Submitted:
18 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Warburg Effect and Tumor Metabolism: The Central Role of NAD+/NADH
2.1. Glycolytic Dependence of Cancer Cells
2.2. The NAD+ Paradox in Oncology vs Anti-Aging
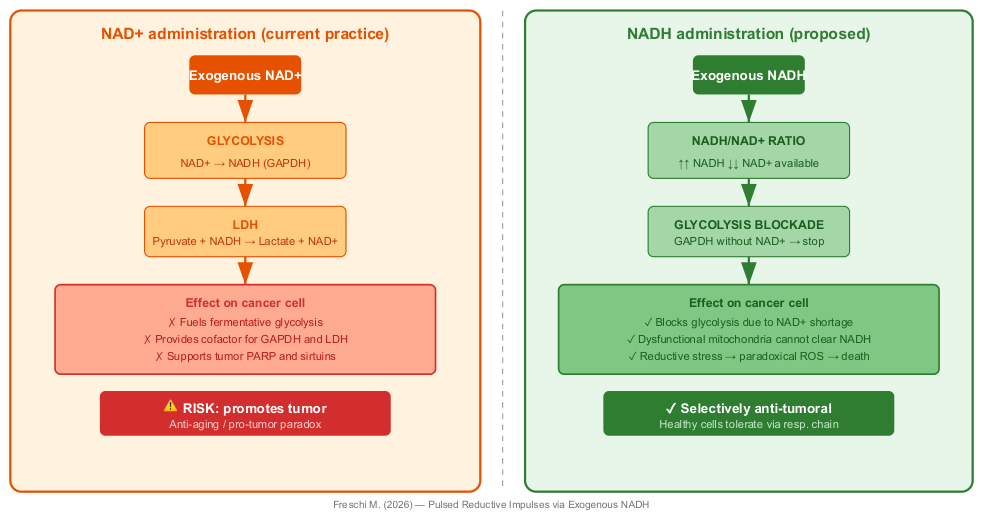
2.3. Exogenous Reducing Equivalents vs NAD+: A Fundamental Distinction
3. Redox State and Tumor Microenvironment
3.1. The Oxidative-Inflammatory Environment as a Tumor Substrate
3.2. The Transmembrane Redox Interface
- (a)
- Transplasma membrane redox systems (ECTO-NOX): membrane proteins that transfer electrons from intracellular NADH to extracellular acceptors; in a reduced external environment, these systems are blocked, causing intracellular NADH accumulation [18].
- (b)
- Cysteine/cystine system (xCT/SLC7A11): the xCT transporter imports cystine (oxidized form), which is internally reduced to cysteine for glutathione synthesis. A reductive extracellular environment alters the cysteine/cystine ratio, modifying the intracellular glutathione pool [19].
- (c)
- Redox-sensitive receptors: EGFR, IGFR, and integrins possess extracellular domains with cysteine residues whose oxidation is required for dimerization and activation. A reductive environment suppresses these proliferative signals [20].
- (d)
- Direct ROS diffusion: hydrogen peroxide (H₂O₂) crosses the cell membrane, including through aquaporins, acting as a transmembrane redox messenger [21].
- (e)
- Lactate/pH gradients: lactate transport through MCT transporters is dependent on the pH gradient; alterations in the extracellular environment modify this gradient and the metabolic shuttle between cancer cells and stroma [22].
- (f)
- Intercellular communication: gap junctions (connexins) allow the direct passage of NADH, NAD+, glutathione, and second messengers between adjacent cells [23].
3.3. Reductive Stress: A Mirror Concept to Oxidative Stress
4. Limitations of Conventional Antioxidants
4.1. Lack of Targeting
4.2. Secondary Pro-Oxidation
4.3. Inadequate Compartmentalization
4.4. The Advantage of Reducing Equivalents as Electron Vehicles
5. Hypothesis: Cyclic Reductive Impulses via Exogenous Reducing Equivalents
5.1. Rationale for Intrinsic Metabolic Selectivity
- (a)
- Glycolytic blockade: NAD+ shortage arrests GAPDH, blocking glycolysis—the primary source of ATP for the cancer cell.
- (b)
- Blockade of mitochondrial substrate level phosphorylation (mSLP): the alpha-ketoglutarate dehydrogenase reaction, which converts alpha-ketoglutarate to succinyl-CoA in the glutaminolysis pathway, requires NAD+ as an electron acceptor. The depletion of NAD+ caused by NADH accumulation blocks this reaction, cutting off the substrate supply to succinyl-CoA ligase and halting mSLP-derived ATP production [12,13]. This means that the cancer cell cannot use glutamine as an alternative energy source to bypass the glycolytic blockade.
- (c)
- Metabolic congestion: NADH excess allosterically inhibits key Krebs cycle enzymes (isocitrate dehydrogenase, alpha-ketoglutarate dehydrogenase), halting intermediary metabolism.
- (d)
- Paradoxical ROS generation: excess electrons leak from the saturated and dysfunctional respiratory chain, generating superoxide that the cancer cell—already with antioxidant systems at their limit—cannot neutralize.
- (e)
- Bioenergetic collapse: without ATP from glycolysis, without functional oxidative phosphorylation, and without mSLP from glutaminolysis, the cancer cell undergoes a total energy crisis with consequent plasma membrane depolarization and activation of cell death pathways. The cancer cell is thus subjected to a simultaneous blockade of all three known ATP-generating pathways.
5.1.1. Unifying the Extracellular and Intracellular Reductive Stress Mechanisms
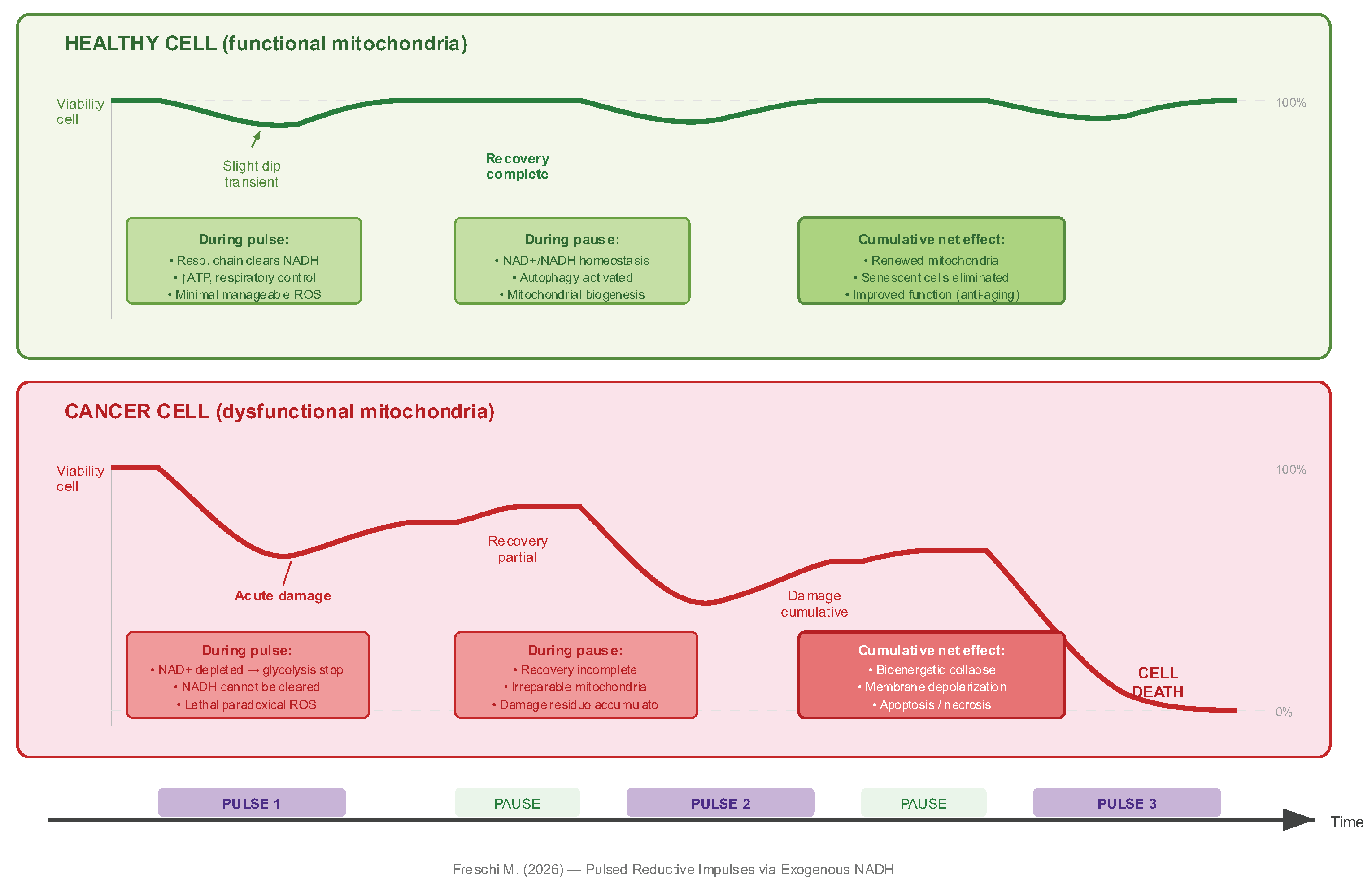
5.2. Press-Pulse Logic Applied to Redox State
5.3. Synergy with Glucose Restriction
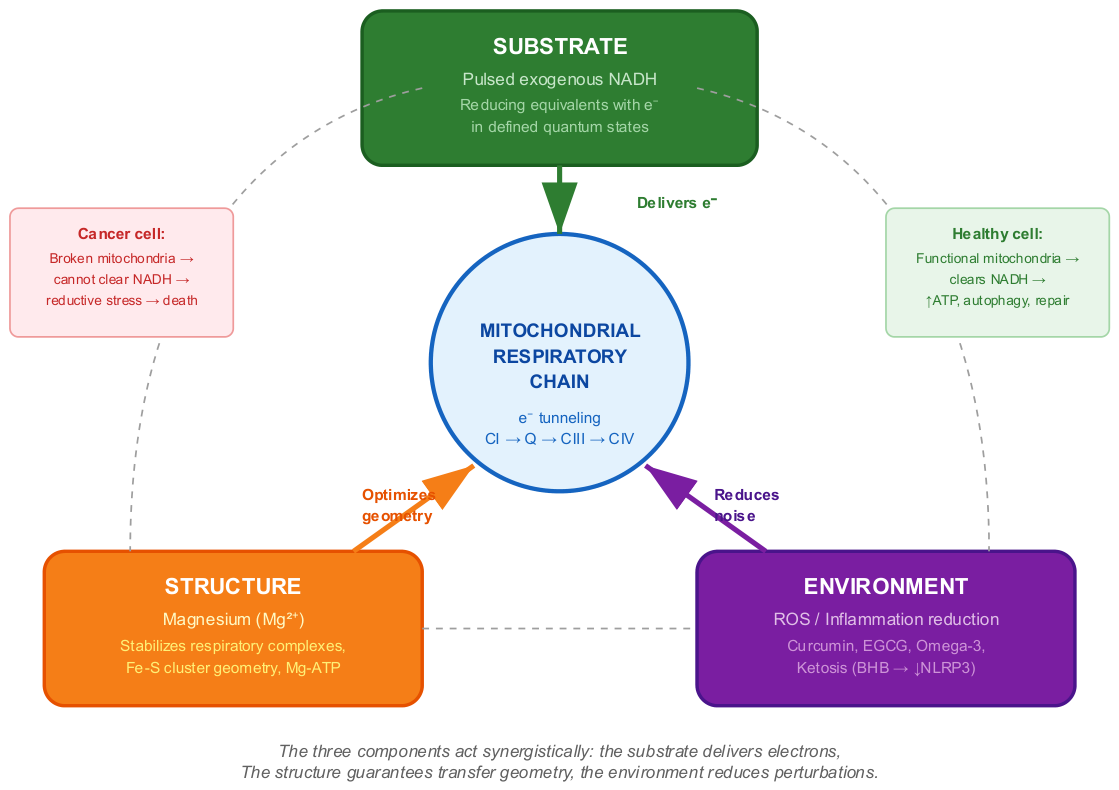
5.4. The Therapeutic Triad: Substrate, Structure, Environment
5.5. Dual-Compartment Rationale: Extracellular Blockade and Intracellular Stress
6. Proposed Experimental Protocol
6.1. Preclinical In Vitro Phase
- Warburg-dominant tumor lines with documented glycolytic dependence and compromised OxPhos: PC-3 and LNCaP (prostate), HeLa (cervical), MCF-7 (breast). These lines test the extracellular blockade mechanism via MCT-mediated lactate export.
- OxPhos-competent tumor lines with elevated reductive stress vulnerability, as characterized by Weiss-Sadan et al. [34]: KEAP1-mutant NSCLC lines (A549, H1299) and, as a complementary model, A549 treated with the KEAP1 inhibitor KI696 to induce NRF2-driven reductive strain. These lines test whether extracellular NADH saturation recapitulates the selective vulnerability previously demonstrated only with Complex I inhibition.
- Healthy control lines to document the therapeutic window: RWPE-1 (prostate), MCF-10A (breast), BEAS-2B (bronchial epithelium).
- Cell viability: MTT assay, trypan blue exclusion, clonogenic survival.
- Cytosolic NADH/NAD⁺ ratio: enzymatic assays and genetically encoded fluorescent reporters (SoNar, Peredox) to capture the predicted rise in intracellular NADH, which is central to the hypothesis.
- Extracellular lactate and intracellular lactate accumulation: to document MCT-mediated export blockade.
- ATP levels: total cellular ATP and compartment-specific quantification, including separate assessment of glycolytic and OxPhos contributions via selective inhibitors.
- ROS generation: DCFDA probe and mitochondrial superoxide-specific probes (MitoSOX) to detect the paradoxical ROS generation from saturated electron transport chains.
- Mitochondrial membrane potential: JC-1 or TMRM.
- Spare respiratory capacity and basal oxygen consumption: Seahorse extracellular flux analysis, as a direct measure of the redox buffering capacity hypothesized to underlie the differential tolerance between healthy and transformed cells.
- Pulsed NADH (2h exposure + 22h recovery, repeated for 5 cycles).
- Continuous NADH (same total dose distributed over 120h).
- Pulsed IACS-010759 (Complex I inhibitor, as a direct test of the Weiss-Sadan mechanism).
- Combined pulsed NADH + IACS-010759 (dual-compartment strategy, Section 5.5).
- Pulsed NADH in glucose-free medium (ketosis/fasting simulation, testing the synergy with substrate restriction per Section 5.3).
- Untreated control.
6.2. Modalities for Reducing Equivalent Delivery
6.3. Pulse Protocol Parameters (Preliminary Clinical Proposal)
- Pulse duration: 2–4 hours of slow intravenous infusion
- Frequency: 2–3 sessions per week during intensive cycles
- Cycle duration: 2–3 weeks
- Inter-cycle pause: 4–6 weeks
- Metabolic combination: sessions should ideally be conducted during fasting periods (≥16 hours) to maximize the metabolic vulnerability of cancer cells
- Supplementation: magnesium glycinate/citrate (400–800 mg/day), reduced CoQ10 (ubiquinol, 200–400 mg/day)
- Mandatory monitoring: plasma NAD+/NADH ratio, blood lactate, blood glucose, ketones, continuous ECG during infusion, electrolytes (Na+, K+, Mg²⁺), inflammatory markers (CRP, IL-6), complete blood count
6.4. Safety Criteria
- Cardiac risk: acute alterations of the NAD+/NADH ratio may affect cardiomyocyte membrane potential. Continuous ECG monitoring during infusion is mandatory.
- Metabolic risk: an excessive infusion rate could cause paradoxical lactic acidosis or excessively activate respiratory control, reducing ATP production in healthy cells. Gradual dose titration is recommended.
- Electrolyte risk: reductive stress could transiently affect the Na+/K+-ATPase pump. Monitoring of electrolytes before, during, and after infusion is essential.
7. Anti-Cancer and Anti-Aging Convergence
7.1. Shared Mechanisms
7.2. The Nocturnal Window as a Physiological Reductive Impulse
7.3. Integrated Preventive Protocol
8. Limitations and Future Directions
8.1. Limitations of the Present Hypothesis
- (a)
- Absence of specific clinical data: no clinical trials exist that test pulsed NADH administration with the metabolic selectivity rationale described here. The experiences of the Hyperthermia Centre Hannover [8] and Birkmayer [7] use NADH in a different therapeutic framework (general support) without press-pulse logic.
- (b)
- Pharmacokinetics of exogenous NADH: the intracellular bioavailability of intravenously administered NADH is not fully characterized. NADH could be metabolized in plasma before reaching target cells. Dedicated pharmacokinetic studies are necessary. It should be noted, however, that even the fraction of exogenous NADH that undergoes oxidation to NAD+ in the plasma does not represent a significant risk of fueling tumor glycolysis. Extracellular NAD+ is rapidly hydrolyzed by ectonucleotidases, principally CD38 and CD157, to nicotinamide and ADP-ribose, with a plasma half-life on the order of minutes [44]. Importantly, CD38 is overexpressed in the tumor microenvironment, particularly on immunosuppressive myeloid cells, meaning that NAD+ is degraded even more rapidly in the peritumoral space than in the systemic circulation. The degradation products—nicotinamide and ADP-ribose—do not directly fuel glycolysis and require slow intracellular resynthesis via NAMPT to regenerate NAD+, a process too slow to compensate for the acute NAD+ depletion induced by the reductive impulse.
- (c)
- Tumor heterogeneity: not all tumors exhibit the same degree of mitochondrial dysfunction. Tumors with partially preserved oxidative phosphorylation might better tolerate the reductive impulse. Patient selection based on metabolic imaging (FDG-PET, as an indicator of the Warburg effect) could improve selectivity.
- (d)
- Therapeutic window: the distance between the dose effective against cancer cells and the dose toxic to healthy cells—considering the respiratory control mechanism that also limits the clearance capacity of healthy cells—could be narrow. Only preclinical experimentation can define this window.
- (e)
- Interaction with conventional therapies: the effect of the reductive impulse on cells treated with chemotherapy, radiotherapy, or immunotherapy is unknown and requires specific study. Of particular interest would be the combination with PARP inhibitors (e.g., olaparib), which deplete intracellular NAD+ through a complementary mechanism.
- (f)
- Need for structural turnover: the administration of exogenous reducing equivalents does not repair damaged respiratory complexes. Its effect is to bypass metabolic bottlenecks and, through cyclic impulses, to stimulate the mitophagy and mitochondrial biogenesis that replace structurally compromised components.
8.2. Future Directions
- (a)
- Preclinical in vitro studies with the protocol described in Section 6.1 to validate the selectivity of the reductive impulse, with specific measurement of the simultaneous blockade of glycolysis, OxPhos, and mSLP.
- (b)
- Development of NADH formulations in liposomes or nanoparticles to improve intracellular delivery.
- (c)
- Combined study of pulsed NADH + fasting + PARP inhibitors to maximize NAD+ depletion in cancer cells through complementary mechanisms.
- (d)
- Experimental validation of the dual-compartment redox blockade concept: systematic comparison of extracellular-only perturbation (NADH) versus combined extracellular + intracellular perturbation using agents with different membrane permeability profiles, to determine whether dual-compartment approaches enhance selectivity and efficacy.
- (e)
- Quantum biology studies (see Appendix) to characterize the quantum states of electrons in exogenous vs endogenous NADH and their functional relevance.
- (f)
- Development of predictive response biomarkers based on tumor metabolic profile (NAD+/NADH ratio, complex I expression, LDH activity, succinate export as indicator of mSLP dependence).
- (g)
- Comparative evaluation of different modalities for exogenous reducing equivalent delivery (NADH, ubiquinol, H₂, and future catalytic redox vehicles) within the press-pulse protocol, with attention to the coherence of the quantum states of the delivered electrons.
- (h)
- Investigation of the potential immunometabolic effects of reductive impulses on tumor-associated macrophage polarization and T lymphocyte function in the tumor microenvironment.
- (i)
- Investigation of the combination of pulsed reducing equivalent infusions with CD38 inhibitors (e.g., daratumumab, already approved for multiple myeloma). CD38 overexpression in the tumor microenvironment depletes extracellular NAD+, contributing to T cell exhaustion. Inhibition of CD38 during the reductive impulse could simultaneously preserve extracellular NAD+ for immune cell function while the NADH-mediated thermodynamic blockade disrupts tumor metabolism—creating a three-pronged attack combining metabolic blockade, immune restoration via CD38 inhibition, and immune unmasking via ROS neutralization.
- (j)
- Exploration of non-molecular electron delivery methods capable of simultaneously achieving both extracellular and intracellular redox perturbation, bypassing the membrane permeability and metabolic byproduct limitations inherent to molecular reducing vehicles. Such approaches could represent a significant advancement over carrier-based strategies by providing direct reducing equivalents to all cellular compartments without the constraints of enzymatic recognition or transmembrane transport.
9. Conclusions
Funding
Use of Artificial Intelligence
Acknowledgments
Conflicts of Interest
Appendix A. Quantum Biology Perspectives
Appendix A.1. Preamble
Appendix A.2. Quantum Tunneling in the Respiratory Chain
Appendix A.3. Quantum Coherence in Biological Systems
Appendix A.4. Quantum States of Electrons in Exogenous vs Endogenous Reducing Equivalents
Appendix A.5. Decoherence as a Component of Aging
Appendix A.6. Experimental Verification
- (a)
- Pulsed EPR spectroscopy and ENDOR: to compare the spin states and relaxation times (T1, T2) of electrons in fresh synthetic NADH vs NADH extracted from young tissues vs NADH extracted from aged tissues.
- (b)
- High-resolution oximetry (Oroboros O2k): to measure respiratory chain efficiency in mitochondria isolated from young vs aged tissues, before and after incubation with exogenous NADH.
- (c)
- Time-resolved fluorescence spectroscopy: to characterize the dynamics of enzyme-bound NADH in different age and pathology contexts.
- (d)
- Comparative measurement of electronic coherence in alternative vehicles: pulsed EPR T1/T2 characterization of candidate non-NADH electron donors (coordinated metal complexes, protected catalytic systems, biocompatible quantum dots) in aqueous and lipid-mimetic media, correlated with functional electron-transfer efficiency to isolated Complex I or submitochondrial particles. This would establish whether vehicles with longer intrinsic coherence times yield measurably greater transfer efficiency, directly testing the central prediction of this appendix.
References
- Warburg O. On the origin of cancer cells. Science. 1956;123(3191):309-314. [CrossRef]
- Seyfried TN. Cancer as a Metabolic Disease: On the Origin, Management, and Prevention of Cancer. Hoboken, NJ: Wiley; 2012.
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: an expanding universe. Cell. 2023;186(2):243-278. [CrossRef]
- Massudi H, Grant R, Braidy N, Guest J, Farnsworth B, Guillemin GJ. Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. PLoS One. 2012;7(7):e42357. [CrossRef]
- Grant R, Berg J, Mestayer R, Braidy N, Bennett J, Broom S, Watson J. A pilot study investigating changes in the human plasma and urine NAD+ metabolome during a 6 hour intravenous infusion of NAD+. Front Aging Neurosci. 2019;11:257. [CrossRef]
- Yaku K, Okabe K, Hikosaka K, Nakagawa T. NAD metabolism in cancer therapeutics. Front Oncol. 2018;8:622. [CrossRef]
- Birkmayer JGD, Vrecko C, Volc D, Birkmayer W. Nicotinamide adenine dinucleotide (NADH)—a new therapeutic approach to Parkinson’s disease. Comparison of oral and parenteral application. Acta Neurol Scand Suppl. 1993;146:32-35. [CrossRef]
- Hyperthermia Centre Hannover. NADH infusions in oncology. Available at: https://www.hyperthermia-centre-hannover.com. Accessed April 2026.
- Seyfried TN, Yu G, Maroon JC, D’Agostino DP. Press-pulse: a novel therapeutic strategy for the metabolic management of cancer. Nutr Metab (Lond). 2017;14:19. [CrossRef]
- Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science. 2009;324(5930):1029-1033. [CrossRef]
- Verdin E. NAD+ in aging, metabolism, and neurodegeneration. Science. 2015;350(6265):1208-1213. [CrossRef]
- Seyfried TN, Lee DC, Duraj T, Ta NL, Mukherjee P, Kiebish M, Arismendi-Morillo G, Chinopoulos C. The Warburg hypothesis and the emergence of the mitochondrial metabolic theory of cancer. J Bioenerg Biomembr. 2025;57(2-3):57-83. [CrossRef]
- Lee DC, Ta L, Mukherjee P, Duraj T, Domin M, Greenwood B, Karmacharya S, Narain NR, Kiebish M, Chinopoulos C, Seyfried TN. Amino acid and glucose fermentation maintain ATP content in mouse and human malignant glioma cells. ASN Neuro. 2024;16(1):2422268. [CrossRef]
- Lucena-Cacace A, Otero-Albiol D, Jimenez-Garcia MP, Muñoz-Galvan S, Carnero A. NAMPT overexpression induces cancer stemness and defines a novel tumor signature for glioma prognosis. Oncotarget. 2017;8(59):99514-99530. [CrossRef]
- Hasmann M, Schemainda I. FK866, a highly specific noncompetitive inhibitor of nicotinamide phosphoribosyltransferase, represents a novel mechanism for induction of tumor cell apoptosis. Cancer Res. 2003;63(21):7436-7442.
- Sabharwal SS, Schumacker PT. Mitochondrial ROS in cancer: initiators, amplifiers or an Achilles’ heel? Nat Rev Cancer. 2014;14(11):709-721. [CrossRef]
- Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436-444. [CrossRef]
- Morré DJ, Morré DM. ECTO-NOX Proteins: Growth, Cancer, and Aging. New York: Springer; 2013.
- Lewerenz J, Hewett SJ, Huang Y, Lambros M, Gout PW, Kalivas PW, Massie A, Smolders I, Methner A, Pergande M, Smith SB, Ganapathy V, Maher P. The cystine/glutamate antiporter system xc- in health and disease. Antioxid Redox Signal. 2013;18(5):522-555. [CrossRef]
- Paulsen CE, Carroll KS. Cysteine-mediated redox signaling: chemistry, biology, and tools for discovery. Chem Rev. 2013;113(7):4633-4679. [CrossRef]
- Bienert GP, Schjoerring JK, Jahn TP. Membrane transport of hydrogen peroxide. Biochim Biophys Acta. 2006;1758(8):994-1003. [CrossRef]
- Halestrap AP, Wilson MC. The monocarboxylate transporter family—role and regulation. IUBMB Life. 2012;64(2):109-119. [CrossRef]
- Aasen T, Mesnil M, Naus CC, Lampe PD, Laird DW. Gap junctions and cancer: communicating for 50 years. Nat Rev Cancer. 2016;16(14):775-788. [CrossRef]
- Xiao W, Loscalzo J. Metabolic responses to reductive stress. Antioxid Redox Signal. 2020;32(20):1330-1347. [CrossRef]
- Aon MA, Cortassa S, O’Rourke B. Redox-optimized ROS balance: a unifying hypothesis. Biochim Biophys Acta. 2010;1797(6-7):865-877. [CrossRef]
- Halliwell B. The antioxidant paradox: less paradoxical now? Br J Clin Pharmacol. 2013;75(3):637-644. [CrossRef]
- Carr AC, Frei B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am J Clin Nutr. 1999;69(6):1086-1107. [CrossRef]
- Traber MG, Atkinson J. Vitamin E, antioxidant and nothing more. Free Radic Biol Med. 2007;43(1):4-15. [CrossRef]
- The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N Engl J Med. 1994;330(17):1029-1035. [CrossRef]
- Klein EA, Thompson IM Jr, Tangen CM, Crowley JJ, Lucia MS, Goodman PJ, Minasian LM, Ford LG, Parnes HL, Gaziano JM, Karp DD, Lieber MM, Walther PJ, Klotz L, Parsons JK, Chin JL, Darke AK, Lippman SM, Goodman GE, Meyskens FL Jr, Baker LH. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011;306(16):1549-1556. [CrossRef]
- Murphy MP, Smith RAJ. Targeting antioxidants to mitochondria by conjugation to lipophilic cations. Annu Rev Pharmacol Toxicol. 2007;47:629-656. [CrossRef]
- Skulachev VP. An attempt to prevent senescence: a mitochondrial approach. Biochim Biophys Acta. 2009;1787(5):437-461. [CrossRef]
- Chance B, Williams GR. Respiratory enzymes in oxidative phosphorylation. III. The steady state. J Biol Chem. 1955;217(1):409-427.
- Weiss-Sadan T, Ge M, Hayashi M, Gohar M, Yao CH, de Groot A, Harry S, Carlin A, Fischer H, Shi L, Wei TY, Adelmann CH, Wolf K, Vornbäumen T, Dürr BR, Takahashi M, Richter M, Zhang J, Yang TY, Vijay V, Fisher DE, Hata AN, Haigis MC, Mostoslavsky R, Bardeesy N, Papagiannakopoulos T, Bar-Peled L. NRF2 activation induces NADH-reductive stress, providing a metabolic vulnerability in lung cancer. Cell Metab. 2023;35(3):487-503.e7. [CrossRef]
- Joiner MC, van der Kogel AJ, eds. Basic Clinical Radiobiology. 5th ed. Boca Raton: CRC Press; 2018.
- Sonveaux P, Végran F, Schroeder T, Wergin MC, Verrax J, Rabbani ZN, De Saedeleer CJ, Kennedy KM, Diepart C, Jordan BF, Kelley MJ, Gallez B, Wahl ML, Feron O, Dewhirst MW. Targeting lactate-fueled respiration selectively kills hypoxic tumor cells in mice. J Clin Invest. 2008;118(12):3930-3942. [CrossRef]
- de Baaij JHF, Hoenderop JGJ, Bindels RJM. Magnesium in man: implications for health and disease. Physiol Rev. 2015;95(1):1-46. [CrossRef]
- Ohsumi Y. Historical landmarks of autophagy research. Cell Res. 2014;24(1):9-23. [CrossRef]
- Youle RJ, Narendra DP. Mechanisms of mitophagy. Nat Rev Mol Cell Biol. 2011;12(1):9-14. [CrossRef]
- Hardie DG, Ross FA, Hawley SA. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2012;13(4):251-262. [CrossRef]
- Campisi J. Aging, cellular senescence, and cancer. Annu Rev Physiol. 2013;75:685-705. [CrossRef]
- Youm YH, Nguyen KY, Grant RW, Goldberg EL, Bodogai M, Kim D, D’Agostino D, Planavsky N, Lupfer C, Kanneganti TD, Kang S, Horvath TL, Fahmy TM, Crawford PA, Biragyn A, Alnemri E, Dixit VD. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat Med. 2015;21(3):263-269. [CrossRef]
- Saha T, Dash C, Jayabalan R, Khiste S, Kulkarni A, Kurmi K, Mondal J, Majumder PK, Bardia A, Jang HL, Sengupta S. Intercellular nanotubes mediate mitochondrial trafficking between cancer and immune cells. Nat Nanotechnol. 2022;17(1):98-106. [CrossRef]
- Camacho-Pereira J, Tarragó MG, Chini CCS, Nin V, Escande C, Warner GM, Puranik AS, Schoon RA, Reid JM, Galina A, Chini EN. CD38 dictates age-related NAD decline and mitochondrial dysfunction through an SIRT3-dependent mechanism. Cell Metab. 2016;23(6):1127-1139. [CrossRef]
- Moser CC, Keske JM, Warncke K, Farid RS, Dutton PL. Nature of biological electron transfer. Nature. 1992;355(6363):796-802. [CrossRef]
- Marcus RA, Sutin N. Electron transfers in chemistry and biology. Biochim Biophys Acta. 1985;811(3):265-322. [CrossRef]
- Engel GS, Calhoun TR, Read EL, Ahn TK, Mančal T, Cheng YC, Blankenship RE, Fleming GR. Evidence for wavelike energy transfer through quantum coherence in photosynthetic systems. Nature. 2007;446(7137):782-786. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



