Submitted:
08 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction: Toward a Paradigm Refinement
2. Three Persistent Misconceptions in Applying Thermodynamics to Body Weight Regulation
2.1. Calories Cannot Be Eaten – Nor Oxidized
2.2. The Irrelevance of Einstein’s E=mc² to Human Metabolism
2.3. The First Law of Thermodynamics Concerns Only Energy, Not Mass
2.4. Why These Distinctions Matter
4. The Inefficiency of the Two-Step Conversion Process in EBM
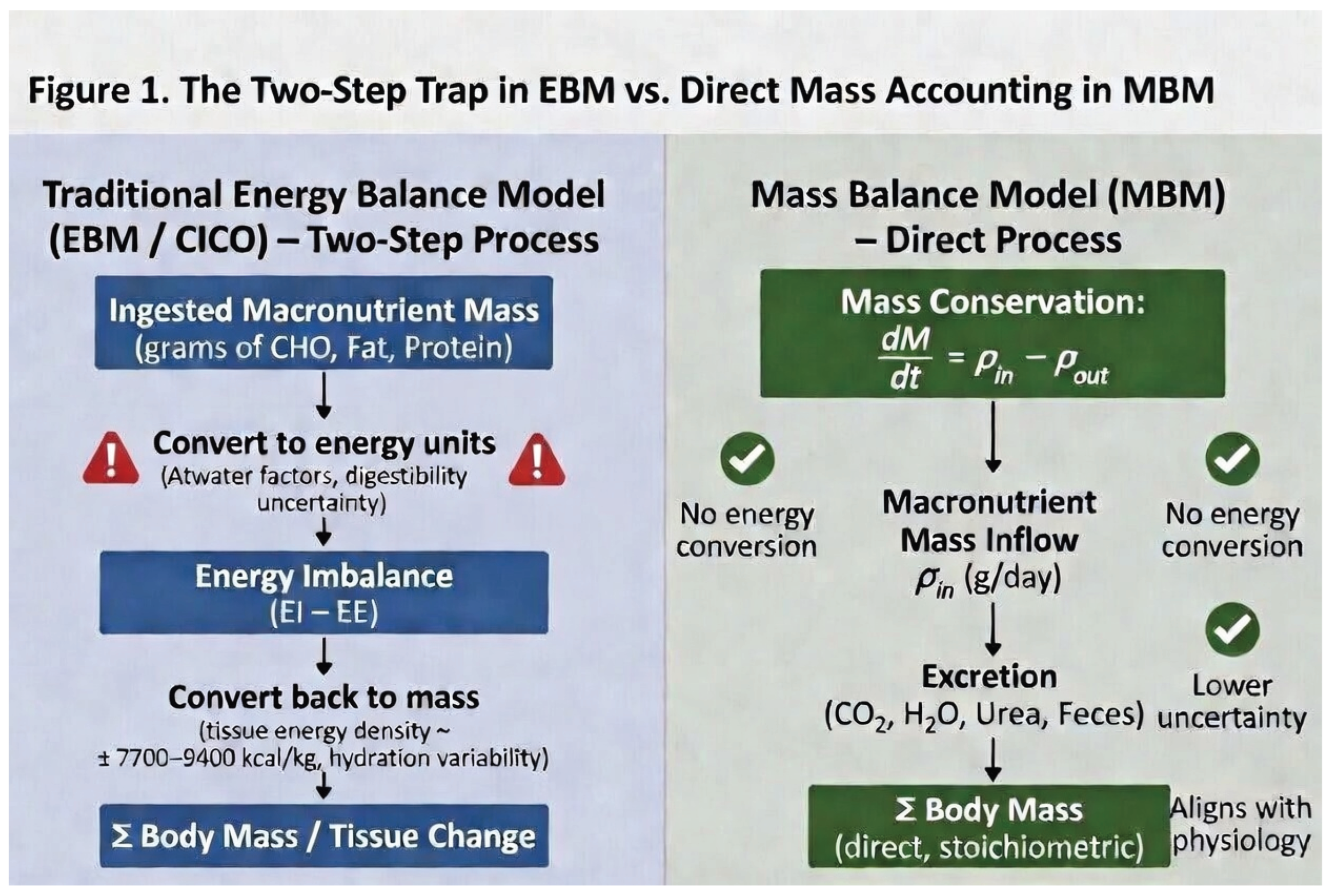
4. Conclusion
Author Contributions
Funding
Ethics approval and consent to participate
Consent for publication
Availability of data
Acknowledgments
Conflicts of Interest
References
- Hall KD, Farooqi IS, Friedman JM, Klein S, Loos RJF, Mangelsdorf DJ, O’Rahilly S, Ravussin E, Redman LM, Ryan DH, Speakman JR, Tobias DK. The energy balance model of obesity: beyond calories in, calories out. Am J Clin Nutr. 2022 May 1;115(5):1243-1254. [CrossRef] [PubMed] [PubMed Central]
- Nutrition and Metabolic Management Branch of China International Exchange and Promotive Association for Medical and Health Care, Clinical Nutrition Branch of Chinese Nutrition Society, Chinese Diabetes Society, Chinese Society for Parenteral and Enteral Nutrition, Chinese Clinical Nutritionist Center of Chinese Medical Doctor Association. Guidelines for medical nutrition treatment of overweight/obesity in China (2021). Asia Pac J Clin Nutr. 2022;31(3):450-482. [CrossRef] [PubMed]
- Hassapidou M, Vlassopoulos A, Kalliostra M, Govers E, Mulrooney H, Ells L, Salas XR, Muscogiuri G, Darleska TH, Busetto L, Yumuk VD, Dicker D, Halford J, Woodward E, Douglas P, Brown J, Brown T. European Association for the Study of Obesity Position Statement on Medical Nutrition Therapy for the Management of Overweight and Obesity in Adults Developed in Collaboration with the European Federation of the Associations of Dietitians. Obes Facts. 2023;16(1):11-28. [CrossRef] [PubMed] [PubMed Central]
- Raynor HA, Morgan-Bathke M, Baxter SD, Halliday T, Lynch A, Malik N, Garay JL, Rozga M. Position of the Academy of Nutrition and Dietetics: Medical Nutrition Therapy Behavioral Interventions Provided by Dietitians for Adults With Overweight or Obesity, 2024. J Acad Nutr Diet. 2024 Mar;124(3):408-415. [CrossRef] [PubMed]
- Arencibia-Albite F. Serious analytical inconsistencies challenge the validity of the energy balance theory. Heliyon. 2020 Jul 10;6(7):e04204. Erratum in: Heliyon. 2020 Sep 14;6(9):e04609. doi: 10.1016/j.heliyon.2020.e04609. [CrossRef] [PubMed]
- Arencibia-Albite F. The energy balance theory is an inconsistent paradigm. J Theor Biol. 2022 Oct 7;550:111240. [CrossRef] [PubMed]
- Manninen AH. Chronic positive mass balance is the actual etiology of obesity: A living review. Global Transl Med. 2023, 2(1), 222. [CrossRef]
- Manninen AH. Mass balance and energy balance in body weight regulation: A response to Theodorakis’ comments. Global Transl Med. 2025, 4(3), 119–120. [CrossRef]
- Arencibia-Albite F. Empirical Evaluation of the Mass Balance Model in Human Bodyweight Regulation, 13 January 2026, PREPRINT (Version 1) available at Research Square. [CrossRef]
- Arencibia-Albite F. Quantitative Foundations of Energy Balance Are Not Empirically Reliable Under Ideal Conditions, 16 January 2026, PREPRINT (Version 1) available at Research Square. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



