Submitted:
09 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Background
2. Overview of Pancreatic Cystic Lesions
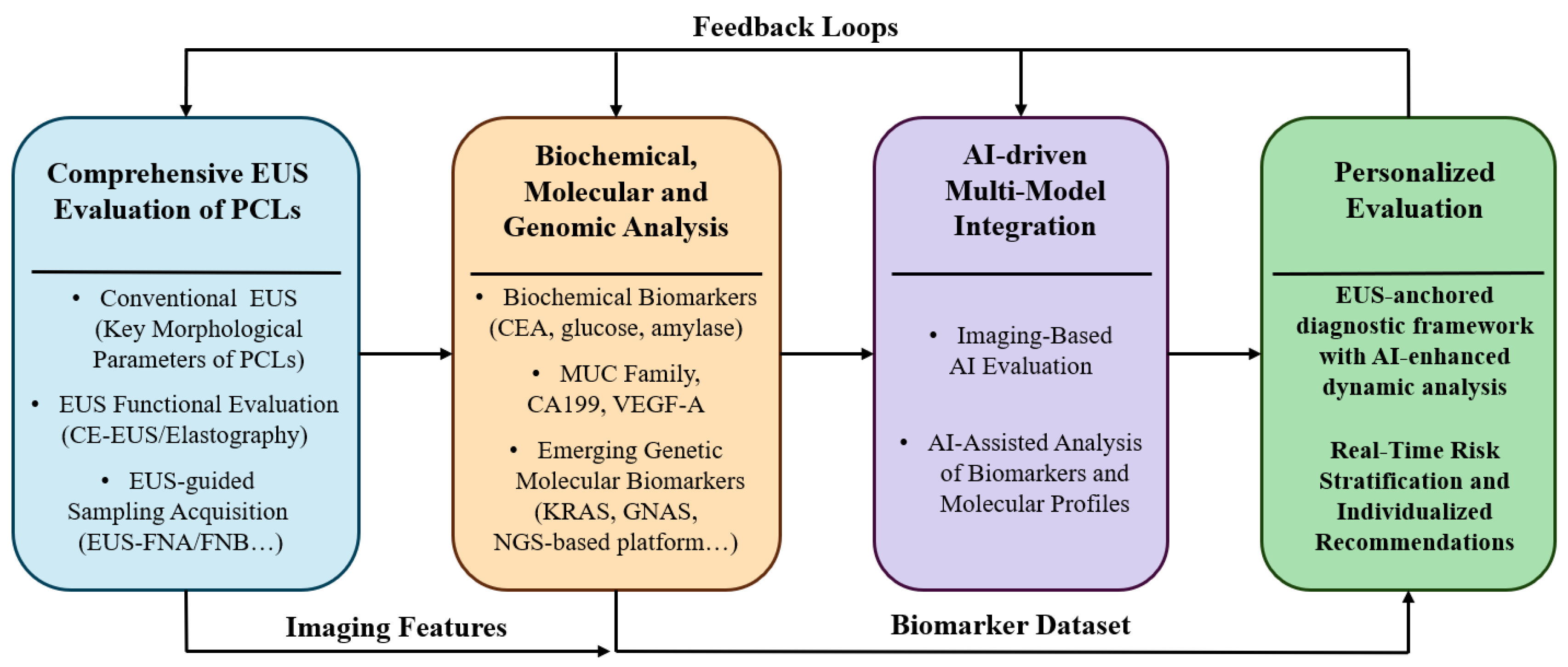
3. Comprehensive EUS Evaluation of Pancreatic Cystic Lesions
3.1. Morphological Evaluation of PCLs
3.2. Functional Evaluation of PCLs
3.3. EUS-Guided Tissue and Fluid Acquisition
4. Molecular Profiling and Biomarker Characterization of Pancreatic Cystic Lesions
4.1. Conventional Biochemical Biomarkers
4.1.1. CEA
4.1.2. Cyst-Fluid Glucose
4.1.3. Cyst-Fluid Amylase
4.2. Cyst-Fluid Biomarkers with Potential Clinical Application Value
4.2.1. Mucin-Related Glycoproteins
4.2.2. CA19-9
4.2.3. VEGF-A
4.3. Emerging Genetic Molecular Testing
4.3.1. Diagnostic Mutations for Cyst Typing
4.3.2. NGS-Based Platforms
4.3.3. Liquid-Biopsy Markers
5. Artificial Intelligence in the Assessment of PCLs
5.1. Imaging-Based Artificial Intelligence for PCLs Evaluation
5.2. AI-Assisted Analysis of Biomarkers and Molecular Profiles in PCLs
5.2.1. AI in Biochemical Biomarker Evaluation
5.2.2. AI in Emerging Molecular and Genomic Biomarker Analysis
6. Multi-Model Approach for Risk Stratification and Personalized Evaluation of PCLs
7. Summary
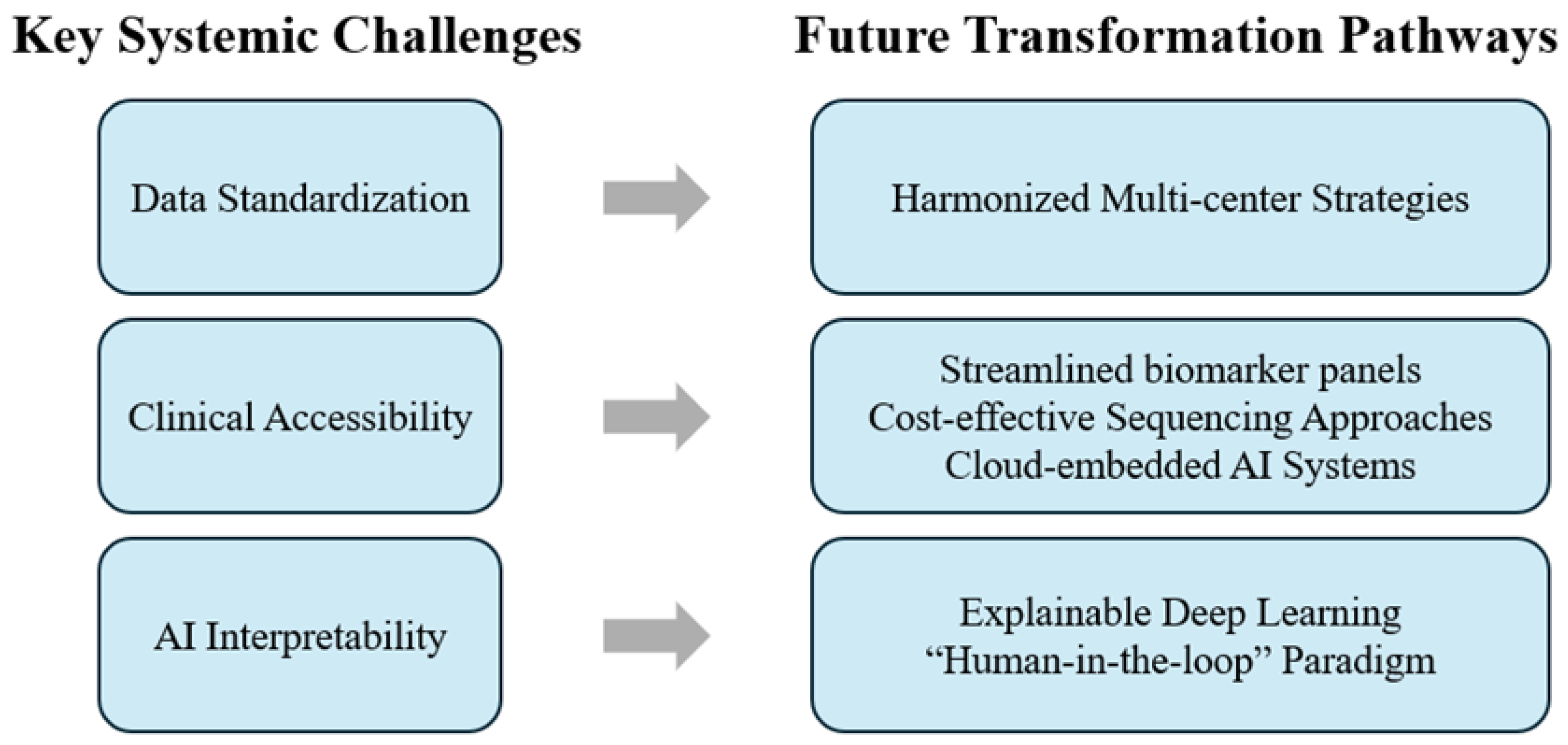
8. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PCLs EUS AI CT MRI IPMNs MCNs SCNs SPNs cNETs CE-EUS EUS-FNA EUS-FNB EUS-TTNB CEA MUC VEGF NGS HGD IC cfDNA miRNA CNNs AUC XAI ML SVM |
Pancreatic Cystic Lesions Endoscopic Ultrasound Artificial Intelligence Computed Tomography Magnetic Resonance Imaging Intraductal Papillary Mucinous Neoplasms Mucinous Cystic Neoplasms Serous Cystic Neoplasms Solid Pseudopapillary Neoplasms cystic Neuroendocrine Tumors Contrast-Enhanced EUS EUS-guided Fine-needle Aspiration EUS-guided Fine-needle Biopsy EUS through-the-needle micro-forceps Biopsy Carcinoembryonic Antigen Mucin-related glycoprotein Vascular Endothelial Growth Factor Next-generation Sequencing High-grade Dysplasia Invasive Cancer cell-free DNA microRNA Convolutional Neural Networks Area Under the curve Explainable AI Machine Learning Support Vector Machines |
| MDPI PCLs EUS AI CT MRI IPMNs MCNs SCNs SPNs cNETs CE-EUS EUS-FNA EUS-FNB EUS-TTNB CEA MUC VEGF NGS HGD IC cfDNA miRNA CNNs AUC XAI ML SVM |
Multidisciplinary Digital Publishing Institute Pancreatic Cystic Lesions Endoscopic Ultrasound Artificial Intelligence Computed Tomography Magnetic Resonance Imaging Intraductal Papillary Mucinous Neoplasms Mucinous Cystic Neoplasms Serous Cystic Neoplasms Solid Pseudopapillary Neoplasms cystic Neuroendocrine Tumors Contrast-Enhanced EUS EUS-guided Fine-needle Aspiration EUS-guided Fine-needle Biopsy EUS through-the-needle micro-forceps Biopsy Carcinoembryonic Antigen Mucin-related glycoprotein Vascular Endothelial Growth Factor Next-generation Sequencing High-grade Dysplasia Invasive Cancer cell-free DNA microRNA Convolutional Neural Networks Area Under the curve Explainable AI Machine Learning Support Vector Machines |
| MDPI PCLs EUS AI CT MRI IPMNs MCNs SCNs SPNs cNETs CE-EUS EUS-FNA EUS-FNB EUS-TTNB CEA MUC VEGF NGS HGD IC cfDNA miRNA CNNs AUC XAI ML SVM |
Multidisciplinary Digital Publishing Institute Pancreatic Cystic Lesions Endoscopic Ultrasound Artificial Intelligence Computed Tomography Magnetic Resonance Imaging Intraductal Papillary Mucinous Neoplasms Mucinous Cystic Neoplasms Serous Cystic Neoplasms Solid Pseudopapillary Neoplasms cystic Neuroendocrine Tumors Contrast-Enhanced EUS EUS-guided Fine-needle Aspiration EUS-guided Fine-needle Biopsy EUS through-the-needle micro-forceps Biopsy Carcinoembryonic Antigen Mucin-related glycoprotein Vascular Endothelial Growth Factor Next-generation Sequencing High-grade Dysplasia Invasive Cancer cell-free DNA microRNA Convolutional Neural Networks Area Under the curve Explainable AI Machine Learning Support Vector Machines |
| MDPI PCLs EUS AI CT MRI IPMNs MCNs SCNs SPNs cNETs CE-EUS EUS-FNA EUS-FNB EUS-TTNB CEA MUC VEGF NGS HGD IC cfDNA miRNA CNNs AUC XAI ML SVM |
Multidisciplinary Digital Publishing Institute Pancreatic Cystic Lesions Endoscopic Ultrasound Artificial Intelligence Computed Tomography Magnetic Resonance Imaging Intraductal Papillary Mucinous Neoplasms Mucinous Cystic Neoplasms Serous Cystic Neoplasms Solid Pseudopapillary Neoplasms cystic Neuroendocrine Tumors Contrast-Enhanced EUS EUS-guided Fine-needle Aspiration EUS-guided Fine-needle Biopsy EUS through-the-needle micro-forceps Biopsy Carcinoembryonic Antigen Mucin-related glycoprotein Vascular Endothelial Growth Factor Next-generation Sequencing High-grade Dysplasia Invasive Cancer cell-free DNA microRNA Convolutional Neural Networks Area Under the curve Explainable AI Machine Learning Support Vector Machines |
References
- Galgano, S.J.; Kambadakone, A.R.; van der Pol, C.B. A Review of Pancreatic Cystic Lesions on Imaging. Advances in Clinical Radiology 2024, 6, 19–30. [Google Scholar] [CrossRef]
- Pan, Y.; Ran, T.; Zhang, X.; Qin, X.; Zhang, Y.; Zhou, C.; Zou, D. Adequacy of EUS–guided fine-needle aspiration and fine-needle biopsy for next-generation sequencing in pancreatic malignancies: A systematic review and meta-analysis. Endoscopic Ultrasound 2024, 13, 366–375. [Google Scholar] [CrossRef]
- Jiang, M.; Peng, L.; Sun, Y.; Li, S.; Li, Z.; Sun, L.; Huang, H.; Zhendong, J. EUS-guided ablation for pancreatic cystic lesions: An updated review. Endoscopic Ultrasound 2025, 10, 1097. [Google Scholar] [CrossRef]
- Quingalahua, E.; Al-Hawary, M.M.; Machicado, J.D. The role of magnetic resonance imaging (MRI) in the diagnosis of pancreatic cystic lesions (PCLs). Diagnostics 2023, 13, 585. [Google Scholar] [CrossRef]
- Yasrab, M.; Kwak, S.J.; Khoshpouri, P.; Fishman, E.K.; Zaheer, A. Misdiagnosis of pancreatic intraductal papillary mucinous neoplasms and the challenge of mimicking lesions: imaging diagnosis and differentiation strategies. Abdominal Radiology 2025, 50, 2241–2257. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.L.; Pitman, M.B. Cytology of cystic lesions of the pancreas: Practical insights, pearls, and pitfalls. Cancer Cytopathology 2025, 133, e70011. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.; Du, C.; He, Z.; Feng, X.; Li, H.; Wang, Z.; Gao, F.; Zhao, Y.; Chai, N.; Linghu, E. Value of EUS-guided through-the-needle biopsy in the diagnosis of pancreatic cystic neoplasms: An 8-year experience. Endoscopic Ultrasound 2024, 13, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Krishna, S.; Abdelbaki, A.; Hart, P.A.; Machicado, J.D. Endoscopic ultrasound-guided needle-based confocal endomicroscopy as a diagnostic imaging biomarker for intraductal papillary mucinous neoplasms. Cancers 2024, 16, 1238. [Google Scholar] [CrossRef]
- Dahiya, D.S.; Shah, Y.R.; Ali, H.; Chandan, S.; Gangwani, M.K.; Canakis, A.; Ramai, D.; Hayat, U.; Pinnam, B.S.M.; Iqbal, A. Basic principles and role of endoscopic ultrasound in diagnosis and differentiation of pancreatic cancer from other pancreatic lesions: a comprehensive review of endoscopic ultrasound for pancreatic cancer. Journal of Clinical Medicine 2024, 13, 2599. [Google Scholar] [CrossRef]
- Moond, V.; Mohan, B.P.; Diehl, D.; Adler, D.G. EUS-guided through-the-needle microforceps biopsy for pancreatic cysts: Why no widespread adoption? Endoscopic Ultrasound 2024, 13, 127–128. [Google Scholar] [CrossRef]
- Hickman, K.; Sadler, T.; Zhang, T.; Boninsegna, E.; Majcher, V.; Godfrey, E. Pancreatic cystic lesions and the role of contrast enhanced endoscopic ultrasound. Clinical Radiology 2022, 77, 418–427. [Google Scholar] [CrossRef]
- Lipkin, J.; McGrath, K. EUS for pancreatic cysts. Endoscopic Ultrasonography 2024, 150–160. [Google Scholar]
- Pușcașu, C.I.; Rimbaş, M.; Mateescu, R.B.; Larghi, A.; Cauni, V. Advances in the diagnosis of pancreatic cystic lesions. Diagnostics 2022, 12, 1779. [Google Scholar] [CrossRef]
- Rogowska, J.; Semeradt, J.; Durko, Ł.; Małecka-Wojciesko, E. Diagnostics and Management of Pancreatic Cystic Lesions—New Techniques and Guidelines. Journal of Clinical Medicine 2024, 13, 4644. [Google Scholar] [CrossRef]
- Singh, R.R.; Gopakumar, H.; Sharma, N.R. Diagnosis and management of pancreatic cysts: a comprehensive review of the literature. Diagnostics 2023, 13, 550. [Google Scholar] [CrossRef]
- Ciaravino, V.; Calbi, R.; Giugliano, A.; Geraci, L.; Tomaiuolo, L.; Longo, C.; De Robertis, R.; D’Onofrio, M. Pseudocysts and Other Cystic Lesions. In Imaging and Pathology of Pancreatic Neoplasms: A Pictorial Atlas; Springer, 2022; pp. 393–413. [Google Scholar]
- Ozcan, K.; Klimstra, D.S. A review of mucinous cystic and Intraductal neoplasms of the Pancreatobiliary tract: recent advances. Archives of Pathology & Laboratory Medicine 2022, 146, 298–311. [Google Scholar]
- Coban, S.; Basar, O.; Brugge, W.R. Pancreatic cystic neoplasms. Gastroenterology Clinics 2022, 51, 537–559. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Ahmed, N.; Krishna, S.G. Pancreatic cystic lesions: a focused review on cyst clinicopathological features and advanced diagnostics. Diagnostics 2022, 13, 65. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Chennatt, J.J.; Mandal, C.; Gupta, J.; Krishnasamy, S.; Bose, B.; Solanki, P.; Singh, S.K.; Gupta, S.; Bose, B., Sr. Approach to cystic lesions of the pancreas: Review of literature. Cureus 2023, 15. [Google Scholar] [CrossRef]
- Aunan, J.R.; Al-Saiddi, M.S.; Stutchfield, B.; Jamieson, N.B.; Søreide, K. Pancreatic cystic lesions and risk of cancer. In Textbook of pancreatic cancer: principles and practice of surgical oncology; Springer, 2021; pp. 777–797. [Google Scholar]
- Buerlein, R.C.; Shami, V.M. Management of pancreatic cysts and guidelines: What the gastroenterologist needs to know. Therapeutic Advances in Gastrointestinal Endoscopy 2021, 14, 26317745211045769. [Google Scholar] [CrossRef]
- Ogura, T.; Hijioka, S.; Hara, K.; Nishioka, N.; Okuda, A.; Ueno, S.; Nishikawa, H.; Yamada, M.; Nagashio, Y.; Hisada, Y.; et al. Multicenter, randomized controlled trial of EUS-guided fine-needle biopsy using a fork-tip needle with macroscopic or rapid on-site evaluation for pancreatic lesions (H2O trial). Endoscopic Ultrasound 2024, 13, 300–305. [Google Scholar] [CrossRef]
- Yang, S.; Chen, T.; Cui, M.; Hu, Y. Genomic alterations in pancreatic cystic neoplasms: from molecular characterization to precision clinical management-conventional review. International Journal of Surgery 2025, 10, 7053–7071. [Google Scholar] [CrossRef]
- Pawłowski, W.; Stefański, M.; Włodarczyk, B.; Durko, Ł.; Małecka-Wojciesko, E. Pancreatic Cancer Detection in Intraductal Papillary Mucinous Neoplasm (IPMN)—New Insights. Cancers 2025, 17, 3341. [Google Scholar] [CrossRef]
- Mulqui, M.V.; Caillol, F.; Ratone, J.P.; Hoibian, S.; Dahel, Y.; Meunier, É.; Archimbaud, C.; Giovannini, M. Detective flow imaging versus contrast-enhanced EUS in solid pancreatic lesions. Endoscopic Ultrasound 2024, 13, 248–252. [Google Scholar] [CrossRef]
- Conti, C.B.; Mulinacci, G.; Salerno, R.; Dinelli, M.E.; Grassia, R. Applications of endoscopic ultrasound elastography in pancreatic diseases: From literature to real life. World Journal of Gastroenterology 2022, 28, 909. [Google Scholar] [CrossRef] [PubMed]
- Bruni, A.; Tuccillo, L.; Dell’Anna, G.; Mandarino, F.V.; Lisotti, A.; Maida, M.; Ricci, C.; Fuccio, L.; Eusebi, L.H.; Marasco, G. Endoscopic Ultrasound-Guided Pancreatic Cystic Fluid Biochemical and Genetic Analysis for the Differentiation Between Mucinous and Non-Mucinous Pancreatic Cystic Lesions. Journal of Clinical Medicine 2025, 14, 3825. [Google Scholar] [CrossRef] [PubMed]
- Rift, C.V.; Scheie, D.; Toxværd, A.; Kovacevic, B.; Klausen, P.; Vilmann, P.; Hansen, C.P.; Lund, E.L.; Hasselby, J.P. Diagnostic accuracy of EUS-guided through-the-needle-biopsies and simultaneously obtained fine needle aspiration for cytology from pancreatic cysts: A systematic review and meta-analysis. Pathology-Research and Practice 2021, 220, 153368. [Google Scholar] [CrossRef] [PubMed]
- Iwashita, T.; Uemura, S.; Shimizu, M. Endoscopic ultrasound-guided fine-needle aspiration for pancreatic cystic lesions: a comprehensive review. Journal of medical ultrasonics 2024, 51, 219–226. [Google Scholar] [CrossRef]
- Yousri, M.; Abusinna, E.; Tahoun, N.; Okasha, H.H.; El-Habashi, A.H. A comparative study of the diagnostic utility of endoscopic ultrasound-guided fine needle aspiration cytology (EUS-FNA) versus endoscopic ultrasound-guided fine needle biopsy (EUS-FNB) in pancreatic and non-pancreatic lesions. Asian Pacific Journal of Cancer Prevention: APJCP 2022, 23, 2151. [Google Scholar] [CrossRef]
- Ding, C; Yang, J-f; Wang, X; Zhou, Y-f; Gu, Y; Liu, Q; Shen, H-z; Zhang, X-f. Diagnostic yield of endoscopic ultrasound-guided fine-needle aspiration-based cytology for distinguishing malignant and benign pancreatic cystic lesions: A systematic review and meta-analysis. PloS one 2025, 20, e0314825. [Google Scholar] [CrossRef]
- Mota, J.; Ribeiro, T.; Bellocchi, M.C.C.; De Pretis, N.; Frulloni, L.; Lopes, J.; Sina, S.; Dhar, J.; Samanta, J.; Macedo, G. Endoscopic Ultrasound Through-The-Needle Biopsy of Pancreatic Cystic Neoplasms: Update on Indications, Safety Profile, and Research Directions. Best Practice & Research Clinical Gastroenterology 2025, 102058. [Google Scholar] [CrossRef]
- Sbeit, W.; Kadah, A.; Shahin, A.; Khoury, T. The yield of string sign in differentiating mucinous from non-mucinous pancreatic cysts: A retrospective cross-sectional study. Medicina 2021, 57, 716. [Google Scholar] [CrossRef] [PubMed]
- Triantopoulou, C.; Gourtsoyianni, S.; Karakaxas, D.; Delis, S. Intraductal papillary mucinous neoplasm of the pancreas: a challenging diagnosis. Diagnostics 2023, 13, 2015. [Google Scholar] [CrossRef]
- Nista, E.C.; Schepis, T.; Candelli, M.; Giuli, L.; Pignataro, G.; Franceschi, F.; Gasbarrini, A.; Ojetti, V. Humoral predictors of malignancy in IPMN: a review of the literature. International Journal of Molecular Sciences 2021, 22, 12839. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; He, Z.; Gao, F.; Li, L.; Han, K.; Feng, X.; Wang, X.; Tang, P.; Chai, N.; Linghu, E. Factors affecting the diagnostic value of liquid-based cytology by EUS-FNA in the diagnosis of pancreatic cystic neoplasms. Endoscopic Ultrasound 2024, 13, 94–99. [Google Scholar] [CrossRef]
- Rogowska, J.O.; Durko, Ł.; Malecka-Wojciesko, E. The latest advancements in diagnostic role of endosonography of pancreatic lesions. Journal of Clinical Medicine 2023, 12, 4630. [Google Scholar] [CrossRef]
- Oppong, K.W.; Bekkali, N.L.; Leeds, J.S.; Johnson, S.J.; Nayar, M.K.; Darné, A.; Egan, M.; Bassett, P.; Haugk, B. Fork-tip needle biopsy versus fine-needle aspiration in endoscopic ultrasound-guided sampling of solid pancreatic masses: a randomized crossover study. Endoscopy 2020, 52, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Noh, D.H.; Choi, K.; Gu, S.; Cho, J.; Jang, K.-T.; Woo, Y.S.; Lee, K.T.; Lee, J.K.; Lee, K.H. Comparison of 22-gauge standard fine needle versus core biopsy needle for endoscopic ultrasound-guided sampling of suspected pancreatic cancer: a randomized crossover trial. Scandinavian Journal of Gastroenterology 2018, 53, 94–99. [Google Scholar] [CrossRef]
- Luthra, A.K.; Krishna, S.G. Through-the-needle forceps biopsy for pancreatic cystic lesions: multiple meta-analyses but limited prospective data. Endoscopy International Open 2020, 8, E1134–E1136. [Google Scholar] [CrossRef]
- Facciorusso, A.; Ramai, D.; Gkolfakis, P.; Shapiro, A.; Arvanitakis, M.; Lisotti, A.; Triantafyllou, K.; Fusaroli, P.; Papanikolaou, I.S.; Crinò, S.F. Through-the-needle biopsy of pancreatic cystic lesions: Current evidence and implications for clinical practice. Expert Review of Medical Devices 2021, 18, 1165–1174. [Google Scholar] [CrossRef]
- Deris Zayeri, Z.; Parsi, A.; Shahrabi, S.; Kargar, M.; Davari, N.; Saki, N. Epigenetic and metabolic reprogramming in inflammatory bowel diseases: diagnostic and prognostic biomarkers in colorectal cancer. Cancer Cell International 2023, 23, 264. [Google Scholar] [CrossRef]
- Shockley, K.E.; To, B.; Chen, W.; Lozanski, G.; Cruz-Monserrate, Z.; Krishna, S.G. The role of genetic, metabolic, inflammatory, and immunologic mediators in the progression of intraductal papillary mucinous neoplasms to pancreatic adenocarcinoma. Cancers 2023, 15, 1722. [Google Scholar] [CrossRef] [PubMed]
- Faias, S.; Cravo, M.; Chaves, P.; Pereira, L. Comparative analysis of glucose and carcinoembryonic antigen in the diagnosis of pancreatic mucinous cysts: a systematic review and meta-analysis. Gastrointestinal endoscopy 2021, 94, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Gorris, M.; Dijk, F.; Farina, A.; Halfwerk, J.B.; Hooijer, G.K.; Lekkerkerker, S.J.; Voermans, R.P.; Wielenga, M.C.; Besselink, M.G.; van Hooft, J.E. Validation of combined carcinoembryonic antigen and glucose testing in pancreatic cyst fluid to differentiate mucinous from non-mucinous cysts. Surgical Endoscopy 2023, 37, 3739–3746. [Google Scholar] [CrossRef]
- Williet, N.; Caillol, F.; Karsenti, D.; Abou-Ali, E.; Camus, M.; Belle, A.; Chaput, U.; Levy, J.; Ratone, J.-P.; Tournier, Q.; et al. The level of glucose in pancreatic cyst fluid is more accurate than carcinoembryonic antigen to identify mucinous tumors: A French multicenter study. Endoscopic Ultrasound 2023, 12, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Krasinskas, A.M.; Oakley, G.J.; Bagci, P.; Jang, K.-T.; Kuan, S.-F.; Reid, M.D.; Erbarut, I.; Adsay, V. “Simple mucinous cyst” of the pancreas: a clinicopathologic analysis of 39 examples of a diagnostically challenging entity distinct from intraductal papillary mucinous neoplasms and mucinous cystic neoplasms. The American Journal of Surgical Pathology 2017, 41, 121–127. [Google Scholar] [CrossRef]
- You, L.; Ma, L.; Zhao, W.; Zhao, Y.; Dai, M. Emerging role of tumor markers and biochemistry in the preoperative invasive assessment of intraductal papillary mucinous neoplasm of the pancreas. Clinica Chimica Acta 2016, 454, 89–93. [Google Scholar] [CrossRef]
- Goonetilleke, K.; Siriwardena, A. Systematic review of carbohydrate antigen (CA 19-9) as a biochemical marker in the diagnosis of pancreatic cancer. European Journal of Surgical Oncology (EJSO) 2007, 33, 266–270. [Google Scholar] [CrossRef]
- Keane, M.G. The development of novel diagnostic markers and treatments for cystic lesions of the pancreas. UCL (University College London) 2025. [Google Scholar]
- Carr, R.A.; Yip-Schneider, M.T.; Dolejs, S.; Hancock, B.A.; Wu, H.; Radovich, M.; Schmidt, M.C. Pancreatic cyst fluid vascular endothelial growth factor A and carcinoembryonic antigen: a highly accurate test for the diagnosis of serous cystic neoplasm. Journal of the American College of Surgeons 2017, 225, 93–100. [Google Scholar] [CrossRef]
- Yip-Schneider, M.T.; Wu, H.; Dumas, R.P.; Hancock, B.A.; Agaram, N.; Radovich, M.; Schmidt, C.M. Vascular endothelial growth factor, a novel and highly accurate pancreatic fluid biomarker for serous pancreatic cysts. Journal of the American College of Surgeons 2014, 218, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Singhi, A.D.; McGrath, K.; Brand, R.E.; Khalid, A.; Zeh, H.J.; Chennat, J.S.; Fasanella, K.E.; Papachristou, G.I.; Slivka, A.; Bartlett, D.L. Preoperative next-generation sequencing of pancreatic cyst fluid is highly accurate in cyst classification and detection of advanced neoplasia. Gut 2018, 67, 2131–2141. [Google Scholar] [CrossRef]
- Ribeiro, P.M.G.M. The Contribution of Endoscopic Ultrasound and Biomarkers in the Management of Pancreatic Adenocarcinoma and its Precursor Lesions. In Universidade do Porto (Portugal); 2022. [Google Scholar]
- McCarty, T.R.; Paleti, S.; Rustagi, T. Molecular analysis of EUS-acquired pancreatic cyst fluid for KRAS and GNAS mutations for diagnosis of intraductal papillary mucinous neoplasia and mucinous cystic lesions: a systematic review and meta-analysis. Gastrointestinal Endoscopy 2021, 93, 1019–1033. e1015. [Google Scholar] [CrossRef] [PubMed]
- Nikas, I.P.; Mountzios, G.; Sydney, G.I.; Ioakim, K.J.; Won, J.-K.; Papageorgis, P. Evaluating pancreatic and biliary neoplasms with small biopsy-based next generation sequencing (NGS): doing more with less. Cancers 2022, 14, 397. [Google Scholar] [CrossRef]
- Baydogan, S.; Mohindroo, C.; Hasanov, M.; Montiel, M.F.; Quesada, P.; Cazacu, I.M.; Luzuriaga Chavez, A.A.; Mork, M.E.; Dong, W.; Feng, L.; et al. New-onset diabetes is a predictive risk factor for pancreatic lesions in high-risk individuals: An observational cohort study. Endoscopic Ultrasound 2024, 13, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.R.; Bardhi, O.; Polanco, P.; Ellis, D.; Bishop, C.; Coleman, V.; Foley, B.; Khatri, G.; Goldschmiedt, M.; Mansour, J.; et al. Clinical utility of incorporating next-generation sequencing results in the management algorithm of pancreatic cysts. Gastrointestinal Endoscopy 2025, 102, 223–232.e3. [Google Scholar] [CrossRef]
- Paziewska, A.; Polkowski, M.; Goryca, K.; Karczmarski, J.; Wiechowska-Kozlowska, A.; Dabrowska, M.; Mikula, M.; Ostrowski, J. Mutational mosaics of cell-free DNA from pancreatic cyst fluids. Digestive Diseases and Sciences 2020, 65, 2294–2301. [Google Scholar] [CrossRef]
- Lee, L.S.; Szafranska-Schwarzbach, A.E.; Wylie, D.; Doyle, L.A.; Bellizzi, A.M.; Kadiyala, V.; Suleiman, S.; Banks, P.A.; Andruss, B.F.; Conwell, D.L. Investigating microRNA expression profiles in pancreatic cystic neoplasms. Clinical and translational gastroenterology 2014, 5, e47. [Google Scholar] [CrossRef]
- Seyithanoglu, D.; Durak, G.; Keles, E.; Medetalibeyoglu, A.; Hong, Z.; Zhang, Z.; Taktak, Y.B.; Cebeci, T.; Tiwari, P.; Velichko, Y.S. Advances for Managing Pancreatic Cystic Lesions: Integrating Imaging and AI Innovations. Cancers 2024, 16, 4268. [Google Scholar] [CrossRef]
- Alnaggar, O.A.M.F.; Jagadale, B.N.; Saif, M.A.N.; Ghaleb, O.A.; Ahmed, A.A.; Aqlan, H.A.A.; Al-Ariki, H.D.E. Efficient artificial intelligence approaches for medical image processing in healthcare: comprehensive review, taxonomy, and analysis. Artificial Intelligence Review 2024, 57, 221. [Google Scholar] [CrossRef]
- Nguon, L.S.; Seo, K.; Lim, J.-H.; Song, T.-J.; Cho, S.-H.; Park, J.-S.; Park, S. Deep learning-based differentiation between mucinous cystic neoplasm and serous cystic neoplasm in the pancreas using endoscopic ultrasonography. Diagnostics 2021, 11, 1052. [Google Scholar] [CrossRef]
- Yashika, J.; Ohno, E.; Ishikawa, T.; Iida, T.; Suzuki, H.; Uetsuki, K.; Yamada, K.; Yoshikawa, M.; Gibo, N.; Shimoyama, Y. Utility of multiphase contrast enhancement patterns on CEH-EUS for the differential diagnosis of IPMN-derived and conventional pancreatic cancer. Pancreatology 2021, 21, 390–396. [Google Scholar] [CrossRef]
- Lee, L.S.; Andersen, D.K.; Ashida, R.; Brugge, W.R.; Canto, M.I.; Chang, K.J.; Chari, S.T.; DeWitt, J.; Hwang, J.H.; Khashab, M.A. Endoscopic ultrasound and related technologies for the diagnosis and treatment of pancreatic disease-Research gaps and opportunities: Summary of a National Institute of Diabetes and Digestive and Kidney Diseases Workshop. Pancreas 2017, 46, 1242–1250. [Google Scholar] [CrossRef]
- Cui, S.; Tang, T.; Su, Q.; Wang, Y.; Shu, Z.; Yang, W.; Gong, X. Radiomic nomogram based on MRI to predict grade of branching type intraductal papillary mucinous neoplasms of the pancreas: a multicenter study. Cancer Imaging 2021, 21, 26. [Google Scholar] [CrossRef]
- Ma, Z. Towards reliable radiomics modeling: a multi-institutional multi-modality feature repeatability study on head and neck cancer patients. 2025. [Google Scholar]
- Alharthi, A.; Alqurashi, A.; Alharbi, T.; Alammar, M.; Aldosari, N.; Bouchekara, H.; Shaaban, Y.; Shahriar, M.S.; Ayidh, A.A. The Role of Explainable AI in Revolutionizing Human Health Monitoring: A Review. arXiv. 2024.
- Mahajan, S.; Siyu, S.; Bhutani, M.S. What can artificial intelligence do for EUS? Endoscopic Ultrasound 2025, 14, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, K.; Lingasamy, P.; Khatun, M.; Sudhakaran, S.L.; Salumets, A.; Sundararajan, V.; Modhukur, V. Artificial Intelligence in cancer epigenomics: a review on advances in pan-cancer detection and precision medicine. Epigenetics & Chromatin 2025, 18, 35. [Google Scholar] [PubMed]
- Huang, W.; Xu, Y.; Li, Z.; Li, J.; Chen, Q.; Huang, Q.; Wu, Y.; Chen, H. Enhancing noninvasive pancreatic cystic neoplasm diagnosis with multimodal machine learning. Scientific Reports 2025, 15, 16398. [Google Scholar] [CrossRef] [PubMed]
- Tacelli, M.; Lauri, G.; Tabacelia, D.; Tieranu, C.G.; Arcidiacono, P.G.; Săftoiu, A. Integrating artificial intelligence with endoscopic ultrasound in the early detection of bilio-pancreatic lesions: Current advances and future prospects. Best Practice & Research Clinical Gastroenterology 2025, 74, 101975. [Google Scholar] [CrossRef]
- Yoon, J.; Yang, J.; Lee, H.S.; Kim, M.J.; Park, V.Y.; Rho, M.; Yoon, J.H. AI analytics can be used as imaging biomarkers for predicting invasive upgrade of ductal carcinoma in situ. Insights into Imaging 2024, 15, 100. [Google Scholar] [CrossRef] [PubMed]
- Shipley, L.C.; Ahmed, A.M. New and emerging technology in the diagnosis and treatment of pancreatic cysts. Translational Gastroenterology and Hepatology 2022, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Giannone, F.; Crippa, S.; Aleotti, F.; Palumbo, D.; Belfiori, G.; Partelli, S.; Lena, M.S.; Capurso, G.; Petrone, M.C.; De Cobelli, F. Improving diagnostic accuracy and appropriate indications for surgery in pancreatic cystic neoplasms: the role of EUS. Gastrointestinal Endoscopy 2022, 96, 648–656. e642. [Google Scholar] [CrossRef] [PubMed]
- Overbeek, K.A.; Cahen, D.L.; Bruno, M.J. The role of endoscopic ultrasound in the detection of pancreatic lesions in high-risk individuals. Familial Cancer 2024, 23, 279–293. [Google Scholar] [CrossRef]
- Gonda, T.A.; Cahen, D.L.; Farrell, J.J. Pancreatic cysts. New England Journal of Medicine 2024, 391, 832–843. [Google Scholar] [CrossRef]
| Features | BD-IPMNs | MD-IPMN or MT- IPMNs |
MCNs | SCNs | cNETs | SPNs |
|---|---|---|---|---|---|---|
| Age (years) | 50-70 | 50-70 | 30-50 | 60-80 | 50-60 | 20-40 |
| Gender | F=M | F=M | F (>95%) | F (70%) | M>F | F (>80%) |
| Location in pancreas | 50% in head and uncinate |
Any | 90% in body and tail |
Any | Any | Any |
| Clinical symptoms | Mostly no symptom when lesions are small | Mostly no symptom when lesions are small | Up to 50% of cases (might associate with compressive symptoms) | Up to 50% of cases (might associate compressive symptoms) | 90% asymptomatic. Functional symptoms (depend on secreted hormone) |
Mostly no. Abdominal pain or discomfort (37%) |
| Calcification | No | No | Rare peripheral calcification | Central calcification in 30-40% | No | Irregular |
| Malignant potential | Yes | Yes | Yes | None | Irregular | Irregular |
| EUS morphological features | Anechoic multiloculated lesion |
MD-IPMN: dilation of MPD (segmental or diffuse) MT-IPMN: dilation of MPD and BD- IPMN |
Single well- defined round shape with visible wall (pseudocapsule) and clear contours ‘orange-like’ or ‘cyst-in-cyst’ |
Multi- loculated lesions with irregular thin capsule |
Round and well-demarcated hypoechoic solid lesions with an anechoic area of cystic degeneration |
Single well- defined encapsulated with regular margins, hypoechoic solid or mixed |
| Glucose | Low | Low | Low | Normal | - | - |
| Amylase(U/L) | >250 | >250 | <250 | <250 | - | >250 |
| CEA (ng/mL) | >192 | >192 | >192 | <5 | <5 | <5 |
| KRAS mutation | Yes | Yes | Yes | Very rare | None | |
| GNAS mutation | - | Yes | Yes | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




