Submitted:
07 April 2026
Posted:
08 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Risk Factors for CRS-1 Development in Patients with ADHFpEF
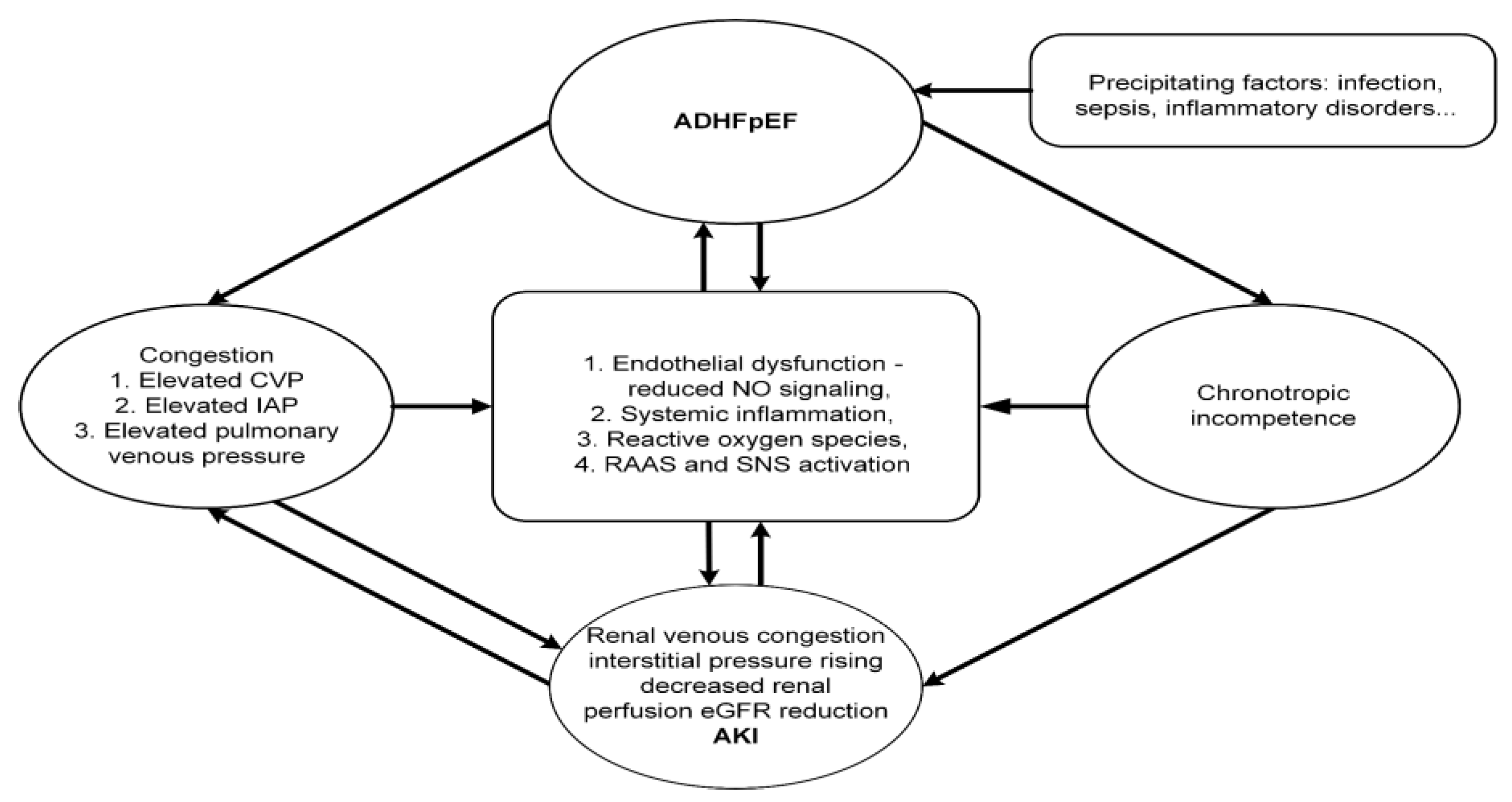
Pathophysiology
Diagnosis
Therapeutic Options
The Difference Between Transitory Worsening of Renal Function (WRF) and CRS-1, and Their Prognostic Impact
Future Directions
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bedo D, Beaudrey T, Florens N. Unraveling Chronic Cardiovascular and Kidney Disorder through the Butterfly Effect. Diagnostics (Basel). 2024 Feb 20;14(5):463. 2 iz med. istrživanja.
- Ronco C. The Cardiorenal Syndrome: Basis and Common Ground for a Multidisciplinary Patient-Oriented Therapy. Cardiorenal Med 2011;1(1):3-4.
- Ronco C, House AA, Haapio M. Cardiorenal syndrome: refining the definition of a complex symbiosis gone wrong. Intensive Care Med 2008;34(5):957-62.
- Rangaswami J, Bhalla V, Blair JEA, Chang TI, Costa S, Lentine KL, et al. Cardiorenal Syndrome: Classification, Pathophysiology, Diagnosis, and Treatment Strategies: A Scientific Statement From the American Heart Association. Circulation 2019;139(16):e840-e878.
- Zoccali C. A New Clinical Entity Bridging the Cardiovascular System and the Kidney: The Chronic Cardiovascular-Kidney Disorder. Cardiorenal Med 2025;15(1):21-8.
- Georgopoulou T, Petrakis I, Dermitzaki K, Pleros C, Drosataki E, Aletras G, et al. Cardiorenal Syndrome: Challenges in Everyday Clinical Practice and Key Points towards a Better Management. J Clin Med. 2023 Jun 18;12(12):4121.
- Buliga-Finis ON, Ouatu A, Badescu MC, Dima N, Tanase DM, Richter P, et al. Beyond the Cardiorenal Syndrome: Pathophysiological Approaches and Biomarkers for Renal and Cardiac Crosstalk. Diagnostics (Basel). 2022 Mar 22;12(4):773.
- Aletras G, Bachlitzanaki M, Stratinaki M, Lamprogiannakis E, Panagoutsos S, Kantartzi K, et la. Unraveling Acute Cardiorenal Syndrome: Predictors and Consequences in Acute Heart Failure. J Clin Med. 2025 Mar 26;14(7):2270.
- Sharma K, Hill T, Grams M, Daya NR, Hays AG, Fine D, et al. Outcomes and worsening renal function in patients hospitalized with heart failure with preserved ejection fraction. Am J Cardiol 2015;116(10):1534-40.
- Caetano F, Barra S, Faustino A, Botelho A, Mota P, Costa M, Leitão Marques A. Cardiorenal syndrome in acute heart failure: a vicious cycle? Rev Port Cardiol 2014;33(3):139-46.
- Nijst P, Mullens W. The acute cardiorenal syndrome: burden and mechanisms of disease. Curr Heart Fail Rep 2014;11(4):453-62.
- McCallum W, Sarnak MJ. Cardiorenal Syndrome in the Hospital. Clin J Am Soc Nephrol 2023;18(7):933-45.
- Ahmed A, Rich MW, Sanders PW, Perry GJ, Bakris GL, Zile MR, et al. Chronic kidney disease associated mortality in diastolic versus systolic heart failure: a propensity matched study. Am J Cardiol 2007;99(3):393-8.
- Gottlieb SS, Abraham W, Butler J, Forman DE, Loh E, Massie BM, et al. The prognostic importance of different definitions of worsening renal function in congestive heart failure. J Card Fail 2002;8(3):136-41.
- Newsome BB, Warnock DG, McClellan WM, Herzog CA, Kiefe CI, Eggers PW, et al. Long-term risk of mortality and end-stage renal disease among the elderly after small increases in serum creatinine level during hospitalization for acute myocardial infarction. Arch Intern Med 2008;168(6):609-16.
- Uduman J. Epidemiology of Cardiorenal Syndrome. Adv Chronic Kidney Dis 2018;25(5):391-399.
- Grilo RM, Falcão LM. Heart Failure With Preserved Ejection Fraction and Chronic Kidney Disease: From Pathophysiology to Treatment. Am J Cardiol 2026;258:287-301.
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 2022;24(1):4-131.
- Roh J, Hill JA, Singh A, Valero-Muñoz M, Sam F. Heart Failure With Preserved Ejection Fraction: Heterogeneous Syndrome, Diverse Preclinical Models. Circ Res. 2022;130(12):1906-25.
- Agrawal A, Naranjo M, Kanjanahattakij N, Rangaswami J, Gupta S. Cardiorenal syndrome in heart failure with preserved ejection fraction-an under-recognized clinical entity. Heart Fail Rev 2019;24(4):421-37.
- Ananthram MG, Gottlieb SS. Renal Dysfunction and Heart Failure with Preserved Ejection Fraction. Heart Fail Clin 2021;17(3):357-67.
- Tuttle ML, Fang JC, Sarnak MJ, McCallum W. Epidemiology and Management of Patients With Kidney Disease and Heart Failure With Preserved Ejection Fraction. Semin Nephrol. 2024 Mar;44(2):151516.
- Kajimoto K, Sato N, Keida T, Sakata Y, Takano T; Acute Decompensated Heart Failure Syndromes (ATTEND) Investigators. Associations of anemia and renal dysfunction with outcomes among patients hospitalized for acute decompensated heart failure with preserved or reduced ejection fraction. Clin J Am Soc Nephrol 2014;9(11):1912-21.
- Masuda T, Kariro K, Morishita Y. Why are SGLT2 inhibitors effective in HFpEF? Implications for interstitial fluid retention and cardio-renal interaction. Hypertens Res. 2026 Jan 6. doi: 10.1038/s41440-025-02516-1. Epub ahead of print.
- Núñez-Marín G, Santas E. Cardiorenal Disease and Heart Failure with Preserved Ejection Fraction: Two Sides of the Same Coin. Cardiorenal Med 2025;15(1):108-21.
- Gori M, Senni M, Gupta DK, Charytan DM, Kraigher-Krainer E, Pieske B, et al. Association between renal function and cardiovascular structure and function in heart failure with preserved ejection fraction. Eur Heart J 2014;35(48):3442-51.
- Khandait H, Sodhi SS, Khandekar N, Bhattad VB. Cardiorenal Syndrome in Heart Failure with Preserved Ejection Fraction: Insights into Pathophysiology and Recent Advances. Cardiorenal Med 2025;15(1):41-60.
- Lazzeri C, Valente S, Tarquini R, Gensini GF. Cardiorenal syndrome caused by heart failure with preserved ejection fraction. Int J Nephrol. 2011 Feb 7;2011:634903.
- Ronco C, Cicoira M, McCullough PA. Cardiorenal syndrome type 1: pathophysiological crosstalk leading to combined heart and kidney dysfunction in the setting of acutely decompensated heart failure. J Am Coll Cardiol 2012;60(12):1031-42.
- Hunley TE, Ma LJ, Kon V. Scope and mechanisms of obesity-related renal disease. Curr Opin Nephrol Hypertens 2010;19(3):227-34.
- Ferris M, Hogan SL, Chin H, Shoham DA, Gipson DS, Gibson K, et al. Obesity, albuminuria, and urinalysis findings in US young adults from the Add Health Wave III study. Clin J Am Soc Nephrol 2007;2(6):1207-14.
- Aune D, Sen A, Norat T, Janszky I, Romundstad P, Tonstad S, et al. Body Mass Index, Abdominal Fatness, and Heart Failure Incidence and Mortality: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Circulation 2016;133(7):639-49.
- Fu K, Hu Y, Zhang H, Wang C, Lin Z, Lu H, et al. Insights of Worsening Renal Function in Type 1 Cardiorenal Syndrome: From the Pathogenesis, Biomarkers to Treatment. Front Cardiovasc Med. 2021 Dec 14;8:760152.
- Méndez AB, Azancot MA, Olivella A, Soler MJ. New aspects in cardiorenal syndrome and HFpEF. Clin Kidney J. 2022 May 6;15(10):1807-15.
- Binanay C, Califf RM, Hasselblad V, O’Connor CM, Shah MR, Sopko G, et al. Evaluation study of congestive heart failure and pulmonary artery catheterization effectiveness: the ESCAPE trial. JAMA 2005;294(13):1625-33.
- Damman K, Masson S, Hillege HL, Voors AA, van Veldhuisen DJ, Rossignol P, et al. Tubular damage and worsening renal function in chronic heart failure. JACC Heart Fail 2013;1(5):417-24.
- Sowers JR, Whaley-Connell A, Hayden MR. The Role of Overweight and Obesity in the Cardiorenal Syndrome. Cardiorenal Med 2011;1(1):5-12.
- Ahmad T, Jackson K, Rao VS, Tang WHW, Brisco-Bacik MA, Chen HH, et al. Worsening Renal Function in Patients With Acute Heart Failure Undergoing Aggressive Diuresis Is Not Associated With Tubular Injury. Circulation 2018;137(19):2016-28.
- Miller WL, Mullan BP. Volume Overload Profiles in Patients With Preserved and Reduced Ejection Fraction Chronic Heart Failure: Are There Differences? A Pilot Study. JACC Heart Fail 2016;4(6):453-9.
- Takei M, Kohsaka S, Shiraishi Y, Goda A, Izumi Y, Yagawa M, et al. Effect of estimated plasma volume reduction on renal function for acute heart failure differs between patients with preserved and reduced ejection fraction. Circ Heart Fail 2015;8(3):527-32.
- Mullens W, Damman K, Harjola VP, Mebazaa A, Brunner-La Rocca HP, Martens P, et al. The use of diuretics in heart failure with congestion - a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail 2019;21(2):137-155.
- Aletras G, Bachlitzanaki M, Stratinaki M, Foukarakis E, Petrakis I, Pantazis Y, et al. Diuretic resistance in cardiorenal syndrome: mechanisms, monitoring and phenotype-tailored management. Front Cardiovasc Med. 2026 Jan 5;12:1731305.
- Felker GM, Ellison DH, Mullens W, Cox ZL, Testani JM. Diuretic Therapy for Patients With Heart Failure: JACC State-of-the-Art Review. J Am Coll Cardiol 2020;75(10):1178-95.
- Al-Shammari AS, Hamzah KA, Masanabadi M, Asif M, Budhiraja M, Shafi N, et al. Urinary Sodium-Guided Diuretic Therapy in Acute Decompensated Heart Failure: A Grade Assessed Systematic Review and Meta-Analysis with Trial Sequential Analysis. Curr Probl Cardiol. 2026 Jan 20:103272.
- Prata AA, Prata AA, Coan ACC, Scardini PG, Fernandes JM, Neto WF, et al. Protocolized natriuresis-guided diuretic therapy in acute heart failure: a systematic review and meta-analysis. Crit Care. 2025 Nov 5;29(1):474.
- Riccardi M, Pagnesi M, Lombardi CM, Metra M. Severe acute kidney injury in the intensive care unit: step-to-step management. Eur Heart J Acute Cardiovasc Care 2025;14(10):618-30.
- Ter Maaten JM, Beldhuis IE, van der Meer P, Krikken JA, Postmus D, Coster JE, et al. Natriuresis-guided diuretic therapy in acute heart failure: a pragmatic randomized trial. Nat Med 2023;29(10):2625-32.
- Georgopoulou T, Stratinaki M, Petrakis I, Aletras G, Foukarakis E, Marketou M, et al. Chloride dynamics in Heart Failure: The Clinical Implications of Serum Levels and the Emerging Significance of Urinary Chloride. Journal of CardioRenal Medicine. 2025; 1(1):6.
- Zaheen M, Ferdous F, Amarasekera AT, Petutschnigg J, Edelmann F, Tan TC. Mineralocorticoid Receptor Antagonists in Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis. J Clin Med. 2025 May 21;14(10):3598. doi: 10.3390/jcm14103598.
- Sabina M, Trube J, Shah S, Lurie A, Grimm M, Bizanti A. Finerenone: A Third-Generation MRA and Its Impact on Cardiovascular Health-Insights from Randomized Controlled Trials. J Clin Med. 2024 Oct 25;13(21):6398. doi: 10.3390/jcm13216398.
- McCallum W, Testani JM. Updates in Cardiorenal Syndrome Med Clin North Am 2023;107(4):763-80.
- Hida Y, Imamura T, Kinugawa K. Impact of Finerenone in Patients with Heart Failure and Reduced/Mildly Reduced Ejection Fraction, Diabetes Mellitus, and Chronic Kidney Disease. J Clin Med. 2025 Nov 11;14(22):7997. doi: 10.3390/jcm14227997.
- Felker GM, Lee KL, Bull DA, Redfield MM, Stevenson LW, Goldsmith SR, et al. NHLBI Heart Failure Clinical Research Network. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med 2011;364(9):797-805.
- Costanzo MR, Guglin ME, Saltzberg MT, Jessup ML, Bart BA, Teerlink JR, et al. Ultrafiltration versus intravenous diuretics for patients hospitalized for acute decompensated heart failure. J Am Coll Cardiol 2007;49(6):675-83.
- Verbrugge FH, Grieten L, Mullens W. New insights into combinational drug therapy to manage congestion in heart failure. Curr Heart Fail Rep 2014;11(1):1-9.
- Mullens W, Abrahams Z, Skouri HN, Francis GS, Taylor DO, Starling RC, Paganini E, Tang WH. Elevated intra-abdominal pressure in acute decompensated heart failure: a potential contributor to worsening renal function? J Am Coll Cardiol 2008;51(3):300-6.
- Mullens W, Abrahams Z, Francis GS, Taylor DO, Starling RC, Tang WH. Prompt reduction in intra-abdominal pressure following large-volume mechanical fluid removal improves renal insufficiency in refractory decompensated heart failure. J Card Fail 2008;14(6):508-14.
- Tran P, Khweir L, Kuehl M, Joshi M, Appunu K, Ayub W, et al. A Pragmatic Approach to Acute Cardiorenal Syndrome: Diagnostic Strategies and Targeted Therapies to Overcome Diuretic Resistance. J Clin Med. 2025 Apr 26;14(9):2996. doi: 10.3390/jcm14092996.
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 2024 ;26(1):5-17.
- Cannatà A, Anastasia G, De Marzo V, Caspi O, Bromage D, Porto I, et al. Diuretic strategies in acute heart failure: a systematic review and network meta-analysis of randomized clinical trials. Eur Heart J Cardiovasc Pharmacother 2026;12(1):6-14.
- Kapelios CJ, Vazir A, Lund LH, Filippatos G, Fang JC. Pharmacological options to relieve congestion in acute heart failure. Heart Fail Rev 2025;30(6):1275-90.
- Cox ZL, Collins SP, Hernandez GA, McRae AT 3rd, Davidson BT, Adams K, et al. Efficacy and Safety of Dapagliflozin in Patients With Acute Heart Failure. J Am Coll Cardiol 2024;83(14):1295-306.
- Neuen BL, Young T, Heerspink HJL, Neal B, Perkovic V, Billot L, et al. SGLT2 inhibitors for the prevention of kidney failure in patients with type 2 diabetes: a systematic review and meta-analysis. Lancet Diabetes Endocrinol 2019;7(11):845-854.
- Beldhuis IE, Streng KW, Ter Maaten JM, Voors AA, van der Meer P, Rossignol P, et al. Renin-Angiotensin System Inhibition, Worsening Renal Function, and Outcome in Heart Failure Patients With Reduced and Preserved Ejection Fraction: A Meta-Analysis of Published Study Data. Circ Heart Fail. 2017 Feb;10(2):e003588.
- Solomon SD, Rizkala AR, Gong J, Wang W, Anand IS, Ge J, et al. Angiotensin Receptor Neprilysin Inhibition in Heart Failure With Preserved Ejection Fraction: Rationale and Design of the PARAGON-HF Trial. JACC Heart Fail. 2017 Jul;5(7):471-82.
- Al-Jarallah M, Rajan R, Al-Zakwani I, Dashti R, Bulbanat B, Sulaiman K, et al. Incidence and impact of cardiorenal anaemia syndrome on all-cause mortality in acute heart failure patients stratified by left ventricular ejection fraction in the Middle East. ESC Heart Fail 2019;6(1):103-10.
- Forman DE, Butler J, Wang Y, Abraham WT, O’Connor CM, Gottlieb SS, et al. Incidence, predictors at admission, and impact of worsening renal function among patients hospitalized with heart failure. J Am Coll Cardiol 2004;43(1):61-7.
- Owan TE, Hodge DO, Herges RM, Jacobsen SJ, Roger VL, Redfield MM. Secular trends in renal dysfunction and outcomes in hospitalized heart failure patients. J Card Fail 2006;12(4):257-62.
- Lala A, Coca S, Feinman J, Hamo CE, Fiuzat M, Abraham WT, et al. Standardized Definitions of Changes in Kidney Function in Trials of Heart Failure: JACC Expert Panel From the HF-ARC. J Am Coll Cardiol 2025;85(7):766-81.
- Kenneally LF, Lorenzo M, Romero-González G, Cobo M, Núñez G, Górriz JL, et al. Kidney function changes in acute heart failure: a practical approach to interpretation and management. Clin Kidney J 2023;16(10):1587-99.
- Mitsas AC, Elzawawi M, Mavrogeni S, Boekels M, Khan A, Eldawy M, et al. Heart Failure and Cardiorenal Syndrome: A Narrative Review on Pathophysiology, Diagnostic and Therapeutic Regimens-From a Cardiologist’s View. J Clin Med. 2022 Nov 28;11(23):7041. doi: 10.3390/jcm11237041.
- Filippatos G, Teerlink JR, Farmakis D, Cotter G, Davison BA, Felker GM, et al. Serelaxin in acute heart failure patients with preserved left ventricular ejection fraction: results from the RELAX-AHF trial. Eur Heart J 2014 ;35(16):1041-50.
- Metra M, Teerlink JR, Cotter G, Davison BA, Felker GM, Filippatos G, et al. Effects of Serelaxin in Patients with Acute Heart Failure. N Engl J Med 2019 22;381(8):716-26.
- Voors AA, Metra M, Postmus D, Greenberg BH, Cotter G, Davison BA, et al. End-organ protective effect of serelaxin in patients hospitalized for heart failure: Results of the biomarker substudy of Relaxin in Acute Heart Failure-2 (RELAX-AHF-2). Eur J Heart Fail 2025;27(7):1215-23.
- Nassiri S, Voordes GHD, van Heerebeek L, van Raalte DH, Verhaar MC, Touw DJ, et al. Effects of geranylgeranylacetone on diastolic and microvascular function in patients with heart failure with a preserved ejection fraction: A phase 2, randomized, placebo-controlled, crossover trial. Eur J Heart Fail 2025 ;27(12):3304-11.
- Chiuariu T, Șalaru D, Ureche C, Vasiliu L, Lupu A, Lupu VV, et al. Cardiac and Renal Fibrosis, the Silent Killer in the Cardiovascular Continuum: An Up-to-Date. J Cardiovasc Dev Dis. 2024 Feb 16;11(2):62. doi: 10.3390/jcdd11020062.
- Nathan S, Basir MB. Emerging Device Therapies for Cardiorenal Syndrome. J Soc Cardiovasc Angiogr Interv 2023 Dec 4;2(6Part B):101210.
- Adamson PB, Abraham WT, Aaron M, Aranda JM Jr, Bourge RC, Smith A, et al. CHAMPION trial rationale and design: the long-term safety and clinical efficacy of a wireless pulmonary artery pressure monitoring system. J Card Fail 2011;17(1):3-10.
- Shah SJ, Borlaug BA, Chung ES, Cutlip DE, Debonnaire P, Fail PS, et al. Atrial shunt device for heart failure with preserved and mildly reduced ejection fraction (REDUCE LAP-HF II): a randomised, multicentre, blinded, sham-controlled trial. Lancet. 2022 Mar 19;399(10330):1130-1140.
| CRS type | Name | Etiology/primarily failing organ |
|---|---|---|
| 1 | Acute cardiorenal syndrome | Acute decompensated heart failure (ADHF)/cardiogenic shock (different causes), including progression of heart failure; acute myocardial infarction (AMI); acute myocarditis, etc. |
| 2 | Chronic cardiorenal syndrome | Chronic heart failure (CHF) resulting in chronic kidney disease (CKD) |
| 3 | Acute renocardiac syndrome | Acute kidney injury (AKI) (different causes) resulting in HF |
| 4 | Chronic renocardiac syndrome | Chronic kidney disease (CKD) resulting in CKD-associated cardiomyopathy and HF |
| 5 | Secondary cardiorenal syndrome | Systemic condition/disease resulting in simultaneous kidney and heart dysfunction/failure (e.g., amyloidosis, sepsis, cirrhosis) |
| Therapy | Comments |
|---|---|
| Loop diuretics | - The mainstay of therapy for decongestion - Can be used alone or in combination with other diuretics as a part of sequential nephron blockade - Can cause enhanced RAAS activation and WRF/AKI |
| Mineralocorticosteroid receptor antagonists (MRAs) | - Successful in combination with loop diuretics, as a part of sequential nephron blockade - May have positive effects on the heart and kidneys in chronic settings - Caution: hyperkalemia |
| SGLT2 inhibitors | - In the acute setting: may enhance diuresis when combined with loop diuretics - In the chronic setting: disease-modifying agents that improve prognosis in patients with HFpEF - Initial WRF is transitory; it is followed by kidney function recovery and has no adverse prognostic impact. |
| ACEi inhibitors/ARB/ARNI | - No prognostic impact in patients with HFpEF - Used for treatment of concomitant conditions HTN, DM, etc. - Cautions: vasodilation and reduced preload may further cause a decline in kidney function. |
| Renal replacement therapy (RRT) | - If all previous measures do not lead to effective decongestion; mandatory in patients with volume overload, oliguria, electrolyte and/or metabolic acid-base disbalance |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




