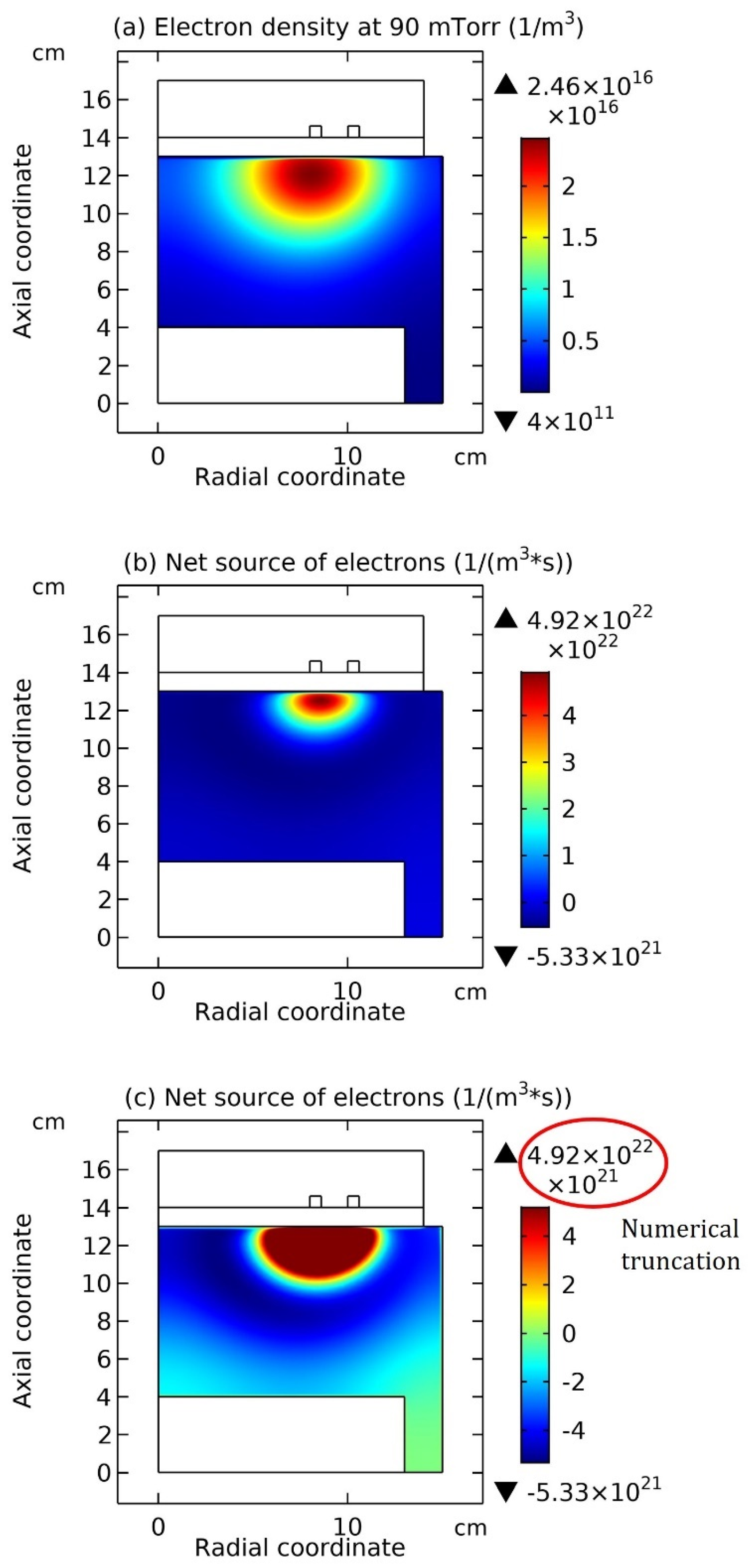
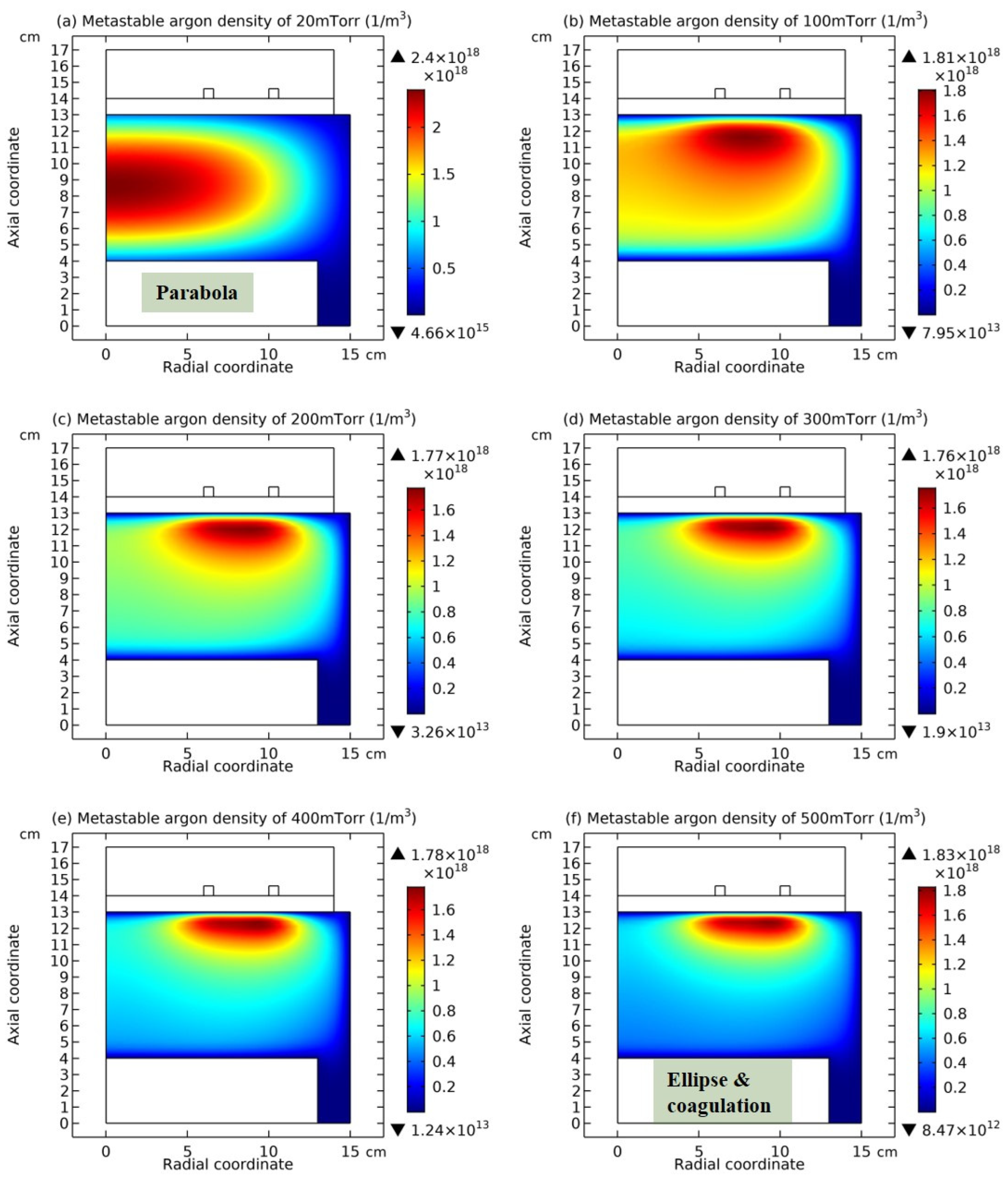
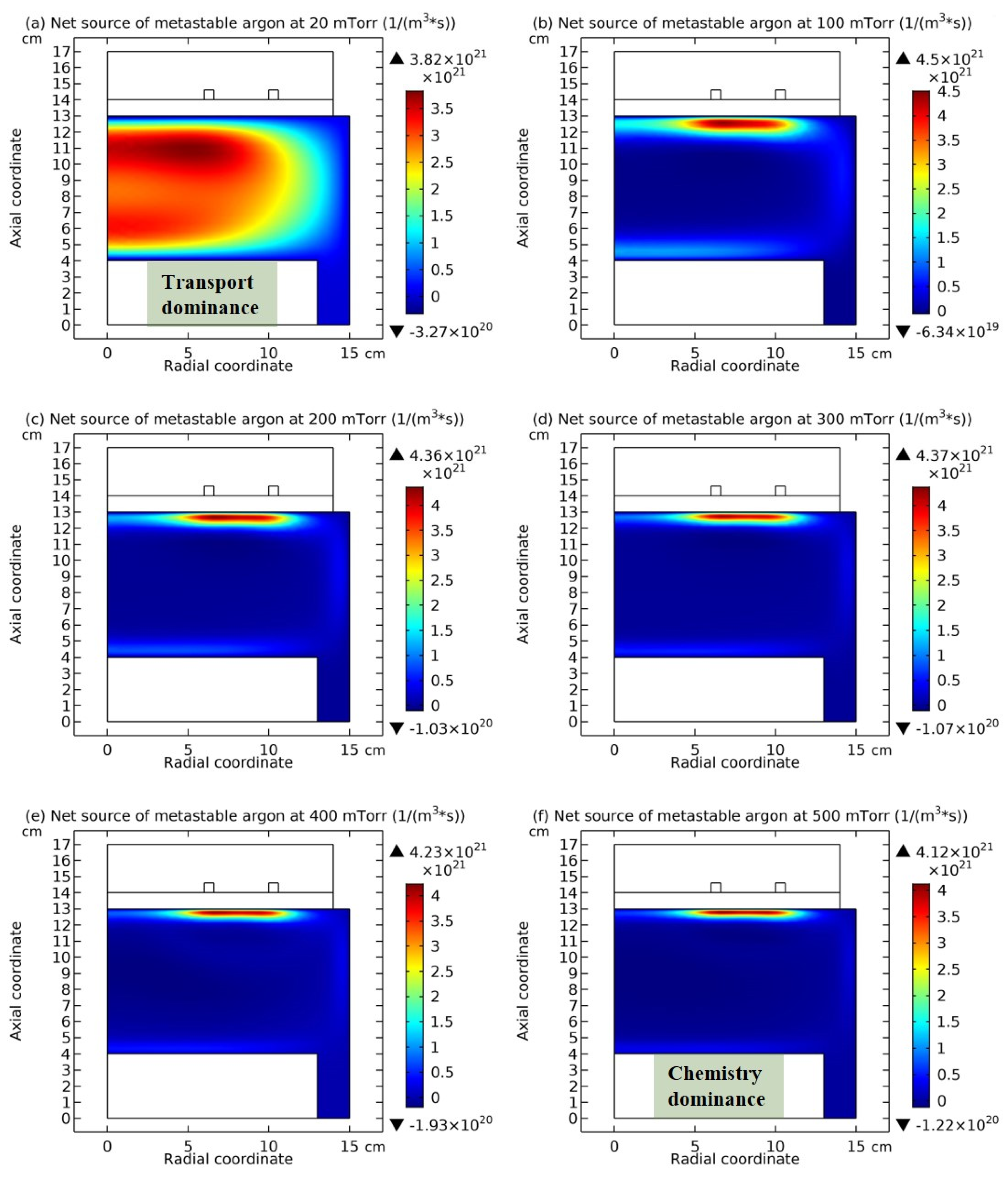
3.2. Chemically Ambi-Polar Self-Coagulations of Ions in Highly Electronegative Plasma at High Pressure and Its Implication on the Quantum and Particle Physics
(a) Mixed structure of self-coagulation and ellipse background.
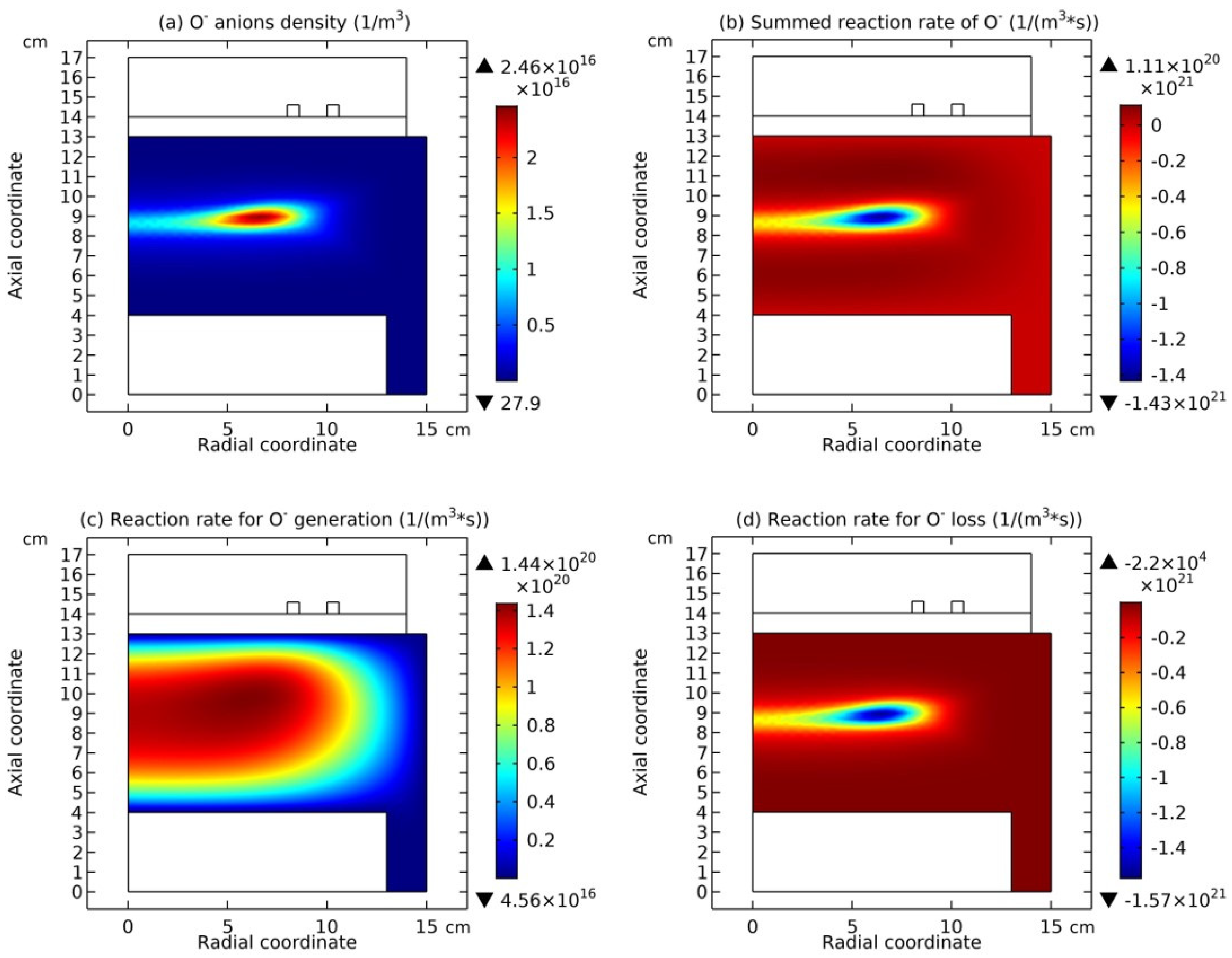
In
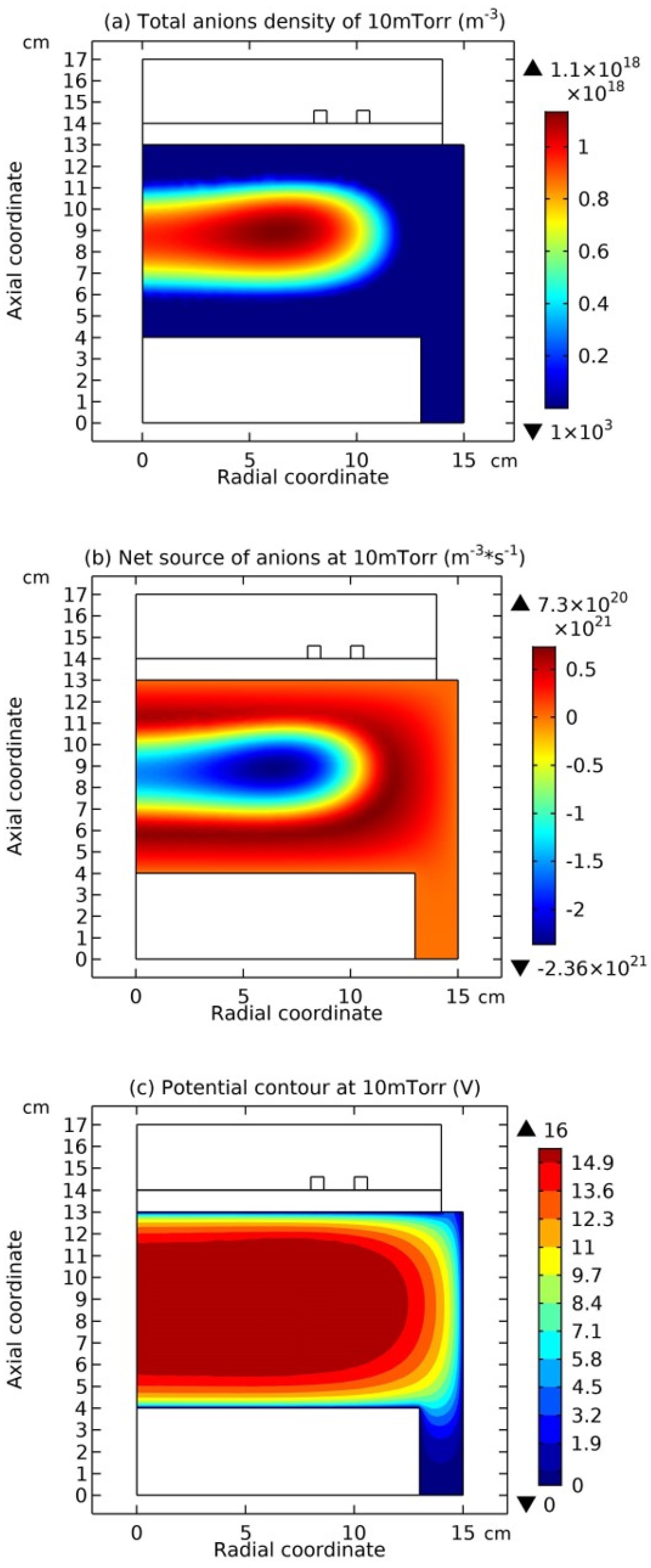
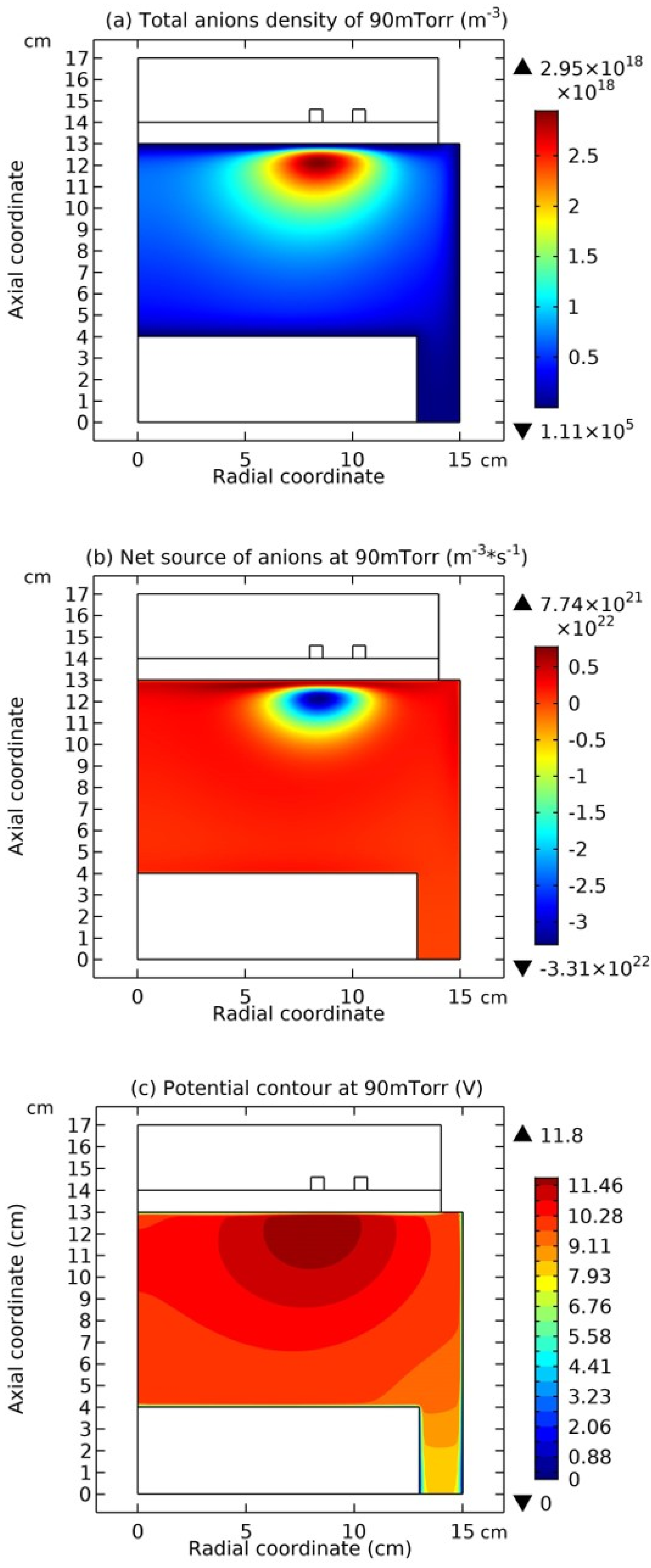
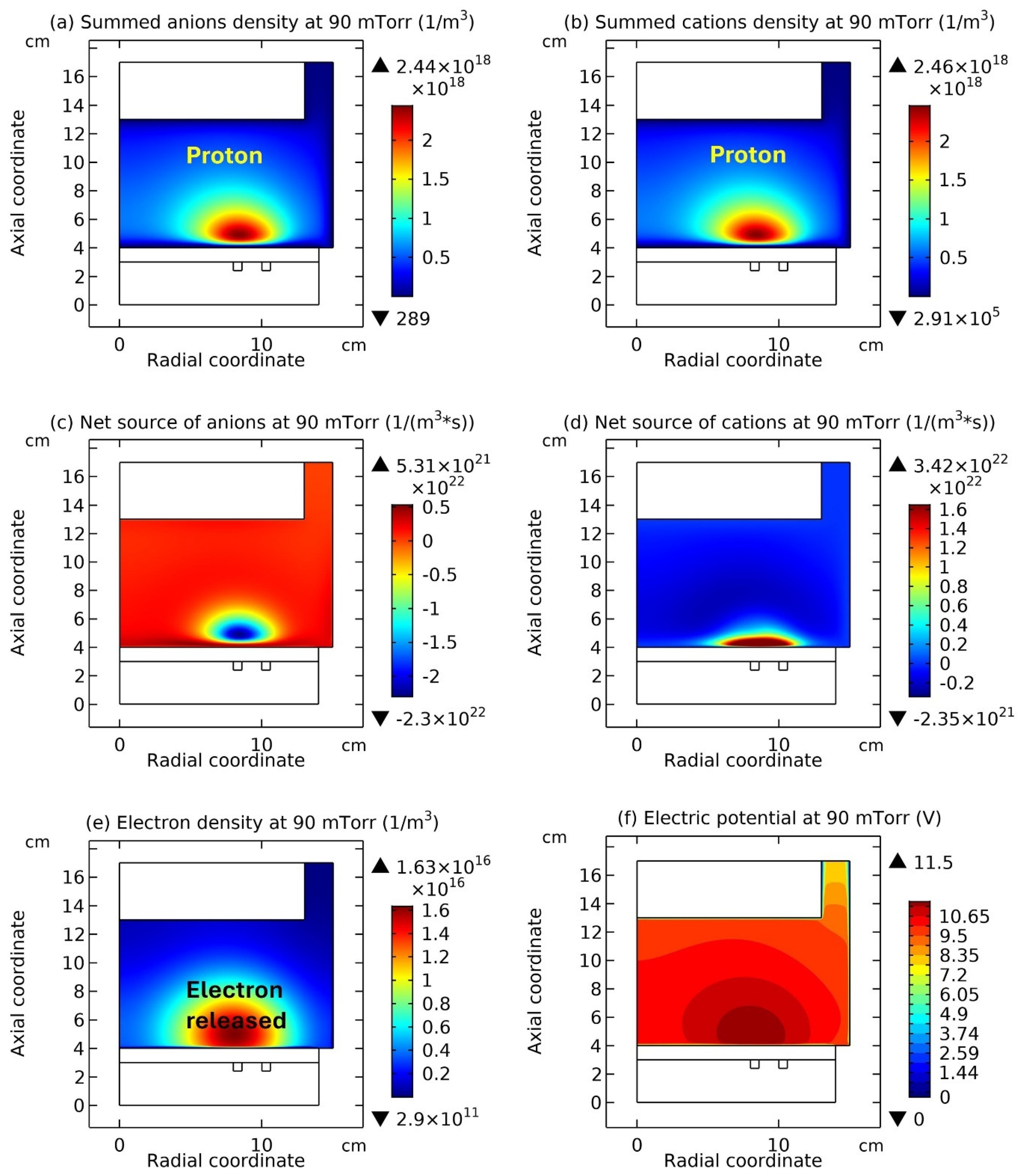
Figure 7, the summed anions density, the net source of anions, and the electric potential of Ar/SF
6 ICP at the high pressure of 90 mTorr are presented. It is seen from
Figure 5(a) and
Figure 7(a) as increasing the pressure, the discharge structure of Ar/SF
6 ICP changes. The stratification disappears and a mixed structure of the localized self-coagulation of anions under the coil and the peripherical anions ellipse background, i.e., flattened profile, is formed. Moreover, at the position of coagulated structure, the chemical source of anions in
Figure 7(b) is negative, and in
Figure 7(c), the profile of potential at the coagulated position is also flat, which implies the free diffusion of anions. So, these two conditions are satisfied again, and the self-coagulation described in
Section 2.3c works and the anions coagulates under the coil. Besides, it is noticed the appearance of ellipse profile implies that the discharge shifts from the regime of transport to the regime of chemistry.
(b) Mass balance of cations at the chemically ambi-polar coagulation scheme.
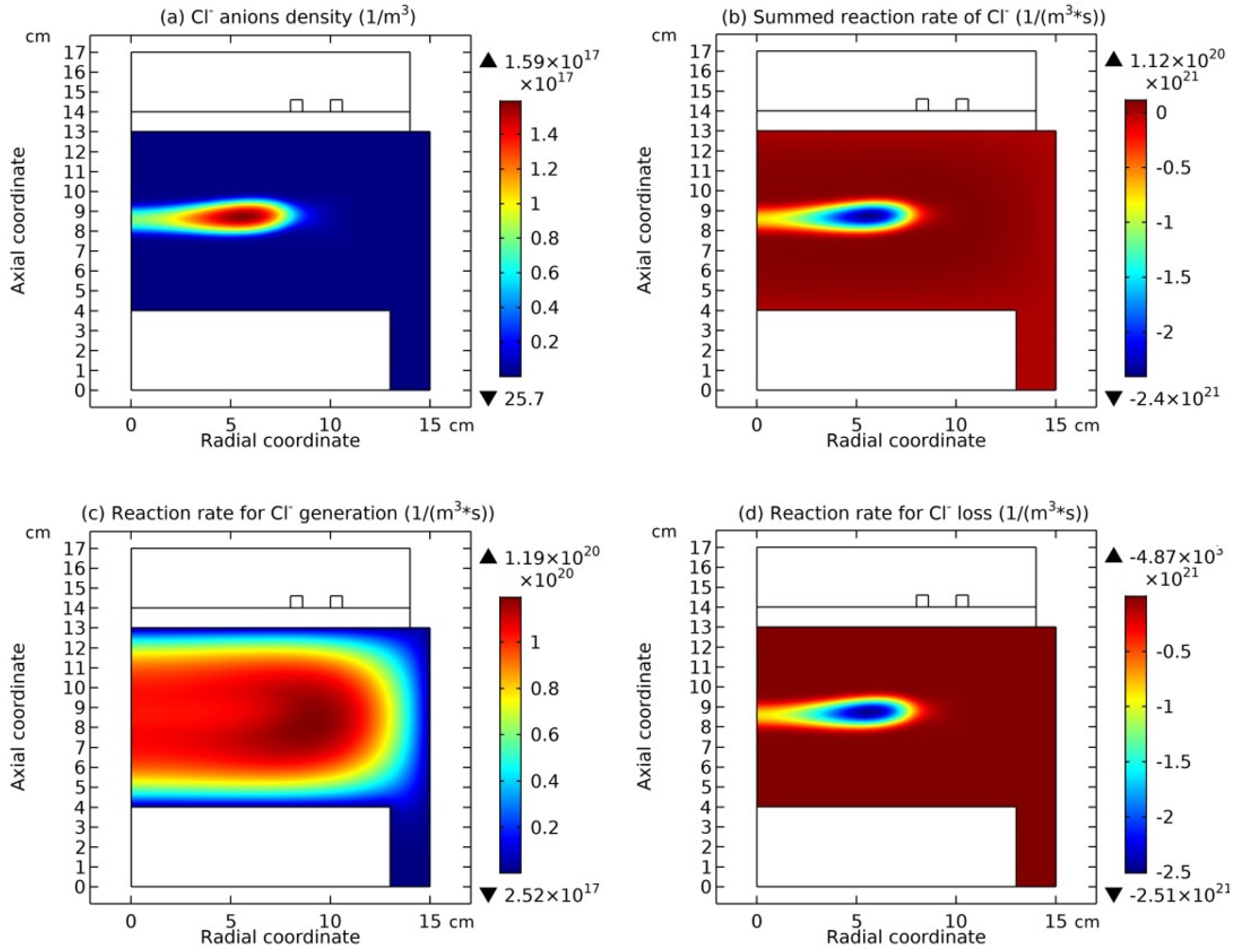
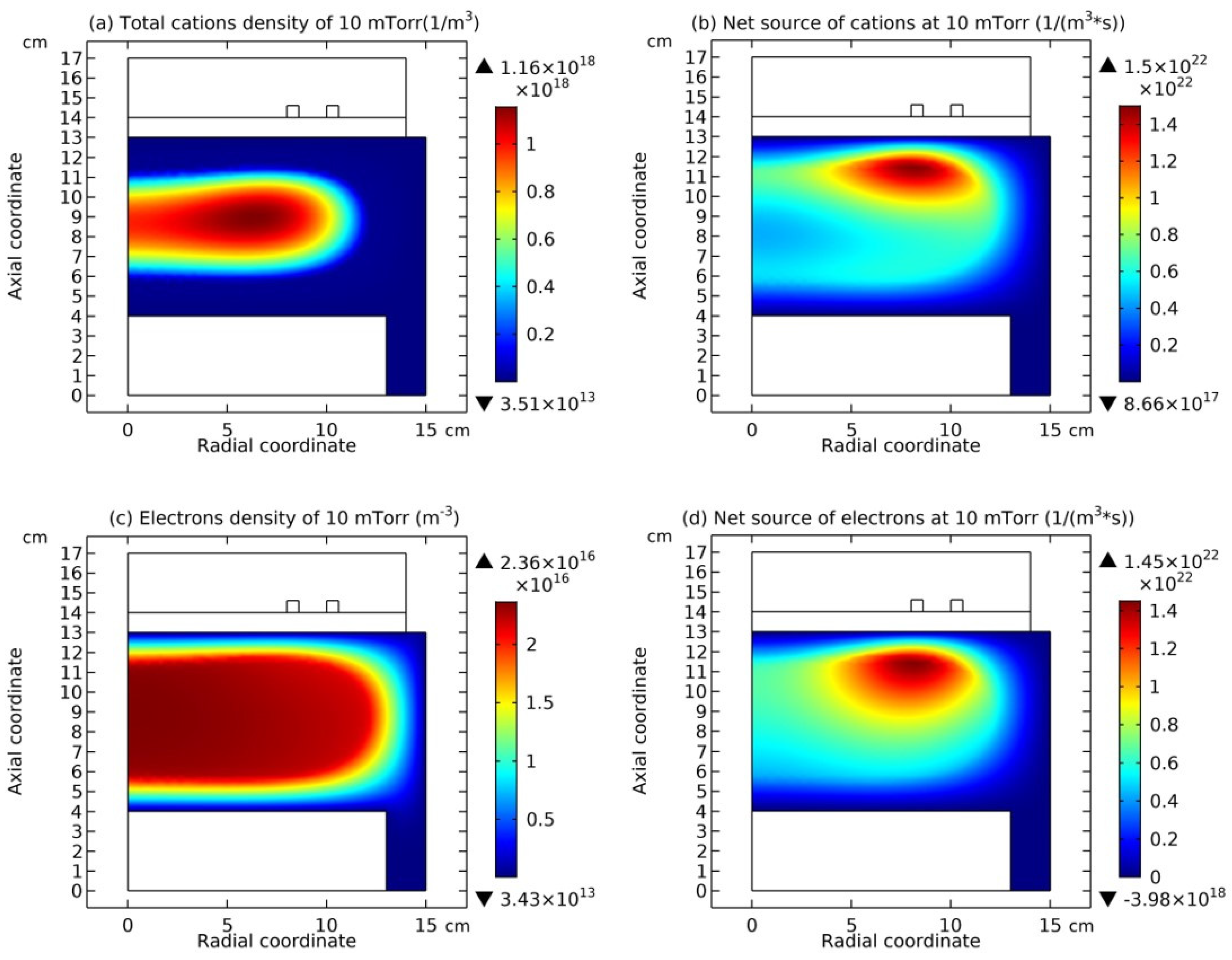
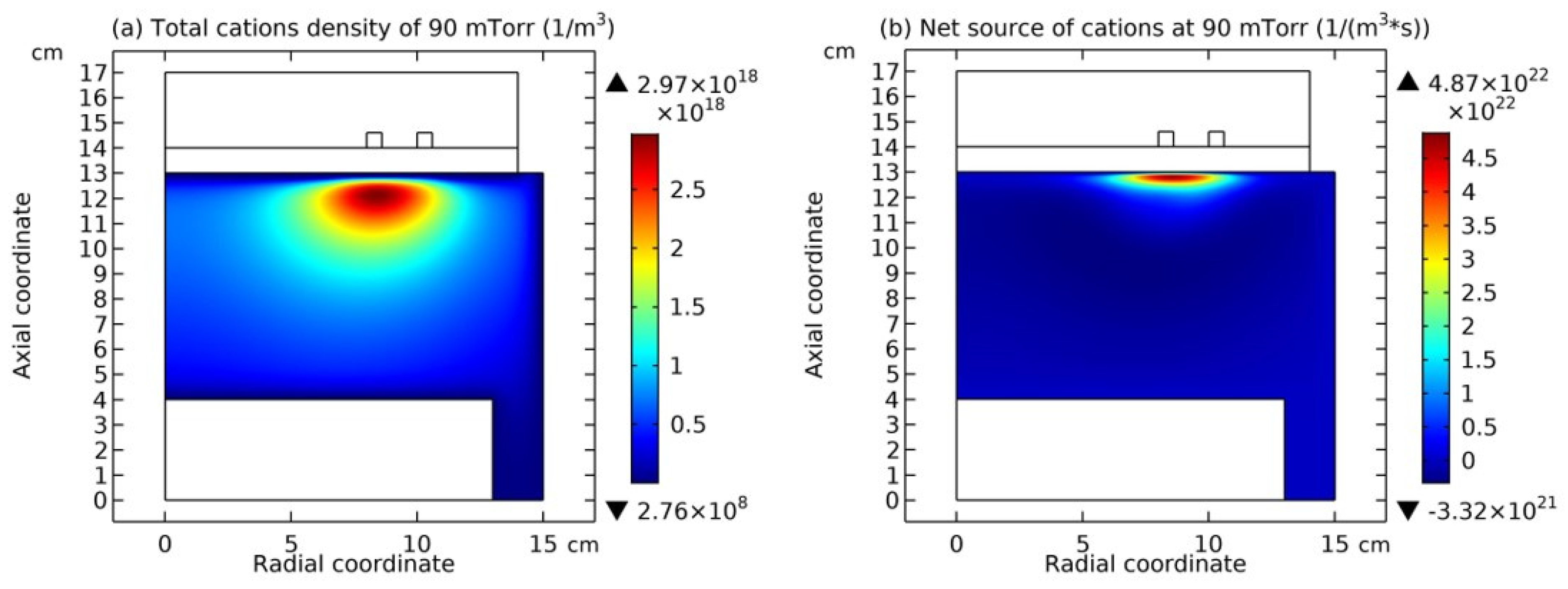
In
Figure 8, the summed cations density and the net source of them in the Ar/SF
6 ICP at the high pressure of 90 mTorr are presented. It is seen that the cations density in
Figure 8(a) holds the same profile as anions in
Figure 7(a), and meanwhile, in
Figure 8(b), the net source of cations in the regime of chemistry exhibits different characteristics, i.e., with a red and positive delta type of chemical source under the coil and the peripherical negative wide-spread type of chemical source. As stated below, at such a characterized profile of chemical source, the cations exhibit the self-coagulation as well, separately.
In Equation (56), the general continuity equation of cation density is given. Herein,
are
the summed cations density and flux, respectively.
and
are the generation and loss sources of them, respectively.
In Equation (57), the specific continuity equation of cation density in the chemical scheme is given, where the mass transport is controlled exclusively by the chemical kinetics and the mass flux is thereby zero. Here,
and
represent the ionization rate and recombination rate, respectively. They are both assumed constant for simplicity. The introduced functions,
and
, are explained in Equations (58) and (59), respectively.
As illustrated in Equations (58) and (59),
is strictly a Dirac function and
is a quasi- Step function.
In Equation (60), the steady state case of cation continuity equation in the chemical scheme is given. Here,
is the background cation density and
is the recombination frequency of cations with anions.
In Equation (61), the balance between the generation and loss rates of cations is given. The two sides of Equation (61) are integrated and then in Equation (63) the background cation density,
, is calculated. As seen, it depends on both the localized ionization rate and the widespread recombination frequency. Here,
is the volume of chamber and
is the occupied volume of localized ionization rate.
Finally, in Equation (63), the localized cation density peak,
, is predicted. As seen, it tends to infinite naturally and its magnitude is proportional to the ionization rate logically. To summarize the self-coagulation of anions in
Figure 7 and
Section 2.3c and the self-coagulation of cation in
Figure 8 and Equations (56)–(63), it is believed a chemically ambi-polar self-coagulation scheme can be defined at the high pressure of 90 mTorr, where both chemistries of anions and cations dominate over the transport. In
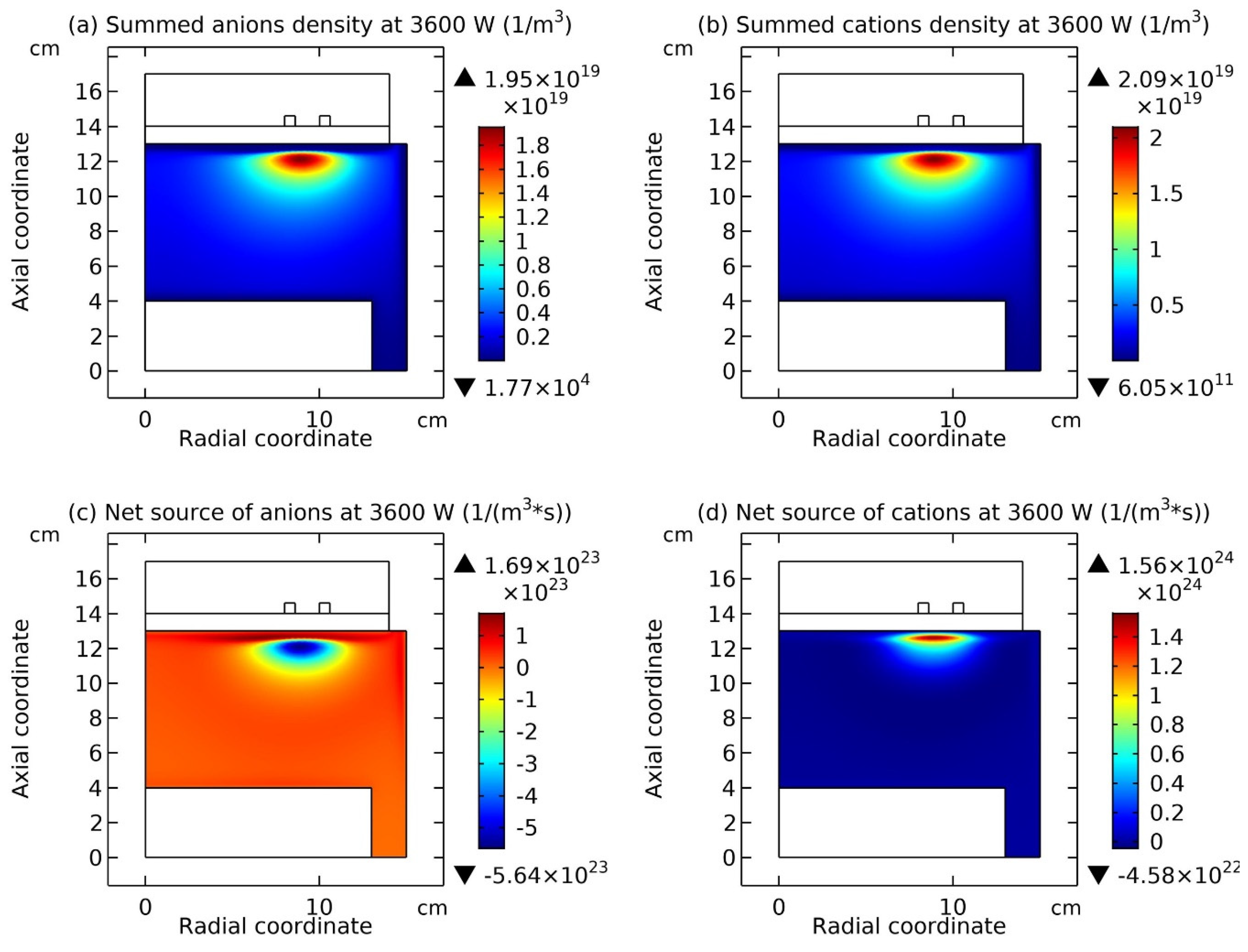
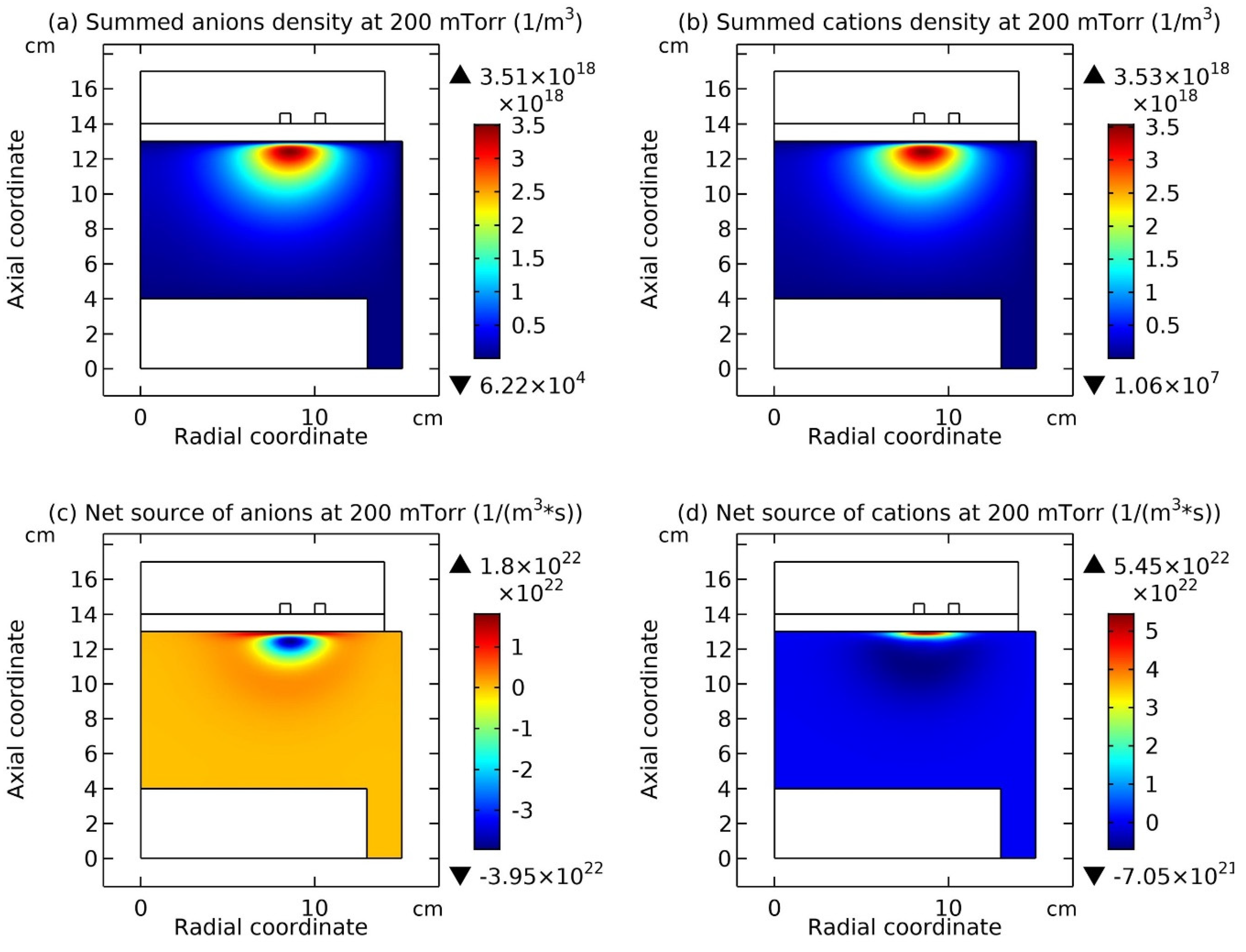
Figure 9 and
Figure 10, it is seen that this type of chemically ambi-polar self-coagulation scheme becomes stronger at the high power, 3600W, and the high pressure, 200mTorr, respectively.
(c) Implication of coagulation and ellipse mixed profile on wave-particle duality.
In Ref. [
12], it is predicted that the mixed structure of ellipse and self-coagulation represents the wave-particle duality, based on the wave dynamic equation and quasi-particle model equation, when considering the role of inertia term. In the next section, it is further predicted the Schrodinger equation can be used to find out the eigen state of coagulated anions in an assumed semi-circle and infinitely high potential barrier.
(d) Schrodinger equation of anions in the semi-circle infinite high potential barrier
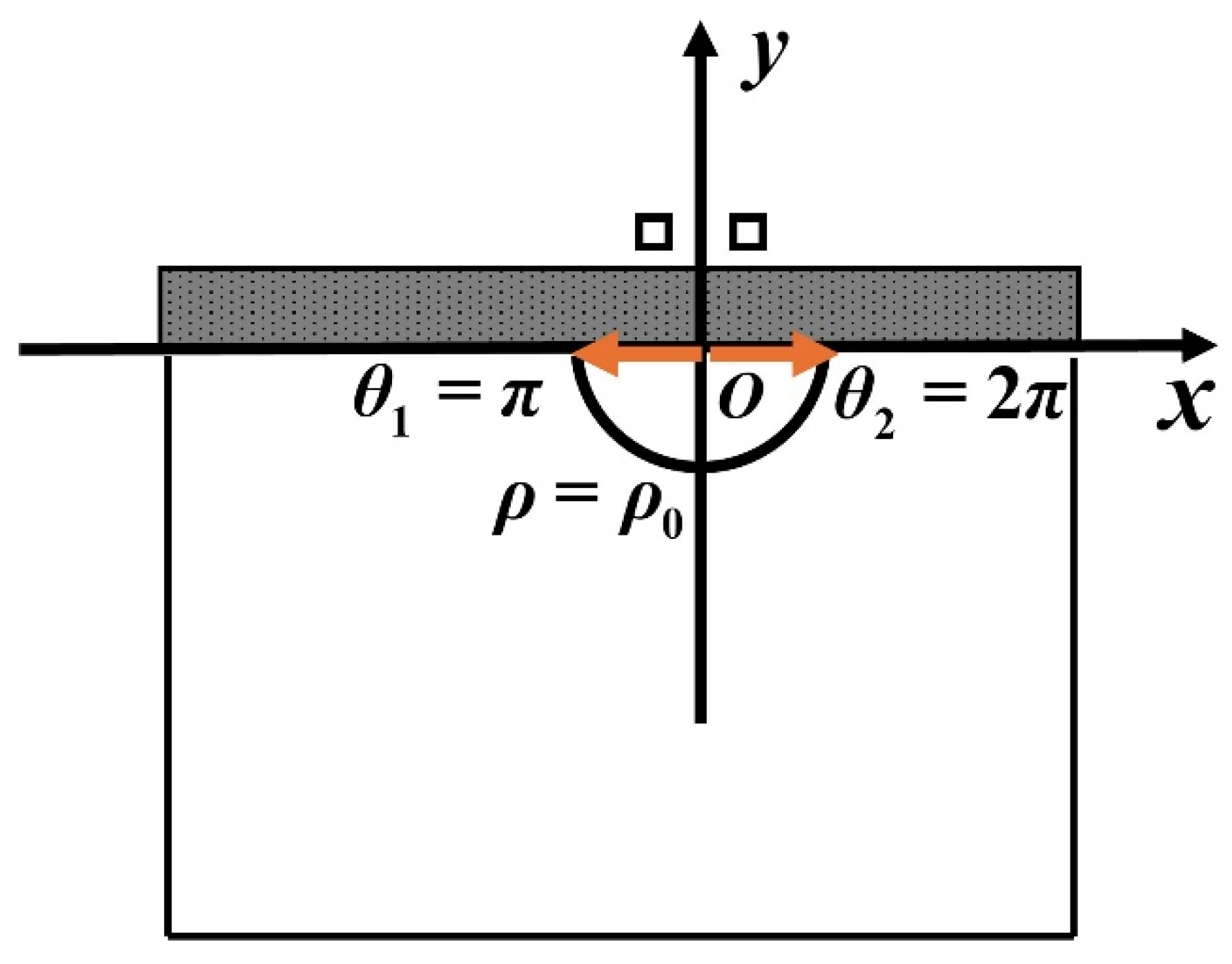
Figure 11.
Polar coordinate system for the semi-circle and infinitely high potential barrier model.
Figure 11.
Polar coordinate system for the semi-circle and infinitely high potential barrier model.
In this section, the Schrodinger equation of anions in the semi-circle and infinitely high potential barrier is described and the probabilities of finding the anion particle is given.
In Equations (64) and (65), the potential model is given.
In Equation (66), the Schrodinger equation is given. Herein,
is the energy of particle in the potential barrier and
is the probability function.
When
,
When
,
With the method of variable separation, we have
Substituting
in Equation (69) with the above expression of Equation (70), we have
Reform Equation (71), we have
Introducing the parameter,
, into Equation (72), we have
Assume,
Then, we have
Assuming
, we have
As seen, Equation (77) represents the Bessel’s equation. The solution of Equation (73) is
Utilizing the periodical boundary condition,
, we have
Hence, the eigen values of introduced parameter are
. So, the Equation (77) is the
integer order Bessel equation. Further utilizing the homogeneous boundary condition of
at
, we have the another set of eigen values illustrated as
Here,
represents all the zero points of
order Bessel function. By correlating Equation (75), we finally have the eigen energy as follows,
It is seen that the energy level of particle in such a barrier depends on the value of
. When the order number of Bessel function, i.e., the value of
, is fixed, the energy interval of two energy levels is determined by the deduct of squares of two adjacent zero points of
order Bessel function, illustrated below,
Since the Planck’s constant,
, is quite small and the square deduct of zero points of Bessel’s function is limited value, the energy interval will tend to zero if the size of semi-circle and infinitely high potential barrier is large enough, like in an order of centimeter as designed in the fluid simulation of Ar/SF
6 ICP. As the energy level tends to zero in the size of centimeter, the quantum scheme transitions to the continuity scheme, and the probability of finding the particle in the potential barrier is one.
(e) Implication on particle physics,
decay.
In this sub-section, the implication of self-coagulation and related hierarchy on the decay in nucleus is elaborated. In Ref. [
12], the two main structures of hierarchy at the high pressure of 90 mTorr, coagulated body and ellipse background, are correlated to the neutron and proton models, respectively, since the blue sheath that connects the two structures is expressed with the Yukawa potential[
18], which represents a meson[
19]. Herein, it is believed that the
and
decays occurring in the nucleus[
20] as listed in
Table 1, can be interpreted by a change of coil position from the top to bottom, verse vice, as illustrated by
Figure 12,
Figure 13,
Figure 14 and
Figure 15. In
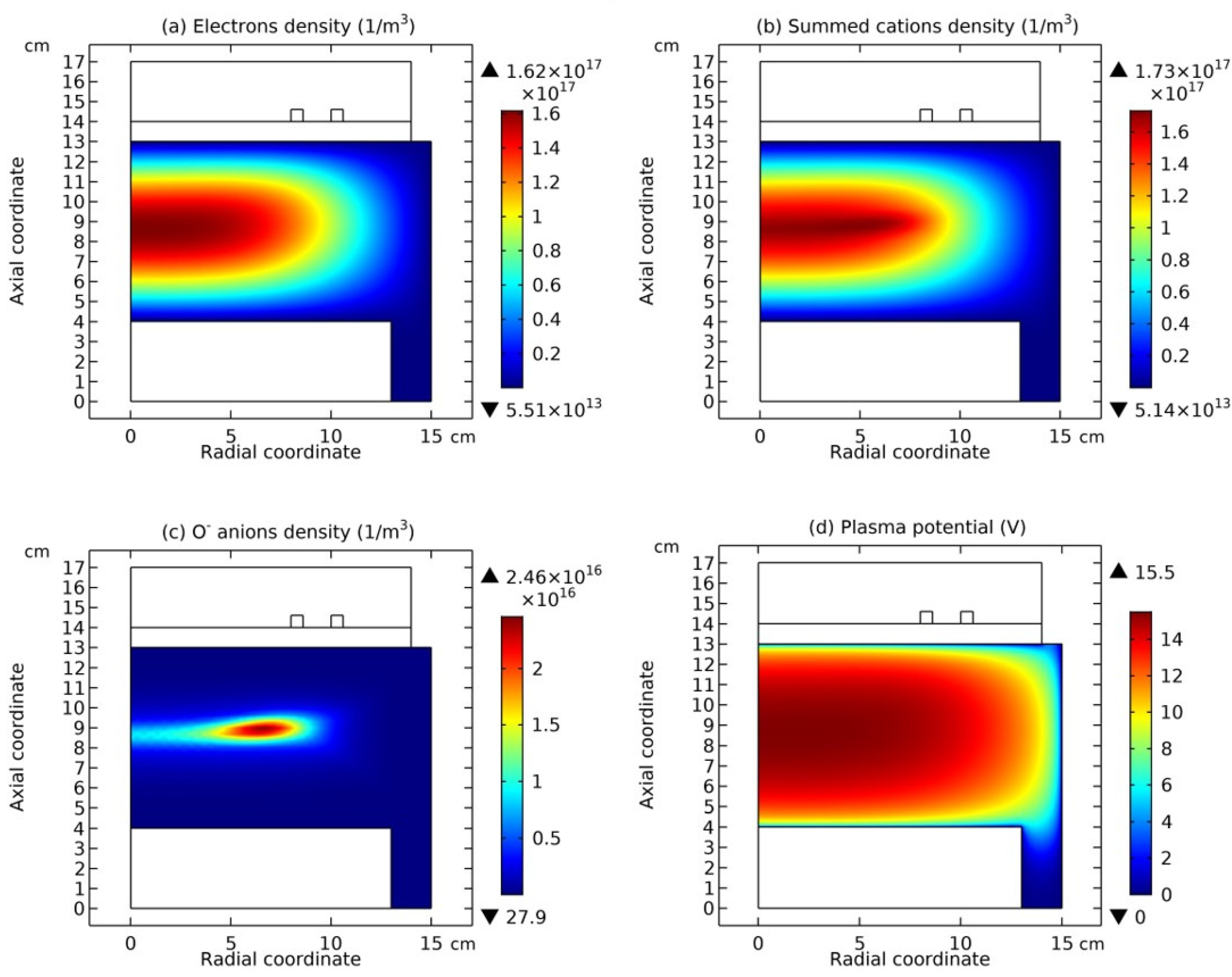
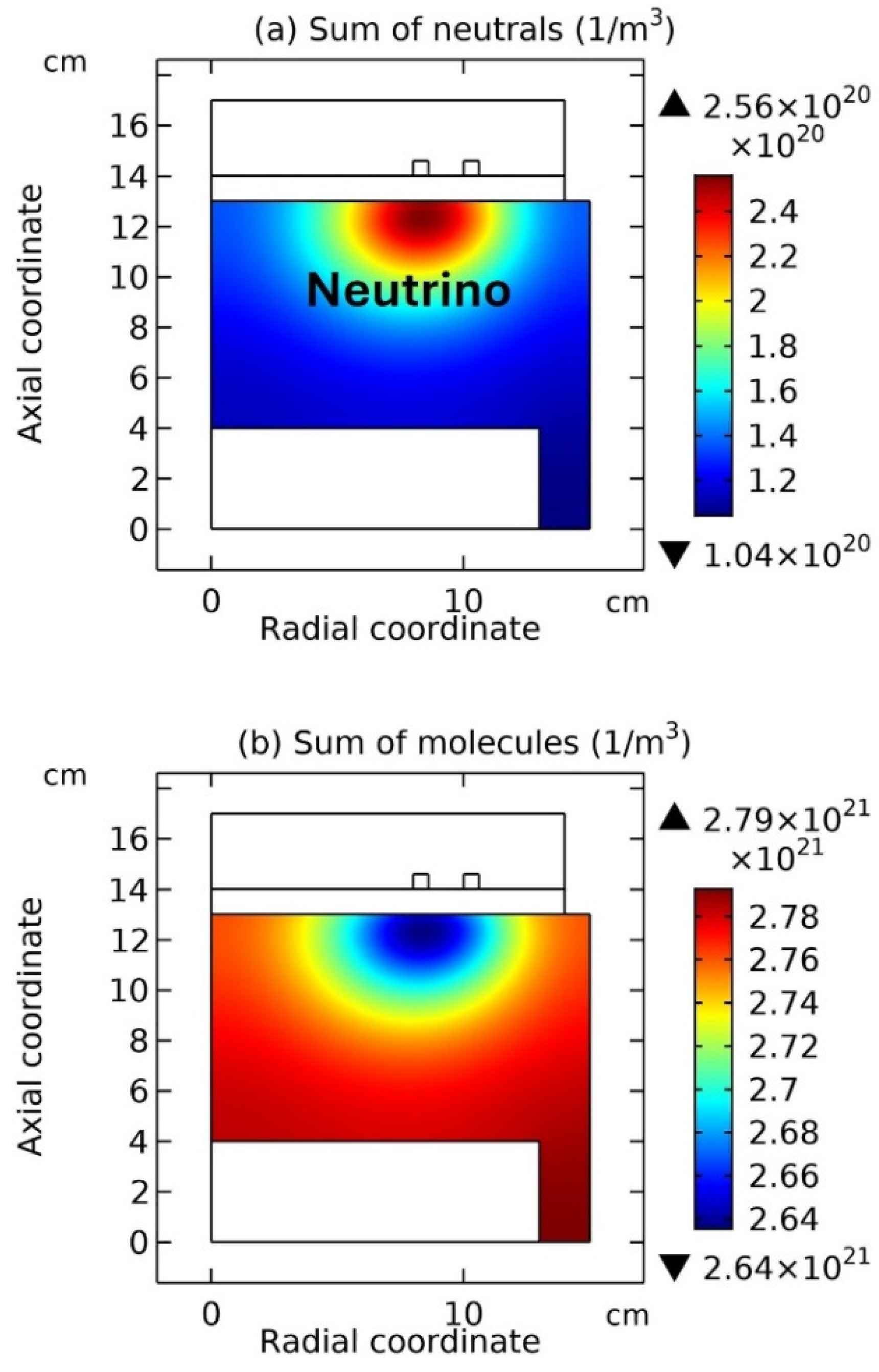
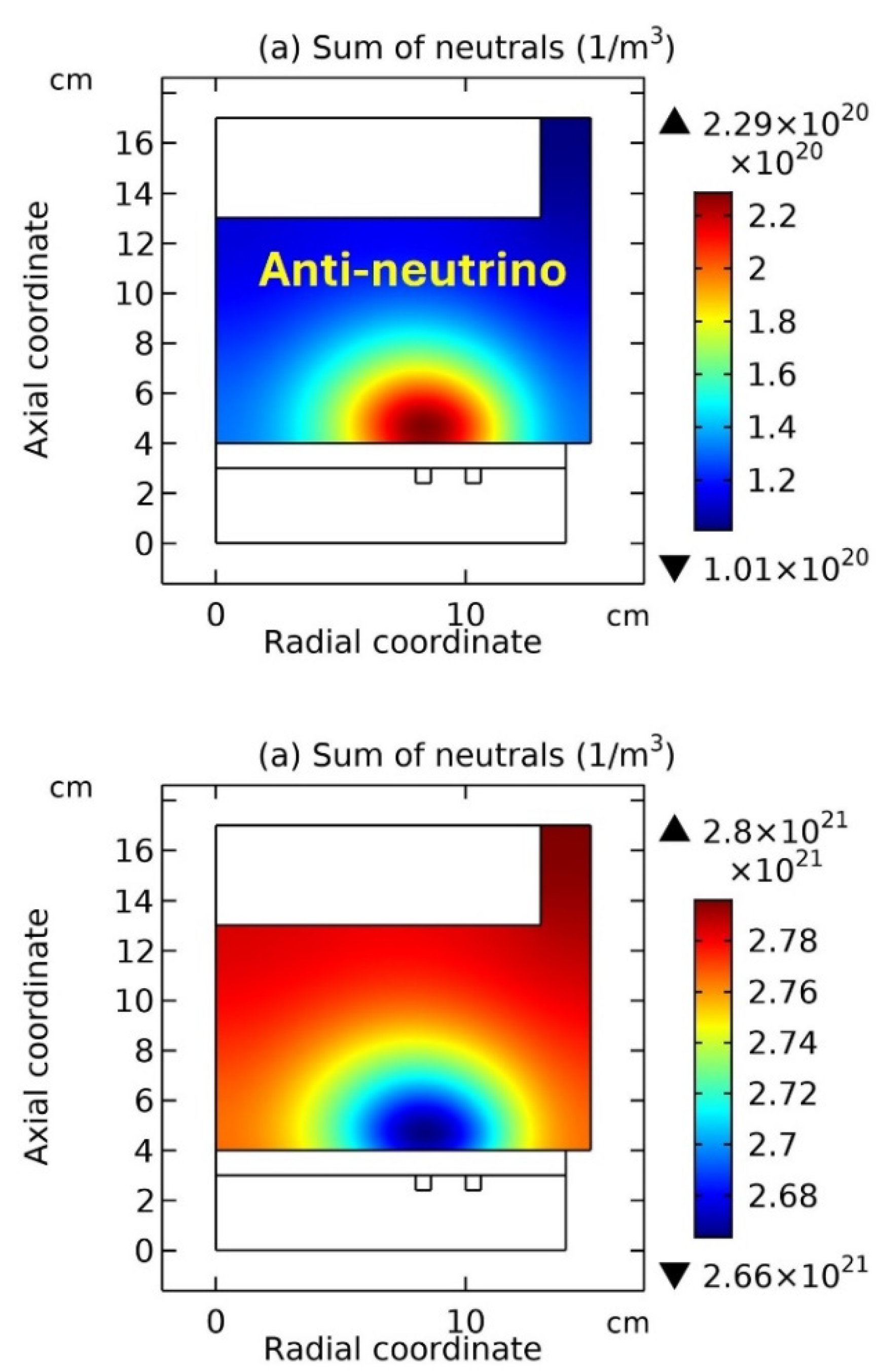
Figure 12 and
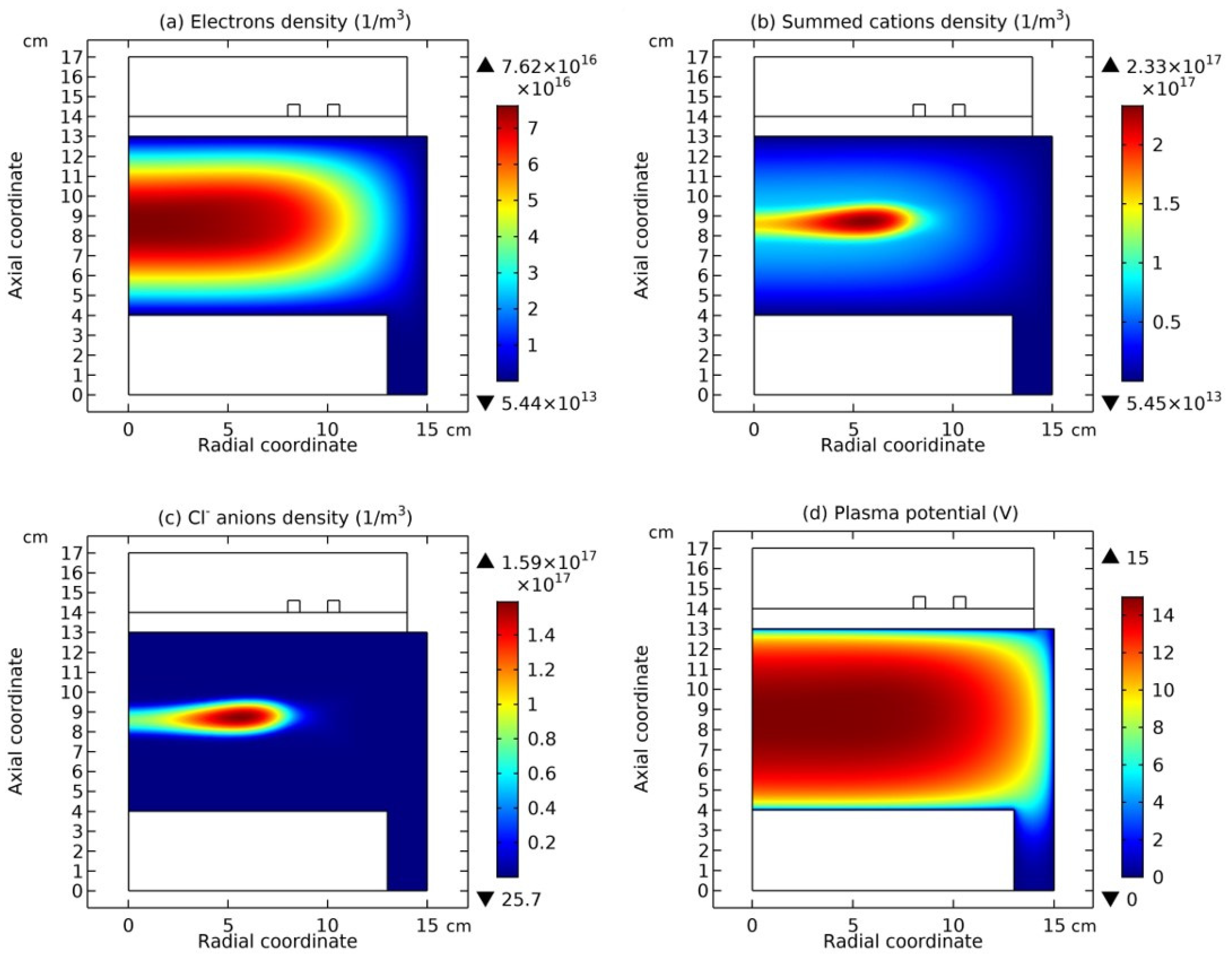
Figure 14, the coil is on the top, while in
Figure 13 and
Figure 15, the coil is on the bottom, i.e., a reversed chamber that is clockwise rotated
. In
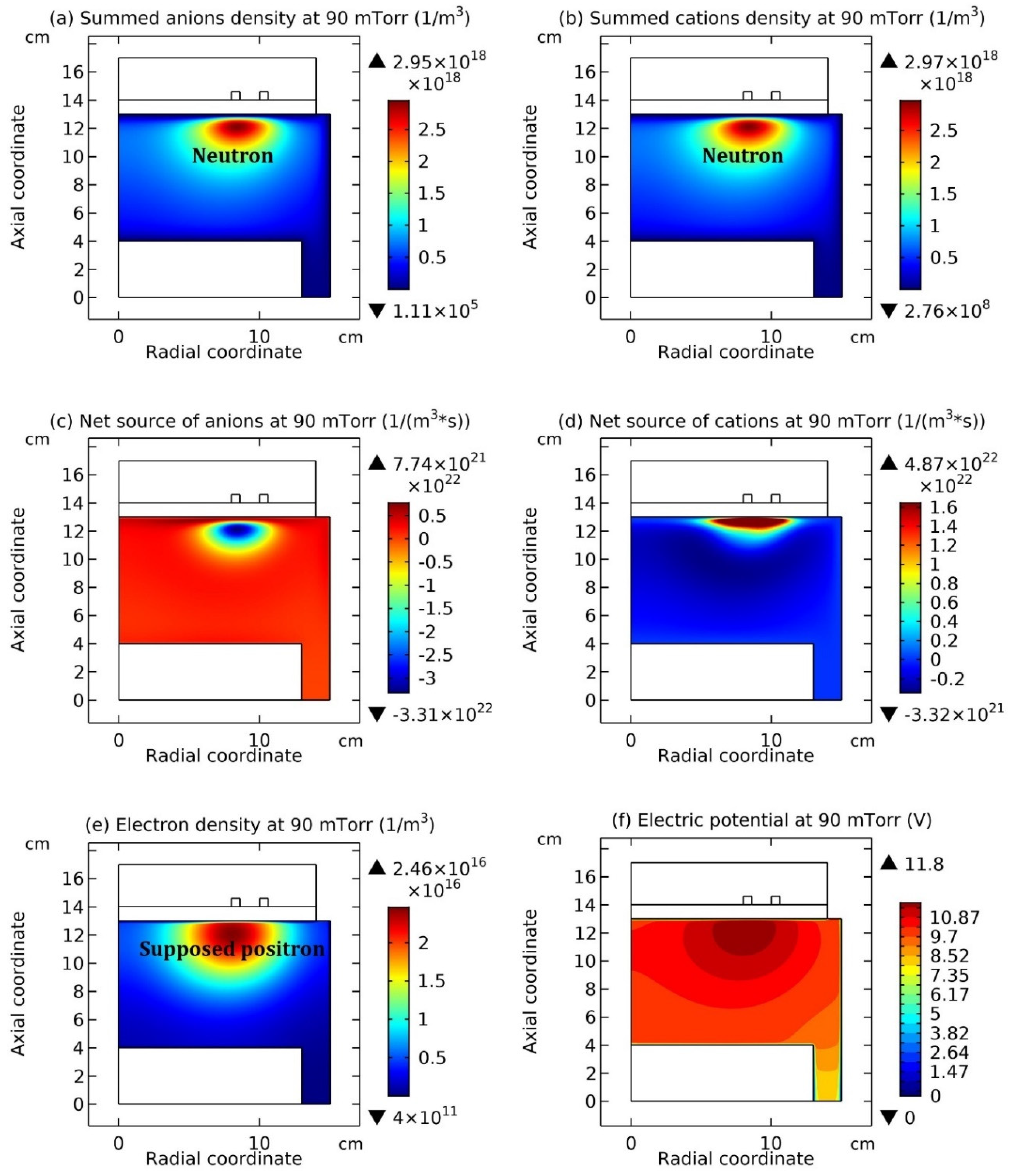
Figure 12 and
Figure 13, the summed anions density, summed cations density, net source of anions, net source of cations, electron density, and electric potential, at the top and bottom coil positions are plotted, respectively. And in
Figure 14 and
Figure 15, the summed neutrals and molecules, at the two coil positions, are plotted, respectively. It’s analyzed from the
Figure 12(a, b), and
Figure 13(a, b), as the coil is changed from the top to the bottom, the neutron under the coil turns into the proton. Meanwhile, the electron released in this
decay is represented by the coagulated electrons in
Figure 13 (e), and the anti-neutrino released is represented by the coagulated neutrals in
Figure 15 (a).
Reversely, as the coil is changed from the bottom to the top, i.e., seen from
Figure 13 to
Figure 12, the proton in
Figure 13 (a, b) turns into the neutron under the coil in
Figure 12(a, b). Accordingly, the supposed positron is released, which is represented by the coagulated electrons, as shown in
Figure 12 (e), and the neutrino released is represented by the coagulated neutrals under the coil in
Figure 14 (a). It’s noted the positron is presently not included in the simulation, and we have tried to include the positron and photon as species in the present simulation, but all failed lastly due to the lack of self-consistency. However, in the next two
Section 3.3 and
Section 3.4, it is predicted that the mono-polar mass, including the electron and positron, can be self-coagulated, and even the non-polar mass, e.g., metastable argon, can be self-coagulated too, which somehow represent the positron (was is existed) and the neutrino. In the real
and
decays in the nucleus, all the needed components, i.e., neutron, proton, electron, positron, neutrino, and anti-neutrino are existed. In the present simulation environment, only five components are involved, and the positron is excluded due to its strict condition for production. Nevertheless, it is believed was it included in the present framework, can it be self-coagulated, since self-coagulation does no requires a specialty on the polarity, but a special requirement on chemical reaction source term; see
Section 3.3 and
Section 3.4 for reference.
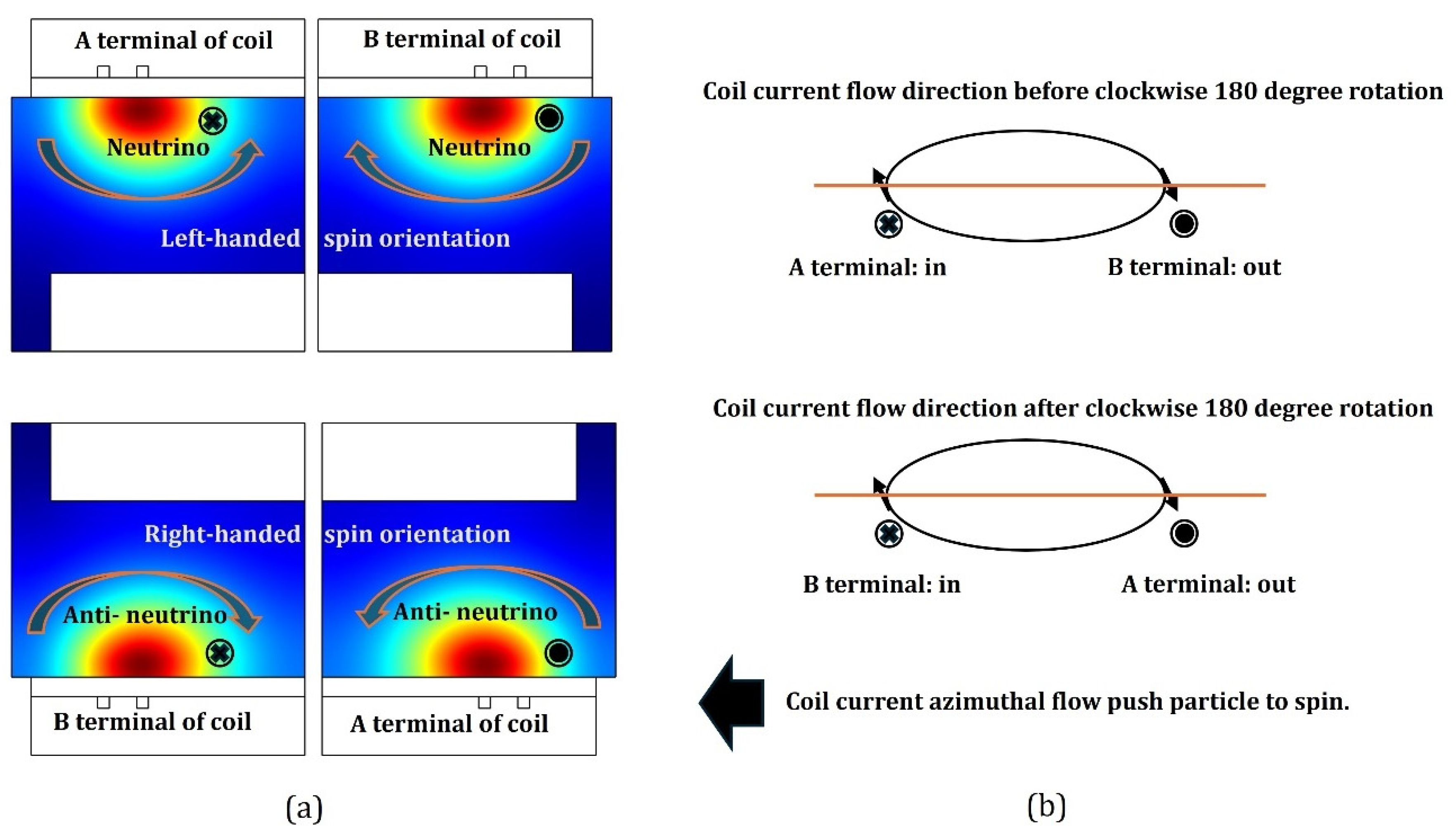
Besides, the neutrino holds the left-hand spin orientation, and the anti-neutrino holds the right-hand spin orientation[
21]. Assume the neutrino model in
Figure 14(a) flights outside, then its spin is left-handed since the left side is faced with the space but the right side is against the wall. Moreover, assume the anti-neutrino model in
Figure 15(a) flights outside as well, then its spin is right-handed, since the right side is against the wall but the left side is faced with the space. The assumed flighting directions of neutrino in
Figure 14(a) and of anti-neutrino in
Figure 15(a) are both out of page but the spin orientations of them are contradictory, because of the asymmetry caused by the
clockwise rotation of chamber. In
Figure 16, more details of neutrino and anti-neutrino spin characteristics and the origin of spin, coil current and its azimuthal rotation are given. The whole story of
and
decays depicted herein originates from the asymmetry, i.e., the coil that represents the particle coagulation position changes from the top to the bottom.
The view of three-dimension chamber configuration and the cylindrical symmetry of simulation assumed are needed to understand the asymmetry of chamber clockwise rotation in
Figure 16. In the upper panel of
Figure 16(a), the corresponding left profile of simulated intersection of chamber in the radial and axial dimensions that is always presented on the right side, is duplicated based on the cylindrical symmetry, when the coil is at the top. Accordingly, in the down panel of
Figure 16(a), the corresponding left profile of right-side simulated intersection in the two dimensions is duplicated as well based on the symmetry, when the coil is at the bottom. In the upper and left panel of
Figure 16(a) the term, A terminal of coil, is defined above the dielectric window, while in the upper and right panel of
Figure 16(a) the term, B terminal of coil, is defined above the dielectric. And in the upper panel of
Figure 16(b), assume the coil current flows as illustrated, A terminal in and B terminal out, at the moment of chamber clockwise rotation. Then, after this clockwise rotation, in the down panel of
Figure 16(a), B terminal of coil is shifted into the left profile and A terminal of coil is shifted into the right profile. Furthermore, refer to the coil current flowing direction after the clockwise rotation in the down panel of
Figure 16(b) and it changes into, A terminal out and B terminal in, due to the asymmetry introduced. Namely, the coil current flow changes its direction when it rotates a
. Actually, the right two sub-panels of
Figure 16(a) correspond to the
Figure 14(a) and
Figure 15(a), separately. Moreover, the left two sub-panels of
Figure 16(a) correspond to the left-hand spin of neutrino and the right-hand spin of anti-neutrino, respectively, but with coil current flowing into the page.
It is believed that in Ref. [
17], at the self-coagulation role that condenses scattering mass into particles, the macroscopic plasma mass does not hold spin property, but has the self-diffusion or strong ambi-polar diffusion to constitute a dynamic balance between the mass compression and dispersion. Nevertheless, for microscopic plasma mass, such as neutrino and anti-neutrino, it is believed that self-coagulation exists as well to sustain the particle property, but transport phenomena does not exist anymore due to the tiny mass, size and space. Instead, the spin is needed to construct the dynamic balance of mass compression and dispersion. The predictions in
Figure 16 about the spin properties are based on the macroscopic view; nevertheless, this macroscopic analysis based on the transport is in accord to the real spin property, again illustrating that the microscopic quantum property will comply with the real body particle, along with the space size increasing. Besides, the thereby depicted “spins” in
Figure 16(a) all orient toward the space, which validates that a spin in the microscopic particles is to abandon mass to the space, so against the self-coagulation that accumulate mass from the space. Lastly, the summed molecules, SF
6 and Ar in
Figure 14(b) and
Figure 15(b), are not coagulated but sunk, because of a mass balance that all plasma species, e.g., electrons, anions, cations, neutrals, and even further the positron, all arise from the mixed feedstock gases.