Submitted:
06 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
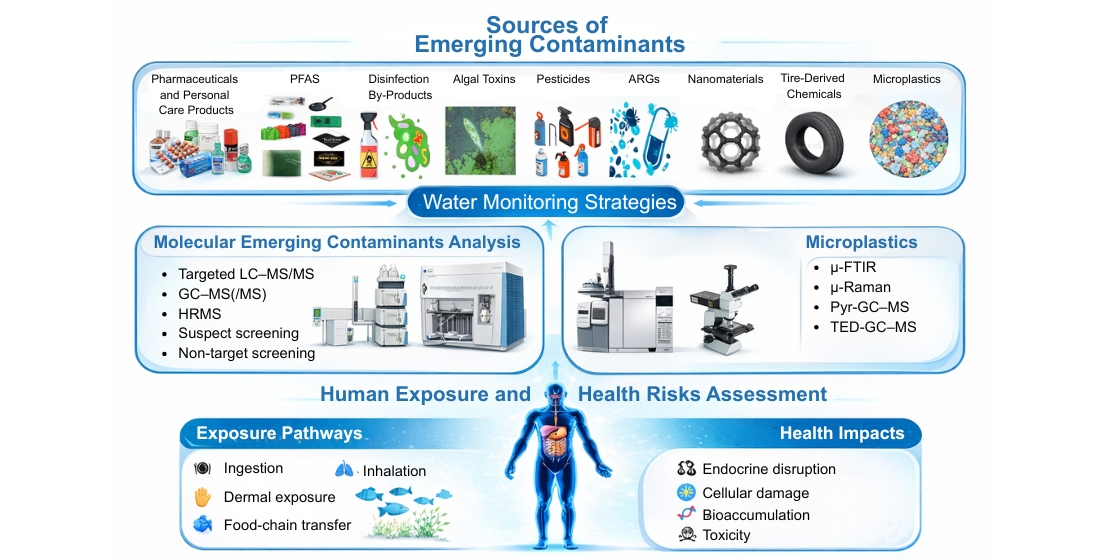
Keywords:
1. Introduction
2. Occurrence and Sources of Emerging Contaminants and Microplastics in Water Systems
2.1. Molecular Emerging Contaminants
2.2. Particulate Emerging Contaminants (Microplastics)
3. Sampling and Sample Preparation Strategies
3.1. Sampling Considerations for Water Matrices
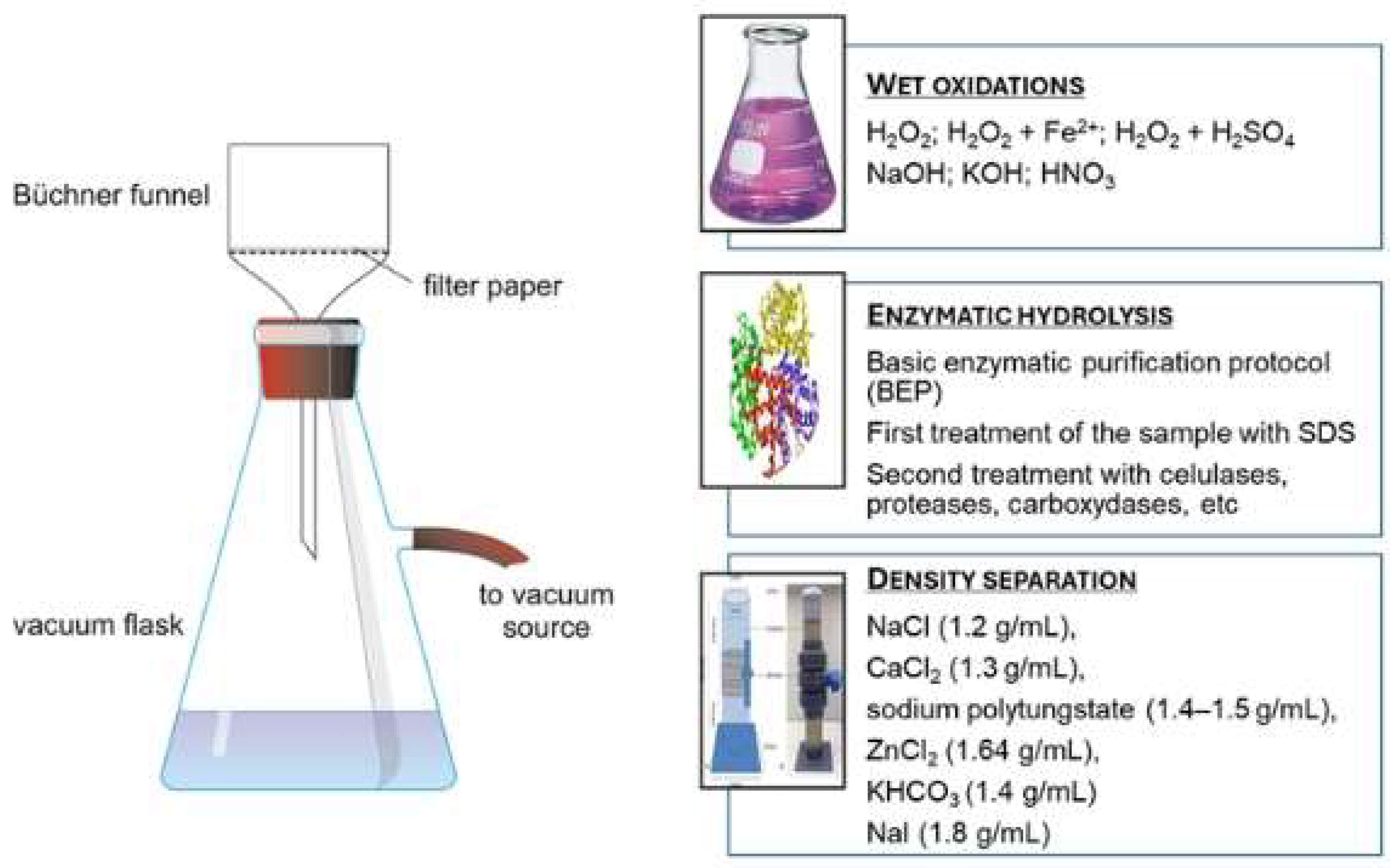
3.2. Sample Preparation for Molecular Emerging Contaminants
3.3. Sample Preparation for Particulate Emerging Contaminants (Microplastics)
4. Analytical Strategies for Molecular Emerging Contaminants
4.1. Targeted LC-MS/MS Approaches
4.2. High-Resolution Mass Spectrometry (HRMS)
4.3. Non-Target and Suspect Screening Approaches
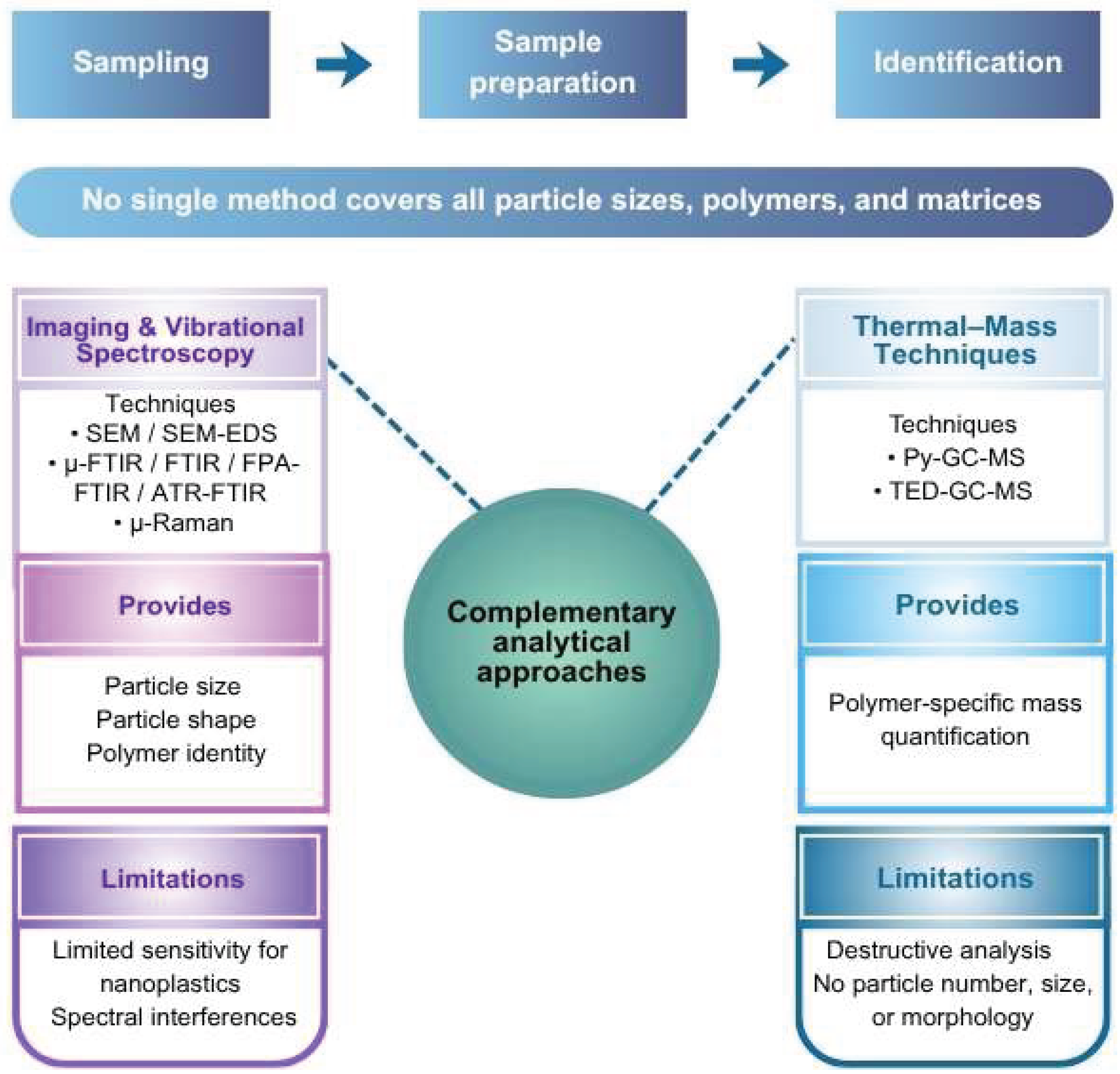
5. Analytical Strategies for Particulate Emerging Contaminants (Microplastics)
Spectroscopic and Thermal–Mass-Based Techniques
6. Environmental and Human Health Risk Assessment
6.1. Environmental Risks of Molecular Emerging Contaminants
6.2. Human Health Risks
6.3. Risks Associated with Microplastics
7. Knowledge Gaps, Analytical Challenges, and Future Perspectives
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mallek, M.; Barceló, D. Assessment of Removal Technologies for Microplastics in Surface Waters and Wastewaters. Curr. Opin. Chem. Eng. 2025, 49, 101170. [Google Scholar] [CrossRef]
- Mallek, M.; Barceló, D. Advancing Water Treatment and Reuse Technologies to Address the Nexus of Climate Change, Water Scarcity, and Pharmaceutical Contamination. J. Environ. Chem. Eng. 2025, 13, 119284. [Google Scholar] [CrossRef]
- Li, X.; Shen, X.; Jiang, W.; Xi, Y.; Li, S. Comprehensive Review of Emerging Contaminants: Detection Technologies, Environmental Impact, and Management Strategies. Ecotoxicol. Environ. Saf. 2024, 278, 116420. [Google Scholar] [CrossRef]
- Chen, H.; Xie, J.; Huang, C.; Liang, Y.; Zhang, Y.; Zhao, X.; Ling, Y.; Wang, L.; Zheng, Q.; Yang, X. Database and Review of Disinfection By-Products since 1974: Constituent Elements, Molecular Weights, and Structures. J. Hazard. Mater. 2024, 462, 132792. [Google Scholar] [CrossRef]
- Mallek, M.; Barceló, D. Analysis and Fate of Per- and Polyfluoroalkyl Substances (PFAS) in the Global Aquatic Environment: Perspectives and Combined Risks with Microplastics. Anal. Bioanal. Chem. 2025. [Google Scholar] [CrossRef]
- Du, J.; Xu, J.; Luo, Y.; Li, X.; Zhao, L.; Liu, S.; Jia, X.; Wang, Z.; Ge, L.; Cui, K.; et al. High-Throughput Monitoring of 323 Pharmaceuticals and Personal Care Products (PPCPs) and Pesticides in Surface Water for Environmental Risk Assessment. Environ. Sci. Technol. 2025, 59, 11275–11285. [Google Scholar] [CrossRef]
- Sahai, H.; Hernando, M.D.; Martínez Bueno, M.J.; Aguilera del Real, A.M.; Fernández-Alba, A.R. Evaluation of the Sorption/Desorption Processes of Pesticides in Biodegradable Mulch Films Used in Agriculture. Chemosphere 2024, 351, 141183. [Google Scholar] [CrossRef]
- Kazmi, S.S.U.H.; Xu, Q.; Tayyab, M.; Pastorino, P.; Barceló, D.; Yaseen, Z.M.; Khan, Z.H.; Li, G. Navigating the Environmental Dynamics, Toxicity to Aquatic Organisms and Human Associated Risks of an Emerging Tire Wear Contaminant 6PPD-Quinone. Environ. Pollut. 2024, 356, 124313. [Google Scholar] [CrossRef]
- Liao, C.; Kim, U. A Review of Environmental Occurrence, Fate, Exposure, and Toxicity of Benzothiazoles. Environ. Sci. Technol. 2018, 52, 5007–5026. [Google Scholar] [CrossRef] [PubMed]
- Rapp-Wright, H.; Rodríguez-Mozaz, S.; Alvarez-Muñoz, D.; Barceló, D.; Regan, F.; Barron, L.; White, B. International Comparison, Risk Assessment, and Prioritisation of 26 Endocrine Disrupting Compounds in Three European River Catchments in the UK, Ireland, and Spain. Molecules 2023, 28, 5994. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Liu, S.; Zhang, T.; Liu, Q.; Alvarez, P.; Chen, W. Current Methods and Prospects for Analysis and Characterization of Nanomaterials in the Environment. Environ. Sci. Technol. 2022, 56, 7426–7447. [Google Scholar] [CrossRef]
- Dong, H.; Aziz, Md.T.; Richardson, S. Transformation of Algal Toxins during the Oxidation/Disinfection Processes of Drinking Water: From Structure to Toxicity. Environ. Sci. Technol. 2023, 57, 12944–12957. [Google Scholar] [CrossRef]
- Jaén-Gil, A.; Tisserand, A.; Santos, L.; Rodríguez-Mozaz, S.; Gomiero, A.; Langeland, E.; Khan, F. Legacy of Chemical Pollution from an Underwater Tire Dump in Alver Municipality, Norway: Implication for the Persistence of Tire-Derived Chemicals and Site Remediation. Environments 2025, 12, 356. [Google Scholar] [CrossRef]
- Qaiser, Z.; Aqeel, M.; Sarfraz, W.; Fatima Rizvi, Z.; Noman, A.; Naeem, S.; Khalid, N. Microplastics in Wastewaters and Their Potential Effects on Aquatic and Terrestrial Biota. Case Stud. Chem. Environ. Eng. 2023, 8, 100536. [Google Scholar] [CrossRef]
- González Fernández, D.; Cózar, A.; Hanke, G.; Viejo, J.; Morales-Caselles, C.; Bakiu, R.; Barceló, D.; Bessa, F.; Bruge, A.; Cabrera Fernández, M.; et al. Floating Macrolitter Leaked from Europe into the Ocean. Nat. Sustain. 2021, 4, 474–483. [Google Scholar] [CrossRef]
- Johannessen, C.; Helm, P.; Lashuk, B.; Yargeau, V.; Metcalfe, C.D. The Tire Wear Compounds 6PPD-Quinone and 1,3-Diphenylguanidine in an Urban Watershed. Arch. Environ. Contam. Toxicol. 2022, 82, 171–179. [Google Scholar] [CrossRef]
- Nam, WWF-Viet. Report on Plastic Waste Generation in 2022; Thanh Nien Publishing House: Ha Noi, Viet Nam, 2023. [Google Scholar]
- Li, J.; Aziz, Md.T.; Granger, C.O.; Richardson, S.D. Halocyclopentadienes: An Emerging Class of Toxic DBPs in Chlor(Am)Inated Drinking Water. Environ. Sci. Technol. 2022, 56, 11387–11397. [Google Scholar] [CrossRef]
- Yu, L.; Lin, Y.; Li, J.; Deng, C.; Zhang, R.; Liu, A.; Wang, L.; Li, Y.; Wei, X.; Lu, D.; et al. Suspect Screening of Pharmaceuticals and Their Transformation Products (TPs) in Wastewater during COVID-19 Infection Peak: Identification of New TPs and Elevated Risks. Environ. Sci. Technol. 2025, 59, 4893–4905. [Google Scholar] [CrossRef]
- Wang, C.; Guo, R.; Guo, C.; Yin, H.; Xu, J. Photodegradation of Typical Psychotropic Drugs in the Aquatic Environment: A Critical Review. Environ. Sci. Process. Impacts 2025, 27, 320–354. [Google Scholar] [CrossRef]
- Boahen, E.; Owusu, L.; Adjei-Anim, S. A Comprehensive Review of Emerging Environmental Contaminants of Global Concern. Discover Environment 2025, 3, 144. [Google Scholar] [CrossRef]
- Ma, W.; Li, F.; Tian, Y.; Xu, H.; Luo, J.; Shi, D.; Dai, J.; Tang, J.; Pan, Y. Simultaneous Analysis of Ultrashort- to Long-Chain PFAS in Multisalinity Aquatic Systems: Methodology, Spatial Profiling, and Risk Prioritization. Environ. Sci. Technol. 2025, 59, 26770–26780. [Google Scholar] [CrossRef]
- Cao, G.; Wang, W.; Zhang, J.; Wu, P.; Qiao, H.; Li, H.; Huang, G.; Yang, Z.; Cai, Z. Occurrence and Fate of Substituted p-Phenylenediamine-Derived Quinones in Hong Kong Wastewater Treatment Plants. Environ. Sci. Technol. 2023, 57, 15635–15643. [Google Scholar] [CrossRef]
- Wang, X.; Yang, J.; Jiao, Z.; Liang, J.; Chen, Y.; Gao, Q.; Shi, W.; Zhang, X.; Yu, H.; Wei, S.; et al. Uncovering Overlooked Fluorinated Compounds by Multi-Strategy Nontarget Screening in an Urban River System in China. Environ. Sci. Technol. 2026, 60, 3567–3579. [Google Scholar] [CrossRef]
- Wang, W.; Cao, G.; Zhang, J.; Chang, W.; Sang, Y.; Cai, Z. Fragmentation Pattern-Based Screening Strategy Combining Diagnostic Ion and Neutral Loss Uncovered Novel p-Phenylenediamine Quinone Contaminants in the Environment. Environ. Sci. Technol. 2024, 58, 5921–5931. [Google Scholar] [CrossRef]
- Qian, Y.; Ke, Y.; Wang, L.; Yu, N.; He, Y.; Yu, Q.; Wei, S.; Ren, H.; Geng, J. Entropy Similarity-Driven Transformation Reaction Molecular Networking Reveals Transformation Pathways and Potential Risks of Emerging Contaminants in Wastewater: The Example of Sartans. Environ. Sci. Technol. 2025, 59, 4153–4164. [Google Scholar] [CrossRef]
- Lara-Martín, P.A.; Schinkel, L.; Eberhard, Y.; Giger, W.; Berg, M.; Hollender, J. Suspect and Nontarget Screening of Organic Micropollutants in Swiss Sewage Sludge: A Nationwide Survey. Environ. Sci. Technol. 2025, 59, 7688–7698. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Georgi, A.; Huaccallo-Aguilar, Y.; Meier, M.; Kryk, H.; Reinecke, S.F.; Hampel, U. Degradation and Defluorination of Perfluorooctane Sulfonate (PFOS) Forever Chemical in Water Using Hydrodynamic Cavitation Treatment. Chem. Eng. J. Adv. 2026, 25, 101046. [Google Scholar] [CrossRef]
- Mallek, M.; Barceló, D. Sustainable Analytical Approaches for Microplastics in Wastewater, Sludge, and Landfills: Challenges, Fate, and Green Chemistry Perspectives. Adv. Sample Prep. 2025, 14, 100178. [Google Scholar] [CrossRef]
- van Mourik, L.M.; Crum, S.; Martinez-Frances, E.; van Bavel, B.; Leslie, H.A.; de Boer, J.; Cofino, W.P. Results of WEPAL-QUASIMEME/NORMAN’s First Global Interlaboratory Study on Microplastics Reveal Urgent Need for Harmonization. Sci. Total Environ. 2021, 772, 145071. [Google Scholar] [CrossRef]
- Sumpter, J.P.; Runnalls, T.J.; Johnson, A.C.; Barceló, D. A ‘Limitations’ Section Should Be Mandatory in All Scientific Papers. Sci. Total Environ. 2023, 857, 159395. [Google Scholar] [CrossRef]
- Boisseaux, P.; Delignette-Muller, M.L.; Galloway, T. A Quantitative Environmental Risk Assessment for Microplastics in Sewage Sludge Applied to Land. Environ. Sci. Technol. 2025, 59, 26526–26538. [Google Scholar] [CrossRef]
- Hayes, A.; Zhang, L.; Snape, J.; Feil, E.; Kasprzyk-Hordern, B.; Gaze, W.H.; Murray, A.K. Common Non-Antibiotic Drugs Enhance Selection for Antimicrobial Resistance in Mixture with Ciprofloxacin. ISME Commun. 2025, 5, ycaf169. [Google Scholar] [CrossRef]
- Liu, X.; Wei, W.; Chen, Z.; Wu, L.; Duan, H.; Zheng, M.; Wang, D.; Ni, B.-J. The Threats of Micro- and Nanoplastics to Aquatic Ecosystems and Water Health. Nat. Water 2025, 3, 764–781. [Google Scholar] [CrossRef]
- Pestana, C.J.; Moura, D.S.; Capelo-Neto, J.; Edwards, C.; Dreisbach, D.; Spengler, B.; Lawton, L.A. Potentially Poisonous Plastic Particles: Microplastics as a Vector for Cyanobacterial Toxins Microcystin-LR and Microcystin-LF. Environ. Sci. Technol. 2021, 55, 15940–15949. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Lázaro, J.M.; González-Pleiter, M.; Leganés, F.; Martínez Bueno, M.J.; Fernández-Alba, A.R.; Rosal, R.; Fernández-Piñas, F. The Role of Greenhouse Agricultural Plastic Waste as a Reservoir of Antibiotic Resistance Genes and Vector of Their Environmental Dissemination. Environ. Chem. Ecotoxicol. 2025, 7, 1545–1559. [Google Scholar] [CrossRef]
- Fu, Q.; Malchi, T.; Carter, L.J.; Li, H.; Gan, J.; Chefetz, B. Pharmaceutical and Personal Care Products: From Wastewater Treatment into Agro-Food Systems. Environ. Sci. Technol. 2019, 53, 14083–14090. [Google Scholar] [CrossRef]
- Andreasidou, E.; Kovačič, A.; Manzano-Sánchez, L.; Heath, D.; Kosjek, T.; Pintar, M.; Maršič, N.K.; Blaznik, U.; Fernández-Alba, A.R.; Hernando, M.D.; et al. Uptake of Emerging Contaminants in Tomato Plants: A Field Study on Treated Wastewater Reuse. Environ. Int. 2025, 205, 109916. [Google Scholar] [CrossRef] [PubMed]
- Sanchís, J.; Jiménez-Lamana, J.; Abad, E.; Szpunar, J.; Farré, M. Occurrence of Cerium-, Titanium-, and Silver-Bearing Nanoparticles in the Besòs and Ebro Rivers. Environ. Sci. Technol. 2020, 54, 3969–3978. [Google Scholar] [CrossRef]
- Zheng, Z.; Zhang, R.; Hong, W.; Yang, S.; Lin, X.; Shu, W.; Price, G.W.; Song, L. Landfills as Hotspots of Multidrug Resistance Genes: Profiles, Drivers, and Hosts. Environ. Sci. Technol. 2025, 59, 25724–25737. [Google Scholar] [CrossRef]
- Yu, X.; Sui, Q.; Lyu, S.; Zhao, W.; Liu, J.; Cai, Z.; Yu, G.; Barceló, D. Municipal Solid Waste Landfills: An Underestimated Source of Pharmaceutical and Personal Care Products in the Water Environment. Environ. Sci. Technol. 2020, 54, 9757–9768. [Google Scholar] [CrossRef]
- Andrews, W.J.; Masoner, J.R.; Cozzarelli, I.M. Emerging Contaminants at a Closed and an Operating Landfill in Oklahoma. Groundw. Monit. Remediat. 2012, 32, 120–130. [Google Scholar] [CrossRef]
- Yu, X.; Sui, Q.; Lyu, S.; Zhao, W.; Wu, D.; Yu, G.; Barceló, D. Rainfall Influences Occurrence of Pharmaceutical and Personal Care Products in Landfill Leachates: Evidence from Seasonal Variations and Extreme Rainfall Episodes. Environ. Sci. Technol. 2021, 55, 4822–4830. [Google Scholar] [CrossRef]
- Ehalt Macedo, H.; Lehner, B.; Nicell, J.A.; Khan, U.; Klein, E.Y. Antibiotics in the Global River System Arising from Human Consumption. PNAS Nexus 2025, 4, pgaf096. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Y.; Zhang, Z.; Tian, L.; Zhu, T.; Zhao, Y.; Tong, Y.; Yang, Y.; Sun, P.; Liu, Y. Effect, Fate and Remediation of Pharmaceuticals and Personal Care Products (PPCPs) during Anaerobic Sludge Treatment: A Review. Environ. Sci. Technol. 2024, 58, 19095–19114. [Google Scholar] [CrossRef]
- Ashfaq, M.; Li, Y.; Wang, Y.; Chen, W.; Wang, H.; Chen, X.; Wu, W.; Huang, Z.; Yu, C.-P.; Sun, Q. Occurrence, Fate, and Mass Balance of Different Classes of Pharmaceuticals and Personal Care Products in an Anaerobic-Anoxic-Oxic Wastewater Treatment Plant in Xiamen, China. Water Res. 2017, 123, 655–667. [Google Scholar] [CrossRef]
- Narumiya, M.; Nakada, N.; Yamashita, N.; Tanaka, H. Phase Distribution and Removal of Pharmaceuticals and Personal Care Products during Anaerobic Sludge Digestion. J. Hazard. Mater. 2013, 260, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yin, M.; Li, S.; Zhang, S.; Han, G. The Removal of Erythromycin and Its Effects on Anaerobic Fermentation. Int. J. Environ. Res. Public Health 2022, 19, 7256. [Google Scholar] [CrossRef]
- Xiang, Y.; Xiong, W.; Yang, Z.; Xu, R.; Zhang, Y.; Wu, M.; Ye, Y.; Peng, H.; Sun, W.; Wang, D. Metagenomic Insights into the Toxicity of Carbamazepine to Functional Microorganisms in Sludge Anaerobic Digestion. Sci. Total Environ. 2024, 919, 170780. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Sallach, J.B.; Zhang, W.; Boyd, S.A.; Li, H. Characterization of Plant Accumulation of Pharmaceuticals from Soils with Their Concentration in Soil Pore Water. Environ. Sci. Technol. 2022, 56, 9346–9355. [Google Scholar] [CrossRef]
- Kah, M.; Wilson, S.C.; Carter, L. The Fundamental Role of Healthy Soil in Maintaining Water Quality. Nat. Water 2025, 3, 1365–1375. [Google Scholar] [CrossRef]
- Molins-Delgado, D.; Díaz-Cruz, M.S.; Barceló, D. Introduction: Personal Care Products in the Aquatic Environment. In Personal Care Products in the Aquatic Environment; Díaz-Cruz, M.S., Barceló, D., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–34. [Google Scholar]
- Sang, Z.; Jiang, Y.; Tsoi, Y.-K.; Leung, K.S.-Y. Evaluating the Environmental Impact of Artificial Sweeteners: A Study of Their Distributions, Photodegradation and Toxicities. Water Res. 2014, 52, 260–274. [Google Scholar] [CrossRef]
- Pastorino, P.; Barceló, D.; Prearo, M. Alps at Risk: High-Mountain Lakes as Reservoirs of Persistent and Emerging Contaminants. J. Contam. Hydrol. 2024, 264, 104361. [Google Scholar] [CrossRef]
- Sanchís, J.; Berrojalbiz, N.; Caballero, G.; Dachs, J.; Farré, M.; Barceló, D. Occurrence of Aerosol-Bound Fullerenes in the Mediterranean Sea Atmosphere. Environ. Sci. Technol. 2012, 46, 1335–1343. [Google Scholar] [CrossRef] [PubMed]
- Yuan, B.; Letcher, R.J. Evolving Accumulation of a Complex Profile of Polychlorinated Alkanes in Canadian Polar Bears. Environ. Sci. Technol. Lett. 2024, 11, 591–597. [Google Scholar] [CrossRef]
- Elidrissi El Yallouli, N.; Lahrouni, M.; Mugani, R.; Oudra, B.; Poté, J. Insight in Limited Research on Environmental Factors and Health Implications of Toxic Cyanobacteria Bloom in African Freshwater Bodies. Discover Public Health 2024, 21, 202. [Google Scholar] [CrossRef]
- Marques dos Santos, M.; Snyder, S.A. Occurrence of Polymer Additives 1,3-Diphenylguanidine (DPG), N-(1,3-Dimethylbutyl)-N′-Phenyl-1,4-Benzenediamine (6PPD), and Chlorinated Byproducts in Drinking Water: Contribution from Plumbing Polymer Materials. Environ. Sci. Technol. Lett. 2023, 10, 885–890. [Google Scholar] [CrossRef]
- Seiwert, B.; Nihemaiti, M.; Troussier, M.; Weyrauch, S.; Reemtsma, T. Abiotic Oxidative Transformation of 6PPD and 6PPD-Quinone from Tires and Occurrence of Their Products in Snow from Urban Roads and in Municipal Wastewater. Water Res. 2022, 212, 118122. [Google Scholar] [CrossRef]
- Richardson, S.D.; Manasfi, T. Water Analysis: Emerging Contaminants and Current Issues. Anal. Chem. 2024, 96, 8184–8219. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.N.; Hu, X.; Gonzalez, M.; Rideout, C.A.; Hobby, G.C.; Fisher, M.F.; McCormick, C.J.; Dodd, M.C.; Kim, K.E.; Tian, Z.; et al. Screening p-Phenylenediamine Antioxidants, Their Transformation Products, and Industrial Chemical Additives in Crumb Rubber and Elastomeric Consumer Products. Environ. Sci. Technol. 2023, 57, 2779–2791. [Google Scholar] [CrossRef] [PubMed]
- Reddy, C.M.; Quinn, J.G. Environmental Chemistry of Benzothiazoles Derived from Rubber. Environ. Sci. Technol. 1997, 31, 2847–2853. [Google Scholar] [CrossRef]
- Ni, H.-G.; Lu, F.-H.; Luo, X.-L.; Tian, H.-Y.; Zeng, E.Y. Occurrence, Phase Distribution, and Mass Loadings of Benzothiazoles in Riverine Runoff of the Pearl River Delta, China. Environ. Sci. Technol. 2008, 42, 1892–1897. [Google Scholar] [CrossRef] [PubMed]
- Franklin, E.B.; Alves, M.R.; Moore, A.N.; Kilgour, D.B.; Novak, G.A.; Mayer, K.; Sauer, J.S.; Weber, R.J.; Dang, D.; Winter, M.; et al. Atmospheric Benzothiazoles in a Coastal Marine Environment. Environ. Sci. Technol. 2021, 55, 15705–15714. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Dai, Z.; Tang, Z.; Zhu, G.; Liu, W.; Zhang, C.; Li, J.; Yang, Y.; Yu, Z.; Lin, B.; et al. Workplace Presence and Exposure of p-Phenylenediamines and Their Quinones in Rubber Production. Environ. Sci. Technol. Lett. 2025, 12, 1418–1425. [Google Scholar] [CrossRef]
- Chen, X.; He, T.; Yang, X.; Gan, Y.; Qing, X.; Wang, J.; Huang, Y. Analysis, Environmental Occurrence, Fate and Potential Toxicity of Tire Wear Compounds 6PPD and 6PPD-Quinone. J. Hazard. Mater. 2023, 452, 131245. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zhao, H.N.; Tian, Z.; Peter, K.T.; Dodd, M.C.; Kolodziej, E.P. Chemical Characteristics, Leaching, and Stability of the Ubiquitous Tire Rubber-Derived Toxicant 6PPD-Quinone. Environ. Sci. Process. Impacts 2023, 25, 901–911. [Google Scholar] [CrossRef]
- Zoroufchi Benis, K.; Behnami, A.; Minaei, S.; Brinkmann, M.; McPhedran, K.N.; Soltan, J. Environmental Occurrence and Toxicity of 6PPD Quinone, an Emerging Tire Rubber-Derived Chemical: A Review. Environ. Sci. Technol. Lett. 2023, 10, 815–823. [Google Scholar] [CrossRef]
- Li, C.; Yang, Y.; Tian, Z.; Huang, Z.; Huang, Y.; Hong, Y. Residues of 6PPD-Q in the Aquatic Environment and Toxicity to Aquatic Organisms: A Review. Fishes 2025, 10, 146. [Google Scholar] [CrossRef]
- Zhao, H.N.; Peter, K.T.; Gonzalez, M.; Rideout, C.A.; Hu, X.; Tian, Z.; Kolodziej, E.P. Temporal Dynamics of PPD-Class Antioxidants and Transformation Products in a Small Roadway-Runoff-Impacted Watershed. Environ. Sci. Technol. 2025, 59, 18358–18371. [Google Scholar] [CrossRef]
- Steinhoff, B.; Müller, J.; Mozhayeva, D.; Spelz, B.T.F.; Engelhard, C.; Butz, B.; Schönherr, H. Investigation of the Fate of Silver and Titanium Dioxide Nanoparticles in Model Wastewater Effluents via Selected Area Electron Diffraction. Environ. Sci. Technol. 2020, 54, 8681–8689. [Google Scholar] [CrossRef]
- Ma, R.; Levard, C.; Judy, J.D.; Unrine, J.M.; Durenkamp, M.; Martin, B.; Jefferson, B.; Lowry, G.V. Fate of Zinc Oxide and Silver Nanoparticles in a Pilot Wastewater Treatment Plant and in Processed Biosolids. Environ. Sci. Technol. 2014, 48, 104–112. [Google Scholar] [CrossRef]
- Stegemeier, J.P.; Avellan, A.; Lowry, G.V. Effect of Initial Speciation of Copper- and Silver-Based Nanoparticles on Their Long-Term Fate and Phytoavailability in Freshwater Wetland Mesocosms. Environ. Sci. Technol. 2017, 51, 12114–12122. [Google Scholar] [CrossRef]
- Lowry, G.V.; Espinasse, B.P.; Badireddy, A.R.; Richardson, C.J.; Reinsch, B.C.; Bryant, L.D.; Bone, A.J.; Deonarine, A.; Chae, S.; Therezien, M.; et al. Long-Term Transformation and Fate of Manufactured Ag Nanoparticles in a Simulated Large Scale Freshwater Emergent Wetland. Environ. Sci. Technol. 2012, 46, 7027–7036. [Google Scholar] [CrossRef]
- Windler, L.; Lorenz, C.; von Goetz, N.; Hungerbühler, K.; Amberg, M.; Heuberger, M.; Nowack, B. Release of Titanium Dioxide from Textiles during Washing. Environ. Sci. Technol. 2012, 46, 8181–8188. [Google Scholar] [CrossRef]
- Weir, A.; Westerhoff, P.; Fabricius, L.; Hristovski, K.; von Goetz, N. Titanium Dioxide Nanoparticles in Food and Personal Care Products. Environ. Sci. Technol. 2012, 46, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Gondikas, A.P.; von der Kammer, F.; Reed, R.B.; Wagner, S.; Ranville, J.F.; Hofmann, T. Release of TiO2 Nanoparticles from Sunscreens into Surface Waters: A One-Year Survey at the Old Danube Recreational Lake. Environ. Sci. Technol. 2014, 48, 5415–5422. [Google Scholar] [CrossRef] [PubMed]
- Verwold, C.; Tremblay, C.; Patron, M.; Kimura, S. Total Organic Halogen (TOX) in Treated Wastewaters: Method Development and Comparison with Target Analysis. 2023. [Google Scholar] [CrossRef]
- Zhou, X.; Zhao, X.; Chen, T.; Lv, J.; Guo, J.; Song, B.; Li, J.; Wang, Y.; Jiang, G. Comprehensive Assessment of Poly- and Perfluoroalkyl Substances in China: Flows, Stocks, and Emissions from Polytetrafluoroethylene. Environ. Sci. Technol. 2026, 60, 3886–3898. [Google Scholar] [CrossRef]
- Thompson, K.A.; Mortazavian, S.; Gonzalez, D.J.; Bott, C.; Hooper, J.; Schaefer, C.E.; Dickenson, E.R.V. Poly- and Perfluoroalkyl Substances in Municipal Wastewater Treatment Plants in the United States: Seasonal Patterns and Meta-Analysis of Long-Term Trends and Average Concentrations. ACS ES&T Water 2022, 2, 690–700. [Google Scholar]
- Baqar, M.; Zhao, M.; Saleem, R.; Cheng, Z.; Fang, B.; Dong, X.; Chen, H.; Yao, Y.; Sun, H. Identification of Emerging Per- and Polyfluoroalkyl Substances (PFAS) in E-Waste Recycling Practices and New Precursors for Trifluoroacetic Acid. Environ. Sci. Technol. 2024, 58, 16153–16163. [Google Scholar] [CrossRef]
- Kudzin, G.P.; Dodds, J.N.; Kirkwood-Donelson, K.I.; Schiffenbauer, A.; Sarkar, K.; Noroozi Farhadi, P.; Miller, F.W.; Johnson, D.; Chappel, J.; Jarmusch, A.K.; et al. Evaluating Legacy and Emerging PFAS in Human Blood Collected from 2003 to 2021. Environ. Sci. Technol. 2026, 60, 3068–3079. [Google Scholar] [CrossRef]
- Teymoorian, T.; Delon, L.; Munoz, G.; Sauvé, S. Target and Suspect Screening Reveal PFAS Exceeding European Union Guideline in Various Water Sources South of Lyon, France. Environ. Sci. Technol. Lett. 2025, 12, 327–333. [Google Scholar] [CrossRef]
- Llamas, M.I.; Fernández-Valenzuela, P.J.; Vadillo, I.; Sanmiguel-Martí, M.; Rambla-Nebot, J.; Aranda-Mares, J.L.; Jiménez-Gavilán, P. Study of the Presence and Environmental Risk of Organic Contaminants Policed by the European Union and Other Organic Compounds in the Water Resources of a Region Overlapping Protected Areas: The Guadiaro River Basin (Southern Spain). J. Environ. Manage. 2023, 345, 118903. [Google Scholar] [CrossRef]
- Liu, N.; Jin, X.; Johnson, A.C.; Zhou, S.; Liu, Y.; Hou, L.; Meng, F.; Wu, F. Pharmaceutical and Personal Care Products (PPCPs) in Global Surface Waters: Risk and Drivers. Environ. Sci. Technol. 2025, 59, 19146–19159. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Lv, M.; Song, L.; Ding, J.; Man, M.; Fu, L.; Song, Z.; Li, B.; Chen, L. Occurrence, Distribution, and Trophic Transfer of Pharmaceuticals and Personal Care Products in the Bohai Sea. Environ. Sci. Technol. 2023, 57, 21823–21834. [Google Scholar] [CrossRef]
- Schildroth, S.; Bethea, T.N.; Wesselink, A.K.; Friedman, A.; Fruh, V.; Calafat, A.M.; Wegienka, G.; Gaston, S.; Baird, D.D.; Wise, L.A.; et al. Personal Care Products, Socioeconomic Status, and Endocrine-Disrupting Chemical Mixtures in Black Women. Environ. Sci. Technol. 2024, 58, 3641–3653. [Google Scholar] [CrossRef] [PubMed]
- Subedi, B.; Du, B.; Chambliss, C.K.; Koschorreck, J.; Rüdel, H.; Quack, M.; Brooks, B.W.; Usenko, S. Occurrence of Pharmaceuticals and Personal Care Products in German Fish Tissue: A National Study. Environ. Sci. Technol. 2012, 46, 9047–9054. [Google Scholar] [CrossRef]
- Bexfield, L.M.; Toccalino, P.L.; Belitz, K.; Foreman, W.T.; Furlong, E.T. Hormones and Pharmaceuticals in Groundwater Used as a Source of Drinking Water Across the United States. Environ. Sci. Technol. 2019, 53, 2950–2960. [Google Scholar] [CrossRef]
- Ziajahromi, S.; Lu, H.-C.; Drapper, D.; Hornbuckle, A.; Leusch, F.D.L. Microplastics and Tire Wear Particles in Urban Stormwater: Abundance, Characteristics, and Potential Mitigation Strategies. Environ. Sci. Technol. 2023, 57, 12829–12837. [Google Scholar] [CrossRef] [PubMed]
- Rauert, C.; Charlton, N.; Okoffo, E.D.; Stanton, R.S.; Agua, A.R.; Pirrung, M.C.; Thomas, K.V. Concentrations of Tire Additive Chemicals and Tire Road Wear Particles in an Australian Urban Tributary. Environ. Sci. Technol. 2022, 56, 2421–2431. [Google Scholar] [CrossRef]
- Cao, X.; Yue, T.; Meng, W.; Zhang, Y.; Li, J.; Yang, Y.; Su, G. Nontargeted Screening Unveils Structural Diversity and Environmental Pervasiveness of p-Phenylenediamine Antioxidant-Derived Quinones: Evidence from End-of-Life Tires and Road Dust Contamination. Environ. Sci. Technol. 2025, 59, 20630–20641. [Google Scholar] [CrossRef]
- Miller, S.A.; Schmidt, T.S.; Barber, L.B.; Hladik, M.L.; Kolpin, D.W.; Shoda, M.E.; Stackpoole, S.M. Imidacloprid in United States Rivers, 2013–2022: Persistent Presence and Emerging Chronic Hazard. Environ. Sci. Technol. 2025, 59, 26702–26715. [Google Scholar] [CrossRef]
- Hohrenk-Danzouma, L.L.; Vosough, M.; Merkus, V.I.; Drees, F.; Schmidt, T.C. Non-Target Analysis and Chemometric Evaluation of a Passive Sampler Monitoring of Small Streams. Environ. Sci. Technol. 2022, 56, 5466–5477. [Google Scholar] [CrossRef]
- León-Morán, L.O.; Pastor-Belda, M.; Viñas, P.; Arroyo-Manzanares, N.; Sánchez-Fernández, O.; Pérez-Ruzafa, Á.; Campillo, N. Targeted and Untargeted Approaches Using Liquid Chromatography with High-Resolution Mass Spectrometry for the Determination of Lipophilic Marine Biotoxins in Seawater and Mussel Samples. Mar. Pollut. Bull. 2026, 222, 118688. [Google Scholar] [CrossRef]
- Méndez, S.; Montes, R.; Raimundo, J.; López-Castillo, D.; López-Vázquez, J.; Caetano, M.; Lopes, C.; Figueiredo, C.; Pinheiro, M.; Alves, N.; et al. Screening of Organic Pollutants in Mollusc and Fish Samples from the Portuguese Coast by Combining Liquid and Gas Chromatography with High Resolution Mass Spectrometry. Mar. Pollut. Bull. 2026, 222, 118598. [Google Scholar] [CrossRef] [PubMed]
- The Handbook of Environmental Chemistry; Barceló, D., Kostianoy, A.G., Eds.; Springer: Cham, Switzerland, 2015; Vol. 36. [Google Scholar]
- Yu, X.; Zhao, W.; Lyu, S.; Cai, Z.; Yu, G.; Wang, H.; Barceló, D.; Sui, Q. Estimating the Mass of Pharmaceuticals Harbored in Municipal Solid Waste Landfills by Analyzing Refuse Samples at Various Ages and Depths. Environ. Sci. Technol. 2023, 57, 6063–6071. [Google Scholar] [CrossRef] [PubMed]
- Mehrabi, K.; Nowack, B.; Arroyo Rojas Dasilva, Y.; Mitrano, D.M. Improvements in Nanoparticle Tracking Analysis To Measure Particle Aggregation and Mass Distribution: A Case Study on Engineered Nanomaterial Stability in Incineration Landfill Leachates. Environ. Sci. Technol. 2017, 51, 5611–5621. [Google Scholar] [CrossRef]
- Pettibone, J.M.; Liu, J. In Situ Methods for Monitoring Silver Nanoparticle Sulfidation in Simulated Waters. Environ. Sci. Technol. 2016, 50, 11145–11153. [Google Scholar] [CrossRef]
- Smalling, K.L.; Romanok, K.M.; Bradley, P.M.; Morriss, M.C.; Gray, J.L.; Kanagy, L.K.; Gordon, S.E.; Williams, B.M.; Breitmeyer, S.E.; Jones, D.K.; et al. Per- and Polyfluoroalkyl Substances (PFAS) in United States Tapwater: Comparison of Underserved Private-Well and Public-Supply Exposures and Associated Health Implications. Environ. Int. 2023, 178, 108033. [Google Scholar] [CrossRef] [PubMed]
- Wicks, A.; Brady, S.; Whitehead, H.D.; Hedman, T.; Zachritz, A.; Venier, M.; Peaslee, G.F. Per- and Polyfluoroalkyl Substances in Reusable Feminine Hygiene Products. Environ. Sci. Technol. Lett. 2025, 12, 924–929. [Google Scholar] [CrossRef]
- Xu, Y.; Ou, Q.; Wang, X.; Hou, F.; Li, P.; van der Hoek, J.P.; Liu, G. Assessing the Mass Concentration of Microplastics and Nanoplastics in Wastewater Treatment Plants by Pyrolysis Gas Chromatography–Mass Spectrometry. Environ. Sci. Technol. 2023, 57, 3114–3123. [Google Scholar] [CrossRef]
- Yang, T.; Luo, J.; Nowack, B. Characterization of Nanoplastics, Fibrils, and Microplastics Released during Washing and Abrasion of Polyester Textiles. Environ. Sci. Technol. 2021, 55, 15873–15881. [Google Scholar] [CrossRef] [PubMed]
- Paterson, K.; Beckingham, B.; Momplaisir, G.-M.; Varner, K. Adapting Methods for Isolation and Enumeration of Microplastics to Quantify Tire Road Wear Particles with Confirmation by Pyrolysis GC–MS. Environ. Sci. Technol. 2025, 59, 1769–1779. [Google Scholar] [CrossRef]
- Regan, F.; Allen, C.; Lawler, J. Demonstrating the Application of a Liquid Chromatography-Mass Spectrometry Method for the Determination of Phthalate Diesters in Complex Solid and Liquid Environmental Samples. Anal. Methods 2025, 17, 8877–8888. [Google Scholar] [CrossRef]
- Kazmi, S.S.U.H.; Xu, Q.; Saqib, H.S.A.; Azeem, M.; Pastorino, P.; Barceló, D.; Yang, Q.; Li, G. Pollutant-Driven Humification of Dissolved Organic Matter Fails to Mitigate the Chronic Toxicity of Tire-Wear Contaminants in Freshwater Bioindicator. Chem. Eng. J. 2026, 529, 173274. [Google Scholar] [CrossRef]
- Gu, Y.; Feuerstein, M.L.; Lloyd, D.T.; Patel, C.J.; Johnson, C.H.; Warth, B. Quantitative Exposomics Targeting over 200 Toxicants and Key Biomarkers at the Picomolar Level. Environ. Sci. Technol. 2025, 59, 21818–21829. [Google Scholar] [CrossRef]
- Miller, T.R.; Xiong, A.; Deeds, J.R.; Stutts, W.L.; Samdal, I.A.; Løvberg, K.E.; Miles, C.O. Microcystin Toxins at Potentially Hazardous Levels in Algal Dietary Supplements Revealed by a Combination of Bioassay, Immunoassay, and Mass Spectrometric Methods. J. Agric. Food Chem. 2020, 68, 8016–8025. [Google Scholar] [CrossRef]
- Mahony, A.K.; Clair, T.A.; Mendez, M.A.; McNamara, P.J.; Arnold, W.A. Quaternary Ammonium Compounds in Wastewater Influents, Effluents, and Biosolids: Analysis from Twelve Wastewater Treatment Plants from 2020 to 2023. Environ. Sci. Technol. 2025, 59, 27929–27942. [Google Scholar] [CrossRef]
- Mills, C.; Dillon, M.J.; Kulabhusan, P.K.; Senovilla-Herrero, D.; Campbell, K. Multiplex Lateral Flow Assay and the Sample Preparation Method for the Simultaneous Detection of Three Marine Toxins. Environ. Sci. Technol. 2022, 56, 12210–12217. [Google Scholar] [CrossRef]
- Zhang, W.; Dixon, M.B.; Saint, C.; Teng, K.S.; Furumai, H. Electrochemical Biosensing of Algal Toxins in Water: The Current State-of-the-Art. ACS Sens. 2018, 3, 1233–1245. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.-H.; Law, C.F.J.; Leung, K. Degradation of Acesulfame in UV/Monochloramine Process: Kinetics, Transformation Pathways and Toxicity Assessment. J. Hazard. Mater. 2021, 403, 123935. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Law, J.C.-F.; Leung, K.S.-Y. Chlorination of Bisphenols in Water: Understanding the Kinetics and Formation Mechanism of 2-Butene-1,4-Dial and Analogues. J. Hazard. Mater. 2023, 459, 132128. [Google Scholar] [CrossRef]
- Hofstetter, T.B.; Bakkour, R.; Buchner, D.; Eisenmann, H.; Fischer, A.; Gehre, M.; Haderlein, S.B.; Höhener, P.; Hunkeler, D.; Imfeld, G.; et al. Perspectives of Compound-Specific Isotope Analysis of Organic Contaminants for Assessing Environmental Fate and Managing Chemical Pollution. Nat. Water 2024, 2, 14–30. [Google Scholar] [CrossRef]
- Boatman, A.K.; Chappel, J.R.; Kirkwood-Donelson, K.I.; Fleming, J.F.; Reif, D.M.; Schymanski, E.L.; Rager, J.E.; Baker, E.S. Updated Guidance for Communicating PFAS Identification Confidence with Ion Mobility Spectrometry. Environ. Sci. Technol. 2025, 59, 17711–17721. [Google Scholar] [CrossRef] [PubMed]
- Shafique, I.; Ahmad, R.; Shin, K.-H.; Cho, Y.; Oh, N.-H.; Hur, J.; Kim, S. Uncovering Hidden Pollution: Diffuse Contaminant Sources in a Sparsely Industrialized Estuarine System. ACS ES&T Water 2025, 5, 6994–7004. [Google Scholar] [CrossRef]
- Yao, J.; Sheng, N.; Guo, Y.; Yeung, L.W.Y.; Dai, J.; Pan, Y. Nontargeted Identification and Temporal Trends of Per- and Polyfluoroalkyl Substances in a Fluorochemical Industrial Zone and Adjacent Taihu Lake. Environ. Sci. Technol. 2022, 56, 7986–7996. [Google Scholar] [CrossRef] [PubMed]
- Arp, H.P.H.; Gredelj, A.; Glüge, J.; Scheringer, M.; Cousins, I.T. The Global Threat from the Irreversible Accumulation of Trifluoroacetic Acid (TFA). Environ. Sci. Technol. 2024, 58, 19925–19935. [Google Scholar] [CrossRef]
- Celma, A.; Sancho, J.V.; Schymanski, E.L.; Fabregat-Safont, D.; Ibáñez, M.; Goshawk, J.; Barknowitz, G.; Hernández, F.; Bijlsma, L. Improving Target and Suspect Screening High-Resolution Mass Spectrometry Workflows in Environmental Analysis by Ion Mobility Separation. Environ. Sci. Technol. 2020, 54, 15120–15131. [Google Scholar] [CrossRef]
- Luo, Y.-S.; Aly, N.; McCord, J.; Strynar, M.; Chiu, W.; Dodds, J.; Baker, E.; Rusyn, I. Rapid Characterization of Emerging Per- and Polyfluoroalkyl Substances in Aqueous Film-Forming Foams Using Ion Mobility Spectrometry–Mass Spectrometry. Environ. Sci. Technol. 2020, 54, 15024–15034. [Google Scholar] [CrossRef]
- Camdzic, D.; Dickman, R.A.; Joyce, A.S.; Wallace, J.S.; Ferguson, P.L.; Aga, D.S. Quantitation of Total PFAS Including Trifluoroacetic Acid with Fluorine Nuclear Magnetic Resonance Spectroscopy. Anal. Chem. 2023, 95, 5484–5488. [Google Scholar] [CrossRef]
- Smolinski, R.; Koelmel, J.P.; Stelben, P.; Weil, D.; Godri, D.; Schiessel, D.; Kummer, M.; Stow, S.M.; Mohsin, S.; Royer, L.; et al. FluoroMatch IM: An Interactive Software for PFAS Analysis by Ion Mobility Spectrometry. Environ. Sci. Technol. 2025, 59, 6636–6648. [Google Scholar] [CrossRef]
- Motteau, S.; Dervilly, G.; Cariou, R.; Margalef, M.; Lamoree, M.; Hamers, T.; König, M.; Escher, B.I.; Vinggaard, A.M.; Rørbye, C.; et al. Determination of Chemical Mixtures in Environmental, Food, and Human Samples Using High-Resolution Mass Spectrometry-Based Suspect Screening Approaches. Environ. Sci. Technol. 2025, 59, 21265–21277. [Google Scholar] [CrossRef]
- Graves, L.G.; Zarfl, C.; Hirsch, T.; Vitale, G.A.; Petras, D.; Spahr, S. Target and Nontarget Analyses Reveal Similar Dissolved Organic Contaminant Patterns Relative to Quantified Catchment Characteristics along Two German Rivers. Environ. Sci. Technol. 2026, 60, 3580–3591. [Google Scholar] [CrossRef]
- Zou, Q.; Zhang, Q.; Yang, R.; Li, Y.; Pei, Z.; Liu, M.; Zhang, G.; Ji, F.; Zhang, X.; Yang, X.; et al. Non-Negligible Polyhalogenated Carbazoles in Arctic Soils and Sediments: Occurrence, Target and Suspect Screening, and Potential Sources. Environ. Sci. Technol. 2024, 58, 23169–23179. [Google Scholar] [CrossRef]
- Schymanski, E.L.; Jeon, J.; Gulde, R.; Fenner, K.; Ruff, M.; Singer, H.P.; Hollender, J. Identifying Small Molecules via High Resolution Mass Spectrometry: Communicating Confidence. Environ. Sci. Technol. 2014, 48, 2097–2098. [Google Scholar] [CrossRef]
- Nanusha, M.Y.; Frøkjær, E.E.; Søndergaard, J.; Mørk Larsen, M.; Schwartz Glottrup, C.; Bruun Nicolaisen, J.; Hansen, M. Quantitative Non-Targeted Screening to Profile Micropollutants in Sewage Sludge Used for Agricultural Field Amendments. Environ. Sci. Technol. 2024, 58, 9850–9862. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhou, Y.; Chen, Y.; Xie, Z.; Qiu, J.; Zhu, F.; Ouyang, G.; Yang, X. Stable Isotopic Labeling and Automated Reactivity-Directed Approach Accelerating Discovery of Toxic Disinfection Byproducts in Chlorinated and Chloraminated Waters. Environ. Sci. Technol. 2026, 60, 3486–3496. [Google Scholar] [CrossRef] [PubMed]
- Maurer, L.; Carmona, E.; Machate, O.; Schulze, T.; Krauss, M.; Brack, W. Contamination Pattern and Risk Assessment of Polar Compounds in Snow Melt: An Integrative Proxy of Road Runoffs. Environ. Sci. Technol. 2023, 57, 4143–4152. [Google Scholar] [CrossRef] [PubMed]
- Black, G.P.; Anderson, B.N.; Wong, L.; Alaimo, C.P.; He, G.; Denison, M.S.; Bennett, D.H.; Tancredi, D.; Durbin-Johnson, B.; Hammock, B.D.; et al. Comprehensive Nontargeted Analysis of Drinking Water Supplies to Identify Chemicals Associated with Estrogen Receptor Agonism or Present in Regions of Elevated Breast Cancer Occurrence. Environ. Sci. Technol. 2025, 59, 5237–5248. [Google Scholar] [CrossRef]
- Liu, X.; Xu, C.; Wu, J.; Foo, Y.H.; Zhou, J.; Wu, B.; Chan, L.L. Automatic MS/MS Data Mining Strategy for Rapid Screening of Polyether Toxins Derived from Gambierdiscus Species. Anal. Chem. 2025, 97, 5643–5652. [Google Scholar] [CrossRef]
- Li, Q.; Lai, Y.; Li, P.; Liu, X.; Yao, Z.; Liu, J.; Yu, S. Evaluating the Occurrence of Polystyrene Nanoparticles in Environmental Waters by Agglomeration with Alkylated Ferroferric Oxide Followed by Micropore Membrane Filtration Collection and Py-GC/MS Analysis. Environ. Sci. Technol. 2022, 56, 8255–8265. [Google Scholar] [CrossRef]
- Okoffo, E.D.; Chan, C.M.; Rauert, C.; Kaserzon, S.; Thomas, K.V. Identification and Quantification of Micro-Bioplastics in Environmental Samples by Pyrolysis–Gas Chromatography–Mass Spectrometry. Environ. Sci. Technol. 2022, 56, 13774–13785. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Lai, Y.; Zheng, R.; Li, Q.; Sheng, X.; Yu, S.; Hao, Z.; Cai, Y.; Liu, J. Extraction of Common Small Microplastics and Nanoplastics Embedded in Environmental Solid Matrices by Tetramethylammonium Hydroxide Digestion and Dichloromethane Dissolution for Py-GC-MS Determination. Environ. Sci. Technol. 2023, 57, 12010–12018. [Google Scholar] [CrossRef] [PubMed]
- Gahn, M.B.; Wharton, M.; Mortuza, A.; Hala, D.; Marshall, C.D.; Kaiser, K. Rapid and Sensitive Quantification of Nano- and Microplastics in Water, Sediment, and Biological Tissue by Pyrolysis-Gas Chromatography Tandem Mass Spectrometry with Dynamic Reaction Monitoring. Anal. Chem. 2026, 98, 633–641. [Google Scholar] [CrossRef]
- Wang, Y.; Davis, I.; Shin, I.; Wherritt, D.J.; Griffith, W.P.; Dornevil, K.; Colabroy, K.L.; Liu, A. Biocatalytic Carbon–Hydrogen and Carbon–Fluorine Bond Cleavage through Hydroxylation Promoted by a Histidyl-Ligated Heme Enzyme. ACS Catal. 2019, 9, 4764–4776. [Google Scholar] [CrossRef]
- Dalmijn, J.; Glüge, J.; Scheringer, M.; Cousins, I.T. Emission Inventory of PFAS and Other Fluorinated Organic Substances for the Fluoropolymer Production Industry in Europe. Environ. Sci. Process. Impacts 2023, 25, 1806–1819. [Google Scholar]
- Zhang, Y.; Guo, C.; Wu, R.; Hou, S.; Liu, Y.; Zhao, J.; Jiang, M.; Xu, J.; Wu, F. Global Occurrence, Distribution, and Ecological Risk Assessment of Psychopharmaceuticals and Illicit Drugs in Surface Water Environment: A Meta-Analysis. Water Res. 2024, 263, 122165. [Google Scholar] [CrossRef]
- Sanchís, J.; Olmos, M.; Vincent, P.; Farré, M.; Barceló, D. New Insights on the Influence of Organic Co-Contaminants on the Aquatic Toxicology of Carbon Nanomaterials. Environ. Sci. Technol. 2016, 50, 961–969. [Google Scholar] [CrossRef]
- Forster, A.L.B.; Wiskur, S.L.; Richardson, S.D. Formation of Eight Classes of DBPs from Chlorine, Chloramine, and Ozone: Mechanisms and Formation Pathways. Environ. Sci. Technol. 2025, 59, 15594–15611. [Google Scholar] [CrossRef]
- McIntyre, J.K.; Prat, J.; Cameron, J.; Wetzel, J.; Mudrock, E.; Peter, K.T.; Tian, Z.; Mackenzie, C.; Lundin, J.; Stark, J.D.; et al. Treading Water: Tire Wear Particle Leachate Recreates an Urban Runoff Mortality Syndrome in Coho but Not Chum Salmon. Environ. Sci. Technol. 2021, 55, 11767–11774. [Google Scholar] [CrossRef]
- Süßmuth, R.; Rosenberger, T.; Schweyen, P.; Dierkes, G.; Bell, A.M.; Wick, A.; Buchinger, S.; Ternes, T.A. Deciphering the Estrogenic Activity of Aqueous Leachates from Elastomers by Effect-Directed Analysis. Environ. Sci. Technol. 2025, 59, 21442–21453. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, C.; Sun, S.; Xie, M.; Wang, X.; Liu, S.; Xu, J.; Wu, F. Cigarette Filters—A Neglected Source of Phthalate Exposure to Humans. Environ. Sci. Technol. Lett. 2025, 12, 137–143. [Google Scholar] [CrossRef]
- Rodgers, T.F.M.; Drew, S.; Brown, T.; Hiki, K.; Hiroshi, Y.; King, M.; Kolodziej, E.P.; Krogh, E.T.; McIntyre, J.K.; Miller, K.; et al. Turning the Corner on Hazardous Tire Compounds: A Management Framework for Tire Additive Pollution. Environ. Sci. Technol. Lett. 2025, 12, 869–880. [Google Scholar] [CrossRef]
- Thomas, J.; Patil, R. The Road to Sustainable Tire Materials: Current State-of-the-Art and Future Prospectives. Environ. Sci. Technol. 2023, 57, 2209–2216. [Google Scholar] [CrossRef]
- Eckelman, M.J.; Mauter, M.S.; Isaacs, J.A.; Elimelech, M. New Perspectives on Nanomaterial Aquatic Ecotoxicity: Production Impacts Exceed Direct Exposure Impacts for Carbon Nanotubes. Environ. Sci. Technol. 2012, 46, 2902–2910. [Google Scholar] [CrossRef] [PubMed]
- Pu, Y.; Tang, F.; Adam, P.-M.; Laratte, B.; Ionescu, R.E. Fate and Characterization Factors of Nanoparticles in Seventeen Subcontinental Freshwaters: A Case Study on Copper Nanoparticles. Environ. Sci. Technol. 2016, 50, 9370–9379. [Google Scholar] [CrossRef] [PubMed]
- Conway, J.R.; Adeleye, A.S.; Gardea-Torresdey, J.; Keller, A.A. Aggregation, Dissolution, and Transformation of Copper Nanoparticles in Natural Waters. Environ. Sci. Technol. 2015, 49, 2749–2756. [Google Scholar] [CrossRef]
- Holden, P.A.; Gardea-Torresdey, J.L.; Klaessig, F.; Turco, R.F.; Mortimer, M.; Hund-Rinke, K.; Cohen Hubal, E.A.; Avery, D.; Barceló, D.; Behra, R.; et al. Considerations of Environmentally Relevant Test Conditions for Improved Evaluation of Ecological Hazards of Engineered Nanomaterials. Environ. Sci. Technol. 2016, 50, 6124–6145. [Google Scholar] [CrossRef]
- Dong, H.; Aziz, Md.T.; Richardson, S.D. Transformation of Algal Toxins during the Oxidation/Disinfection Processes of Drinking Water: From Structure to Toxicity. Environ. Sci. Technol. 2023, 57, 12944–12957. [Google Scholar] [CrossRef]
- Bei, E.; Li, X.; Wu, F.; Li, S.; He, X.; Wang, Y.; Qiu, Y.; Wang, Y.; Wang, C.; Wang, J.; et al. Formation of N-Nitrosodimethylamine Precursors through the Microbiological Metabolism of Nitrogenous Substrates in Water. Water Res. 2020, 183, 116055. [Google Scholar] [CrossRef]
- Agathokleous, E.; Peñuelas, J.; Azevedo, R.A.; Rillig, M.C.; Sun, H.; Calabrese, E.J. Low Levels of Contaminants Stimulate Harmful Algal Organisms and Enrich Their Toxins. Environ. Sci. Technol. 2022, 56, 11991–12002. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Xia, W.; Liu, H.; Liu, J.; Cao, S.; Fang, X.; Li, S.; Li, Y.; Chen, C.; Xu, S. Trihalomethanes in Global Drinking Water: Distributions, Risk Assessments, and Attributable Disease Burden of Bladder Cancer. J. Hazard. Mater. 2024, 469, 133760. [Google Scholar] [CrossRef]
- Chen, R.; Zhou, Y.; Wang, Y.; Cheng, R.; Li, P.; Wang, Y.; Jiang, Y.; Wang, J.; Fu, Y.; Ni, B.; et al. Benzotriazoles and Benzothiazoles, Maternal and Neonatal Thyroid Hormones, and Childhood Growth: A Longitudinal Cohort Study. Environ. Sci. Technol. 2025, 59, 20967–20977. [Google Scholar] [CrossRef]
- Moulton, C.; Baroni, A.; Tasciotti, E. Micro- and Nanoplastics Exposure Across the Lifespan: One Health Implications for Aging and Longevity. J. Xenobiot. 2026, 16, 52. [Google Scholar] [CrossRef] [PubMed]
- Bayattork, M.; Rahman, M.; Hossain, M.I.; Zhang, Y.; Haque, A.N.M.A.; Kim, B.; Naebe, M. Impact of Textile-Derived Micro- and Nanoplastics on Brain Health: An Emerging Environmental Risk. Environ. Sci. Technol. 2026, 60, 2863–2895. [Google Scholar] [CrossRef] [PubMed]
- Šmídová, K.; Soinne, H.; Kim, S.W.; Tirronniemi, J.; Meffe, R.; Redondo-Hasselerharm, P.E.; Braun, M.; Rillig, M.C.; Fritze, H.; Adamczyk, B.; et al. Conventional and Biodegradable Agricultural Microplastics: Effects on Soil Properties and Microbial Functions across a European Pedoclimatic Gradient. Environ. Pollut. 2025, 386, 127212. [Google Scholar] [CrossRef] [PubMed]
| Analytical strategy | Operational size range | Polymer identification | Quantification capability | Additional information obtained | Main strengths | Main limitations | Reference |
| Agglomeration with alkylated Fe₃O₄ + membrane filtration + Pyr-GC/MS | Nanoplastics; validated down to 20 nm for PS | PS and PMMA via characteristic pyrolysis products | Detection limits 0.02–0.03 µg L⁻¹ for PS and PMMA; environmental PS detected in 11/15 samples at <0.07–0.73 µg L⁻¹ | Polymer-specific mass concentrations in environmental waters | Very low detection limits; efficient nanoplastic enrichment; magnetic agglomeration reduces filtration resistance | Destructive; no particle-size, shape, or count information; validated for limited polymer scope | [133] |
| Pressurized liquid extraction (PLE) + thermochemolysis (TMAH) + Pyr-GC/MS | Mass-based MP fraction (<5 mm); no particle-size resolution; applicable to biodegradable polymers and nanoplastic-containing extracts | PLA, PCL, PBS, PBAT, and PHAs | Recoveries 74–116%; LOQs 0.02–0.05 mg g⁻¹; MDLs 0.06–0.17 µg injection⁻¹; calibration R ≥ 0.95 | Polymer-specific mass quantification with internal-standard correction | Simultaneous identification and quantification; suitable for complex matrices; thermochemolysis improves specificity | Destructive; no particle-number or morphology information; requires extraction and derivatization steps | [134] |
| TMAH digestion + dichloromethane dissolution + Pyr-GC/MS for solid matrices | Small MPs/NPs <150 µm; validated down to 50 nm for PS and PMMA | PS, PMMA, PE, PP, PVC, PET | Recoveries 79.6–91.4%; size-dependent recoveries 69–101%; LODs 2.3–29.2 µg g⁻¹; linearity R² ≥ 0.97 | Polymer-specific mass concentrations in soil, sediment, and sludge; total MNP levels 4.6–51.4 µg g⁻¹ in real samples | Efficient extraction of small particles embedded in solid matrices; limited size effect; suitable for NOM-rich samples | Destructive; possible PVC-related benzene interference; centrifugation may cause minor nanoparticle losses | [135] |
| Pyr-GC-QqQ-MS in dynamic MRM mode with internal-standard calibration | Mass-based MP fraction (<5 mm); operational water fraction typically >5 µm after filtration; also applicable to nanoplastic-containing fractions | Twelve polymers including PMMA, PP, PVC, PA, PC, PA66, PE, PET, ABS, SBR, PUR, and PS | Nanogram-level sensitivity (1–126 ng depending on polymer); example LODs: 1 ng for PA, PUR, and PS; 7 ng for PET; 10 ng for PC; 12 ng for PP; 60 ng for PE | Polymer concentrations reported in multiple matrices: 2–35 µg L⁻¹ in surface waters, 6.9–185.9 µg g⁻¹ dw in sediments, and 0.1–13.3 mg g⁻¹ dw in biota | Very high sensitivity and selectivity; internal-standard calibration; suitable for multiple matrices; CaCO₃-assisted pyrolysis improves signal intensity | Destructive; no particle-number, size, or morphology information; requires specialized pyrolyzer–triple quadrupole instrumentation | [136] |
| Stormwater workflow combining filtration, FTIR, and Pyr-GC/MS for MPs and TWPs | Operational fraction >25 µm | FTIR: PET and PP; Pyr-GC/MS: poly(butadiene) rubber and styrene–butadiene rubber (SBR) for TWPs | Inlet concentrations 3.8–59 MPs L⁻¹ and 2.5–58 TWPs L⁻¹ (0.4–4 mg L⁻¹ TWPs); outlet concentrations 1.8–32 MPs L⁻¹ and 1.3–32 TWPs L⁻¹ (0.2–1.1 mg L⁻¹ TWPs); removal 35–88% | Fragments dominated the MP profile (>94%); ~92% of fragments were black rubbery TWPs confirmed by Pyr-GC/MS; supports treatment-performance assessment | Combines particle counting with chemical confirmation; explicitly captures dense TWPs often missed by optical methods alone; directly useful for stormwater treatment benchmarking | Misses smaller MPs and NPs because of size cut-off; Pyr-GC/MS is destructive and increases analytical cost/time; FTIR is limited for black rubber; mass estimates remain method-dependent | [90] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




