Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
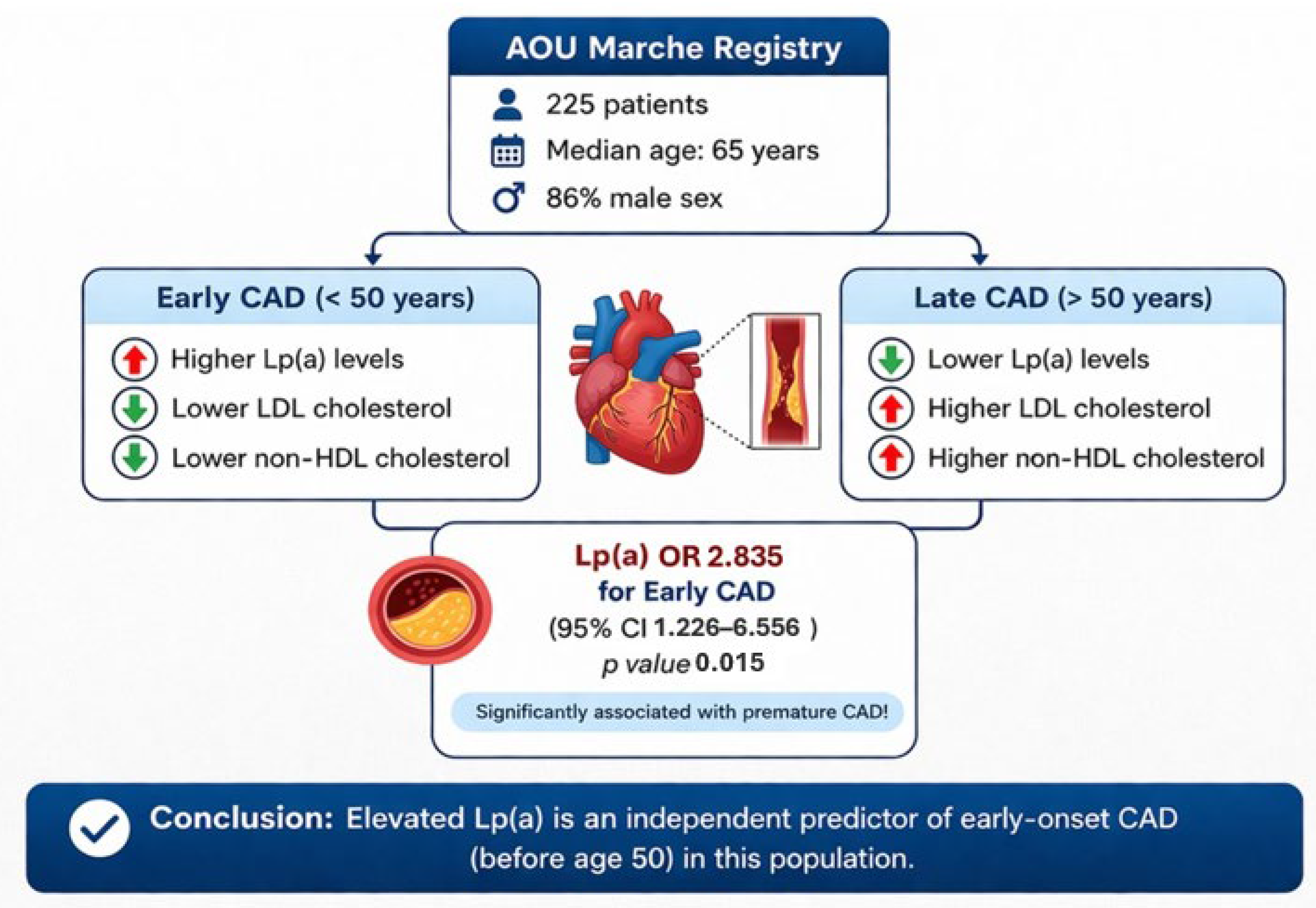
2.1. Study Population
2.2. Statistical Analysis
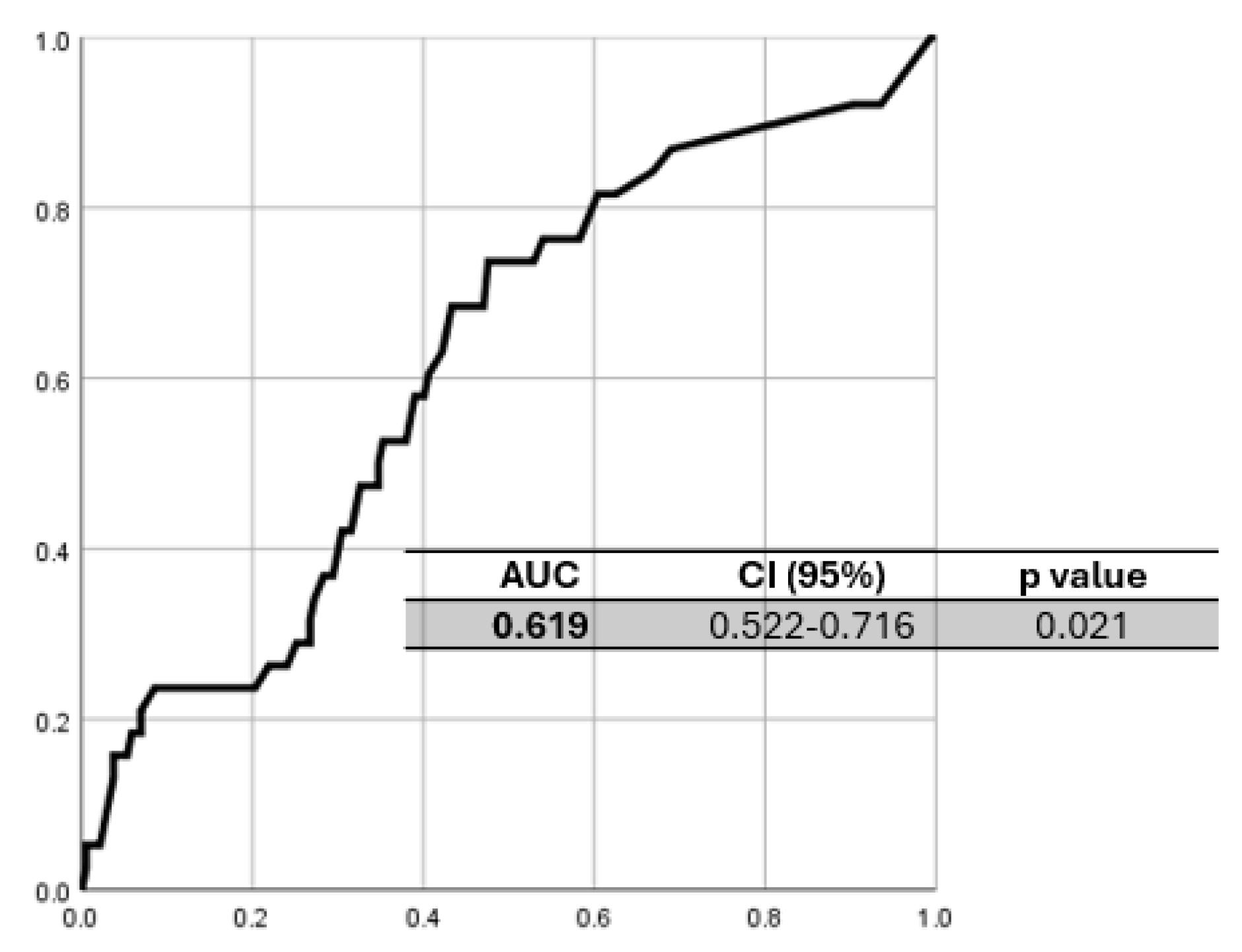
3. Results
4. Discussion
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Abbreviations
| ACS — Acute Coronary Syndrome |
| ASO — Antisense Oligonucleotide |
| AUC — Area Under the Curve |
| CABG — Coronary Artery Bypass Grafting |
| CAD — Coronary Artery Disease |
| CCS — Chronic Coronary Syndrome |
| CI — Confidence Interval |
| CVD — Cardiovascular Disease |
| HDL — High-Density Lipoprotein |
| IQR — Interquartile Range |
| ISR — In-Stent Restenosis |
| LDL — Low-Density Lipoprotein |
| Lp(a) — Lipoprotein(a) |
| NPV — Negative Predictive Value |
| OR — Odds Ratio |
| PCI — Percutaneous Coronary Intervention |
| PCSK9 — Proprotein Convertase Subtilisin/Kexin Type 9 |
| PPV — Positive Predictive Value |
| ROC — Receiver Operating Characteristic |
| siRNA — Small Interfering Ribonucleic Acid |
| SPSS — Statistical Package for the Social Sciences |
References
- Schmidt, K.; Noureen, A.; Kronenberg, F.; Utermann, G. Structure, function, and genetics of lipoprotein (a). J Lipid Res 2016, 57, 1339–1359. [Google Scholar] [CrossRef]
- Mack, S.; et al. A genome-wide association meta-analysis on lipoprotein (a) concentrations adjusted for apolipoprotein (a) isoforms. J Lipid Res 2017, 58, 1834–1844. [Google Scholar] [CrossRef] [PubMed]
- Patel, A. P.; et al. Lp(a) (Lipoprotein[a]) Concentrations and Incident Atherosclerotic Cardiovascular Disease: New Insights From a Large National Biobank. Arterioscler Thromb Vasc Biol 2021, 41, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Marcovina, S. M. Ancestry, Lipoprotein(a), and Cardiovascular Risk Thresholds: JACC Review Topic of the Week. J Am Coll Cardiol 2022, 80, 934–946. [Google Scholar] [CrossRef] [PubMed]
- Dzobo, K. E.; Kraaijenhof, J. M.; Stroes, E. S. G.; Nurmohamed, N. S.; Kroon, J. Lipoprotein(a): An underestimated inflammatory mastermind. Atherosclerosis 2022, 349, 101–109. [Google Scholar] [CrossRef]
- Greco, A.; et al. Lipoprotein(a) as a Pharmacological Target: Premises, Promises, and Prospects. Circulation 2025, 151, 400–415. [Google Scholar] [CrossRef]
- Wong, N. D.; et al. Lipoprotein(a) and Long-Term Cardiovascular Risk in a Multi-Ethnic Pooled Prospective Cohort. J Am Coll Cardiol 2024, 83, 1511–1525. [Google Scholar] [CrossRef]
- Madsen, C. M.; Kamstrup, P. R.; Langsted, A.; Varbo, A.; Nordestgaard, B. G. Lipoprotein(a)-Lowering by 50 mg/dL (105 nmol/L) May Be Needed to Reduce Cardiovascular Disease 20% in Secondary Prevention: A Population-Based Study. ATVB 2020, 40, 255–266. [Google Scholar] [CrossRef]
- Viney, N. J.; et al. Antisense oligonucleotides targeting apolipoprotein(a) in people with raised lipoprotein(a): two randomised, double-blind, placebo-controlled, dose-ranging trials. Lancet 2016, 388, 2239–2253. [Google Scholar] [CrossRef]
- Cho, L.; et al. Design and Rationale of Lp(a)HORIZON Trial: Assessing the Effect of Lipoprotein(a) Lowering With Pelacarsen on Major Cardiovascular Events in Patients With CVD and Elevated Lp(a). Am Heart J 2025, 287, 1–9. [Google Scholar] [CrossRef]
- O’Donoghue, M. L.; et al. The Off-Treatment Effects of Olpasiran on Lipoprotein(a) Lowering: OCEAN(a)-DOSE Extension Period Results. J Am Coll Cardiol 2024, 84, 790–797. [Google Scholar] [CrossRef]
- O’Donoghue, M. L.; et al. Small Interfering RNA to Reduce Lipoprotein(a) in Cardiovascular Disease. N Engl J Med 2022, 387, 1855–1864. [Google Scholar] [CrossRef]
- Nicholls, S. J.; et al. Oral Muvalaplin for Lowering of Lipoprotein(a): A Randomized Clinical Trial. JAMA 2025, 333, 222–231. [Google Scholar] [CrossRef]
- de Boer, L. M.; et al. Statin therapy and lipoprotein(a) levels: a systematic review and meta-analysis. Eur. J. Prev. Cardiol. 2022, 29, 779–792. [Google Scholar] [CrossRef]
- Khera, A. V.; et al. Lipoprotein(a) concentrations, rosuvastatin therapy, and residual vascular risk: an analysis from the JUPITER Trial (Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin). Circulation 2014, 129, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.; De Irala, J.; Palacios, M. Lipoprotein(a), tobacco use, and risk of coronary heart disease. International Journal of Cardiology 2000, 74, 233–234. [Google Scholar] [CrossRef] [PubMed]
- Gossett, L. K.; et al. Smoking Intensity and Lipoprotein Abnormalities in Active Smokers. J Clin Lipidol 2009, 3, 372–378. [Google Scholar] [CrossRef]
- Kauss, A. R.; et al. Smoking and apolipoprotein levels: A meta-analysis of published data. Toxicol Rep 2022, 9, 1150–1171. [Google Scholar] [CrossRef]
- Vrints, C.; et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. European Heart Journal 2024, 45, 3415–3537. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; et al. Association between lipoprotein(a) and premature atherosclerotic cardiovascular disease: a systematic review and meta-analysis. Eur Heart J Open 2024, 4, oeae031. [Google Scholar] [CrossRef]
- Cesaro, A.; et al. Role of LipoprotEin(a) in CardiovascuLar diseases and premature acute coronary syndromes (RELACS study): Impact of Lipoprotein(a) levels on the premature coronary event and the severity of coronary artery disease. Nutr Metab Cardiovasc Dis 2025, 35, 103843. [Google Scholar] [CrossRef] [PubMed]
| Table 1 | Total (n = 225) |
First PCI (n = 181) |
CABG (n = 13) |
In-stent restenosis (n = 31) |
p value |
|---|---|---|---|---|---|
| Age, years | 65 (55–74) | 62 (55–73)* | 73 (69–79) | 70 (63–73)* | 0.010 |
| Lp(a), mg/dL | 24 (10–67) | 21 (10–69) | 43 (19–70) | 29 (10–58) | 0.689 |
| Non–HDL cholesterol, mg/dL | 99 (69–132) | 107 (75–141) *° | 70 (58–108) * | 66 (54–79) ° | <0.001 |
| Triglycerides, mg/dL | 109 (87–144) | 110 (90–148) | 108 (71–148) | 99 (72–149) | 0.369 |
| LDL cholesterol, mg/dL | 71 (49–106) | 82 (55–108) * | 54 (39–76) | 50 (34–76) * | <0.001 |
| ACS at admission, n (%) | 96 (43) | 82 (45) * | 1 (8) *° | 13 (42) ° | 0.030 |
| High-intensity statin therapy, n (%) | 82 (37) | 57 (32) * | 6 (50) | 19 (66) * | 0.002 |
| Ezetimibe therapy, n (%) | 83 (38) | 59 (33) * | 6 (50) | 18 (60) * | 0.013 |
| PCSK9 inhibitors, n (%) | 4 (2) | 3 (2) | 0 (0) | 1 (3) | 0.731 |
| Bempedoic acid, n (%) | 3 (1) | 3 (2) | 0 (0) | 0 (0) | 0.697 |
| Inclisiran, n (%) | 1 (0) | 0 (0) * | 0 (0) ° | 1 (3) *° | 0.043 |
| Smoking history, n (%) | 136 (61) | 108 (60) | 8 (67) | 20 (65) | 0.799 |
| Hypertension, n (%) | 161 (72) | 123 (68) * | 9 (70) ° | 29 (94) *° | 0.014 |
| Diabetes mellitus, n (%) | 57 (25) | 40 (22) | 4 (31) | 13 (42) | 0.057 |
| ACS: acute coronary syndrome; HDL:high density lipoprotein; LDL: low density lipoprotein; PCSK9: Proprotein Convertase Subtilisin/Kexin type 9 | |||||
| Table 2 | <50 years (n = 38) | ≥50 years (n = 187) | p value |
|---|---|---|---|
| Lp(a), mg/dL | 42 (19–75) | 21 (10–66) | 0.020 |
| Non–HDL cholesterol, mg/dL | 84 (60–115) | 106 (74–136) | 0.010 |
| Triglycerides, mg/dL | 108 (83–152) | 111 (88–143) | 0.578 |
| LDL cholesterol, mg/dL | 61 (41–82) | 78 (53–107) | 0.012 |
| ACS at presentation, n (%) | 15 (40) | 81 (43) | 0.662 |
| High-intensity statin therapy, n (%) | 23 (60) | 59 (33) | 0.001 |
| Ezetimibe therapy, n (%) | 19 (50) | 64 (35) | 0.086 |
| PCSK9 inhibitors, n (%) | 2 (5) | 2 (1) | 0.081 |
| Bempedoic acid, n (%) | 3 (8) | 0 (0) | <0.001 |
| Inclisiran, n (%) | 0 (0) | 1 (1) | 0.651 |
| Smoking history, n (%) | 29 (76) | 107 (58) | 0.031 |
| Hypertension, n (%) | 21 (55) | 140 (75) | 0.015 |
| Diabetes mellitus, n (%) | 5 (13) | 52 (29) | 0.058 |
| ACS: acute coronary syndrome; HDL:high density lipoprotein; LDL: low density lipoprotein; PCSK9: Proprotein Convertase Subtilisin/Kexin type 9 | |||
| Table 3 | |||
|---|---|---|---|
| Variable | OR | 95% CI | p value |
| Lp(a), mg/dl | 2.671 | 1.160-6.149 | 0.021 |
| Non-HDL, mg/dl | 0.991 | 0.981–1.000 | 0.046 |
| TG, mg/dl | 0.999 | 0.994–1.004 | 0.688 |
| LDL, mg/dl | 0.990 | 0.980–1.000 | 0.047 |
| ACS, % | 0.853 | 0.419–1.739 | 0.663 |
| High-intensity statin, % | 3.171 | 1.542–6.520 | 0.002 |
| Smoking, % | 2.379 | 1.066–5.307 | 0.034 |
| Hypertension, % | 0.416 | 0.202–0.852 | 0.017 |
| Diabetes, % | 0.393 | 0.146–1.062 | 0.066 |
| ACS: acute coronary syndrome; LDL: low density lipoprotein; TG: triglycerides | |||
| Table 4 | |||
|---|---|---|---|
| Variable | OR | 95% CI | p value |
| Lp(a), mg/dl | 2.835 | 1.226–6.556 | 0.015 |
| Smoking, % | 2.516 | 1.116–5.673 | 0.026 |
| Table 5 | Lipoprotein | Coefficient | p value |
|---|---|---|---|
| High intensity statin | LDL | -0.322 | 0.000 |
| Non-HDL | -0.342 | 0.000 | |
| Lp (a) | 0.044 | 0.510 |
| Table 6 | |
| Sensibilità | 74% |
| Specificità | 52% |
| PPV | 24% |
| NPV | 91% |
| NPV: negative predictive value; PPV: positive predictive value | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




