Submitted:
03 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
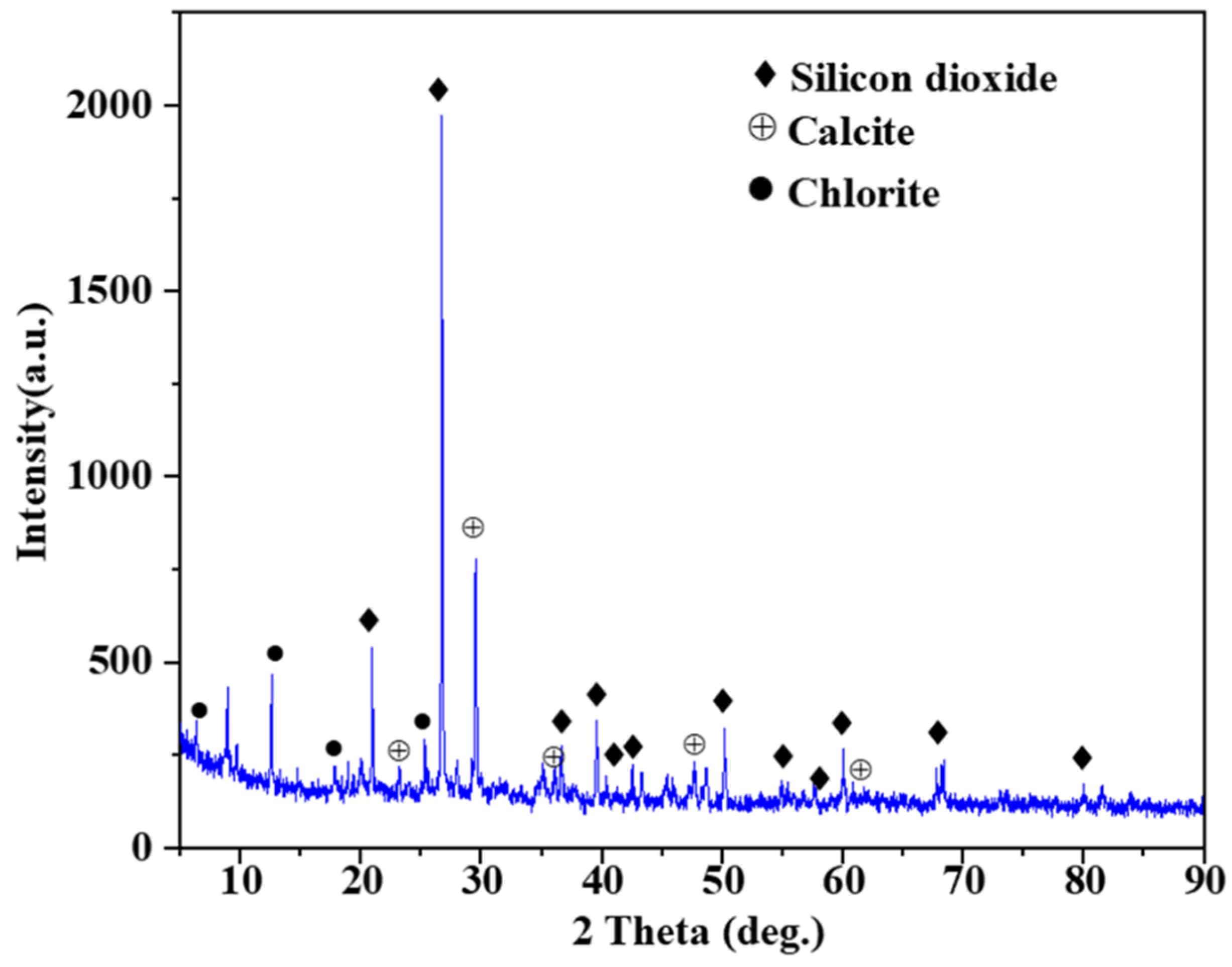
2.1. Raw Materials

2.2. Methods
2.2.1. Mix Proportion Design, Molding, and Curing of Stabilized Soil

2.2.2. Unconfined Compressive Strength (UCS) Test
2.2.3. Water Stability Test
2.2.4. Freeze-Thaw Cycle Test
2.2.5. Material Analytical Methods
3. Results and Discuss
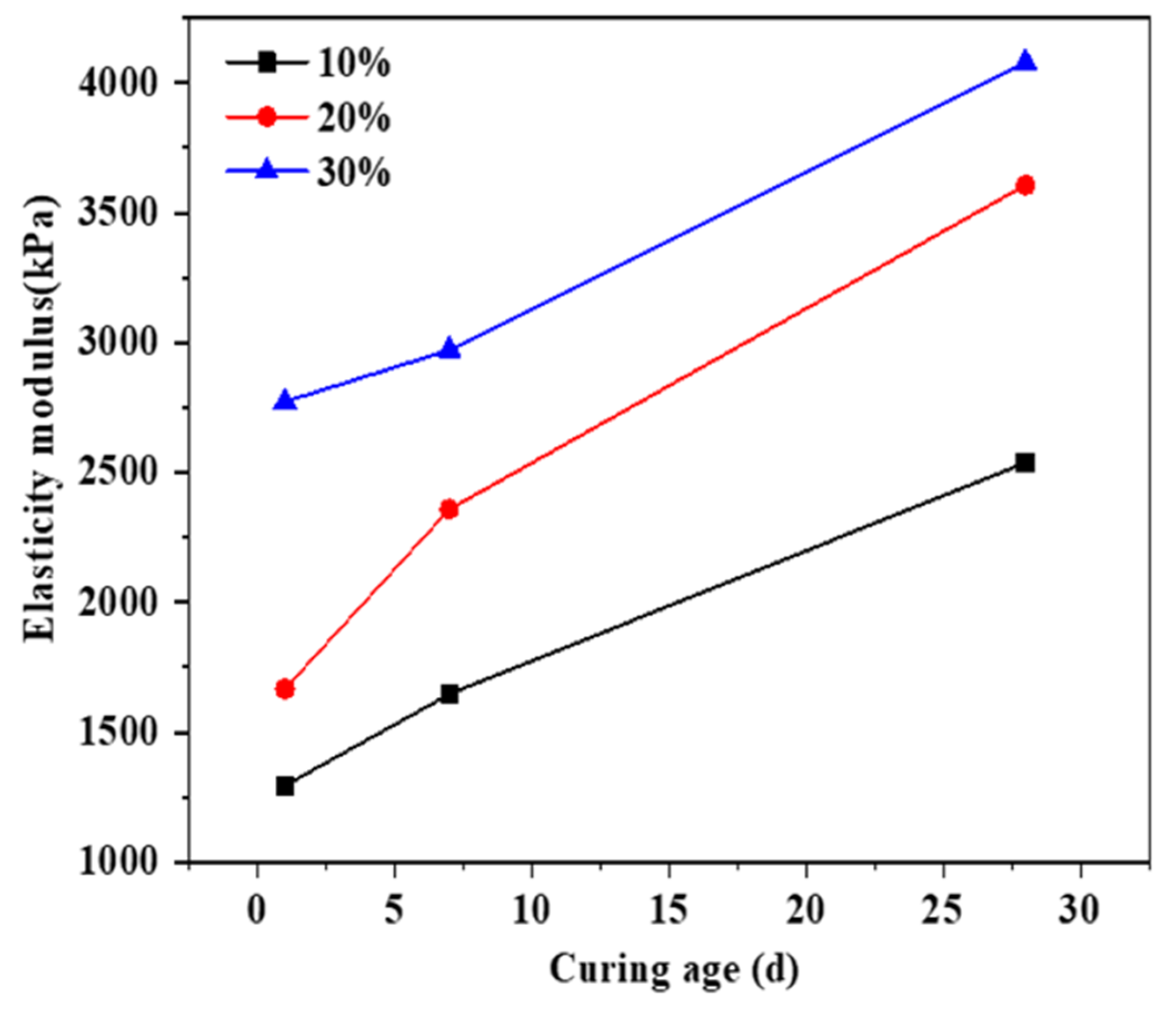
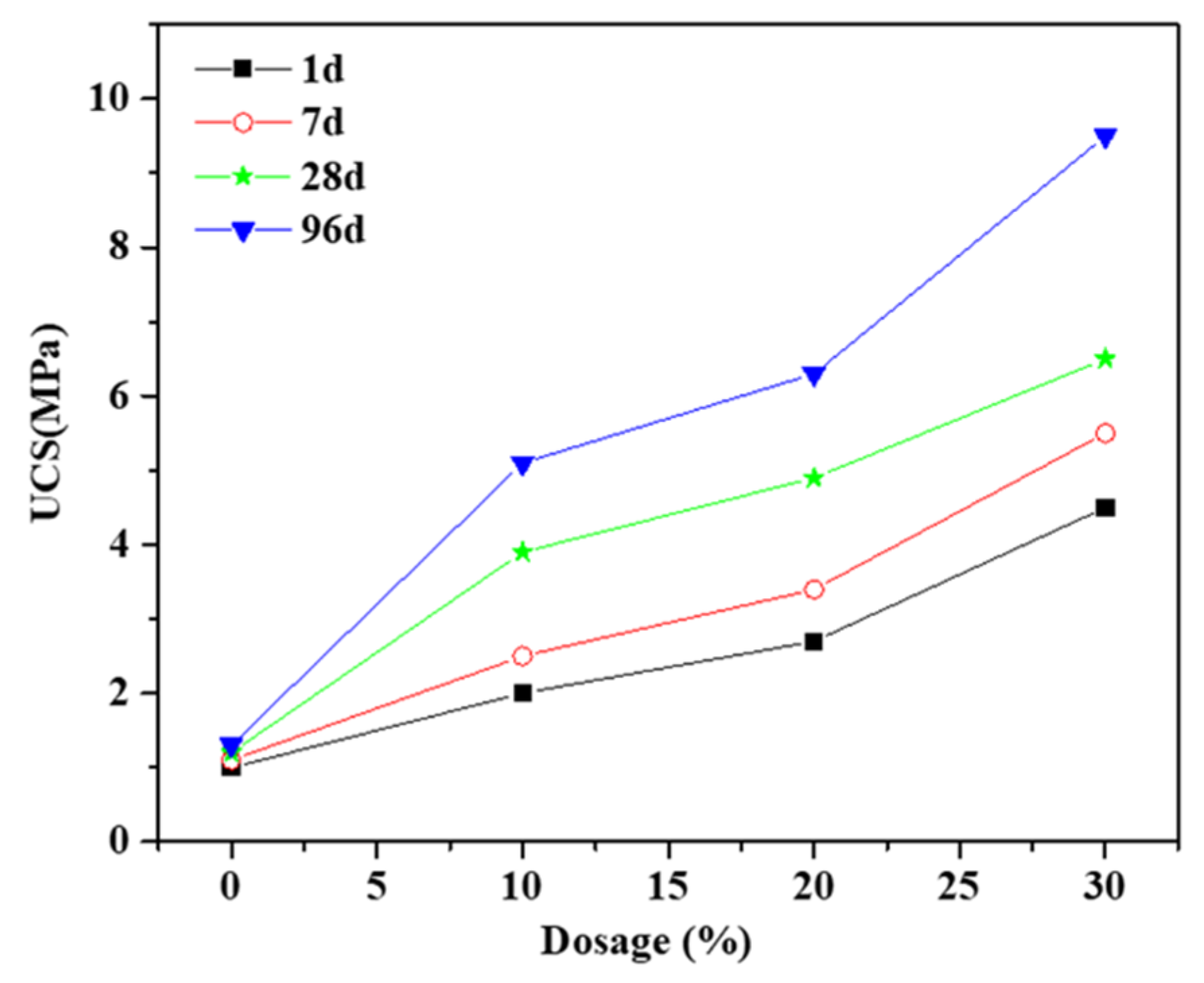
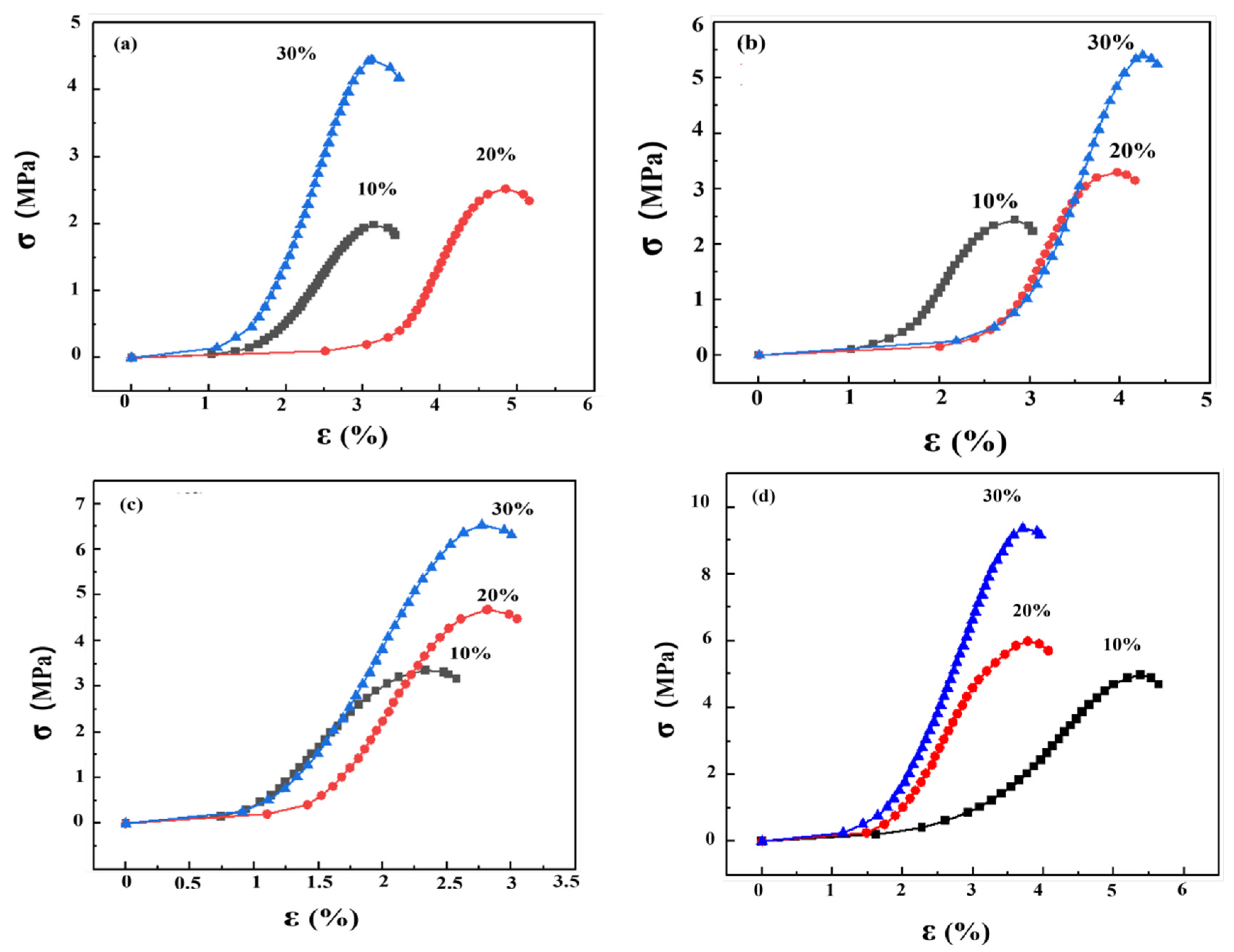
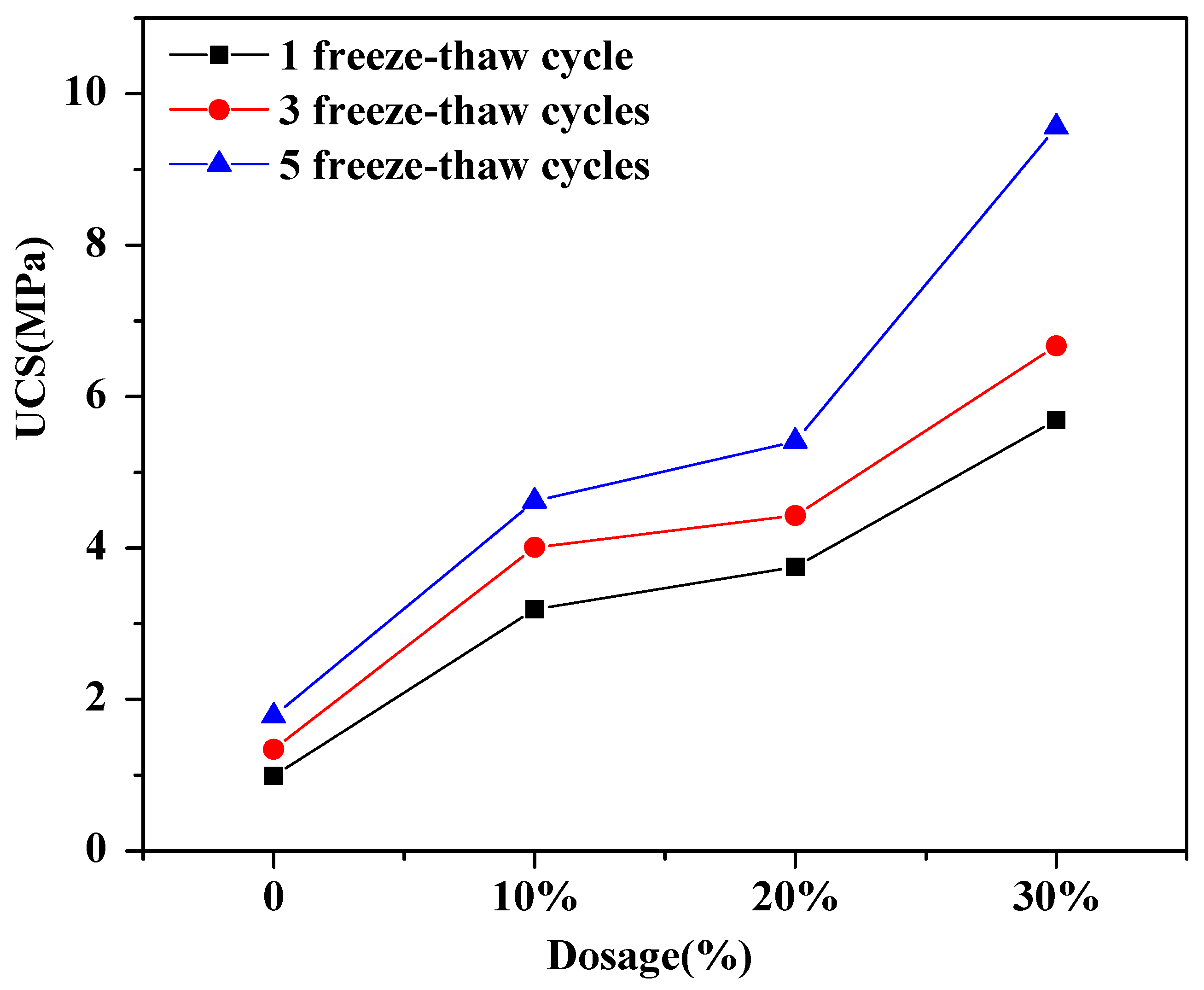
3.1. The Effect of Soil Stabilizer on the Mechanical Properties of Stabilized Gravelly Soil
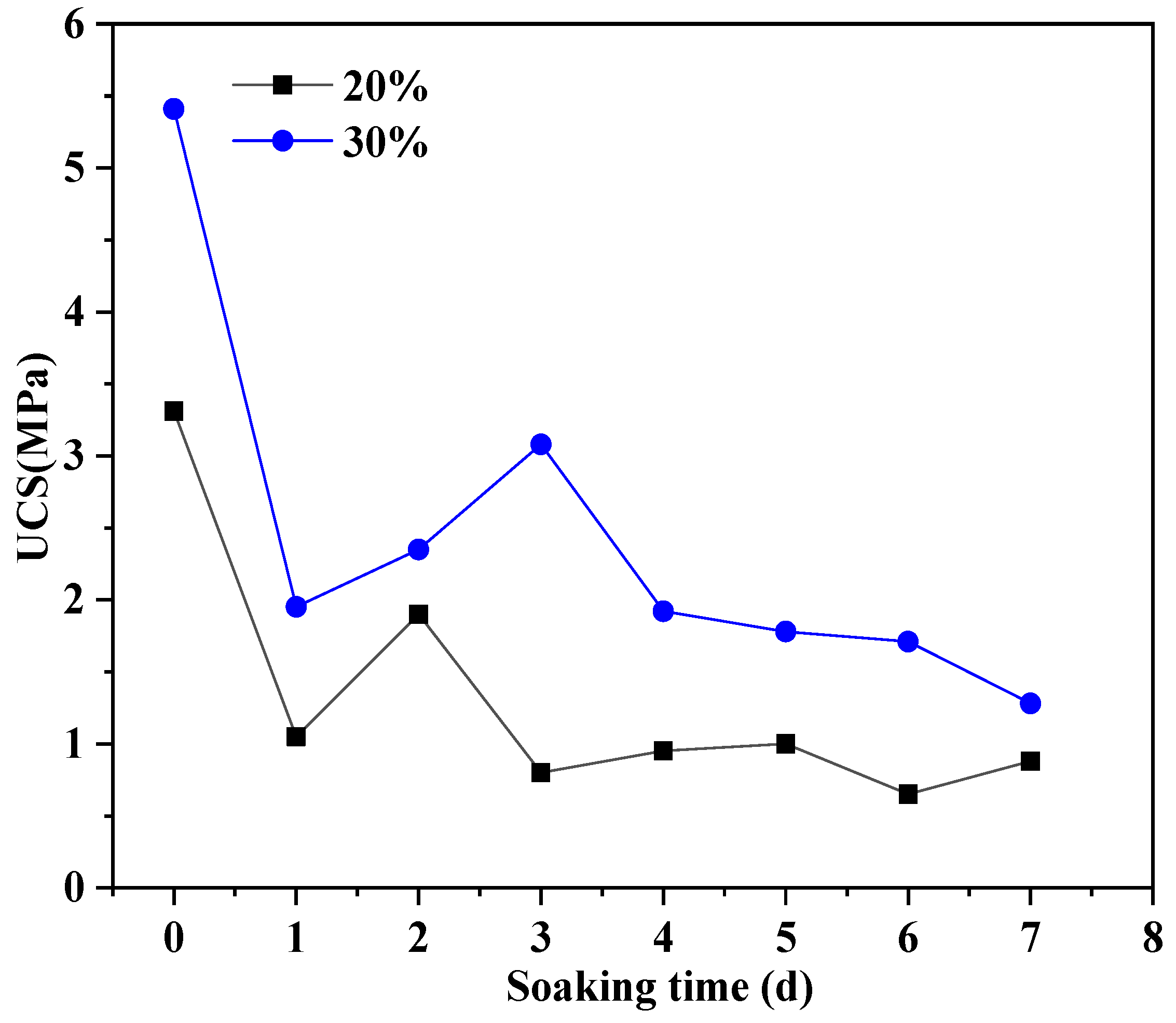
3.2. The Effect of Soil Stabilizer on the Water Stability of Stabilized Gravelly Soil
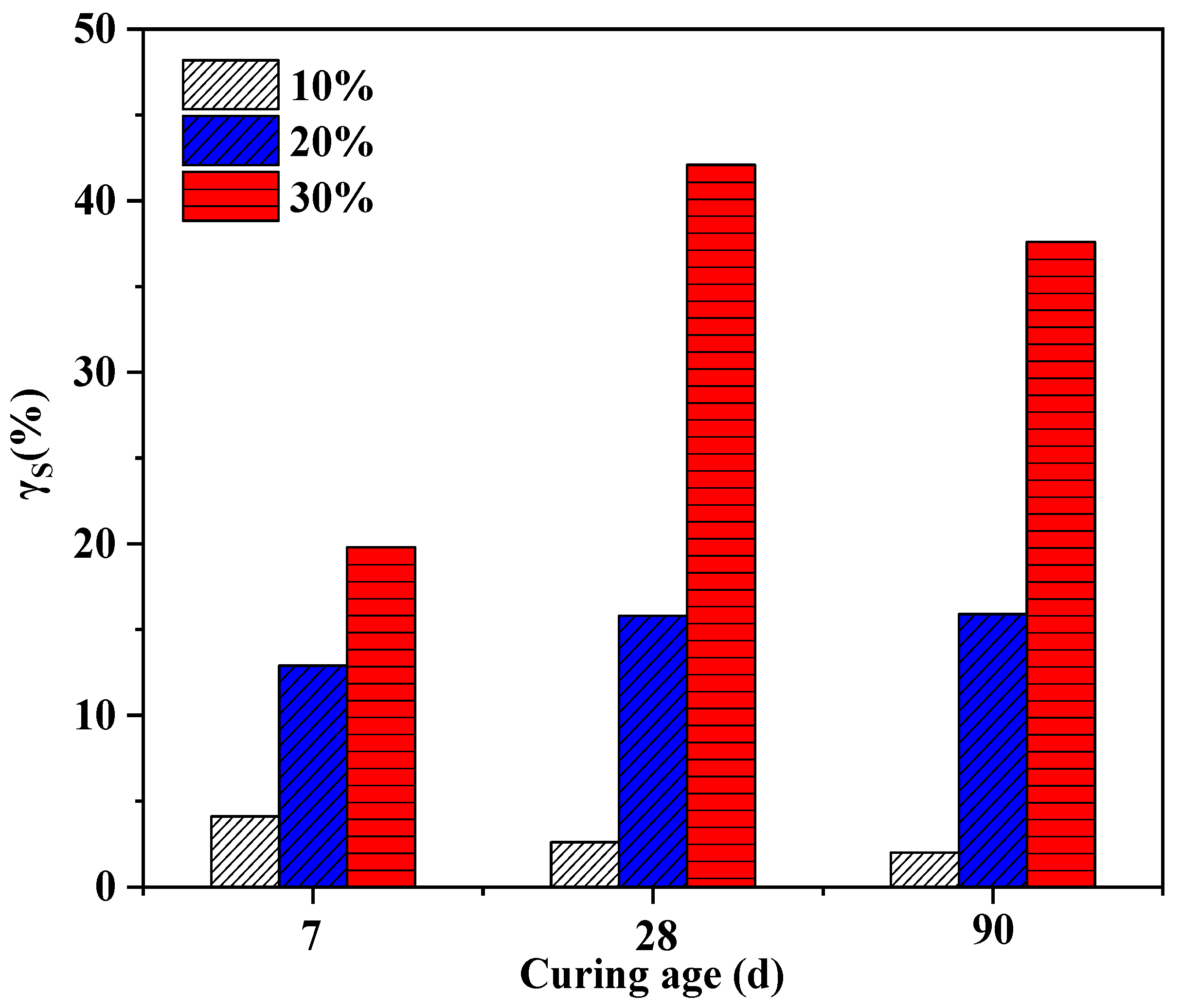
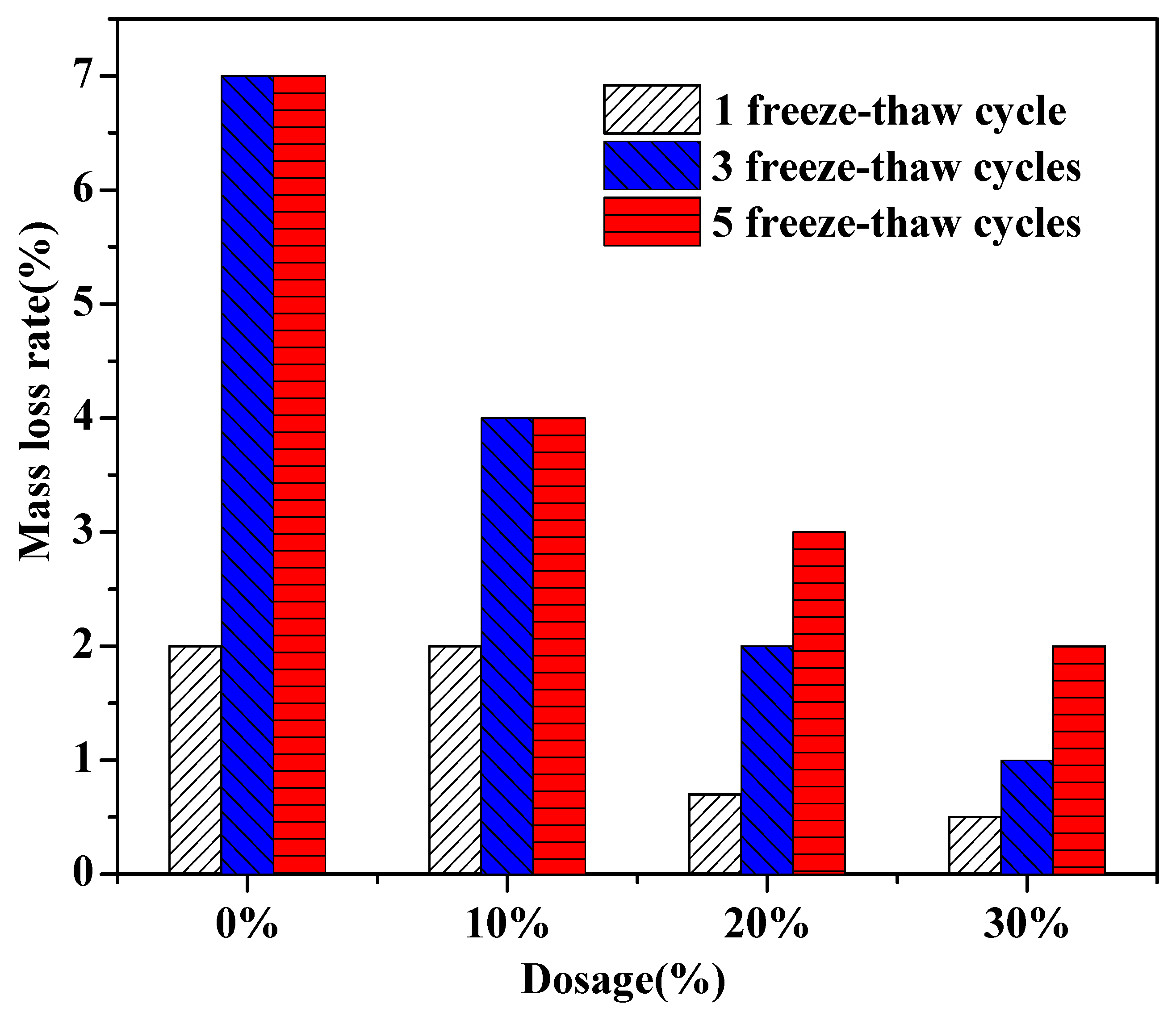
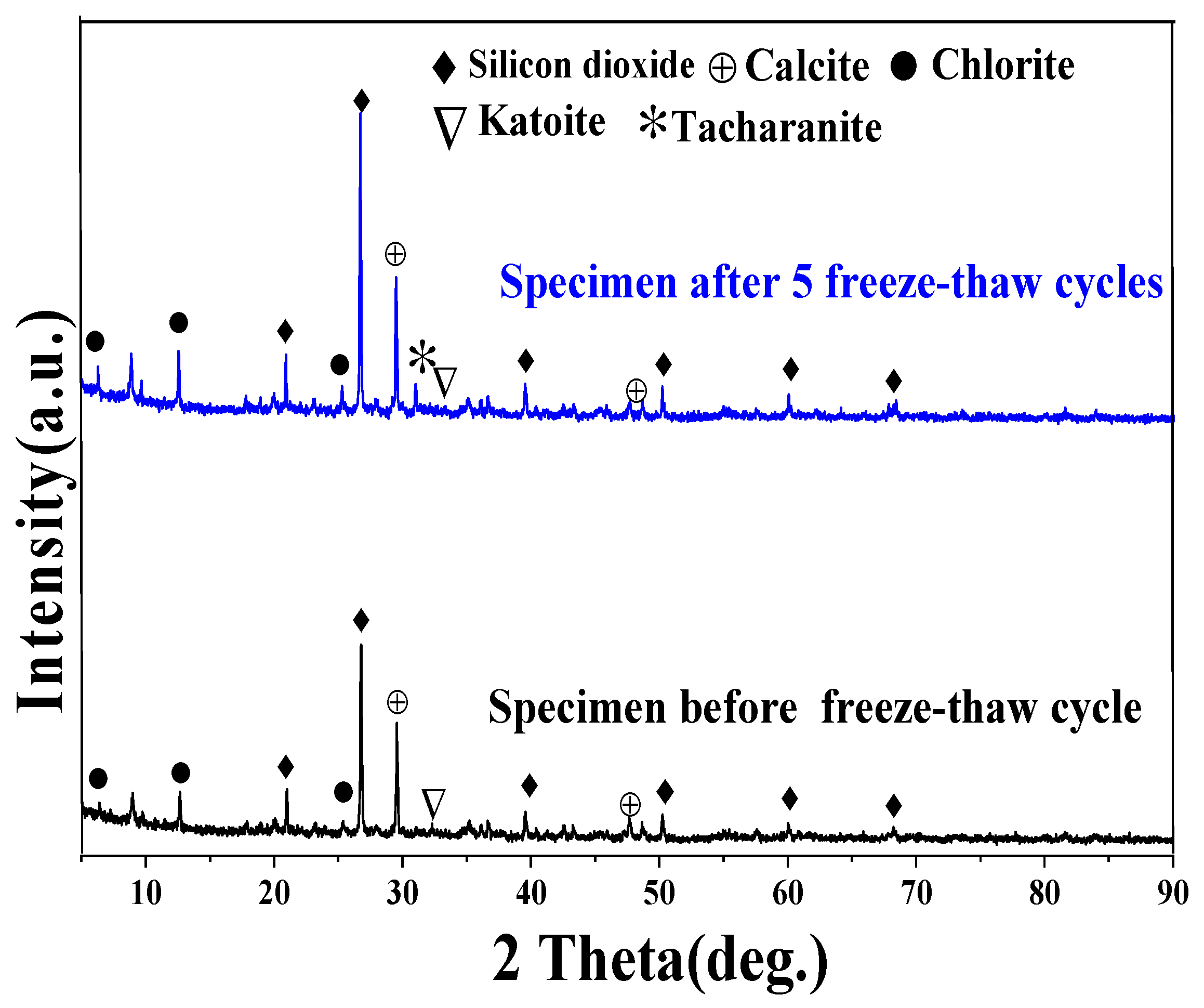
3.3. The Effect of Soil Stabilizer on the Freeze - Thaw Resistance of Stabilized Gravelly Soil
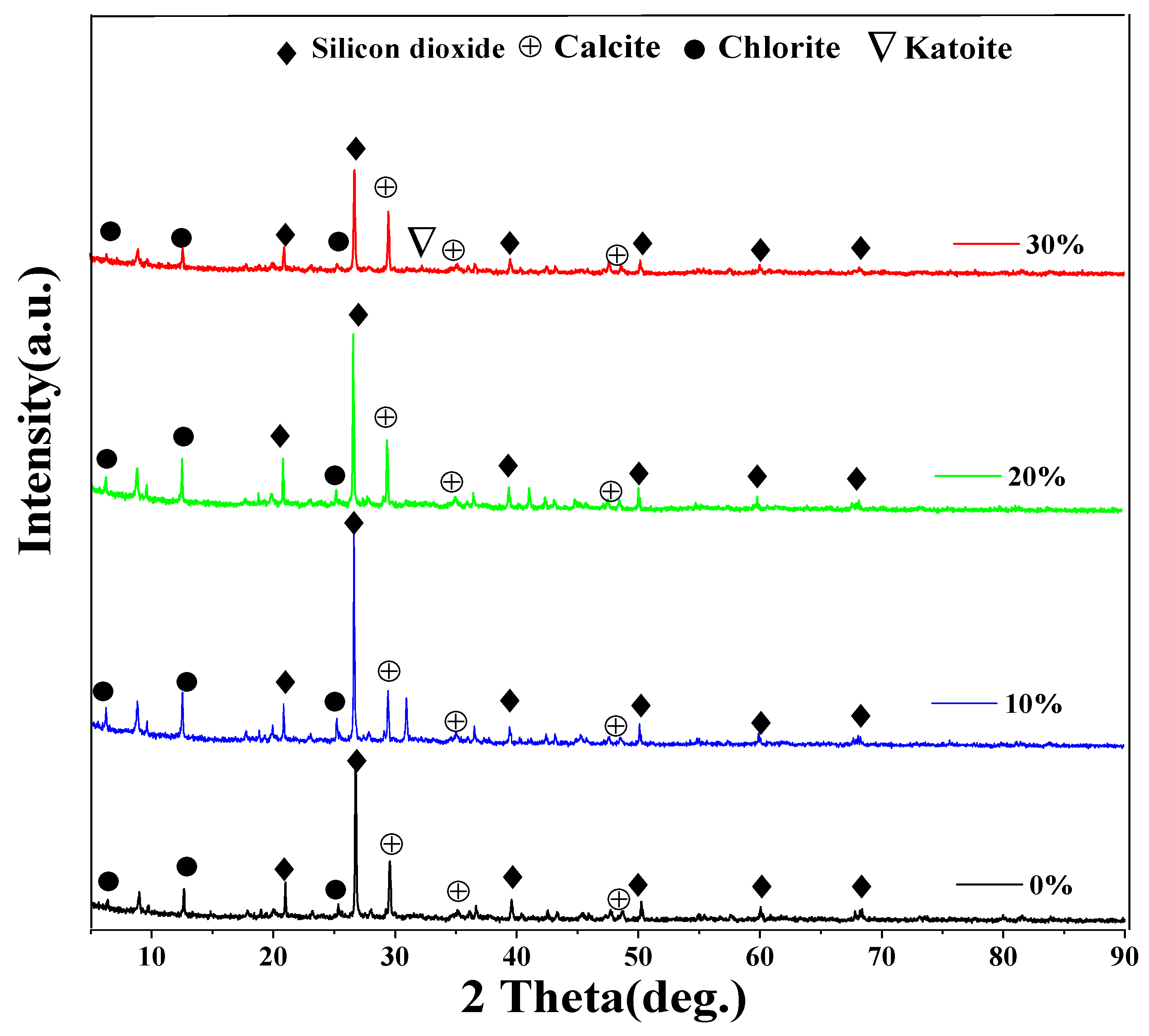
3.4. Discuss on the Mechanism of the Soil Stabilizer on Gravelly Soil Stabilization
4. Conclusion
Author Contributions
Acknowledgments
References
- ZHAO M Z, LIU G, DENG L X, et al. Optimizing the compaction characteristics and strength properties of gravelly soils in terms of fine contents[J]. Advances in Materials Science and Engineering, 2021, 2021: 6634237.
- WANG T, YUE Z R. Influence of fines content on frost heaving properties of coarse grained soil[J]. Rock and Soil Mechanics, 2013, 34(2): 359–363.
- PASTOR J L, CHAI J, SÁNCHEZ I. Strength and microstructure of a clayey soil stabilized with natural stone industry waste and lime or cement[J]. Applied Sciences, 2023, 13(4): 2583.
- ONYELOWE K C, MOGHAL A A B, EBID A, et al. Estimating the strength of soil stabilized with cement and lime at optimal compaction using ensemble-based multiple machine learning[J]. Scientific Reports, 2024, 14(1): 15308.
- ZHUANG S, XIE H, LING J. Study on the performance of cement-lime stabilized soil for road subgrade[J]. Journal of Highway and Transportation Research and Development, 2001, 18(3): 25–29.
- BOAVENTURA N F, SOUSA T F D P, CASAGRANDE M D T. The application of an eco-friendly synthetic polymer as a sandy soil stabilizer[J]. Polymers, 2023, 15(24): 4626.
- LI H, JIA J, LU X, et al. The effect of ionic soil stabilizer on cement and cement-stabilized iron tailings soil: Hydration difference and mechanical properties[J]. Materials, 2025, 18(7): 1444.
- WU Y, LIU X, YANG J, et al. Influence of the wide range cement content on the properties of cement–treated marine soft soil and bound method of semi–solidified soil[J]. Case Studies in Construction Materials, 2024, 20: 03180.
- WANG J, LI X, WEN H. Shrinkage cracking model for cementitiously stabilized layers for use in the mechanistic-empirical pavement design guide[J]. Construction and Building Materials, 2020, 260: 119–130.
- SAHLABADI S H, BAYAT M, MOUSIVAND M, et al. Freeze–thaw durability of cement-stabilized soil reinforced with polypropylene/basalt fibers[J]. Journal of Materials in Civil Engineering, 2021, 33(9): 04021232.
- SHIBI T, KAMEI T. Effect of freeze–thaw cycles on the strength and physical properties of cement-stabilised soil containing recycled bassanite and coal ash[J]. Cold Regions Science and Technology, 2014, 106: 36–45.
- LAVEGLIA A, SAMBATARO L, UKRAINCZYK N, et al. Hydrated lime life-cycle assessment: Current and future scenarios in four EU countries[J]. Journal of Cleaner Production, 2022, 369: 133224.
- KOGBARA R B, MASAD E A, LITTLE D N, et al. A state-of-the-art review of polymers used in soil stabilization[J]. Construction and Building Materials, 2021, 305: 124685.
- LIU Y, ZHAO Z, AMIN M N, et al. Foam concrete for lightweight construction applications: A comprehensive review of the research development and material characteristics[J]. Reviews on Advanced Materials Science, 2024, 63(1): 20240022.
- LIU J, LIU Y, MA X, et al. Thermal performance optimisation of foam concrete for energy-efficient construction: A state-of-the-art review[J]. Journal of Building Engineering, 2025, 113899.
- FU Y, WANG X, WANG L, et al. Foam concrete: a state-of-the-art and state-of-the-practice review[J]. Advances in Materials Science and Engineering, 2020, 2020: 6153602.
- JUST A, MIDDENDORF B. Microstructure of high-strength foam concrete[J]. Materials Characterization, 2009, 60(7): 741–748.
- HUANG W, LIU J, SHI Q, et al. Mechanical and pore properties of foam concrete under salt erosion environment[J]. Materials, 2025, 18: 2810.
- HUANG B, WANG X, KUA H W, et al. Construction and demolition waste management: China’s lessons[J]. Resources, Conservation and Recycling, 2018, 129: 36–44.
- ENGELSEN C J, VAN DER SLOOT H A, PETKOVIC G. Leaching behaviour of recycled aggregate used in unbound road layers–A laboratory study[J]. Waste Management, 2017, 61: 408–416.
- WEBER R, MÜLLER J. Landfilling of construction and demolition waste: A review of environmental issues[J]. Waste Management & Research, 2013, 31(10): 1005–1016.
- ZUHEIR B, AL-MULALI M Z. An innovative approach on recycle foam concrete as a sustainable alternative with the addition of nano titanium dioxide TiO2 on the properties of foam concrete[J]. Engineering, Technology & Applied Science Research, 2025, 15(1): 19196–19199.
- TURKEY F A, BEDDU S, AL-HUBBOUBI S K, et al. Recycled foam concrete masonry and porcelanite rocks-based lightweight geo-polymer concrete at elevated temperatures[J]. Alexandria Engineering Journal, 2024, 105: 171–180.
- ZUHAIR B, AMEER M Z A. Innovative approach to foam concrete production by utilizing recycled foam concrete as a sustainable alternative[J]. Journal of Engineering, 2025, 31(4): 159–174.
- GOŁASZEWSKI J, CYGAN G, GOŁASZEWSKA M, et al. Utilization of recycled foam concrete powder with phase-change material as a cement or sand replacement: Impact on mortar properties and superplasticizer performance[J]. Sustainability, 2025, 18(2): 1004.
- SONG Y, LANGE D. Crushing performance of ultra-lightweight foam concrete with fine particle inclusions[J]. Applied Sciences, 2019, 9(5): 876.
- PIZOŃ J. Fresh, mechanical, and thermal properties of cement composites containing recycled foam concrete as partial replacement of cement and fine aggregate[J]. Materials, 2023, 16(22): 7169.
- OHEMENG E A, EKOLU S O. A review on the reactivation of hardened cement paste and treatment of recycled aggregates[J]. Magazine of Concrete Research, 2020, 72(10): 526–539.
- CUNHA S, KAPTAN K, HARDY E, et al. Assessment of the efficiency of mechanical grinding and calcination processes for construction and demolition waste as binder replacement in cement pastes: Mechanical properties evaluation[J]. Sustainability, 2025, 17: 5248.
- STEPKOWSKA E T, BLANES J M, FRANCO F, et al. Phase transformation on heating of an aged cement paste[J]. Thermochimica Acta, 2004, 420(1-2): 79–87.
- JI X, JI D, YANG Z, et al. Study on the phase composition and structure of hardened cement paste during heat treatment[J]. Construction and Building Materials, 2021, 310: 125267.
- HASANIN T H A, ALSAHLI S A, ALTALLEB H A, et al. Hydration characteristics of cement blended with thermally reactivated recycled concrete demolition waste[J]. Scientific Reports, 2026, 16: 1499.
- XI X, ZHENG Y, DU C, et al. Study on the hydration characteristics, mechanical properties, and microstructure of thermally activated low-carbon recycled cement[J]. Construction and Building Materials, 2024, 447: 138042.
- XU R, XIE Y, CHEN Y, et al. Mechanical properties and micro-mechanisms of soft soil stabilized with rice husk ash and multi-source solid waste-based cementitious materials[J]. Frontiers in Built Environment, 2025, 11: 1759978.
- ZHUANG S, WANG Q, LUO T. Effect of C₁₂A₇ in steel slag on the early-age hydration of cement[J]. Cement and Concrete Research, 2022, 162: 107010.
- JIANG N, WANG C, WANG Z, et al. Strength characteristics and microstructure of cement stabilized soft soil admixed with silica fume[J]. Materials, 2021, 14(8): 1929.
- LAI Z, CHEN Y. Enhancing the mechanical and environmental performance of solidified soil using construction waste and glass micro-powder[J]. Heliyon, 2024, 10(22): e40187.
- ZHANG W, MU F Y, XUE Q, et al. Solidification/stabilization mechanisms of uncalcined waste concrete powder and ground granulated blast slag on lead-contaminated soil: From nanoscale to macroscale[J]. Journal of Environmental Chemical Engineering, 2025, 13(5): 117776.
- TIAN H, WEI C, TAN L. Effect of freezing-thawing cycles on the microstructure of soils: A two-dimensional NMR relaxation analysis[J]. Cold Regions Science and Technology, 2019, 158: 106–116.
- REN H, ZHANG D, ZHANG L, et al. Research advances in the freeze–thaw resistance of cement-stabilized soils[J]. Journal of Cold Regions Engineering, 2026, 40(2): 03126001.


|
Natural moisture content (%) |
Maximum dry density (g·cm-3) |
Optimum moisture content(%) | Coefficient of uniformity (Cu) |
Coefficient of curvature (Cc) |
| 11.5 | 2.22 | 10.6 | 8.37 | 1.21 |
| Particle size range (mm) | (0.25,0.5) | (0.5,10) | (1,2) | (2,5) | (5,10) | (10,20) | (20,40) |
| Particle content (%) | 1.2 | 13.8 | 8.5 | 28.2 | 22.0 | 16.3 | 10.0 |
| Samples | dosage of stabilizer(%) |
Maximum dry density (g/cm3) |
Optimum moisture content(%) |
| SGS0 | 0 | 2.22 | 10.60 |
| SGS10 | 10 | 1.98 | 10.01 |
| SGS20 | 20 | 1.80 | 10.32 |
| SGS30 | 30 | 1.83 | 10.77 |
|
dosage of soil stabilizer (%) |
soaking time(d) | ||||||
| 1d | 2d | 3d | 4d | 5d | 6d | 7d | |
| 0 | disintegrate | - | - | - | - | - | - |
| 10 | disintegrate | - | - | - | - | - | - |
| 20 | stable | stable | stable | stable | stable | stable | stable |
| 30 | stable | stable | stable | stable | stable | stable | stable |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




