Submitted:
03 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
2.1. Catalyst Preparation
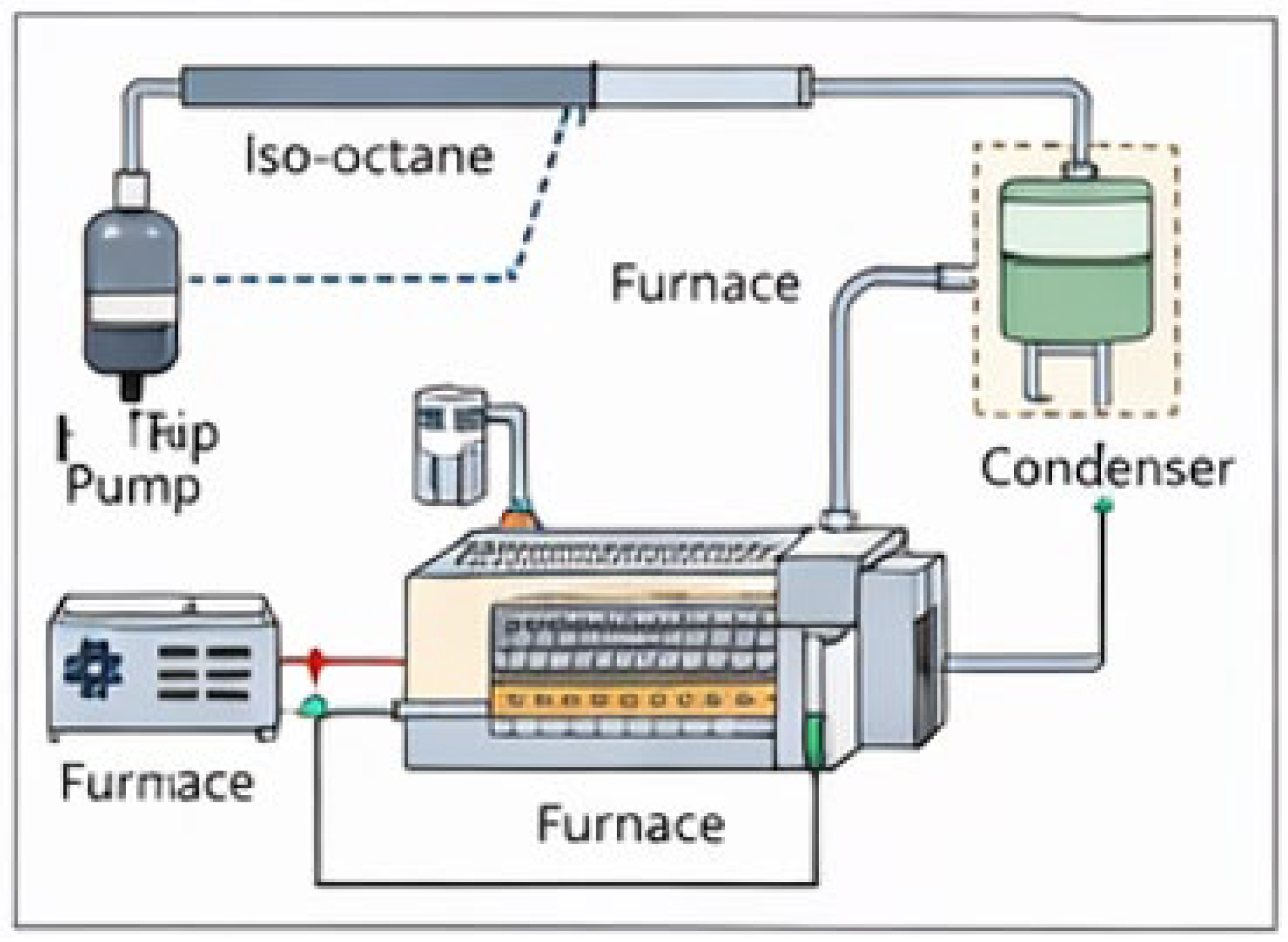
2.2. Catalytic Reforming Experiments
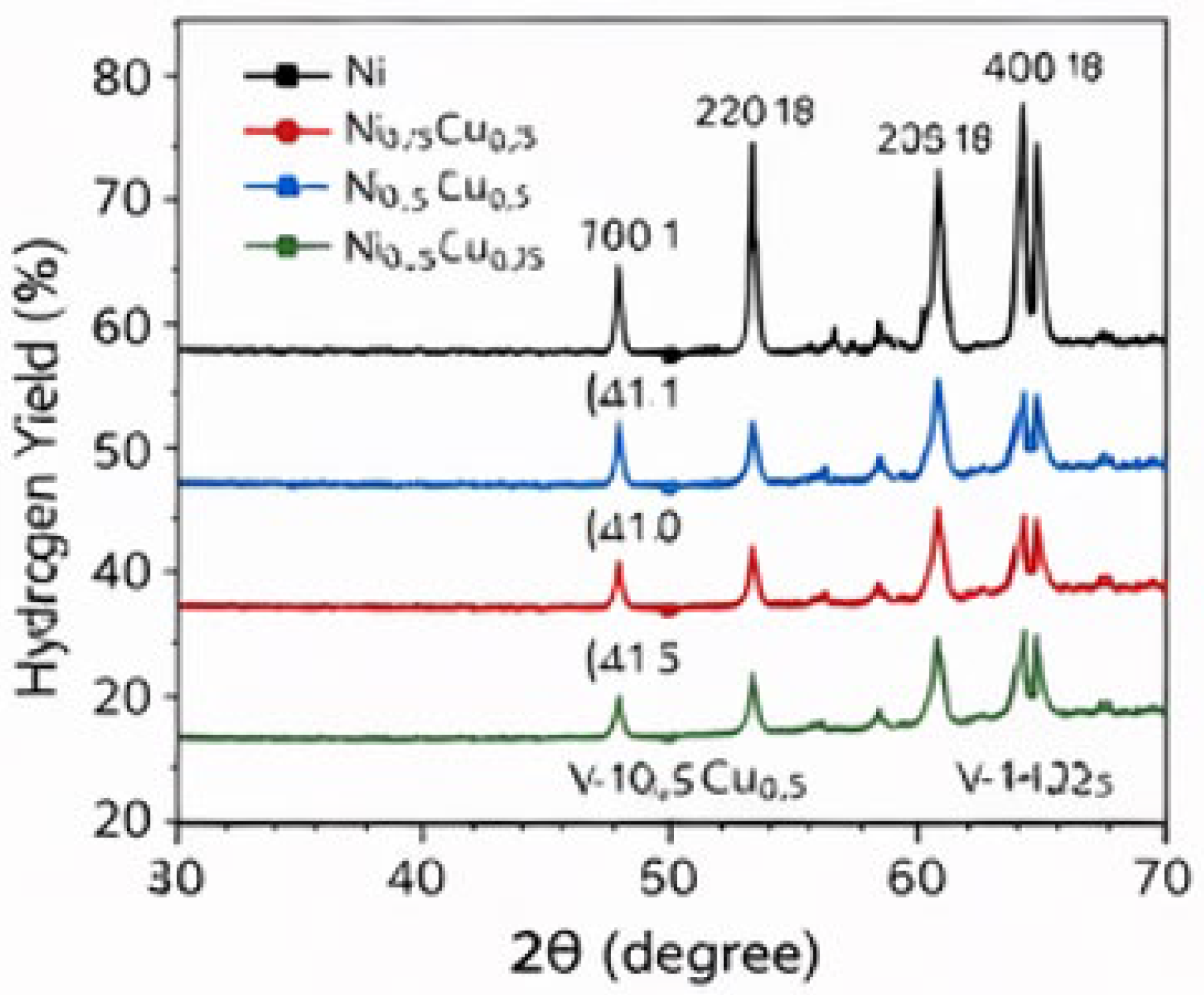
2.3. Catalyst Characterization
3. Results and Discussion
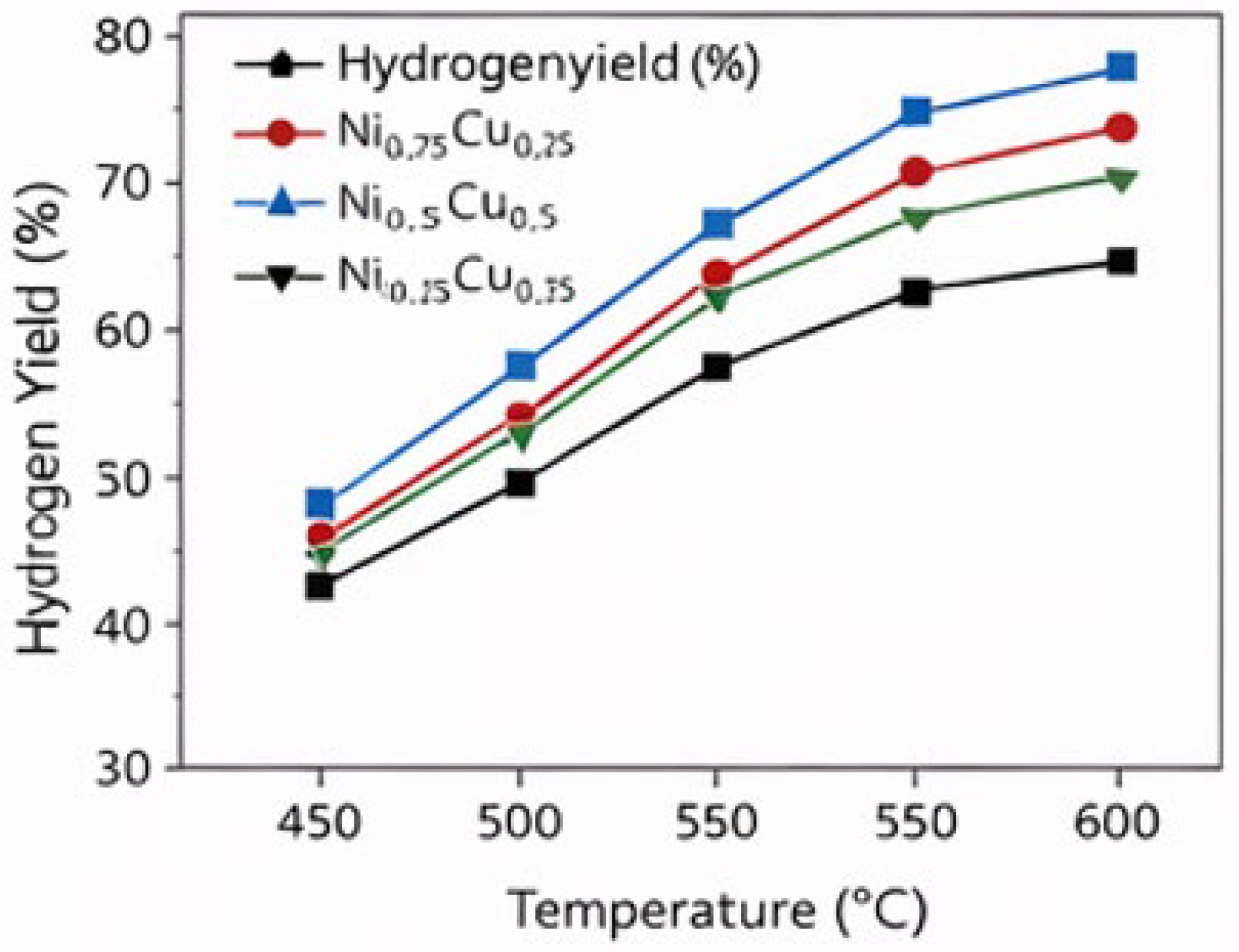
3.1. Effect of Temperature on Hydrogen Production
3.2. Influence of Ni–Cu Composition
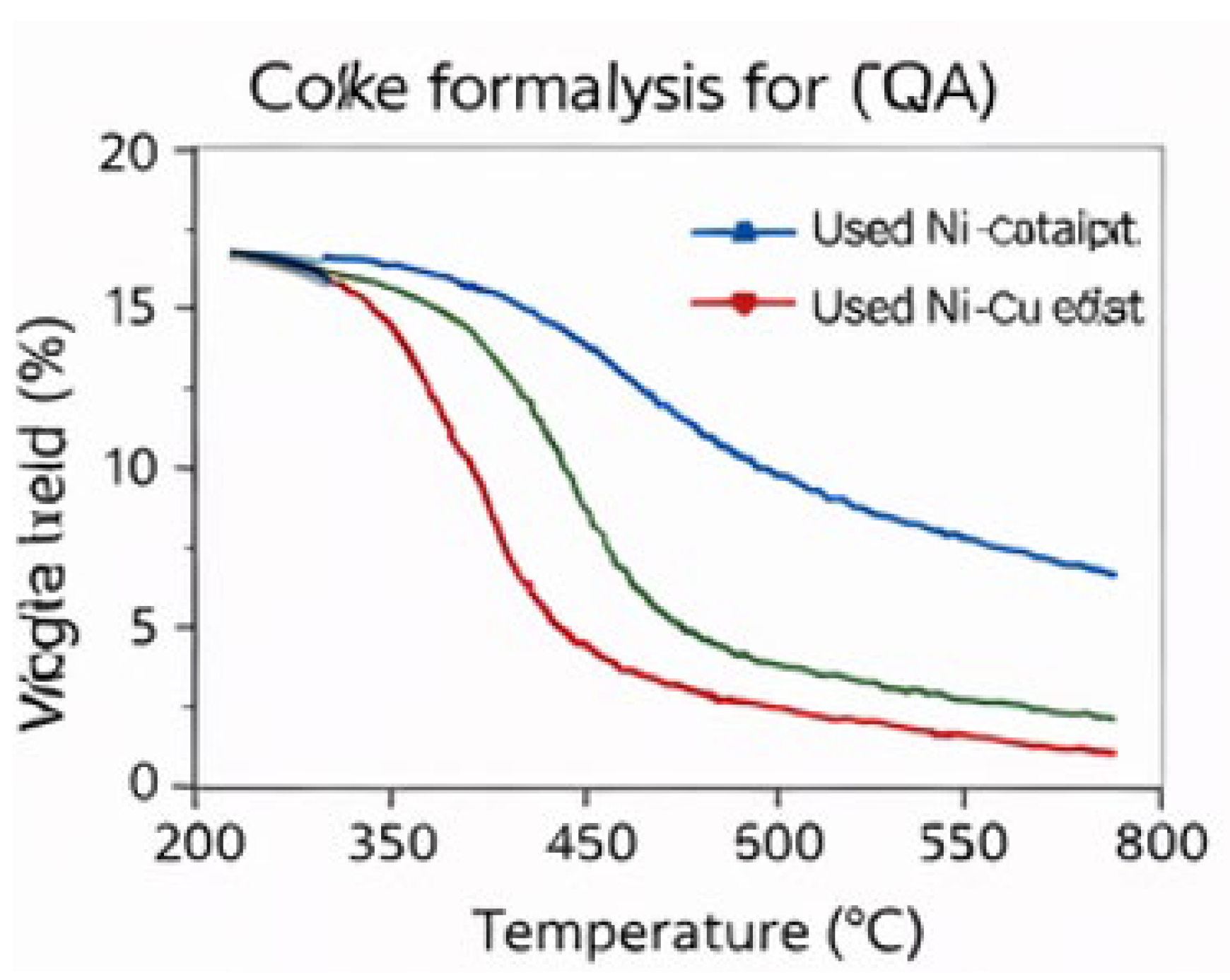
3.3. Catalyst Stability and Coke Suppression
4. Conclusions
References
- Zhang Y. et al. (2022). Hydrogen production via hydrocarbon steam reforming over bimetallic catalysts. International Journal of Hydrogen Energy, 47, 15678–15690.
- Song, C. Fuel processing for low-temperature and high-temperature fuel cells. Catalysis Today, 2002, 77, 17–49. [CrossRef]
- Armor J. N. (1999). The multiple roles for catalysis in the production of H2. Applied Catalysis A, 176, 159-176.
- Rostrup-Nielsen, J.R. Production of synthesis gas. Catalysis Today, 2000, 63, 159–164. [CrossRef]
- Rostrup-Nielsen, J.R.; Sehested, J.; Nørskov, J.K. Hydrogen and synthesis gas by steam- and CO2 reforming. Advances in Catalysis, 2002, 47, 65–139.
- Wang L. et al. (2021). Catalytic performance of Ni–Cu catalysts for hydrogen production from hydrocarbon reforming. Fuel, 292, 120309.
- Rostrup-Nielsen J. (2002). Catalytic steam reforming. Catalysis Today, 63, 159-164.
- Sehested J. (2006). Four challenges for nickel steam-reforming catalysts. Catalysis Today, 111, 103-110. [CrossRef]
- Bartholomew C. H. (2001). Mechanisms of catalyst deactivation. Applied Catalysis A, 212, 17-60.
- Sehested, J. Four challenges for nickel steam-reforming catalysts. Catalysis Today, 2006, 111, 103–110. [CrossRef]
- Li X. et al. (2023). Advances in nickel-based catalysts for hydrogen production. Applied Catalysis A: General, 651, 118896.
- Trimm D. L. (1999). Coke formation and minimisation during steam reforming reactions. Catalysis Today, 49, 3-10. [CrossRef]
- Trimm, D.L. Coke formation and minimisation during steam reforming reactions. Catalysis Today, 1997, 37, 233–238. [CrossRef]
- Ashok J. et al. (2015). Bimetallic Ni catalysts for reforming reactions. Fuel Processing Technology, 134, 341-350.
- Liu, D.; Quek, X.Y.; Cheo, W.N.E.; Lau, R.; Borgna, A.; Yang, Y. MCM-41 supported nickel catalysts for hydrogen production from steam reforming of hydrocarbons. Applied Catalysis B: Environmental, 2009, 92, 385–392.
- Song H., Ozkan U. (2010). Effects of Cu addition in Ni catalysts. Journal of Catalysis, 261, 66-74.
- Hou Z., Hughes R. (2001). Hydrogen production by methane reforming. International Journal of Hydrogen Energy, 26, 849-856.
- Busca G. (2014). Alumina-supported catalysts. Catalysis Today, 226, 2-13.
- Rostrup-Nielsen J. R. (2004). Hydrogen and synthesis gas. Catalysis Today, 106, 293-296.
- Xu, J.; Froment, G.F. Methane steam reforming, methanation and water–gas shift: I. Intrinsic kinetics. AIChE Journal, 1989, 35, 88–96.
- Chen Y. et al. (2022). Bimetallic catalysts for hydrocarbon reforming reactions. Catalysis Today, 389, 45–56.
- Chen D. et al. (2008). Carbon deposition in hydrocarbon reforming. Applied Catalysis A, 346, 1-8.
- Singh R. et al. (2021). Catalytic hydrogen production from hydrocarbon fuels. Renewable Energy, 178, 735–748.
- Ruckenstein, E.; Wang, H.Y. Carbon deposition and catalytic deactivation during CO2 reforming of methane over Ni catalysts. Applied Catalysis A: General, 2000, 204, 257–263.
- Bradford, M.C.J.; Vannice, M.A. CO2 reforming of CH4 over supported Ni catalysts. Catalysis Reviews, 1999, 41, 1–42.


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



