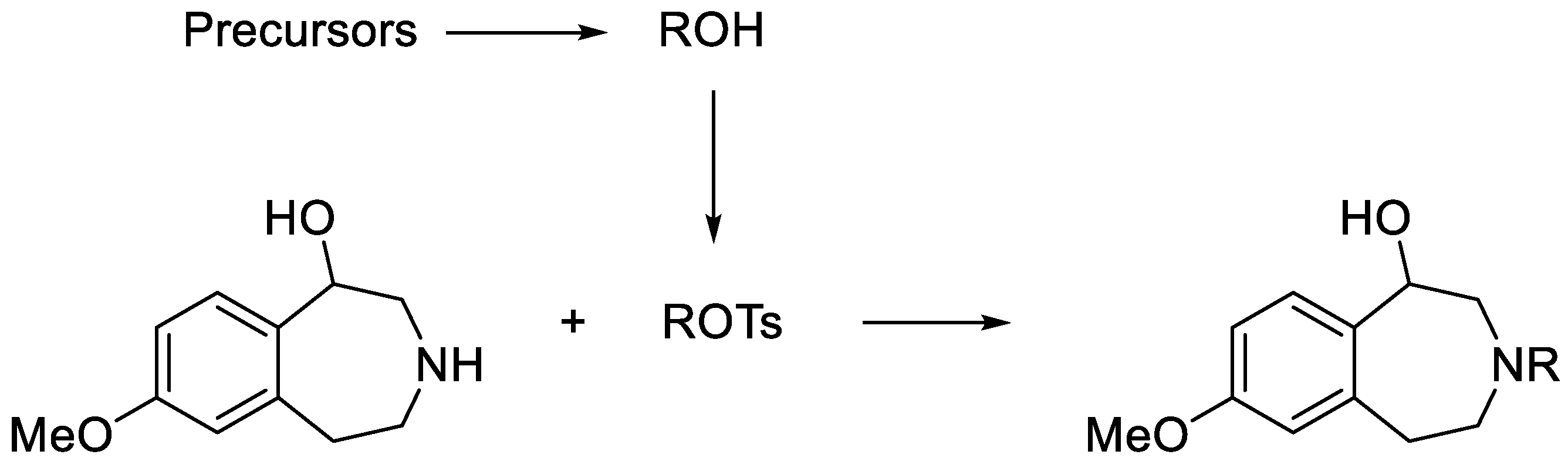
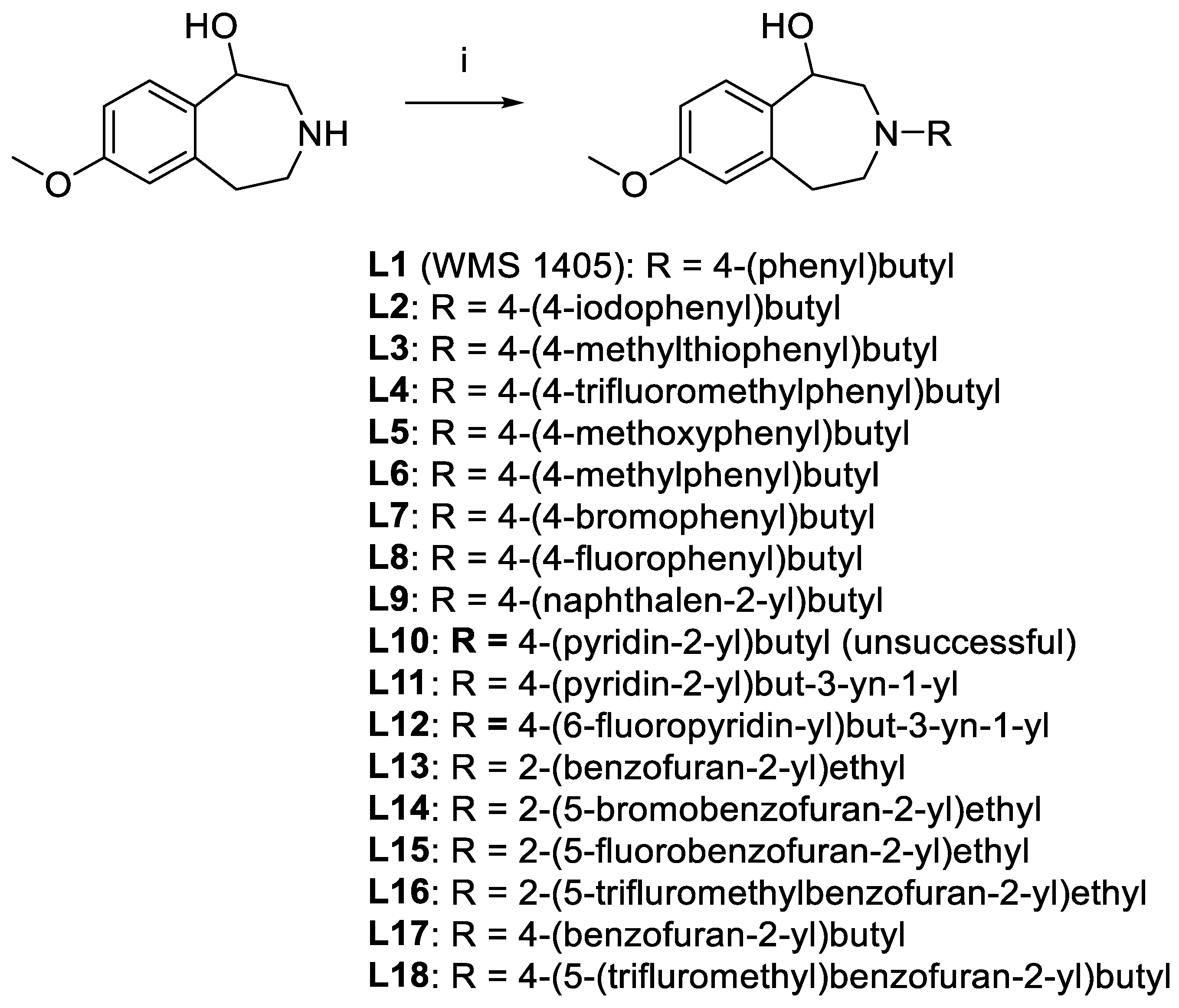
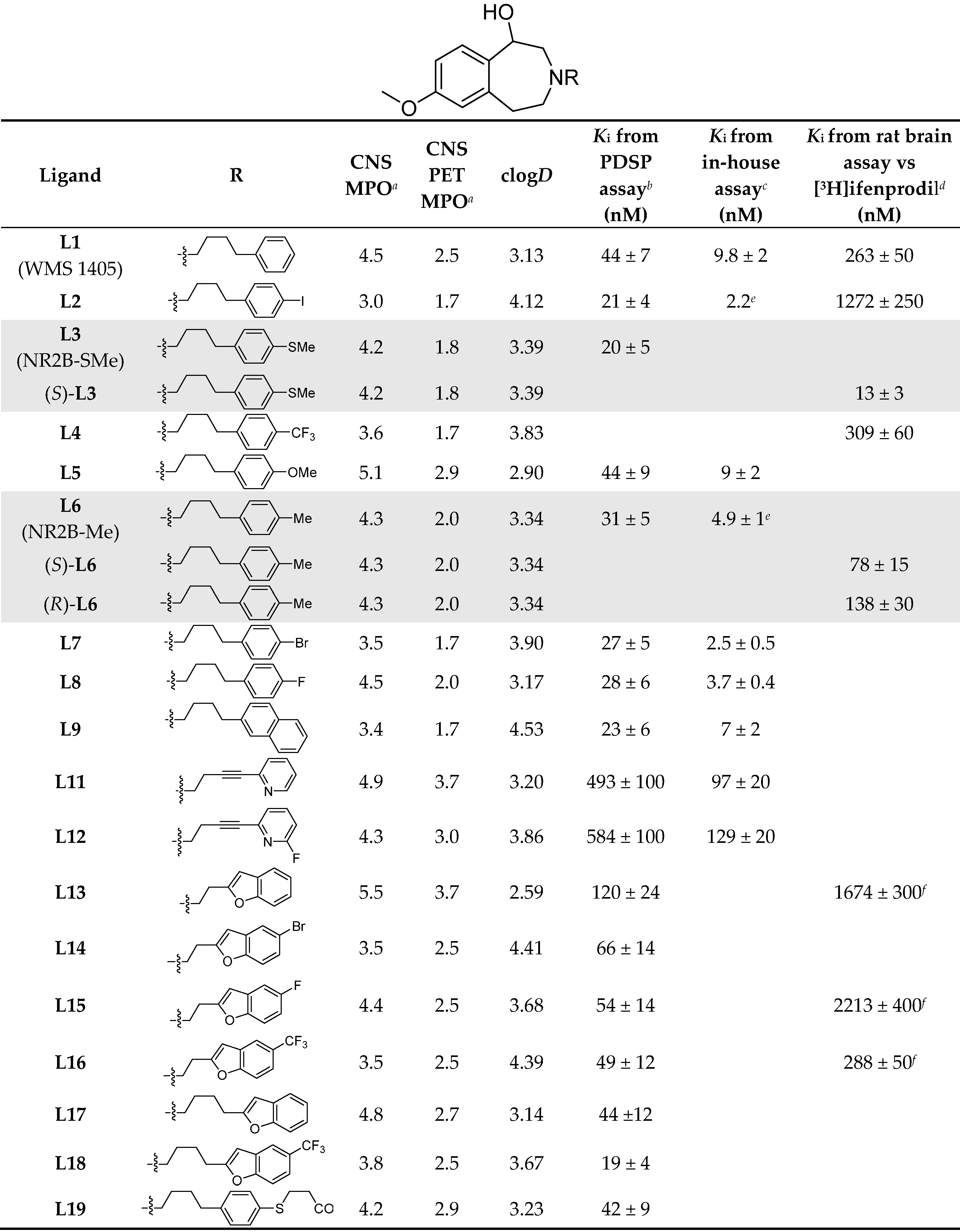
7-Methoxy-3-(4-(phenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L1). 7-Methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (190 mg, 0.98 mmol), X1 (390 mg, 1.28 mmol), and K2CO3 (610 mg, 4.4 mmol) were suspended in acetonitrile (10 mL) and refluxed for 6 d. Reaction progress was monitored with HPLC (general method A). The reaction mixture was cooled to RT and passed through a 0.2 μm syringe filter. The reaction mixture was then purified with reversed phase HPLC (general method C). The solvent was then removed under vacuum and the product was redissolved in acetonitrile, passed through a 0.2 μm syringe filter, and then dried with Centrifan to give L1 as a white crystalline solid (0.23 g, yield 72%). Mp: 78–79 °C. 1H-NMR (CDCl3): δ 7.28 (t, 3JHH = 8.0 Hz, 2H, m-Ar-H), 7.18 (t, 3JHH = 8.0 Hz, 1H, p-Ar-H), 7.18 (d, 3JHH = 8.0 Hz, 2H, o-Ar-H), 7.10 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.64 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.63 (brs, 1H, Ar-H), 4.59 (d, 3JHH = 4.0 Hz, 1H, CH-OH), 3.76 (s, 3H, OCH3), 3.26 (vt, 2JHH = 12 Hz, 1H, CH2), 3.18–3.14 (m, 1H, CH2), 3.02–2.97 (m, 1H, CH2), 2.68–2.59 (m, 5H, CH2), 2.53 (d, 2JHH = 12 Hz, 1H, CH2), 2.43 (vt, 2JHH = 12 Hz, 1H, CH2), 1.69–1.62 (m, 2H, CH2), 1.61–1.54 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 159.09, 142.43, 141.25, 135.57, 129.86, 128.57 (CH), 128.52 (CH), 125.97 (CH), 116.74 (CH), 110.39 (CH), 72.42 (CHO), 60.88 (CN), 59.76 (CN), 56.19 (CN), 55.38 (OCH3), 36.52, 35.92, 29.28, 26.66. HRMS calcd for C21H28NO2 [M + H]+: m/z = 326.2120, found 326.2126; error: 1.8 ppm. HPLC (general method A): tR = 5.14 min, purity 99.62%.
7-Methoxy-3-(4-(4-iodophenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L2) and enantiomers. 7-Methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (420 mg, 2.17 mmol), X2 (1.13 g, 2.63 mmol), and Na2HPO4 (1.30 g, 9.2 mmol) were suspended in acetonitrile (8 mL) and heated at 75 oC for 5 d. Reaction progress was monitored with HPLC (general method A). Next, the reaction mixture was cooled to RT and passed through a 0.2 μm syringe filter and then purified with reversed phase HPLC (general method C). The solvent was then removed under vacuum. The residue was redissolved in ethanol, passed through a 0.2 µm syringe filter and then dried with Centrifan to give L2 as a white crystalline solid (0.714 g, yield 73%). Mp: 100–101 °C. 1H-NMR (CDCl3): δ 7.60 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.16 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.92 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 6.69 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.65 (d, 4JHH = 4.0 Hz, 1H, Ar-H), 4.76 (brs, 1H, CHOH), 3.78 (s, 3H, OCH3), 3.39–3.32 (m, 2H, CH2), 3.16–3.12 (m, 1H, CH2), 2.75 (brs, 4H, CH2), 2.60–2.58 (m, 3H, CH2), 1.63 (m, 4H, CH2). 13C{1H}-NMR (CDCl3): δ 159.34, 141.60, 140.31, 137.64 (CH), 134.50, 130.67 (CH), 129.80, 116.62 (CH), 110.89 (CH), 91.16 (CAr-I), 71.47 (CHO), 60.33 (CN), 59.45 (CN), 55.79 (CN), 55.46 (OCH3), 42.47, 35.20, 28.81, 25.46, 11.26. HRMS calcd for C21H27INO2 [M + H]+: m/z = 452.1086, found 452.1092; error: 1.3 ppm. HPLC (general method A): tR = 7.31 min, purity 100%. Chiral HPLC analysis (general method E): (S)-L2 (tR = 6.97 min), 50.35%; (R)-L2 (tR = 10.96 min), 47.92%.
The enantiomers were resolved and purified with chiral HPLC (general method F). (S)-L2: chiral HPLC (general method E): tR = 6.89 min, purity 98.14%; [α]D²⁰ = –39.02° (c 1.0, CHCl3). [α]D²⁰ = +8.89° (c 1.0, EtOH). (R)-L2: chiral HPLC (general method E): tR = 11.24 min, HPLC purity 99.2%; [α]D²⁰ = +34.03° (c 1.0, CHCl3), [α]D²⁰ = –3.67° (c 1.0, EtOH).
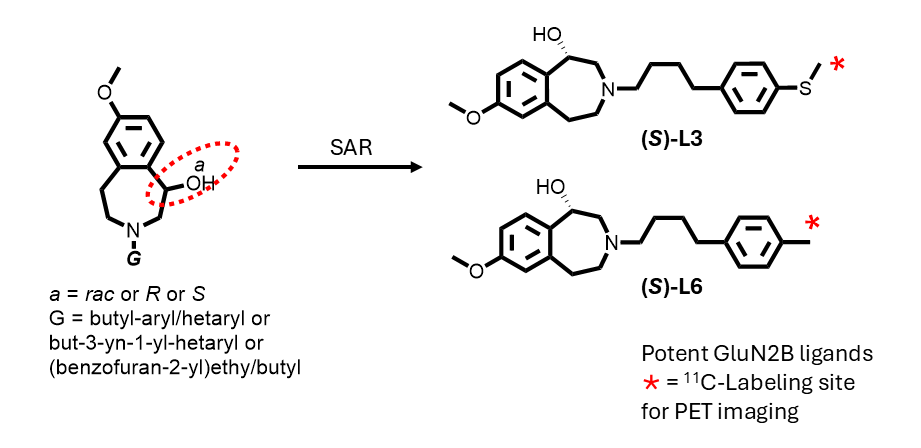
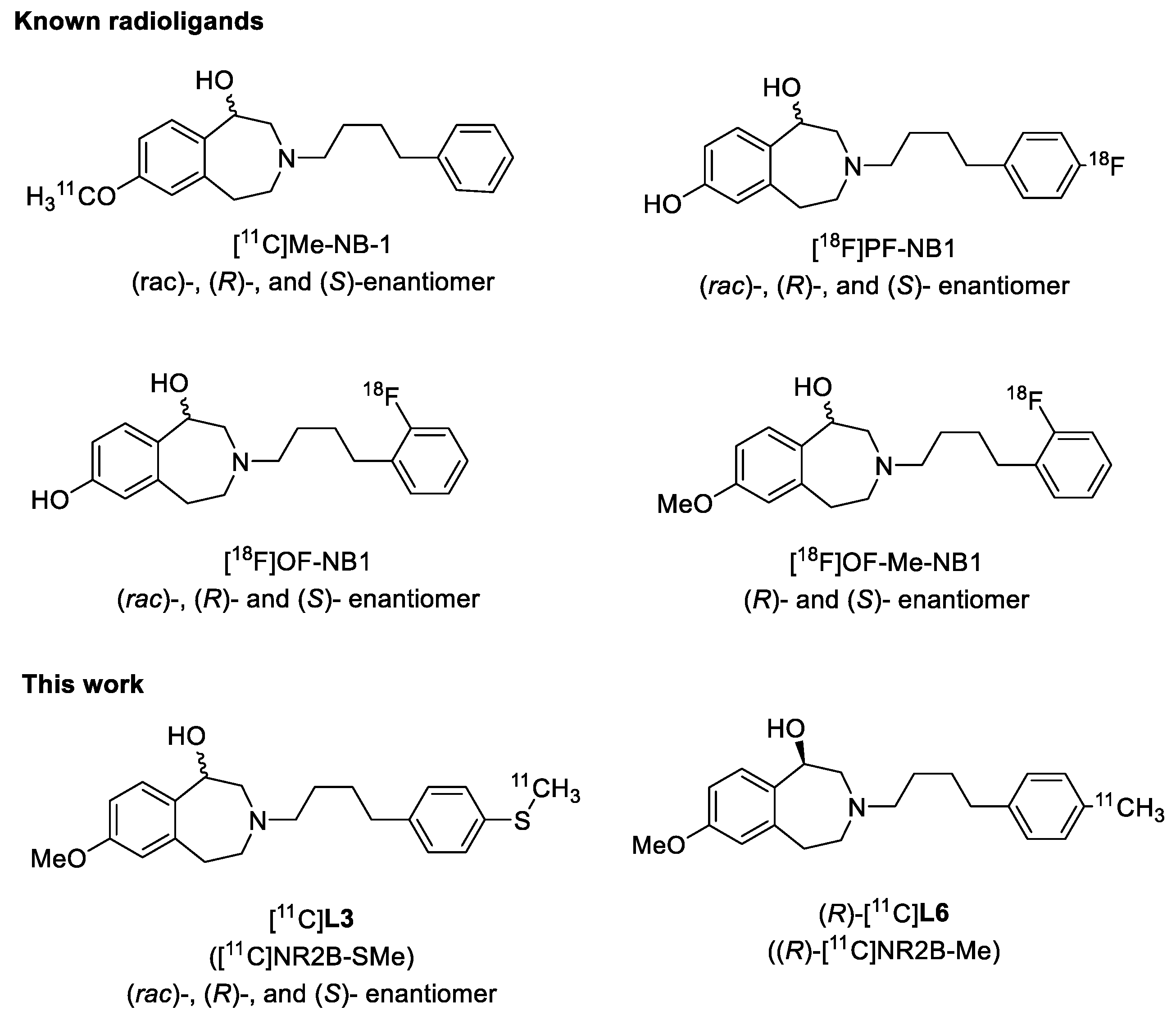
7-Methoxy-3-(4-(4-(methylthio)phenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L3), and its enantiomers. 7-Methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (200 mg, 1.03 mmol), X3 (450 mg, 1.28 mmol), and Na2HPO4 (620 mg, 4.4 mmol) were suspended in acetonitrile (2 mL) and heated at 75 oC for 2 d. Reaction progress was monitored with HPLC (general method A) . Next, the reaction mixture was cooled to RT and passed through a 0.2 μm syringe filter and then purified with reversed phase HPLC (general method C). The solvent was removed under vacuum, the product was redissolved in ethanol and Na2CO3 (1.0 g, 9.4 mmol) was added. The mixture was sonicated for 5 min and then stirred at RT for 1 min before finally being passed through a 0.2 µm syringe filter and then dried with a Centrifan to give L3 as a white crystalline solid (0.288 g, yield 75%). Mp: 89–91 °C. 1H-NMR (CDCl3): δ 7.18 (AB, 3JHH = 8.0 Hz, 2H, Ar-H), 7.09 (AB, 3JHH = 8.0 Hz, 1H, Ar-H), 7.08 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.63 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.62 (brs, 1H, Ar-H), 4.56 (d, 3JHH = 4.0 Hz, 1H, CHOH), 3.75 (s, 3H, OCH3), 3.25 (vt, 2JHH = 12 Hz, 1H, CH2), 3.17–3.12 (m, 1H, CH2), 3.00–2.95 (m, 1H, CH2), 2.66–2.60 (m, 1H, CH2), 2.58 (t, 3JHH = 8.0 Hz, 4H, CH2), 2.50 (d, 2JHH = 12 Hz, 1H, CH2), 2.45 (s, 3H, SCH3), 2.39 (vt, 2JHH = 12 Hz, 1H, CH2), 1.62 (quint, 3JHH = 8.0 Hz, 2H, CH2), 1.54 (quint, 3JHH = 8.0 Hz, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 159.14, 141.36, 139.64, 135.69, 135.45, 129.94, 129.15 (CH), 127.39 (CH), 116.82 (CH), 110.40 (CH), 72.60 (CHO), 60.97 (CN), 59.79 (CN), 56.31 (CN), 55.42 (OCH3), 37.04, 35.40, 29.28, 26.77, 16.57 (SCH3). HRMS calcd for C22H30NO2S [M + H]+: m/z = 372.1997, found 372.1999; error: 0.51 ppm. HPLC (general method A): tR = 7.31 min, purity 100%. Chiral HPLC (general method E): (S)-L3: tR = 8.05 min, 48.03%; (R)-L3: tR = 13.22 min, 48.06%.
The enantiomers were resolved and purified with chiral HPLC (general method F). (S)-L3 chiral HPLC (general method E): tR = 8.10 min, 98.01%. [α]D²⁰ = –39.02° (c 1.0, CHCl3), [α]D²⁰ = +8.89° (c 1.0, EtOH). (R)-L3: chiral HPLC (general method E: tR = 13.27 min, 99.19%. [α]D²⁰ = +34.03° (c 1.0, CHCl3), [α]D²⁰ = –3.67° (c 1.0, EtOH).
7-Methoxy-3-(4-(4-trifluoromethylphenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L4). Use of the method for L3 in the same molar proportions to X4 (240 mg, 0.64 mmol) gave L4 as a white crystalline solid (0.232 g, yield 71%). Mp: 79–81 °C. 1H-NMR (CDCl3): δ 7.52 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.26 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.14 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.67 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.63 (d, 4JHH = 4.0 Hz, 1H, Ar-H), 4.74 (brs, 1H, CHOH), 3.76 (s, 3H, OCH3), 3.35‒3.27 (m, 2H, CH2), 3.17–3.08 (m, 1H, CH2), 2.73-2.49 (m, 7H, CH2), 1.65 (brs, 4H, CH2). 13C{1H}-NMR (CD3OD): δ 160.26, 148.55, 141.78, 137.22, 130.23 (2C, CH),129.29 (q, 2JCF = 31.9 Hz, 1C, CCF), 127.67 (CH), 126.34 (q,3JCF = 3.9 Hz, 2C, CHCHCF), 126.08 (q, 1JCF = 271.0 Hz, 1C, CF), 116.57 (CH), 111.72 (CH), 72.37 (OCH), 63.71 (CN), 60.11 (CN), 56.53 (CN), 55.75 (OCH3), 36.58 (CH2), 30.31 (CH2), 27.24 (CH2). HRMS calcd for C22H27F3NO2 [M + H]+: m/z = 394.1994, found 394.1998; error: 1.0 ppm. HPLC (general method A): tR = 7.21 min, purity 100%.
7-Methoxy-3-(4-(4-methoxyphenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L5). 7-Methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (100 mg, 0.52 mmol), X5 (270 mg, 0.81 mmol), and Na2HPO4 (430 mg, 3.0 mmol) were suspended in acetonitrile (2 mL) and heated at 90oC for 2 d. Reaction progress was monitored with HPLC (general method A). The reaction mixture was cooled to RT, passed through a 0.2 μm syringe filter, and then purified with reversed phase HPLC (general method C). The solvent was then removed under vacuum. The recovered product was then redissolved in acetonitrile, passsed through a 0.2 μm syringe filter, and dried with Centrifan to give L5 as a white crystalline solid (0.21 g, yield 87%). Mp: 86–89 °C. 1H-NMR (CDCl3): δ 7.18 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 7.06 (3JHH = 8 Hz, 2H, Ar-H), 6.81 (3JHH = 8.0 Hz, 2H, Ar-H), 6.69 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.62 (d, 4JHH = 4.0 Hz, 1H, Ar-H), 4.83 (d, 3JHH = 4.0 Hz, 1H, CHOH), 3.77 (s, 3H, OCH3), 3.76 (s, 3H, OCH3), 3.37 (vt, 2JHH = 12 Hz, 1H, CH2), 3.20–3.29 (m, 1H, CH2), 3.15 (m, 1H, CH2), 2.86–2.76 (m, 5H, CH2), 2.57 (vt, 2JHH = 12 Hz, 2H, CH2), 1.61 (m, 4H, CH2). 13C{1H}-NMR (CDCl3): δ 159.31, 158.02, 139.94, 134.24, 133.88, 129.56, 129.40 (2C, CH), 116.43 (CH), 114.00 (2C, CH), 111.01 (CH), 70.85 (CHO), 60.08 (CN), 59.39 (CN), 55.48 (CN), 55.41 (OCH3), 34.65, 34.33, 29.09, 24.92, 21.20. HRMS calcd for C22H30NO3 [M + H]+: m/z = 356.2226, found 356.2223; error: –0.8 ppm. HPLC (general method A): tR = 5.05 min, purity 99.52%.
7-Methoxy-3-(4-(4-methylphenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L6) and enantiomers. Use of the method for L5 in the same molar proportions with 7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (130 mg, 0.67 mmol), X6 (260 mg, 0.82 mmol), and Na2HPO4 (630 mg, 4.4 mmol) in acetonitrile (10 mL) gave L6 as a white crystalline solid (88 mg, 37%). Mp: 86–87 °C. 1H-NMR (CDCl3): δ 7.11 (d, 3JHH = 12 Hz, 1H, Ar-H), 7.10 (AB, 3JHH = 8 Hz, 2H, Ar-H), 7.07 (AB, 3JHH = 8.0 Hz, 2H, Ar-H), 6.66 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.54 (d, 4JHH = 4.0 Hz, 1H, Ar-H), 4.60 (d, 3JHH = 4.0 Hz, 1H, CHOH), 3.78 (s, 3H, OCH3), 3.28 (vt, 2JHH = 12 Hz, 1H, CH2), 3.20–3.16 (m, 1H, CH2), 3.04–2.99 (m, 1H, CH2), 2.69–2.58 (m, 5H, CH2), 2.54 (d, 2JHH = 12 Hz, 1H, CH2), 2.43 (vt, 2JHH = 12 Hz, 1H, CH2), 2.32 (s, 3H, CH3), 1.68–1.61 (m, 2H, CH2), 1.59–1.53 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 159.14, 141.31, 139.36, 135.63, 135.41, 129.91, 129.23 (CH), 128.46 (CH), 116.79 (CH), 110.43 (CH), 72.49 (CHO), 60.92 (CN), 59.83 (CN), 56.24 (CN), 55.42 (OCH3), 35.89, 35.48, 29.42, 26.70, 21.20 (CH3). HRMS calcd for C22H30NO2 [M + H]+: m/z = 340.2277, found 340.2271; error: –1.8 ppm. HPLC (general method A): tR = 6.27 min, purity 100%.
The enantiomers were resolved and purified with HPLC (general method F): Chiral HPLC (general method E): (S)-L6, tR = 5.76 min, 44.43%; (R)-L6, tR = 9.44 min, 51.46%.
(S)-L6: chiral HPLC (general method E): tR = 5.93min, 98.28%; [α]D²⁰ = –24.23° (c 1.0, CHCl3). (R)-L6: chiral HPLC (general method E): tR = 9.57 min, 99.28%. [α]D²⁰ = +28.23° (c 1.0, CHCl3).
7-Methoxy-3-(4-(4-bromophenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L7). Use of the method for L5 in the same molar proportions with 7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (210 mg, 1.1 mmol), X7 (400 mg, 1.04 mmol), and Na2HPO4 (700 mg, 4.9 mmol) in acetonitrile (2 mL) heated at 90 oC for 5 d gave L7 as a white crystalline solid (0.35 g, yield 62%). Mp: 93–96 °C. 1H-NMR (CDCl3): δ 7.40 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.11 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 7.05 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 6.66 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.64 (d, 4JHH = 4.0 Hz, 1H, Ar-H), 4.62 (d, 3JHH = 4.0 Hz, 1H, CHOH), 3.77 (s, 3H, OCH3), 3.28 (vt, 2JHH = 12 Hz, 1H, CH2), 3.20–3.15 (m, 1H, CH2), 3.04–2.99 (m, 1H, CH2), 2.70–2.55 (m, 6H, CH2), 2.46 (vt, 2JHH = 12 Hz, 1H, CH2), 1.66–1.60 (m, 2H, CH2), 1.58–1.53 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 159.16, 141.31, 141.13, 135.44, 131.59 (CH), 130.34 (CH), 129.87, 119.72 (CH), 116.76 (CH), 110.49 (CH), 72.36 (CHO), 60.85 (CN), 59.65 (CN), 56.19 (CN), 55.41 (OCH3), 36.64, 35.29, 29.08, 26.48. HRMS calcd for C21H27BrNO2 [M + H]+: m/z = 404.1225, found 404.1229; error: 1.0 ppm. HPLC (general method A): tR = 6.90 min, purity 96.64%.
7-Methoxy-3-(4-(4-fluorophenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L8). Use of the method for L5 in the same molar proportions with 7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (120 mg, 0.62 mmol), X8 (260 mg, 0.81 mmol), and Na2HPO4 (450 mg, 3.2 mmol) in acetonitrile (2 mL) gave L8 as a brown oil (0.23 g, yield 79%). 1H-NMR (CDCl3): δ 7.13 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 7.11 (d, 3JHH = 8.0 Hz, 2H, Ar-H), 6.96 (t, 3JHH = 3JHF = 8.0 Hz, 2H, Ar-H), 6.65 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.63 (brs, 1H, Ar-H), 4.62 (d, 3JHH = 4.0 Hz, 1H, CHOH), 3.77 (s, 3H, OCH3), 3.27 (vt, 2JHH = 12 Hz, 1H, CH2), 3.19‒3.15 (m, 1H, CH2), 3.03‒2.99 (m, 1H, CH2), 2.70–2.55 (m, 6H, CH2), 2.46 (vt, 2JHH = 12 Hz, 1H, CH2), 1.67–1.60 (m, 2H, CH2), 1.58‒1.53 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 162.64 (C-F), 160.23 (C-F), 159.16, 141.13, 137.97, 137.94, 135.46, 129.89 (CH, JCF), 129.81 (CH, JCF), 116.75 (CH), 115.36 (CH, JCF), 115.15 (CH, JCF), 110.49 (CH), 72.34 (CHO), 60.86 (CN), 59.69 (CN), 56.17 (CN), 55.41 (OCH3), 36.65, 35.07, 29.37, 26.47. HRMS calcd for C21H27NO2F [M + H]+: m/z = 344.2026, found 344.2025; error: –0.3 ppm. HPLC (general method A): tR = 5.55 min, purity 98.74%.
7-Methoxy-3-(4-(naphthalen-2-yl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L9). Use of the method for L7 in the same molar proportions with7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (100 mg, 0.52 mmol), X9 (250 mg, 0.71 mmol), and Na2HPO4 (280 mg, 2.0 mmol) in acetonitrile (10 mL) gave L9 as a white crystalline solid (0.14 g, yield 74%). Mp: 86–88 °C. 1H-NMR (CDCl3): δ 7.81 (d, 3JHH = 8.0 Hz, 1H, Nap-H), 7.78 (d, 3JHH = 8.0 Hz, 1H, Nap-H), 7.77 (d, 3JHH = 8.0 Hz, 1H, Nap-H), 7.60 (s, 1H, Nap-H), 7.44 (p, 3JHH = 7.0 Hz, 2H, Nap-H), 7.31 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 7.14 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.68 (dd, 3JHH = 8.0 Hz, 4JHH = 4.0 Hz, 1H, Ar-H), 6.63 (d, 4JHH = 4.0 Hz, 1H, Ar-H), 4.72 (d, 3JHH = 4.0 Hz, 1H, CHOH), 3.76 (s, 3H, OCH3), 3.34 (vt, 2JHH = 12 Hz, 1H, CH2), 3.29–3.24 (m, 1H, CH2), 3.11–3.07 (m, 1H, CH2), 2.81–2.66 (m, 6H, CH2), 2.57 (vt, 2JHH = 12 Hz, 1H, CH2), 1.78–1.70 (m, 2H, CH2), 1.69‒1.62 (m, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 159.20, 139.69, 134.96, 133.78, 132.19, 129.79 (CH), 129.09, 128.12 (CH), 127.79 (CH), 127.61 (CH), 127.40 (CH), 126.68 (CH), 126.11 (CH), 125.34 (CH), 116.61 (CH), 110.67 (CH), 71.66 (CHO), 60.57 (CN), 59.62 (CN), 55.91 (CN), 55.40 (OCH3), 35.89, 35.69, 28.91, 25.89. HRMS calcd for C25H30NO2 [M + H]+: m/z = 376.2277, found 376.2272; error: –1.3 ppm. HPLC (general method A): tR = 5.69 min, purity 99.13%.
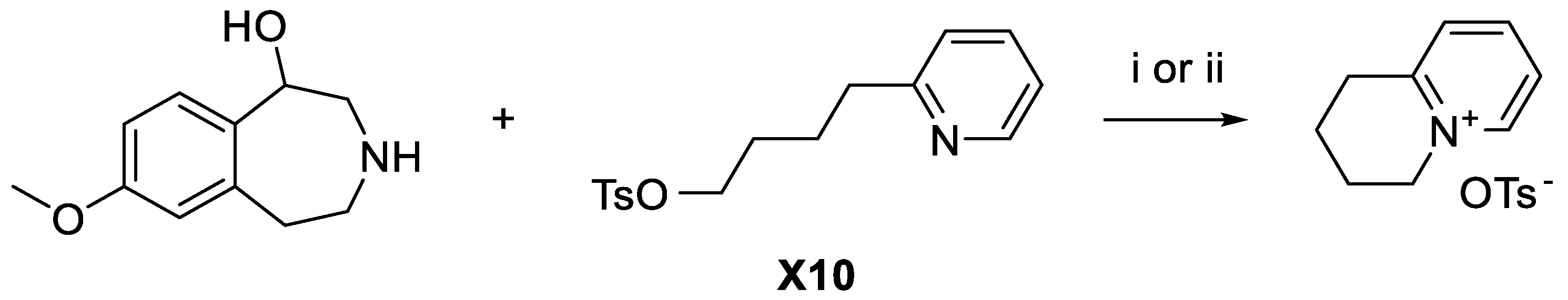
3-(4-(Pyridin-2-yl)butyl)-7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L10). Method 1. 7-Methoxy-2,3,4,5-tetrahydro-1
H-benzo[
d]azepin-1-ol (15.3 mg, 0.52 mmol),
X10 (39.7 mg, 0.66 mmol), and Na
2HPO
4 (73.3 mg, 2.0 mmol) were suspended in DMSO (1.0 mL). The reaction mixture was then heated in a microwave reactor (three conditions were utilized: i) 80 °C 10 min, 30 W, 250 psi; ii) 120 °C, 10 min, 50 W, 250 psi; iii) 150 °C, 10 min, 60 W, 250 psi). A cyclized by-product (
Scheme 7) due to cyclization of
X10 was the only product observed when analyzed with LC-MS.
Method 2. 7-Methoxy-2,3,4,5-tetrahydro-1
H-benzo[
d]azepin-1-ol (150 mg, 0.52 mmol),
X10 (300 mg, 0.66 mmol) and Na
2HPO
4 (510 mg, 2.0 mmol) were suspended in acetonitrile (10 ml) and heated at 90
oC for 22 d. Again, a by-product formed by cyclization of
X10 (
Scheme 7) was the only product observed when analyzed with LC-MS.
7-Methoxy-3-(4-(pyridin-2-yl)but-3-yn-1-yl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L11). Use of the method for L7 in the same molar proportions with X11 (240 mg, 0.80 mmol) gave L11 a brown oil (0.268 g, yield 77%). 1H-NMR (CD3OD): δ 8.34 (ddd, 3JHH = 5.0 Hz, 1 H, Ar-H), 7.68 (td, 3JHH = 7.8 Hz, 4JHH = 1.8 Hz, 1H, Ar-H), 7.37 (vt, 3JHH = 7.9 Hz, 1H, Ar-H), 7.23 (dd, 3JHH = 7.7 Hz ,1 H, Ar-H), 7.17 (d, 3JHH = 8.4 Hz, 1H, Ar-H), 6.62 (dd, 3JHH = 8.3 Hz, 4JHH = 2.7 Hz, 1H, Ar-H), 6.57 (d, 4JHH = 2.6 Hz, 1H, Ar-H), 4.67 (d, 3JHH = 7.6 Hz, 1H, CHOH), 3.66 (s, 3H, OCH3), 2.92 (m, 1H, CH2), 2.83 (m, 3H, CH2), 2.73 (m, 3H, CH2), 2.59 (m, 3H, CH2). 13C{1H}-NMR in (CD3OD): δ 160.35, 150.42 (CH), 144.54, 142.03, 138.66, 137.06, 128.66 (CH), 128.37 (CH), 124.42 (CH), 116.76 (CH), 111.69 (CH), 91.08 81. 69, 72.97 (CHO), 63.09 (CN), 58.77 (CN), 56.21 (CN), 55.76 (OCH3), 36.99 (CH2), 18.12 (CH2). HRMS calcd for C20H23N2O2 [M + H]+: m/z = 323.1760, found 323.1757; error: ‒0.9 ppm. HPLC (general method B): tR = 6.50 min, purity 100%.
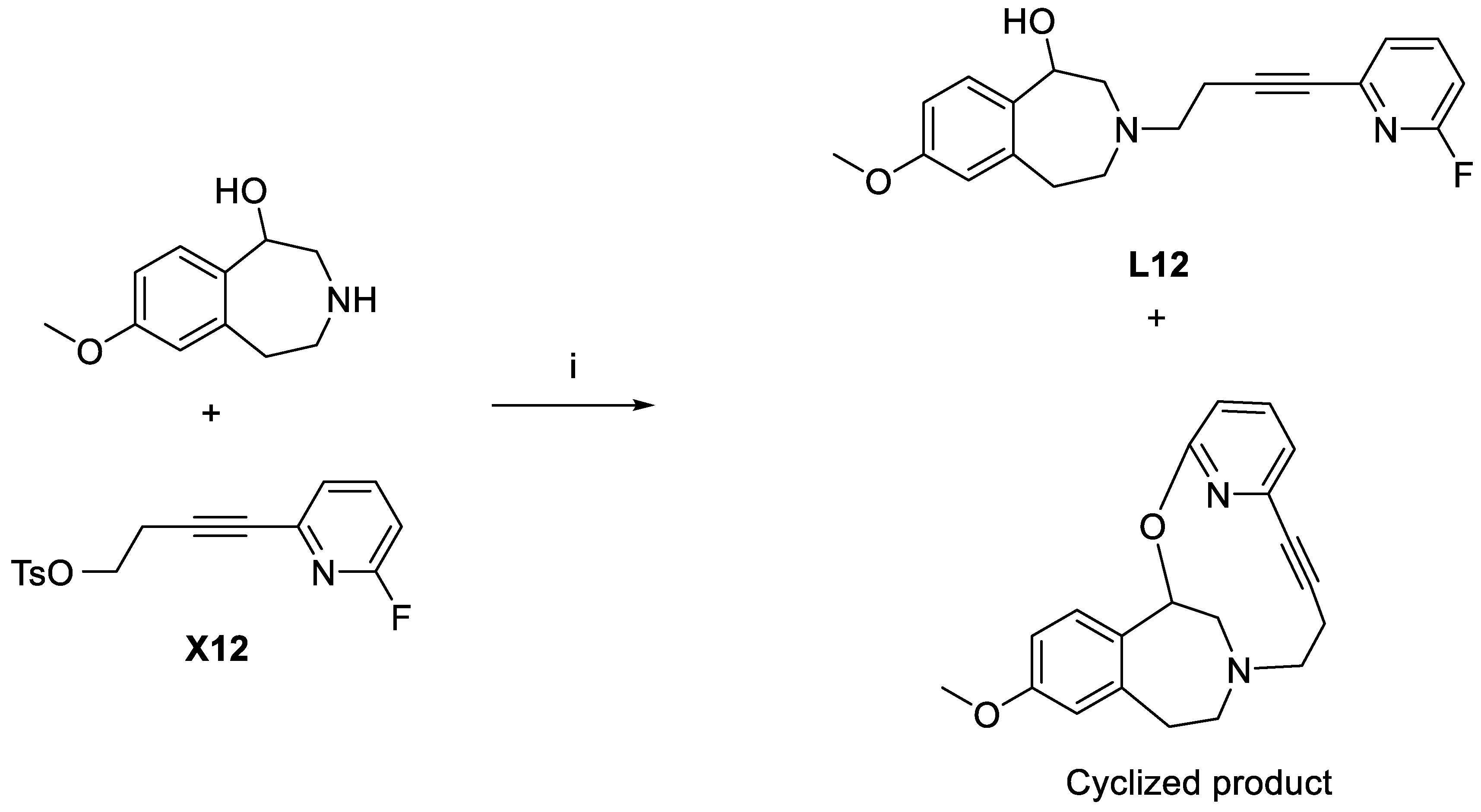
3-(4-(6-Fluoropyridin-2-yl)but-3-yn-1-yl)-7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L12). 7-Methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (134 mg, 0.69 mmol) and X12 (274 mg, 0.86 mmol) were mixed and then heated in a microwave reactor (135 °C,10 min, 60 W, 250psi) to give a black solid upon cooling to RT. The solid was dissolved in DMF (3 mL), passed through a 0.2 μm syringe, and then purified with HPLC (general method C). The solvent was then removed under vacuum. The product was redissolved in acetonitrile, passed through a 0.2 μm syringe filter, and then dried with Centrifan to give L12 as a brown oil (29 mg, yield 12%). 1H-NMR (CDCl3) δ 7.70 (dd, 3JHH = 8.0 Hz, 3JHF = 16.0, 1H, Ar-H), 7.28 (dd, 3JHH = 7.1 Hz, 4JHH = 1.8 Hz, 1H, Ar-H), 7.11 (d, 3JHH = 7.9 Hz, 1H, Ar-H), 6.86 (dd, 3JHH = 8.3 Hz, 4JHH = 2.5 Hz, 1H, Ar-H), 6.67 (dd, 3JHH = 8.3 Hz, 4JHH = 2.6 Hz, 1H, Ar-H), 6.65 (s, 1H, Ar-H), 4.63 (d, 3JHH = 6.8 Hz, 1H, CH-OH), 3.78 (s, 3H, OCH3), 3.27 (m, 2H, CH2), 3.09 (m, 1H, CH2), 2.96 (t, 3JHH = 7.1 Hz, 2H, CH2), 2.68 (m, 4H, CH2), 2.58 (t, 3JHH = 12.0 Hz, 1H, CH2). 13C{1H}-NMR(CDCl3): δ 163.08 (d, 1JCF = 240 Hz, 1C, CF), 159.18, 141.62 (d, 3JCF = 15.1 Hz, 1C, CNCF), 141.37 (d, 3JCF = 8.2 Hz, 1C, CHCHCF), 141.19, 135.50, 130.06, 124.54 (d, 4JCF = 4.2 Hz, 1C, CHCHCHCF), 116.87 (CH), 110.50 (CH), 109.22 (d, 2JCF = 36.7 Hz, 1C, CHCF), 90.15, 80.64, 72.73 (CHO), 60.70 (CN), 58.07 (CN), 55.96 (CN), 55.42 (OCH3), 37.32 (CH2), 18.27 (CH2). HRMS calcd for C20H22N2O2F [M + H]+: m/z = 341.1665, found 323.1667. error: 0.6 ppm. HPLC (general method B): tR = 7.60 min, purity 98.69%.
3-(2-(Benzofuran-2-yl)ethyl)-7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L13). 7-Methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (100 mg, 0.52 mmol), X13 (184 mg, 0.582 mmol) and Na2HPO4 (250 mg, 1.76 mmol) were suspended in acetonitrile (10 mL) and heated at 90 oC for 22 d to give L13 as a brown oil (139 mg, yield 71%). 1H-NMR (CD3OD): δ 7.48 (dd, 3JHH = 7.1 Hz, 4JHH = 1.4, 1H, Ar-H), 7.39 (d, 3JHH = 8.1 Hz, 1H, Ar-H), 7.28 (d, 3JHH = 8.3 Hz, 1H, Ar-H), 7.20 (td, 3JHH = 7.3 Hz, 4JHH = 1.4 Hz, 1H, Ar-H), 7.16 (td, 3JHH = 7.4 Hz, 4JHH = 1.2 Hz, 1H, Ar-H), 6.73 (dd, 3JHH = 8.3 Hz, 4JHH = 2.6 Hz, 1H, Ar-H), 6.68 (d, 4JHH = 2.6 Hz, 1H, Ar-H), 6.53 (s, 1H, Ar-H), 4.79 (d, 3JHH = 8.0 Hz, 1H, CHOH), 3.77 (s, 3H, OCH3), 3.03 (t, 3JHH = 3.1 Hz, 4H, CH2), 2.97 (m, 2H, CH2), 2.86 (m, 2H, CH2), 2.78 (t, 3JHH = 11.7 Hz, 1H, CH2), 2.66 (t, 3JHH = 10.5 Hz, 1H, CH2). 13C{1H}-NMR (CD3OD): δ 160.31, 158.78, 156.28, 141.96, 137.06, 130.40, 128.27 (CH), 124.54 (CH), 123.72 (CH), 121.51 (CH), 116.75 (CH), 111.67 (CH), 103.90 (CH), 72.82 (CHO), 63.21 (CN), 58.41 (CN), 56.28 (CN) 55.75 (OCH3), 36.87 (CH2), 27.00 (CH2). HRMS calcd for C21H24NO3 [M + H]+: m/z = 338.1756, found 338.1751. error: ‒1.5 ppm. HPLC (general method A): tR = 4.59 min, purity 97.52%
3-(2-(5-Bromobenzofuran-2-yl)ethyl)-7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L14). Use of the method for L12 in the same molar proportions with X14 (142 mg, 0.359 mmol) gave L14 as a brown oil (76 mg, yield 51%). 1H-NMR (CDCl3): δ 7.61 (s, 1H, Ar-H), 7.33 (AB, 3JHH = 8.0 Hz, 1H, Ar-H), 7.27 (AB, 3JHH = 8.0 Hz, 1H, Ar-H), 7.21 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 7.13 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.72 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.65 (s, 1H, Ar-H), 6.46 (s, 1H, Ar-H), 4.92 (brs, 1H, CHOH), 3.78 (s, 3H, OCH3), 3.34 (brs, 2H, CH2), 3.21 (brs, 2H, CH2), 3.13 (q, 3JHH = 8.0 Hz, 2H, CH2), 2.75‒2.69 (m, 2H, CH2), 1.32 (t, 3JHH = 8.0 Hz, 2H, CH2). 13C{1H}-NMR (CDCl3): δ 159.30, 153.68, 130.84 (CH), 130.07, 129.12, 126.67 (CH), 126.13, 123.35 (CH), 116.77 (CH), 115.95 (CH), 112.46 (CH), 110.73 (CH), 103.10 (CBr), 72.03 (CHO), 60.52 (CN), 57.72 (CN), 56.00 (CN), 55.45 (OCH3), 45.90, 26.33. HRMS calcd for C21H23BrNO3 [M + H]+: m/z = 416.0861, found 416.0865; error: 1.0 ppm. HPLC (general method A): tR = 6.52 min, purity 100%.
3-(2-(5-Fluorobenzofuran-2-yl)ethyl)-7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L15). Use of the method for L12 in the same molar proportions with X15 (214 mg, 0.640 mmol) gave L16 as a brown wax (134 mg, yield 59%). 1H-NMR (CD3OD): δ 7.38 (dd, 3JHH = 3.9 Hz, 1H, Ar-H), 7.26 (d, 3JHH = 8.3 Hz, 1H, Ar-H), 7.20 (dd, 3JHH = 8.8, 4JHH = 2.4 Hz, 1H, Ar-H), 6.96 (td, 3JHH = 9.3 Hz, 4JHH = 2.9 Hz, 1H, Ar-H), 6.74 (dd, 3JHH = 8.4 Hz, 4JHH = 2.4 Hz, 1H, Ar-H), 6.71 (d, 4JHH = 2.4 Hz, 1H, Ar-H), 6.58 (s, 1H, Ar-H), 4.82 (t, 3JHH = 4.6 Hz CH-OH), 3.77 (s, 3H, O-CH3), 3.13 (br, 5H, CH2), 2.95 (br, 5H, CH2). 13C{1H}-NMR (CD3OD): δ 160.78 (d, 1J CF = 235 Hz, 1C, CF), 160.66, 159.93, 152.60, 141.64, 136.24, 131.29 (d, 3JCF = 11.0 H, 1C, CHCHCF), 129.17, 116.94 (CH), 112.53 (d, 3JCF = 9.9 Hz, 1C, CHCHCF), 112.20 (CH), 111.95 (CH), 107.06 (d, 2J CF = 25.2 Hz, 1C, CHCF), 104.71 (d, 4JCF = 3.7 Hz, 1C, CHCHCHCF), 72.34 (CHO), 62.21 (CN), 58.09 (CN), 56.62 (CN), 55.80 (OCH3), 35.75 (CH2), 26.43 (CH2). HRMS calcd for C21H23NO3F [M + H]+: m/z = 356.1662, found 356.1658; error: –1.1 ppm. HPLC (general method A): tR = 5.38 min, purity 99.57%.
3-(2-(5-Trifluoromethylbenzofuran-2-yl)ethyl)-7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L16). Use of the method for L11 in the same molar proportions with X16 (178 mg, 0.463 mmol) gave L16 as a thick brown oil (120 mg, yield 64%). 1H-NMR (CD3OD): δ 7.85 (s, 1H, Ar-H), 7.58 (AB, 3JHH = 8.6 Hz, 1H, Ar-H), 7.52 (AB, 3JHH = 8.6 Hz, 4JHH = 1.5 Hz, 1H, Ar-H), 7.28 (d, 3JHH = 8.4 Hz, 1H, Ar-H), 6.73 (dd, 3JHH = 8.4 Hz, 4JHH = 2.6 Hz, 1H, Ar-H), 6.68 (m, 3JHH = 4.0 Hz, 2H, Ar-H), 4.78 (d, 3JHH = 7.6 Hz, 1H, CH-OH), 3.77 (s, 3H, OCH3), 3.06 (m, 4H, CH2), 2.99 (m, 1H, CH2), 2.95 (m, 1H, CH2), 2.83 (m, 2H, CH2), 2.77 (m, 1H, CH2), 2.67 (m, 1H, CH2). 13C{1H}-NMR (CD3OD): δ 161.42, 160.34, 157.71 (d, 4JCF = 1.67 Hz, 1C, CHCHCHCF), 141.96, 130.75, 128.28 (CH), 126.41 (q, 1JCF = 270.0 Hz, 1C, CF), 126.41 (q, 2JCF = 31.4 Hz, 1C, CHCF), 121.64 (q, 3JCF = 3.6 Hz, 1C, CHCHCF), 119.15 (q, 3JCF = 4.3 Hz, 1C, CHCHCF), 116.74 (CH), 112.35 (CH), 111.68 (CH), 104.31 (CH), 72.89 (CHO), 63.23 (CN), 58.14 (CN), 56.28 (CN), 55.75 (OCH3), 36.87 (CH2), 27.03 (CH2). HRMS calcd for C22H23NO3F3 [M + H]+: m/z = 406.1630, found 406.1625; error: ‒1.2 ppm. HPLC (general method A): tR = 6.81 min, purity 96.43%
3-(4-(Benzofuran-2-yl)butyl)-7-methoxy-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L17). Use of the method for L12 in same molar proportions with X17 (257 mg, 0.746 mmol) gave L17 as a white crystalline solid (194 mg, yield 71%). Mp: 90–92 °C. 1H-NMR (CD3OD): δ 7.47 (dd, 3JHH = 7.0 Hz, 4JHH = 1.9 Hz, 1H, Ar-H), 7.37 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 7.29 (d, 3JHH = 8.5 Hz, 1H, Ar-H), 7.18 (td, 3J HH = 7.2 Hz, 4JHH = 1.5 Hz, 1H, Ar-H), 7.14 (td, 3JHH = 7.3 Hz, 4JHH = 1.3 Hz, 1H, Ar-H), 6.72 (dd, 3JHH = 8.4 Hz, 4JHH = 2.6 Hz, 1H, Ar-H), 6.66 (d, 4JHH = 2.6 Hz, 1H, Ar-H), 6.47 (d, 4JHH = 0.6 Hz, 1H, Ar-H), 4.79 (d, 3JHH = 8.1 Hz, 1H, CH-OH), 3.75 (s, 3H, OCH3), 2.89 (m, 6H, CH2), 2.60 (m, 3H, CH2), 2.44 (br, 1H, CH2), 1.79 (quint, 2H, CH2), 1.65 (quint, 2H, CH2). 13C{1H}-NMR (CD3OD): δ 160.59, 160.24, 156.28, 141.79, 137.22, 130.50 (CH), 127.68 (CH), 124.38, 123.64, 121.41 (CH), 116.57 (CH), 111.71 (CH), 111.62 (CH), 103.26 (CH), 72.37 (CHO), 63.70 (CN), 60.01 (CN), 56.52 (CN), 55.75 (OCH3), 36.51 (CH2), 29.18 (CH2), 27.20 (CH2), 26.89 (CH2). HRMS: calcd for C23H28NO3 [M + H]+: m/z = 366.2069, found 366.2069; error: 0.0 ppm. HPLC (general method A): tR = 6.34 min, purity 99.00%.
7-Methoxy-3-(4-(5-(trifluoromethyl)benzofuran-2-yl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L18). Use of the method for L12 in same molar proportions with X18 (197 mg, 0.478 mmol) gave L18 as a thick brown oil (141 mg, yield 68%). 1H-NMR (CD3OD): δ 7.83 (s, 1H, Ar-H), 7.56 (AB, 3JHH = 8.6 Hz, 1H, Ar-H), 7.51 (AB, 3JHH = 8.6 Hz, 4JHH = 1.3 Hz, 1 H, Ar-H), 7.29 (d, 3JHH = 8.4 Hz, 1 H, Ar-H), 6.72 (dd, 3JHH = 8.4 Hz, 4JHH = 2.6 Hz, 1 H, Ar-H), 6.67 (d, 4JHH = 2.5 Hz, 1 H, Ar-H), 6.63 (d, 4JHH = 0.8 Hz, 1 H, Ar-H), 4.79 (d, 3JHH = 7.8 Hz, 1H, CH-OH), 3.76 (s, 3H, OCH3), 2.90 (m, 6H, CH2), 2.61 (br, 3H, CH2), 2.51 (br, 1H, CH2), 1.82 (quint, 3JHH = 7.8 Hz, 2H, CH2), 1.67 (quint, 3JHH = 7.8 Hz, 2H, CH2). 13C{1H}-NMR (CD3OD): δ 163.13, 160.28, 157.71, 144.77, 137.15, 130.83, 127.78 (CH), 126.44 (q, 1JCF = 270.8 Hz, 1C, CF), 126.35 (q, 2JCF = 31.6 Hz, 1C, CHCF), 121.52 (q, 3JCF = 3.7 Hz, 1C, CHCHCF), 119.06 (q, 3JCF = 4.1 Hz, 1C, CHCHCF), 116.59 (CH), 112.30 (CH), 111.73 (CH), 103.64 (CH), 72.39 (CHO), 63.60 (CN), 59.94 (CN), 56.56 (CN), 55.75 (OCH3), 36.46 (CH2), 29.14 (CH2), 27.15 (CH2), 26.70 (CH2). HRMS calcd for C23H28NO3 [M + H]+: m/z = 434.1943, found 434.1948; error: 1.2 ppm. HPLC (general method A): tR = 8.06 min, purity 98.37%.
Methyl 3-((4-(4-(1-hydroxy-7-methoxy-1,2,4,5-tetrahydro-3H-benzo[d]azepin-3-yl)butyl)phenyl)thio)propanoate (L19) and enantiomers. L2 (0.2067 g, 0.458 mmol), methyl 3-mercaptopropanoate (62 µL, d = 1.085 g/mL, 0.560 mmol), N,N,1,1,1-pentamethylstannanamine (90 µL, d =1.274 g/mL, 0.552 mmol), and Pd(PPh3)4 (120 mg, 0.104 mmol) were dissolved in DMSO (4.0 mL). The reaction mixture was then heated in a microwave reactor (110 °C, 10 min, 50 W, 250 psi). LC-MS analysis showed consumption of the starting material L2, and this was confirmed with HPLC (general method A). The reaction mixture was purified with reversed phase HPLC (general method D). The product fractions were collected and dried under vacuum. The product was then redissolved in ethanol, passed through a 0.2 μm syringe filter, and then dried with Centrifan to give L19 a brown waxy material (174.3 mg, yield 86%). 1H-NMR (CD3OD): δ 7.30 (d, 3JHH = 6.7 Hz, 2H, Ar-H), 7.30 (t, 3JHH = 4.5 Hz, 1H, Ar-H), 7.16 (d, 3JHH = 8.2 Hz, 2H, Ar-H), 6.73 (dd, 3JHH = 8.3 Hz, 4JHH = 2.6 Hz, 1H, Ar-H), 6.67 (d, 4JHH = 2.5 Hz, 1H. Ar-H), 4.79 (d, 3JHH = 8.0 Hz, 1H, CHOH), 3.76 (s, 3H, OCH3), 3.65 (s, 3H, OCH3), 3.11 (t, 3JHH = 7.1 Hz, 2H, CH2), 2.88 (m, 4H, CH2), 2.61 (m, 8H, CH2), 1.63 (m, 4H, CH2). 13C{1H}-NMR (CD3OD): δ 174.10 (C=O), 160.31, 142.71, 141.71, 138.65, 137.04, 133.59 (CH), 132.03 (CH), 130.43 (CH), 127.88 (CH), 116.61 (CH), 111.76 (CH), 72.24 (OCH), 63.45 (CN), 60.18 (CN), 56.55 (CN), 55.77 (OCH3), 52.36 (OCH3), 36.29 (CH2), 35.34 (CH2), 30.92 (CH2), 30.62 (CH2), 30.44 (CH2), 27.04 (CH2). HRMS calcd for C25H34NO4S [M + H]+: m/z = 444.2209, found 444.2203. error: ‒1.4 ppm. HPLC (general method A): tR = 6.46 min, purity 97.56%. Chiral HPLC (general method E): (S)-L19, tR = 13.91 min, 50.24%; (R)-L19, tR = 21.48 min, 48.23%.
The enantiomers were resolved and purified with HPLC (general method F). (S)-L19: tR = 14.04 min, 97.24%. [α]D²⁰ = +2.47° (c 1.0, CHCl3). (R)-L19: tR = 20.25 min, 97.71%. [α]D²⁰ = –1.81° (c 1.0, CHCl3).
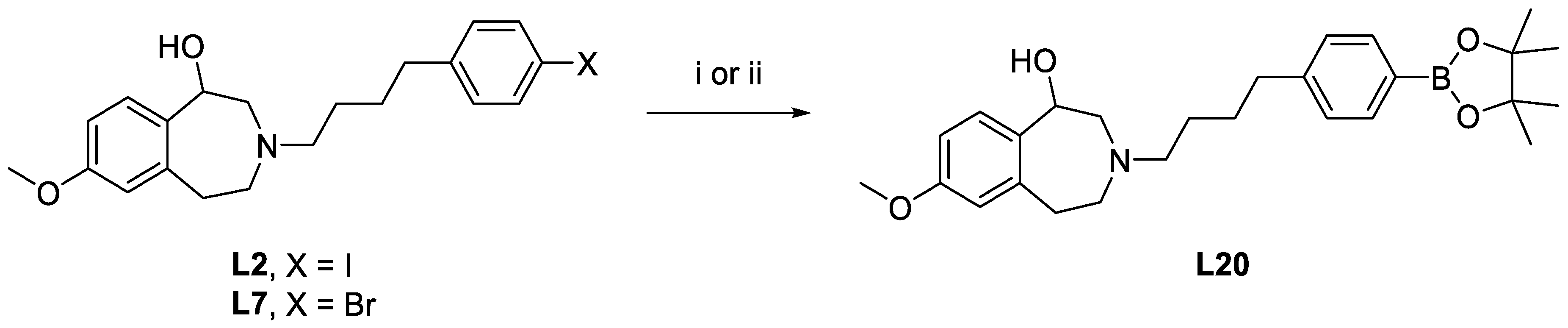
7-Methoxy-3-(4-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)butyl)-2,3,4,5-tetrahydro-1H-benzo[d]azepin-1-ol (L20). Method 1. L2 (105.7 mg, 0.234 mmol), 4,4,4’,4’,5,5,5’,5’-octamethyl-2,2’-bi(1,3,2-dioxaborolane) (86.9 mg, 0.342 mmol), KOAc (83.7 mg, 0.853 mmol), and DPPF-PdCl2·CH2Cl2 (34.8 mg, 0.0426 mmol) were dissolved in DMSO (3.0 mL). The reaction mixture was then heated in a microwave reactor (80 °C, 60 min, 60 W, 250 psi). LC-MS analysis showed consumption of the starting material L2 and this was confirmed with HPLC (general method A). The reaction mixture was purified with reversed phase HPLC (general method D). A saturated NaCl solution was immediately added to each product fraction as they eluted, followed by a saturated NaHCO3 solution to prevent decomposition. The aqueous phase was then extracted with acetonitrile and dried (MgSO4), filtered, and then dried under vacuum. The product was redissolved in ethanol, passed through a 0.2 μm syringe filter, and then dried with Centrifan to give L20 purple wax (39.0 mg, yield 37%). 1H-NMR (CD3CN): δ 7.63 (d, 3JHH = 7.7 Hz, 2H, Ar-H), 7.23 (d, 3JHH = 7.6 Hz, 2H, Ar-H), 7.17 (d, 3JHH = 8.0 Hz, 1H, Ar-H), 6.68 (m, 2H, Ar-H), 4.61 (d, 3JHH = 7.3 Hz, 1H, CHOH), 3.74 (s, 3H, OCH3), 2.85 (m, 1H, CH2), 2.73 (m, 2H, CH2), 2.65 (m, 2H, CH2), 2.58 (m, 2H, CH2), 1.64 (q, 3JHH = 7.6 Hz, 2H, CH2), 1.54 (m, 2H, CH2), 1.31 (s, 12H, CH3). 13C{1H}-NMR (CD3CN): δ 159.27, 146.85, 136.67, 135.12, 128.70 (CH), 128.57 (CH), 117.88 (CH), 116.35 (CH), 110.76 (CH), 84.23 (COC3), 72.02 (COH), 62.06 (CN), 59.21 (CN), 55.98 (CN), 55.35 (OCH3), 36.29 (CH2), 35.94 (CH2), 29.27 (CH2), 26.68 (CH2), 24.77 (CH3). HRMS calcd for C27H39BNO4 [M+H]+: m/z = 452.2972, found 452.2976; error: 0.9 ppm. HPLC (general method A): tR = 7.64 min, purity 76.67%.
Method 2. Compound L7 (18.7 mg, 0.0462 mmol), 4,4,4’,4’,5,5,5’,5’-octamethyl-2,2’-bi(1,3,2-dioxaborolane) (57 mg, 0.224 mmol), KOAc (7.9 mg, 0.0805 mmol), and DPPF-PdCl2·CH2Cl2 (13.2 mg, 0.0162 mmol) were dissolved in DMSO (0.5 mL). The reaction mixture was then heated in a microwave reactor (110 °C, 30 min, 50 W, 250 psi). LC-MS analysis showed consumption of the starting material L7and this was confirmed with HPLC (general method A). The reaction mixture was purified with reversed phase HPLC (general method D) and the collected fractions were dried under vacuum. The product was redissolved in ethanol, passed through a 0.2 μm syringe filter, and then dried with Centrifan to give L20 as a purple wax (11.2 mg, yield 54%).
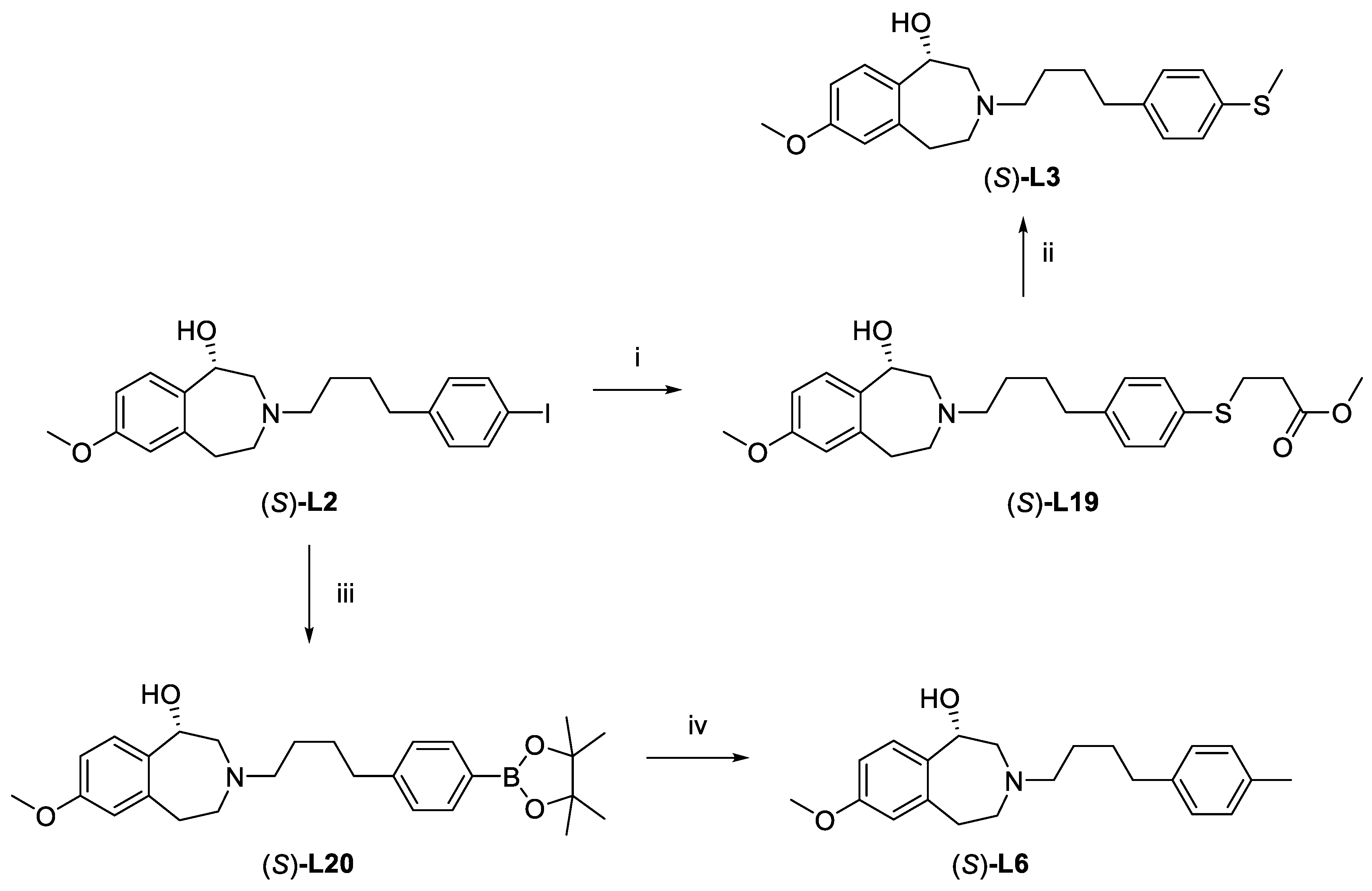
Infrared/Vibrational Circular Dichroism. The enantiomers of L3 were subjected to a computational infrared (IR) and vibrational circular dichroism (VCD) study to allow comparison of computed spectra with those determined experimentally, and to define the absolute configurations of the enantiomers (see Supplementary Information). The first eluting enantiomer in chiral HPLC analysis (general method E) was found to be the S-enantiomer.
Verification of absolute configuration of enantiomers of L6, L2, L19, and L20 by chemical transformations originating from (S)-L3.
Synthesis of (S)-L19 from (S)-L2. Compound (S)-L2 (46.8 mg, 103.7 μmol), methyl 3-((trimethylstannyl)thio)propanoate (26.7 μL, 124.4 μmol), triethylamine (14.5 μL, 103.7 μmol), and [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II) (35.5 mg, 48.52 μmol) were dissolved in acetonitrile (1 mL). The reaction mixture was then heated in a microwave reactor (90 °C. 20 min, 120 W, 250 psi) and then purified with normal phase HPLC (method G) to give compound (S)-L19 as a brown oil (38.8 mg, 84.4%). HPLC (general method E): tR = 14.04 min, purity 97.24%.
Synthesis of (S)-L3 from (S)-L19. Compound (S)-L19 (27.4 mg, 61.77 μmol) in DCM (1 mL) was mixed with methyl iodide (3.86 μL, 61.77 μmol) and 1 M tetra-n-butylammonium hydroxide (TBAOH;129.7 μL, 129.7 μmol) and stirred at rt for 20 min. The reaction mixture was then diluted with DCM (5 mL) and washed thrice with water (10 mL). The solvent was removed by rotary evaporator, redissolved in DCM (1 mL) and purified with HPLC (general method F) to give compound (S)-L3 as a colorless oil (17.3 mg, 75.4%). HPLC (general method E): tR = 8.10 min, purity 98.01%.
Synthesis of (S)-L20 from (S)-L2. Compound (S)-L2 (138 mg, 306 μmol), bis(pinacolato)diborane (200 mg, 788 μmol), potassium acetate (51.2 mg, 521 μmol), and [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II) (62.8 mg, 85.8 μmol) were dissolved in acetonitrile (3 mL). The reaction mixture was then heated in a microwave reactor (80 °C, 20 min, 150 W, 250 psi). and passed through a 0.2 μm syringe filter and purified with normal phase HPLC in three equal portions (method G). Fractions containing the product were immediately neutralized with saturated aqueous sodium bicarbonate (1 mL), extracted with ethyl acetate, and dried to give (S)-L20 (43 mg, 31%) as a purple wax. HPLC (general method E): tR = 6.0 min, purity 93.5%.
Synthesis of (S)-L6 from (S)-L20. Compound (S)-L20 (20.0mg, 44.3 μmol) was dissolved in methanol (0.4 mL). Cesium fluoride (199.4 μmol) in methanol (1 M; 199.4 μL), methyl iodide (3.04 μL, 48.8 μmol) and tetrakis(triphenylphosphine)palladium(0) (5.2 mg, 4.50 μmol) were dissolved in acetonitrile (0.6 mL). The reaction mixture was then heated in a microwave reactor (90 °C, 20 min, 80 W, 250 psi) and then passed through a 0.2 μm syringe filter and purified by reverse phased HPLC (method G) to give (S)-L6 as a brown oil (12.6mg, 83.9%). HPLC (general method E): tR = 5.93 min, purity 98.28%
Methylation of L19 to synthesize L3 under conditions mimicking a11C-labeling reaction. A stock solution of L19 in DMF (1.25 mg/mL) was prepared and 0.4 mL of this solution was transferred to a V-vial. A stock solution of iodomethane in DMF (1% v/v; 1 mL) was prepared and 7.0 µL of this solution was added to the L19 solution and stirred for 2 min. TBAOH in methanol (1 M; 5.0 µL) was then added and this was stirred at RT for 5 min. LC-MS showed complete conversion of L19 into L3. The solution was then dried with Centrifan and the reaction mixture redissolved in ethanol (0.1 mL) for chiral HPLC (general method E) and LC-MS analysis.
Methylation of L20 to L6 under conditions mimicking a11C-labeling reaction. Pd2(dba)3 and P(o,p-tol)3 were mixed in the weight ratio of 1: 1.33. A portion of this mixture (0.6 mg; 0.562 µmol Pd) was taken and placed in a vial. A solution of L20 in methanol (1.25 mg/mL; 0.40 mL) was added, followed by a solution of iodomethane in methanol (10% w/w; 7 µL). A stock solution (20 µL) of cesium fluoride in methanol (1 M) was then added to the reaction mixture, which was heated at 80 oC for 90 min. LC-MS showed consumption of the starting material (L20) and product formation (L6). The reaction mixture was then purified with reversed phase HPLC (general method C) and the product dried with Centrifan. The purified product was analyzed with chiral HPLC (general method E) and LC-MS.