Submitted:
08 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
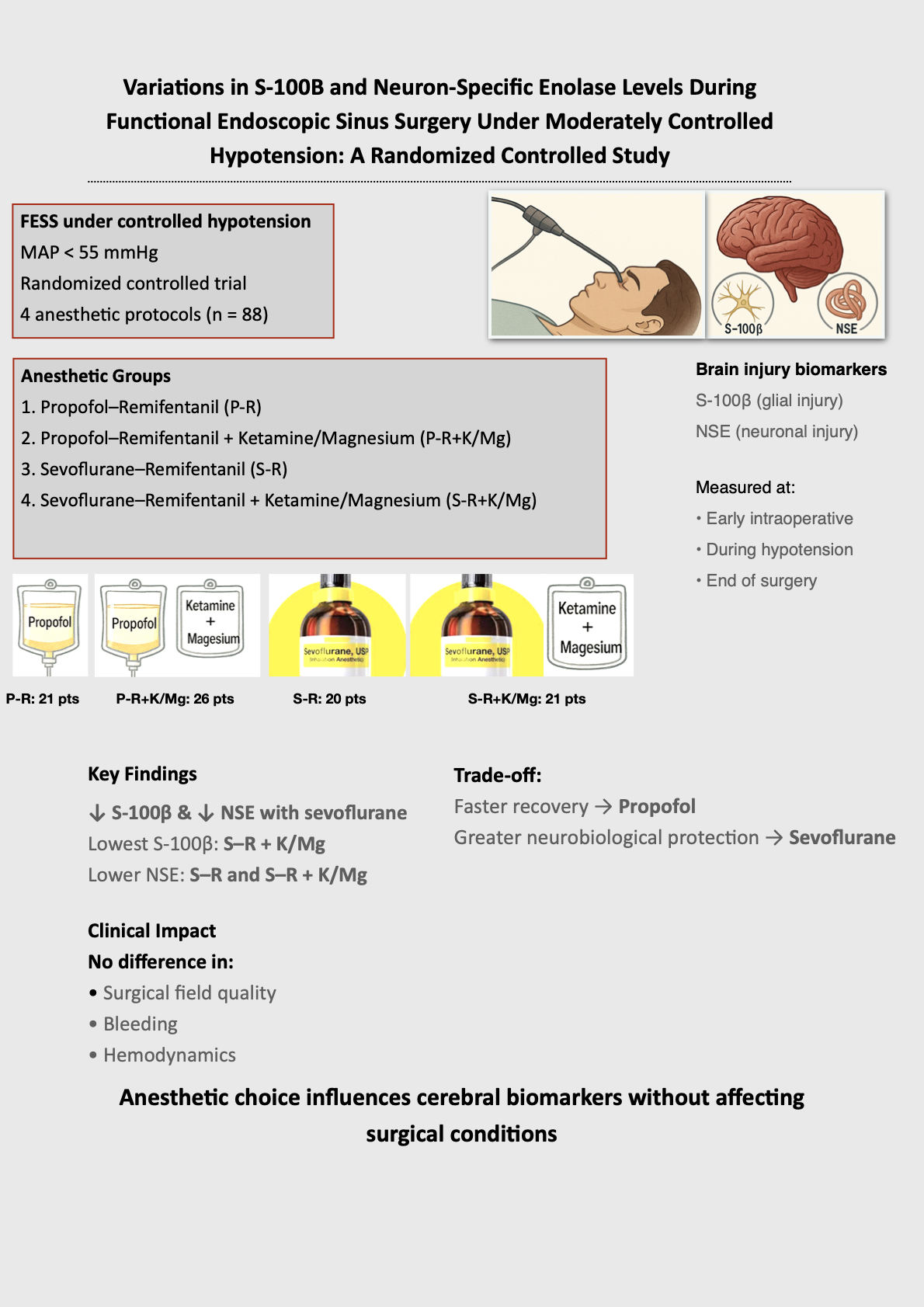
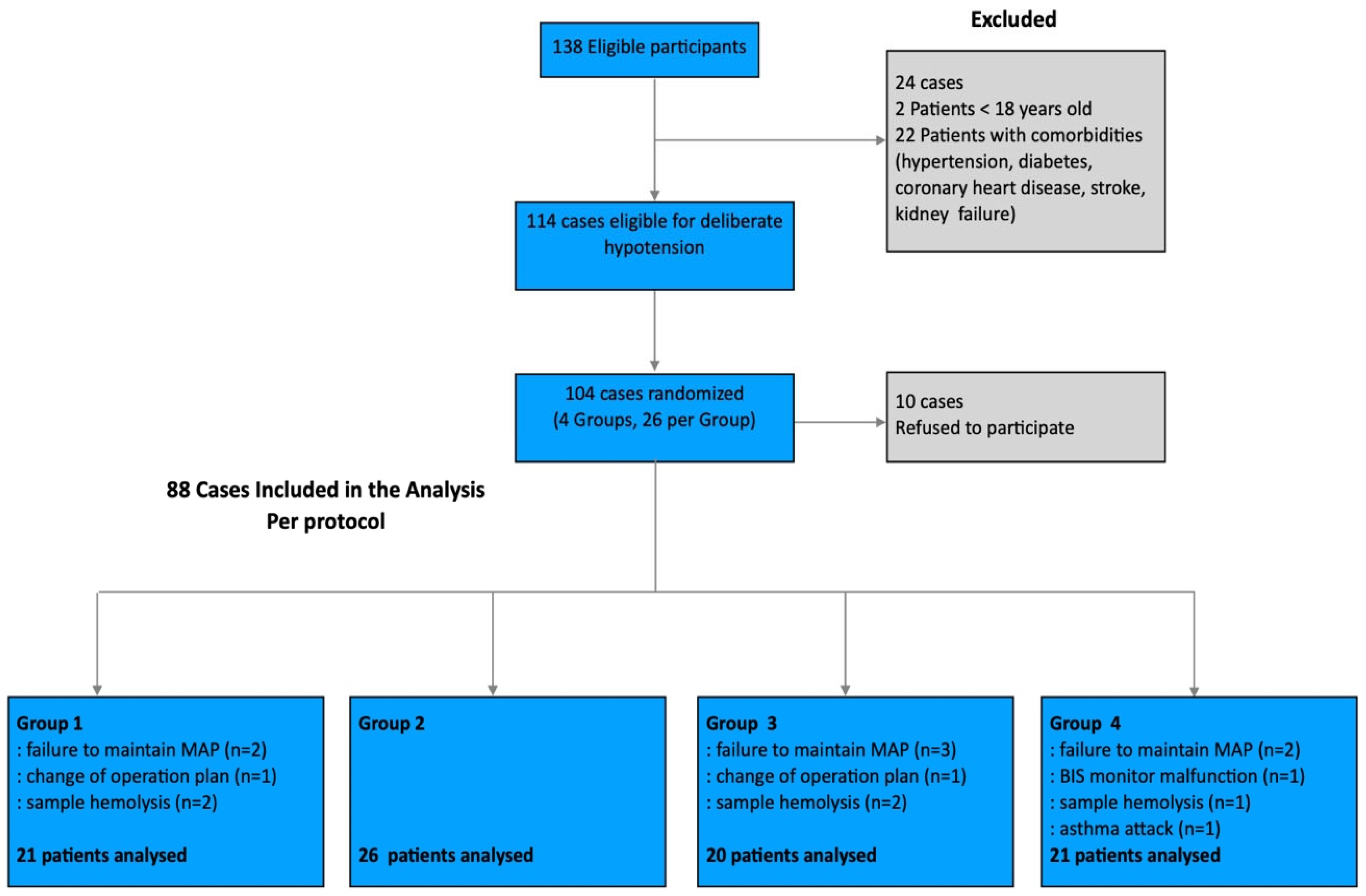
2.1. Study Design and Patient Selection
2.2. Randomization and Group Allocation
2.3. Perioperative Monitoring and Procedures
2.4. Anesthesia Induction and Sample Collection Procedures
2.5. Statistical Analysis and Calculation of Patient Sample
2.6. Outcomes Measures
3. Results
3.1. Demographics and Baseline Characteristics
3.2. Primary Outcomes
3.3. Secondary Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of Variance |
| ASA | American Society of Anesthesiologists |
| BIS | Bispectral index |
| BMI | Body mass index |
| CI | Confidence interval |
| CNS | Central Neuron System |
| ETCO₂ | End-tidal carbon dioxide |
| FESS | Functional endoscopic sinus surgery |
| HR | Heart rate |
| IV | Intravenous |
| LOD | Limit of detection |
| MAC | Minimum alveolar concentration |
| MAP | Mean arterial pressure |
| Mg+ | Magnesium sulfate |
| NSE | Neuron-specific enolase |
| NMDA | N-Methyl-D-aspartate |
| PACU | Post-anesthesia care unit |
| PCO₂ | Partial pressure of carbon dioxide |
| REML | Restricted maximum likelihood |
| S-100B | S-100 beta protein |
| SAS | Sedation–Agitation Scale |
| SD | Standard deviation |
References
- Chhabra, A.; Saini, P.; Sharma, K.; Chaudhary, N.; Singh, A.; Gupta, S. Controlled hypotension for FESS: A randomised double-blinded comparison of magnesium sulphate and dexmedetomidine. Indian J. Anaesth. 2020, 64, 24–30. [Google Scholar] [CrossRef]
- Giriyapur, P.; Madhusudhana, R. Controlled Hypotension for Functional Endoscopic Sinus Surgery With Two Different Doses of Fentanyl. Cureus 2023. [Google Scholar] [CrossRef]
- relationship_between_intraoperative_mean_arterial.6. p. 6.
- Hallqvist, L.; Granath, F.; Huldt, E.; Bell, M. Intraoperative hypotension is associated with acute kidney injury in noncardiac surgery. Eur. J. Anaesthesiol. 2018, 35, 273–9. [Google Scholar] [CrossRef]
- Van Waes, J.A.R.; Van Klei, W.A.; Wijeysundera, D.N.; Van Wolfswinkel, L.; Lindsay, T.F.; Beattie, W.S. Association between intraoperative hypotension and myocardial injury after vascular surgery. Anesthesiology 2016, 124, 35–44. [Google Scholar] [CrossRef]
- Mugabo, E.N.; Kulimushi, Y.M.; Pollach, G.; Sabra, R.A.; Beltagy, R.S.; Blaise Pascal, F.N. Clonidine and dexmedetomidine for controlled hypotension during functional endoscopic sinus surgery: a comparative study. BMC Anesthesiol. 2024, 24. [Google Scholar] [CrossRef]
- Kumar, B.R.P.; Venkatesh, C.B.; Hirguppe Somashekhar, A. EUROPEAN JOURNAL OF CARDIOVASCULAR MEDICINE European Journal of Cardiovascular Medicine Dexmedetomidine versus Esmolol for Induced Hypotension during Functional Endoscopic Sinus Surgery-A Prospective Randomised Comparative Study Open Access Article Title: Dexmedetomidine versus Esmolol for Induced Hypotension during Functional Endoscopic Sinus Surgery-A Prospective Randomised Comparative Study. Available online: https://www.healthcare-bulletin.co.uk/.
- Sahu, B.P.; Nayak, L.K.; Mohapatra, P.S.; Mishra, K. Induced Hypotension in Functional Endoscopic Sinus Surgery: A Comparative Study of Dexmedetomidine and Esmolol. Cureus 2021. [Google Scholar] [CrossRef]
- Kenny, J.D.; Westover, M.B.; Ching, S.N.; Brown, E.N.; Solt, K. Propofol and sevoflurane induce distinct burst suppression patterns in rats. Front. Syst. Neurosci. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, R.; Panda, N.; Bhagat, H.; et al. Comparison of propofol and sevoflurane on cerebral oxygenation using juglar venous oximetery (SjVo2) in patients undergoing surgery for traumatic brain injury. Asian J. Neurosurg. 2020, 15, 614–9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shan, G.J.; Zhang, Y.X.; et al. Propofol compared with sevoflurane general anaesthesia is associated with decreased delayed neurocognitive recovery in older adults. Br. J. Anaesth. 2018, 121, 595–604. [Google Scholar] [CrossRef]
- Jang, J.S.; Kwon, Y.; Hwang, S.M.; et al. Comparison of the effect of propofol and desflurane on S-100Β and GFAP levels during controlled hypotension for functional endoscopic sinus surgery: A randomized controlled trial. Medicine 2019, 98, e17957. [Google Scholar] [CrossRef] [PubMed]
- Zanza, C.; Piccolella, F.; Racca, F.; et al. Ketamine in Acute Brain Injury: Current Opinion Following Cerebral Circulation and Electrical Activity. Healthcare (Switzerland) 2022. [Google Scholar] [CrossRef] [PubMed]
- Gregers, M.C.T.; Mikkelsen, S.; Lindvig, K.P.; Brøchner, A.C. Ketamine as an Anesthetic for Patients with Acute Brain Injury: A Systematic Review. Neurocritical Care 2020. [Google Scholar] [CrossRef]
- Choudhury, D.; Autry, A.E.; Tolias, K.F.; Krishnan, V. Ketamine: Neuroprotective or Neurotoxic? Frontiers in Neuroscience 2021. [Google Scholar] [CrossRef]
- Hakim, K.Y.K.; Wahba, W.Z. Effect of N-methyl-d-aspartate (NMDA) receptor antagonist combination as adjuvant to general anesthesia in functional endoscopic sinus surgery. Ain-Shams Journal of Anesthesiology 2019, 11. [Google Scholar] [CrossRef]
- Sen, A.P.; Gulati, A. Use of Magnesium in Traumatic Brain Injury. [CrossRef]
- Suk Lee, J.; Min Han, Y.; Sung Yoo, D.; et al. A Molecular Basis for the Efficacy of Magnesium Treatment Following Traumatic Brain Injury in Rats. 2004. [Google Scholar]
- Nutrition and Traumatic Brain Injury : Improving Acute and Subacute Health Outcomes in Military Personnel. National Academies Press, 2011.
- Jitsinthunun, T.; Raksakietisak, M.; Pantubtim, C.; Mahatnirunkul, P. Effects of Magnesium Sulfate on Intraoperative Blood Loss and Anesthetic Requirement in Meningioma Patients Undergoing Craniotomy with Tumor Removal: A Prospective Randomized Study. J. Neuroanaesth. Crit. Care 2022, 9, 168–76. [Google Scholar] [CrossRef]
- Makovec, M.; Skitek, M.; Šimnovec, L.; Jerin, A. Neuron-Specific Enolase and S100B as Biomarkers of Ischemic Brain Injury During Surgery. Clinics and Practice 2025. [Google Scholar] [CrossRef]
- Khanduja, S.; Kim, J.; Kang, J.K.; et al. Hypoxic-Ischemic Brain Injury in ECMO: Pathophysiology, Neuromonitoring, and Therapeutic Opportunities. Cells 2023. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Yao, Z. Relationships of serum VILIP-1, NSE, and ADP levels with postoperative cognitive dysfunction in elderly patients undergoing general anesthesia: a retrospective, observational study. Journal of International Medical Research 2023, 51. [Google Scholar] [CrossRef] [PubMed]
- Eyglóardóttir, K.; Michaëlsson, I.; Hallén, T.; Jakola, A.; Skoglund, T. Circulating Brain Injury Biomarkers for Predicting Outcomes Following Elective Neurosurgery: A Scoping Review. World Neurosurgery 2024. [Google Scholar] [CrossRef]
- Fletcher-Sandersjöö, A.; Lindblad, C.; Thelin, E.P.; et al. Serial S100B sampling detects intracranial lesion development in patients on extracorporeal membrane oxygenation. Front. Neurol. 2019, 10. [Google Scholar] [CrossRef]
- Michetti, F.; Clementi, M.E.; Di Liddo, R.; et al. The S100B Protein: A Multifaceted Pathogenic Factor More Than a Biomarker. International Journal of Molecular Sciences 2023. [Google Scholar] [CrossRef]
- Komarnicki, P.; Musiałkiewicz, J.; Stańska, A.; et al. Circulating Neuroendocrine Tumor Biomarkers: Past, Present and Future. Journal of Clinical Medicine 2022. [Google Scholar] [CrossRef]
- Zhi, M.; Huang, J.; Jin, X. Clinical value of serum neuron-specific enolase in sepsis-associated encephalopathy: a systematic review and meta-analysis. Syst. Rev. 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Hodge, D.R.; Gillespie, D.F. Phrase Completion Scales. [PubMed]
- Reis, C.; Wang, Y.; Akyol, O.; et al. What’s new in traumatic brain injury: Update on tracking, monitoring and treatment. Int. J. Mol. Sci. 2015, 16, 11903–65. [Google Scholar] [CrossRef] [PubMed]
- Stefanović, B.; Durić, O.; Stanković, S.; et al. Elevated Serum Protein S100B and Neuron Specific Enolase Values as Predictors of Early Neurological Outcome after Traumatic Brain Injury. J. Med. Biochem. 2017, 36, 314–21. [Google Scholar] [CrossRef]
- Dragas, M.; Koncar, I.; Opacic, D.; et al. Fluctuations of serum neuron specific enolase and protein S-100B concentrations in relation to the use of shunt during carotid endarterectomy. PLoS One 2015, 10. [Google Scholar] [CrossRef]
- Dash, P.K.; Zhao, J.; Hergenroeder, G.; Moore, A.N. Biomarkers for the Diagnosis, Prognosis, and Evaluation of Treatment Efficacy for Traumatic Brain Injury. [CrossRef]
- Clausen, N.G.; Antonsen, S.; Spielmann, N.; Hansen, T.G.; Weiss, M.; Ringer, S.K. Hypotension and Hypocapnia during General Anesthesia in Piglets: Study of S100b as an Acute Biomarker for Cerebral Tissue Injury. J. Neurosurg. Anesthesiol. 2020, 32, 273–8. [Google Scholar] [CrossRef]
- INTERNATIONAL COUNCIL FOR HARMONISATION OF TECHNICAL REQUIREMENTS FOR PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED GUIDELINE GOOD CLINICAL PRACTICE (GCP) E6(R3).
- Kwon, Y.; Jang, J.S.; Hwang, S.M.; et al. Range of S-100Β levels during functional endoscopic sinus surgery with moderately controlled hypotension. European Archives of Oto-Rhino-Laryngology 2017, 274, 3527–32. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.; Stapelfeldt, W.H.; Khanna, A.K.; et al. Intraoperative Hypotension Is Associated with Adverse Clinical Outcomes after Noncardiac Surgery. Anesth. Analg. 2021, 132, 1654–65. [Google Scholar] [CrossRef]
- Bijker, J.B.; Persoon, S.; Peelen, L.M.; et al. Intraoperative Hypotension and Perioperative Ischemic Stroke after General Surgery A Nested Case-Control Study. 2012. [Google Scholar]
- Thelin, E.P.; Nelson, D.W.; Bellander, B.M. A review of the clinical utility of serum S100B protein levels in the assessment of traumatic brain injury. Acta Neurochirurgica 2017. [Google Scholar] [CrossRef] [PubMed]
- Park, H.P.; Jeong, E.J.; Kim, M.H.; et al. Effects of sevoflurane on neuronal cell damage after severe cerebral ischemia in rats. Korean J. Anesthesiol. 2011, 61, 327–31. [Google Scholar] [CrossRef]
- Park, S. Volatile anesthetics as a neuroprotective agent. Korean Journal of Anesthesiology 2011. [Google Scholar] [CrossRef]
| Variable | Group 1 (n=21) N [%] Mean [SD] |
Group 2 (n=26) N [%] Mean [SD] |
Group 3 (n=20) N [%] Mean [SD] |
Group 4 (n=21) N [%] Mean [SD] |
p-value |
|---|---|---|---|---|---|
|
Gender male female |
12 [57] 9 [42.9] |
20 [71.4] 6 [28.6] |
12 [60] 8 [40] |
15[ 71.4] 6 [28.6] |
0.64 |
| Age (years) | 43.047 [18.94] | 40.53 [13.39] |
44.47 [13.41] |
43.23 [17.38] |
0.85 |
| Weight (kg) | 77.19 [17.48] |
81.17 [15.79] |
84.45 [18.3] |
81.19 [17.07] |
0.6 |
| B.M.I (kg/m²) | 25.76 [3.7] |
27.18 [4.13] |
28.31 [5.69] |
27.27 [5.41] |
0.43 |
|
ASA 1 2 3 |
11 [55] 8 [40] 1 [5] |
15 [53,6] 13 [46.4]0 |
11 [61.1] 5 [27.8] 2 [11.1] |
8[38.1] 10[47,6] 3 [14,3] |
0.37 |
|
Smoking Smokers Non-smokers |
8 [40] 12 [60] |
15[53.6] 13[46.4] |
4[23.5] 13[76.5] |
12[57.1] 9 [42.9] |
0.2 |
|
Surgery Duration (min) |
98.4 [28.6] |
103.85 [21.9] |
99.52 [29.17] |
114.23 [34.13] |
0.43 |
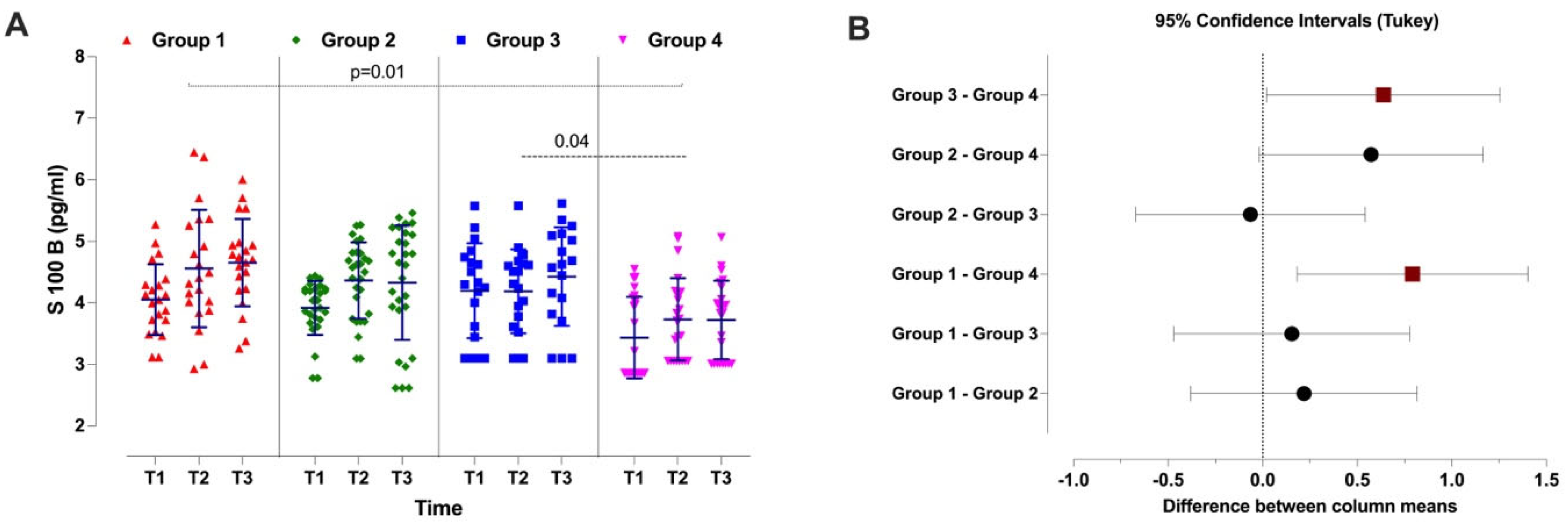
| S100B – Comparisons Between Groups | |||
|---|---|---|---|
| Tukey’s multiple comparisons test | Mean diff. | 95% CI of diff. | Adjusted P Value |
| Group 1 vs. Group 2 | 0.22 | -0.38 to 0.82 | 0.66 |
| Group 1 vs. Group 3 | 0.15 | -0.47 to 0.78 | 0.85 |
| Group 1 vs. Group 4 | 0.79 | 0.18 to 1.40 | 0.01 |
| Group 2 vs. Group 3 | -0.06 | -0.67 to 0.54 | 0.98 |
| Group 2 vs. Group 4 | 0.57 | -0.02 to 1.20 | 0.06 |
| Group 3 vs. Group 4 | 0.64 | 0.02 to 1.30 | 0.04 |
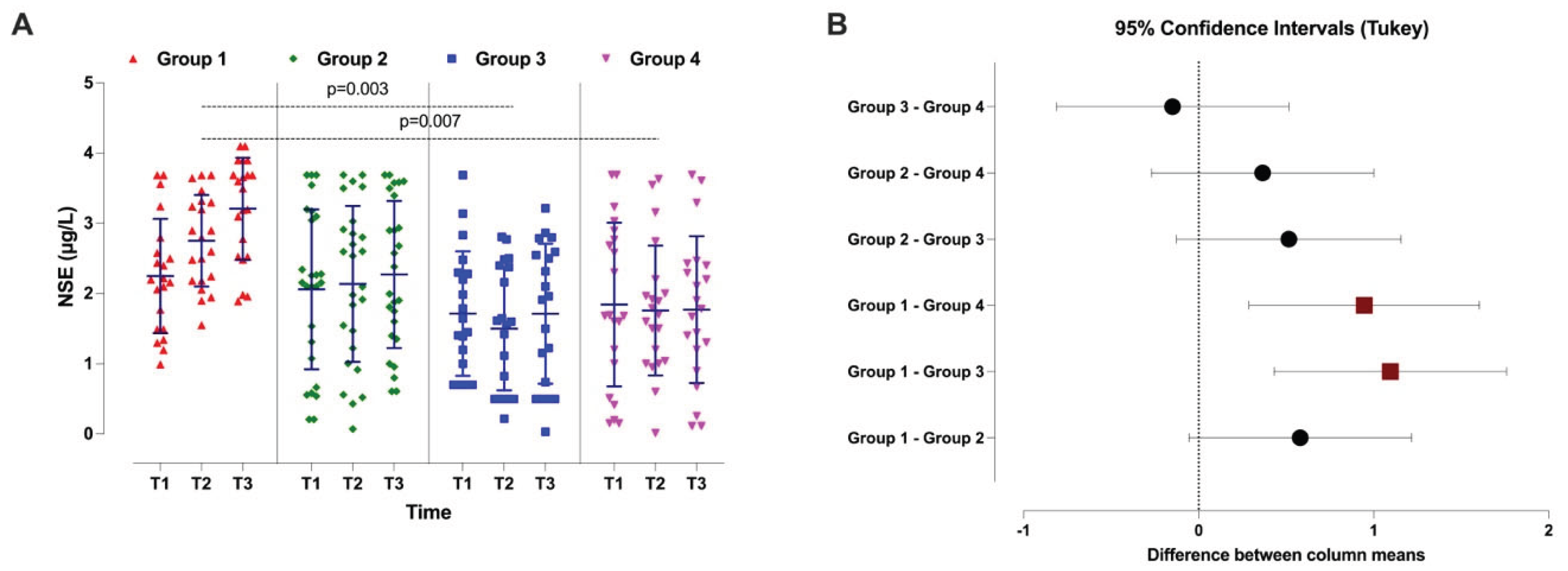
| Neuron Specific Enolase (NSE)—Comparisons Between Groups | |||
|---|---|---|---|
| Tukey’s multiple comparisons test | Mean diff. | 95% CI of diff. | Adjusted P Value |
| Group 1 vs. Group 2 | 0.58 | -0.05 to 1.21 | 0.07 |
| Group 1 vs. Group 3 | 1.09 | 0.43 to 1.76 | 0.003 |
| Group 1 vs. Group 4 | 0.94 | 0.28 to 1.60 | 0.007 |
| Group 2 vs. Group 3 | 0.51 | -0.12 to 1.15 | 0.12 |
| Group 2 vs. Group 4 | 0.36 | -0.27 to 1.00 | 0.32 |
| Group 3 vs. Group 4 | -0.15 | -0.81 to 0.51 | 0.88 |
| Recovery Time (Time to Verbal Response, min) | U | Z | p-value |
|---|---|---|---|
| Group1 vs Group2 | 113.5 | −3.49 | <0.001 |
| Group1 vs Group3 | 126.0 | −1.34 | 0.18 |
| Group1 vs Group4 | 100.0 | −2.88 | 0.004 |
| Group2 vs Group3 | 151.0 | −2.04 | 0.04 |
| Group2 vs Group4 | 290.0 | −0.08 | 0.93 |
| Group3 vs Group4 | 122.5 | −1.65 | 0.10 |
| Extubation Time(min) | |||
| Group1 vs Group2 | 115.5 | −3.44 | <0.001 |
| Group1 vs Group3 | 122.0 | −1.46 | 0.14 |
| Group1 vs Group4 | 102.5 | −2.81 | 0.005 |
| Group2 vs Group3 | 128.5 | −2.57 | 0.01 |
| Group2 vs Group4 | 280.0 | −0.28 | 0.77 |
| Group3 vs Group4 | 112.0 | −1.95 | 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




