Submitted:
03 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. The DLI Axis: Established Mechanistic Evidence
1.1. DUOX Enzymes and H2O2 Generation
1.2. Lactoperoxidase and Hypoiodous Acid
1.3. Iodine Deficiency: Global Burden
1.4. HIF-1α Biology and Activation Threshold
1.5. Thyroid Dysfunction and IL-6-Driven Iodide Sequestration
1.6. Silent Hypoxia and SpO2 Trajectories
1.7. Blood pH: A Downstream Marker of Cascade Initiation
1.8. NHE9 and Endosomal pH
2. The Novel Synthesis: Oxygen as the Dual Control Variable
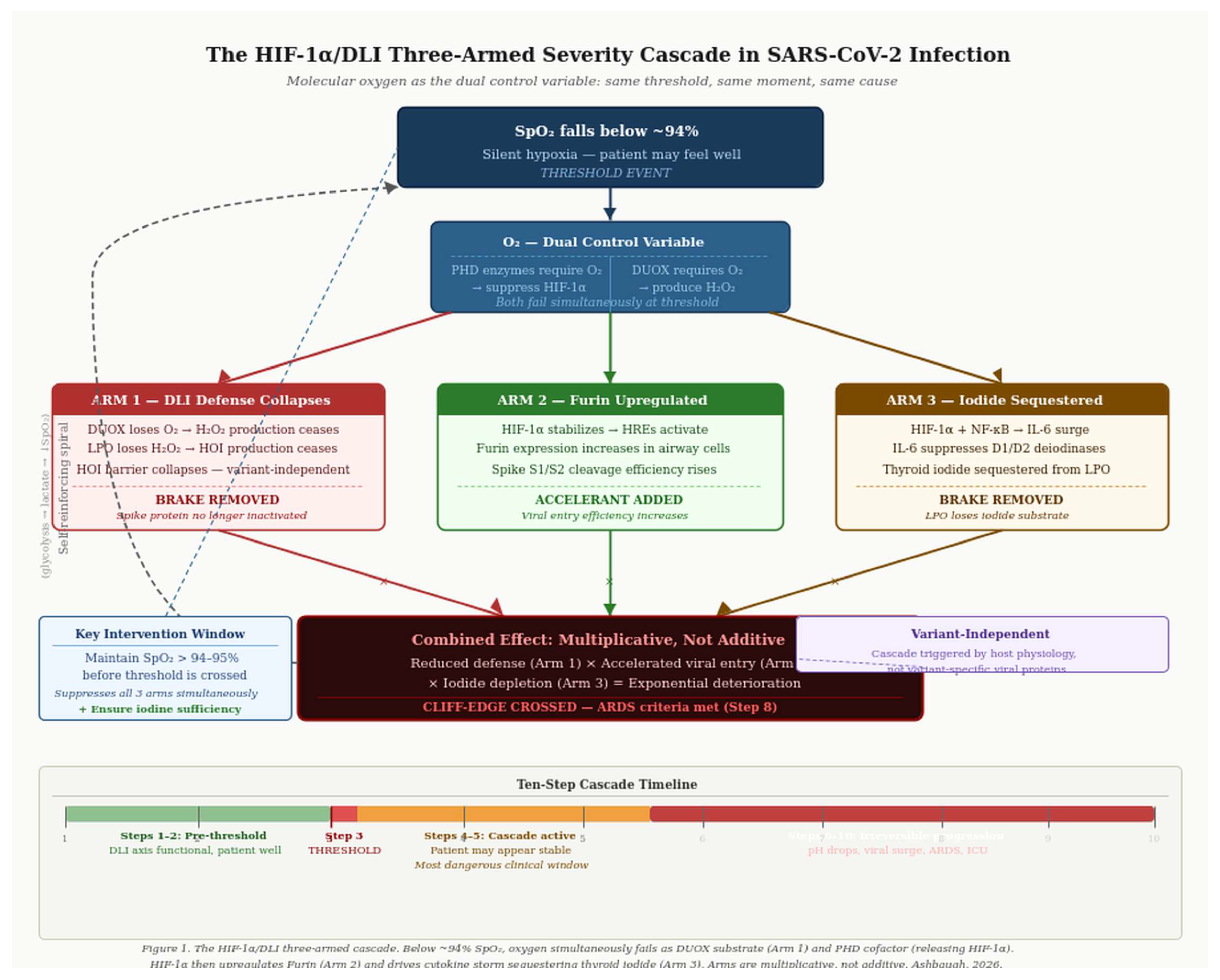
3. The Three-Armed Cascade
3.1. Arm 1: HIF-1α Dismantles the DLI Defense
3.2. Arm 2: HIF-1α Upregulates Furin, Accelerating Viral Entry
3.3. Arm 3: HIF-1α Drives Cytokine Storm and Iodide Depletion
| Arm | Trigger | Substrate / Target | Mechanism | Stages Active | Interaction Role |
| Arm 1 DLI Defense Collapse | SpO2 < ~94% (PHD fails; DUOX loses O2) | O2 (DUOX electron acceptor) | DUOX loses H2O2 output → LPO loses substrate → HOI production ceases. Spike protein no longer inactivated. | Steps 3–10 (immediate at threshold) | BRAKE REMOVED Reduces defense (multiplicative denominator) |
| Arm 2 Furin Upregulation | HIF-1α binds HREs in Furin promoter | Spike S1/S2 cleavage efficiency | Furin expression rises; more efficient spike cleavage → faster viral entry → more AT2 destruction → deeper SpO2 decline (feedback loop). | Steps 4–10 (lag ~hours post-threshold) | ACCELERANT ADDED Increases viral entry efficiency (multiplicative numerator) |
| Arm 3 Iodide Depletion | HIF-1α → NF-κB → IL-6 surge suppresses D1/D2 deiodinases | Thyroid iodide reserve (LPO substrate) | IL-6 suppresses D1/D2 → T4→T3 conversion blocked → iodide sequestered in thyroid → LPO loses I− substrate → HOI ceases even if DUOX recovered. | Steps 5–10 (2–4 day lag post-threshold) | BRAKE REMOVED Depletes iodide substrate (compounds Arm 1 multiplicatively) |
3.4. Why Exponential, Not Linear, Kinetics
3.5. The Ten-Step Cascade Summary
3.6. Computational Support for the Multiplicative Architecture
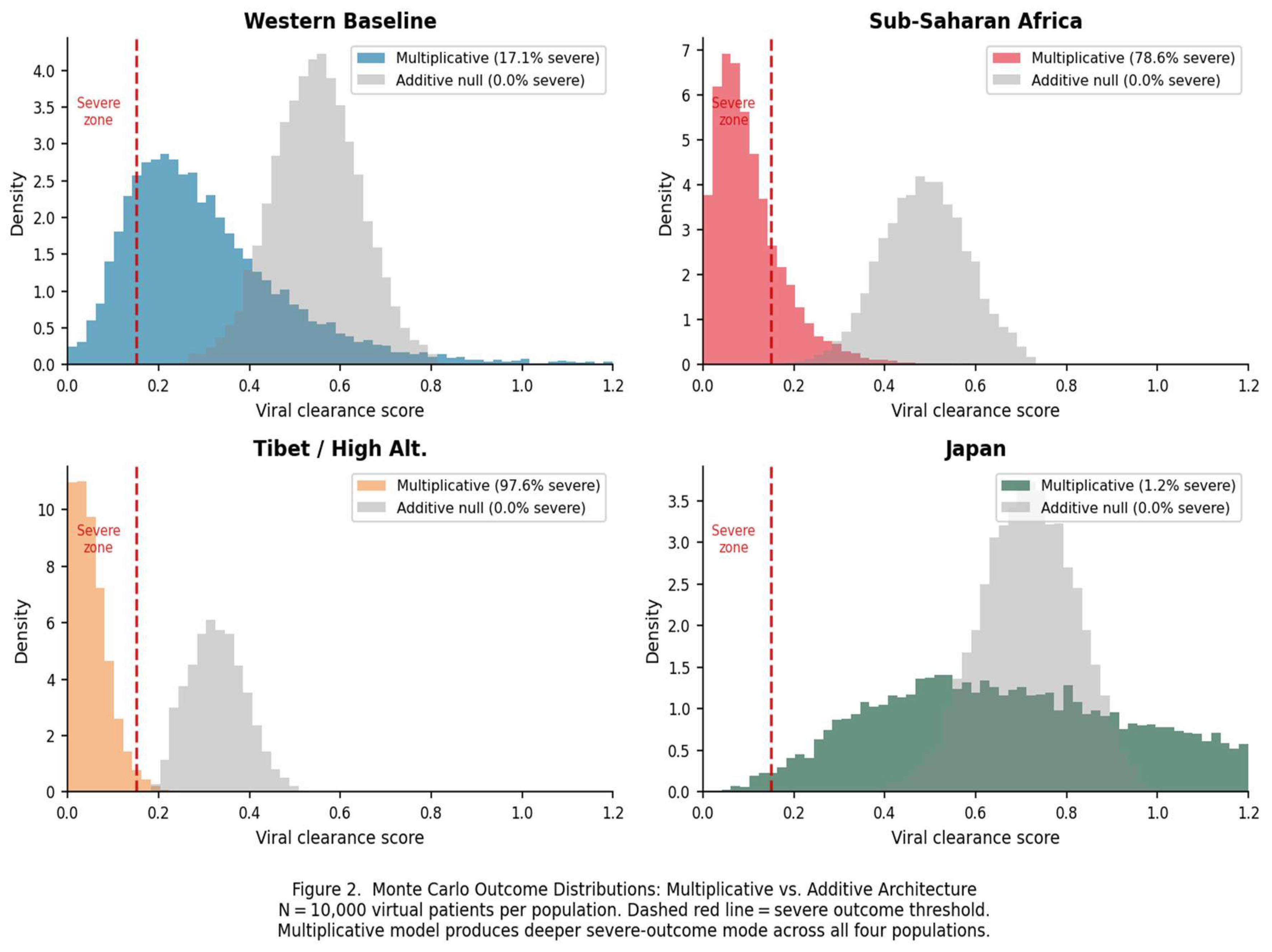
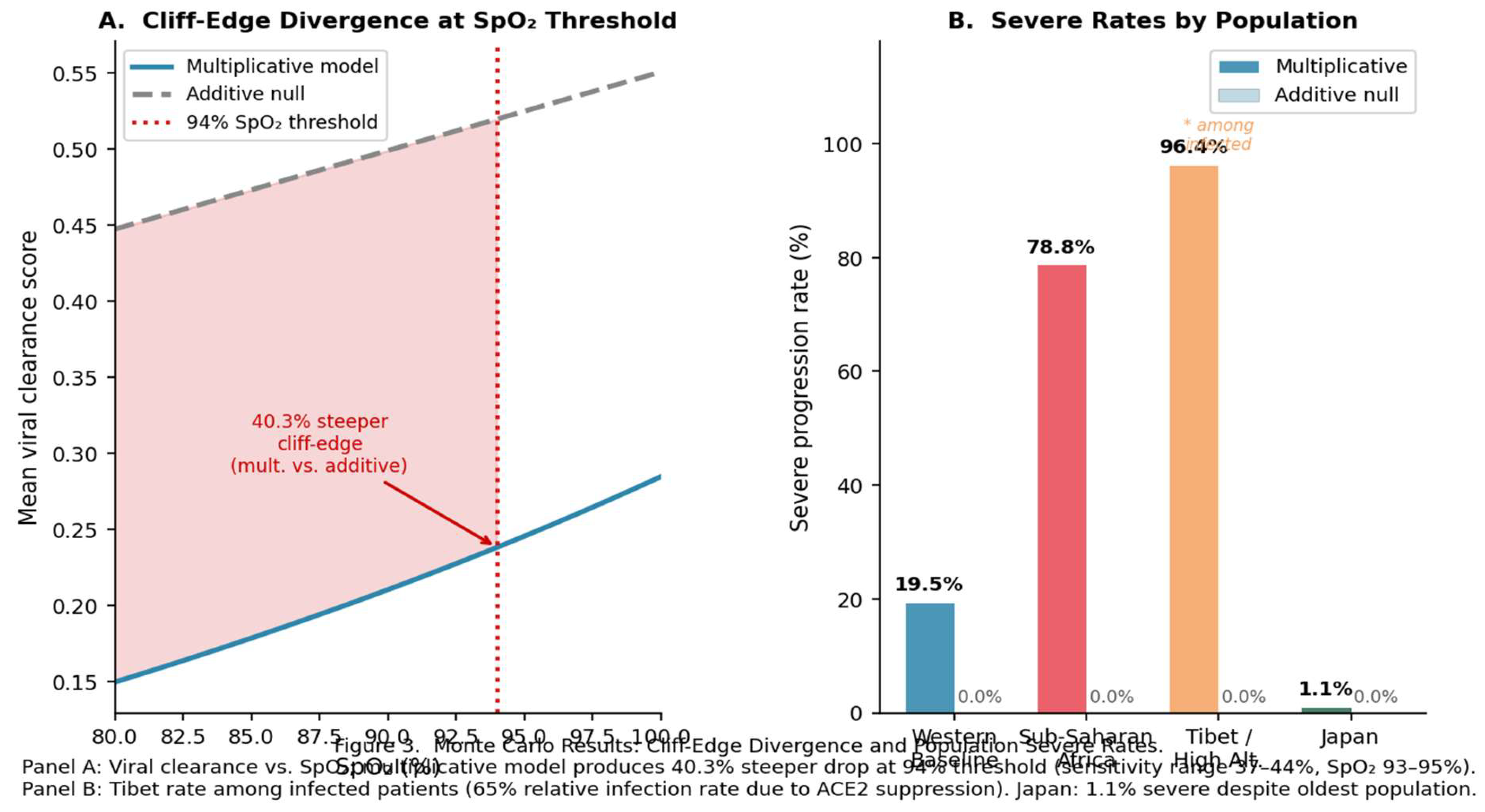
3.7. How the Computational Evidence Supports the HIF-1α Hypothesis
3.7.1. What the Monte Carlo Establishes
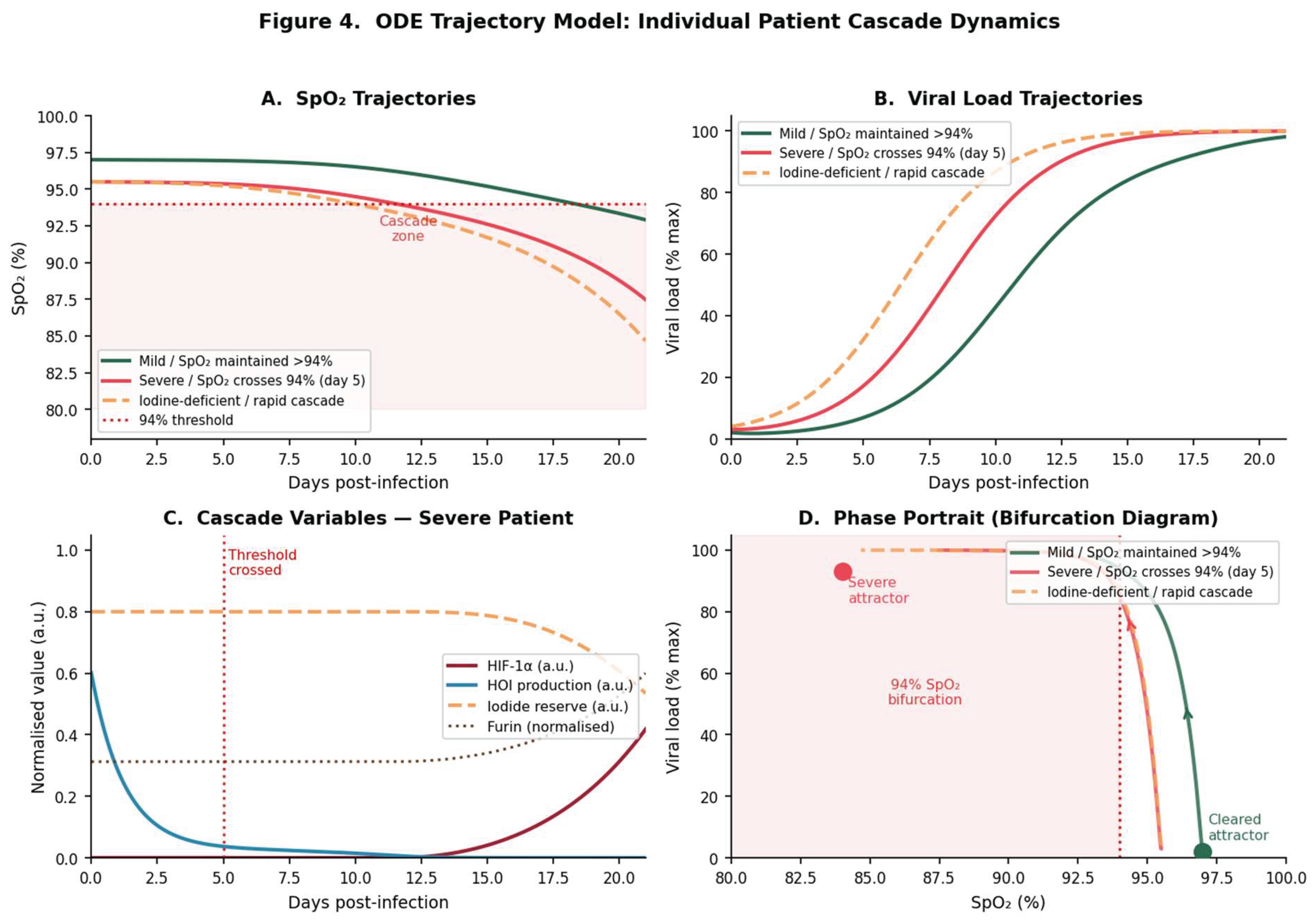
3.7.2. What the ODE Model Adds
3.7.3. Limitations of the Computational Evidence
3.7.4. Where Clinical Trials Would Resolve the Key Uncertainties
4. Counter-Evidence and Mechanistic Complications
4.1. The High-Altitude Dissociation: Incidence Without Fatality Reduction
4.2. The Sub-Saharan Africa Paradox
4.3. Povidone-Iodine Clinical Trial Heterogeneity
4.4. HIF-1α as a Protective Signal in Some Contexts
4.5. What Would Falsify This Framework
- If serum HIF-1α at admission shows no dose-dependent correlation with DLI markers, IL-6, and 28-day mortality in a prospective cohort (Prediction 1 fails), the cascade’s central molecular pivot is not operationally measurable as proposed.
- If early oxygen therapy at 94–95% SpO2 produces no measurable superiority over the standard ≤92% threshold in an RCT (Prediction 2 fails), the 94% threshold is not the mechanistic tipping point this framework claims.
- If iodine status at admission has no independent effect on time-to-cascade-progression after controlling for age and comorbidity (Prediction 3 fails), the iodide depletion arm is not clinically operative at the magnitude the framework predicts.
- If direct measurement of airway surface liquid O2 tension at SpO2 94% shows that DUOX activity is not yet meaningfully impaired — i.e., the threshold is wrong — the entire cascade timing model requires recalibration.
4.6. The Vaccination Question: Does the Framework Apply Post-Vaccination?
5. Three Testable Clinical Predictions
Prediction 1: HIF-1α as an Admission Biomarker of Cascade Stage
Prediction 2: Early Oxygen Therapy at the 94–95% SpO2 Threshold
Prediction 3: Iodine Status as an Independent Modifier of Cascade Progression
6. Clinical Implications
6.1. Oxygen Therapy: The Case for a Revised Threshold
6.2. Iodine Supplementation: Prophylactic and Therapeutic Rationale
6.3. The DLI Competency Index
6.4. At-Risk Populations
7. Limitations
- 5.
- The integrated cascade has not been tested as a unified model in a clinical cohort. Each component is supported independently; their combination as a self-amplifying cascade is proposed, not yet demonstrated.
- 6.
- The ~94% SpO2 threshold for HIF-1α activation is an approximation derived from PHD enzyme kinetics and indirect data. The precise threshold likely varies between cell types, tissues, individuals, and degree of chronic hypoxic pre-conditioning.
- 7.
- Direct measurement of circulating iodide dynamics in COVID-19 patients relative to cytokine storm progression has not been reported. The iodide depletion mechanism is inferred from established deiodinase biology, not from dedicated COVID-19 iodide kinetic studies.
- 8.
- The blood pH – endosomal transfer rate connection is a deferred hypothesis requiring direct experimental evidence before it can be incorporated as a mechanistic claim.
- 9.
- The sub-Saharan Africa paradox (high iodine deficiency + lower COVID-19 mortality) is mechanistically addressable but not fully resolved with existing data. Confounders — especially age structure and cross-reactive immunity — likely dominate the iodine signal in this population.
- 10.
- The high-altitude case-fatality dissociation (lower incidence but equal fatality at altitude) is consistent with the revised framework but represents a boundary condition that limits the framework’s predictive power for altitude-adapted populations.
- 11.
- Clinical recommendations for early oxygen intervention assume pulse oximetry and supplemental oxygen availability, which is not universal globally.
- 12.
- The Monte Carlo simulation is a theoretical consistency demonstration, not empirical validation. It shows that the multiplicative architecture produces outcome patterns consistent with clinical observations across four populations; it does not prove the mechanism. The cumulative viral clearance score is a model construct that cannot be directly validated against a single published dataset.
- 13.
- The ODE trajectory model has not been fitted to patient data and has not been validated prospectively. It is a forward model (mechanistic prediction) rather than an inverse model (data-fitted). Rate constants are estimated from qualitative biochemical knowledge and calibrated to produce physiologically plausible trajectories; they are not independently measured in COVID-19 patients.
- 14.
- The single largest source of model uncertainty is the iodide depletion kinetics during acute COVID-19. No published study has directly measured airway or serum iodide dynamics during active SARS-CoV-2 infection. The depletion curve used in the simulation is derived from non-thyroidal illness syndrome literature and IL-6 magnitude data.
8. Future Research Directions
- 15.
- Prospective cohort study measuring serum HIF-1α, urinary iodide, airway H2O2, and IL-6 at admission in COVID-19 patients, correlated with 28-day outcomes (Prediction 1).
- 16.
- RCT of supplemental oxygen initiated at SpO2 94–95% versus standard ≤92% threshold (Prediction 2).
- 17.
- Prospective measurement of circulating iodide dynamics in COVID-19 patients across cascade stages, directly correlating iodide depletion rate with cytokine storm markers and DLI surrogate measures.
- 18.
- DUOX activity measurement in airway surface liquid at varying SpO2 levels in controlled human challenge models, to directly confirm the oxygen dependency threshold.
- 19.
- Genetic studies linking DUOX2 SNP status to COVID-19 severity in large multi-ethnic cohorts, including sub-Saharan African and South Asian populations where both DUOX2 variants and iodine deficiency are prevalent.
- 20.
- Prospective study of urinary iodide at admission in COVID-19 patients across iodine-replete (Japan, Scandinavia) and iodine-deficient (inland Africa, Central Asia) populations, to test whether iodine status independently predicts cascade timeline after controlling for age and comorbidity.
- 21.
- Mechanistic investigation of whether systemic blood pH acidosis alters endosomal pH in airway epithelial cells, to test the deferred blood pH – endosomal transfer rate hypothesis.
- 22.
- Assessment of DLICI as a composite bedside staging tool, with formal validation against clinical outcomes in a prospective cohort.
- 23.
- Evaluation of Furin inhibition as a targeted Arm 2 intervention. Small-molecule Furin inhibitors — including decanoyl-RVKR-chloromethylketone (CMK) and the engineered serpin α1-antitrypsin Portland (α1-PDX) — have demonstrated suppression of SARS-CoV-2 spike S1/S2 cleavage and reduction of viral production in cell models [35,36]. The HIF-1α/DLI framework provides a novel rationale for the therapeutic window in which Furin inhibition would be most beneficial: Steps 4–6 of the cascade, after HIF-1α has upregulated Furin but before viral load has surged. Furin inhibition cannot substitute for Arms 1 and 3 interventions but represents a direct pharmacological approach to the accelerant arm of the cascade.
- 24.
- Formal Bayesian parameter estimation for the ODE cascade model. Once patient-level data are available — specifically serial serum iodide measurements, SpO2 trajectories, and DUOX2 genotypes from a prospective COVID-19 cohort — Bayesian inference methods (e.g., Markov Chain Monte Carlo) could be applied to estimate posterior distributions for the key rate constants. A cohort of 50–100 patients with serial SpO2, IL-6, and urinary iodide measurements over the first 14 days of admission would be sufficient for initial parameter estimation.
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
AI Collaboration Disclosure
Abbreviations
| Abbreviation | Definition |
| HIF-1α | Hypoxia-inducible factor 1-alpha |
| DUOX | Dual oxidase |
| LPO | Lactoperoxidase |
| HOI | Hypoiodous acid |
| DLI | DUOX-Lactoperoxidase-Iodine (axis) |
| PHD | Prolyl hydroxylase domain enzyme |
| SpO2 | Peripheral oxygen saturation |
| AT2 | Alveolar type II (pneumocyte) |
| V/Q | Ventilation-perfusion (ratio) |
| ARDS | Acute respiratory distress syndrome |
| NTIS | Non-thyroidal illness syndrome |
| NHE9 | Na+/H+ exchanger isoform 9 |
| DLICI | DLI Competency Index |
| ODE | Ordinary differential equation |
| RCT | Randomised controlled trial |
| ICU | Intensive care unit |
Appendix A. Computational Methods: ODE Trajectory Model and Bifurcation Analysis
A.1. State Variables and System Overview
A.2. Differential Equations
A.3. Bifurcation Analysis
A.4. Clinical Prediction Engine
Appendix B. Monte Carlo Simulation: Full Parameter Table
Appendix C. Supplementary Code
- 25.
- cascade_arm_dynamics.html — Monte Carlo simulation with interactive population explorer
- 26.
- sensitivity_analysis.html — sensitivity analysis across key parameters
- 27.
- cascade_model.py — Python implementation of the ODE trajectory model and clinical prediction engine
Appendix D. Data Summary Tables
Appendix E. Interactive Visualisation Tools
- 28.
- cascade_visualization.html: Monte Carlo Explorer — visualises population-level outcome distributions under multiplicative and additive architectures across four simulated populations.
- 29.
- cascade_ode_model.html: ODE Model — visualises individual patient cascade trajectories, bifurcation diagram, and phase portrait for the six-variable ODE system.
- 30.
- clinical_predictor.html: Clinical Prediction Engine — accepts patient-specific parameters and generates predicted cascade timeline.
- 31.
- population_model.html: Population Explorer — compares cascade dynamics across iodine-replete and iodine-deficient populations.
References
- Alsaleh, S.; Alhussein, A.; Alyamani, A.; Alhussain, F.; Alhijji, A.; Binkhamis, K.; Khan, A.; Javer, A.; Alshahrani, F.S. Efficacy of povidone-iodine nasal rinse and mouth wash in COVID-19 management: a prospective, randomized pilot clinical trial. BMC Infect. Dis. 2024, 24, 271. [Google Scholar] [CrossRef] [PubMed]
- Boer, M.L.; Smith, M.L.; Singh, T.P. Iodide supplementation of the anti-viral duox-lactoperoxidase activity may prevent some SARS-CoV-2 infections. Eur. J. Clin. Nutr. 2022, 76, 629–630. [Google Scholar] [CrossRef]
- Cummings, M.J.; Baldwin, M.R.; Abrams, D.; Jacobson, S.D.; Meyer, B.J.; Balough, E.M.; Aaron, J.G.; Claassen, J.; Rabbani, L.E.; Hastie, J.; et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet 2020, 395, 1763–1770. [Google Scholar] [CrossRef] [PubMed]
- Dhamija, P.; Bhargava, R. “Silent hypoxemia” leads to vicious cycle of infection, coagulopathy and cytokine storm in COVID-19: can prophylactic oxygen therapy prevent it? Indian J. Physiol. Pharmacol. 2021, 65, 10–17. [Google Scholar] [CrossRef]
- Effraimidis, G.; Konstantinos, P.; Brix, T.H.; Hegedus, L. Cytokine storm-induced thyroid dysfunction in COVID-19: insights into mechanisms and clinical management. Drug Des. Dev. Ther. 2024, 18, 4303–4320. [Google Scholar] [CrossRef]
- Elsersy, H.E.; Zahran, M.A.H.; Elbakry, A.-E.; Abd-Elwahab, M.; Ahmed, M.M.; Elgandy, M.S.; Mohammed, E.H.M.; Elewa, N.M. Combined nasal, oropharyngeal povidone iodine plus glycyrrhizic acid sprays accelerate clinical and laboratory recovery and reduce household transmission of SARS-CoV-2. Front. Med. 2022, 9, 863917. [Google Scholar] [CrossRef]
- Fischer, A.J.; Lennemann, N.J.; Krishnamurthy, S.; Pocza, P.; Durairaj, L.; Launspach, J.L.; Rhein, B.A.; Wohlford-Lenane, C.; Lorentzen, D.; Banfi, B.; et al. Enhancement of respiratory mucosal antiviral defenses by the oxidation of iodide. Am. J. Respir. Cell Mol. Biol. 2011, 45, 874–881. [Google Scholar] [CrossRef]
- Friedland, P.; Tucker, S.; Goodall, S.; Hale, S.J.M.; Lux, C.A. Phase II trial of the impact 0.5% povidone-iodine nasal spray (Nasodine) on shedding of SARS-CoV-2. Laryngoscope 2024. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Zevallos-Concha, A.; Tapia, V.; Gasco, M. High altitude reduces infection rate of COVID-19 but not case-fatality rate. Respir. Physiol. Neurobiol. 2020, 281, 103494. [Google Scholar] [CrossRef]
- Harper, R.W.; Xu, C.; Soucek, K.; Setiadi, H.; Eiserich, J.P. Dual oxidase 2 bidirectional promoter polymorphisms confer differential immune responses in airway epithelia. Am. J. Respir. Cell Mol. Biol. 2012, 46, 289–297. [Google Scholar] [CrossRef]
- Iftikhar, A.; Islam, M.; Shepherd, S.; Jones, S.; Ellis, I. An in vitro antiviral activity of iodine complexes against SARS-CoV-2. Arch. Microbiol. 2021, 203, 4233–4241. [Google Scholar] [CrossRef] [PubMed]
- Iodine Global Network. Global Scorecard of Iodine Nutrition 2025. Available online: https://ign.org/scorecard/ (accessed on April 2026).
- Kasumba, D.M.; Huot, S.; Caron, E.; Larouche, A.; Tremblay, M.G.; Emond-Rheault, J.G.; Bhatt, D.L. DUOX2 regulates secreted factors in virus-infected respiratory epithelial cells that contribute to neutrophil attraction and activation. FASEB J. 2023, 37, e22765. [Google Scholar] [CrossRef]
- Luo, Z.; Ke, C.; Lai, Y. Global burden of iodine deficiency: insights and projections to 2050 using GBoost and SHAP. Adv. Nutr. 2025. [Google Scholar] [CrossRef]
- Machado, C.; Gonzalez-Quevedo, A. Hypoxemia and cytokine storm in COVID-19: clinical implications. MEDICC Rev. 2021, 23, 54–59. [Google Scholar] [CrossRef]
- Matsuyama, A.; Okura, H.; Hashimoto, S.; et al. A prospective, randomized, open-label trial of early versus late povidone-iodine gargling in patients with COVID-19. Sci. Rep. 2022, 12, 20449. [Google Scholar] [CrossRef] [PubMed]
- Mertens, L.; Boer, M.L.; Smith, M.L.; Pham, K.; Noorlander, C.W.; Boer, R. Iodine increases pulmonary type I interferon responses and decreases COVID-19 disease severity: results from an open-label randomized clinical trial. PLOS ONE 2026, 21, e0341126. [Google Scholar] [CrossRef]
- NIH COVID-19 Treatment Guidelines Panel. Oxygenation and Ventilation for Adults: COVID-19 Treatment Guidelines. National Institutes of Health, 2020. Available online: https://www.covid19treatmentguidelines.nih.gov.
- Olagnier, D.; Farahani, E.; Thyrsted, J.; Blay-Cadanet, J.; Herengt, A.; Idorn, M.; Hait, A.; Maurin, B.; Sobanski, V.; Moya, D.; et al. Mucosal reactive oxygen species are required for antiviral response: role of Duox in influenza A virus infection. Antioxid. Redox Signal. 2014, 20, 2695–2709. [Google Scholar] [CrossRef]
- Prasad, V.; Keller, M.; Engberg, N.; Lindqvist, R.; Oberg, M.; Hober, S.; Punga, T.; Strom, A. Acidic pH of early endosomes governs SARS-CoV-2 transport in host cells. J. Biol. Chem. 2024. [Google Scholar] [CrossRef]
- Ruggeri, R.M.; Campenni, A.; Giuffrida, G.; Casciaro, M.; Bonanno, A.M.; Alibrandi, A.; Trimarchi, F.; Gangemi, S. The cytokine storm and thyroid hormone changes in COVID-19. J. Endocrinol. Invest. 2021, 44, 891–897. [Google Scholar] [CrossRef]
- Salinas-Assumpao, R.; Ferreira, M.L.; Bezerra, C.S. HIF-1α pathway in COVID-19: a scoping review of its modulation and related treatments. Int. J. Mol. Sci. 2025, 26, 4202. [Google Scholar] [CrossRef]
- Siebert, M.; Barreto, G.; Barreto, P.; Pereira, S.M.M.; Gomes, K.B.; Paixao, H.H.T. Blood pH analysis in combination with molecular medical tools in relation to COVID-19 symptoms. Biomedicines 2023, 11, 1421. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.L.; Sharma, S.; Singh, T.P. Iodide supplementation of the anti-viral duox-lactoperoxidase activity may prevent some SARS-CoV-2 infections. Scand. J. Immunol. 2022, 95, 196–198. [Google Scholar] [CrossRef]
- Smith, M.L.; Sharma, S.; Singh, T.P. High dietary iodine intake may contribute to the low death rate from COVID-19 infection in Japan with activation by the lactoperoxidase system. Scand. J. Immunol. 2023, 97, e13269. [Google Scholar] [CrossRef]
- Strengert, M.; Jennings, R.; Davanture, S.; Hayes, P.; Gabriel, G.; Knaus, U.G. Dual oxidase 1 promotes antiviral innate immunity. Proc. Natl. Acad. Sci. USA 2021, 118, e2017130118. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y.; Wang, Q.; Yang, C.; Zhang, M.; Yu, J.; Li, X.; Ji, M.; Ren, J. Virucidal effect of povidone-iodine against SARS-CoV-2 in vitro. J. Int. Med. Res. 2021, 49. [Google Scholar] [CrossRef]
- Waruru, A.; Wambua, J.M.; Otieno, F.V.; Mbai, J.; Mwangome, M.; Munyua, P.; Waruiru, W.; Jonnalagadda, S.; Ngunu, C.; Muange, A.K.; et al. Hypoxia as a predictor of mortality among patients admitted with COVID-19 disease in three referral hospitals in Kenya. medRxiv 2024. [Google Scholar] [CrossRef]
- WHO European Region; Iodine Global Network. Prevention and Control of Iodine Deficiency in the WHO European Region: Adapting to Changes in Diet and Lifestyle; WHO Regional Office for Europe, 2024. [Google Scholar]
- World Health Organization. COVID-19 Dashboard; WHO. 2023. Available online: https://covid19.who.int.
- Zhang, R.; Wu, Y.; Zhao, M.; Liu, C.; Zhou, L.; Shen, S.; Liao, S.; Yang, K.; Li, Q.; Wan, H. Role of HIF-1alpha in the regulation of ACE and ACE2 expression in hypoxic human pulmonary artery smooth muscle cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2009, 297, L631–L640. [Google Scholar] [CrossRef]
- Zava, T.T.; Zava, D.T. Assessment of Japanese iodine intake based on seaweed consumption in Japan: a literature-based analysis. Thyroid Res. 2011, 4, 14. [Google Scholar] [CrossRef]
- Nitipir, C.; Orlov, C.; Marin, A.; Miron, A.; Balescu, I.; Bacalbasa, N. Repurposing povidone-iodine to reduce the risk of SARS-CoV-2 infection and transmission: a narrative review. Ann. Med. 2022, 54, 1236–1247. [Google Scholar] [CrossRef]
- Zarabanda, D.; Vukkadala, N.; Phillips, K.M.; Qian, Z.J.; Mfuh, K.O.; Hatter, M.J.; Lee, I.T.; Rao, V.K.; Hwang, P.H.; Domb, G.; et al. The effect of povidone-iodine nasal spray on nasopharyngeal SARS-CoV-2 viral load: a randomized control trial. Laryngoscope 2022, 132, 2089–2095. [Google Scholar] [CrossRef] [PubMed]
- Devi, K.P.; Pourkarim, M.R.; Thijssen, M.; Sureda, A.; Khayatkashani, M.; Cismaru, C.A.; Berindan Neagoe, I.; Habtemariam, S.; Razmjouei, S.; Khayat Kashani, H.R. A perspective on the applications of furin inhibitors for the treatment of SARS-CoV-2. Pharmacol. Rep. 2022, 74, 425–430. [Google Scholar] [CrossRef]
- Thomas, G.; Couture, F.; Kwiatkowska, A. The path to therapeutic furin inhibitors: from yeast pheromones to SARS-CoV-2. Int. J. Mol. Sci. 2022, 23, 3435. [Google Scholar] [CrossRef]
- Afsar, B.; Kanbay, M.; Elsurer Afsar, R. Hypoxia inducible factor-1 protects against COVID-19: a hypothesis. Med. Hypotheses 2020, 143, 109857. [Google Scholar] [CrossRef] [PubMed]
- Arias-Reyes, C.; Zubieta-DeUrioste, N.; Poma-Machicao, L.; Aliaga-Raduan, F.; Carvajal-Rodriguez, F.; Dutschmann, M.; Schneider-Gasser, E.M.; Zubieta-Calleja, G.; Soliz, J. Does the pathogenesis of SARS-CoV-2 virus decrease at high-altitude? Respir. Physiol. Neurobiol. 2020, 277, 103443. [Google Scholar] [CrossRef]
- Mayr, U.; Londhe, P.; Abegg, K.; Schotten, T.; Schultze, T.; Braunisch, M.; Zimmermann, G.; Schmid, R.M. HIF1alpha cardioprotection in COVID-19 patients. medRxiv 2021. [Google Scholar] [CrossRef]
- ANTICOV-IMMUNO Study Group. Determinants of long-term SARS-CoV-2 immune responses in asymptomatic-to-moderate COVID-19 patients in sub-Saharan Africa. BMC Med. 2026, 23. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Pohlmann, S. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 2021, 22, 517–534. [Google Scholar] [CrossRef]
- Kwizera, A.; Kabatoro, D.; Sendagire, C.; Nakibuuka, J.; Owachi, D.; Nsereko, C.; Ochieng, J.P.; Nampiina, M.G.; Nampaawu, M.J.; Kakaire, D.; et al. The epidemiology and outcomes of adults with acute hypoxaemic respiratory failure in a low-income country in the context of the COVID-19 pandemic. BMJ Glob. Health 2025, 10, e017949. [Google Scholar] [CrossRef] [PubMed]
- Mikami, Y.; Grubb, B.R.; Rogers, T.D.; Dang, H.; Asakura, T.; Kota, P.; Gilmore, R.C.; Okuda, K.; Morton, L.C.; Sun, L.; et al. Chronic airway epithelial hypoxia exacerbates injury in muco-obstructive lung disease through mucus hyperconcentration. Sci. Transl. Med. 2023, 15, eabo7728. [Google Scholar] [CrossRef]
- Derscheid, R.J.; van Geelen, A.; Berkebile, A.R.; Gallup, J.M.; Hostetter, S.J.; Banfi, B.; McCray, P.B., Jr.; Ackermann, M.R. Increased concentration of iodide in airway secretions is associated with reduced respiratory syncytial virus disease severity. Am. J. Respir. Cell Mol. Biol. 2014, 50, 389–397. [Google Scholar] [CrossRef]
- Bastani, M.N.; Jalilian, S. Unraveling the enigma: the emerging significance of pulmonary surfactant proteins in predicting, diagnosing, and managing COVID-19. Immun. Inflamm. Dis. 2024, 12, e1302. [Google Scholar] [CrossRef]
- Fontela, P.C.; Berwanger, O.; Cavalcanti, A.B.; Tanaka, L.M.S.; Daltro-Oliveira, R.; Lobo, S.M.; Lacerda, M.V.G.; Moura, A.D.T. New and personalized ventilatory strategies in patients with COVID-19. Front. Med. 2023, 10, 1194773. [Google Scholar] [CrossRef]
- Abou-Ismail, M.Y.; Diamond, A.; Kapoor, S.; Arafah, Y.; Nayak, L. The hypercoagulable state in COVID-19: incidence, pathophysiology, and management. Thromb. Res. 2020, 194, 101–115. [Google Scholar] [CrossRef]
- Genzel, L.; Akgul, G.; Abel, T.; Bhatt, D.L.; Bhatt, S.; Birmingham, K.; Bishnoi, I.R.; Bhatt, M.; Bhatt, V.; et al. Animal models for the study of SARS-CoV-2-induced respiratory disease and pathology. Comp. Med. 2023, 73, 85–103. [Google Scholar] [CrossRef]
- Zhong, M.; Gao, Y.; Hu, H.; Zhu, X.; Gan, L.; Li, L.; Xiang, C.; Yan, Y.; Dai, Z. Transient low T3 syndrome in patients with COVID-19: a new window for prediction of disease severity. Front. Endocrinol. 2023, 14, 1154007. [Google Scholar] [CrossRef]
- Colella, C.; Baldelli, R.; Saba, L.; Granata, A. Non-thyroidal illness syndrome and thyroid autoimmunity in hospitalized COVID-19 patients: a retrospective study. J. Clin. Med. 2025, 14, 6784. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Zhang, S.; Peng, F.; Zhang, Q.; Li, Y.; Zhong, Y. The association between fT3 with the outcome and inflammation/coagulopathy/fibrinolysis of COVID-19. Front. Endocrinol. 2022, 13, 877010. [Google Scholar] [CrossRef] [PubMed]
- Rada, B.; Gardina, P.; Myers, T.G.; Trinchieri, G. Antimicrobial actions of dual oxidases and lactoperoxidase. J. Microbiol. 2018, 56, 373–386. [Google Scholar] [CrossRef] [PubMed]
| Step | Phase | Arms Active | Event / Cascade State |
| 1 | Pre-threshold | None (DLI intact) | Viral entry; initial replication; DLI axis functional; patient asymptomatic or mildly symptomatic. |
| 2 | Pre-threshold | None (DLI intact) | SpO2 begins falling via AT2 cell destruction (surfactant loss), V/Q mismatch, and microvascular thrombosis. Silent hypoxia. HIF-1α still suppressed by PHD enzymes above threshold. |
| 3 | THRESHOLD EVENT | Arms 1+2+3 initiated | SpO2 crosses ~94%; PHD activity falls; HIF-1α accumulates; DUOX O2 substrate depleted. All three cascade arms activate simultaneously. |
| 4 | Early cascade | Arm 1 + Arm 2 | HIF-1α activates anaerobic glycolysis and upregulates Furin; Furin-mediated viral entry accelerates AT2 destruction, steepening SpO2 decline. Viral amplification feedback loop begins. HOI falls below protective concentration. |
| 5 | Early cascade | Arm 3 initiating | NF-κB co-activation; IL-6 surge begins; thyroid iodide sequestration starts. Patient may still appear stable. |
| 6 | Mid cascade | All 3 arms | Anaerobic glycolysis lowers blood pH; SpO2 worsens; self-reinforcing spiral accelerates. |
| 7 | Mid cascade | All 3 arms | Viral load surges via efficient Furin-mediated entry; HOI barrier gone; iodide reserves exhausted. |
| 8 | Cliff-edge | All 3 arms (peak) | Cytokine storm peaks; ARDS criteria met; cliff-edge crossed. |
| 9 | Irreversible | All 3 arms | Alveolar flooding; diffuse alveolar damage; loss of hypoxic vasoconstriction reflex; mechanical ventilation required; viral amplification loop irreversible. |
| 10 | Irreversible | All 3 arms | Multi-organ failure; coagulopathy; ICU escalation; high mortality risk. |
| Cascade Element | Finding That Would Falsify | What It Would Mean | Confirmatory Prediction |
| Arm 1 (DUOX/HOI) | HOI production does not decline at SpO2 < 94% | DUOX O2 dependency not clinically relevant at airway O2 tensions | DUOX H2O2 output falls measurably below SpO2 94% in airway epithelial cells |
| Arm 2 (Furin) | HIF-1α activation does not upregulate Furin at the 94% SpO2 threshold | Furin expression uncoupled from HIF-1α in respiratory epithelium during COVID-19 | Furin expression increases in COVID-19 patients as SpO2 falls below 94% |
| Arm 3 (Iodide) | Serum iodide does not fall during acute COVID-19 | D1/D2 suppression by IL-6 does not produce airway iodide depletion on a clinically relevant timescale | Serum/urinary iodide falls ≥30% within 7 days in severe COVID-19 patients with high IL-6 |
| Threshold (94% SpO2) | Severe COVID-19 occurs equally in patients with SpO2 consistently above 95% | The cascade has no SpO2 threshold and proceeds regardless of oxygenation | Threshold crossing at 94% correlates with abrupt deterioration in prospective cohort (Prediction 2) |
| Architecture (Multiplicative) | A single-arm model explains the cliff-edge as well as the three-arm framework | Multiplicative combination rule not necessary to explain outcome distributions | Additive model produces outcome distribution statistically indistinguishable from multiplicative across four populations |
| Iodide Protection | Japan’s COVID-19 mortality advantage disappears after controlling for age and healthcare access | Dietary iodide has no independent effect on COVID-19 severity | Urinary iodide ≥100 μg/L at admission not associated with longer time to cascade progression (Prediction 3) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




