Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction and Clinical Significance
2. Case Presentation
2.1. Medications
2.2. Present Illness 2.3. Physical Examination
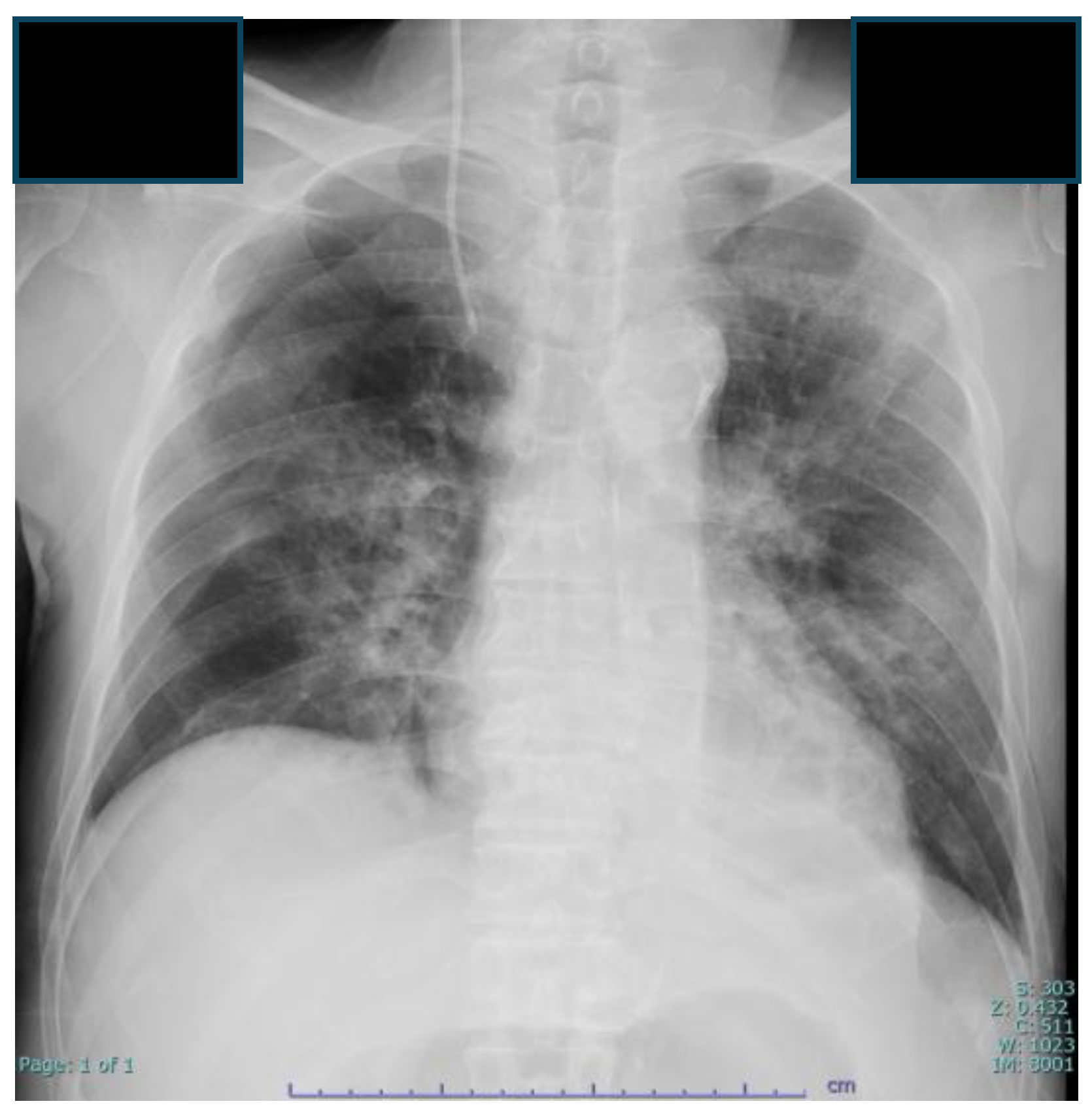
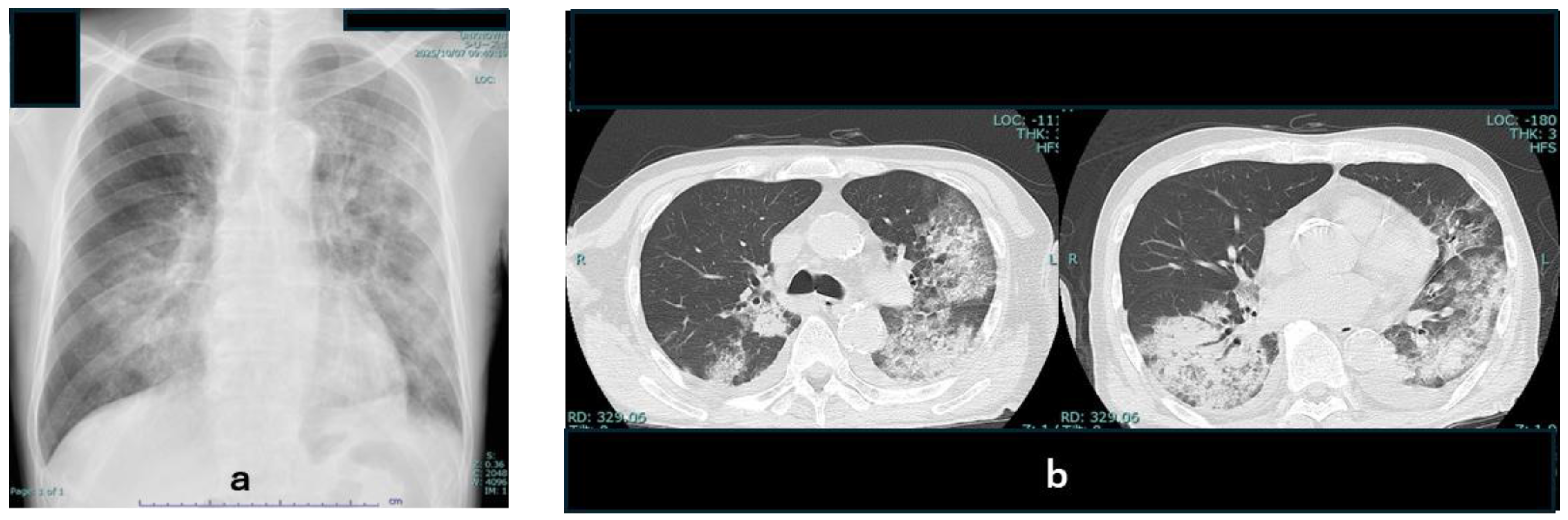
2.4. Laboratory and Imaging Findings
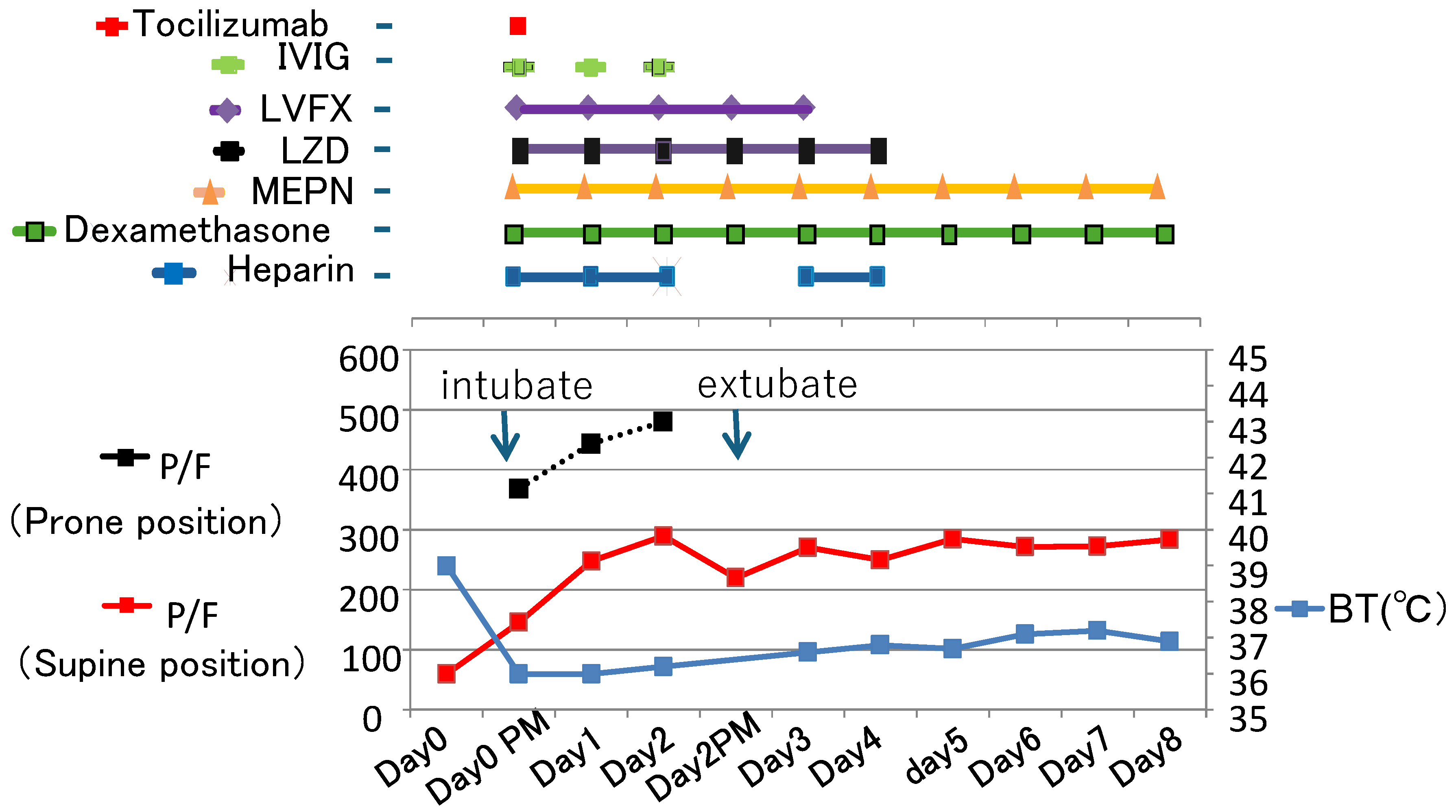
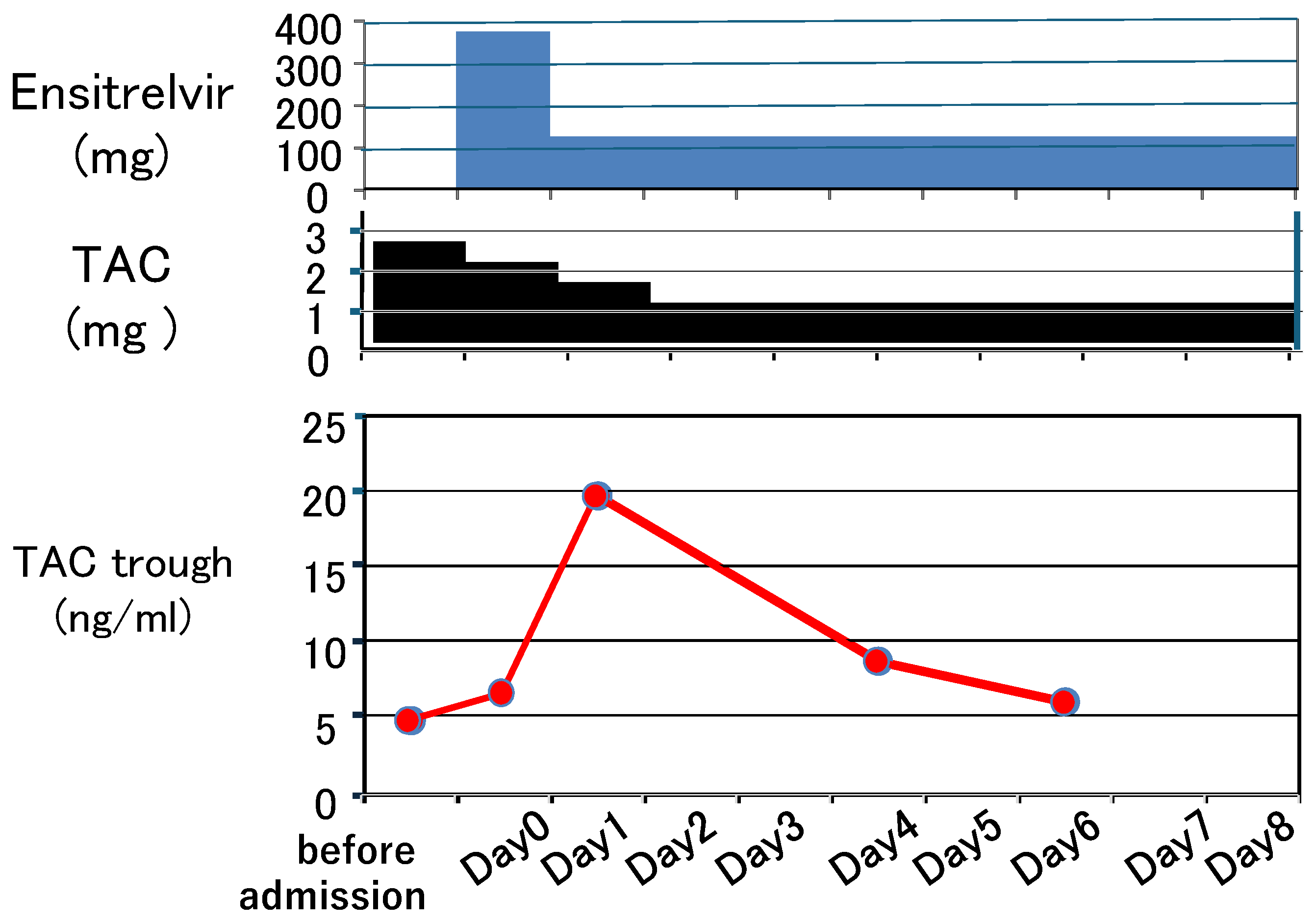
3. Clinical Course and Management (Figure 2)
| Assay primer/probe | Reference Range | Pre | post |
|---|---|---|---|
| Ct value Gene E | >40 | 22.2 | 28.2 |
| Ct value GeneN2 | >40 | 27.9 | 36.4 |
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TCA | tacrolimus |
| ESV | Ensitrelvir |
| CYP | cytochrome P450 |
| AUC | area under the blood concentration time curve |
| ARDS | Acute Respiratory Distress Syndrome |
References
- Wei, M.; Yang, N.; Wang, F.; Zhao, G.; Gao, H.; Li, Y. Epidemiology of Coronavirus Disease 2019 (COVID-19) Caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Disaster Med Public Health Prep 2020, 14, 796–804. [CrossRef]
- WHO. With the international public health emergency ending, WHO/Europe launches its transition plan for COVID-19. 2023, doi:https://www.who.int/europe/news/item/12-06-2023-with-the-international-public-health-emergency-ending.
- WHO. Six-years-after-covid-19-s-global-alarm-is-the-world-better prepared-for-the-next-pandemic. 2026.
- CDC. Clinical-care/underlying-condition. doi:clinical-care/underlying-condition.
- Bank, D. DB00864 – Acetaminophen (Paracetamol). doi:https://go.drugbank.com/drugs/DB00864.
- Barten, M.J.; van Gelder, T.; Gummert, J.F.; Boeke, K.; Shorthouse, R.; Billingham, M.E.; Morris, R.E. Pharmacodynamics of mycophenolate mofetil after heart transplantation: new mechanisms of action and correlations with histologic severity of graft rejection. Am J Transplant 2002, 2, 719–732. [CrossRef]
- Vrij, C.; Bogaerts, K.; Vermeersch, P.; Lagrou, K.; Molenberghs, G.; Rega, F.; Ceulemans, L.J.; Van Raemdonck, D.; Jochmans, I.; Monbaliu, D.; et al. Risk factors for SARS-CoV-2 infection and severe COVID-19 in unvaccinated solid organ transplant recipients. Sci Rep 2024, 14, 26465. [CrossRef]
- Lai, Q.; Spoletini, G.; Bianco, G.; Graceffa, D.; Agnes, S.; Rossi, M.; Lerut, J. SARS-CoV2 and immunosuppression: A double-edged sword. Transpl Infect Dis 2020, 22, e13404. [CrossRef]
- IDSA. IDSA Guidelines on the Treatment and Management of Patients with COVID-19; 2025.
- Brosh-Nissimov, T. Refining COVID-19 care for immunocompromised patients. Lancet Infect Dis 2025, 25, 1167–1168. [CrossRef]
- Miyata, Y.; Yamaguchi, R.; Yamamoto, T.; Kishida, T.; Ikeuchi, K.; Harada, H.; Tsutsumi, T.; Fujio, K.; Takada, T. Drug-drug interaction between ensitrelvir and tacrolimus in a patient undergoing treatment for COVID-19: a case report. J Pharm Health Care Sci 2025, 11, 3. [CrossRef]
- Naganawa, H.; Katada, Y.; Nakagawa, S.; Umemura, K.; Ishimura, H.; Kajiwara, M.; Endo, H.; Sugimoto, M.; Katsube, Y.; Kotani, K.; et al. Influence of ensitrelvir or nirmatrelvir/ritonavir on tacrolimus clearance in kidney transplant recipients: a single-center case series. J Pharm Health Care Sci 2024, 10, 37. [CrossRef]
- Shimizu, R.; Matsuzaki, T.; Oka, R.; Sonoyama, T.; Fukuhara, T.; Kuwata, A.; Matsuo, Y.; Kubota, R. Evaluation of Drug-Drug Interactions of Ensitrelvir, a SARS-CoV-2 3CL Protease Inhibitor, With Transporter Substrates Based on In Vitro and Clinical Studies. J Clin Pharmacol 2023, 63, 918–927. [CrossRef]
- Ohno, Y.; Hisaka, A.; Suzuki, H. General framework for the quantitative prediction of CYP3A4-mediated oral drug interactions based on the AUC increase by coadministration of standard drugs. Clin Pharmacokinet 2007, 46, 681–696. [CrossRef]
- Gastine, S.; Obiero, C.; Kane, Z.; Williams, P.; Readman, J.; Murunga, S.; Thitiri, J.; Ellis, S.; Correia, E.; Nyaoke, B.; et al. Simultaneous pharmacokinetic/pharmacodynamic (PKPD) assessment of ampicillin and gentamicin in the treatment of neonatal sepsis. J Antimicrob Chemother 2022, 77, 448–456. [CrossRef]
- Tan, C.M.; Juurlink, D.N. Navigating Drug Interactions with Proton Pump Inhibitors. JAMA Netw Open 2024, 7, e2419818. [CrossRef]
- Bonneville, E.; Gautier-Veyret, E.; Ihl, C.; Hilleret, M.N.; Baudrant, M.; Fonrose, X.; Stanke-Labesque, F. Unexpected overdose blood concentration of tacrolimus: Keep in mind the role of inflammation. Br J Clin Pharmacol 2020, 86, 1888–1891. [CrossRef]
- Enokiya, T.; Nishikawa, K.; Hamada, Y.; Ikemura, K.; Sugimura, Y.; Okuda, M. Temporary decrease in tacrolimus clearance in cytochrome P450 3A5 non-expressors early after living donor kidney transplantation: Effect of interleukin 6-induced suppression of the cytochrome P450 3A gene. Basic Clin Pharmacol Toxicol 2021, 128, 525–533. [CrossRef]
- Barry, A.; Levine, M. A systematic review of the effect of CYP3A5 genotype on the apparent oral clearance of tacrolimus in renal transplant recipients. Ther Drug Monit 2010, 32, 708–714. [CrossRef]
- Wang, M.M.; Miao, D.; Cao, X.P.; Tan, L.; Tan, L. Innate immune activation in Alzheimer’s disease. Ann Transl Med 2018, 6, 177. [CrossRef]
- Minotti, C.; Tirelli, F.; Barbieri, E.; Giaquinto, C.; Dona, D. How is immunosuppressive status affecting children and adults in SARS-CoV-2 infection? A systematic review. J Infect 2020, 81, e61–e66. [CrossRef]
- Caillard, S.; French Registry of Solid Organ Transplant Patients, C. COVID-19 and kidney transplantation. Presse Med 2022, 51, 104146. [CrossRef]
- Fix, O.K.; Hameed, B.; Fontana, R.J.; Kwok, R.M.; McGuire, B.M.; Mulligan, D.C.; Pratt, D.S.; Russo, M.W.; Schilsky, M.L.; Verna, E.C.; et al. Clinical Best Practice Advice for Hepatology and Liver Transplant Providers During the COVID-19 Pandemic: AASLD Expert Panel Consensus Statement. Hepatology 2020, 72, 287–304. [CrossRef]
- He, S.; Zhou, C.; Lu, D.; Yang, H.; Xu, H.; Wu, G.; Pan, W.; Zhu, R.; Jia, H.; Tang, X.; et al. Relationship between chest CT manifestations and immune response in COVID-19 patients. Int J Infect Dis 2020, 98, 125–129. [CrossRef]
- Yitbarek, G.Y.; Walle Ayehu, G.; Asnakew, S.; Ayele, F.Y.; Bariso Gare, M.; Mulu, A.T.; Dagnaw, F.T.; Melesie, B.D. The role of C-reactive protein in predicting the severity of COVID-19 disease: A systematic review. SAGE Open Med 2021, 9, 20503121211050755. [CrossRef]
- Bastani, M.N.; Jalilian, S. Unraveling the enigma: The emerging significance of pulmonary surfactant proteins in predicting, diagnosing, and managing COVID-19. Immun Inflamm Dis 2024, 12, e1302. [CrossRef]
- Calkovska, A.; Kolomaznik, M.; Calkovsky, V. Alveolar type II cells and pulmonary surfactant in COVID-19 era. Physiol Res 2021, 70, S195–S208. [CrossRef]
- Almubarak, A.; Tanagala, K.K.K.; Papapanou, P.N.; Lalla, E.; Momen-Heravi, F. Disruption of Monocyte and Macrophage Homeostasis in Periodontitis. Front Immunol 2020, 11, 330. [CrossRef]
- Ozawa, T. Effects of FK506 on ca release channels (review). Perspect Medicin Chem 2008, 2, 51–55. [CrossRef]
- Hattori, T. Neutrophil-Galectin-9 Axis Linking Innate and Adaptive Immunity in ATL, Sezary Syndrome, COVID-19, and Psoriasis: An AI-Assisted Integrative Review. Reports (MDPI) 2025, 9. [CrossRef]
- Sun, J.; Sui, Y.; Wang, Y.; Song, L.; Li, D.; Li, G.; Liu, J.; Shu, Q. Galectin-9 expression correlates with therapeutic effect in rheumatoid arthritis. Sci Rep 2021, 11, 5562. [CrossRef]
- Iwasaki-Hozumi, H.; Chagan-Yasutan, H.; Ashino, Y.; Hattori, T. Blood Levels of Galectin-9, an Immuno-Regulating Molecule, Reflect the Severity for the Acute and Chronic Infectious Diseases. Biomolecules 2021, 11. [CrossRef]
- Gross, N.J.; Narine, K.R. Experimental radiation pneumonitis. Corticosteroids increase the replicative activity of alveolar type 2 cells. Radiat Res 1988, 115, 543–549.
- Castelnovo, L.; Tamburello, A.; Lurati, A.; Zaccara, E.; Marrazza, M.G.; Olivetti, M.; Mumoli, N.; Mastroiacovo, D.; Colombo, D.; Ricchiuti, E.; et al. Anti-IL6 treatment of serious COVID-19 disease: A monocentric retrospective experience. Medicine (Baltimore) 2021, 100, e23582. [CrossRef]
- Watts, N.; Amann, M.; Arnell, N.; Ayeb-Karlsson, S.; Beagley, J.; Belesova, K.; Boykoff, M.; Byass, P.; Cai, W.; Campbell-Lendrum, D.; et al. The 2020 report of The Lancet Countdown on health and climate change: responding to converging crises. Lancet 2021, 397, 129–170. [CrossRef]
| CompleteBloodCellcount anddifferential | Reference Range | Result | Urine Routin Test | Reference Range | Result |
|---|---|---|---|---|---|
| Whitecellunit(/µL) | 3700–8500 | 8900 | Color | Yellow | Yellow |
| Neutrophils(%) | 44.0–68.0 | 87.5 | Clatity | Clear | Clear |
| Lymphocytes(%) | 27.0–44.0 | 5 | Specific gravity | 1.009-1.025 | 1.020 |
| Monocytes(%) | 3.0–12.0 | 4.5 | pH | 4.8-7.5 | 6.5 |
| Eosinophils(%) | 0.0–10.0 | 0.5 | Protein | - | 1+ |
| Basophils(%) | 0.0–3.0 | 0.0 | Sugar | - | 4+ |
| Hematocrit(%) | 42.0–53 | 30.1 | White cells | - | - |
| Hemoglobin(g/dL) | 13.5–17.5 | 9.8 | Red cell | - | - |
| Plateletcount×103(/µL) | 150–355 | 193 | |||
| Redcellcount×106(/µL) | 3.90–5.30 | 3.50 | Immunoserological Test | Reference Range | Result |
| Endotoxin | 0.0-5.0 | <3.5 | |||
| Biochemical Test | Reference Range | Result | (1→3)-β-D-glucan | 0.0-11.0 | <6.5 |
| Urea nitrogen(mg/dl) | 8-20 | 24 | Procalcitonin | 0.00-0.05 | 0.43 |
| Creatinine(mg/dl) | 0.65-1.07 | 1.35 | |||
| Alanine aminotransferase (U/L) | 3-40 | 19 | Coagulationtest | Reference Range | |
| Aspartate aminotransferase (U/L) | 8-35 | 23 | prothrombin time(s) | 10.0–13.5 | 13.1 |
| Lactate dehydrogenase (U/L) | 124-222 | 258 | Prothrombin activity (%) | 80.0-120.0 | 107.3 |
| Ferrtin (ng/mL) | 14-304 | 550 | PT-International Normalized Ratio | 0.90-1.10 | 0.97 |
| C-reactive protein (mg/dl) | 0.00-0.30 | 19.85 | Activated Partial Thromboplastin Time (s) | 24.0–39.0 | 46.5 |
| Total protein (g/dl) | 6.6-8.4 | 5.2 | D-dimer(µg/mL) | 0.00–1.00 | 3.27 |
| Albumin (g/dl) | 3.8-5.2 | 2.7 | Fibrinogen(mg/dL) | 200–400 | 894 |
| Result | |||
|---|---|---|---|
| Respiratory pathogen panel test PCR (1) Nasal swab |
SARS-Cov-2-PCR | + | |
| InfluenzaA-PCR | - | ||
| InfluenzaB-PCR | - | ||
| Adenovirus-PCR | - | ||
| RS Virus -PCR | - | ||
| Human Metapeuumo-PCR | - | ||
| Myco pneumoia-PCR | - | ||
| Culture Test | sputum | - | |
| blood | - | ||
| urine | - | ||
| Antigen test | blood | cytomegaloviruspp65 antigenemia (2) |
- |
| Nasal swab | InfluenzaA (3) | - | |
| InfluenzaB (3) | - | ||
| SARS-Cov2 (3) | + | ||
| urine | Pneumococcal antigen (4) | - | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




