Submitted:
05 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Periods and Data Sources
2.2. Sample
2.3. Data Extraction and Comparisons
2.4. Data Analysis
3. Results
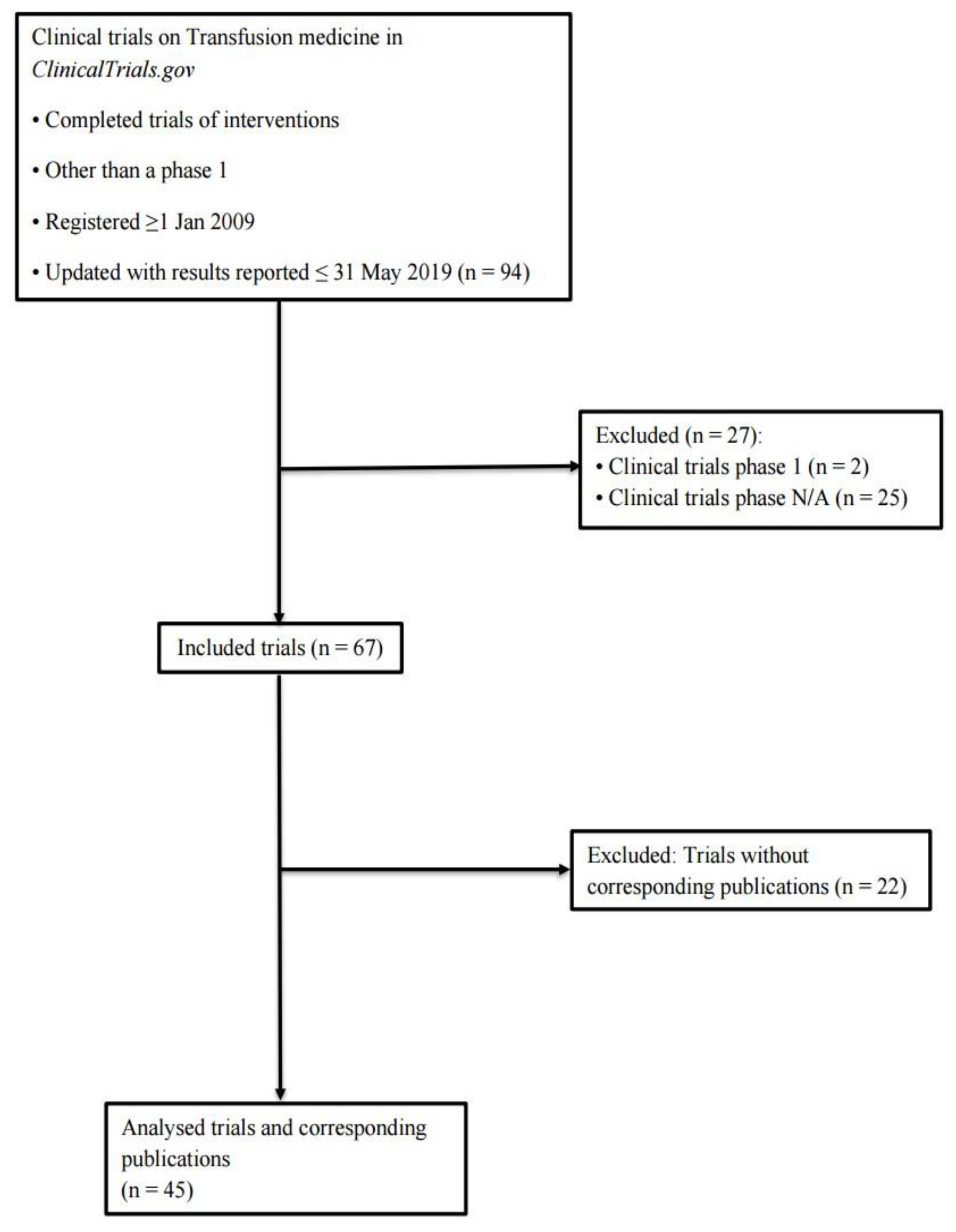
3.1. General Characteristics of Transfusion Medicine Clinical Trials from ClinicalTrials.gov
3.2. Completeness of WHO TRDS Reporting at Initial Registration Entry, Final Registration Update, and Corresponding Peer-Reviewed Publications in Transfusion Medicine Clinical Trials from ClinicalTrials.gov
3.3. Changes in WHO TRDS Reporting Between the Initial Registration Entry and Final Registration Update in Transfusion Medicine Clinical Trials from ClinicalTrials.gov
3.4. Changes in WHO TRDS Reporting Between the Final Registration Update and Corresponding Peer-Reviewed Journal Publications in Transfusion Medicine Clinical Trials from ClinicalTrials.gov
3.5. Adverse Event Reporting in Transfusion Medicine Clinical Trials from ClinicalTrials.gov and Corresponding Publications
3.6. Discrepancies in Serious Adverse Event Reporting for Transfusion Medicine Clinical Trials from ClinicalTrials.gov and Corresponding Publications
3.7. Discrepancies in Other Adverse Event Reporting for Transfusion Medicine Clinical Trials from ClinicalTrials.gov and Corresponding Publications
4. Discussion
5. Conclusion
Funding
Ethics approval and consent to participate
Consent for publication
Competing interests
Authors contributions
Data Availability Statement
References
- World Health Organization. Global Status Report on Blood Safety and Availability 2021; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/publications/i/item/9789240051683 (accessed on 16 January 2026).
- Jacobs, J.W.; Diaz, M.; Arevalo Salazar, D.E.; Tang, A.; Stephens, L.D.; Booth, G.S.; et al. United States blood pricing: A cross-sectional analysis of charges and reimbursement at 200 US hospitals. Am. J. Hematol. 2023, 98, E179–E182. [Google Scholar] [CrossRef]
- Lozano, M.; Cid, J. How do we forecast tomorrow’s transfusion: Non-transfusional hemotherapy. Transfus. Clin. Biol. 2023, 30, 282–286. [Google Scholar] [CrossRef]
- U.S. National Library of Medicine. Noninfectious Complications of Blood Transfusion. National Library of Medicine: Bethesda, MD, USA; Available online: https://www.ncbi.nlm.nih.gov/books/NBK574536/ (accessed on 16 January 2026).
- Garraud, O. How to reposition the benefit–risk balance to safely transfuse patients in non-vital situations? Transfus. Clin. Biol. 2019, 26, 171–173. [Google Scholar] [CrossRef] [PubMed]
- Goodnough, L.T.; Levy, J.H.; Murphy, M.F. Concepts of blood transfusion in adults. Lancet 2013, 381, 1845–1854. [Google Scholar] [CrossRef]
- Kleinman, S.; Caulfield, T.; Chan, P.; Davenport, R.; McFarland, J.; McPhedran, S.; et al. Toward an understanding of transfusion-related acute lung injury: Statement of a consensus panel. Transfusion 2004, 44, 1774–1789. [Google Scholar] [CrossRef] [PubMed]
- Carson, J.L.; Stanworth, S.J.; Roubinian, N.; Fergusson, D.A.; Triulzi, D.; Doree, C.; et al. Transfusion thresholds and other strategies for guiding allogeneic red blood cell transfusion. Cochrane Database Syst. Rev. 2016, 10, CD002042. [Google Scholar] [CrossRef] [PubMed]
- Garraud, O. Do we need more clinical trials in transfusion medicine and hemotherapy? Transfus. Apher. Sci. 2016, 55, 262–263. [Google Scholar] [CrossRef]
- Concato, J.; Shah, N.; Horwitz, R.I. Randomized, controlled trials, observational studies, and the hierarchy of research designs. N. Engl. J. Med. 2000, 342, 1887–1892. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D.; CONSORT Group. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef]
- Hopewell, S.; Clarke, M.; Moher, D.; Wager, E.; Middleton, P.; Altman, D.G.; et al. CONSORT for reporting randomized controlled trials in journal and conference abstracts: Explanation and elaboration. PLoS Med. 2008, 5, e20. [Google Scholar] [CrossRef]
- Wong, E.K.; Lachance, C.C.; Page, M.J.; Watt, J.; Veroniki, A.; Straus, S.E.; et al. Selective reporting bias in randomised controlled trials from two network meta-analyses: Comparison of clinical trial registrations and their respective publications. BMJ Open 2019, 9, e031138. [Google Scholar] [CrossRef]
- Talebi, R.; Redberg, R.F.; Ross, J.S. Consistency of trial reporting between ClinicalTrials.gov and corresponding publications: One decade after FDAAA. Trials 2020, 21, 675. [Google Scholar] [CrossRef]
- Jurić, D.; Pranić, S.; Tokalić, R.; Milat, A.M.; Mudnić, I.; Pavličević, I.; et al. Clinical trials on drug–drug interactions registered in ClinicalTrials.gov reported incongruent safety data in published articles: An observational study. J. Clin. Epidemiol. 2018, 104, 35–45. [Google Scholar] [CrossRef]
- Paladin, I.; Pranić, S.M. Reporting of the safety from allergic rhinitis trials registered on ClinicalTrials.gov and in publications: An observational study. BMC Med. Res. Methodol. 2022, 22, 262. [Google Scholar] [CrossRef]
- Krešo, A.; Grahovac, M.; Znaor, L.; Marušić, A. Safety reporting in trials on glaucoma interventions registered in ClinicalTrials.gov and corresponding publications. Sci. Rep. 2024, 14, 27762. [Google Scholar] [CrossRef]
- Pavić, M.; Tokalić, R.; Marušić, A. Poor registration and publication practices in clinical trials of targeted therapeutics for endocrine and metabolic diseases: An observational study. J. Clin. Epidemiol. 2024, 176, 111570. [Google Scholar] [CrossRef]
- Murphy, M.F.; Brunskill, S.; Estcourt, L.; Stanworth, S.; Dorée, C. How to further develop the evidence base for transfusion medicine. Blood Transfus. 2012, 10, 436–439. [Google Scholar] [CrossRef]
- Song, F.; Parekh, S.; Hooper, L.; Loke, Y.K.; Ryder, J.; Sutton, A.J.; et al. Dissemination and publication of research findings: An updated review of related biases. Health Technol. Assess. 2010, 14, 1–193. [Google Scholar] [CrossRef] [PubMed]
- Sim, I.; Chan, A.-W.; Gülmezoglu, A.M.; Evans, T.; Pang, T. Clinical trial registration: Transparency is the watchword. Lancet 2006, 367, 1631–1633. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. About the Results Database; U.S. National Library of Medicine: Bethesda, MD, USA; Available online: https://classic.clinicaltrials.gov/ct2/about-site/results (accessed on 16 January 2026).
- Zarin, D.A.; Tse, T. Trust but verify: Trial registration and determining fidelity to the protocol. Ann. Intern. Med. 2013, 159, 65–67. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health; Department of Health and Human Services. Clinical Trials Registration and Results Information Submission: Final Rule. Fed. Regist. 2016, 81, 64981–65157. Available online: https://www.federalregister.gov/documents/2016/09/21/2016-22129/clinical-trials-registration-and-results-information-submission (accessed on 16 January 2026).
- Mathieu, S.; Boutron, I.; Moher, D.; Altman, D.G.; Ravaud, P. Comparison of registered and published primary outcomes in randomized controlled trials. JAMA 2009, 302, 977–984. [Google Scholar] [CrossRef]
- Enrico, D.; Waisberg, F.; Burton, J.; Mandó, P.; Chacón, M. Analysis of adverse events attribution and reporting in cancer clinical trials: A systematic review. Crit. Rev. Oncol. Hematol. 2021, 160, 103296. [Google Scholar] [CrossRef] [PubMed]
- Madi, K.; Flumian, C.; Olivier, P.; Sommet, A.; Montastruc, F. Quality of reporting of adverse events in clinical trials of COVID-19 drugs: Systematic review. BMJ Med. 2023, 2, e000352. [Google Scholar] [CrossRef]
- Gates, A.; Caldwell, P.; Curtis, S.; Dans, L.; Fernandes, R.M.; Hartling, L.; et al. Reporting of data monitoring committees and adverse events in paediatric trials: A descriptive analysis. BMJ Paediatr. Open 2019, 3, e000426. [Google Scholar] [CrossRef] [PubMed]
- Junqueira, D.R.; Zorzela, L.; Golder, S.; Loke, Y.; Gagnier, J.J.; Julious, S.A.; et al. CONSORT Harms 2022 statement, explanation, and elaboration: Updated guideline for the reporting of harms in randomised trials. BMJ 2023, 381, e073725. [Google Scholar] [CrossRef] [PubMed]
- Hopewell, S.; Chan, A.-W.; Collins, G.S.; Hróbjartsson, A.; Moher, D.; Schulz, K.F.; et al. CONSORT 2025 statement: Updated guideline for reporting randomised trials. BMJ 2025, 389, e081123. [Google Scholar] [CrossRef]
- Jhaveri, P.; Bozkurt, S.; Moyal, A.; Belov, A.; Anderson, S.; Shan, H.; et al. Analyzing real-world data of blood transfusion adverse events: Opportunities and challenges. Transfusion 2022, 62, 1019–1026. [Google Scholar] [CrossRef]
- Gresham, G.; Meinert, J.L.; Gresham, A.G.; Piantadosi, S.; Meinert, C.L. Update on the clinical trial landscape: Analysis of ClinicalTrials.gov registration data, 2000–2020. Trials 2022, 23, 858. [Google Scholar] [CrossRef]
- STROBE Statement. Strengthening the Reporting of Observational Studies in Epidemiology; STROBE Initiative. Available online: https://www.strobe-statement.org (accessed on 16 January 2026).
- ClinicalTrials.gov. FDAAA 801 and the Final Rule; U.S. National Library of Medicine: Bethesda, MD, USA; Available online: https://clinicaltrials.gov/policy/fdaaa-801-final-rule (accessed on 16 January 2026).
- U.S. National Library of Medicine. Clinical Trial Registry Numbers in MEDLINE/PubMed Records. National Library of Medicine: Bethesda, MD, USA, 2019; Available online: https://www.nlm.nih.gov/pubs/techbull/mj05/mj05_ct_beta.html (accessed on 16 January 2026).
- World Health Organization. WHO Trial Registration Data Set (TRDS); World Health Organization: Geneva, Switzerland; Available online: https://www.who.int/tools/clinical-trials-registry-platform/network/who-data-set (accessed on 25 January 2026).
- Hartung, D.M.; Zarin, D.A.; Guise, J.M.; McDonagh, M.; Paynter, R.; Helfand, M. Reporting discrepancies between the ClinicalTrials.gov results database and peer-reviewed publications. Ann. Intern. Med. 2014, 160, 477–483. [Google Scholar] [CrossRef]
- Chen, R.; Desai, N.R.; Ross, J.S.; Zhang, W.; Chau, K.H.; Wayda, B.; et al. Publication and reporting of clinical trial results: Cross-sectional analysis across academic medical centers. BMJ 2016, 352, i637. [Google Scholar] [CrossRef]
- Ross, J.S.; Mulvey, G.K.; Hines, E.M.; Nissen, S.E.; Krumholz, H.M. Trial publication after registration in ClinicalTrials.gov: A cross-sectional analysis. PLoS Med. 2009, 6, e1000144. [Google Scholar] [CrossRef]
- Viergever, R.F.; Ghersi, D. The quality of registration of clinical trials. PLoS ONE 2011, 6, e14701. [Google Scholar] [CrossRef]
- Li, G.; Abbade, L.P.F.; Nwosu, I.; Jin, Y.; Leenus, A.; Maaz, M.; et al. A systematic review of comparisons between protocols or registrations and full reports in primary biomedical research. BMC Med. Res. Methodol. 2018, 18, 9. [Google Scholar] [CrossRef]
- Jones, C.W.; Keil, L.G.; Holland, W.C.; Caughey, M.C.; Platts-Mills, T.F. Comparison of registered and published outcomes in randomized controlled trials: A systematic review. BMC Med. 2015, 13, 282. [Google Scholar] [CrossRef]
- Wandalkar, P.; Gandhe, P.; Pai, A.; Limaye, M.; Chauthankar, S.; Gogtay, N.J.; et al. A study comparing trial registry entries of randomized controlled trials with publications of their results in a high impact factor journal: The Journal of the American Medical Association. Perspect. Clin. Res. 2017, 8, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.-W.; Krleža-Jerić, K.; Schmid, I.; Altman, D.G. Outcome reporting bias in randomized trials funded by the Canadian Institutes of Health Research. CMAJ 2004, 171, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Dwan, K.; Gamble, C.; Williamson, P.R.; Kirkham, J.J. Systematic review of the empirical evidence of study publication bias and outcome reporting bias: An updated review. PLoS ONE 2013, 8, e66844. [Google Scholar] [CrossRef]
- Kirkham, J.J.; Dwan, K.M.; Altman, D.G.; Gamble, C.; Dodd, S.; Smyth, R.; et al. The impact of outcome reporting bias in randomised controlled trials on a cohort of systematic reviews. BMJ 2010, 340, c365. [Google Scholar] [CrossRef] [PubMed]
- Golder, S.; Loke, Y.K.; Wright, K.; Norman, G. Reporting of adverse events in published and unpublished studies of health care interventions: A systematic review. PLoS Med. 2016, 13, e1002127. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A.; Lau, J. Completeness of safety reporting in randomized trials: An evaluation of 7 medical areas. JAMA 2001, 285, 437–443. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A.; Evans, S.J.W.; Gøtzsche, P.C.; O’Neill, R.T.; Altman, D.G.; Schulz, K.; et al. Better reporting of harms in randomized trials: An extension of the CONSORT statement. Ann. Intern. Med. 2004, 141, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Zorzela, L.; Loke, Y.K.; Ioannidis, J.P.A.; Golder, S.; Santaguida, P.; Altman, D.G.; et al. PRISMA harms checklist: Improving harms reporting in systematic reviews. BMJ 2016, 352, i157. [Google Scholar] [CrossRef]
- Vamvakas, E.C.; Blajchman, M.A. Transfusion-related mortality: The ongoing risks of allogeneic blood transfusion and the available strategies for their prevention. Blood 2009, 113, 3406–3417. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A. Meta-research: Why research on research matters. PLoS Biol. 2018, 16, e2005468. [Google Scholar] [CrossRef]
- Wieseler, B.; Wolfram, N.; McGauran, N.; Kerekes, M.F.; Vervölgyi, V.; Kohlepp, P.; et al. Completeness of reporting of patient-relevant clinical trial outcomes: Comparison of unpublished clinical study reports with publicly available data. PLoS Med. 2013, 10, e1001526. [Google Scholar] [CrossRef]
- Prayle, A.P.; Hurley, M.N.; Smyth, A.R. Compliance with mandatory reporting of clinical trial results on ClinicalTrials.gov: Cross-sectional study. BMJ 2012, 344, d7373. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, S.; Bouillon-Minois, J.B.; Renard Triché, L.; et al. Protocol publication rate and comparison between article, registry and protocol in RCTs. BMC Med. Res. Methodol. 2025, 25, 31. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Primary Registries in the WHO Registry Network; World Health Organization: Geneva, Switzerland; Available online: https://www.who.int/clinical-trials-registry-platform/network/primary-registries (accessed on 25 January 2026).
- Anderson, M.L.; Chiswell, K.; Peterson, E.D.; Tasneem, A.; Topping, J.; Califf, R.M. Compliance with results reporting at ClinicalTrials.gov. N. Engl. J. Med. 2015, 372, 1031–1039. [Google Scholar] [CrossRef]
- Riveros, C.; Dechartres, A.; Perrodeau, E.; Haneef, R.; Boutron, I.; Ravaud, P. Timing and completeness of trial results posted at ClinicalTrials.gov and published in journals. PLoS Med. 2013, 10, e1001566. [Google Scholar] [CrossRef]
- Goldacre, B.; Drysdale, H.; Dale, A.; Milosevic, I.; Slade, E.; Hartley, P.; et al. COMPare: A prospective cohort study correcting and monitoring 58 misreported trials in real time. Trials 2019, 20, 118. [Google Scholar] [CrossRef] [PubMed]
- Jerčić Martinić-Cezar, I.; Pranić, S.; Tavra, A.; Marušić, A. WHO TRDS Reporting and Adverse Event Comparison Dataset in Transfusion Medicine Clinical Trials. Zenodo. To be deposited upon publication.
| Baseline characteristics | No. of trials (%)a |
|---|---|
| Trial phase: | |
| 1/2 | 5 (7) |
| 2 | 28 (42) |
| 2/3 | 4 (6) |
| 3 | 20 (30) |
| 4 | 10 (15) |
| Maskingb: | |
| None (Open-label) | 34 (51) |
| Single-blind | 9 (13) |
| Double-blind | 7 (10) |
| Triple-blind | 7 (10) |
| Quadruple-blind | 10 (15) |
| Intervention model: | |
| Parallel assignment | 41 (61) |
| Single assignment | 22 (33) |
| Crossover assignment | 4 (6) |
| Primary sponsorc: | |
| National Institutes of Health | 2 (3) |
| Industry | 27 (40) |
| Community-based organization | 9 (13) |
| University | 29 (43) |
| Type of intervention: | |
| Drug | 39 (58) |
| Drug & Procedure | 5 (7) |
| Drug & Biological | 1 (1) |
| Drug & Radiation | 1 (1) |
| Drug & Other investigational products | 3 (4) |
| Procedure | 5 (7) |
| Procedure & Biological | 1 (1) |
| Biological | 9 (13) |
| Biological & Other investigational products | 1 (1) |
| Biological & Device | 1 (1) |
| Other investigational products | 1 (1) |
| Allocationd: | |
| Nonrandomized | 10 (15) |
| Randomized | 46 (69) |
| Not applicable | 11 (16) |
| Primary purpose: | |
| Treatment | 56 (84) |
| Prevention | 7 (10) |
| Supportive care | 1 (1) |
| Basic science | 1 (1) |
| Other | 1 (1) |
| Missing | 1 (1) |
| Main categoriesa | No. of trials (%)b |
|---|---|
| Blood components | 13 (19) |
| Haemostasis & Coagulation | 10 (15) |
| Immunohematology | 3 (4) |
| Patient Blood Management (PBM)c | 24 (36) |
| Chelation & Iron overload | 15 (22) |
| Transplantation | 2 (3) |
| Different time points for the included clinical trials | Median months (95% CI) |
|---|---|
| Initial registration entry to Study start datea | 0.00 (0.00 to 0.00) |
| Study start date to Study completion datea | 38.00 (33.00 to 49.71) |
| Primary completion date to Results posting datea | 19.77 (15.55 to 25.87) |
| Study completion date to Study publication dateb | 12.43 (8.97 to 21.18) |
| Results posting date to Study publication dateb | -3.77 (-16.52 to 3.50) |
| WHO TRDS items missing | Initial registration entry (n = 67) | Final registration update (n = 67) | Journal publication (n = 45) |
|---|---|---|---|
| NCT identifier | 0 (0) | 0 (0) | 6 (13) |
| Primary sponsor | 0 (0) | 0 (0) | 4 (9) |
| Public titlea | 0 (0) | 0 (0) | – |
| Scientific title | 4 (6) | 0 (0) | 0 (0) |
| Countries of recruitment | 9 (13) | 3 (4) | 4 (9) |
| Health condition studied | 0 (0) | 0 (0) | 0 (0) |
| Interventions | 0 (0) | 0 (0) | 0 (0) |
| Key inclusion criteria | 0 (0) | 0 (0) | 0 (0) |
| Key exclusion criteria | 2 (3) | 0 (0) | 0 (0) |
| Study typeb | 0 (0) | 0 (0) | 18 (40) |
| Date of first enrolment | 9 (13)c | 0 (0) | 25 (56)d |
| Sample size | 4 (6) | 0 (0) | 0 (0) |
| Key primary outcomes | 10 (15) | 0 (0) | 0 (0) |
| Key secondary outcomes | 17 (25) | 12 (18) | 0 (0) |
| Completion date | 22 (33)e | 0 (0) | 29 (64)f |
| IPD sharing statementg | – | – | 2 (25) |
| Registration stage | Trials with complete WHO TRDSa, n | Industry-sponsored, n (%) | Prospectively registered, n (%) |
|---|---|---|---|
| Initial registration | 22 | 5 (23) | 14 (64) |
| Final registration update | 52 | 25 (48) | 28 (54) |
| WHO TRDS items changed | Initial registration entry to final registration update (maximum n = 67), no. (%)ᵃ |
|---|---|
| Primary sponsor | 11/67 (16) |
| Public title | 19/67 (28) |
| Scientific title | 14/63 (22) |
| Countries of recruitment | 14/57 (25) |
| Health condition studied | 15/67 (22) |
| Interventions | 45/67 (67) |
| Key inclusion criteria | 27/67 (40) |
| Key exclusion criteria | 19/65 (29) |
| Study type | 0 (0) |
| Date of first enrolment | 20/58 (34) |
| Sample size | 55/63 (87) |
| Key primary outcomes | 49/57 (86) |
| Key secondary outcomes | 41/46 (89) |
| Completion date | 40/45 (89) |
| IPD sharing statement | ‒ |
| WHO TRDS items changed | Final registration entry to publication (maximum n = 45), no. (%) |
|---|---|
| NCT number | 0/39 (0) |
| Primary sponsorᵃ | 18/41 (44) |
| Countries of recruitment | 6/41 (15) |
| Health condition studied | 1/45 (2) |
| Interventions | 1/45 (2) |
| Key inclusion criteria | 31/45 (69) |
| Key exclusion criteria | 35/45 (78) |
| Study typeb | 22/45 (49) |
| Date of first enrolmentc | 10/20 (50) |
| Sample size | 17/45 (38) |
| Key primary outcomesd | 8/45 (18) |
| Key secondary outcomese | 30/45 (67) |
| Completion datef | 8/16 (50) |
| ClinicalTrials.gov, no. (%) | Publications, no. (%) | |
| AEs >0 reported (n = 45)ᵃ | ||
| SAEs | 31 (69) | 26 (58) |
| OAEs | 32 (71) | 29 (64) |
| SAEs and OAEs not separately reported or not explicitly reported | 1 (2)b | 9 (20)c |
| Deaths | 20 (44) | 27 (60) |
| Deaths reported in ClinicalTrials.gov (n = 20) | ||
| In the All-Cause mortality field | 8 (40) | – |
| In outcome results or participant flow | 8 (40) | – |
| In the adverse event module | 4 (20) | – |
| AEs reported as zero (n = 45) | ||
| SAEs | 12 (27) | 4 (9) |
| OAEs | 11 (24) | 5 (11) |
| Deaths | 6 (13) | 3 (7) |
| No deaths reported | 19 (42) | 15 (33) |
| Number of patients with AEs per trial (median, IQR/range)ᵈ,ᵉ | ||
| SAEs | 17, 0–72 / 0–365 | 16, 3–66 / 0–1025 |
| OAEs | 33, 0–130 / 0–607 | 40, 0–168 / 0–607 |
| Number of patients with SAEs (n = 45) | no. (%) |
|---|---|
| Yes | 21 (47) |
| No | 13 (29) |
| Unable to determine or not applicablea | 11 (24) |
| Among trials with differences (n = 21) | |
| More in the registry | 12 (57) |
| More in the publication | 9 (43) |
| Number of SAEs that differ between sources (n = 45) | |
| Yes | 24 (53) |
| No | 11 (24) |
| Unable to determine or not applicableb | 10 (22) |
| Among trials with differences (n = 24) | |
| More in the registry | 19 (79) |
| More in the publication | 5 (21) |
| Different description of SAEs (n = 45) | |
| Yes | 21 (47) |
| No | 8 (18) |
| Unable to determine or not applicablec | 16 (35) |
| Omission of 1 or more registered SAEs in publications (n = 45) | |
| Yes | 14 (31) |
| No | 22 (49) |
| Unable to determine or not applicabled | 9 (20) |
| Number of patients with OAEs (n = 45) | no. (%) |
|---|---|
| Yes | 13 (29) |
| No | 9 (20) |
| Unable to determine or not applicablea | 23 (51) |
| Among trials with differences (n = 13) | |
| More in the registry | 4 (31) |
| More in the publication | 9 (69) |
| Number of OAEs that differ between sources (n = 45) | |
| Yes | 14 (31) |
| No | 9 (20) |
| Unable to determine or not applicableb | 22 (49) |
| Among trials with differences (n = 14) | |
| More in the registry | 8 (57) |
| More in the publication | 6 (43) |
| Different description of OAEs (n = 45) | |
| Yes | 14 (31) |
| No | 7 (16) |
| Unable to determine or not applicablec | 24 (53) |
| Type of reporting (n = 45) | |
| Reported as an AE with quantifiable values | 30 (67) |
| Reported as an AE without quantifiable values | 10 (22) |
| Reported as TEAE only | 4 (9) |
| Reported as ADR only | 1 (2) |
| Frequency threshold (n = 45) | |
| Same in both sources | 2 (4) |
| Higher in registry | 1 (2) |
| Higher in publication | 2 (4) |
| Unstated in publications | 40 (89) |
| Omission of 1 or more registered OAEs in publications (n = 45) | |
| Yes | 18 (40) |
| No | 15 (33) |
| Unable to determine or not applicabled | 12 (27) |
| Among trials with omission (n = 18) | |
| Threshold reporting | |
| Yes | 4 (22) |
| No | 14 (77) |
| TEAE-only reporting | |
| Yes | 6 (33) |
| No | 12 (66) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




