Submitted:
02 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
Supplemental Oxygen
Clinical Delivery and Hemodynamic Impact
Hyperoxia
Types of Hyperoxia
Systemic Toxicity and Organ Dysfunction
Impact of Supplemental Oxygen on Lung Physiology
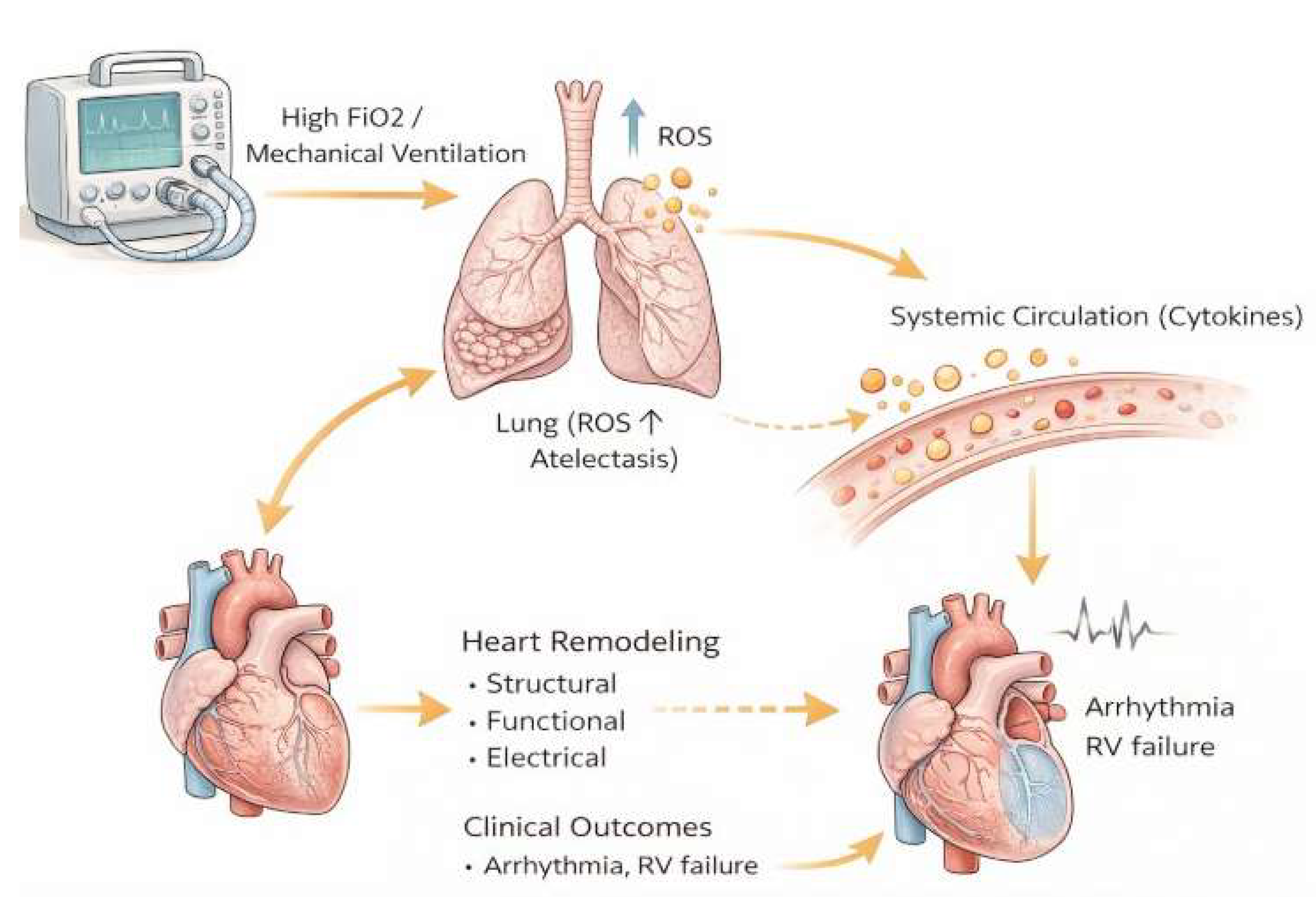
Supplemental Oxygen Induced Cardiac Pathophysiology
I. Mechanical Ventilation as a Structural Stressor of the Myocardium
II. Functional Cardiac Consequences of Mechanical Ventilation
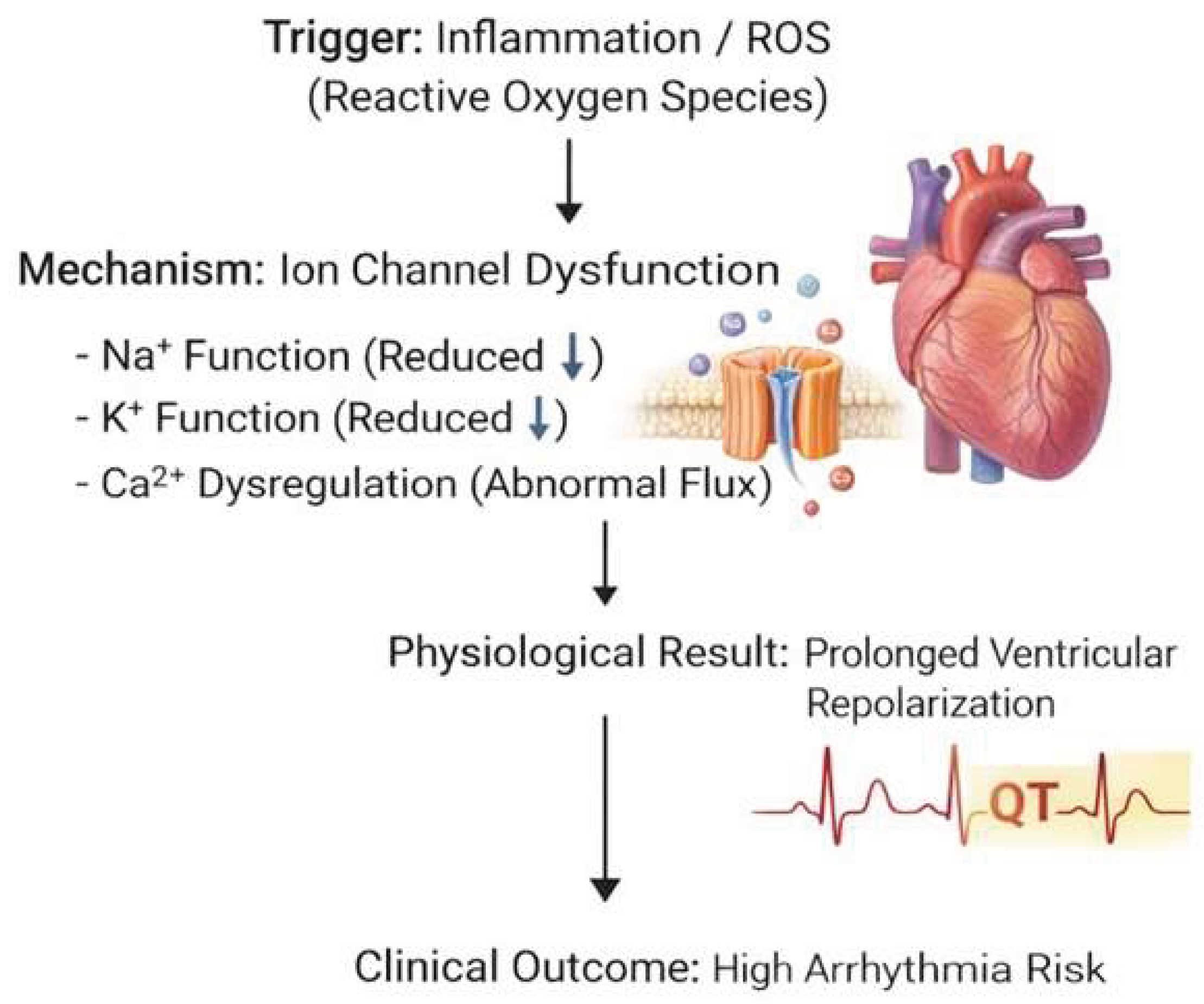
III. Electrical Remodeling During Mechanical Ventilation
IV Supplemental Oxygen Exacerbates Cardiac Injury in COVID-19 Patients
Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abbreviation | Full Term |
| AKI | Acute kidney injury |
| ARDS | Acute respiratory distress syndrome |
| AT1 | Angiotensin II type 1 receptor |
| ATP | Adenosine triphosphate |
| COPD | Chronic obstructive pulmonary disease |
| COVID-19 | Coronavirus disease 2019 |
| ECM | Extracellular matrix |
| EF | Ejection fraction |
| FiO₂ | Fraction of inspired oxygen |
| HALI | Hyperoxic acute lung injury |
| HDLVEF | Hyperdynamic left ventricular ejection fraction |
| HFNC | High-flow nasal cannula |
| HPV | Hypoxic pulmonary vasoconstriction |
| ICU | Intensive Care Unit |
| IL-6 | Interleukin-6 |
| I_K1 | Inward rectifier potassium current |
| I_Kr | Rapid delayed rectifier potassium current |
| I_Ks | Slow delayed rectifier potassium current |
| I_Na | Fast inward sodium current |
| I_to | Transient outward potassium current |
| LV | Left ventricle/left ventricular |
| LVOT VTI | Left ventricular outflow tract velocity-time integral |
| MAP | Mean arterial pressure |
| MI | Myocardial infarction |
| mRNA | Messenger RNA |
| MV | Mechanical ventilation |
| Nav1.5 | Voltage-gated sodium channel isoform 1.5 |
| NF-κB | Nuclear factor kappa B |
| NFAT | Nuclear factor of activated T-cells |
| NIV | Non-invasive ventilation |
| PaO₂ | Partial pressure of arterial oxygen |
| PEEP | Positive end-expiratory pressure |
| PI3K–Akt | Phosphoinositide 3-kinase – protein kinase B pathway |
| PVR | Pulmonary vascular resistance |
| RAAS | Renin–angiotensin–aldosterone system |
| ROS | Reactive oxygen species |
| RV | Right ventricle/right ventricular |
| RyR | Ryanodine receptor |
| SaO₂ | Arterial oxygen saturation |
| SCN5A | Gene encoding Nav1.5 sodium channel |
| SERCA | Sarco/endoplasmic reticulum calcium ATPase |
| SpO₂ | Peripheral oxygen saturation |
| T1DM | Type 1 diabetes mellitus |
| TAPSE | Tricuspid annular plane systolic excursion |
| TNF-α | Tumor necrosis factor alpha |
| VILI | Ventilator-induced lung injury |
| V/Q | Ventilation-perfusion |
References
- Lius, E.E.; Syafaah, I. Hyperoxia in the Management of Respiratory Failure: A Literature Review. Ann Med Surg (Lond) 2022, 81, 104393. [Google Scholar] [CrossRef] [PubMed]
- Mart, M.F.; Sendagire, C.; Ely, E.W.; Riviello, E.D.; Twagirumugabe, T. Oxygen as an Essential Medicine. Crit Care Clin 2022, 38(no. 4), 795–808. [Google Scholar] [CrossRef]
- Bhandari, V. Molecular Mechanisms of Hyperoxia-Induced Acute Lung Injury. Front Biosci 2008, 13, 6653–6661. [Google Scholar] [CrossRef] [PubMed]
- Busani, S.; Sarti, M.; Serra, F.; Gelmini, R.; Venturelli, S.; Munari, E.; Girardis, M. Revisited Hyperoxia Pathophysiology in the Perioperative Setting: A Narrative Review. Front Med (Lausanne) 2021, 8, 689450. [Google Scholar] [CrossRef]
- Chapalamadugu, K.C.; Panguluri, S.K.; Bennett, E.S.; Kolliputi, N.; Tipparaju, S. M. High Level of Oxygen Treatment Causes Cardiotoxicity with Arrhythmias and Redox Modulation. Toxicol Appl Pharmacol 2015, 282(no. 1), 100–107. [Google Scholar] [CrossRef]
- Duan, Y.; Li, X.; Han, S.; Guan, J.; Chen, Z.; Li, W.; Meng, R.; Ding, J. Normobaric Hyperoxia Therapy in Treating Stroke. Clin Interv Aging 2025, 20, 969–81. [Google Scholar] [CrossRef]
- Hochberg, C.H.; Semler, M.W.; Brower, R. G. Oxygen Toxicity in Critically Ill Adults. Am J Respir Crit Care Med 2021, 204(no. 6), 632–41. [Google Scholar] [CrossRef]
- Horn, Andrew G.; Kunkel, Olivia N.; Schulze, Kiana M.; Baumfalk, Dryden R.; Weber, Ramona E.; Poole, David C.; Behnke, Bradley J. Supplemental Oxygen Administration during Mechanical Ventilation Reduces Diaphragm Blood Flow and Oxygen Delivery. Journal of Applied Physiology 2022, 132(no. 5), 1190–200. [Google Scholar] [CrossRef]
- Horncastle, E.; Lumb, A. B. Hyperoxia in Anaesthesia and Intensive Care. BJA Educ 2019, 19(no. 6), 176–82. [Google Scholar] [CrossRef] [PubMed]
- Li, L.F.; Liao, S.K.; Ko, Y.S.; Lee, C.H.; Quinn, D. A. Hyperoxia Increases Ventilator-Induced Lung Injury Via Mitogen-Activated Protein Kinases: A Prospective, Controlled Animal Experiment. Crit Care 2007, 11(no. 1), R25. [Google Scholar] [CrossRef]
- Minkove, S.; Dhamapurkar, R.; Cui, X.; Li, Y.; Sun, J.; Cooper, D.; Eichacker, P.Q.; Torabi-Parizi, P. Effect of Low-to-Moderate Hyperoxia on Lung Injury in Preclinical Animal Models: A Systematic Review and Meta-Analysis. Intensive Care Med Exp 2023, 11(no. 1), 22. [Google Scholar] [CrossRef]
- Singer, M.; Young, P.J.; Laffey, J.G.; Asfar, P.; Taccone, F.S.; Skrifvars, M.B.; Meyhoff, C.S.; Radermacher, P. Dangers of Hyperoxia. Crit Care 2021, 25(no. 1), 440. [Google Scholar] [CrossRef]
- Ayalasomayajula, Y.; Hesaraghatta, A.; Dantuluri, N.; Yassine, J.; Saleem, F.; Mansour, N.; Chayawatto, C.; Rangarajan, N.; Rangarajan, S.; Krishnan, S.; Panguluri, S. K. Influence of Age and Sex on Physical, Cardiac Electrical and Functional Alterations in Progressive Hyperoxia Treatment: A Time Course Study in a Murine Model. Exp Gerontol 2024, 191, 112435. [Google Scholar] [CrossRef] [PubMed]
- Bojkovic, K.; Rodgers, J.L.; Vichare, R.; Nandi, A.; Mansour, N.; Saleem, F.; Abidin, Z.U.; Vanthenapalli, S.; Cheng, F.; Panguluri, S. K. The Implications of Hyperoxia, Type 1 Diabetes and Sex on Cardiovascular Physiology in Mice. Sci Rep 2021, 11(no. 1), 23086. [Google Scholar] [CrossRef] [PubMed]
- Panguluri, S.K.; Tur, J.; Fukumoto, J.; Deng, W.; Sneed, K.B.; Kolliputi, N.; Bennett, E.S.; Tipparaju, S. M. Hyperoxia-Induced Hypertrophy and Ion Channel Remodeling in Left Ventricle. Am J Physiol Heart Circ Physiol 2013, 304(no. 12), H1651–61. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, J.L.; Iyer, D.; Rodgers, L.E.; Vanthenapalli, S.; Panguluri, S. K. Impact of Hyperoxia on Cardiac Pathophysiology. J Cell Physiol 2019, 234(no. 8), 12595–603. [Google Scholar] [CrossRef]
- Rodgers, J.L.; Rodgers, L.E.; Tian, Z.; Allen-Gipson, D.; Panguluri, S. K. Sex Differences in Murine Cardiac Pathophysiology with Hyperoxia Exposure. J Cell Physiol 2019, 234(no. 2), 1491–501. [Google Scholar] [CrossRef]
- Rodgers, J.L.; Samal, E.; Mohapatra, S.; Panguluri, S. K. Hyperoxia-Induced Cardiotoxicity and Ventricular Remodeling in Type-Ii Diabetes Mice. Heart Vessels 2018, 33(no. 5), 561–72. [Google Scholar] [CrossRef]
- Rodgers, J.L.; Vanthenapalli, S.; Panguluri, S. K. Electrical Remodeling and Cardiotoxicity Precedes Structural and Functional Remodeling of Mouse Hearts under Hyperoxia Treatment. J Cell Physiol 2021, 236(no. 6), 4482–95. [Google Scholar] [CrossRef]
- Saleem, F.; Mansour, N.; Vichare, R.; Ayalasomayajula, Y.; Yassine, J.; Hesaraghatta, A.; Panguluri, S. K. Influence of Age on Hyperoxia-Induced Cardiac Pathophysiology in Type 1 Diabetes Mellitus (T1dm) Mouse Model. Cells 2023, 12(no. 11). [Google Scholar] [CrossRef]
- Vichare, R.; Saleem, F.; Mansour, N.; Bojkovic, K.; Cheng, F.; Biswal, M.; Panguluri, S. K. Impact of Age and Sex on Hyperoxia-Induced Cardiovascular Pathophysiology. Mech Ageing Dev 2022, 208, 111727. [Google Scholar] [CrossRef]
- Vysotskaya, Z.; Chidipi, B.; Rodgers, J.L.; Tang, X.; Samal, E.; Kolliputi, N.; Mohapatra, S.; Bennett, E.S.; Panguluri, S. K. Elevated Potassium Outward Currents in Hyperoxia Treated Atrial Cardiomyocytes. J Cell Physiol 2018, 233(no. 5), 4317–26. [Google Scholar] [CrossRef]
- Durlinger, E.M.J.; Spoelstra-de Man, A.M.E.; Smit, B.; de Grooth, H.J.; Girbes, A.R.J.; Oudemans-van Straaten, H.M.; Smulders, Y. M. Hyperoxia: At What Level of Spo2 Is a Patient Safe? A Study in Mechanically Ventilated Icu Patients. J Crit Care 2017, 39, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.N.; Kvietys, P. R. Reperfusion Injury and Reactive Oxygen Species: The Evolution of a Concept. Redox Biol 2015, 6, 524–51. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, S.S.; Pinsky, M. R. Heart-Lung Interactions during Mechanical Ventilation: The Basics. Ann Transl Med 2018, 6(no. 18), 349. [Google Scholar] [CrossRef]
- Weekley, M.S.; Lobo, C.M.; Bland, L. E. Oxygen Administration. In Statpearls; StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.: Treasure Island (FL), 2025. [Google Scholar]
- Fuentes, S.; Chowdhury, Y. S. Fraction of Inspired Oxygen. In Statpearls; StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.: Treasure Island (FL), 2025. [Google Scholar]
- Mas, A.; Masip, J. Noninvasive Ventilation in Acute Respiratory Failure. Int J Chron Obstruct Pulmon Dis 2014, 9, 837–52. [Google Scholar]
- Oczkowski, S.; Ergan, B.; Bos, L.; Chatwin, M.; Ferrer, M.; Gregoretti, C.; Heunks, L.; Frat, J.P.; Longhini, F.; Nava, S.; Navalesi, P.; Ozsancak Uğurlu, A.; Pisani, L.; Renda, T.; Thille, A.W.; Winck, J.C.; Windisch, W.; Tonia, T.; Boyd, J.; Sotgiu, G.; Scala, R. Ers Clinical Practice Guidelines: High-Flow Nasal Cannula in Acute Respiratory Failure. Eur Respir J 2022, 59(no. 4). [Google Scholar] [CrossRef] [PubMed]
- Kallet, Richard H; Diaz, Janet V. The Physiologic Effects of Noninvasive Ventilation. Respiratory Care 2009, 54(no. 1), 102–15. [Google Scholar] [CrossRef]
- Moore, D.P.; Weston, A.R.; Hughes, J.M.; Oakley, C.M.; Cleland, J. G. Effects of Increased Inspired Oxygen Concentrations on Exercise Performance in Chronic Heart Failure. Lancet 1992, 339(no. 8797), 850–3. [Google Scholar] [CrossRef]
- Haque, W.A.; Boehmer, J.; Clemson, B.S.; Leuenberger, U.A.; Silber, D.H.; Sinoway, L. I. Hemodynamic Effects of Supplemental Oxygen Administration in Congestive Heart Failure. J Am Coll Cardiol 1996, 27(no. 2), 353–7. [Google Scholar] [CrossRef]
- Daly, W.J.; Bondurant, S. Effects of Oxygen Breathing on the Heart Rate, Blood Pressure, and Cardiac Index of Normal Men--Resting, with Reactive Hyperemia, and after Atropine. J Clin Invest 1962, 41(no. 1), 126–32. [Google Scholar] [CrossRef] [PubMed]
- Barazzone-Argiroffo, C.; Muzzin, P.; Donati, Y.R.; Kan, C.D.; Aubert, M.L.; Piguet, P. F. Hyperoxia Increases Leptin Production: A Mechanism Mediated through Endogenous Elevation of Corticosterone. Am J Physiol Lung Cell Mol Physiol 2001, 281(no. 5), L1150–6. [Google Scholar] [CrossRef] [PubMed]
- Jaffal, K.; Six, S.; Zerimech, F.; Nseir, S. Relationship between Hyperoxemia and Ventilator Associated Pneumonia. Ann Transl Med 2017, 5(no. 22), 453. [Google Scholar] [CrossRef] [PubMed]
- Ray, Komal; Bodenham, Andrew; Paramasivam, Elankumaran. Pulmonary Atelectasis in Anaesthesia and Critical Care. Continuing Education in Anaesthesia Critical Care & Pain 2014, 14(no. 5), 236–45. [Google Scholar]
- Dunham-Snary, K.J.; Wu, D.; Sykes, E.A.; Thakrar, A.; Parlow, L.R.G.; Mewburn, J.D.; Parlow, J.L.; Archer, S. L. Hypoxic Pulmonary Vasoconstriction: From Molecular Mechanisms to Medicine. Chest 2017, 151(no. 1), 181–92. [Google Scholar] [CrossRef]
- Verhoeff, K.; Mitchell, J. R. Cardiopulmonary Physiology: Why the Heart and Lungs Are Inextricably Linked. Adv Physiol Educ 2017, 41(no. 3), 348–53. [Google Scholar] [CrossRef]
- Delle Femine, F.C.; D’Arienzo, D.; Liccardo, B.; Pastore, M.C.; Ilardi, F.; Mandoli, G.E.; Sperlongano, S.; Malagoli, A.; Lisi, M.; Benfari, G.; Russo, V.; Cameli, M.; D’Andrea, A. Echocardiography in the Ventilated Patient: What the Clinician Has to Know. J Clin Med 2024, 14(no. 1). [Google Scholar] [CrossRef]
- Shekerdemian, L.; Bohn, D. Cardiovascular Effects of Mechanical Ventilation. Arch Dis Child 1999, 80(no. 5), 475–80. [Google Scholar] [CrossRef]
- Berger, D.; Takala, J. Determinants of Systemic Venous Return and the Impact of Positive Pressure Ventilation. Ann Transl Med 2018, 6(no. 18), 350. [Google Scholar] [CrossRef]
- Lansdorp, B.; Hofhuizen, C.; van Lavieren, M.; van Swieten, H.; Lemson, J.; van Putten, M.J.; van der Hoeven, J.G.; Pickkers, P. Mechanical Ventilation-Induced Intrathoracic Pressure Distribution and Heart-Lung Interactions*. Crit Care Med 2014, 42(no. 9), 1983–90. [Google Scholar] [CrossRef]
- Pitoulis, F.G.; Hasan, W.; Papadaki, M.; Clavere, N.G.; Perbellini, F.; Harding, S.E.; Kirk, J.A.; Boateng, S.Y.; de Tombe, P.P.; Terracciano, C. M. Intact Myocardial Preparations Reveal Intrinsic Transmural Heterogeneity in Cardiac Mechanics. J Mol Cell Cardiol 2020, 141, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T. Clinical Assessment of Ventricular Wall Stress in Understanding Compensatory Hypertrophic Response and Maladaptive Ventricular Remodeling. J Cardiovasc Dev Dis 2021, 8(no. 10). [Google Scholar] [CrossRef]
- McCain, M.L.; Parker, K. K. Mechanotransduction: The Role of Mechanical Stress, Myocyte Shape, and Cytoskeletal Architecture on Cardiac Function. Pflugers Arch 2011, 462(no. 1), 89–104. [Google Scholar] [CrossRef]
- Reed, A.; Kohl, P.; Peyronnet, R. Molecular Candidates for Cardiac Stretch-Activated Ion Channels. Glob Cardiol Sci Pract 2014, 9–25. [Google Scholar] [CrossRef]
- Tan, P.M.; Buchholz, K.S.; Omens, J.H.; McCulloch, A.D.; Saucerman, J. J. Predictive Model Identifies Key Network Regulators of Cardiomyocyte Mechano-Signaling. PLoS Comput Biol 2017, 13(no. 11), e1005854. [Google Scholar] [CrossRef] [PubMed]
- Sayed, D.; He, M.; Yang, Z.; Lin, L.; Abdellatif, M. Transcriptional Regulation Patterns Revealed by High Resolution Chromatin Immunoprecipitation during Cardiac Hypertrophy. J Biol Chem 2013, 288(no. 4), 2546–58. [Google Scholar] [CrossRef]
- Rysa, J.; Tokola, H.; Ruskoaho, H. Mechanical Stretch Induced Transcriptomic Profiles in Cardiac Myocytes. Sci Rep 2018, 8(no. 1), 4733. [Google Scholar] [CrossRef]
- Valdez-Jasso, D.; Simon, M.A.; Champion, H.C.; Sacks, M. S. A Murine Experimental Model for the Mechanical Behaviour of Viable Right-Ventricular Myocardium. J Physiol 2012, 590(no. 18), 4571–84. [Google Scholar] [CrossRef]
- Widrich, J.; Shetty, M. Physiology, Pulmonary Vascular Resistance. In Statpearls; StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.: Treasure Island (FL), 2025. [Google Scholar]
- Grübler, M.R.; Wigger, O.; Berger, D.; Blöchlinger, S. Basic Concepts of Heart-Lung Interactions during Mechanical Ventilation. Swiss Med Wkly 2017, 147, w14491. [Google Scholar] [CrossRef]
- Pan, P.; Li, L.; Xie, F.; Hu, X.; Guo, Y.; Xie, L.; Su, L. Physiological Regulation of Pulmonary Microcirculation under Mechanical Ventilation at Different Cardiac Outputs and Positive End-Expiratory Pressures in a Porcine Model. J Pers Med 2023, 13(no. 1). [Google Scholar] [CrossRef] [PubMed]
- Annone, U.; Bocchino, P.P.; Marra, W.G.; D’Ascenzo, F.; Magnino, C.; Montefusco, A.; Omedè, P.; Veglio, F.; Milan, A.; de Ferrari, G. M. Echocardiographic Estimation of Right Ventricular Wall Tension: Haemodynamic Comparison and Long-Term Follow-Up. Pulm Circ 2019, 9(no. 4), 2045894019895420. [Google Scholar] [CrossRef]
- Kret, M.; Arora, R. Pathophysiological Basis of Right Ventricular Remodeling. J Cardiovasc Pharmacol Ther 2007, 12(no. 1), 5–14. [Google Scholar] [CrossRef]
- Simon, M.A.; Pinsky, M. R. Right Ventricular Dysfunction and Failure in Chronic Pressure Overload. Cardiol Res Pract 2011, 2011, 568095. [Google Scholar] [CrossRef]
- Naeije, R.; Badagliacca, R. The Overloaded Right Heart and Ventricular Interdependence. Cardiovasc Res 2017, 113(no. 12), 1474–85. [Google Scholar] [CrossRef] [PubMed]
- Petit, M.; Vieillard-Baron, A. Ventricular Interdependence in Critically Ill Patients: From Physiology to Bedside. Front Physiol 2023, 14, 1232340. [Google Scholar] [CrossRef] [PubMed]
- Burkett, D.A.; Slorach, C.; Patel, S.S.; Redington, A.N.; Ivy, D.D.; Mertens, L.; Younoszai, A.K.; Friedberg, M. K. Impact of Pulmonary Hemodynamics and Ventricular Interdependence on Left Ventricular Diastolic Function in Children with Pulmonary Hypertension. Circ Cardiovasc Imaging 2016, 9(no. 9). [Google Scholar] [CrossRef]
- Grossman, W.; Paulus, W. J. Myocardial Stress and Hypertrophy: A Complex Interface between Biophysics and Cardiac Remodeling. J Clin Invest 2013, 123(no. 9), 3701–3. [Google Scholar] [CrossRef]
- Selldén, H.; Sjövall, H.; Wallin, B.G.; Häggendal, J.; Ricksten, S. E. Changes in Muscle Sympathetic Nerve Activity, Venous Plasma Catecholamines, and Calf Vascular Resistance during Mechanical Ventilation with Peep in Humans. Anesthesiology 1989, 70(no. 2), 243–50. [Google Scholar] [CrossRef] [PubMed]
- Osadchii, O.E. Cardiac Hypertrophy Induced by Sustained Beta-Adrenoreceptor Activation: Pathophysiological Aspects. Heart Fail Rev 2007, 12(no. 1), 66–86. [Google Scholar] [CrossRef]
- Paradis, P.; Dali-Youcef, N.; Paradis, F.W.; Thibault, G.; Nemer, M. Overexpression of Angiotensin Ii Type I Receptor in Cardiomyocytes Induces Cardiac Hypertrophy and Remodeling. Proc Natl Acad Sci U S A 2000, 97(no. 2), 931–6. [Google Scholar] [CrossRef]
- Brilla, C.G. Aldosterone and Myocardial Fibrosis in Heart Failure. Herz 2000, 25(no. 3), 299–306. [Google Scholar] [CrossRef]
- Jia, G.; Aroor, A.R.; Hill, M.A.; Sowers, J. R. Role of Renin-Angiotensin-Aldosterone System Activation in Promoting Cardiovascular Fibrosis and Stiffness. Hypertension 2018, 72(no. 3), 537–48. [Google Scholar] [CrossRef] [PubMed]
- Sorajja, P.; Borlaug, B.A.; Dimas, V.V.; Fang, J.C.; Forfia, P.R.; Givertz, M.M.; Kapur, N.K.; Kern, M.J.; Naidu, S. S. Scai/Hfsa Clinical Expert Consensus Document on the Use of Invasive Hemodynamics for the Diagnosis and Management of Cardiovascular Disease. Catheter Cardiovasc Interv 2017, 89(no. 7), E233–e47. [Google Scholar]
- Xingzheng, L.; Weiguang, G.; Quanqiu, Y.; Huifen, Z.; Zijun, Z.; Qiming, Z.; Suhua, Y.; Fu, Z.; Zhigang, J. The Impact of Positive End-Expiratory Pressure on Right Ventricular Function in Patients with Moderate-to-Severe Ards: A Prospective Paired-Design Study. Front Med (Lausanne) 2024, 11, 1424090. [Google Scholar] [CrossRef]
- Katira, B.H.; Giesinger, R.E.; Engelberts, D.; Zabini, D.; Kornecki, A.; Otulakowski, G.; Yoshida, T.; Kuebler, W.M.; McNamara, P.J.; Connelly, K.A.; Kavanagh, B. P. Adverse Heart-Lung Interactions in Ventilator-Induced Lung Injury. Am J Respir Crit Care Med 2017, 196(no. 11), 1411–21. [Google Scholar] [CrossRef]
- dos Santos, C.C.; Slutsky, A. S. The Contribution of Biophysical Lung Injury to the Development of Biotrauma. Annu Rev Physiol 2006, 68, 585–618. [Google Scholar] [CrossRef]
- Liu, J.; Han, D.; Wang, T.; Liu, P.; Zhao, Y.; Zuo, X. [Role of Inflammation and Apoptosis in Right Ventricular Dysfunction Induced by Injurious Mechanical Ventilation in Rats]. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2022, 34(no. 5), 519–24. [Google Scholar] [PubMed]
- Gordon, J.W.; Shaw, J.A.; Kirshenbaum, L. A. Multiple Facets of Nf-Κb in the Heart: To Be or Not to Nf-Κb. Circ Res 2011, 108(no. 9), 1122–32. [Google Scholar] [CrossRef] [PubMed]
- Boluyt, M.O.; O’Neill, L.; Meredith, A.L.; Bing, O.H.; Brooks, W.W.; Conrad, C.H.; Crow, M.T.; Lakatta, E. G. Alterations in Cardiac Gene Expression during the Transition from Stable Hypertrophy to Heart Failure. Marked Upregulation of Genes Encoding Extracellular Matrix Components. Circ Res 1994, 75(no. 1), 23–32. [Google Scholar] [CrossRef]
- Tadevosyan, A.; Xiao, J.; Surinkaew, S.; Naud, P.; Merlen, C.; Harada, M.; Qi, X.; Chatenet, D.; Fournier, A.; Allen, B.G.; Nattel, S. Intracellular Angiotensin-Ii Interacts with Nuclear Angiotensin Receptors in Cardiac Fibroblasts and Regulates Rna Synthesis, Cell Proliferation, and Collagen Secretion. J Am Heart Assoc 2017, 6(no. 4). [Google Scholar] [CrossRef]
- Díez, J.; González, A.; Kovacic, J. C. Myocardial Interstitial Fibrosis in Nonischemic Heart Disease, Part 3/4: Jacc Focus Seminar. J Am Coll Cardiol 2020, 75(no. 17), 2204–18. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Weber, K. T. Animal Models of Cardiac Fibrosis. Methods Mol Med 2005, 117, 273–90. [Google Scholar] [PubMed]
- Burlew, B.S. Diastolic Dysfunction in the Elderly--the Interstitial Issue. Am J Geriatr Cardiol 2004, 13(no. 1), 29–38. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Shi, M.; Liu, C.; Liu, Q.; Guo, J.; Yu, S.; Jiang, T. Time Course Changes of Oxidative Stress and Inflammation in Hyperoxia-Induced Acute Lung Injury in Rats. Iran J Basic Med Sci 2015, 18(no. 1), 98–103. [Google Scholar]
- Nolfi-Donegan, D.; Braganza, A.; Shiva, S. Mitochondrial Electron Transport Chain: Oxidative Phosphorylation, Oxidant Production, and Methods of Measurement. Redox Biol 2020, 37, 101674. [Google Scholar] [CrossRef]
- Sun, Q.; Karwi, Q.G.; Wong, N.; Lopaschuk, G. D. Advances in Myocardial Energy Metabolism: Metabolic Remodelling in Heart Failure and Beyond. Cardiovasc Res 2024, 120(no. 16), 1996–2016. [Google Scholar] [CrossRef]
- Smith, G.A.; Vandenberg, J.I.; Freestone, N.S.; Dixon, H. B. The Effect of Mg2+ on Cardiac Muscle Function: Is Caatp the Substrate for Priming Myofibril Cross-Bridge Formation and Ca2+ Reuptake by the Sarcoplasmic Reticulum? Biochem J 2001, 354 no. Pt 3, 539–51. [Google Scholar] [CrossRef]
- Pouleur, H. Diastolic Dysfunction and Myocardial Energetics. Eur Heart J 1990, 11 Suppl C, 30–4. [Google Scholar] [CrossRef]
- Nabeebaccus, A.A.; Zoccarato, A.; Hafstad, A.D.; Santos, C.X.; Aasum, E.; Brewer, A.C.; Zhang, M.; Beretta, M.; Yin, X.; West, J.A.; Schröder, K.; Griffin, J.L.; Eykyn, T.R.; Abel, E.D.; Mayr, M.; Shah, A. M. Nox4 Reprograms Cardiac Substrate Metabolism Via Protein O-Glcnacylation to Enhance Stress Adaptation. JCI Insight 2017, 2(no. 24). [Google Scholar] [CrossRef]
- Siani, A.; Tirelli, N. Myofibroblast Differentiation: Main Features, Biomedical Relevance, and the Role of Reactive Oxygen Species. Antioxid Redox Signal 2014, 21(no. 5), 768–85. [Google Scholar] [CrossRef]
- Shaito, A.; Aramouni, K.; Assaf, R.; Parenti, A.; Orekhov, A.; Yazbi, A.E.; Pintus, G.; Eid, A. H. Oxidative Stress-Induced Endothelial Dysfunction in Cardiovascular Diseases. Front Biosci (Landmark Ed) 2022, 27(no. 3), 105. [Google Scholar] [CrossRef]
- Thomas, T.P.; Grisanti, L. A. The Dynamic Interplay between Cardiac Inflammation and Fibrosis. Front Physiol 2020, 11, 529075. [Google Scholar] [CrossRef]
- Wright, S.; Lakin, R. Nature Versus Exposure: Matched Exposure to Circulatory Stressors of Different Natures Elicits Adaptive Remodelling. J Physiol 2015, 593(no. 21), 4691–2. [Google Scholar] [CrossRef]
- Bogaard, H.J.; Abe, K.; Vonk Noordegraaf, A.; Voelkel, N. F. The Right Ventricle under Pressure: Cellular and Molecular Mechanisms of Right-Heart Failure in Pulmonary Hypertension. Chest 2009, 135(no. 3), 794–804. [Google Scholar] [CrossRef]
- Florea, V.G.; Anand, I. S. Troponin T and Plasma Collagen Peptides in Heart Failure. Circ Heart Fail 2012, 5(no. 4), 394–7. [Google Scholar] [CrossRef]
- Asleh, R.; Amir, O.; Kushwaha, S. S. Dynamics of Myocardial Fibrosis after Left Ventricular Assist Device Implantation: Should Speeding up the Scar Have Us Scared Stiff? Eur J Heart Fail 2021, 23(no. 2), 335–38. [Google Scholar] [CrossRef] [PubMed]
- Javan, H.; Szucsik, A.M.; Li, L.; Schaaf, C.L.; Salama, M.E.; Selzman, C. H. Cardiomyocyte P65 Nuclear Factor-Κb Is Necessary for Compensatory Adaptation to Pressure Overload. Circ Heart Fail 2015, 8(no. 1), 109–18. [Google Scholar] [CrossRef] [PubMed]
- Ekbal, N.J.; Dyson, A.; Black, C.; Singer, M. Monitoring Tissue Perfusion, Oxygenation, and Metabolism in Critically Ill Patients. Chest 2013, 143(no. 6), 1799–808. [Google Scholar] [CrossRef] [PubMed]
- Magder, S. Hemodynamic Monitoring in the Mechanically Ventilated Patient. Curr Opin Crit Care 2011, 17(no. 1), 36–42. [Google Scholar] [CrossRef]
- Silva, P.L.; Ball, L.; Rocco, P.R.M.; Pelosi, P. Physiological and Pathophysiological Consequences of Mechanical Ventilation. Semin Respir Crit Care Med 2022, 43(no. 3), 321–34. [Google Scholar] [CrossRef]
- Guyton, A.C.; Lindsey, A.W.; Abernathy, B.; Richardson, T. Venous Return at Various Right Atrial Pressures and the Normal Venous Return Curve. Am J Physiol 1957, 189(no. 3), 609–15. [Google Scholar] [CrossRef] [PubMed]
- Morgan, B.C.; Martin, W.E.; Hornbein, T.F.; Crawford, E.W.; Guntheroth, W. G. Hemodynamic Effects of Intermittent Positive Pressure Respiration. Anesthesiology 1966, 27(no. 5), 584–90. [Google Scholar] [CrossRef] [PubMed]
- LaCombe, P.; Jose, A.; Basit, H.; Lappin, S. L. Physiology, Starling Relationships. In Statpearls; StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.: Treasure Island (FL), 2025. [Google Scholar]
- Vieillard-Baron, A.; Matthay, M.; Teboul, J.L.; Bein, T.; Schultz, M.; Magder, S.; Marini, J. J. Experts’ Opinion on Management of Hemodynamics in Ards Patients: Focus on the Effects of Mechanical Ventilation. Intensive Care Med 2016, 42(no. 5), 739–49. [Google Scholar] [CrossRef]
- Østergaard, L.; Granfeldt, A.; Secher, N.; Tietze, A.; Iversen, N.K.; Jensen, M.S.; Andersen, K.K.; Nagenthiraja, K.; Gutiérrez-Lizardi, P.; Mouridsen, K.; Jespersen, S.N.; Tønnesen, E. K. Microcirculatory Dysfunction and Tissue Oxygenation in Critical Illness. Acta Anaesthesiol Scand 2015, 59(no. 10), 1246–59. [Google Scholar] [CrossRef]
- Jardin, F. The Hemodynamic Consequences of Mechanical Ventilation. Intensive Care Med 1997, 23(no. 10), 1100–1. [Google Scholar]
- Jardin, F.; Delorme, G.; Hardy, A.; Auvert, B.; Beauchet, A.; Bourdarias, J. P. Reevaluation of Hemodynamic Consequences of Positive Pressure Ventilation: Emphasis on Cyclic Right Ventricular Afterloading by Mechanical Lung Inflation. Anesthesiology 1990, 72(no. 6), 966–70. [Google Scholar] [CrossRef]
- Slobod, D.; Assanangkornchai, N.; Alhazza, M.; Mettasittigorn, P.; Magder, S. Right Ventricular Loading by Lung Inflation during Controlled Mechanical Ventilation. Am J Respir Crit Care Med 2022, 205(no. 11), 1311–19. [Google Scholar] [CrossRef]
- Cappio Borlino, S.; Hagry, J.; Lai, C.; Rocca, E.; Fouqué, G.; Rosalba, D.; Fasan, M.; Shi, R.; Recanatini, A.; Cisterna, I.; Barotti, M.; Pham, T.; Teboul, J.L.; Monnet, X. The Effect of Positive End-Expiratory Pressure on Pulmonary Vascular Resistance Depends on Lung Recruitability in Patients with Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med 2024, 210(no. 7), 900–07. [Google Scholar] [CrossRef]
- Pinsky, M.R. The Right Ventricle: Interaction with the Pulmonary Circulation. Crit Care 2016, 20(no. 1), 266. [Google Scholar] [CrossRef]
- Romand, J.A.; Donald, F.A.; Suter, P. M. Acute Right Ventricular Failure, Pathophysiology and Treatment. Monaldi Arch Chest Dis 1995, 50(no. 2), 129–33. [Google Scholar] [PubMed]
- Mitchell, R.; Jamie. Pulmonary Vascular Resistance and Direct Ventricular Interaction during Mechanical Ventilation in an Oleic Acid Induced Acute Lung Injury Model: A Review. Journal of Allergy & Therapy 2012, 01, no. S4. [Google Scholar]
- Repessé, X.; Charron, C.; Vieillard-Baron, A. Acute Cor Pulmonale in Ards: Rationale for Protecting the Right Ventricle. Chest 2015, 147(no. 1), 259–65. [Google Scholar] [CrossRef]
- Dhainaut, J.F.; Devaux, J.Y.; Monsallier, J.F.; Brunet, F.; Villemant, D.; Huyghebaert, M. F. Mechanisms of Decreased Left Ventricular Preload during Continuous Positive Pressure Ventilation in Ards. Chest 1986, 90(no. 1), 74–80. [Google Scholar] [CrossRef]
- Spathoulas, K.; Tsolaki, V.; Zakynthinos, G.E.; Karelas, D.; Makris, D.; Zakynthinos, E.; Papanikolaou, J. The Role of Left Ventricular Ejection Fraction and Left Ventricular Outflow Tract Velocity-Time Integral in Assessing Cardiovascular Impairment in Septic Shock. J Pers Med 2022, 12(no. 11). [Google Scholar] [CrossRef]
- Morimont, P.; Lambermont, B. Left Ventricular Ejection Fraction Depends on Loading Conditions. Asaio j 2019, 65(no. 6), e64. [Google Scholar] [CrossRef] [PubMed]
- Mureddu, G.F.; Faggiano, A. Utility and Limitations of Ejection Fraction and of Diastolic Dysfunction in Heart Failure Patients. Monaldi Arch Chest Dis 2019, 89(no. 1). [Google Scholar] [CrossRef] [PubMed]
- Wolff, C.B. Normal Cardiac Output, Oxygen Delivery and Oxygen Extraction. Adv Exp Med Biol 2007, 599, 169–82. [Google Scholar]
- Selldén, H.; Sjövall, H.; Ricksten, S. E. Sympathetic Nerve Activity and Central Haemodynamics during Mechanical Ventilation with Positive End-Expiratory Pressure in Rats. Acta Physiol Scand 1986, 127(no. 1), 51–60. [Google Scholar] [CrossRef]
- Dünser, M.W.; Takala, J.; Brunauer, A.; Bakker, J. Re-Thinking Resuscitation: Leaving Blood Pressure Cosmetics Behind and Moving Forward to Permissive Hypotension and a Tissue Perfusion-Based Approach. Crit Care 2013, 17(no. 5), 326. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, E.C.; Taha, A.; Tolba, Y.; Hernandez, G.; Pinsky, M. R. Assessment of Tissue Perfusion Pressure in Patients with Septic Shock: Beyond Mean Arterial Pressure. Crit Care Med 2025, 53(no. 11), e2305–e17. [Google Scholar] [CrossRef]
- O’Mahony, D.S.; Liles, W.C.; Altemeier, W.A.; Dhanireddy, S.; Frevert, C.W.; Liggitt, D.; Martin, T.R.; Matute-Bello, G. Mechanical Ventilation Interacts with Endotoxemia to Induce Extrapulmonary Organ Dysfunction. Crit Care 2006, 10(no. 5), R136. [Google Scholar] [CrossRef]
- Papanikolaou, J.; Makris, D.; Saranteas, T.; Karakitsos, D.; Zintzaras, E.; Karabinis, A.; Kostopanagiotou, G.; Zakynthinos, E. New Insights into Weaning from Mechanical Ventilation: Left Ventricular Diastolic Dysfunction Is a Key Player. Intensive Care Med 2011, 37(no. 12), 1976–85. [Google Scholar] [CrossRef]
- Bode, D.; Wen, Y.; Hegemann, N.; Primessnig, U.; Parwani, A.; Boldt, L.H.; Pieske, B.M.; Heinzel, F.R.; Hohendanner, F. Oxidative Stress and Inflammatory Modulation of Ca(2+) Handling in Metabolic Hfpef-Related Left Atrial Cardiomyopathy. Antioxidants (Basel) 2020, 9(no. 9). [Google Scholar]
- Fayssoil, A.; Mansencal, N.; Bergounioux, J.; Wahbi, K.; Stojkovic, T. Cyclic Change in Right and Left Ventricular Systolic and Diastolic Function in Patients with Neuromuscular Disorders on Permanent Mechanical Ventilation. J Pers Med 2022, 12(no. 10). [Google Scholar] [CrossRef] [PubMed]
- Rahman, Annas; Ruge, Max; Hlepas, Alex; Nair, Gatha; Gomez, Joanne; du Fay de Lavallaz, Jeanne; Fugar, Setri; Jahan, Nusrat; Santos Volgman, Annabelle; Williams, Kim A.; Rao, Anupama; Marinescu, Karolina; Suboc, Tisha. Hyperdynamic Left Ventricular Ejection Fraction Is Associated with Higher Mortality in Covid-19 Patients. American Heart Journal Plus: Cardiology Research and Practice 2022, 14, 100134. [Google Scholar] [CrossRef]
- Wright, S.P.; Dawkins, T.G.; Harper, M.I.; Stembridge, M.; Martin-Spencer, H.; Shave, R.; Eves, N. D. Intrathoracic Pressure Deviations Attenuate Left Ventricular Filling and Stroke Volume without Pronounced Myocardial Mechanical Alterations in Healthy Adults. J Appl Physiol (1985) 2025, 138(no. 3), 681–92. [Google Scholar] [CrossRef]
- Mehdi, S.F.; Qureshi, M.H.; Pervaiz, S.; Kumari, K.; Saji, E.; Shah, M.; Abdullah, A.; Zahoor, K.; Qadeer, H.A.; Katari, D.K.; Metz, C.; Mishra, L.; LeRoith, D.; Tracey, K.; Brownstein, M.J.; Roth, J. Endocrine and Metabolic Alterations in Response to Systemic Inflammation and Sepsis: A Review Article. Mol Med 2025, 31(no. 1), 16. [Google Scholar] [CrossRef]
- Varró, A.; Tomek, J.; Nagy, N.; Virág, L.; Passini, E.; Rodriguez, B.; Baczkó, I. Cardiac Transmembrane Ion Channels and Action Potentials: Cellular Physiology and Arrhythmogenic Behavior. Physiol Rev 2021, 101(no. 3), 1083–176. [Google Scholar] [CrossRef] [PubMed]
- Atia, J.; McCloskey, C.; Shmygol, A.S.; Rand, D.A.; van den Berg, H.A.; Blanks, A. M. Reconstruction of Cell Surface Densities of Ion Pumps, Exchangers, and Channels from Mrna Expression, Conductance Kinetics, Whole-Cell Calcium, and Current-Clamp Voltage Recordings, with an Application to Human Uterine Smooth Muscle Cells. PLoS Comput Biol 2016, 12(no. 4), e1004828. [Google Scholar] [CrossRef]
- Marrus, S.B.; Nerbonne, J. M. Mechanisms Linking Short- and Long-Term Electrical Remodeling in the Heart...Is It a Stretch? Channels (Austin) 2008, 2(no. 2), 117–24. [Google Scholar] [CrossRef]
- Buonocunto, M.; Lyon, A.; Delhaas, T.; Heijman, J.; Lumens, J. Electrophysiological Effects of Stretch-Activated Ion Channels: A Systematic Computational Characterization. J Physiol 2024, 602(no. 18), 4585–604. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Tan, H.; Sun, C.; Li, G. Dysfunctional Nav1.5 Channels Due to Scn5a Mutations. Exp Biol Med (Maywood) 2018, 243(no. 10), 852–63. [Google Scholar] [CrossRef] [PubMed]
- King, J.H.; Huang, C.L.; Fraser, J. A. Determinants of Myocardial Conduction Velocity: Implications for Arrhythmogenesis. Front Physiol 2013, 4, 154. [Google Scholar] [CrossRef]
- Weinberg, S.H. Sodium Channel Subpopulations with Distinct Biophysical Properties and Subcellular Localization Enhance Cardiac Conduction. J Gen Physiol 2023, 155(no. 8). [Google Scholar] [CrossRef]
- Le Dinh, M.; Carreira, S.; Obert, J.; Gayan-Ramirez, G.; Riou, B.; Beuvin, M.; Similowski, T.; Coirault, C.; Demoule, A. Prolonged Mechanical Ventilation Worsens Sepsis-Induced Diaphragmatic Dysfunction in the Rat. PLoS One 2018, 13(no. 8), e0200429. [Google Scholar] [CrossRef]
- Obeagu, E.I. Inflammatory Cytokines and Cardiac Arrhythmias: From Pathogenesis to Potential Therapies. Ann Med Surg (Lond) 2025, 87(no. 9), 5607–13. [Google Scholar] [CrossRef]
- Koesters, A.; Engisch, K.L.; Rich, M. M. Decreased Cardiac Excitability Secondary to Reduction of Sodium Current May Be a Significant Contributor to Reduced Contractility in a Rat Model of Sepsis. Crit Care 2014, 18(no. 2), R54. [Google Scholar] [CrossRef] [PubMed]
- Rivaud, M.R.; Agullo-Pascual, E.; Lin, X.; Leo-Macias, A.; Zhang, M.; Rothenberg, E.; Bezzina, C.R.; Delmar, M.; Remme, C. A. Sodium Channel Remodeling in Subcellular Microdomains of Murine Failing Cardiomyocytes. J Am Heart Assoc 2017, 6(no. 12). [Google Scholar] [CrossRef]
- Buchanan, J.W., Jr.; Saito, T.; Gettes, L. S. The Effects of Antiarrhythmic Drugs, Stimulation Frequency, and Potassium-Induced Resting Membrane Potential Changes on Conduction Velocity and Dv/Dtmax in Guinea Pig Myocardium. Circ Res 1985, 56(no. 5), 696–703. [Google Scholar] [CrossRef]
- Quan, W.; Rudy, Y. Unidirectional Block and Reentry of Cardiac Excitation: A Model Study. Circ Res 1990, 66(no. 2), 367–82. [Google Scholar] [CrossRef]
- Antzelevitch, C. Basic Mechanisms of Reentrant Arrhythmias. Curr Opin Cardiol 2001, 16(no. 1), 1–7. [Google Scholar] [CrossRef]
- Boyle, P.M.; Park, C.J.; Arevalo, H.J.; Vigmond, E.J.; Trayanova, N. A. Sodium Current Reduction Unmasks a Structure-Dependent Substrate for Arrhythmogenesis in the Normal Ventricles. PLoS One 2014, 9(no. 1), e86947. [Google Scholar] [CrossRef]
- Rudy, Y. Molecular Basis of Cardiac Action Potential Repolarization. Ann N Y Acad Sci 2008, 1123, 113–8. [Google Scholar] [CrossRef]
- Nerbonne, J.M.; Kass, R. S. Molecular Physiology of Cardiac Repolarization. Physiol Rev 2005, 85(no. 4), 1205–53. [Google Scholar] [CrossRef]
- Zhu, T.G.; Patel, C.; Martin, S.; Quan, X.; Wu, Y.; Burke, J.F.; Chernick, M.; Kowey, P.R.; Yan, G. X. Ventricular Transmural Repolarization Sequence: Its Relationship with Ventricular Relaxation and Role in Ventricular Diastolic Function. Eur Heart J 2009, 30(no. 3), 372–80. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Zhang, Y.; Gao, H.; Nattel, S.; Wang, Z. Impairment of Herg K(+) Channel Function by Tumor Necrosis Factor-Alpha: Role of Reactive Oxygen Species as a Mediator. J Biol Chem 2004, 279(no. 14), 13289–92. [Google Scholar] [CrossRef]
- Fukuda, K.; Davies, S.S.; Nakajima, T.; Ong, B.H.; Kupershmidt, S.; Fessel, J.; Amarnath, V.; Anderson, M.E.; Boyden, P.A.; Viswanathan, P.C.; Roberts, L.J., 2nd; Balser, J. R. Oxidative Mediated Lipid Peroxidation Recapitulates Proarrhythmic Effects on Cardiac Sodium Channels. Circ Res 2005, 97(no. 12), 1262–9. [Google Scholar] [CrossRef] [PubMed]
- Beuckelmann, D.J.; Näbauer, M.; Erdmann, E. Alterations of K+ Currents in Isolated Human Ventricular Myocytes from Patients with Terminal Heart Failure. Circ Res 1993, 73(no. 2), 379–85. [Google Scholar] [CrossRef] [PubMed]
- Ravens, U.; Cerbai, E. Role of Potassium Currents in Cardiac Arrhythmias. Europace 2008, 10(no. 10), 1133–7. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.L.; Hou, C.J.; Lauer, M.R.; Sung, R. J. Electrophysiologic Mechanisms of the Long Qt Interval Syndromes and Torsade De Pointes. Ann Intern Med 1995, 122(no. 9), 701–14. [Google Scholar] [CrossRef]
- Arteyeva, N.V. Dispersion of Ventricular Repolarization: Temporal and Spatial. World J Cardiol 2020, 12(no. 9), 437–49. [Google Scholar] [CrossRef] [PubMed]
- Kléber, A.G.; Rudy, Y. Basic Mechanisms of Cardiac Impulse Propagation and Associated Arrhythmias. Physiol Rev 2004, 84(no. 2), 431–88. [Google Scholar] [CrossRef]
- Qu, Z.; Xie, L.H.; Olcese, R.; Karagueuzian, H.S.; Chen, P.S.; Garfinkel, A.; Weiss, J. N. Early Afterdepolarizations in Cardiac Myocytes: Beyond Reduced Repolarization Reserve. Cardiovasc Res 2013, 99(no. 1), 6–15. [Google Scholar] [CrossRef] [PubMed]
- Timmermann, V.; Dejgaard, L.A.; Haugaa, K.H.; Edwards, A.G.; Sundnes, J.; McCulloch, A.D.; Wall, S. T. An Integrative Appraisal of Mechano-Electric Feedback Mechanisms in the Heart. Prog Biophys Mol Biol 2017, 130, no. Pt B, 404–17. [Google Scholar] [CrossRef]
- Landstrom, A.P.; Dobrev, D.; Wehrens, X. H. T. Calcium Signaling and Cardiac Arrhythmias. Circ Res 2017, 120(no. 12), 1969–93. [Google Scholar] [CrossRef]
- Eisner, D.A.; Caldwell, J.L.; Kistamás, K.; Trafford, A. W. Calcium and Excitation-Contraction Coupling in the Heart. Circ Res 2017, 121(no. 2), 181–95. [Google Scholar] [CrossRef]
- Primeau, J.O.; Armanious, G.P.; Fisher, M.E.; Young, H. S. The Sarcoendoplasmic Reticulum Calcium Atpase. Subcell Biochem 2018, 87, 229–58. [Google Scholar]
- Qin, F.; Siwik, D.A.; Lancel, S.; Zhang, J.; Kuster, G.M.; Luptak, I.; Wang, L.; Tong, X.; Kang, Y.J.; Cohen, R.A.; Colucci, W. S. Hydrogen Peroxide-Mediated Serca Cysteine 674 Oxidation Contributes to Impaired Cardiac Myocyte Relaxation in Senescent Mouse Heart. J Am Heart Assoc 2013, 2(no. 4), e000184. [Google Scholar] [CrossRef]
- Roshan, N.; Pandit, R. Abnormal Calcium Release and Delayed Afterdepolarizations: A Comparison of Two Mathematical Models for Human Ventricular Myocytes. PLoS One 2026, 21(no. 2), e0338873. [Google Scholar] [CrossRef] [PubMed]
- Spencer, C.I.; Sham, J. S. Effects of Na+/Ca2+ Exchange Induced by Sr Ca2+ Release on Action Potentials and Afterdepolarizations in Guinea Pig Ventricular Myocytes. Am J Physiol Heart Circ Physiol 2003, 285(no. 6), H2552–62. [Google Scholar] [CrossRef]
- Xie, L.H.; Chen, F.; Karagueuzian, H.S.; Weiss, J. N. Oxidative-Stress-Induced Afterdepolarizations and Calmodulin Kinase Ii Signaling. Circ Res 2009, 104(no. 1), 79–86. [Google Scholar] [CrossRef]
- Blayney, L.M.; Lai, F. A. Ryanodine Receptor-Mediated Arrhythmias and Sudden Cardiac Death. Pharmacol Ther 2009, 123(no. 2), 151–77. [Google Scholar] [CrossRef]
- Kohl, P.; Day, K.; Noble, D. Cellular Mechanisms of Cardiac Mechano-Electric Feedback in a Mathematical Model. Can J Cardiol 1998, 14(no. 1), 111–9. [Google Scholar]
- Lim, S.; Mangala, M.M.; Holliday, M.; Szappanos, H. Cserne; Barratt-Ross, S.; Li, S.; Thorpe, J.; Liang, W.; Ranpura, G.N.; Vandenberg, J.I.; Semsarian, C.; Hill, A.P.; Hool, L. C. Reduced Connexin-43 Expression, Slow Conduction and Repolarisation Dispersion in a Model of Hypertrophic Cardiomyopathy. Dis Model Mech 2024, 17(no. 8). [Google Scholar] [CrossRef]
- Kohl, P.; Sachs, F. Mechanoelectric Feedback in Cardiac Cells. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 2001, 359(no. 1783), 1173–85. [Google Scholar] [CrossRef]
- Quinn, T.A.; Kohl, P. Cardiac Mechano-Electric Coupling: Acute Effects of Mechanical Stimulation on Heart Rate and Rhythm. Physiol Rev 2021, 101(no. 1), 37–92. [Google Scholar] [CrossRef] [PubMed]
- Nagatomo, T.; Fan, Z.; Ye, B.; Tonkovich, G.S.; January, C.T.; Kyle, J.W.; Makielski, J. C. Temperature Dependence of Early and Late Currents in Human Cardiac Wild-Type and Long Q-T Deltakpq Na+ Channels. Am J Physiol 1998, 275(no. 6), H2016–24. [Google Scholar] [PubMed]
- Wu, Y.; Wang, Q.; Granger, J.; Gaido, O.R.; Aguilar, E.N.; Ludwig, A.; Moroni, A.; Bianchet, M.A.; Anderson, M. E. Hcn Channels Sense Temperature and Determine Heart Rate Responses to Heat. bioRxiv 2023. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, T.; Guo, J.; Yang, T.; Li, W.; Koichopolos, J.; Lamothe, S.M.; Kang, Y.; Ma, A.; Zhang, S. Febrile Temperature Facilitates Herg/Ikr Degradation through an Altered K(+) Dependence. Heart Rhythm 2016, 13(no. 10), 2004–11. [Google Scholar] [CrossRef]
- Cluitmans, M.J.M.; Bayer, J.; Bear, L.R.; Ter Bekke, R.M.A.; Heijman, J.; Coronel, R.; Volders, P. G. A. The Circle of Reentry: Characteristics of Trigger-Substrate Interaction Leading to Sudden Cardiac Arrest. Front Cardiovasc Med 2023, 10, 1121517. [Google Scholar] [CrossRef]
- Remme, C.A.; Bezzina, C. R. Sodium Channel (Dys)Function and Cardiac Arrhythmias. Cardiovasc Ther 2010, 28(no. 5), 287–94. [Google Scholar] [CrossRef]
- Nguyen, M.N.; Kiriazis, H.; Gao, X.M.; Du, X. J. Cardiac Fibrosis and Arrhythmogenesis. Compr Physiol 2017, 7(no. 3), 1009–49. [Google Scholar] [CrossRef]
- Lei, M.; Salvage, S.C.; Jackson, A.P.; Huang, C. L. Cardiac Arrhythmogenesis: Roles of Ion Channels and Their Functional Modification. Front Physiol 2024, 15, 1342761. [Google Scholar] [CrossRef]
- Arango-Ibanez, J.P.; Cordoba-Melo, B.D.; Barbosa Rengifo, M.M.; Tobar-Arteaga, J.D.; Castro-Trujillo, M.L.; Herrera, C.J.; Quintana Da Silva, M.A.; Buitrago Sandoval, A.F.; Coronel Gilio, M.L.; Chon Long, F.P.; Cardenas Aldaz, L.; Valencia, A.; Vesga-Reyes, C.E.; Gomez-Mesa, J. E. Two-Year Follow-up of Patients with Myocardial Injury during Acute Covid-19: Insights from the Cardio Covid 20-21 Registry. Front Cardiovasc Med 2025, 12, 1584732. [Google Scholar] [CrossRef] [PubMed]
- Cardenas-Marin, P.A.; Cordoba-Melo, B.D.; Carrillo-Gomez, D.C.; Leon-Giraldo, H.; Mendoza, I.; Florez, N.; Larrea Gomez, R.E.; Mercedes, J.M.; Herrera, C.J.; Lugo-Pena, J.; Cardenas-Aldaz, L.P.; Rossel, V.; Ramirez, R. Ramirez; Fernandez, H.F.; Retana, A.U.; Sierra-Lara Martinez, J.D.; Figueiredo, E.L.; Yabar Galindo, W.G.; Quintana Da Silva, M.A.; Romero, A.; Silva, P.; Alvarado, A.; Valencia, A.; Gomez-Mesa, J. E. Impact of Myocardial Injury on Cardiovascular Complications in Hospitalized Patients with Covid-19: Insights from Latin America. Front Cardiovasc Med 2025, 12, 1545142. [Google Scholar] [CrossRef]
- Jozwiak, M.; Dupuis, C.; Denormandie, P.; Aurenche Mateu, D.; Louchet, J.; Heme, N.; Mira, J.P.; Doyen, D.; Dellamonica, J. Right Ventricular Injury in Critically Ill Patients with Covid-19: A Descriptive Study with Standardized Echocardiographic Follow-Up. Ann Intensive Care 2024, 14(no. 1), 14. [Google Scholar] [CrossRef] [PubMed]
- Bonnemain, J.; Ltaief, Z.; Liaudet, L. The Right Ventricle in Covid-19. J Clin Med 2021, 10(no. 12). [Google Scholar] [CrossRef]
- D’Alto, M.; Marra, A.M.; Severino, S.; Salzano, A.; Romeo, E.; De Rosa, R.; Stagnaro, F.M.; Pagnano, G.; Verde, R.; Murino, P.; Farro, A.; Ciccarelli, G.; Vargas, M.; Fiorentino, G.; Servillo, G.; Gentile, I.; Corcione, A.; Cittadini, A.; Naeije, R.; Golino, P. Right Ventricular-Arterial Uncoupling Independently Predicts Survival in Covid-19 Ards. Crit Care 2020, 24(no. 1), 670. [Google Scholar] [CrossRef] [PubMed]
- Schjorring, O.L.; Klitgaard, T.L.; Perner, A.; Wetterslev, J.; Lange, T.; Siegemund, M.; Backlund, M.; Keus, F.; Laake, J.H.; Morgan, M.; Thormar, K.M.; Rosborg, S.A.; Bisgaard, J.; Erntgaard, A.E.S.; Lynnerup, A.H.; Pedersen, R.L.; Crescioli, E.; Gielstrup, T.C.; Behzadi, M.T.; Poulsen, L.M.; Estrup, S.; Laigaard, J.P.; Andersen, C.; Mortensen, C.B.; Brand, B.A.; White, J.; Jarnvig, I.L.; Moller, M.H.; Quist, L.; Bestle, M.H.; Schonemann-Lund, M.; Kamper, M.K.; Hindborg, M.; Hollinger, A.; Gebhard, C.E.; Zellweger, N.; Meyhoff, C.S.; Hjort, M.; Bech, L.K.; Grofte, T.; Bundgaard, H.; Ostergaard, L.H.M.; Thyo, M.A.; Hildebrandt, T.; Uslu, B.; Solling, C.G.; Moller-Nielsen, N.; Brochner, A.C.; Borup, M.; Okkonen, M.; Dieperink, W.; Pedersen, U.G.; Andreasen, A.S.; Buus, L.; Aslam, T.N.; Winding, R.R.; Schefold, J.C.; Thorup, S.B.; Iversen, S.A.; Engstrom, J.; Kjaer, M.N.; Rasmussen, B.S.; Hot-Icu Investigators. Lower or Higher Oxygenation Targets for Acute Hypoxemic Respiratory Failure. N Engl J Med 2021, 384(no. 14), 1301–11. [Google Scholar] [CrossRef]
| Category | Driver | Lung Status | ROS/Stress | Cardiac effect | Long-term risk |
|---|---|---|---|---|---|
| Normoxia (baseline) | 21% O2/ATM pressure | Stable Alveoli (N₂ scaffold intact) | Balanced /antioxidants sufficient (glutathione, superoxide dismutase) | Normal hemodynamics | Physiological baseline |
| Supplemental 02 (controlled) | FiO2 21-60% | Blunted HPV reflex → V/Q mismatch ↑ Lung lavage protein (early injury) ↓ N₂ → early alveolar instability ≥7 days at 50–60%: lung “primed” for toxicity ↑ Lung weights in inflammatory models (pulmonary edema) |
Antioxidants sufficient at low FiO₂, increasingly stressed approaching 60% | Vagus-mediated ↓ HR → ↓ cardiac index, ↓ Stroke volume, ↑ Pulmonary capillary wedge pressure, Hemodynamic strain in heart failure patients |
O₂ toxicity, V/Q mismatch, hemodynamic compromise |
| Mechanical Ventilation (Pressure) | Positive pressure/PEEP | N₂ washout → absorption atelectasis, Alveolar overdistension → capillary compression, VILI → biotrauma (TNF-α, IL-6 released systemically) |
Mechanical strain, Neurohormonal activation: sympathetic ↑, RAAS (angiotensin II, aldosterone), Catecholamine-driven ↑ Ca²⁺ cycling |
↓ Venous return / preload, ↑ RV afterload (↑ PVR), Septal shift → ↓ LV filling, Diastolic dysfunction (stiff ventricle → output-limited), EF preserved but stroke volume low — EF misleading in MV, Angiotensin II → hypertrophic gene expression, Aldosterone → collagen synthesis |
RV dilation, Interstitial fibrosis (ECM expansion, reversal limited once established), Electrical instability, Progressive structural remodeling |
| Hyperoxia (Pathological) | FiO2 > 90% | Lipid peroxidation → Type I & II alveolar cell destruction, Surfactant dysfunction → ↑ surface tension, ↓ compliance, HALI → ARDS-like injury → pulmonary fibrosis, Mortality in animal models at 72–96 hrs |
Massive ROS, ↓ Nitric oxide → endothelial dysfunction, NF-κB → myofibroblast differentiation, Mitochondrial dysfunction → ↓ ATP, SERCA↓ + RyR leak → Ca²⁺ dysregulation |
↓ Cardiac output & ejection fraction, Bradycardia, ↑ QTc & JT intervals, Nav1.5↓ → slowed conduction → reentry, K⁺ channel remodeling → repolarization dispersion, Ca²⁺ dysregulation → delayed afterdepolarizations, Septal shift / RV failure, ROS → myofibroblast activation |
Interstitial fibrosis, Arrhythmogenesis, Reversal increasingly limited once fibrosis established, AKI / multi-organ dysfunction (lung–heart–kidney axis), COVID-19: 31% de novo RV dysfunction post-intubation, MV = largest independent mortality risk factor (beyond age >80) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




