Submitted:
04 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
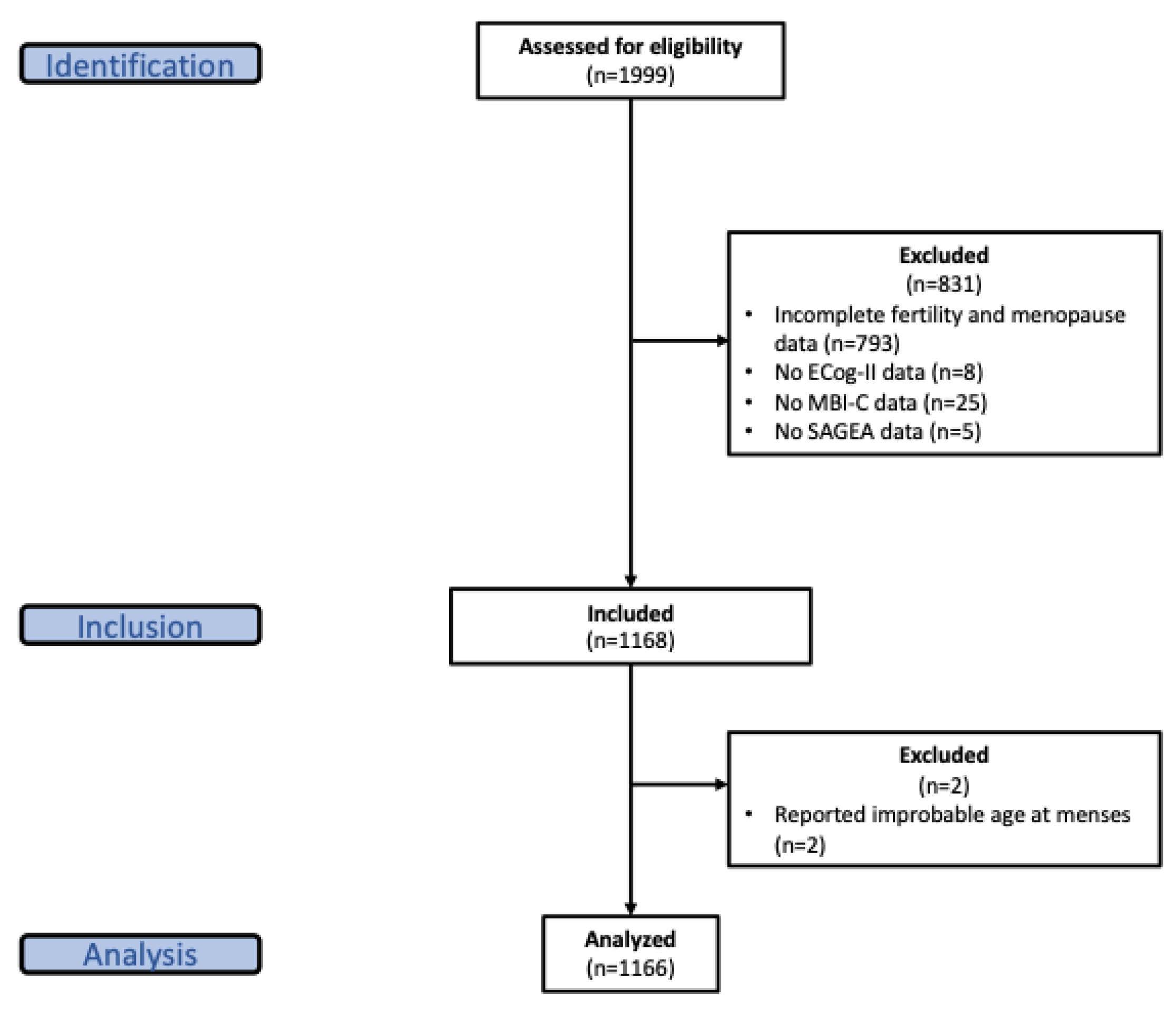
2.1. Study Design
2.2. Participants
2.3. Measures
2.3.1. Lifetime Exposure to Endogenous E2
2.3.2. MHT Use
2.3.3. E2-Related Reproductive Variables
2.3.4. Objective Cognition
2.3.5. Subjective Cognition
2.3.6. Subjective Behaviour
2.3.7. Subjective Function
2.4. Statistical Analysis
3. Results
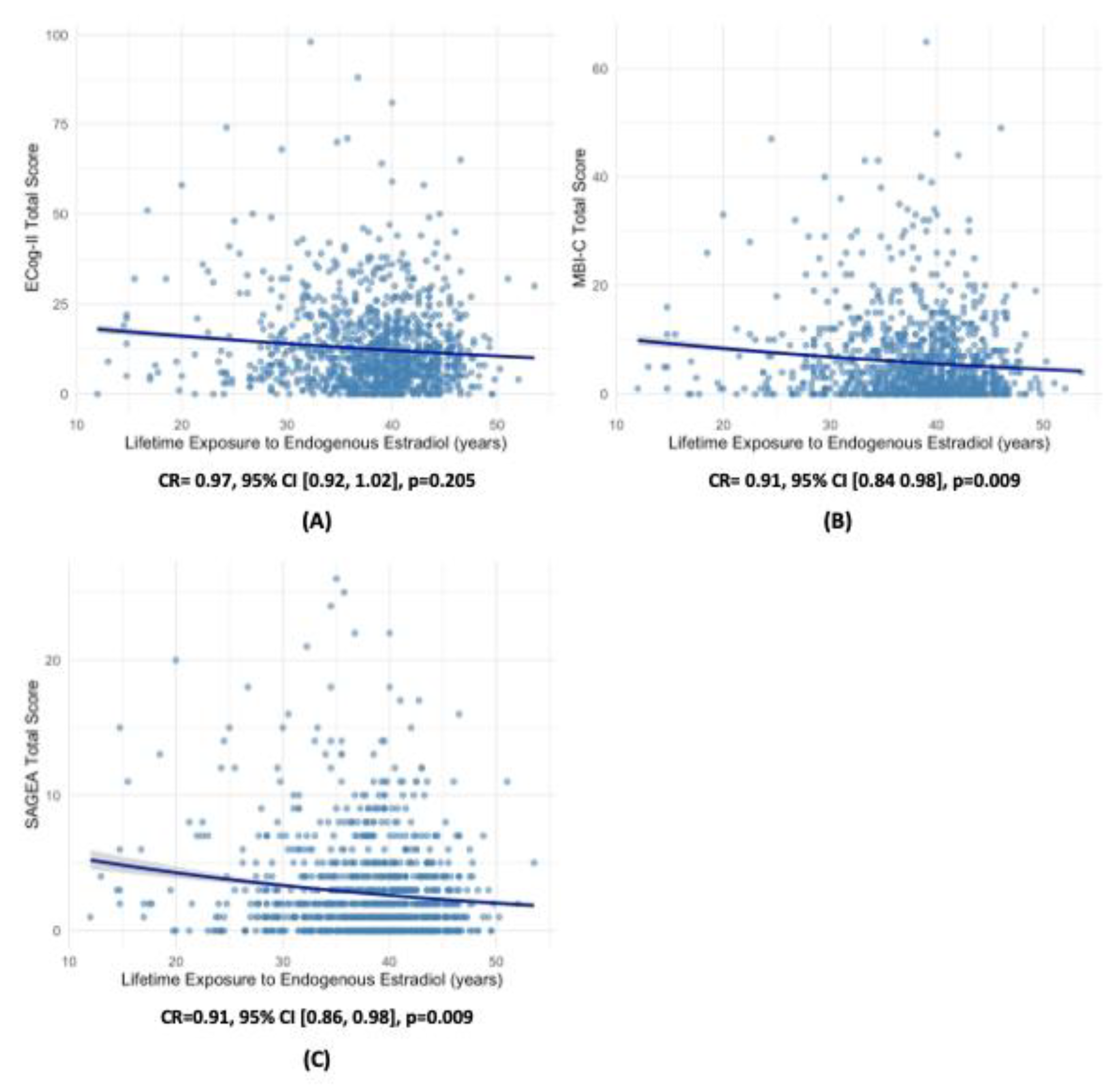
3.1. LEE2 and Cognition
3.2. LEE2 and Behaviour
3.4. LEE2 and Function
3.5. MHT Use
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| AD | Alzheimer disease |
| E2 | Estradiol |
| LEE2 | Lifetime exposure to endogenous estradiol |
| NPS | Neuropsychiatric symptoms |
| MBI | Mild behavioral impairment |
| MHT | Menopause hormone therapy |
| CAN-PROTECT | Canadian Platform for Research Online to Investigate Health, Quality of Life, Cognition, Behaviour, Function, and Caregiving in Aging |
| ECog-II | Revised Everyday Cognition scale |
| MBI-C | Mild Behavioral Impairment Checklist |
| SAGEA | Standard Assessment of Global Everyday Activities |
| CR | Count ratio |
References
- Frisoni, GB; Hansson, O; Nichols, E; et al. New landscape of the diagnosis of Alzheimer’s disease. The Lancet 2025. [Google Scholar] [CrossRef] [PubMed]
- Nichols, E; Steinmetz, JD; Vollset, SE; et al. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: An analysis for the Global Burden of Disease Study 2019. The Lancet Public Health 2022, 7(2), e105–e125. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, MA. Women and the risk of Alzheimer’s disease. Frontiers in Global Women’s Health 2024, 4, 1324522. [Google Scholar] [CrossRef] [PubMed]
- Castro-Aldrete, L; Einsiedler, M; Novakova Martinkova, J; et al. Alzheimer disease seen through the lens of sex and gender. Nature Reviews Neurology 2025, 1–15. [Google Scholar] [CrossRef]
- Zhang, Y; Chen, J; Li, Y; Jiao, B; Luo, S. Disease-modifying therapies for Alzheimer’s disease: Clinical trial progress and opportunity. Ageing research reviews 2025, 103, 102595. [Google Scholar] [CrossRef]
- Smith, EE; Phillips, NA; Feldman, HH; et al. Use of lecanemab and donanemab in the Canadian healthcare system: Evidence, challenges, and areas for future research. The journal of prevention of Alzheimer’s disease 2025, 12(3), 100068. [Google Scholar] [CrossRef]
- Wang, X; Feng, S; Deng, Q; Wu, C; Duan, R; Yang, L. The role of estrogen in Alzheimer’s disease pathogenesis and therapeutic potential in women. Molecular and Cellular Biochemistry 2024, 1–16. [Google Scholar] [CrossRef]
- Thurston, RC; Chang, Y; Wu, M; et al. Reproductive hormones in relation to white matter hyperintensity volumes among midlife women. Alzheimer’s & Dementia 2024, 20(9), 6161–6169. [Google Scholar] [CrossRef]
- Smejkalova, T; Woolley, CS. Estradiol acutely potentiates hippocampal excitatory synaptic transmission through a presynaptic mechanism. Journal of Neuroscience 2010, 30(48), 16137–16148. [Google Scholar] [CrossRef]
- Bendis, PC; Zimmerman, S; Onisiforou, A; Zanos, P; Georgiou, P. The impact of estradiol on serotonin, glutamate, and dopamine systems. Frontiers in neuroscience 2024, 18, 1348551. [Google Scholar] [CrossRef]
- Au, A; Feher, A; McPhee, L; Jessa, A; Oh, S; Einstein, G. Estrogens, inflammation and cognition. Frontiers in neuroendocrinology 2016, 40, 87–100. [Google Scholar] [CrossRef]
- Oughli, HA; Nguyen, SA; Siddarth, P; et al. The effect of cumulative lifetime estrogen exposure on cognition in depressed versus non-depressed older women. Journal of geriatric psychiatry and neurology 2022, 35(6), 832–839. [Google Scholar] [CrossRef] [PubMed]
- Dukic, J; Johann, A; Henninger, M; Ehlert, U. Estradiol and progesterone from pregnancy to postpartum: A longitudinal latent class analysis. Frontiers in Global Women’s Health 2024, 5, 1428494. [Google Scholar] [CrossRef] [PubMed]
- Cote, S; Perron, T-L; Baillargeon, J-P; Bocti, C; Lepage, J-F; Whittingstall, K. Association of cumulative lifetime exposure to female hormones with cerebral small vessel disease in postmenopausal women in the UK biobank. Neurology 2023, 101(20), e1970–e1978. [Google Scholar] [CrossRef] [PubMed]
- Matyi, JM; Rattinger, GB; Schwartz, S; Buhusi, M; Tschanz, JT. Lifetime estrogen exposure and cognition in late life: The Cache County Study. Menopause 2019, 26(12), 1366–1374. [Google Scholar] [CrossRef]
- Park, HK; Marston, L; Mukadam, N. The effects of estrogen on the risk of developing dementia: A cohort study using the UK biobank data. The American Journal of Geriatric Psychiatry 2024, 32(7), 792–805. [Google Scholar] [CrossRef]
- Huang, T; Shafrir, AL; Eliassen, AH; Rexrode, KM; Tworoger, SS. Estimated Number of Lifetime Ovulatory Years and Its Determinants in Relation to Levels of Circulating Inflammatory Biomarkers. American Journal of Epidemiology 2019, 189(7), 660–670. [Google Scholar] [CrossRef]
- Borda, MG; Aarsland, D; Tovar-Rios, DA; et al. Neuropsychiatric symptoms and functional decline in Alzheimerʼs disease and Lewy body dementia. Journal of the American Geriatrics Society 2020, 68(10), 2257–2263. [Google Scholar] [CrossRef]
- Ismail, Z; Smith, EE; Geda, Y; et al. Neuropsychiatric symptoms as early manifestations of emergent dementia: Provisional diagnostic criteria for mild behavioral impairment. Alzheimer’s & Dementia 2016, 12(2), 195–202. [Google Scholar]
- Guan, DX; Rehman, T; Nathan, S; et al. Neuropsychiatric symptoms: Risk factor or disease marker? A study of structural imaging biomarkers of Alzheimer’s disease and incident cognitive decline. Human Brain Mapping 2024, 45(13), e70016. [Google Scholar] [CrossRef]
- Ismail, Z; Leon, R; Creese, B; Ballard, C; Robert, P; Smith, EE. Optimizing detection of Alzheimer’s disease in mild cognitive impairment: A 4-year biomarker study of mild behavioral impairment in ADNI and MEMENTO. Molecular Neurodegeneration 2023, 18(1), 50. [Google Scholar] [CrossRef] [PubMed]
- Leon, R; Ghahremani, M; Guan, DX; Smith, EE; Zetterberg, H; Ismail, Z. Enhancing Alzheimer Disease Detection Using Neuropsychiatric Symptoms: The Role of Mild Behavioural Impairment in the Revised NIA-AA Research Framework. Journal of Geriatric Psychiatry and Neurology 2025, 08919887251366634. [Google Scholar] [CrossRef] [PubMed]
- Ghahremani, M; Leon, R; Smith, EE; Ismail, Z. Exploring the association between mild behavioral impairment and plasma p-tau217: Implications for early detection of Alzheimer’s disease. Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring 2025, 17(2), e70119. [Google Scholar]
- Ghahremani, M; Smith, EE; Ismail, Z. Persistent functional impairment as an early indicator of cognitive decline and dementia in cognitively normal older adults. J Alzheimers Dis 2025, 13872877251406661. [Google Scholar] [CrossRef]
- Ghahremani, M; Smith, EE; Ismail, Z. Persistent Functional Impairment as an Early Indicator of Alzheimer Disease Pathology and Progression. Journal of the American Geriatrics Society 2025. [Google Scholar] [CrossRef]
- Vassilaki, M; Aakre, JA; Kremers, WK; et al. Association between functional performance and Alzheimer’s disease biomarkers in individuals without dementia. Journal of the American Geriatrics Society 2018, 66(12), 2274–2281. [Google Scholar] [CrossRef]
- Arruda, F; Rosselli, M; Greig, MT; et al. The association between functional assessment and structural brain biomarkers in an ethnically diverse sample with normal cognition, mild cognitive impairment, or dementia. Archives of Clinical Neuropsychology 2021, 36(1), 51–61. [Google Scholar] [CrossRef]
- van Zwol-Janssens, C; Louwers, YV; Laven, JS; Schipper, J; Jiskoot, G. Depressive symptoms in women with premature ovarian insufficiency (POI): A cross-sectional observational study. Menopause 2024, 10.1097. [Google Scholar] [CrossRef]
- Velez, MP; Alvarado, BE; Rosendaal, N; et al. Age at natural menopause and physical functioning in postmenopausal women: The Canadian Longitudinal Study on Aging. Menopause 2019, 26(9), 958–965. [Google Scholar] [CrossRef]
- Nerattini, M; Jett, S; Andy, C; et al. Systematic review and meta-analysis of the effects of menopause hormone therapy on risk of Alzheimer’s disease and dementia. Frontiers in aging neuroscience 2023, 15, 1260427. [Google Scholar] [CrossRef]
- Rocca, WA; Kantarci, K; Faubion, SS. Risks and benefits of hormone therapy after menopause for cognitive decline and dementia: A conceptual review. Maturitas 2024, 108003. [Google Scholar] [CrossRef]
- Ismail, Z; Guan, DX; Vellone, D; et al. The Canadian platform for research online to investigate health, quality of life, cognition, behaviour, function, and caregiving in aging (CAN-PROTECT): Study protocol, platform description, and preliminary analyses. Aging and Health Research 2024, 4(4), 100207. [Google Scholar] [CrossRef]
- Guan, DX; Aundhakar, A; Tomaszewski Farias, S; et al. Vascular risk factor associations with subjective cognitive decline and mild behavioural impairment. Brain Communications 2025, 7(3), fcaf163. [Google Scholar] [CrossRef] [PubMed]
- Guan, DX; Peters, ME; Pike, GB; et al. Cognitive, behavioral, and functional outcomes of suspected mild traumatic brain injury in community-dwelling older persons without mild cognitive impairment or dementia. Journal of the Academy of Consultation-Liaison Psychiatry 2025, 66(2), 118–129. [Google Scholar] [CrossRef] [PubMed]
- Mudalige, D; Guan, DX; Ballard, C; et al. The mind and motion: Exploring the interplay between physical activity and Mild Behavioral Impairment in dementia-free older adults. International Review of Psychiatry 2024, 36(3), 196–207. [Google Scholar] [CrossRef]
- Brooker, H; Williams, G; Hampshire, A; et al. FLAME: A computerized neuropsychological composite for trials in early dementia. Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring 2020, 12(1), e12098. [Google Scholar] [CrossRef]
- Farias, ST; Weakley, A; Harvey, D; Chandler, J; Huss, O; Mungas, D. The measurement of Everyday Cognition (ECog): Revisions and updates. Alzheimer Disease & Associated Disorders 2021, 35(3), 258–264. [Google Scholar]
- Ismail, Z; Agüera-Ortiz, L; Brodaty, H; et al. The Mild Behavioral Impairment Checklist (MBI-C): A rating scale for neuropsychiatric symptoms in pre-dementia populations. Journal of Alzheimer’s disease 2017, 56(3), 929–938. [Google Scholar] [CrossRef]
- Marzona, I. The Standard Assessment of Global Activities in the Elderly (SAGE) Scale: Validation process of a new tool for the assessment of disability in older adults; 2011. [Google Scholar]
- Low, L-F; Anstey, K; Jorm, A; Rodgers, B; Christensen, H. Reproductive period and cognitive function in a representative sample of naturally postmenopausal women aged 60–64 years. Climacteric 2005, 8(4), 380–389. [Google Scholar] [CrossRef]
- Georgakis, MK; Kalogirou, EI; Diamantaras, A-A; et al. Age at menopause and duration of reproductive period in association with dementia and cognitive function: A systematic review and meta-analysis. Psychoneuroendocrinology 2016, 73, 224–243. [Google Scholar] [CrossRef]
- Fox, M; Berzuini, C; Knapp, LA. Cumulative estrogen exposure, number of menstrual cycles, and Alzheimer’s risk in a cohort of British women. Psychoneuroendocrinology 2013, 38(12), 2973–2982. [Google Scholar] [CrossRef]
- Ryan, J; Carrière, I; Scali, J; Ritchie, K; Ancelin, M-L. Life-time estrogen exposure and cognitive functioning in later life. Psychoneuroendocrinology 2009, 34(2), 287–298. [Google Scholar] [CrossRef] [PubMed]
- Jett, S; Malviya, N; Schelbaum, E; et al. Endogenous and exogenous estrogen exposures: How women’s reproductive health can drive brain aging and inform Alzheimer’s prevention. Frontiers in Aging Neuroscience 2022, 14, 831807. [Google Scholar] [CrossRef] [PubMed]
- Guan, DX; Mortby, ME; Pike, GB; et al. Linking cognitive and behavioral reserve: Evidence from the CAN-PROTECT study. Alzheimers Dement (N Y) 2024, 10(4), e12497. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q; Yan, Y; La, R; et al. Association of reproductive lifespan and age at menopause with depression: Data from NHANES 2005–2018. Journal of Affective Disorders 2024, 356, 519–527. [Google Scholar] [CrossRef]
- Georgakis, MK; Thomopoulos, TP; Diamantaras, A-A; et al. Association of age at menopause and duration of reproductive period with depression after menopause: A systematic review and meta-analysis. JAMA psychiatry 2016, 73(2), 139–149. [Google Scholar] [CrossRef]
- Crockford, JF; Guan, DX; Einstein, G; et al. Menopausal symptom burden as a predictor of mid-to late-life cognitive function and mild behavioral impairment symptoms: A CAN-PROTECT study. PLoS ONE 2025, 20(3), e0301165. [Google Scholar] [CrossRef]
- Al-Azzawi, F; Palacios, S. Hormonal changes during menopause. Maturitas 2009, 63(2), 135–137. [Google Scholar] [CrossRef]
- Tseng, LA; El Khoudary, SR; Young, EA; et al. The association of menopause status with physical function: The Study of Women’s Health Across the Nation. Menopause 2012, 19(11), 1186–1192. [Google Scholar] [CrossRef]
- Sowers, M; Tomey, K; Jannausch, M; Eyvazzadeh, A; Nan, B; Randolph, J., Jr. Physical functioning and menopause states. Obstetrics & Gynecology 2007, 110(6), 1290–1296. [Google Scholar] [CrossRef]
- Mosconi, L; Berti, V; Dyke, J; et al. Menopause impacts human brain structure, connectivity, energy metabolism, and amyloid-beta deposition. Scientific reports 2021, 11(1), 10867. [Google Scholar] [CrossRef]
- Aittokallio, J; Saaresranta, T; Riskumäki, M; et al. Effect of menopause and age on vascular impairment. Maturitas 2023, 169, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Karlamangla, AS; Burnett-Bowie, S-AM; Crandall, CJ. Bone health during the menopause transition and beyond. Obstetrics and gynecology clinics of North America 2018, 45(4), 695. [Google Scholar] [CrossRef] [PubMed]
- Wright, VJ; Schwartzman, JD; Itinoche, R; Wittstein, J. The musculoskeletal syndrome of menopause. Climacteric 2024, 27(5), 466–472. [Google Scholar] [CrossRef] [PubMed]
- Greendale, GA; Huang, MH; Wight, RG; et al. Effects of the menopause transition and hormone use on cognitive performance in midlife women. Neurology 2009, 72(21), 1850–1857. [Google Scholar] [CrossRef]
- Calvo, N; Einstein, G. Steroid hormones: Risk and resilience in women’s Alzheimer disease. Frontiers in Aging Neuroscience 2023, 15, 1159435. [Google Scholar] [CrossRef]
- Lee, JK; Frank, RD; Christenson, LR; Fields, JA; Rocca, WA; Mielke, MM. Associations of reproductive factors and exogenous estrogens with global and domain-specific cognition in later life. Alzheimer’s & Dementia 2024, 20(1), 63–73. [Google Scholar]
- Watts, A; Donofry, S; Ripperger, H; et al. Lifetime estrogen exposure and domain-specific cognitive performance: Results from the IGNITE study. Frontiers in Aging Neuroscience 2025, 17, 1524474. [Google Scholar] [CrossRef]
- Puri, TA; Gravelsins, LL; Alexander, MW; et al. Association between menopause age and estradiol-based hormone therapy with cognitive performance in cognitively normal women in the CLSA. Neurology 2025, 105(6), e213995. [Google Scholar] [CrossRef]
- Mosconi, L; Andy, C; Nerattini, M; et al. Systematic Review and Meta-analysis of Menopause Hormone Therapy (MHT) and the Risk of Alzheimer’s Disease and All-cause Dementia: Effects of MHT Characteristics, Location, and APOE-4 Status. Current Obstetrics and Gynecology Reports 2025, 14(1), 6. [Google Scholar] [CrossRef]
- Campbell, AJ; Claydon, VE; Liva, S; Cote, AT. Changes in Canadian contraceptive choices: Results of a national survey on hormonal contraceptive use. BMC Women’s Health 2025, 25(1), 147. [Google Scholar] [CrossRef]
- Bae, J; Lipnicki, D; Han, J; et al. Parity and the risk of incident dementia: A COSMIC study. Epidemiology and psychiatric sciences 2020, 29, e176. [Google Scholar] [CrossRef]
- Bae, JB; Lipnicki, DM; Han, JW; et al. Does parity matter in women’s risk of dementia? A COSMIC collaboration cohort study. BMC medicine 2020, 18(1), 210. [Google Scholar] [CrossRef]
- Fu, C; Hao, W; Ma, Y; et al. Number of live births, age at the time of having a child, span of births and risk of dementia: A population-based cohort study of 253,611 UK women. Journal of women’s health 2023, 32(6), 680–692. [Google Scholar] [CrossRef]
| Variable | M(SD), Range | N(%) |
|---|---|---|
| Age (years) | 64.0(7.4), 43.0-88.0 | --- |
| Education (years) | 15.8(4.5), 1.0-30.0 | --- |
| Ethnocultural background | ||
| European origins | --- | 993(85.2) |
| Non-European origins | --- | 173(14.8) |
| Menarche age (years) | 12.7(1.5), 8.0-23.0 | --- |
| Menopause age (years) | 49.6(5.8), 21.0-65.0 | --- |
| Menopause type | ||
| Spontaneous | --- | 887(76.1) |
| Surgical | --- | 187(16.0) |
| Other reasons | --- | 92(7.9) |
| Biological children (number) | ||
| 0 children | --- | 270(23.2) |
| 1 child | --- | 162(13.9) |
| 2 children | --- | 475(40.7) |
| 3 children | --- | 207(17.8) |
| 4 children | --- | 44(3.8) |
| 5 children | --- | 5(0.4) |
| 6+ children | --- | 3(0.3) |
| Time pregnant (years) | 1.3(0.9), 0-4.5 | --- |
| LEE2 (years) | 38.2(5.9), 12.0-53.5 | |
| MHT ever use | --- | 410(35.2) |
| ECog-II score | 12.5(11.7), 0.0-98.0 | --- |
| MBI-C score | 5.9(7.7), 0.0-65.0 | --- |
| SAGE score | 2.8(3.5), 0.0-26.0 | --- |
| Variable | b Coefficient | 95% CI [2.5, 97.5] | p Value |
|---|---|---|---|
| Global score | 0.00 | [-0.03, 0.02] | 0.895 |
| Trail Making B | 0.01 | [-0.02, 0.04] | 0.546 |
| Switching Stroop | 0.02 | [-0.02, 0.07] | 0.349 |
| Self-Ordered Search | 0.00 | [-0.04, 0.05] | 0.915 |
| Paired Associate Learning | -0.03 | [-0.07, 0.01] | 0.111 |
| Verbal Reasoning | -0.01 | [-0.06, 0.04] | 0.643 |
| Digit Span | 0.00 | [-0.04, 0.04] | 0.986 |
| Variable | CR | b | 95% CI [2.5, 97.5] | p-Value |
|---|---|---|---|---|
| Global neuropsychological score | --- | 0.04 | [-0.02, 0.09] | 0.161 |
| Trail Making B | --- | -0.01 | [-0.07, 0.05] | 0.780 |
| Switching Stroop | --- | 0.04 | [-0.05, 0.14] | 0.376 |
| Self-Ordered Search | --- | 0.01 | [-0.10, 0.11] | 0.918 |
| Paired Associate Learning | --- | 0.04 | [-0.05, 0.13] | 0.411 |
| Verbal Reasoning | --- | 0.13 | [0.02, 0.23] | 0.016 |
| Digit Span | --- | 0.03 | [-0.04, 0.11] | 0.407 |
| ECog-II total score | 1.00 | --- | [0.89, 1.12] | 0.969 |
| MBI-C total score | 0.88 | --- | [0.75, 1.03] | 0.104 |
| SAGEA total score | 1.02 | --- | [0.88, 1.18] | 0.782 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




