Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
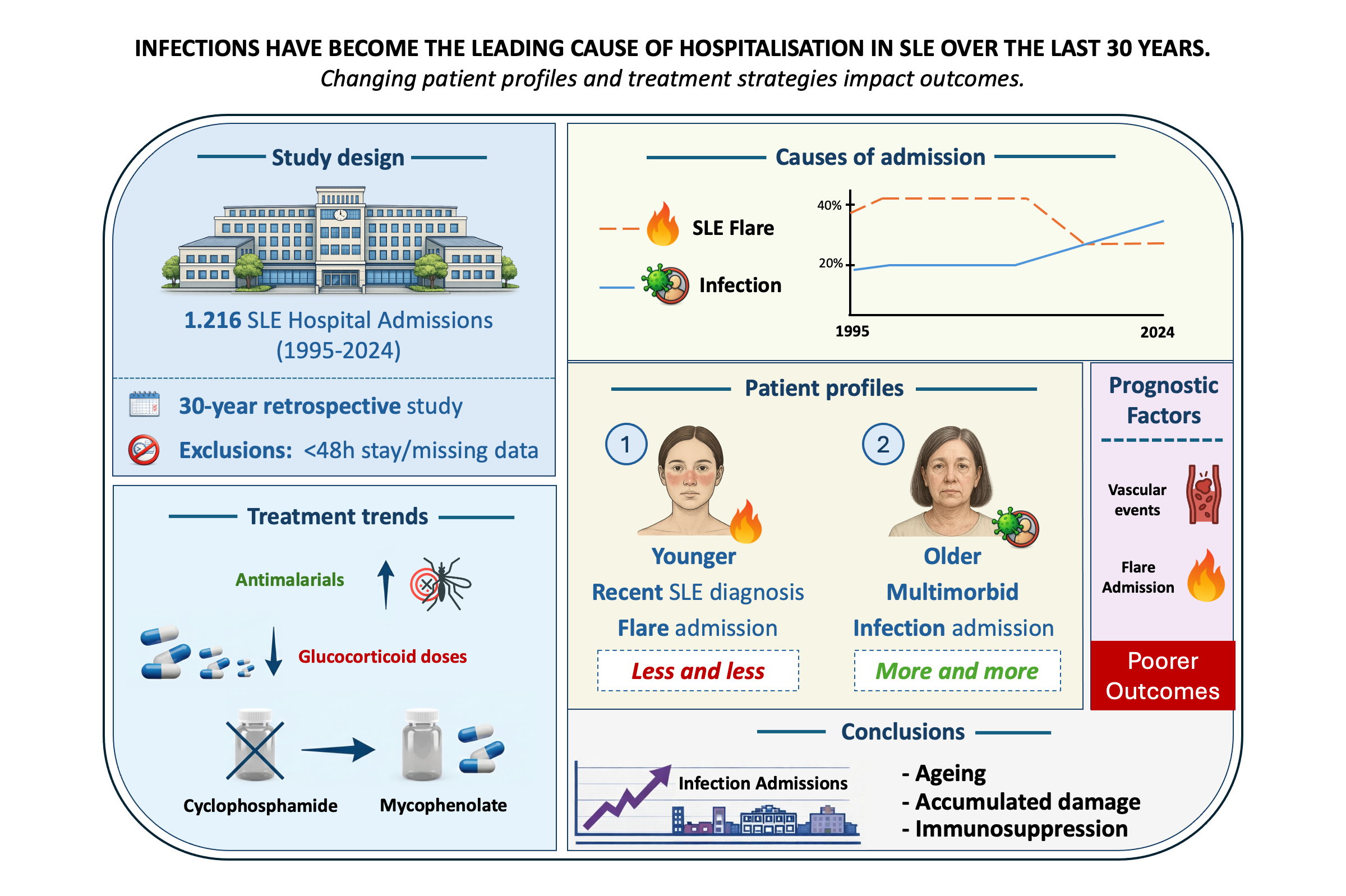
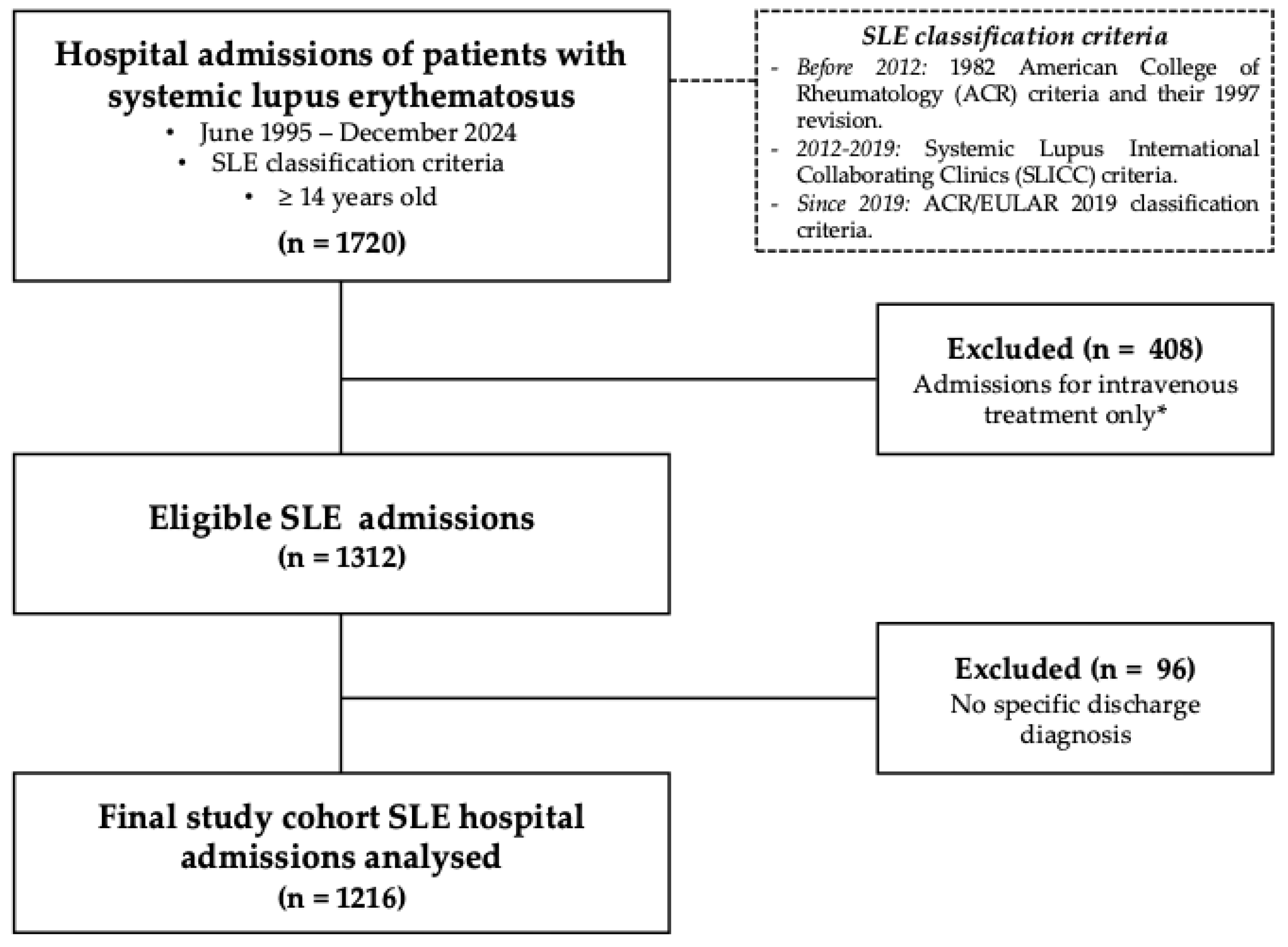
2.1. Study Design and Patient Selection
2.2. Data Collection and Variables
2.3. Statistical Analysis
3. Results
3.1. General Characteristics
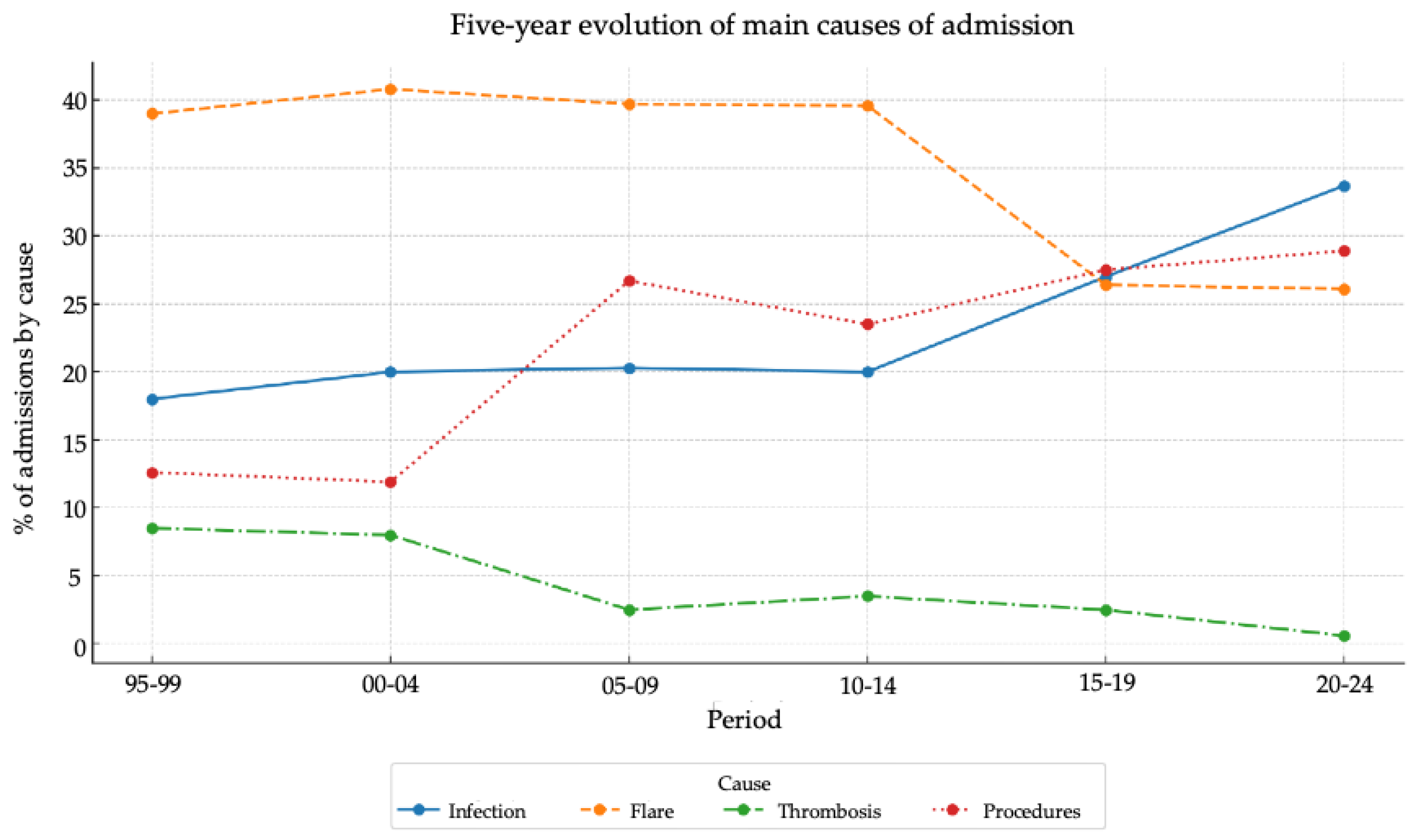
3.2. Causes of Hospital Admission
3.3. Subgroup Characteristics and Risk Factors
3.3.1. SLE Flare-Related Admissions
3.3.2. Infection-Related Admissions
3.4. Composite Outcome: ICU Admission, Readmission, and Prolonged Stay
3.4.1. ICU Admission
3.4.2. Readmission
3.4.3. Mortality
3.5. Risk Factors for the Composite Outcome
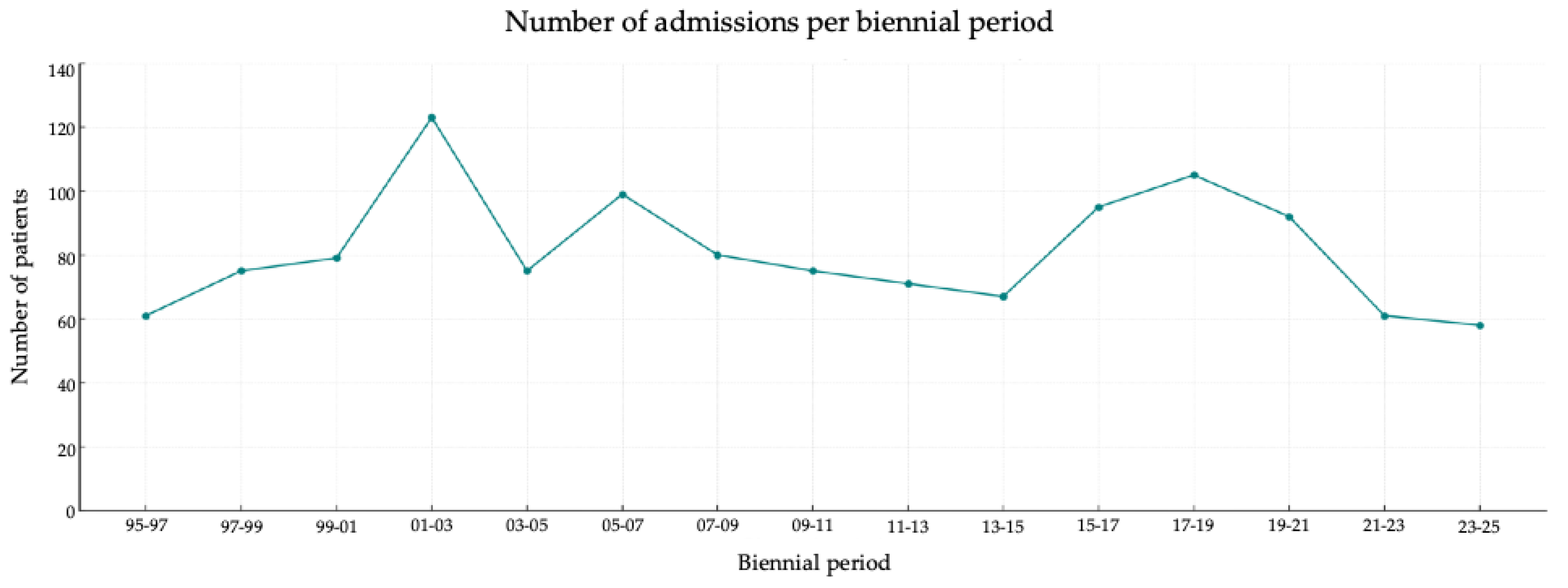
3.6. Temporal Trends Over 30 Years
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACR | American College of Rheumatology |
| ANOVA | Analysis of variance |
| APS | Antiphospholipid syndrome |
| CAPS | Catastrophic antiphospholipid syndrome |
| CI | Confidence interval |
| CRP | C reactive protein |
| DNA | Deoxyribonucleic acid |
| dsDNA | Double-stranded DNA |
| EULAR | European Alliance of Associations for Rheumatology |
| GE | Gerard Espinosa |
| ICU | Intensive care unit |
| IQR | Interquartile range |
| IV | Intravenous |
| JC | Jesús Cívico |
| NS | Non-statistical significance |
| OR | Odds ratio |
| SAD | Systemic autoimmune disease |
| SD | Standard deviation |
| SDI | Systemic Lupus International Collaborating Clinics Damage Index |
| SLE | Systemic lupus erythematosus |
| SLEDAI | SLE Disease Activity Index |
| SLICC | Systemic Lupus International Collaborating Clinics |
Appendix A
Appendix A.1
References
- Lin, D.H.; Murimi-Worstell, I.B.; Kan, H.; Tierce, J.C.; Wang, X.; Nab, H.; Desta, B.; Hammond, E.R.; Alexander, G.C. Health care utilization and costs of systemic lupus erythematosus in the United States: A systematic review. Lupus 2022, 31, 773–807. [Google Scholar] [CrossRef] [PubMed]
- Ng, B.; Zhu, R.; Yap, T.; Gordon, C.; Isenberg, D.; Marjanovic, S. The economic burden of systemic lupus erythematosus in New Zealand: An exploratory study. J. Med. Econ. 2021, 24, 505–513. [Google Scholar] [CrossRef]
- Hill, C.L.; Ngian, G.S.; Proudman, S.M.; Hall, S.; Gill, T.K. Hospitalisation for systemic lupus erythematosus associates with an increased risk of mortality in Australian patients from 1980 to 2014: A longitudinal, population-level, data linkage, cohort study. Lupus Sci. Med. 2023, 10, e000825. [Google Scholar] [CrossRef]
- Edwards, C.J.; Lian, T.Y.; Badsha, H.; Teh, C.L.; Arden, N.; Chng, H.H. Hospitalization of individuals with systemic lupus erythematosus: Characteristics and predictors of outcome. Lupus 2003, 12, 672–676. [Google Scholar] [CrossRef]
- Busch, R.W.; Svanström, H.; Ekstrand, C.; Jacobsen, S.; McDonald, J.C.; Voss, A.; et al. Hospitalization of patients with systemic lupus erythematosus in Denmark: Frequency, causes and outcomes. Lupus 2017, 26, 125–131. [Google Scholar] [CrossRef]
- Zeng, C.; Wang, H.; Liu, J.; Wang, L.; Sun, L.; Gao, J.; et al. Poor prognosis markers among hospitalized patients with systemic lupus erythematosus: A nationwide population-based study in China. Lupus 2022, 31, 935–942. [Google Scholar] [CrossRef]
- Al Arfaj, A.S.; Khalil, N. Clinical and demographic characteristics of hospitalized SLE patients in Saudi Arabia. Lupus 2009, 18, 465–470. [Google Scholar] [CrossRef]
- Pires da Rosa, G.; Fontecha Ortega, M.; Teixeira, A.; Espinosa, G.; Cervera, R. Causes and factors related to hospitalizations in patients with systemic lupus erythematosus: Analysis of a 20-year period (1995–2015) from a single referral centre in Catalonia. Lupus 2019, 28, 1158–1166. [Google Scholar] [CrossRef]
- Moreno-Torres, V.; Martínez-Urbistondo, M.; Gutiérrez-Rojas, A.; Castejón, R.; Sánchez, E.; Calderón-Parra, J.; et al. Impact of severe infections in SLE: An observational study from the Spanish national registry. Lupus Sci. Med. 2022, 9, e000711. [Google Scholar] [CrossRef]
- Pires da Rosa, G.; Cervera, R.; Espinosa, G. Causes of hospitalization in systemic lupus erythematosus: A narrative review. Curr. Rheumatol. Rev. 2021, 17, 29–40. [Google Scholar] [CrossRef]
- Tan, E.M.; Cohen, A.S.; Fries, J.F.; Masi, A.T.; McShane, D.J.; Rothfield, N.F.; et al. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1982, 25, 1271–1277. [Google Scholar] [CrossRef]
- Hochberg, M.C. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997, 40, 1725. [Google Scholar] [CrossRef] [PubMed]
- Aringer, M.; Costenbader, K.; Daikh, D.; Brinks, R.; Mosca, M.; Ramsey-Goldman, R.; et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2019, 71, 1400–1412. [Google Scholar] [CrossRef] [PubMed]
- Petri, M.; Orbai, A.M.; Alarcón, G.S.; Gordon, C.; Merrill, J.T.; Fortin, P.R.; et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2012, 64, 2677–2686. [Google Scholar] [CrossRef] [PubMed]
- Gladman, D.D.; Ibañez, D.; Urowitz, M.B. Systemic lupus erythematosus disease activity index 2000. J. Rheumatol. 2002, 29, 288–291. [Google Scholar]
- Collins, T.C.; Daley, J.; Henderson, W.H.; Khuri, S.F. Risk factors for prolonged length of stay after major elective surgery. Ann. Surg. 1999, 230, 251–259. [Google Scholar] [CrossRef]
- Wang, Y.; Eldridge, N.; Metersky, M.L.; et al. Factors associated with a prolonged length of stay after acute exacerbation of chronic obstructive pulmonary disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2014, 9, 99–107. [Google Scholar] [CrossRef]
- Martins, R.S.; Dawood, Z.S.; Memon, M.K.Y.; Akhtar, S. Prolonged length of stay after surgery for adult congenital heart disease: A single-centre study in a developing country. Cardiol. Young 2020, 30, 1253–1260. [Google Scholar] [CrossRef]
- Canora-Lebrato, J.; Barba-Martín, R.; Perales-Fraile, I.; Marco-Martínez, J.; Plaza-Cantelli, S.; Zapatero-Gaviria, A. Descripción de las altas hospitalarias en pacientes con lupus eritematoso sistémico. Rev. Clin. Esp. 2012, 212, 432–439. [Google Scholar] [CrossRef]
- Moreno-Torres, V.; Tarín, C.; Ruiz-Irastorza, G.; Castejón, R.; Gutiérrez-Rojas, Á.; Royuela, A.; et al. Trends in hospital admissions and death causes in patients with systemic lupus erythematosus: Spanish national registry. J. Clin. Med. 2021, 10, 5749. [Google Scholar] [CrossRef]
- Aldarmaki, R.; Al Khogali, H.I.; Al Dhanhani, A.M. Hospitalization in patients with systemic lupus erythematosus at Tawam Hospital, United Arab Emirates (UAE): Rates, causes, and factors associated with length of stay. Lupus 2021, 30, 845–851. [Google Scholar] [CrossRef]
- Araujo, O.; Espinosa, G.; Hernández-Rodríguez, J.; Hernández-Negrín, H.; Adán, A.; Pelegrín, L.; et al. New insights into the long-term adverse effects of antimalarials in patients with systemic lupus erythematosus. Lupus 2025, 34, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Morand, E.F.; Fernandez-Ruiz, R.; Blazer, A.; Niewold, T.B. Advances in the management of systemic lupus erythematosus. BMJ 2023, 383, e073980. [Google Scholar] [CrossRef] [PubMed]
- Fanouriakis, A.; Kostopoulou, M.; Andersen, J.; Aringer, M.; Arnaud, L.; Bae, S.C.; et al. EULAR recommendations for the management of systemic lupus erythematosus: 2023 update. Ann. Rheum. Dis. 2024, 83, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Van Vollenhoven, R.F.; Mosca, M.; Bertsias, G.; Isenberg, D.; Kuhn, A.; Lerstrøm, K.; et al. Treat-to-target in systemic lupus erythematosus: Recommendations from an international task force. Ann. Rheum. Dis. 2014, 73, 958–967. [Google Scholar] [CrossRef]
- Piga, M.; Parodis, I.; Touma, Z.; Legge, A.; Ugarte-Gil, M.F.; Hmamouchi, I.; et al. Framework for implementing treat-to-target in systemic lupus erythematosus routine clinical care: Consensus statements from an international task force. Autoimmun. Rev. 2025, 24, 103773. [Google Scholar] [CrossRef]
- Jiang, N.; Jin, S.; Yu, C.; Zhao, J.; Wang, Q.; Tian, X.; et al. Efficacy and safety of immunosuppressive agents for adults with lupus nephritis: A systematic review and network meta-analysis. Front. Immunol. 2023, 14, 1232244. [Google Scholar] [CrossRef]
- Mok, C.C. Current role of rituximab in systemic lupus erythematosus. Int. J. Rheum. Dis. 2015, 18, 154–163. [Google Scholar] [CrossRef]
- Drosos, G.C.; Vedder, D.; Houben, E.; Boekel, L.; Atzeni, F.; Badreh, S.; et al. EULAR recommendations for cardiovascular risk management in rheumatic and musculoskeletal diseases, including systemic lupus erythematosus and antiphospholipid syndrome. Ann. Rheum. Dis. 2022, 81, 768–779. [Google Scholar] [CrossRef]
- Antón, J.M.; Castro, P.; Espinosa, G.; Marcos, M.; Gandía, M.; Merchán, R.; et al. Mortality and long-term survival prognostic factors of patients with systemic autoimmune diseases admitted to an intensive care unit: A retrospective study. Clin. Exp. Rheumatol. 2012, 30, 338–344. [Google Scholar]
- Brito-Zerón, P.; Baldini, C.; Bootsma, H.; Bowman, S.J.; Jonsson, R.; Mariette, X.; et al. Sjögren syndrome. Nat. Rev. Dis. Primers 2016, 2, 16047. [Google Scholar] [CrossRef]
| Variable | Value |
| Study population | |
| Female sex, n (%) | 437 (87.4) |
| Age at SLE diagnosis, years mean (SD) | 31.2 (14.8) |
| Age at hospital admission*, years mean (SD) | 42.4 (15.6) |
| Disease duration at admission*, years median [IQR] | 8 [3,4,5,6,7,8,9,10,11,12,13,14,15,16,17] |
| Newly diagnosed SLE, n (%) | 56 (4.6) |
| Cardiovascular risk factors, (n = 500), n (%) | |
| Arterial hypertension | 317 (26.1) |
| Smoking history | 264 (21.7) |
| Dyslipidaemia | 166 (13.7) |
| Diabetes mellitus | 50 (4.1) |
| Associated autoimmune diseases, (n = 500) n (%) | |
| Any overlap syndrome | 167 (33.4) |
| Antiphospholipid syndrome | 88 (17.6) |
| Sjögren’s syndrome | 67 (13.4) |
| Disease status at admission | |
| SDI (n = 500) median [IQR] | 0 [0-1] |
| SLEDAI-2K at admission (n = 426) median [IQR] | 8 [5,6,7,8,9,10,11,12,13] |
| Biomarkers at admission** (n= 331), n (%) | |
| Elevated anti-dsDNA | 284 (85.8) |
| Complement consumption | 271 (77.2) |
| Proteinuria > 0,5 mg/24 h | 221 (56.5) |
| Source of admission* (n = 1216), n (%) | |
| Emergency department | 517 (42.5) |
| Elective admission from home | 334 (27.5) |
| Day hospital | 152 (12.5) |
| Outpatient clinic | 145 (11.9) |
| Other hospital department | 51 (4.2) |
| Intensive care unit | 17 (1.4) |
| Treatment at admission* (n = 1216), n (%) | |
| Antimalarials | 697 (57.3) |
| Immunosuppressants | 516 (42.4) |
| Biologic agents | 50 (4.2) |
| Glucocorticoids | 950 (78.1) |
| Anticoagulants | 178 (14.6) |
| Antiplatelet agents | 248 (20.4) |
| Outcomes, admissions, (n = 1216) | |
| Length of hospital stay, days median [IQR] | 6 [2,3,4,5,6,7,8,9] |
| 30-day readmission, n (%) | 90 (7.4%) |
| ICU admission, n (%) | 66 (5.4%) |
| In-hospital mortality, n (%) | 12 (1.0%) |
| Cause of admission | n (%) |
| SLE flare | 426 (35.0) |
| Renal involvement | 175 (41.1)† |
| Constitutional symptoms | 167 (39.2)† |
| Haematological | 149 (35.0)† |
| Arthritis | 143 (33.6)† |
| Mucocutaneous involvement | 113 (26.5)† |
| Serositis | 79 (18.5)† |
| Neuropsychiatric involvement | 19 (4.5)† |
| Infection | 284 (23.4) |
| Respiratory | 129 (45.4)† |
| Urinary | 59 (20.7)† |
| Gastrointestinal | 44 (15.4)† |
| Skin and soft tissue | 20 (7.0)† |
| Other | 32 (11.2)† |
| Diagnostic procedures | 267 (22.0) |
| Kidney biopsy | 232 (86.8)† |
| Thrombotic events | 50 (4.6) |
| Arterial | 28 (56.0)† |
| Stroke | 12 (42.8)‡ |
| Acute coronary syndrome | 6 (21.4)‡ |
| Digital ischemia | 6 (21.4)‡ |
| Venous | 22 (44.0)† |
| Deep venous thrombosis | 14 (63.6)‡ |
| Pulmonary embolism | 5 (22.7)‡ |
| Musculoskeletal disorders | 35 (2.9) |
| Vertebral fracture | 10 (28.5)† |
| Avascular femoral head necrosis | 5 (14.2)† |
| Glucocorticoid-induced myopathy | 3 (8.5)† |
| Other causes | 154 (12.6) |
| Other systemic autoimmune disease | 34 (2.8)† |
| Neurological disorders | 24 (2.0)† |
| Cardiovascular disease (non-thrombotic) | 24 (2.0)† |
| Others | 72 (5.9)† |
| Variable | SLE flare | Non-SLE flare | p | Infection | Non-Infection | p |
| Number of admissions (patients), n | 426 | 790 | 284 | 932 | ||
| Gender, female, n (%) | 379 (89) | 713 (90.3) | 0.480 | 262 (92.3) | 830 (89.1) | 0.119 |
| Age at SLE diagnosis, years, mean (SD) | 29.3 (13.3) | 32.2 (15.4) | <0.001 | 33.1 (16.4) | 30.6 (14.1) | 0.023 |
| Age at hospitalisation, years, mean (SD) | 37.4 (12.9) | 45.9 (16.1) | <0.001 | 48 (17.2) | 40.7 (14.5) | <0.001 |
| Duration of SLE, years, median [IQR] | 6 [1,2,3,4,5,6,7,8,9,10,11,12,13] | 10 [4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20] | <0.001 | 13 [6-22.2] | 7 [3,4,5,6,7,8,9,10,11,12,13,14,15] | <0.001 |
| Cardiovascular risk factors, n (%) | ||||||
| Arterial hypertension | 83 (19.5) | 234 (29.6) | <0.001 | 98 (34.5) | 219 (23.5) | <0.001 |
| Smoking history | 87 (20.4) | 177 (22.4) | 0.424 | 48 (16.9) | 216 (23.3) | 0.025 |
| Dyslipidaemia | 51 (12) | 115 (14.6) | 0.345 | 40 (14.1) | 126 (13.5) | 0.835 |
| Diabetes mellitus | 13 (3.1) | 37 (4.7) | 0.172 | 17 (6) | 33 (3.5) | 0.069 |
| Associated autoimmune diseases, n (%) | ||||||
| Overlap | 168 (39.4) | 342 (43.3) | 0.194 | 148 (52.1) | 362 (38.8) | <0.001 |
| Antiphospholipid syndrome | 118 (27.7) | 180 (22.8) | 0.057 | 70 (24.6) | 228 (24.5) | 0.950 |
| Sjogren’s syndrome | 67 (15.7) | 166 (21) | 0.025 | 84 (29.6) | 149 (16) | <0.001 |
| SDI at admission, median [IQR] | 0 [0-1] | 0 [0-1] | <0.001a | 1 [0-2] | 0 [0-1] | <0.001 |
| Treatment, n (%) | ||||||
| Antimalarials | 246 (57.7) | 451 (57.1) | 0.825 | 157 (55.3) | 540 (57.9) | 0.428 |
| Immunosuppressants | 168 (39.4) | 348 (44.1) | 0.120 | 119 (41.9) | 397 (42.6) | 0.836 |
| Biologic agents b | 12 (2.8) | 36 (4.6) | 0.126 | 22 (8) | 26 (2.8) | <0.001 |
| Glucocorticoids | 327 (76.8) | 623 (78.9) | 0.398 | 231 (81.3) | 719 (77.1) | 0.135 |
| > 0 ≤ 5mg/24h | 82 (20.1) | 234 (31.1) | <0.001 | 88 (31.8) | 228 (25.9) | <0.001 |
| > 5mg/24h | 226 (55.5) | 351 (46.7) | <0.001 | 136 (49.1) | 441 (50) | <0.001 |
| Anticoagulants | 45 (10.6) | 133 (16.8) | 0.003 | 53 (18.7) | 125 (13.4) | 0.028 |
| Antiplatelets | 76 (17.8) | 172 (21.8) | 0.201 | 68 (23.9) | 180 (19.3) | 0.207 |
| Outcomes c | ||||||
| Days of admission median [IQR] | 7 [4,5,6,7,8,9,10,11] | 5 [2,3,4,5,6,7,8,9] | <0.001 | 6 [4,5,6,7,8,9] | 6 [2,3,4,5,6,7,8,9] | 0.03 |
| Readmission at 30 days n (%) | 29 (7) | 41 (5.3) | 0.271 | 20 (7.3) | 50 (5.5) | 0.271 |
| Need for admission in ICU n (%) | 24 (5.6) | 39 (5) | 0.685 | 23 (8.3) | 40 (4.3) | 0.009 |
| Mortality d n (%) | 3 (0.7) | 8 (1) | 0.756 | 5 (1.8) | 6 (0.6) | 0.140 |
| Univariate analysis | Multivariate analysisa | |||
| Variable | OR (CI 95%) | p | OR (CI 95%) | p |
| SLE Flare as cause of admission | ||||
| Gender (female) | 1.173 (0.793-1.735) | 0.425 | 1.111 (0.733-1.684) | 0.619 |
| Age at SLE diagnosis | 0.985 (0.977-0.993) | <0.001 | NS | NS |
| Age at admission | 0.965 (0.956-0.973) | <0.001 | 0.980 (0.970-0.989) | <0.001 |
| SDI | 0.640 (0.562-.728) | <0.001 | 0.815 (0.708-0.939) | 0.005 |
| Duration of SLE, years | 0.940 (0.926-0.954) | <0.001 | 0.965 (0.948-0.981) | <0.001 |
| Sjogren’s syndrome | 0.677 (0.495-0.925) | 0.014 | 1.030 (0.725-1.464) | 0.868 |
| Present admission is a 30-day readmission | 1.638 (1.008-2.661) | 0.046 | 1.491 (0.879-2.530) | 0.138 |
| Arterial hypertension | 0.570 (0.428-0.759) | <0.001 | 0.845 (0.610-1.170) | 0.310 |
| Anticoagulants | 0.566 (0.394-0.813) | 0.002 | 0.830 (0.556-1.238) | 0.360 |
| Glucocorticoids > 0 ≤ 5mg/24h | 0.591 (0.414-0.844) | 0.004 | 0.727 (0.497-1.063) | 0.100 |
| Infection as cause of admission | ||||
| Gender (female) | 0.652 (0.395-1.076) | 0.094 | 0.749 (0.445-1.261) | 0.276 |
| Age at SLE diagnosis | 1.012 (1.003-1.021) | 0.008 | NS | NS |
| Age at admission | 1.030 (1.021-1.039) | <0.001 | 1.015 (1.005-1.025) | 0.004 |
| SDI | 1.588 (1.428-1.767) | <0.001 | 1.401 (1.248-1.573) | <0.001 |
| Duration of SLE, years | 1.051 (1.037-1.066) | <0.001 | NS | NS |
| Overlap | 1.718 (1.310-2.253) | <0.001 | NS | NS |
| Sjogren’s syndrome | 2.259 (1.655-3.084) | <0.001 | 1.513 (1.075-2.131) | 0.018 |
| Arterial hypertension | 1.799 (1.344-2.408) | <0.001 | 1.442 (1.046-1.990) | 0.026 |
| Smoking history | 0.672 (0.474-0.953) | 0.026 | NS | NS |
| Obesity | 2.051 (1.041-4.043) | 0.038 | NS | NS |
| Azathioprine | 0.553 (0.347-0.881) | 0.013 | 0.690 (0.423-1.123) | 0.135 |
| Biologic agents | 2.932 (1.634-5.260) | <0.001 | 1.825 (1.269-2.624) | 0.001 |
| Anticoagulants | 1.513 (1.061-2.157) | 0.022 | NS | NS |
| Glucocorticoids > 0 ≤ 5mg/24h | 1.541 (1.038-2.287) | 0.032 | NS | NS |
| Sample | Univariate analysis | Multivariate analysisa | ||
| OR (CI 95%) | p | OR (CI 95%) | p | |
| Composite outcome in the whole sample | ||||
| Gender (female) | 1.242 (0.829-1.861) | 0.292 | 1.231 (0.811-1.867) | 0.335 |
| Age at SLE diagnosis | 1.013 (1.005-1.022) | 0.002 | NS | NS |
| Age at admission | 1.010 (1.002-1.018) | 0.011 | 1.000 (1.000-1.016) | 0.324 |
| SDI | 1.159 (1.049-1.281) | 0.004 | NS | NS |
| Overlap | 2.034 (1.581-2.618) | <0.001 | 1.852 (1.422-2.405) | <0.001 |
| Antiphospholipid syndrome | 1.842 (1.396-2.431) | <0.001 | NS | NS |
| Diabetes mellitus | 1.966 (1.104-3.502) | 0.022 | NS | NS |
| Arterial hypertension | 1.635 (1.243-2.151) | <0.001 | 1.553 (1.164-2.099) | 0.003 |
| Dyslipidaemia | 1.436 (1.016-2.030) | 0.040 | NS | NS |
| SLE Flare as cause of admission | 1.417 (1.096-1.833) | 0.008 | 1.768 (1.334-2.331) | <0.001 |
| Vascular events b | 3.050 (1.804-5.155) | <0.001 | 3.112 (1.802-5.382) | <0.001 |
| Antimalarials | 0.699 (0.545-0.898) | 0.005 | 0.784 (0.605-1.011) | 0.057 |
| Anticoagulants | 1.649 (1.182-2.300) | 0.003 | NS | NS |
| Composite outcome in SLE flare admissions | ||||
| Gender (female) | 0.746 (0.457-1.753) | 0.671 | 0.683 (0.322-1.453) | 0.321 |
| SLEDAI-2K at admission | 1.109 (1.068-1.151) | <0.001 | 1.056 (1.005-1.114) | 0.036 |
| Newly diagnosed SLE | 1.891 (1.052-3.399) | 0.033 | 0.926 (0.454-1.877) | 0.813 |
| Antiphospholipid syndrome | 1.594 (1.024-2.482) | 0.039 | 1.837 (1.126-2.998) | 0.015 |
| Constitutional symptoms | 1.527 (1.013-2.303) | 0.043 | 1.653 (1.024-2.673) | 0.043 |
| Haemolytic anaemia | 2.203 (1.280-3.790) | 0.004 | 2.147 (1.164-3.931) | 0.014 |
| Renal flare | 2.600 (1.715-3.943) | <0.001 | 2.448 (1.413-4.230) | 0.001 |
| Neuropsychiatric flare | 4.366 (1.623-11.748) | 0.004 | 5.700 (1.799-18.554) | 0.003 |
| Proteinuria | 1.730 (1.120-2.614) | 0.014 | NS | NS |
| Antimalarials | 0.654 (0.435-0.984) | 0.042 | 0.698 (0.430-1.101) | 0.119 |
| Composite outcome in infection admissions | ||||
| Gender (female) | 0.712 (0.250-2.026) | 0.524 | 0.620 (0.265-2.208) | 0.765 |
| Age at admission | 1.003 (0.988-1.017) | 0.732 | 0.995 (0.979-1.011) | 0.525 |
| Arterial hypertension | 2.148 (1.269-3.634) | 0.004 | 2.263 (1.293-3.962) | 0.004 |
| Variable | 1995-2004 | 2005-2014 | 2015-2024 | p |
| Number of admissions (number of patients) | 403 (196) | 401 (139) | 412 (165) | 0.892 |
| Gender, female, n (%) | 173 (88.3) | 121 (87.1) | 143 (86.7) | 0.89 |
| Age at SLE diagnosis, years, mean (SD) | 32.3 (14.5) | 29.4 (13.5) | 31.9 (15.9) | 0.014 |
| Age at hospitalisation, years, mean (SD) | 41.3 (15.3) | 40.8 (14.3) | 44.9 (16.6) | <0.001 |
| Duration of SLE, years, median [IQR] | 7 [2,3,4,5,6,7,8,9,10,11,12,13,14] | 8 [6,7,8,9,10,11,12] | 10 [4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21] | <0.001 |
| Cardiovascular risk factors, n (%) | ||||
| Arterial hypertension | 126 (31.3) | 98 (24.4) | 93 (22.6) | 0.012 |
| Smoking history | 65 (16.1) | 103 (25.7) | 96 (23.3) | 0.003 |
| Dyslipidaemia | 64 (15.9) | 57 (14.2) | 45 (10.9) | 0.178 |
| Diabetes mellitus | 18 (4.5) | 10 (2.5) | 22 (5.3) | 0.113 |
| Associated autoimmune diseases, n (%) | ||||
| Overlap | 72 (36.7) | 36 (25.9) | 59 (35.8) | 0.860 |
| Antiphospholipid syndrome | 47 (24) | 20 (14.4) | 21 (12.7) | 0.01 |
| SDI at admission, median [IQR] | 0 [0-1] | 0 [0-1] | 0 [0-2] | 0.008 |
| SLEDAI-2K at admissiona, median [IQR] | 8 [5,6,7,8,9,10,11,12] | 8 [6,7,8,9,10,11,12] | 10 [6-17.5] | 0.005 |
| Treatment, n (%) | ||||
| Antimalarials | 182 (45.2) | 214 (53.4) | 287 (69.7) | <0.001 |
| Immunosuppressants | 112 (27.8) | 190 (47.4) | 214 (51.9) | <0.001 |
| Mycophenolate | 17 (4.2) | 96 (23.9) | 113 (27.4) | <0.001 |
| Methotrexate | 12 (3) | 22 (5.5) | 30 (7.3) | 0.022 |
| Cyclophosphamide | 35 (8.7) | 20 (5) | 9 (2.2) | <0.001 |
| Biologic agentsb | 0 | 7 (1.7) | 41 (10.2) | <0.001 |
| Glucocorticoids | 318 (78.9) | 331 (82.5) | 301 (73.1) | 0.004 |
| > 0 and ≤ 5mg/24h | 50 (14.5) | 108 (26.9) | 158 (38.3) | <0.001 |
| > 5mg/24h | 211 (61) | 223 (55.6) | 143 (34.7) | <0.001 |
| Anticoagulants | 50 (12.4) | 55 (13.7) | 73 (17.7) | 0.082 |
| Antiplatelets | 75 (18.6) | 73 (18.2) | 100 (24.3) | 0.098 |
| Outcomes c | ||||
| Days of admission, median [IQR] | 6 [4,5,6,7,8,9,10] | 5 [2,3,4,5,6,7,8,9] | 5 [2,3,4,5,6,7,8,9] | <0.001 |
| Readmission at 30 days, n (%) | 25 (6.3) | 26 (6.7) | 19 (4.8) | 0.498 |
| Need for admission in ICU, n (%) | 14 (3.5) | 18 (4.5) | 31 (7.7) | 0.020 |
| Mortality, n (%) | 4 (1) | 4 (1) | 3 (0.7) | 0.909 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




