Submitted:
06 April 2026
Posted:
08 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell culture and Virus
2.2. Animal Experiments
2.3. Quantitative Real Time PCR (qPCR) for Detection of the ASFV Genome
2.4. ASFV Antibody Detection
2.5. Serum Cytokine Analysis
3. Results
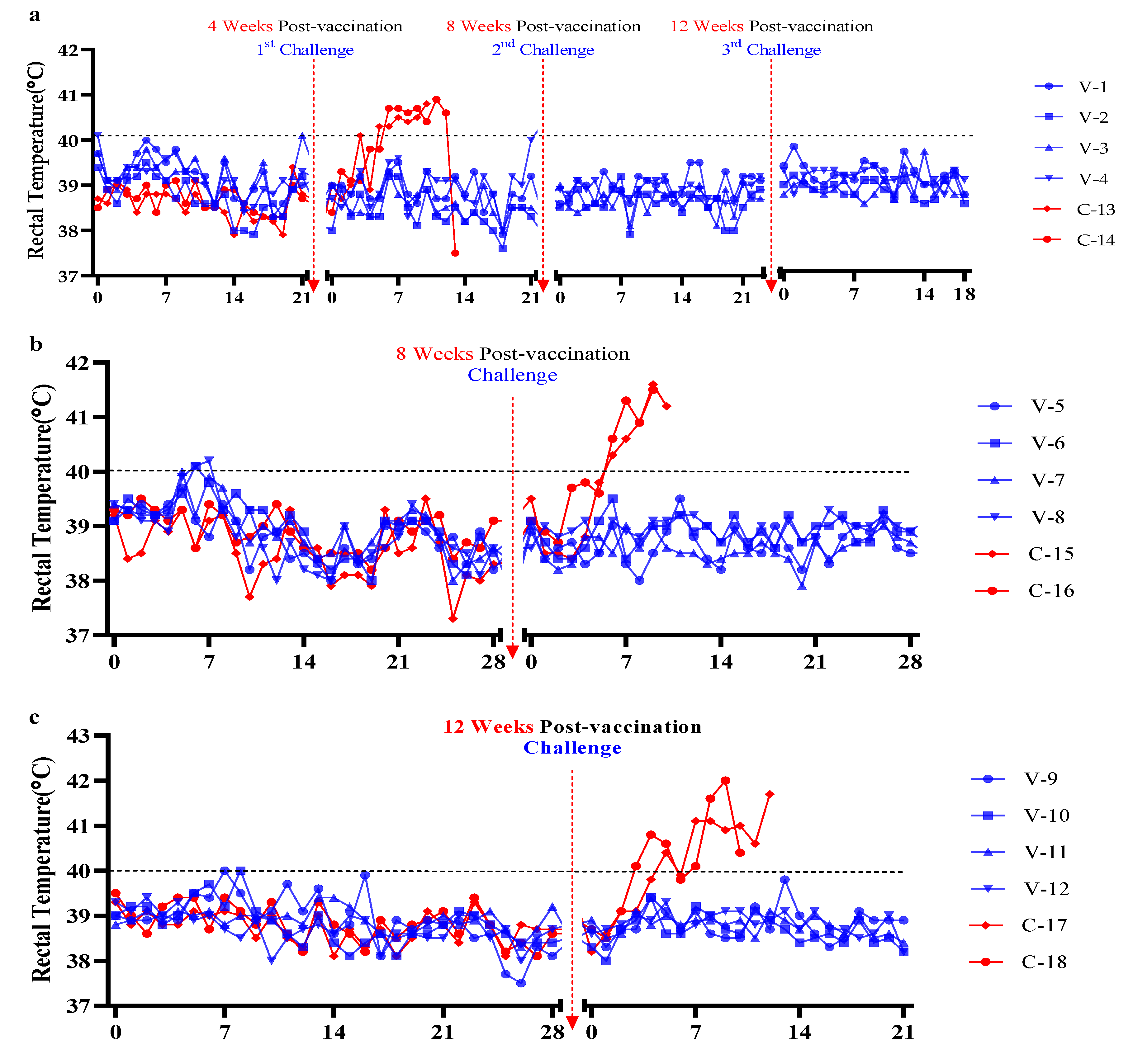
3.1. Protective Efficacy of ASFV-G-ΔI177L/ΔLVR Vaccination Against Multiple and Delayed Challenge in an Experimental Model
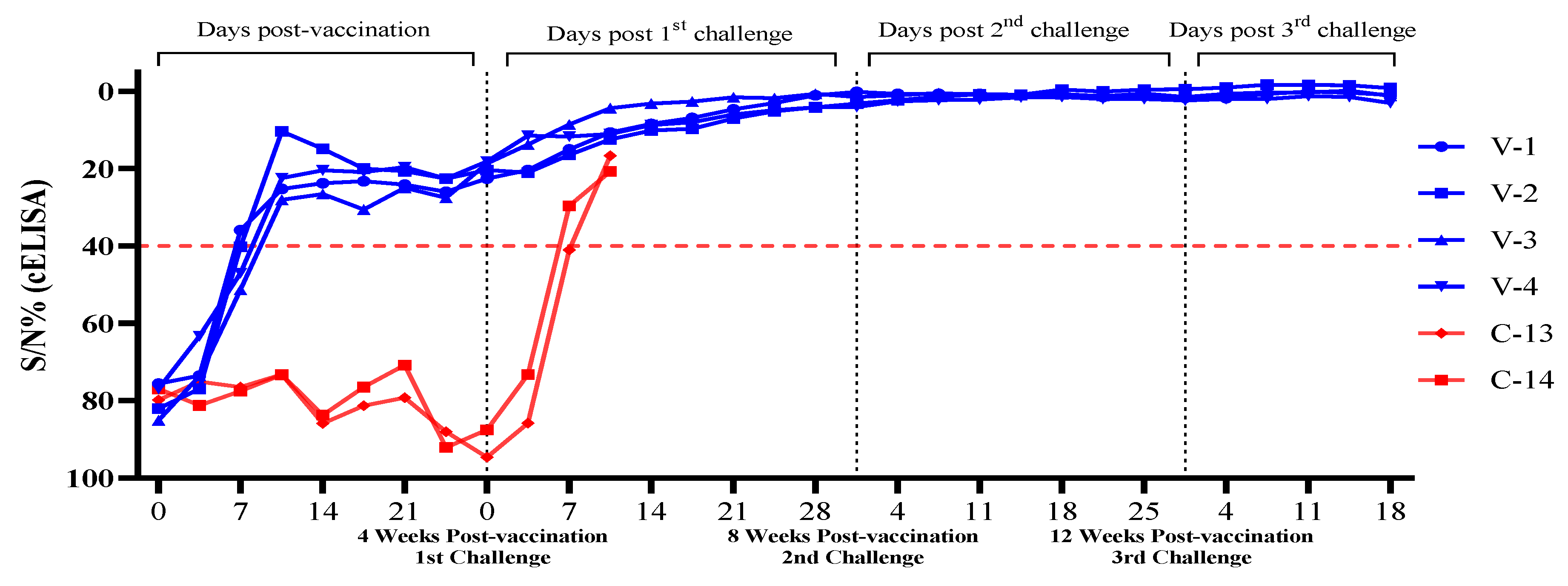
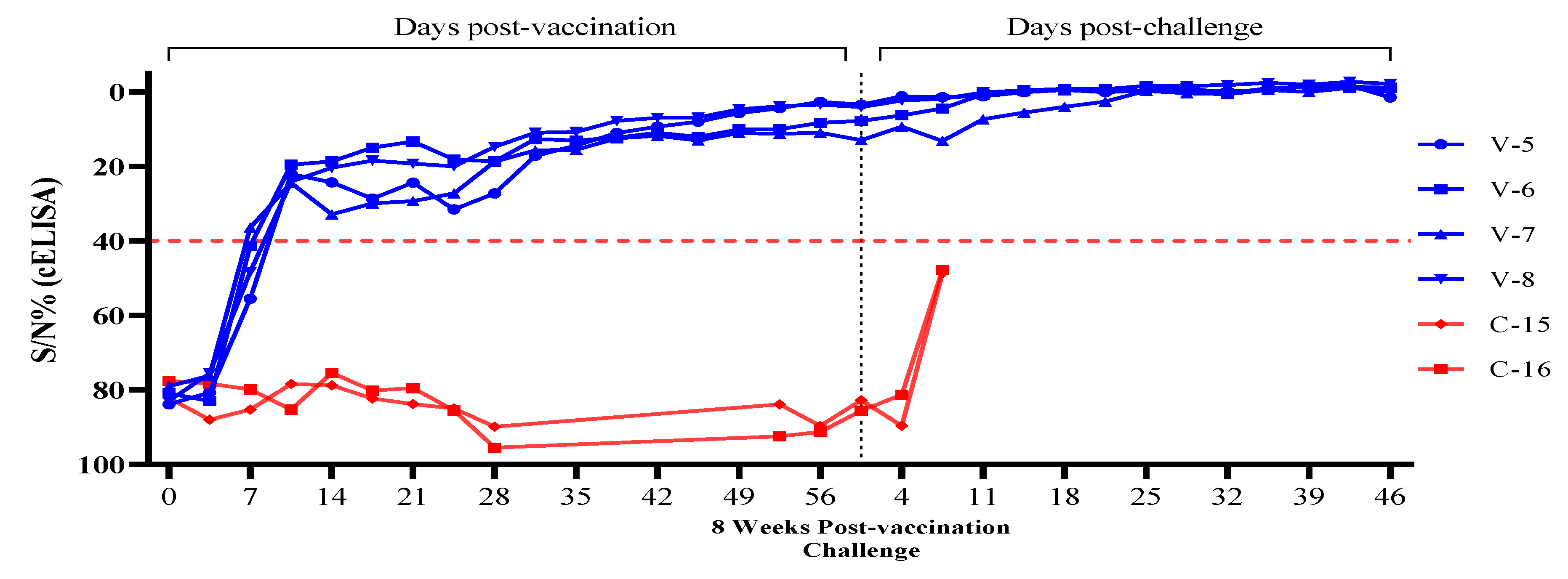
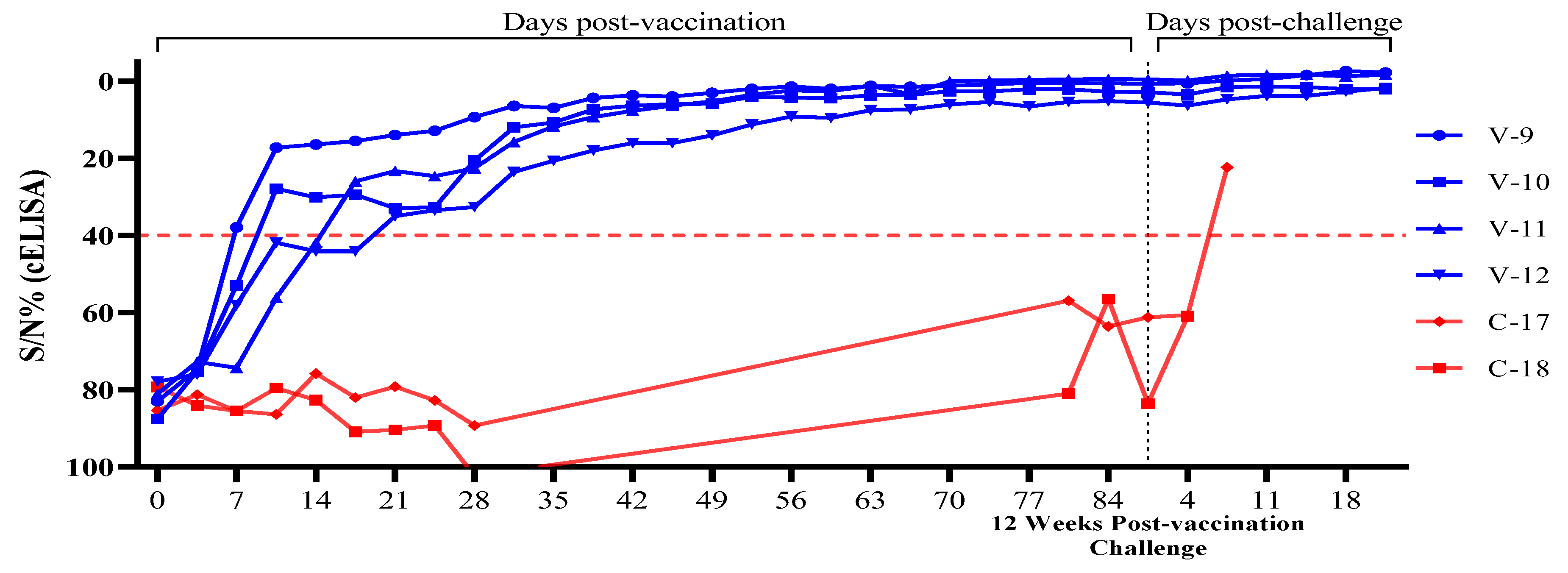
3.2. Evaluation of ASF Vaccine Antibodies in Challenged Pigs After ASFV-G-ΔI177L/ΔLVR Vaccination
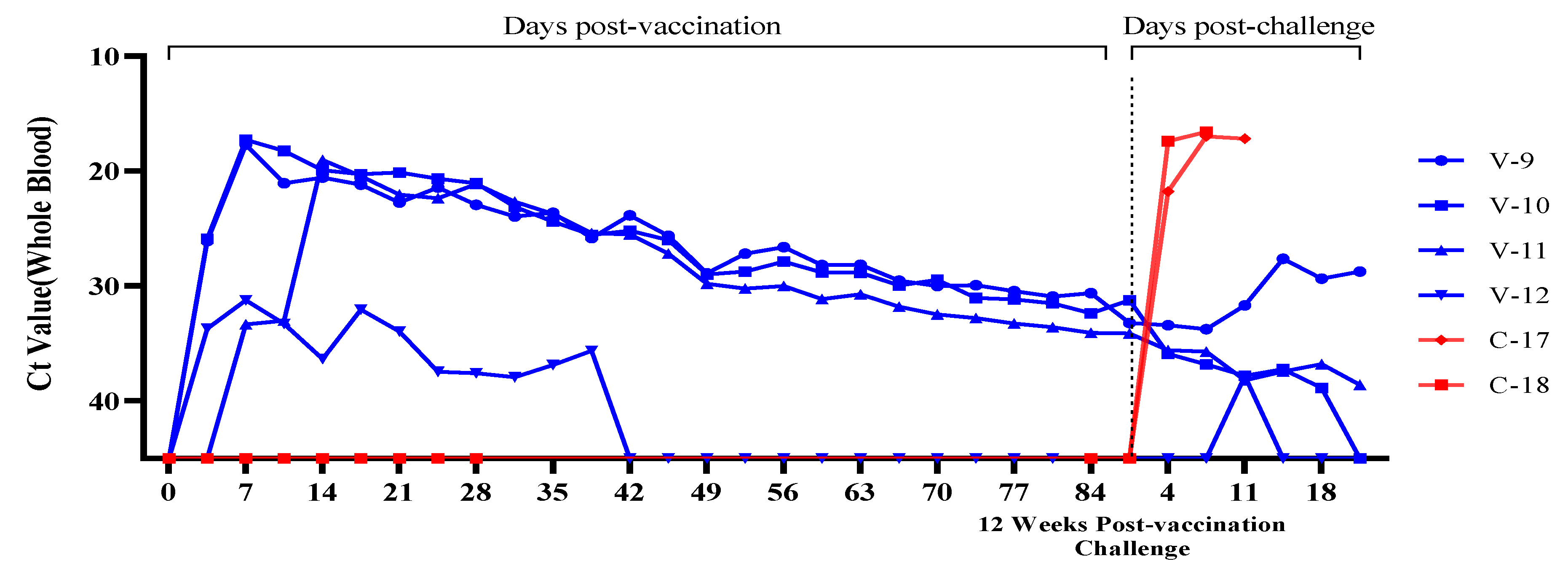
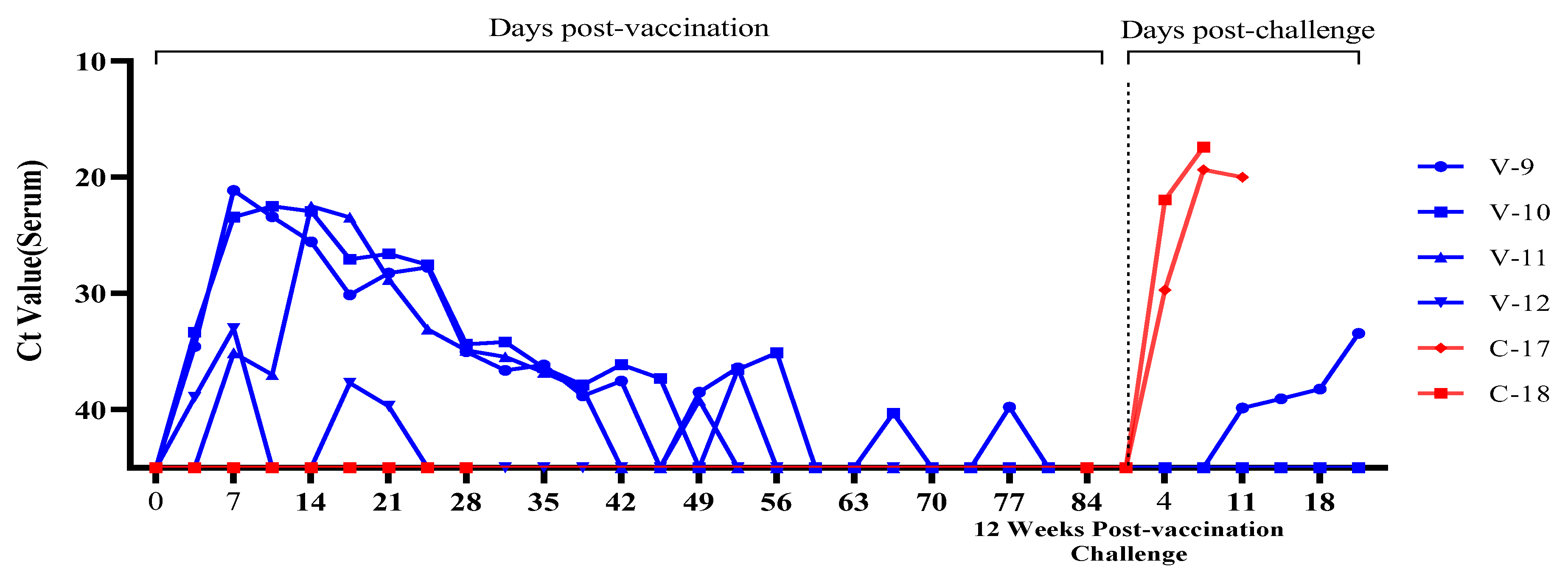
3.3. Evaluation of Viremia Against Multiple and Delayed Challenges Following ASFV-G-ΔI177L/ΔLVR Vaccination
Vaccine-Associated Viremia Assessment
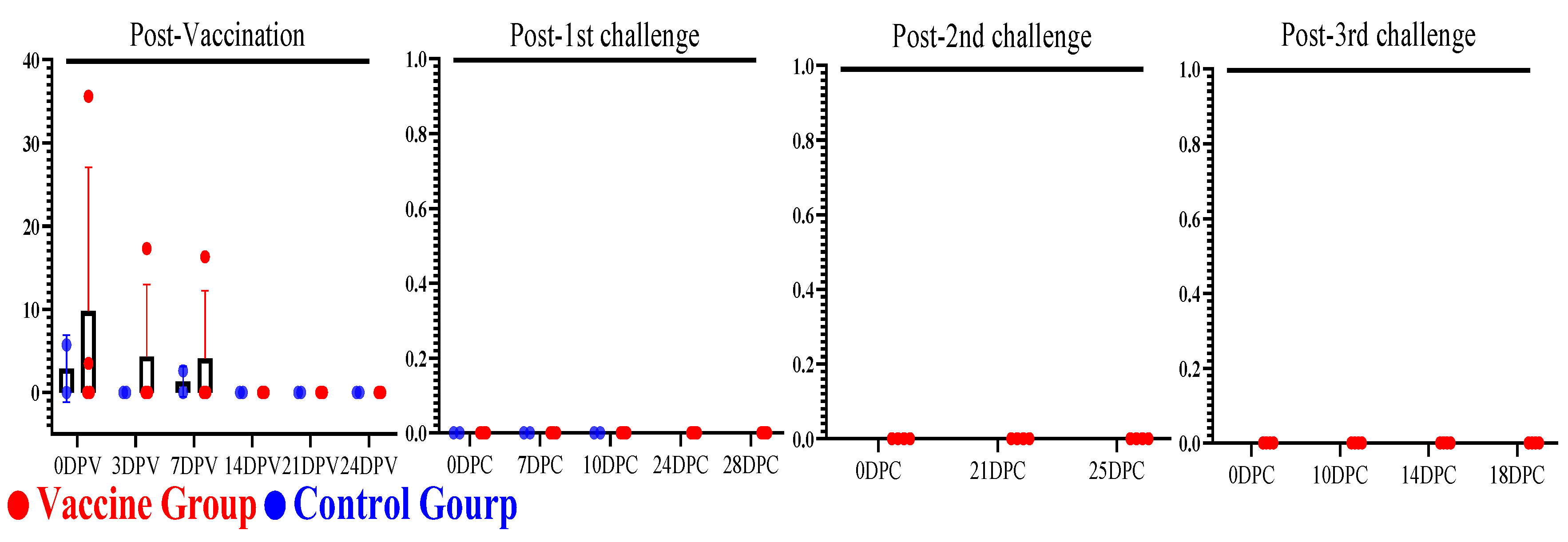
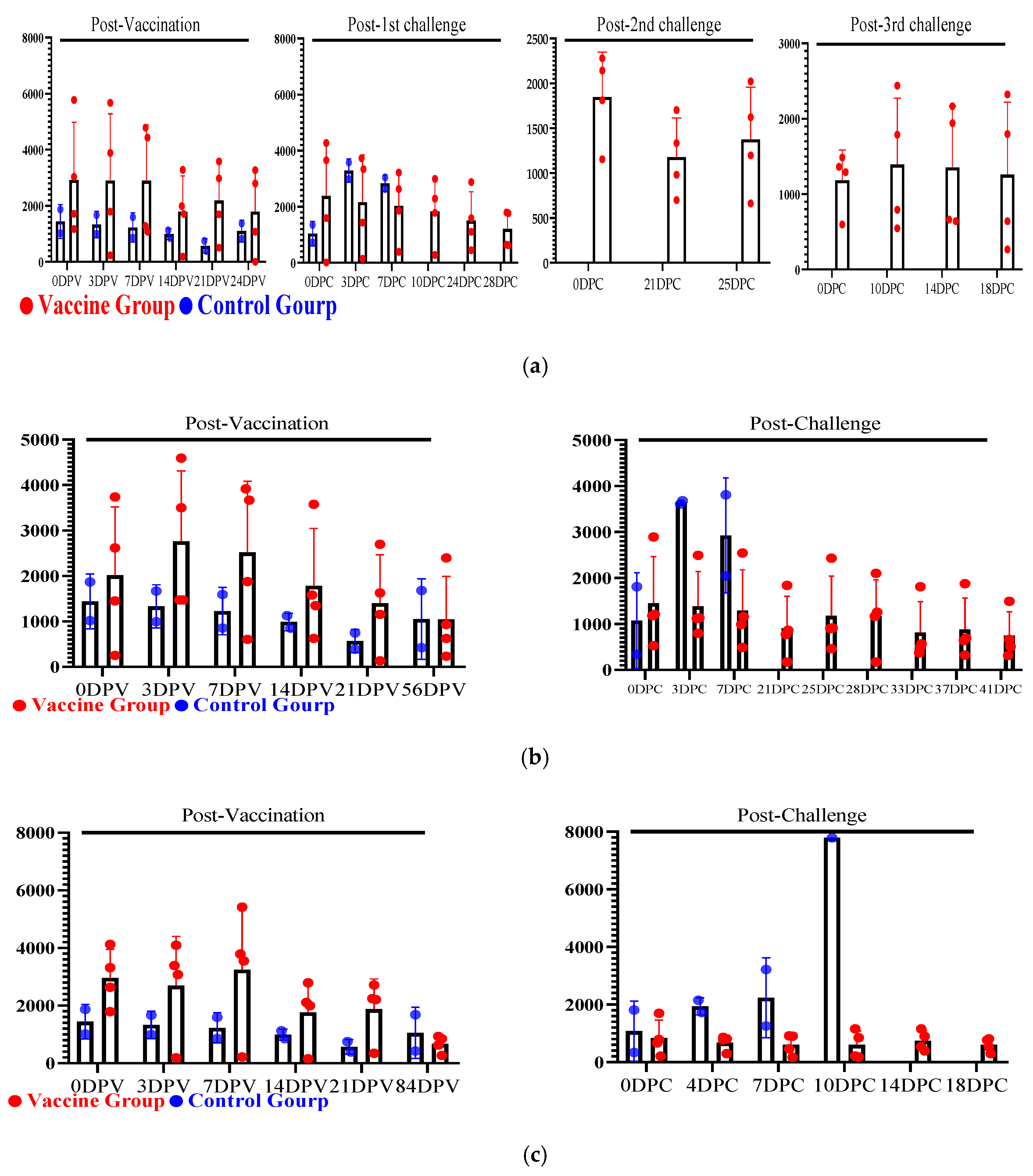
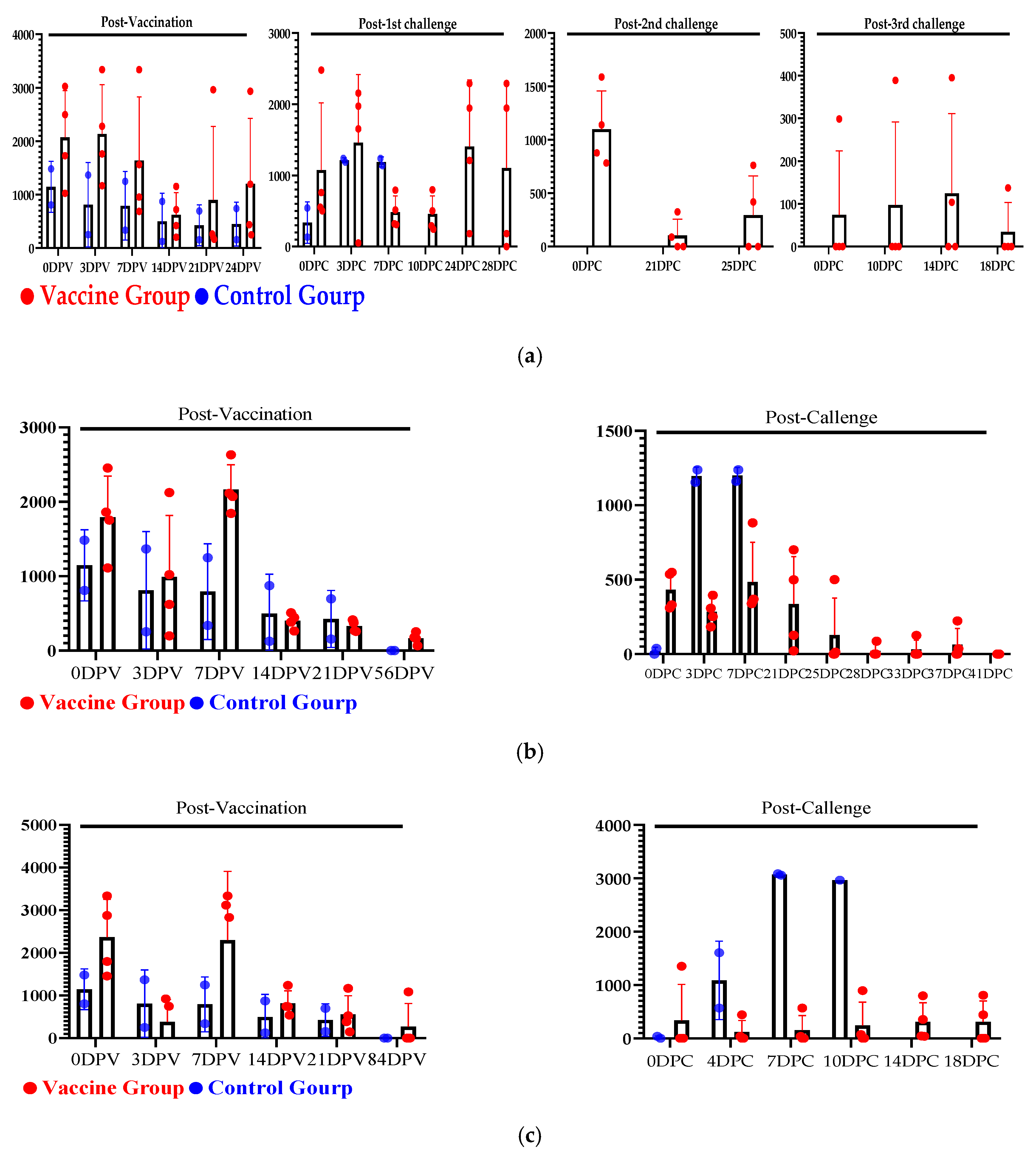
3.4. Evaluation of Serum Cytokines Against Multiple and Delayed Challenges Following ASFV-G-ΔI177L/ΔLVR Vaccination
3.4.1. Post-Vaccination Serum Cytokine Responses
3.4.2. Post-Challenge Serum Cytokine Responses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ASFV | African swine fever virus |
| LAVs | Live attenuated vaccines |
| WOAH | World Organization for Animal Health |
| PIPEC | Plum Island porcine epithelial cells |
| dpv | Days post-vaccination |
| dpc | Days post-challenge |
| WPV | Week post-vaccination |
| IM | Intramuscular |
| qPCR | Quantitative real-time PCR |
| HAD | Hemadsorption dose |
| TCID | Tissue culture infectious dose |
| ELISA | Enzyme-linked immunosorbent assay |
| Ct | Cycle threshold |
References
- Dixon, L.K.; Islam, M.; Nash, R.; Reis, A.L. African swine fever virus evasion of host defences. Virus Res. 2019, 266, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Penrith, M.L.; Vosloo, W. Review of African swine fever: Transmission, spread and control. J. S. Afr. Vet. Assoc. 2009, 80, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Eustace Montgomery, R.E. On a form of swine fever occurring in British East Africa (Kenya Colony). J. Comp. Pathol. Ther. 1921, 34, 159–191. [Google Scholar] [CrossRef]
- Sánchez-Cordón, P.J.; Montoya, M.; Reis, A.L.; Dixon, L.K. African swine fever: A re-emerging viral disease threatening the global pig industry. Vet. J. 2018, 233, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Lv, T.; Xie, X.; Song, N.; Zhang, S.; Ding, Y.; Liu, K.; Diao, L.; Chen, X.; Jiang, S.; Li, T.; et al. Expounding the role of tick in Africa swine fever virus transmission and seeking effective prevention measures: A review. Front. Immunol. 2022, 13, 1093599. [Google Scholar] [CrossRef]
- Sánchez-Vizcaíno, J.M.; Mur, L.; Bastos, A.D.S.; Penrith, M.L. New insights into the role of ticks in African swine fever epidemiology. Rev. Sci. Tech. 2015, 34, 503–511. [Google Scholar] [CrossRef]
- Costard, S.; Mur, L.; Lubroth, J.; Sanchez-Vizcaino, J.M.; Pfeiffer, D.U. Epidemiology of African swine fever virus. Virus Res. 2013, 173, 191–197. [Google Scholar] [CrossRef]
- Busch, F.; Haumont, C.; Penrith, M.L.; Laddomada, A.; Dietze, K.; Globig, A.; Guberti, V.; Zani, L.; Depner, K. Evidence-based African swine fever policies: Do we address virus and host adequately? Front. Vet. Sci. 2021, 8, 637487. [Google Scholar] [CrossRef]
- Penrith, M.-L.; van Heerden, J.; Pfeiffer, D.U.; Oļševskis, E.; Depner, K.; Chenais, E. Innovative research offers New Hope for managing African swine fever better in resource-limited smallholder farming settings: A timely update. Pathogens 2023, 12, 355. [Google Scholar] [CrossRef]
- World Organisation for Animal Health (WOAH). African swine fever. Available online: https://www.woah.org/en/disease/african-swine-fever/ (accessed on 11 March 2026).
- Chandana, M.S.; Nair, S.S.; Chaturvedi, V.K.; Abhishek; Pal, S.; Charan, M.S.S.; Balaji, S.; Saini, S.; Vasavi, K.; Deepa, P. Recent progress and major gaps in the vaccine development for African swine fever. Braz. J. Microbiol. 2024, 55, 997–1010. [Google Scholar] [CrossRef]
- Borca, M.V.; Ramirez-Medina, E.; Silva, E.; Vuono, E.; Rai, A.; Pruitt, S.; Holinka, L.G.; Velazquez-Salinas, L.; Zhu, J.; Gladue, D.P. Development of a highly effective African swine fever virus vaccine by deletion of the I177L gene results in sterile immunity against the current epidemic Eurasia strain. J. Virol. 2020, 94, e02017-19. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, V.; Holinka, L.G.; Gladue, D.P.; Sanford, B.; Krug, P.W.; Lu, X.; Arzt, J.; Reese, B.; Carrillo, C.; Risatti, G.R.; et al. African swine fever virus Georgia isolate harboring deletions of MGF360 and MGF505 genes is attenuated in swine and confers protection against challenge with virulent parental virus. J. Virol. 2015, 89, 6048–6056. [Google Scholar] [CrossRef] [PubMed]
- Boikos, C.; McGovern, I.; Molrine, D.; Ortiz, J.R.; Puig-Barberà, J.; Haag, M. Review of analyses estimating relative vaccine effectiveness of cell-based quadrivalent influenza vaccine in three consecutive US influenza seasons. Vaccines 2022, 10, 896. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.A.; Kim, Y.; Lee, S.J.; Moon, S.C.; Ahn, K.S.; Zheng, X.; Kim, D.S.; Lee, S.Y.; Shin, S.P.; Tark, D.; et al. African swine fever vaccine candidate ASFV-G-ΔI177L/ΔLVR protects against homologous virulent challenge and exhibits long-term maintenance of antibodies. Animals (Basel) 2025, 15, 473. [Google Scholar] [CrossRef]
- Chen, W.; Zhao, D.; He, X.; Liu, R.; Wang, Z.; Zhang, X.; Li, F.; Shan, D.; Chen, H.; Zhang, J.; et al. A live-attenuated virus-based candidate vaccine for African swine fever. Sci. China (Life Sci.) 2020, 63, 790–801. [Google Scholar]
- Ngo, T.T.N.; Oh, T.; Do, D.T. The Prospects and Challenges of Live Attenuated Vaccines Against African Swine Fever Virus in Vietnam. Vaccines 2026, 14(3), 284. [Google Scholar] [CrossRef]
- Attreed, S.E.; Silva, C.; Abbott, S.; Ramirez-Medina, E.; Espinoza, N.; Borca, M.V.; Gladue, D.P.; Diaz-San Segundo, F. A highly effective African swine fever virus vaccine elicits a memory T cell response in vaccinated swine. Pathogens 2022, 11, 1438. [Google Scholar] [CrossRef]
- Lotonin, K.; Brito, F.; Mehinagic, K.; García-Nicolás, O.; Liniger, M.; Donzé, N.; Python, S.; Talker, S.; Ploegaert, T.C.W.; Ruggli, N.; et al. Correlates of protection against African swine fever virus identified by a systems immunology approach. eLife 2025, 107579. [Google Scholar] [CrossRef]
- Wang, Z.; Ai, Q.; Huang, S.; Ou, Y.; Gao, Y.; Tong, T.; Fan, H. Immune escape mechanism and vaccine research progress of African swine fever virus. Vaccines 2022, 10, 344. [Google Scholar] [CrossRef]
- Borca, M.V.; Rai, A.; Ramirez-Medina, E.; Silva, E.; Velazquez-Salinas, L.; Vuono, E.; Pruitt, S.; Espinoza, N.; Gladue, D.P. A cell culture-adapted vaccine virus against the current African swine fever virus pandemic strain. J. Virol. 2021, 95, e0012321. [Google Scholar] [CrossRef]
- Tran, X.H.; Le, T.T.P.; Nguyen, Q.H.; Do, T.T.; Nguyen, V.D.; Gay, C.G.; Borca, M.V.; Gladue, D.P. African swine fever virus vaccine candidate ASFV-G-ΔI177L efficiently protects European and native pig breeds against circulating Vietnamese field strain. Transbound. Emerg. Dis. 2022, 69, e497–e504. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, Y.; Choi, S.A.; Ahn, K.S.; Lee, S.Y.; Zheng, X.; Kim, D.S.; Kim, W.; Shin, Y.; Kim, S.J.; et al. Safety of live attenuated ASFV-G-ΔI177L/ΔLVR vaccination in sows with advanced pregnancies. Transbound. Emerg. Dis. 2025, 2025, 8007143. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Auer, A.; Cattoli, G.; Padungtod, P.; Lamien, C.E.; Oh, Y.; Jayme, S.; Rozstalnyy, A. Challenges in the application of African swine fever vaccines in Asia. Animals (Basel) 2024, 14, 2473. [Google Scholar] [CrossRef]
- Penrith, M.-L.; Depner, K.; Jori, F.; Dione, M.; Alders, R.; Chenais, E. Editorial: African swine fever in smallholder and traditional pig farming systems: Research, challenges and solutions. Front. Vet. Sci. 2022, 9, 878928. [Google Scholar] [CrossRef]
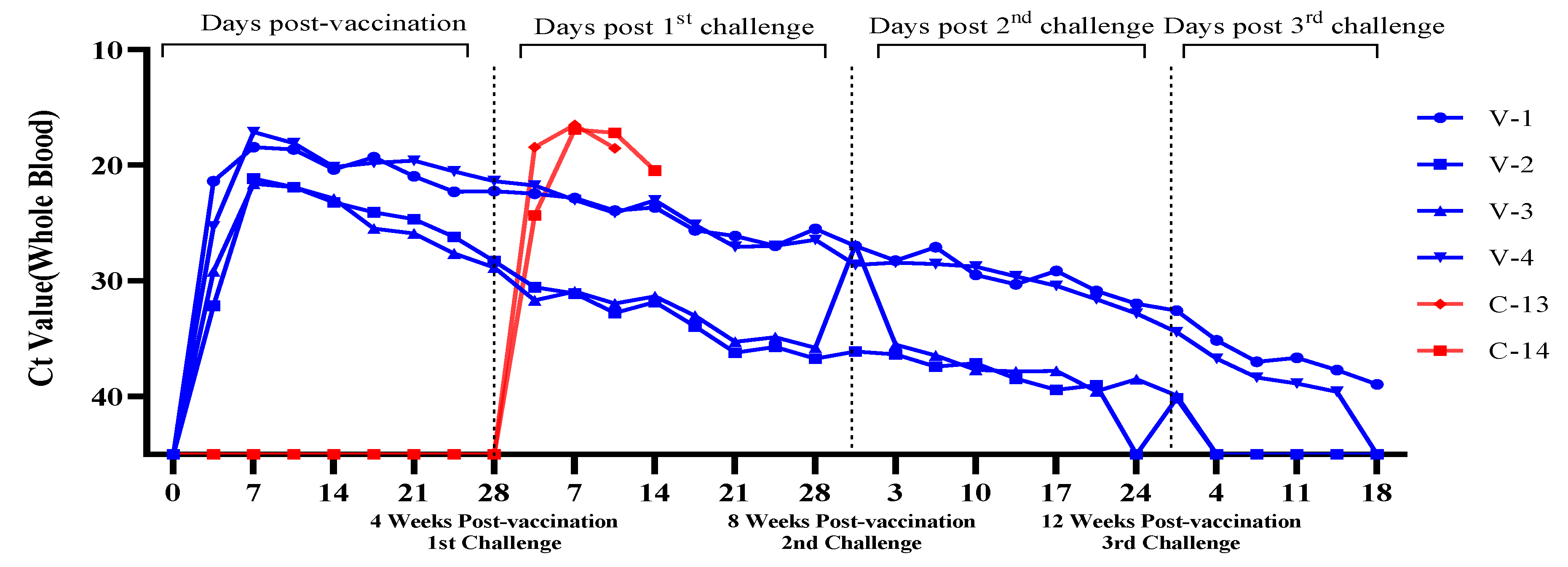
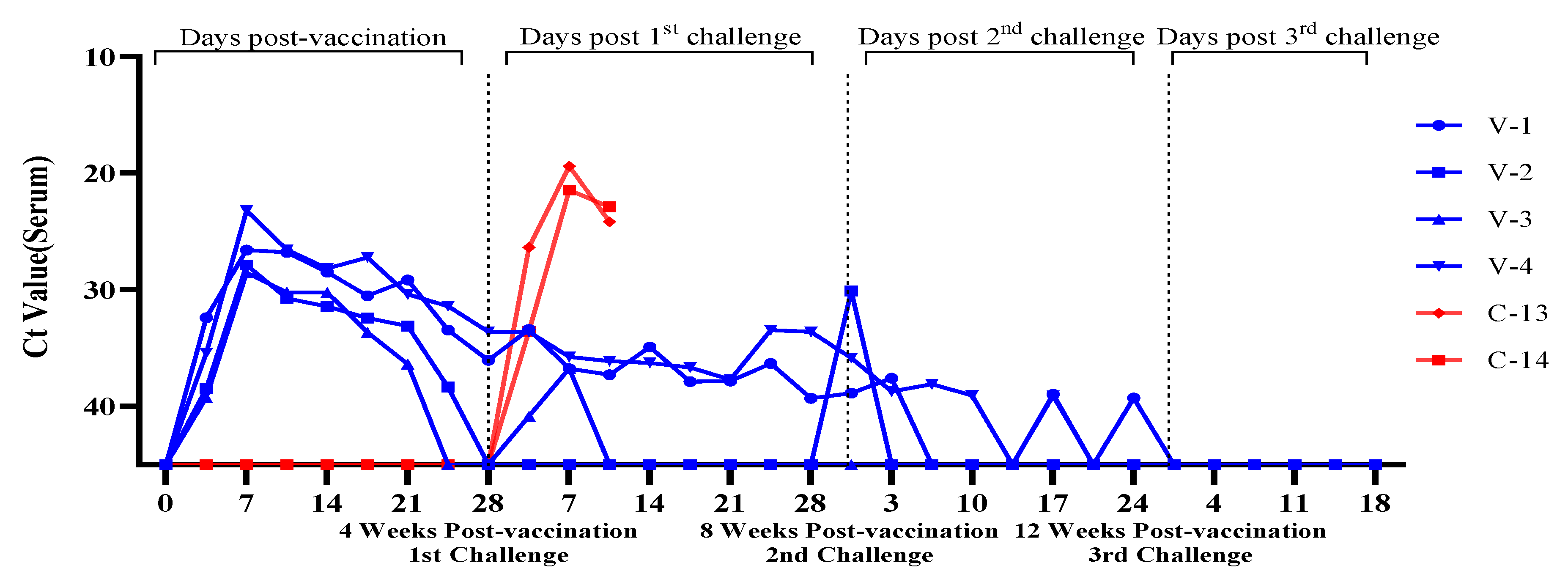
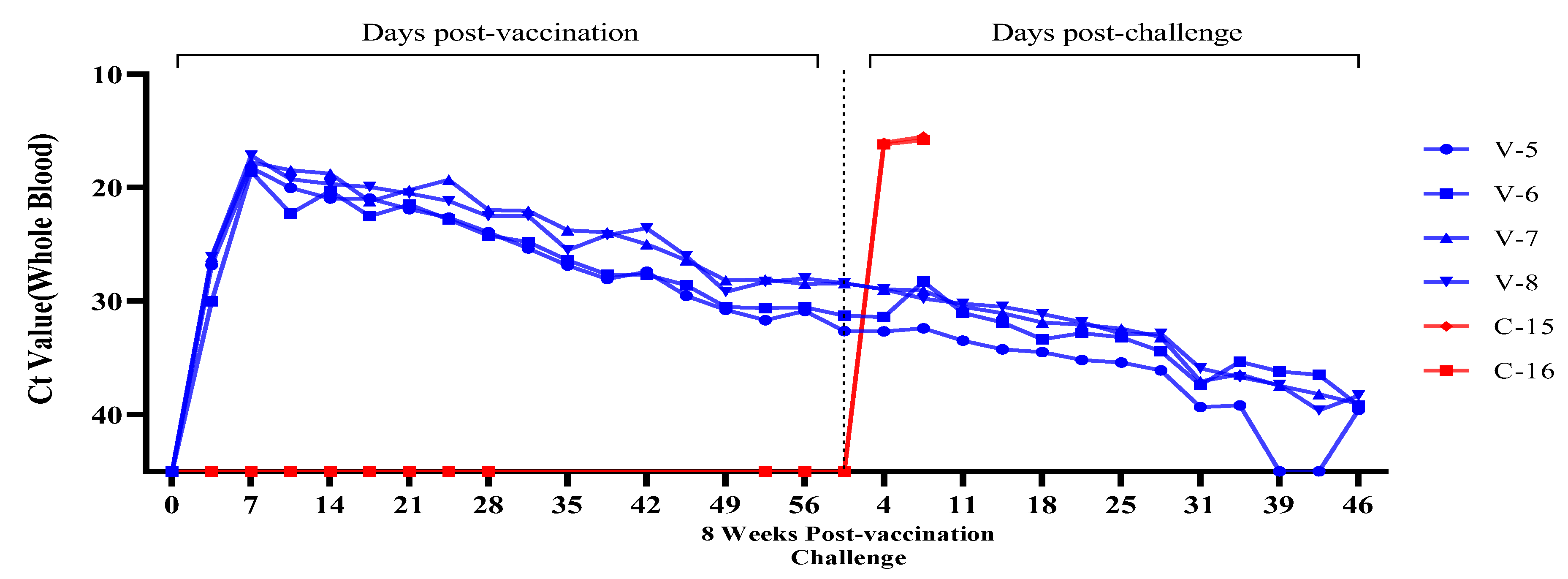
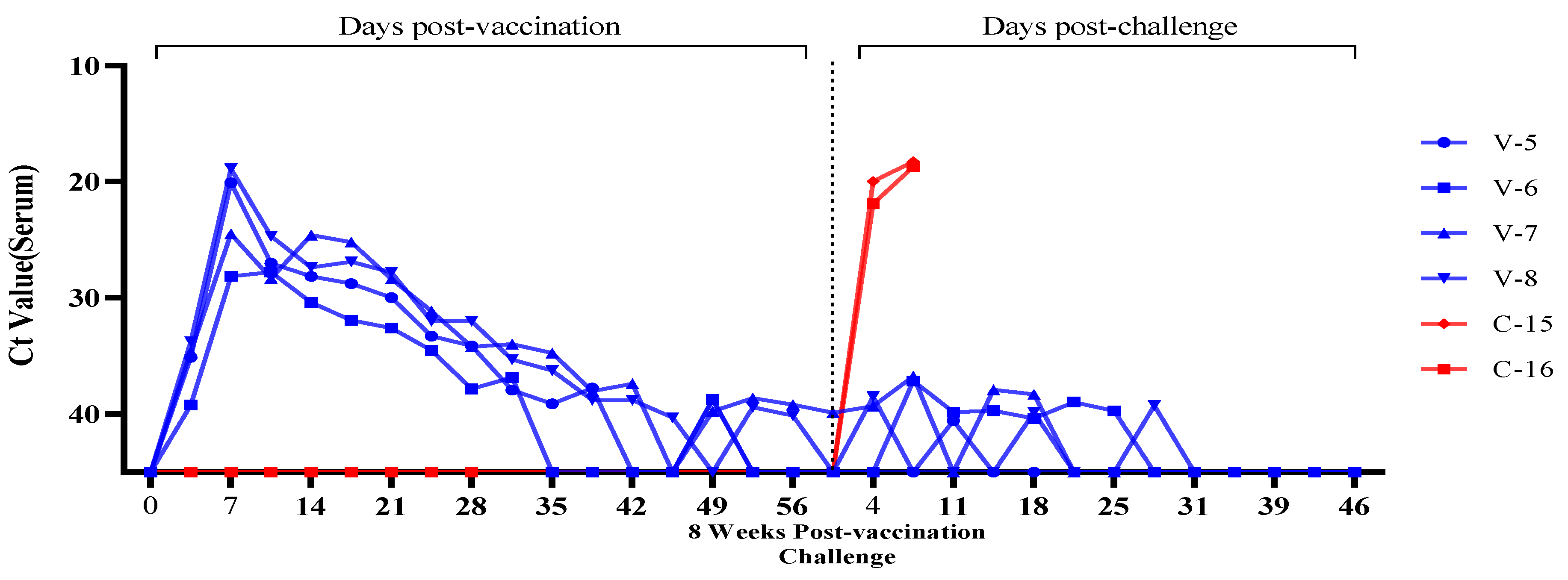
| Trial Group | Animal No. Identification | Number of Challenge & Timing | Vaccine/Dose | Challenge/Dose | Route | ||
|---|---|---|---|---|---|---|---|
| T-1 | 4 (*V1–V4) | 1st | 2nd | 3rd | ASFV-G-ΔI177L/ΔLVR /103TCID50 | ASFV-Hwacheon/2020 (G-II) /102HAD50 |
IM |
| 4WPV | 8WPV | 12WPV | |||||
| T-2 | 4 (V5–V8) | 8WPV | |||||
| T-3 | 4 (V9–V12) | 12WPV | |||||
| C-1 | 2 (§C13–C14) | T-1 Challenge control | Not applicable | ||||
| C-2 | 2 (C15–C16) | T-2 Challenge control | |||||
| C-3 | 2 (C17–C18) | T-3 Challenge control | |||||
| Group | S/N % (cELISA) | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DPV (Days Post-Vaccination) |
1st DPC (1st Days Post-Challenge) |
2nd DPC (2nd Days Post-Challenge) |
3rd DPC (3rd Days Post-Challenge) |
||||||||||||||||||||||||||||
| 0 | 3 | 7 | 10 | 14 | 21 | 24 | 28 | 3 | 7 | 10 | 14 | 17 | 21 | 24 | 28 | 31 | 4 | 7 | 11 | 14 | 18 | 21 | 25 | 28 | 4 | 7 | 11 | 14 | 18 | ||
| T1 | V-1 | 75.6 | 73.5 | 35.9 | 25.2 | 23.7 | 24.1 | 25.9 | 22.5 | 20.3 | 15.0 | 10.6 | 8.4 | 6.8 | 4.6 | 3.0 | 1.0 | 0.1 | 0.6 | 0.6 | 0.6 | 1.0 | 0.6 | 1.4 | 1.2 | 1.6 | 1.7 | 0.6 | 0.0 | 0.3 | 0.9 |
| V-2 | 82.0 | 76.9 | 40.2 | 10.3 | 14.8 | 20.6 | 22.5 | 20.3 | 21.0 | 16.4 | 12.4 | 10.1 | 9.6 | 7.0 | 5.1 | 4.1 | 3.1 | 2.2 | 0.7 | 0.7 | 0.8 | -0.5 | 0.0 | -0.5 | -0.6 | -1.0 | -1.8 | -1.7 | -1.6 | -0.9 | |
| V-3 | 85.1 | 73.6 | 51.3 | 28.0 | 26.5 | 24.9 | 27.5 | 18.6 | 13.7 | 8.5 | 4.3 | 3.1 | 2.6 | 1.5 | 1.7 | 0.6 | 1.4 | 1.0 | 0.9 | 0.9 | 0.9 | 0.7 | 1.2 | 0.5 | 1.4 | 0.6 | 0.2 | 0.2 | -0.2 | 1.1 | |
| V-4 | 76.9 | 63.3 | 47.0 | 22.4 | 20.4 | 19.6 | 22.5 | 18.1 | 11.4 | 11.6 | 11.0 | 8.7 | 7.9 | 5.9 | .8 | 4.1 | 4.0 | 2.5 | 2.2 | 2.1 | 1.5 | 1.4 | 1.9 | 1.9 | 2.3 | 1.9 | 1.9 | 1.2 | 1.4 | 3.0 | |
| C-1 | C-13 | 79.8 | 75.1 | 76.5 | 73.2 | 85.9 | 79.2 | 88.0 | 94.6 | 85.8 | 41.0 | 16.6 | D | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| C-14 | 76.9 | 81.2 | 77.5 | 73.3 | 83.7 | 70.8 | 92.1 | 87.5 | 73.2 | 29.6 | 20.7 | D | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | |
| Group | S/N % (cELISA) | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DPV (Days Post-Vaccination) | DPC (Days Post-Challenge) | |||||||||||||||||||||||||||||||
| 0 | 3 | 7 | 10 | 14 | 17 | 21 | 24 | 28 | 31 | 35 | 38 | 42 | 45 | 49 | 52 | 56 | 59 | 4 | 7 | 11 | 14 | 18 | 21 | 25 | 28 | 32 | 35 | 39 | 42 | 46 | ||
| T2 | V-5 | 83.9 | 80.7 | 55.5 | 22.0 | 24.2 | 28.6 | 24.3 | 31.5 | 27.2 | 17.1 | 14.3 | 11.0 | 9.3 | 7.9 | 5.6 | 4.3 | 2.6 | 3.3 | 1.1 | 1.3 | 1.1 | 0.0 | -0.6 | 0.0 | -0.5 | -0.7 | -0.2 | -0.7 | -1.6 | -1.8 | 1.4 |
| V-6 | 80.9 | 83.0 | 41.3 | 19.5 | 18.6 | 14.9 | 13.3 | 18.1 | 18.6 | 12.6 | 13.0 | 12.2 | 10.9 | 12.1 | 10.3 | 9.9 | 8.2 | 7.7 | 6.2 | 4.4 | 0.6 | -0.3 | -0.5 | -0.7 | -1.7 | -1.1 | 0.2 | -1.0 | -1.4 | -1.5 | -1.2 | |
| V-7 | 79.0 | 76.2 | 36.4 | 24.4 | 32.8 | 29.9 | 29.3 | 27.2 | 18.7 | 15.6 | 15.5 | 12.4 | 11.7 | 12.5 | 10.8 | 11.2 | 10.9 | 12.9 | 9.3 | 13.1 | 7.3 | 5.5 | 3.9 | 2.5 | -0.4 | 0.3 | 0.5 | -0.6 | -0.1 | -1.2 | 0.0 | |
| V-8 | 82.9 | 75.6 | 48.3 | 24.0 | 20.3 | 18.4 | 19.2 | 19.9 | 14.7 | 10.9 | 10.7 | 7.7 | 6.9 | 6.8 | 4.6 | 3.8 | 3.3 | 4.0 | 2.2 | 1.8 | 0.1 | -0.5 | -0.9 | -0.8 | -1.7 | -1.7 | -1.9 | -2.5 | -2.0 | -2.8 | -2.2 | |
| C-2 | C-15 | 82.2 | 88.0 | 85.3 | 78.4 | 78.8 | 82.3 | 83.8 | 85.0 | 89.9 | No Data | 83.9 | 89.7 | 82.8 | 89.7 | 48.6 | D | - | - | - | - | - | - | - | - | - | - | |||||
| C-16 | 77.6 | 78.3 | 79.9 | 85.3 | 75.4 | 80.2 | 79.5 | 85.5 | 95.5 | 92.5 | 91.3 | 85.6 | 81.3 | 47.8 | D | - | - | - | - | - | - | - | - | - | - | |||||||
| Group | S/N % (cELISA) | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DPV (Days Post-Vaccination) | DPC (Days Post-Challenge) |
|||||||||||||||||||||||||||||||||
| 0 | 3 | 7 | 10 | 14 | 17 | 21 | 24 | 28 | 31 | 35 | 38 | 42 | 45 | 49 | 52 | 56 | 59 | 63 | 66 | 70 | 73 | 77 | 80 | 84 | 87 | 4 | 7 | 11 | 14 | 18 | 21 | |||
| T3 | V-9 | 83.0 | 74.8 | 37.9 | 17.2 | 16.4 | 15.5 | 13.9 | 12.8 | 9.3 | 6.4 | 6.9 | 4.3 | 3.6 | 3.9 | 3.0 | 1.9 | 1.3 | 1.9 | 1.2 | 1.4 | 1.1 | 0.8 | 0.4 | 0.5 | 0.5 | 0.6 | 0.5 | -0.2 | -0.6 | -1.6 | -2.7 | -2.3 | |
| V-10 | 87.6 | 75.2 | 52.9 | 27.9 | 30.1 | 29.4 | 32.9 | 32.7 | 20.5 | 11.9 | 10.7 | 7.3 | 6.3 | 5.9 | 5.8 | 4.0 | 4.2 | 4.4 | 3.6 | 3.5 | 2.6 | 2.6 | 2.1 | 2.1 | 2.7 | 2.8 | 3.4 | 1.5 | 1.3 | 1.5 | 2.0 | 2.0 | ||
| V-11 | 81.1 | 72.8 | 74.3 | 56.2 | 41.9 | 25.9 | 23.3 | 24.6 | 22.5 | 15.7 | 11.7 | 9.3 | 7.6 | 6.3 | 5.2 | 3.5 | 2.4 | 2.5 | 1.2 | 3.3 | 0.0 | -0.2 | -0.4 | -0.5 | -0.6 | -0.5 | -0.2 | -1.5 | -1.7 | -1.7 | -1.3 | -1.7 | ||
| V-12 | 77.9 | 76.0 | 58.1 | 41.9 | 44.1 | 44.1 | 35.0 | 33.4 | 32.6 | 23.5 | 20.6 | 17.9 | 16.0 | 15.8 | 13.9 | 11.2 | 9.1 | 9.5 | 7.5 | 7.3 | 6.0 | 5.3 | 6.5 | 5.3 | 5.1 | 5.5 | 6.3 | 4.7 | 3.8 | 3.8 | 2.7 | 1.7 | ||
| C-3 | C-17 | 85.4 | 81.3 | 85.5 | 86.4 | 75.8 | 82.0 | 79.2 | 82.8 | 89.3 | No Data | 56.9 | 63.6 | 61.2 | 60.6 | 22.3 | D | - | - | - | ||||||||||||||
| C-18 | 79.3 | 84.1 | 85.5 | 79.6 | 82.7 | 90.9 | 90.4 | 89.3 | 102.3 | 81.0 | 56.5 | 83.6 | 60.9 | D | - | - | - | - | ||||||||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




