Submitted:
03 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
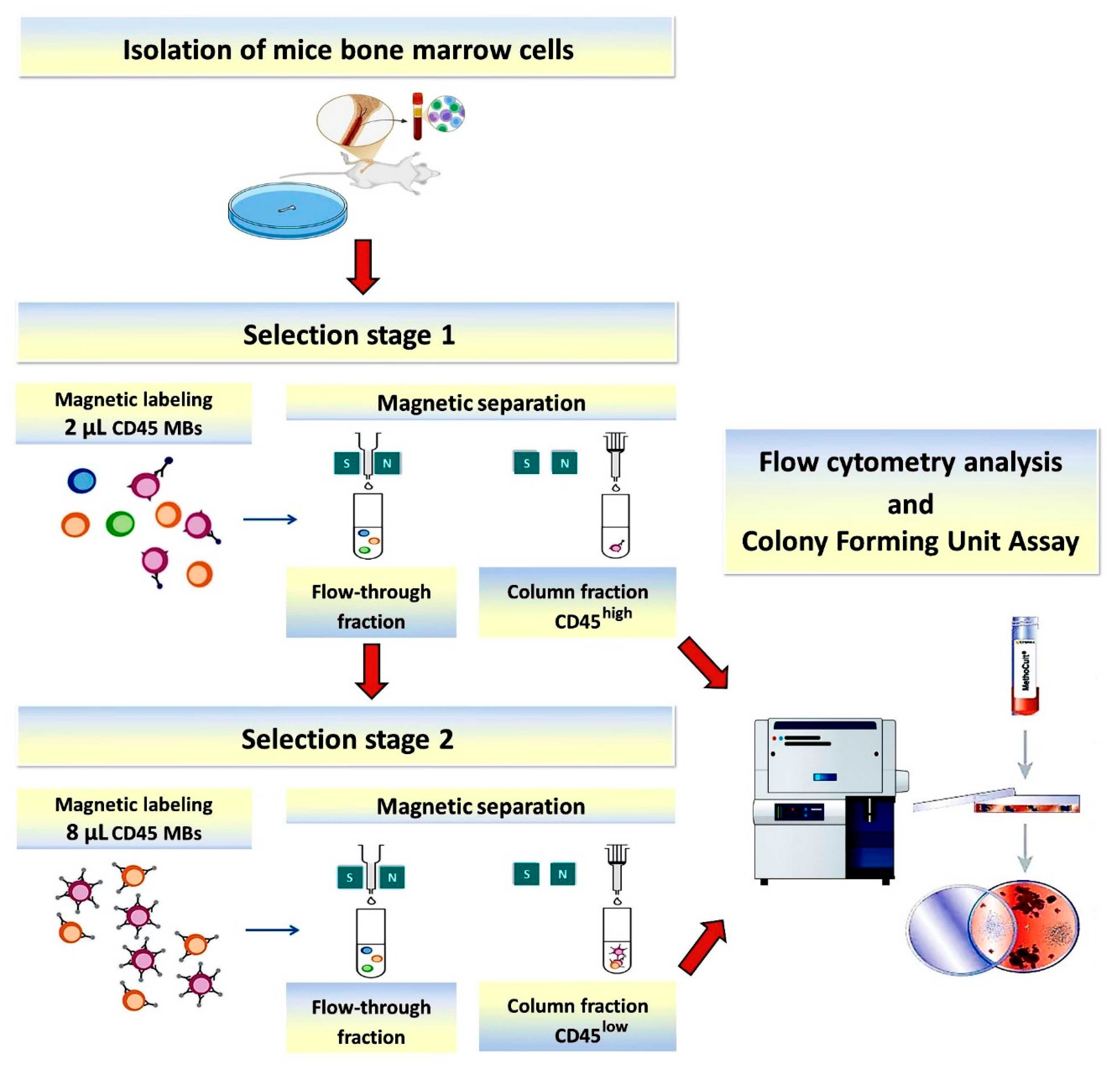
2.1. Isolation of Bone Marrow Cells
2.2. Two-Stage Magnetic Selection of CD45-Positive BM Cells (Basic Protocol)
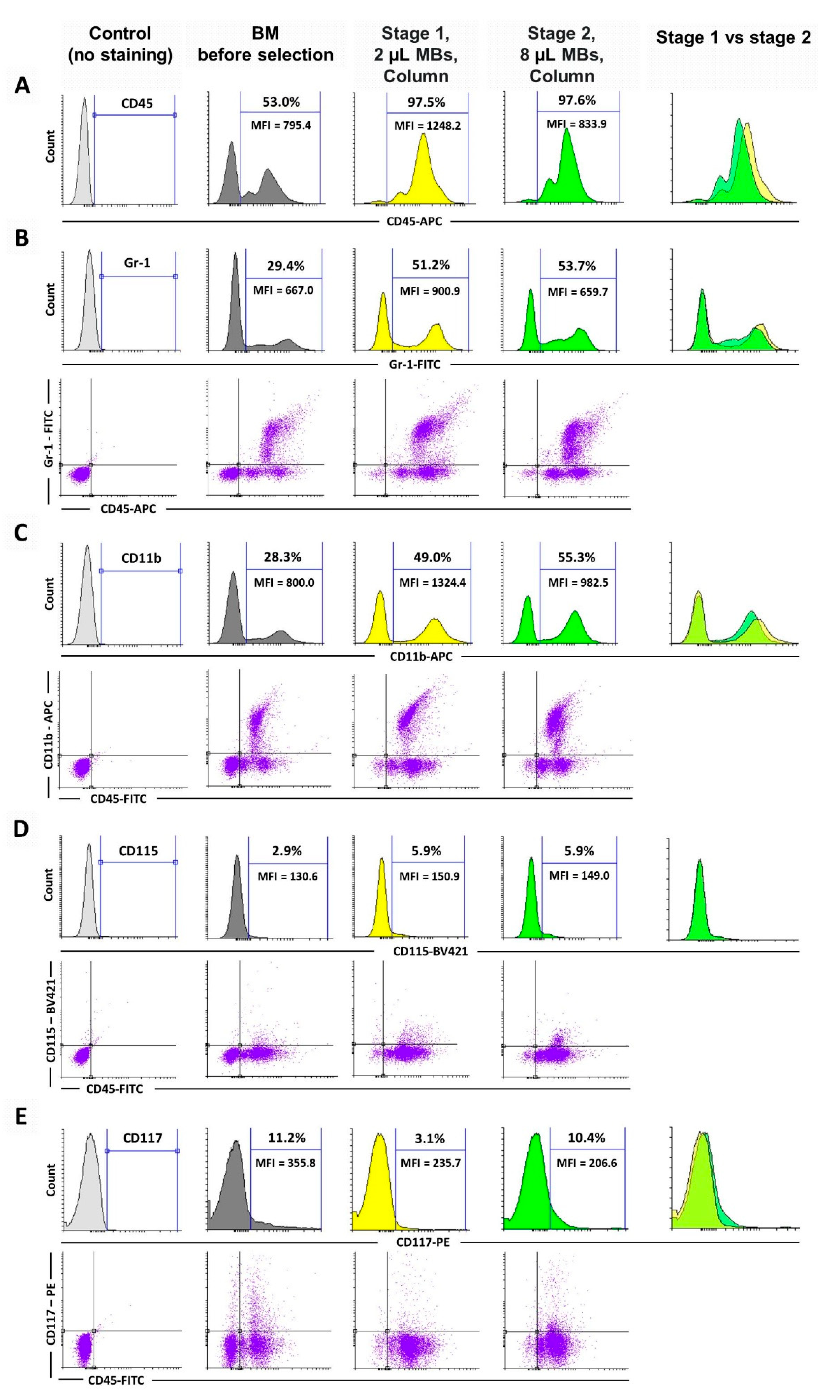
2.3. Flow Cytometry
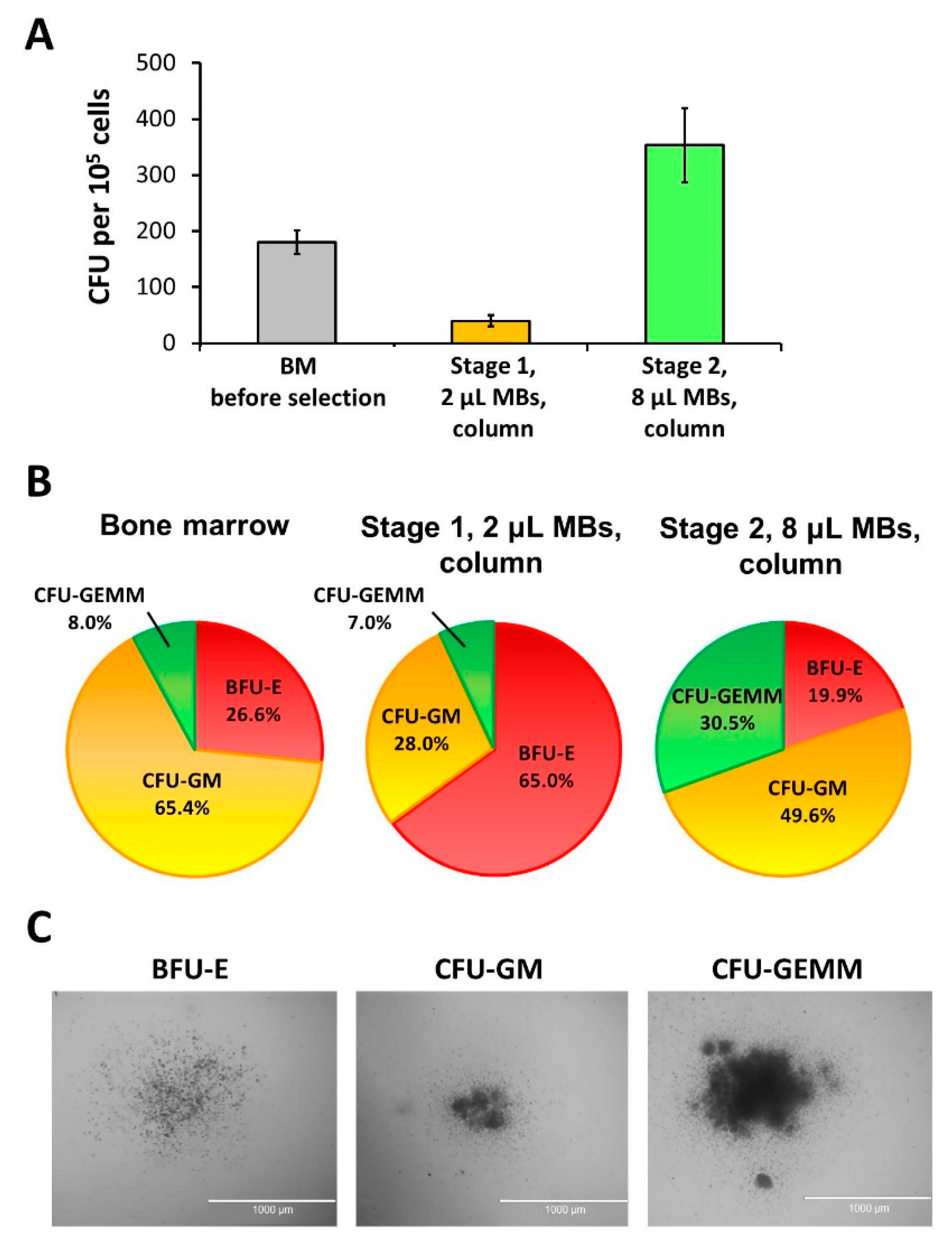
2.4. CFU Analysis
2.5. Statistical Analysis
3. Results
3.1. Basic Protocol: Two-Stage Magnetic Selection of BM Cells
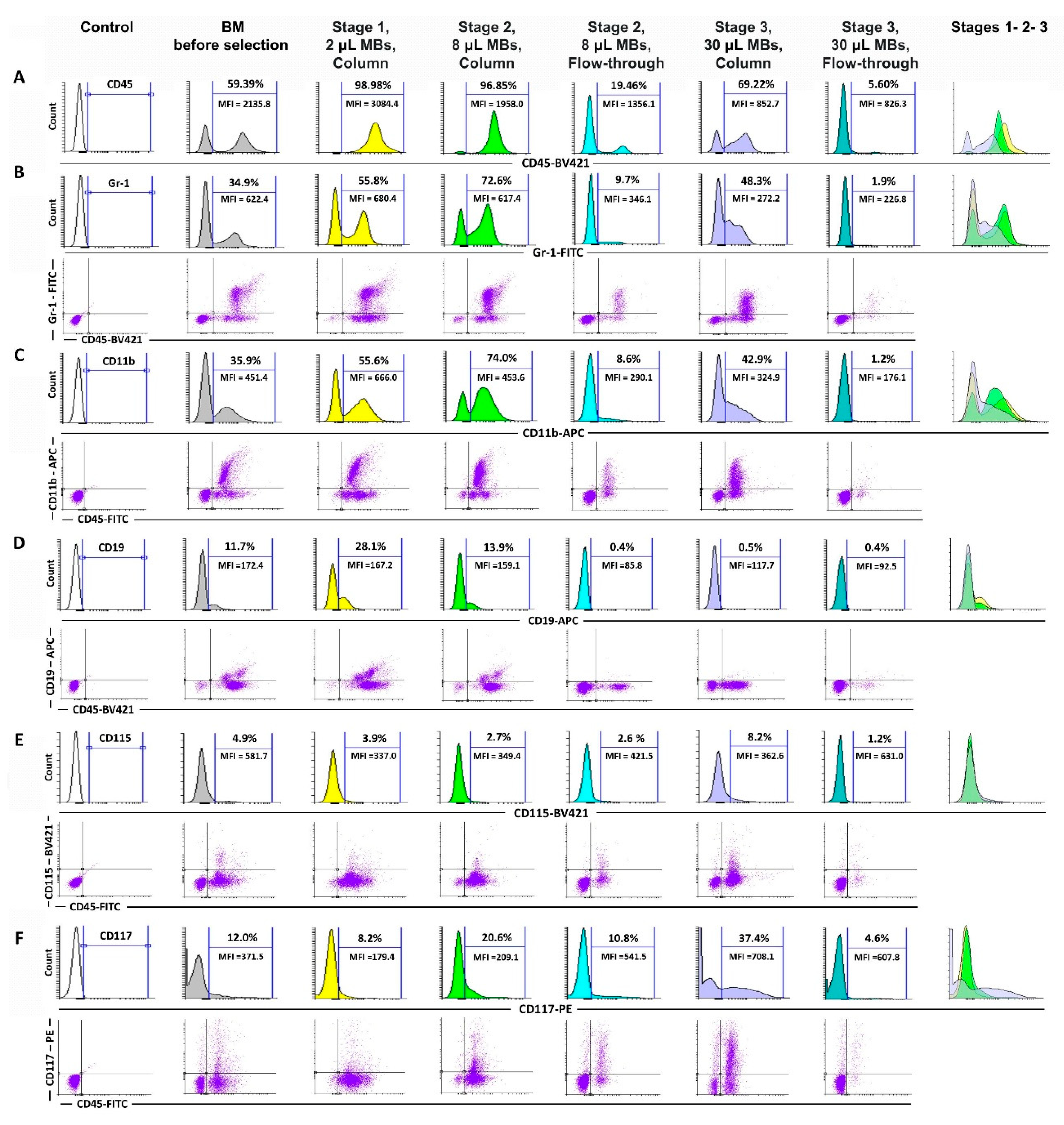
3.2. Basic Protocol: Immunophenotypic Study of the 1st and 2nd Stage Cell Fractions
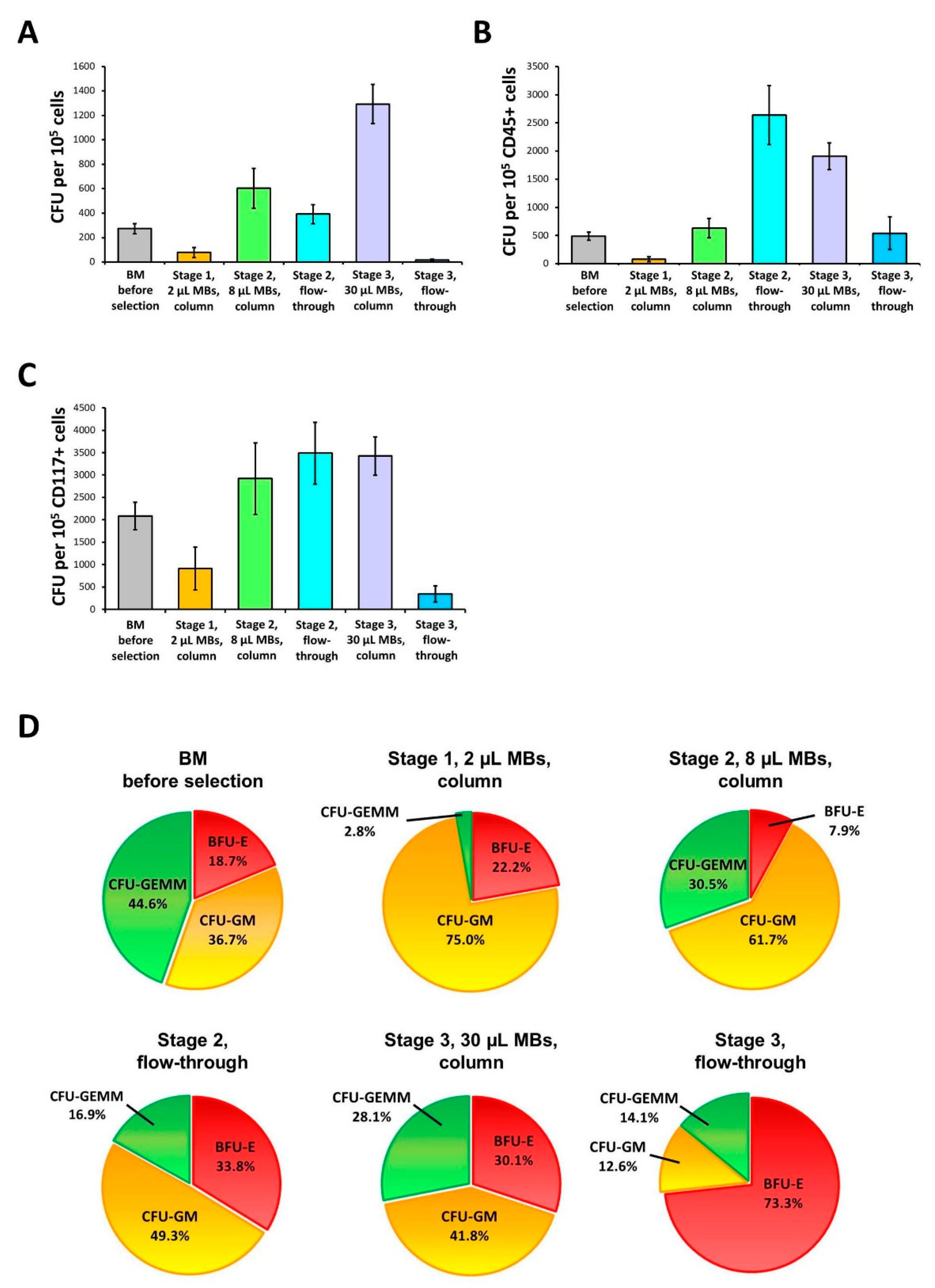
3.3. Basic Protocol: CFU Analysis of the 1st and 2nd Stage Cell Fractions
3.4. Extended Protocol: Immunophenotypic Study of CD45high and CD45low Cell Populations
3.5. Extended Protocol: CFU Analysis of Stage 1-3 Fractions
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ueda, Y.; Kondo, M.; Kelsoe, G. Inflammation and the reciprocal production of granulocytes and lymphocytes in bone marrow. J Exp Med. 2005, 201, 1771–80. [Google Scholar] [CrossRef]
- Osorio, D.; Yu, X.; Zhong, Y.; Li, G.; Yu, P.; Serpedin, E.; Huang, J.Z.; Cai, J.J. Single-Cell Expression Variability Implies Cell Function. Cells 2019, 9, 14. [Google Scholar] [CrossRef]
- Polyakova, N.; Kandarakov, O.; Belyavsky, A. Selection of Cell Populations with High or Low Surface Marker Expression Using Magnetic Sorting. Cells 2023, 12, 1286. [Google Scholar] [CrossRef]
- Pinho, S.; Frenette, P.S. Haematopoietic stem cell activity and interactions with the niche. Nat Rev Mol Cell Biol. 2019, 303–320. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, J.; Kokkaliaris, K.D. Bone marrow niches for hematopoietic stem cells: life span dynamics and adaptation to acute stress. Blood 2024, 144, 21–34. [Google Scholar] [CrossRef]
- Hermiston, M.L.; Xu, Z.; Weiss, A. CD45: a critical regulator of signaling thresholds in immune cells. Annu Rev Immunol. 2003, 21, 107–137. [Google Scholar] [CrossRef]
- Rheinländer, A.; Schraven, B.; Bommhardt, U. CD45 in human physiology and clinical medicine. Immunol Lett. 2018, 196, 22–32. [Google Scholar] [CrossRef]
- Garaudé, S.; Marone, R.; Lepore, R.; Devaux, A.; Beerlage, A.; Seyres, D.; Dell’ Aglio, A.; Juskevicius, D.; Zuin, J.; Burgold, T.; Wang, S.; Katta, V.; Manquen, G.; Li, Y.; Larrue, C.; Camus, A.; Durzynska, I.; Wellinger, L. C.; Kirby, I.; Van Berkel, P. H.; et al. Selective haematological cancer eradication with preserved haematopoiesis. Nature 2024, 630, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wang, X.; To, K.K.W.; Cui, C.; Luo, M.; Wu, S.; Huang, L.; Fu, K.; Pan, C.; Liu, Z.; Fan, T.; Yang, C.; Wang, F.; Fu, L. Circulating tumor cells shielded with extracellular vesicle-derived CD45 evade T cell attack to enable metastasis. Sig. Transduct. Target. Ther. 2024, 9, 84. [Google Scholar] [CrossRef]
- Salehi Farid, A.; Rowley, J.E.; Allen, H.H.; Kruger, I. G.; Tavakolpour, S.; Neeley, K.; Cong, M.; Shahbazian, H.; Dorafshani, N.; Berrada, A.; MacDonagh, A. C.; Padera, R. F.; Brugarolas, P.; Packard, A. B.; Rosenbaum, M. W.; Divakaran, S.; Di Carli, M. F.; Rashidian, M. CD45-PET is a robust, non-invasive tool for imaging inflammation. Nature 2025, 639, 214–224. [Google Scholar] [CrossRef]
- Nobuhisa, I.; Yamasaki, S.; Ramadan, A.; Taga, T. CD45(low)c-Kit(high) cells have hematopoietic properties in the mouse aorta-gonad-mesonephros region. Exp. Сell Res. 2012, 318, 705–715. [Google Scholar] [CrossRef]
- Shah, V.O.; Civin, C.I.; Loken, M.R. Flow cytometric analysis of human bone marrow. IV. Differential quantitative expression of T-200 common leukocyte antigen during normal hemopoiesis. J. Immunol. 1988, 140, 1861–1867. [Google Scholar] [CrossRef] [PubMed]
- Mayani, H.; Dragowska, W.; Lansdorp, P. M. Characterization of functionally distinct subpopulations of CD34+ cord blood cells in serum-free long-term cultures supplemented with hematopoietic cytokines. Blood 1993, 82, 2664–2672. [Google Scholar] [CrossRef] [PubMed]
- Boulais, P. E.; Mizoguchi, T.; Zimmerman, S.; Nakahara, F.; Vivié, J.; Mar, J. C.; van Oudenaarden, A.; Frenette, P. S. The Majority of CD45- Ter119- CD31- Bone Marrow Cell Fraction Is of Hematopoietic Origin and Contains Erythroid and Lymphoid Progenitors. Immunity 2018, 49, 627–639.e6. [Google Scholar] [CrossRef]
- Jackson, K. A.; Mi, T.; Goodell, M. A. Hematopoietic potential of stem cells isolated from murine skeletal muscle. Proc. Natl. Acad. Sci. U S A. 1999, 96, 14482–14486. [Google Scholar] [CrossRef]
- Issarachai, S.; Priestley, G. V.; Nakamoto, B.; Papayannopoulou, T. Cells with hemopoietic potential residing in muscle are itinerant bone marrow-derived cells. Exp. Hematol. 2002, 30, 366–373. [Google Scholar] [CrossRef]
- Cuthbert, R. J.; Giannoudis, P. V.; Wang, X. N.; Nicholson, L.; Pawson, D.; Lubenko, A.; Tan, H. B.; Dickinson, A.; McGonagle, D.; Jones, E. Examining the feasibility of clinical grade CD271+ enrichment of mesenchymal stromal cells for bone regeneration. PLoS One 2015, 10, e0117855. [Google Scholar] [CrossRef]
- Deschaseaux, F.; Gindraux, F.; Saadi, R.; Obert, L.; Chalmers, D.; Herve, P. Direct selection of human bone marrow mesenchymal stem cells using an anti-CD49a antibody reveals their CD45med,low phenotype. Br. J. Haematol. 2003, 122, 506–517. [Google Scholar] [CrossRef]
- Li, H.; Ghazanfari, R.; Zacharaki, D.; Ditzel, N.; Isern, J.; Ekblom, M.; Méndez-Ferrer, S.; Kassem, M.; Scheding, S. Low/negative expression of PDGFR-α identifies the candidate primary mesenchymal stromal cells in adult human bone marrow. Stem Cell Reports 2014, 3, 965–74. [Google Scholar] [CrossRef]
- Yeh, S. P.; Chang, J. G.; Lo, W. J.; Liaw, Y. C.; Lin, C. L.; Lee, C. C.; Chiu, C. F. Induction of CD45 expression on bone marrow-derived mesenchymal stem cells. Leukemia 2006, 20, 894–6. [Google Scholar] [CrossRef]
- Waskow, C.; Paul, S.; Haller, C.; Gassmann, M.; Rodewald, H.R. Viable c-Kit(W/W) mutants reveal pivotal role for c-kit in the maintenance of lymphopoiesis. Immunity 2002, 17, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Edling, C. E.; Hallberg, B. c-Kit--a hematopoietic cell essential receptor tyrosine kinase. Int. J. Biochem. Cell Biol. 2007, 39, 1995–1998. [Google Scholar] [CrossRef]
- Bueno, C.; Barrera, S.; Bataller, A.; Ortiz-Maldonado, V.; Elliot, N.; O’Byrne, S.; Wang, G.; Rovira, M.; Gutierrez-Agüera, F.; Trincado, J. L.; González-González, M.; et al. CD34+CD19-CD22+ B-cell progenitors may underlie phenotypic escape in patients treated with CD19-directed therapies. Blood 2022, 140, 38–44. [Google Scholar] [CrossRef]
- El-Jawhari, J.J.; Cuthbert, R.; McGonagle, D.; Jones, E.; Giannoudis, P.V. The CD45lowCD271high Cell Prevalence in Bone Marrow Samples May Provide a Useful Measurement of the Bone Marrow Quality for Cartilage and Bone Regenerative Therapy. J. Bone Joint Surg. Am. 2017, 99, 1305–1313. [Google Scholar] [CrossRef]
- Passweg, J.R.; Baldomero, H.; Ciceri, F.; Corbacioglu, S.; de la Cámara, R.; Dolstra, H.; Glass, B.; Greco, R.; McLornan, D.P.; Neven, B.; de Latour, R.P.; Perić, Z.; Ruggeri, A.; Snowden, J.A.; Sureda, A. Hematopoietic cell transplantation and cellular therapies in Europe 2021. The second year of the SARS-CoV-2 pandemic. A Report from the EBMT Activity Survey. Bone Marrow Transplant. 2023, 58, 647–658. [Google Scholar] [CrossRef]
- Okalova, J.; Spencer, T.H.; Chandrakasan, S. Next generation targeted non-genotoxic conditioning for hematopoietic stem cell and hematopoietic stem cell-based gene therapy. Front. Immunol. 2025, 16, 1653344. [Google Scholar] [CrossRef]
- Dahlke, M.H.; Larsen, S.R.; Rasko, J.E.; Schlitt, H.J. The biology of CD45 and its use as a therapeutic target. Leuk. Lymphoma 2004, 45, 229–236. [Google Scholar] [CrossRef]
- Forghani, P.; Harris, W.; Li, j.-M.; Khorramizadeh, M.R.; Waller, E. CD11b+ gr-1+ myeloid Derived Suppressor Cells (MDSC) In Normal Bone Marrow Suppress T Cell Proliferation and Enhance T-Regulatory Function Via a IL10-, IL4- and IDO-Independent Mechanism. Blood 2010, 116, 4801. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, T.; Shao, S.; Shi, B.; Zhao, Y. Phenotype, development, and biological function of myeloid-derived suppressor cells. Oncoimmunology 2015, 5, e1004983. [Google Scholar] [CrossRef]
- Gunji, Y.; Nakamura, M.; Osawa, H.; Nagayoshi, K.; Nakauchi, H.; Miura, Y.; Yanagisawa, M.; Suda, T. Human primitive hematopoietic progenitor cells are more enriched in KITlow cells than in KIThigh cells. Blood 1993, 82, 3283–3289. [Google Scholar] [CrossRef] [PubMed]
- Vašíček, J.; Baláži, A.; Bauer, M.; Svoradová, A.; Tirpáková, M.; Ondruška, Ľ.; Parkányi, V.; V. Makarevich, A.; Chrenek, P. Enrichment of Rabbit Primitive Hematopoietic Cells via MACS Depletion of CD45+ Bone Marrow Cells. Magnetochemistry 2021, 7, 11. [Google Scholar] [CrossRef]
- Willasch, A.; Eing, S.; Weber, G.; Kuçi, S.; Schneider, G.; Soerensen, J.; Jarisch, A.; Rettinger, E.; Koehl, U.; Klingebiel, T.; Kreyenberg, H.; Bader, P. Enrichment of cell subpopulations applying automated MACS technique: purity, recovery and applicability for PCR-based chimerism analysis. Bone Marrow Transplant. 2010, 45, 181–189. [Google Scholar] [CrossRef] [PubMed]
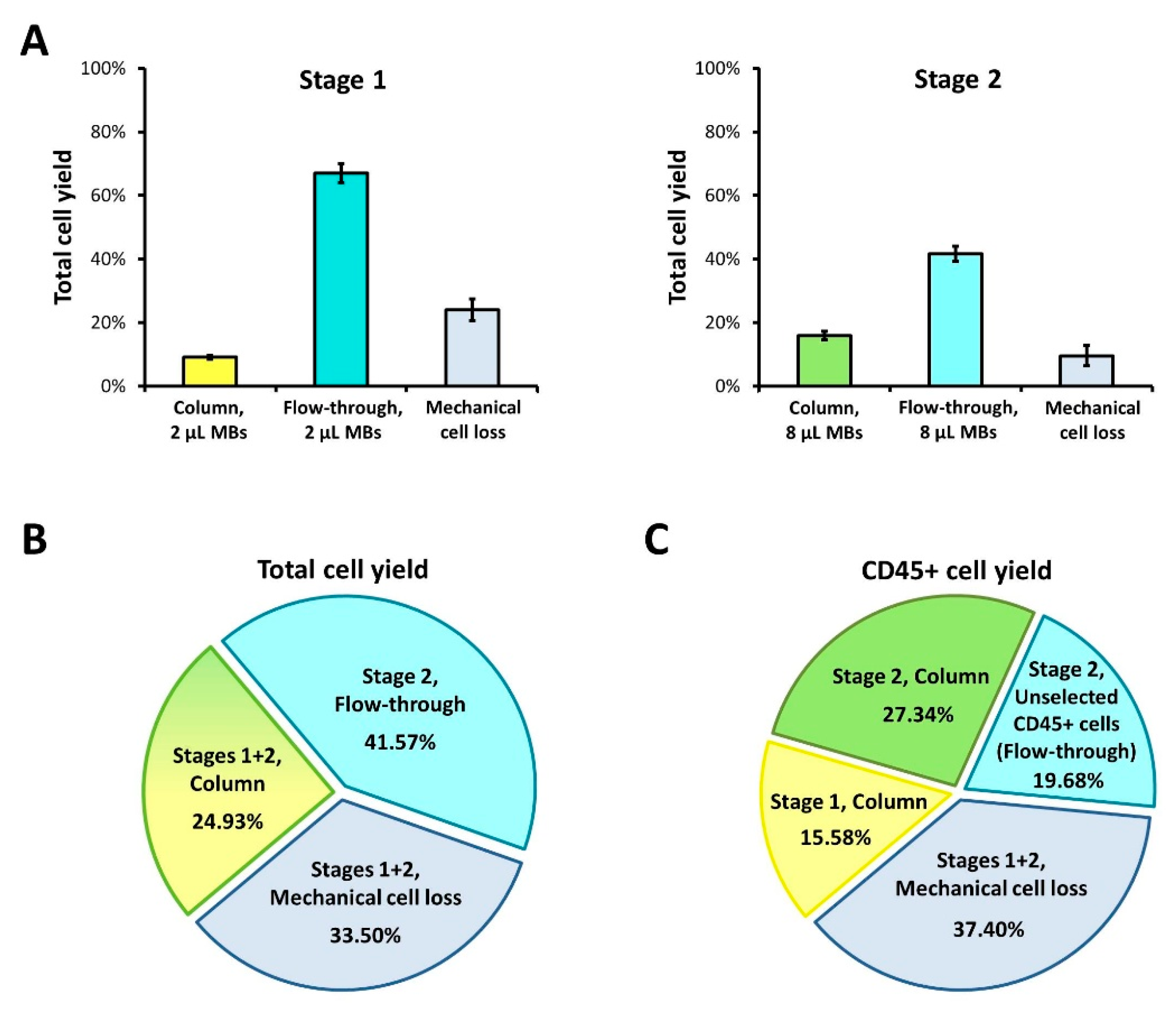
| Stage | Fraction | Total cell yield | |
|---|---|---|---|
| Absolute cell yield | Relative cell yield | ||
| BM | Starting cell number | 100% (2.5*107 cells) | |
| Stage 1 | Column | 14.32% | 14.32% |
| Flow-through | 62.80% | 62.80% | |
| Mechanical loss | 22.88% | 22.88% | |
| Stage 2 | Column | 20.10% | 32.01% |
| Flow-through | 30.66% | 48.82% | |
| Mechanical loss | 12.04% | 19.17% | |
| Stage 3 | Column | 2.76% | 9.02% |
| Flow-through | 24.08% | 78.54% | |
| Mechanical loss | 3.82% | 12.44% | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




