Submitted:
05 April 2026
Posted:
06 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Human Samples
2.2. Experimental Animals
2.3. Ang II Induced Atrial Fibrillation Murine Models with Peptide Treatment
2.4. Isolation and Culture of Primary Rat Atrial Cells
2.5. Drug Treatment Protocol
2.6. Histology and Immunohistochemistry
2.7. Measurement of Intracellular Reactive Oxygen Species (ROS)
2.8. Western Blot Analysis
2.9. Quantitative Real-Time Polymerase Chain Reaction
2.10. Superoxide Dismutase (SOD)
2.11. Transesophageal Programmed Electrical Stimulation
2.12. Cell Proliferation Assay (CCK-8)
2.13. Wound Healing Assay
2.14. Bioinformatic Analysis of Spatial Transcriptomic Data
2.15. RNA Sequencing of Primary Cardiac Fibroblasts
2.16. Outcome Measures
2.17. Data Analysis and Statistical Methods
3. Results
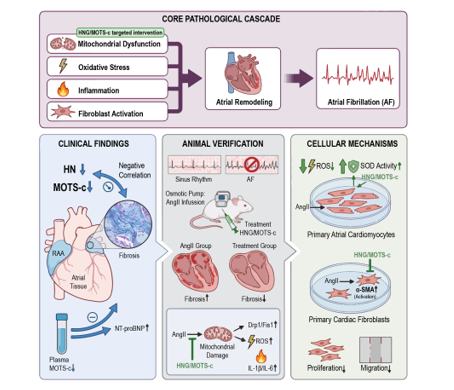
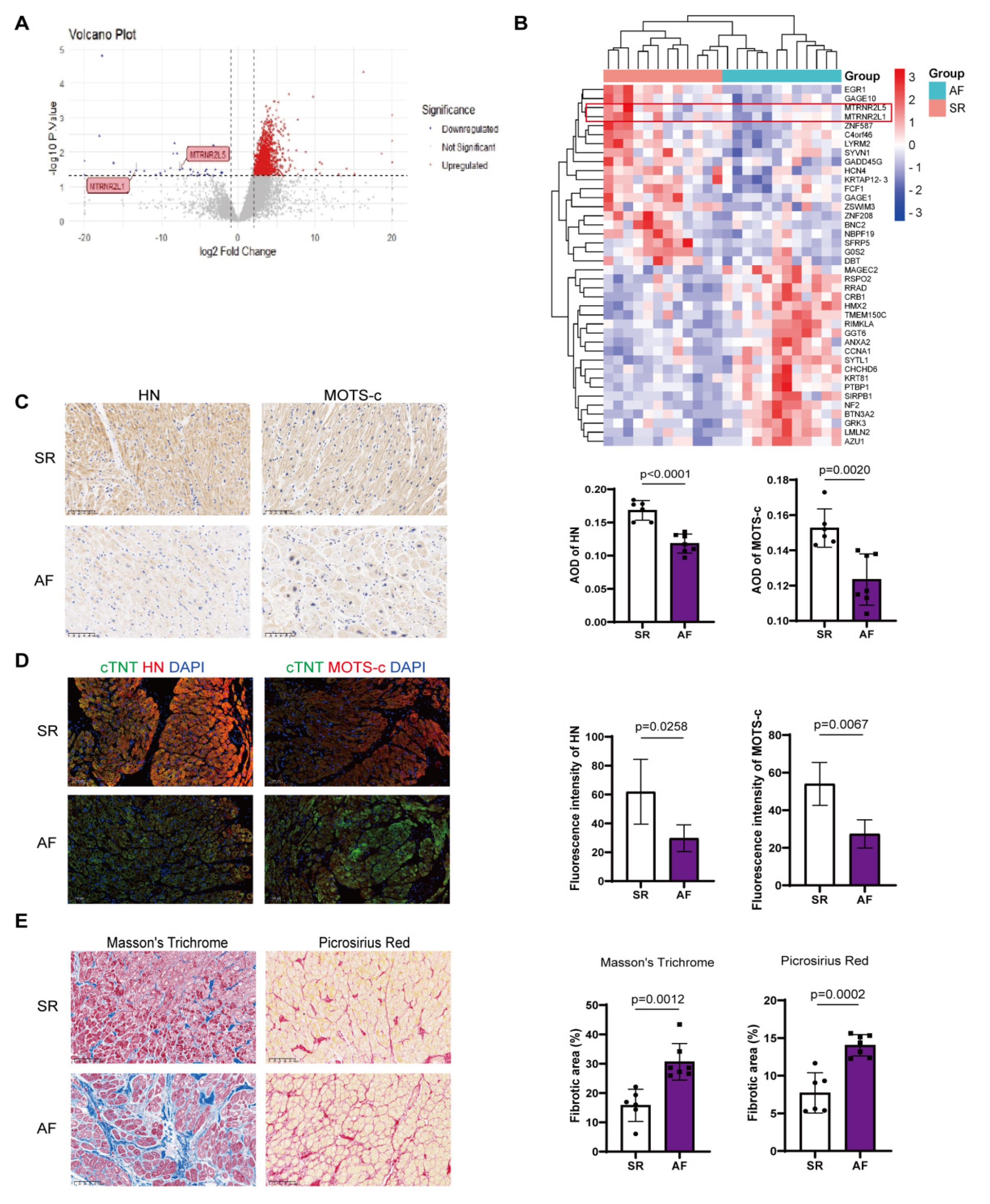
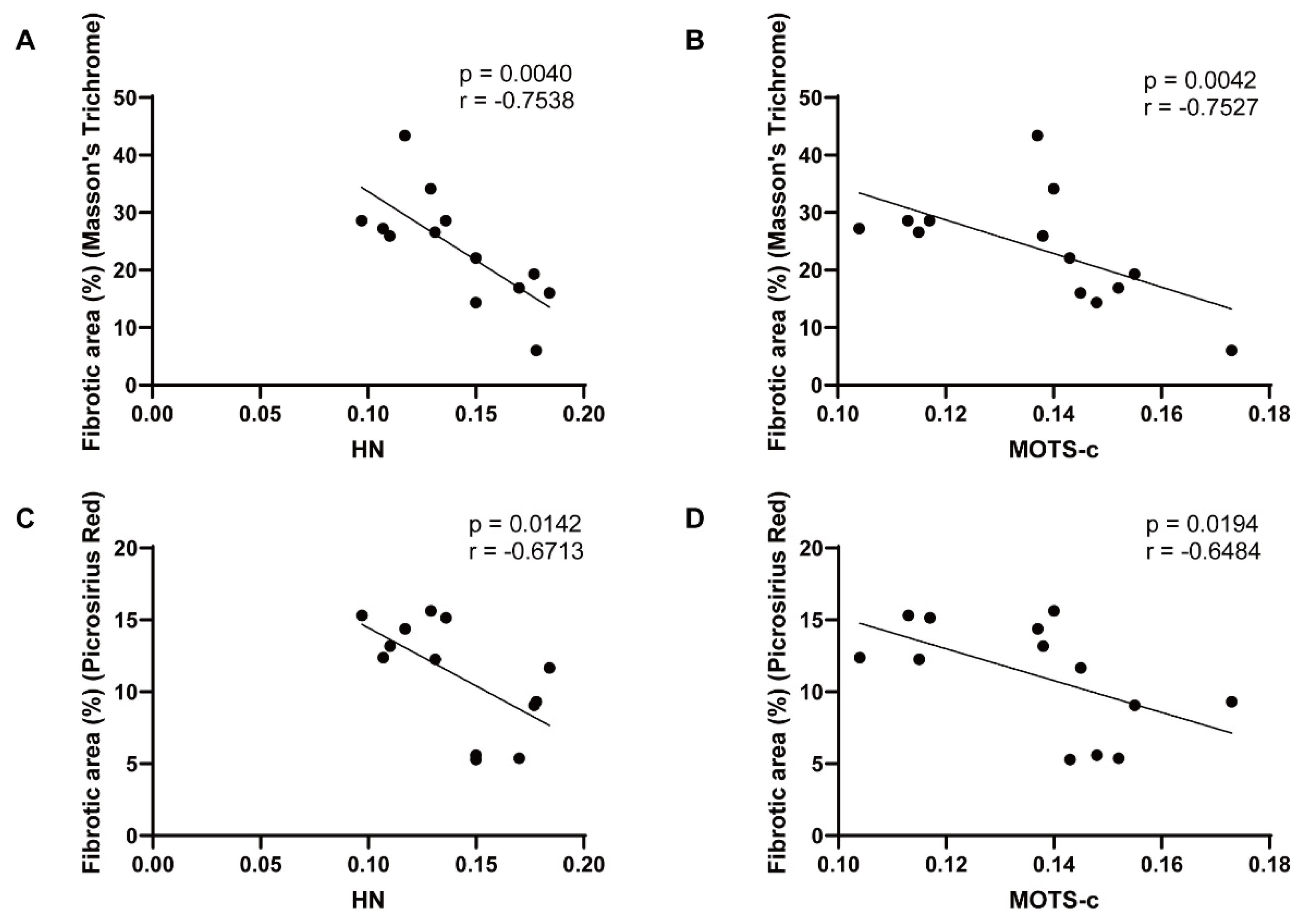
3.1. Both Expressions of HN and MOTS-c Are Downregulated in Human Atrial Fibrillation Tissue and Negatively Correlate with Fibrosis
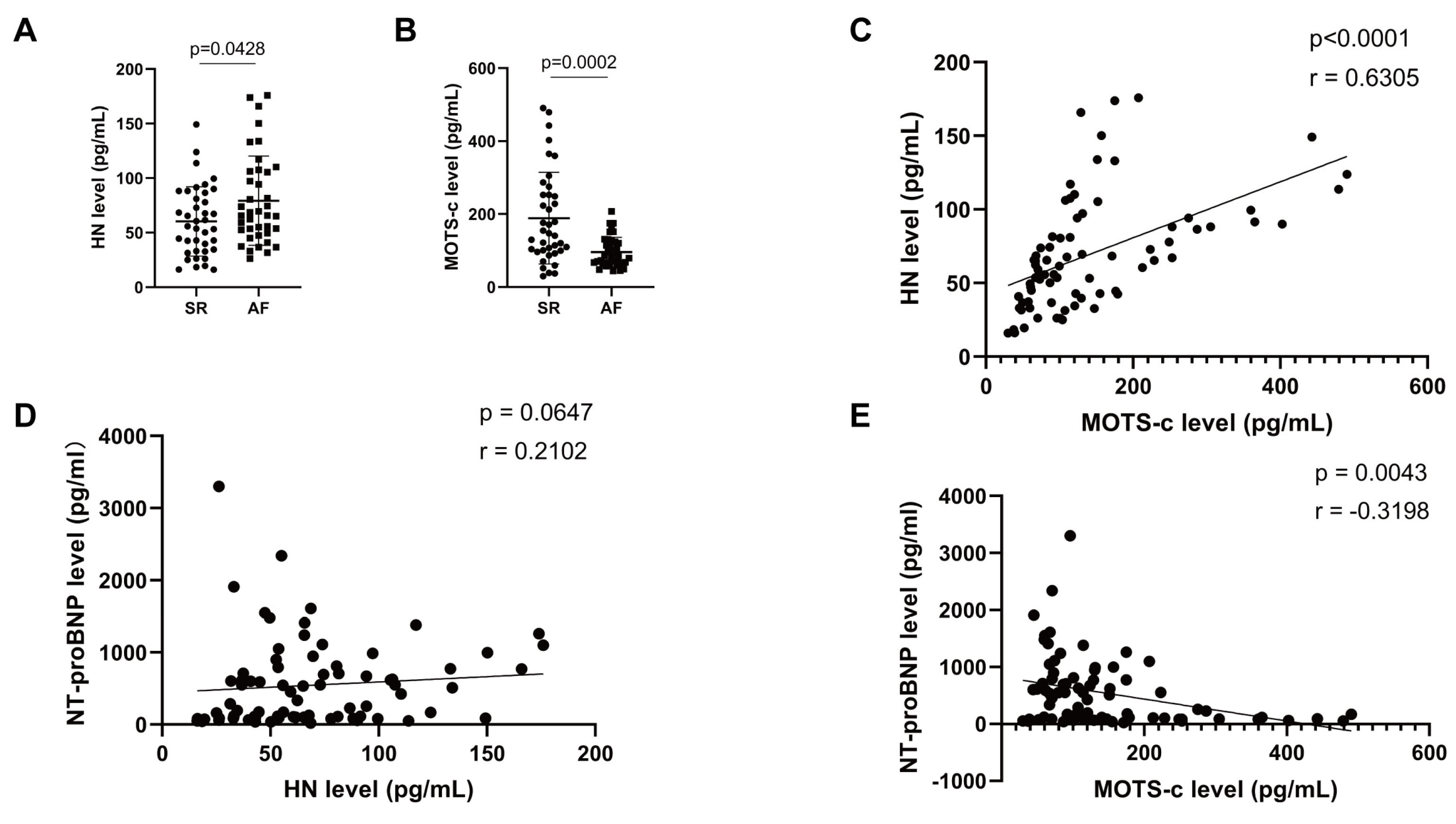
3.2. Plasma MOTS-c Level Decreased in AF Patients and Inversely Correlates with NT-proBNPlevel (N-Terminal pro-B-Type Natriuretic Peptide)
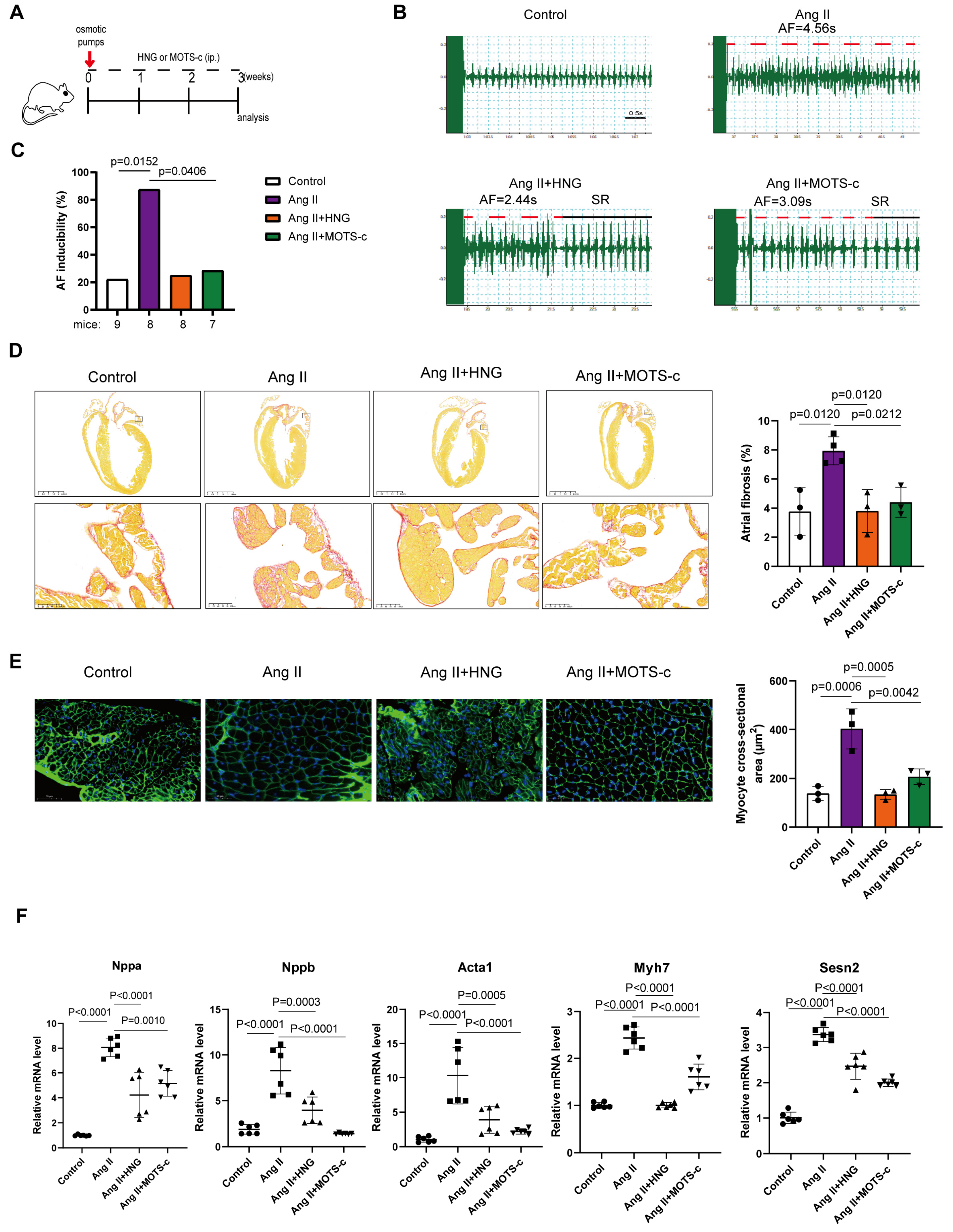
3.3. Administration of HNG or MOTS-c Attenuates AngII-Induced Atrial Fibrillation Susceptibility and Structural Remodeling in Mice
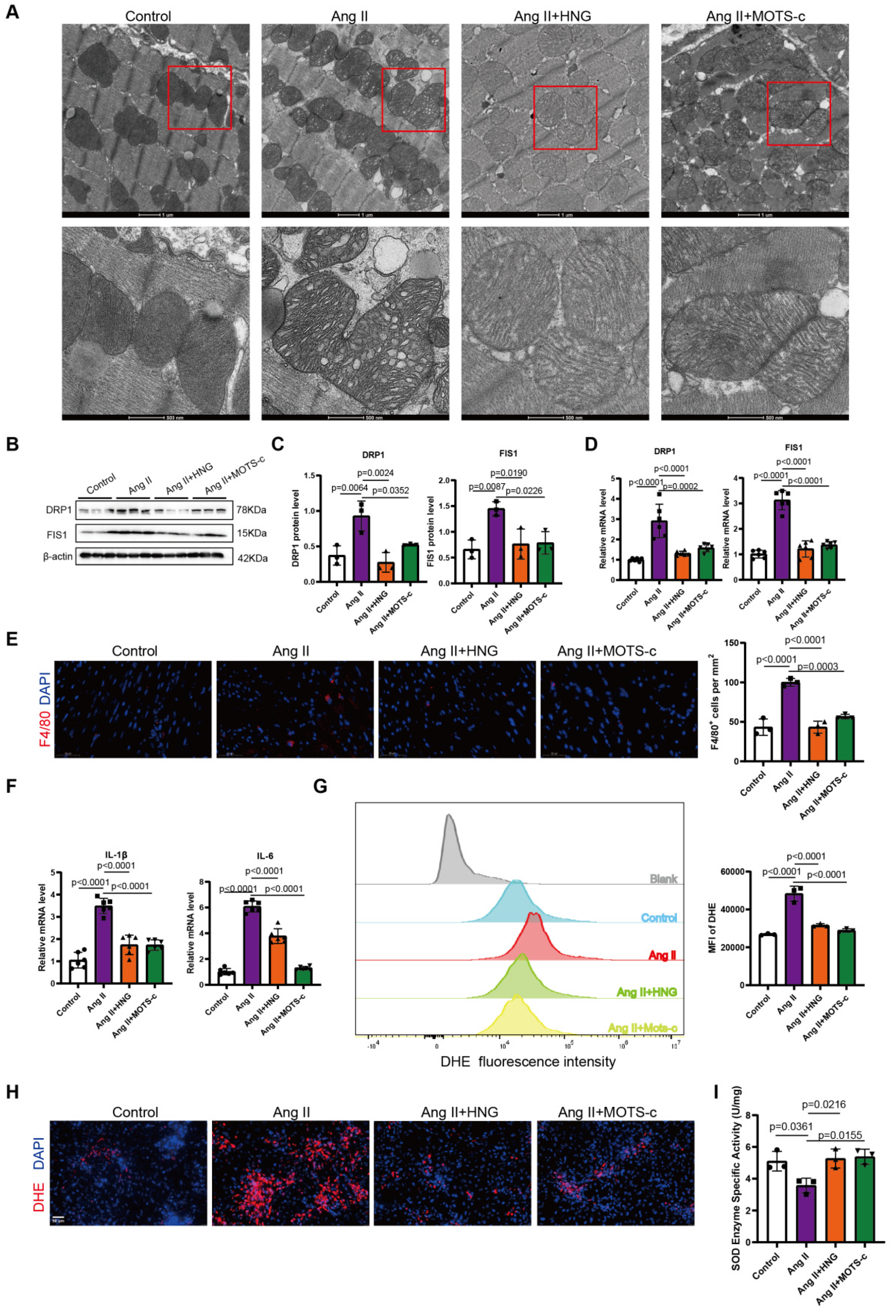
3.4. HNG and MOTS-c Preserve Mitochondrial Integrity and Alleviate Oxidative Stress and Inflammation
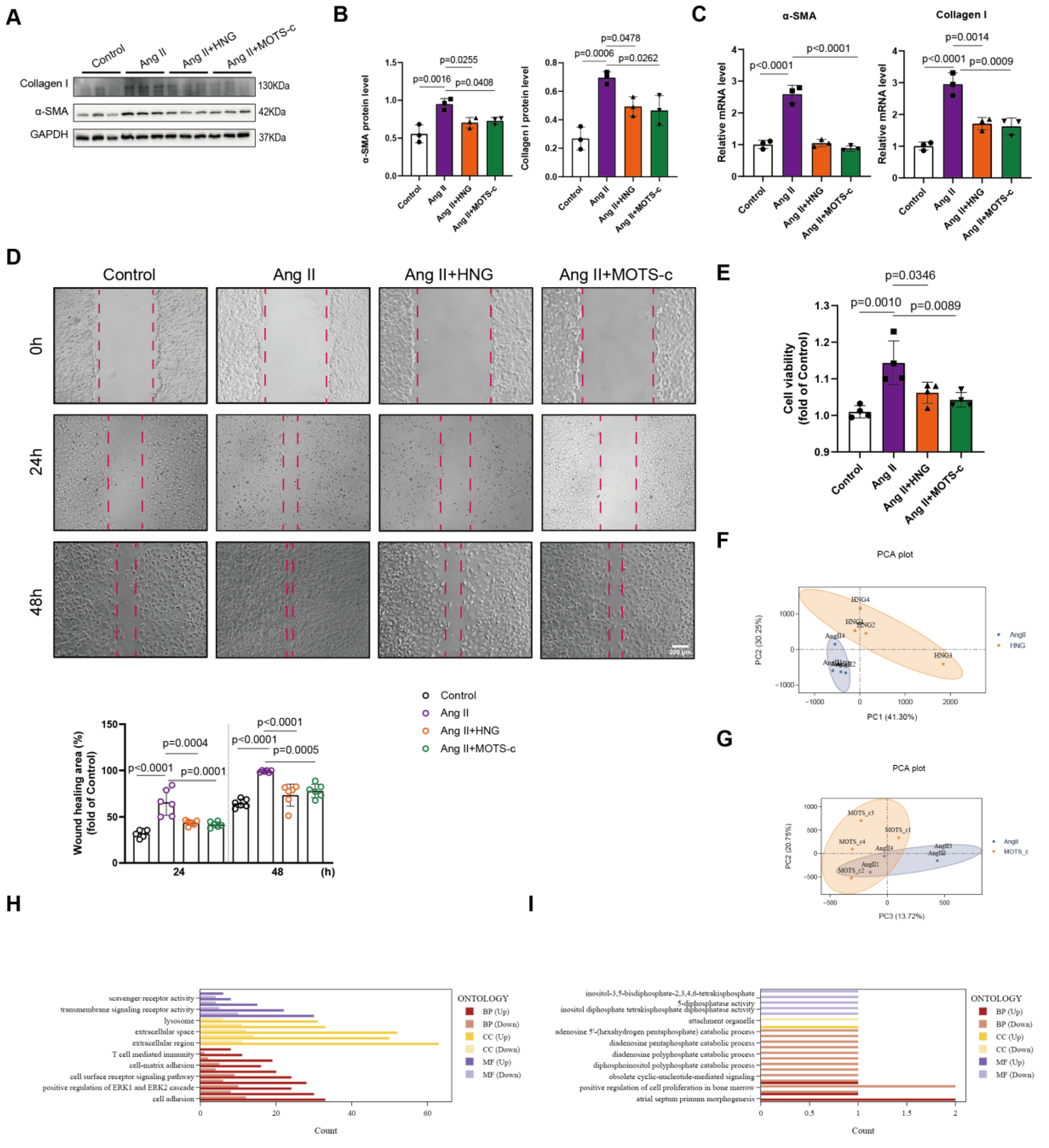
3.5. HNG and MOTS-c Inhibit AngII-Induced Activation, Proliferation, and Migration of Cardiac Fibroblasts
4. Discussion
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AF | Atrial fibrillation |
| α-SMA | Alpha-smooth muscle actin |
| AngII | Angiotensin II |
| ANOVA | Analysis of variance |
| CAD | Coronary artery disease |
| CCK-8 | Cell Counting Kit-8 |
| Col1a1 | Collagen type I alpha 1 chain |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DEGs | Differentially expressed genes |
| DHE | Dihydroethidium |
| DMEM | Dulbecco's Modified Eagle Medium |
| DNA | Deoxyribonucleic acid |
| Drp1 | Dynamin-related protein 1 |
| ECG | Electrocardiogram |
| ELISA | Enzyme-linked immunosorbent assay |
| EPL | Eplerenone |
| FBS | Fetal bovine serum |
| Fis1 | Mitochondrial fission protein 1 |
| GEO | Gene Expression Omnibus |
| GO | Gene Ontology |
| HNG | Gly14-Humanin (Humanin analogue) |
| HN | Humanin |
| IF | Immunofluorescence |
| IHC | Immunohistochemistry |
| IL-1β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| MDPs | Mitochondrial-derived peptides |
| MI | Myocardial infarction |
| MOTS-c | Mitochondrial open reading frame of the 12S rRNA-c |
| MRB | Mineralocorticoid receptor blocker |
| mRNA | Messenger RNA |
| MTRNR2L | MT-RNR2-like (nuclear humanin genes) |
| NRAMs | Neonatal rat atrial myocytes |
| NT-proBNP | N-terminal pro-brain natriuretic peptide |
| ox-LDL | Oxidized low-density lipoprotein |
| PAGE | Polyacrylamide gel electrophoresis |
| PBS | Phosphate-buffered saline |
| PCA | Principal component analysis |
| PCR | Polymerase chain reaction |
| PVDF | Polyvinylidene difluoride |
| qPCR | Quantitative polymerase chain reaction |
| RAAS | Renin-angiotensin-aldosterone system |
| RIPA | Radioimmunoprecipitation assay |
| RNA | Ribonucleic acid |
| ROS | Reactive oxygen species |
| SD | Standard deviation |
| SDS | Sodium dodecyl sulfate |
| SOD | Superoxide dismutase |
| SR | Sinus rhythm |
| TAC | Transverse aortic constriction |
| WGA | Wheat germ agglutinin |
References
- Marek-Iannucci, S.; Ozdemir, A.B.; Moreira, D.; Gomez, A.C.; Lane, M.; Porritt, R.A.; Lee, Y.; Shimada, K.; Abe, M.; Stotland, A.; et al. Autophagy-mitophagy induction attenuates cardiovascular inflammation in a murine model of Kawasaki disease vasculitis. J. Clin. Investig. 2021, 6. [CrossRef]
- Andrade, J., et al., The clinical profile and pathophysiology of atrial fibrillation: relationships among clinical features, epidemiology, and mechanisms.Circ Res, 2014. 114(9): p. 1453-68.
- Dzeshka, M.S., et al., Cardiac Fibrosis in Patients With Atrial Fibrillation: Mechanisms and Clinical Implications.J Am Coll Cardiol, 2015. 66(8): p. 943-59.
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomstrӧm-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Russ. J. Cardiol. 2021, 26, 4701. [CrossRef]
- de Groot, N.M., et al., Electropathological substrate of longstanding persistent atrial fibrillation in patients with structural heart disease: epicardial breakthrough.Circulation, 2010. 122(17): p. 1674-82.
- Krishnan, A.; Chilton, E.; Raman, J.; Saxena, P.; McFarlane, C.; Trollope, A.F.; Kinobe, R.; Chilton, L. Are Interactions between Epicardial Adipose Tissue, Cardiac Fibroblasts and Cardiac Myocytes Instrumental in Atrial Fibrosis and Atrial Fibrillation?. Cells 2021, 10, 2501. [CrossRef]
- Zhu, P.; Li, H.; Zhang, A.; Li, Z.; Zhang, Y.; Ren, M.; Zhang, Y.; Hou, Y. MicroRNAs sequencing of plasma exosomes derived from patients with atrial fibrillation: miR-124-3p promotes cardiac fibroblast activation and proliferation by regulating AXIN1. J. Physiol. Biochem. 2021, 78, 85–98. [CrossRef]
- Karakasis, P.; Theofilis, P.; Vlachakis, P.K.; Korantzopoulos, P.; Patoulias, D.; Antoniadis, A.P.; Fragakis, N. Atrial Fibrosis in Atrial Fibrillation: Mechanistic Insights, Diagnostic Challenges, and Emerging Therapeutic Targets. Int. J. Mol. Sci. 2024, 26, 209. [CrossRef]
- Anné, W.; Willems, R.; Holemans, P.; Beckers, F.; Roskams, T.; Lenaerts, I.; Ector, H.; Heidbüchel, H. Self-terminating AF depends on electrical remodeling while persistent AF depends on additional structural changes in a rapid atrially paced sheep model. J. Mol. Cell. Cardiol. 2007, 43, 148–158. [CrossRef]
- Ehrlich, J.R.; Hohnloser, S.H.; Nattel, S. Role of angiotensin system and effects of its inhibition in atrial fibrillation: clinical and experimental evidence. Eur. Hear. J. 2005, 27, 512–518. [CrossRef]
- Swedberg, K., et al., Eplerenone and atrial fibrillation in mild systolic heart failure: results from the EMPHASIS-HF (Eplerenone in Mild Patients Hospitalization And SurvIval Study in Heart Failure) study.J Am Coll Cardiol, 2012. 59(18): p. 1598-603.
- Takemoto, Y.; Ramirez, R.J.; Kaur, K.; Salvador-Montañés, O.; Ponce-Balbuena, D.; Ramos-Mondragón, R.; Ennis, S.R.; Guerrero-Serna, G.; Berenfeld, O.; Jalife, J. Eplerenone Reduces Atrial Fibrillation Burden Without Preventing Atrial Electrical Remodeling. JACC 2017, 70, 2893–2905. [CrossRef]
- Murphy, E., et al., Mitochondrial Function, Biology, and Role in Disease: A Scientific Statement From the American Heart Association.Circ Res, 2016. 118(12): p. 1960-91.
- Bukowska, A.; Schild, L.; Keilhoff, G.; Hirte, D.; Neumann, M.; Gardemann, A.; Neumann, K.H.; Röhl, F.-W.; Huth, C.; Goette, A.; et al. Mitochondrial Dysfunction and Redox Signaling in Atrial Tachyarrhythmia. Exp. Biol. Med. 2008, 233, 558–574. [CrossRef]
- Lin, P.-H.; Lee, S.-H.; Su, C.-P.; Wei, Y.-H. Oxidative damage to mitochondrial DNA in atrial muscle of patients with atrial fibrillation. Free. Radic. Biol. Med. 2003, 35, 1310–1318. [CrossRef]
- Júnior, A.d.S.M.; de França-E-Silva, A.L.G.; de Oliveira, J.M.; da Silva, D.M. Developing Pharmacological Therapies for Atrial Fibrillation Targeting Mitochondrial Dysfunction and Oxidative Stress: A Scoping Review. Int. J. Mol. Sci. 2023, 25, 535. [CrossRef]
- Xie, W., et al., Mitochondrial oxidative stress promotes atrial fibrillation.Sci Rep, 2015. 5: p. 11427.
- Hashimoto, Y.; Niikura, T.; Tajima, H.; Yasukawa, T.; Sudo, H.; Ito, Y.; Kita, Y.; Kawasumi, M.; Kouyama, K.; Doyu, M.; et al. A rescue factor abolishing neuronal cell death by a wide spectrum of familial Alzheimer's disease genes and Aβ. Proc. Natl. Acad. Sci. USA 2001, 98, 6336–6341. [CrossRef]
- Lee, C.; Zeng, J.; Drew, B.G.; Sallam, T.; Martin-Montalvo, A.; Wan, J.; Kim, S.-J.; Mehta, H.; Hevener, A.L.; de Cabo, R.; et al. The Mitochondrial-Derived Peptide MOTS-c Promotes Metabolic Homeostasis and Reduces Obesity and Insulin Resistance. Cell Metab. 2015, 21, 443–454. [CrossRef]
- Bodzioch, M.; Lapicka-Bodzioch, K.; Zapala, B.; Kamysz, W.; Kiec-Wilk, B.; Dembinska-Kiec, A. Evidence for potential functionality of nuclearly-encoded humanin isoforms. Genomics 2009, 94, 247–256. [CrossRef]
- Bachar, A.R.; Scheffer, L.; Schroeder, A.S.; Nakamura, H.K.; Cobb, L.J.; Oh, Y.K.; Lerman, L.O.; Pagano, R.E.; Cohen, P.; Lerman, A. Humanin is expressed in human vascular walls and has a cytoprotective effect against oxidized LDL-induced oxidative stress. Cardiovasc. Res. 2010, 88, 360–366. [CrossRef]
- Qin, Q.; Mehta, H.; Yen, K.; Navarrete, G.; Brandhorst, S.; Wan, J.; Delrio, S.; Zhang, X.; Lerman, L.O.; Cohen, P.; et al. Chronic treatment with the mitochondrial peptide humanin prevents age-related myocardial fibrosis in mice. Am. J. Physiol. Circ. Physiol. 2018, 315, H1127–H1136. [CrossRef]
- Peng, T.; Wan, W.; Wang, J.; Liu, Y.; Fu, Z.; Ma, X.; Li, J.; Sun, G.; Ji, Y.; Lu, J.; et al. The Neurovascular Protective Effect of S14G-Humanin in a Murine MCAO Model and Brain Endothelial Cells. IUBMB Life 2018, 70, 691–699. [CrossRef]
- Zhong, P.; Peng, J.; Hu, Y.; Zhang, J.; Shen, C. Mitochondrial derived peptide MOTS-c prevents the development of heart failure under pressure overload conditions in mice. J. Cell. Mol. Med. 2022, 26, 5369–5378. [CrossRef]
- Yuan, J.; Wang, M.; Pan, Y.; Liang, M.; Fu, Y.; Duan, Y.; Tang, M.; Laher, I.; Li, S. The mitochondrial signaling peptide MOTS-c improves myocardial performance during exercise training in rats. Sci. Rep. 2021, 11, 1–11. [CrossRef]
- Lu, P.; Li, X.; Li, B.; Li, X.; Wang, C.; Liu, Z.; Ji, Y.; Wang, X.; Wen, Z.; Fan, J.; et al. The mitochondrial-derived peptide MOTS-c suppresses ferroptosis and alleviates acute lung injury induced by myocardial ischemia reperfusion via PPARγ signaling pathway. Eur. J. Pharmacol. 2023, 953, 175835. [CrossRef]
- Kavak, A.G.; Karslioglu, I.; Saracaloglu, A.; Demiryürek, S.; Demiryürek, A.T. Impact of Radiation Therapy on Serum Humanin and MOTS-c Levels in Patients with Lung or Breast Cancer. Curr. Radiopharm. 2024, 17, 229–237. [CrossRef]
- Li, J.; Wang, S.; Zhang, Y.-L.; Bai, J.; Lin, Q.-Y.; Liu, R.-S.; Yu, X.-H.; Li, H.-H. Immunoproteasome Subunit β5i Promotes Ang II (Angiotensin II)–Induced Atrial Fibrillation by Targeting ATRAP (Ang II Type I Receptor–Associated Protein) Degradation in Mice. Hypertension 2019, 73, 92–101. [CrossRef]
- Li, D.-S.; Xue, G.-L.; Yang, J.-M.; Li, C.-Z.; Zhang, R.-X.; Tian, T.; Li, Z.; Shen, K.-W.; Guo, Y.; Liu, X.-N.; et al. Knockout of interleukin-17A diminishes ventricular arrhythmia susceptibility in diabetic mice via inhibiting NF-κB-mediated electrical remodeling. Acta Pharmacol. Sin. 2021, 43, 307–315. [CrossRef]
- Han, W.; Du, C.; Zhu, Y.; Ran, L.; Wang, Y.; Xiong, J.; Wu, Y.; Lan, Q.; Wang, Y.; Wang, L.; et al. Targeting Myocardial Mitochondria-STING-Polyamine Axis Prevents Cardiac Hypertrophy in Chronic Kidney Disease. JACC: Basic Transl. Sci. 2022, 7, 820–840. [CrossRef]
- Schrickel, J.W.; Bielik, H.; Yang, A.; Schimpf, R.; Shlevkov, N.; Burkhardt, D.; Meyer, R.; Grohé, C.; Fink, K.; Tiemann, K.; et al. Induction of atrial fibrillation in mice by rapid transesophageal atrial pacing. Basic Res. Cardiol. 2002, 97, 452–460. [CrossRef]
- Murphy, M.B.; Kim, K.; Kannankeril, P.J.; Subati, T.; Van Amburg, J.C.; Barnett, J.V.; Murray, K.T. Optimizing transesophageal atrial pacing in mice to detect atrial fibrillation. Am. J. Physiol. Circ. Physiol. 2022, 322, H36–H43. [CrossRef]
- Davis, S.; Meltzer, P.S. GEOquery: a bridge between the Gene Expression Omnibus (GEO) and BioConductor. Bioinformatics 2007, 23, 1846–1847. [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [CrossRef]
- Hao, D.; Yang, X.; Li, Z.; Xie, B.; Feng, Y.; Liu, G.; Ren, X. Screening core genes for minimal change disease based on bioinformatics and machine learning approaches. Int. Urol. Nephrol. 2024, 57, 655–671. [CrossRef]
- Chin, Y.-P.; Keni, J.; Wan, J.; Mehta, H.; Anene, F.; Jia, Y.; Lue, Y.-H.; Swerdloff, R.; Cobb, L.J.; Wang, C.; et al. Pharmacokinetics and Tissue Distribution of Humanin and Its Analogues in Male Rodents. Endocrinology 2013, 154, 3739–3744. [CrossRef]
- Wang, W.; Li, E.; Zou, J.; Qu, C.; Ayala, J.; Wen, Y.; Islam, S.; Weintraub, N.L.; Fulton, D.J.; Liang, Q.; et al. Ubiquitin Ligase RBX2/SAG Regulates Mitochondrial Ubiquitination and Mitophagy. Circ. Res. 2024, 135, e39–e56. [CrossRef]
- Zahid, M.A.; Abdelsalam, S.S.; Raïq, H.; Parray, A.; Korashy, H.M.; Zeidan, A.; Elrayess, M.A.; Agouni, A. Sestrin2 as a Protective Shield against Cardiovascular Disease. Int. J. Mol. Sci. 2023, 24, 4880. [CrossRef]
- Cai, H.; Cao, P.; Sun, W.; Shao, W.; Li, R.; Wang, L.; Zou, L.; Forno, E.; Muzumdar, R.; Gong, Z.; et al. Circulating humanin is lower in coronary artery disease and is a prognostic biomarker for major cardiac events in humans. Biochim. et Biophys. Acta (BBA) - Gen. Subj. 2022, 1866, 130010. [CrossRef]
- Conte, M.; Ostan, R.; Fabbri, C.; Santoro, A.; Guidarelli, G.; Vitale, G.; Mari, D.; Sevini, F.; Capri, M.; Sandri, M.; et al. Human Aging and Longevity Are Characterized by High Levels of Mitokines. Journals Gerontol. Ser. A 2018, 74, 600–607. [CrossRef]
- Liu, C.; Gidlund, E.-K.; Witasp, A.; Qureshi, A.R.; Söderberg, M.; Thorell, A.; Nader, G.A.; Barany, P.; Stenvinkel, P.; von Walden, F. Reduced skeletal muscle expression of mitochondrial-derived peptides humanin and MOTS-C and Nrf2 in chronic kidney disease. Am. J. Physiol. Physiol. 2019, 317, F1122–F1131. [CrossRef]
- Qin, Q.; Delrio, S.; Wan, J.; Widmer, R.J.; Cohen, P.; Lerman, L.O.; Lerman, A. Downregulation of circulating MOTS-c levels in patients with coronary endothelial dysfunction. Int. J. Cardiol. 2018, 254, 23–27. [CrossRef]
- Yasar, E.; Cakmak, T.; Bayramoglu, A.; Karaku, Y.; Tekin, S.; Sekerci, G.; Turkoglu, C. MOTS-c as a predictor of coronary lesions and complexity in patients with stable coronary artery disease. 2022, 26, 5676–5682. [CrossRef]
- Woodhead, J.S.T.; D’sOuza, R.F.; Hedges, C.P.; Wan, J.; Berridge, M.V.; Cameron-Smith, D.; Cohen, P.; Hickey, A.J.R.; Mitchell, C.J.; Merry, T.L. High-intensity interval exercise increases humanin, a mitochondrial encoded peptide, in the plasma and muscle of men. J. Appl. Physiol. 2020, 128, 1346–1354. [CrossRef]
- Reynolds, J.C.; Lai, R.W.; Woodhead, J.S.T.; Joly, J.H.; Mitchell, C.J.; Cameron-Smith, D.; Lu, R.; Cohen, P.; Graham, N.A.; Benayoun, B.A.; et al. MOTS-c is an exercise-induced mitochondrial-encoded regulator of age-dependent physical decline and muscle homeostasis. Nat. Commun. 2021, 12, 1–11. [CrossRef]
- Tilokani, L.; Nagashima, S.; Paupe, V.; Prudent, J. Mitochondrial dynamics: overview of molecular mechanisms. Essays Biochem. 2018, 62, 341–360. [CrossRef]
- Quiles, J.M. and B. Gustafsson Å, The role of mitochondrial fission in cardiovascular health and disease. Nat Rev Cardiol, 2022. 19(11): p. 723-736.
- Li, Y., et al., Ibrutinib Promotes Atrial Fibrillation by Disrupting A-Kinase Anchoring Protein 1-Mediated Mitochondrial Quality Surveillance in Cardiomyocytes. Research (Wash D C), 2024. 7: p. 0509.
- Harada, M.; Nattel, S. Implications of Inflammation and Fibrosis in Atrial Fibrillation Pathophysiology. Card. Electrophysiol. Clin. 2021, 13, 25–35. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



