Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Overview of Cardiac Remodeling
2.1. Pathological Cardiac Remodeling
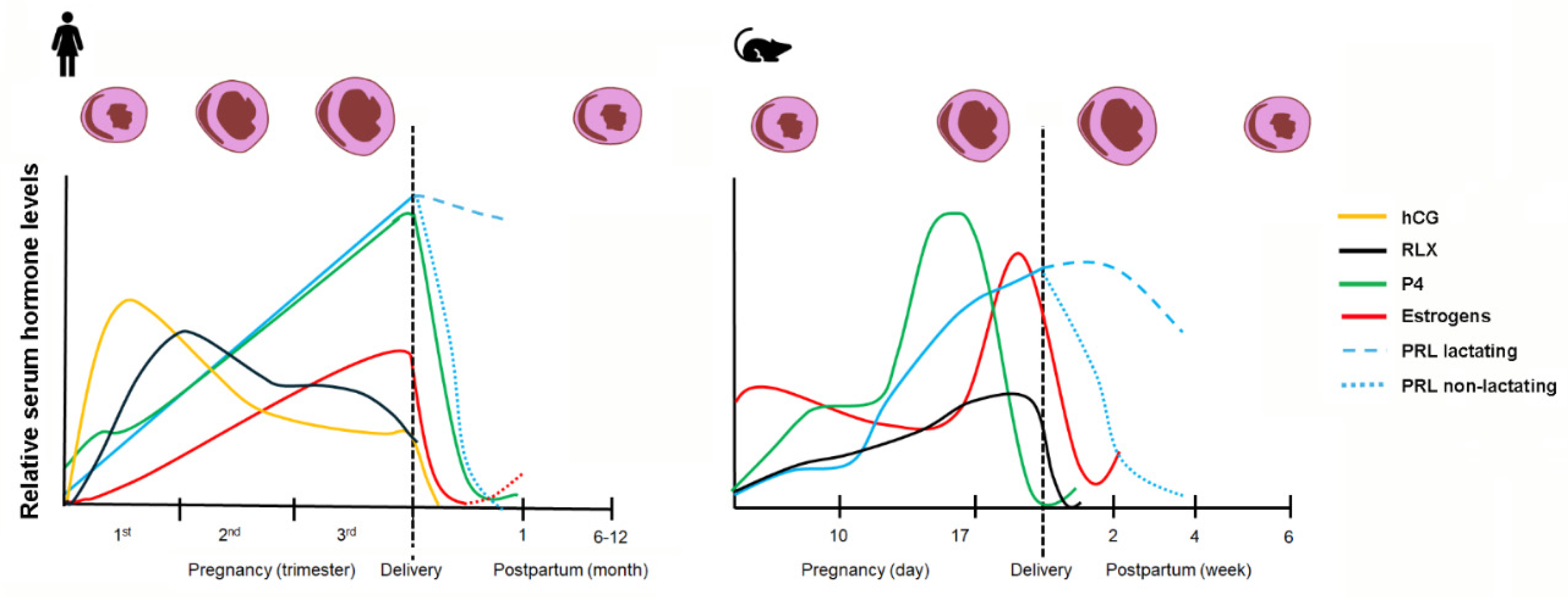
2.2. Physiological Cardiac Remodeling–Non-fibrotic Cardiac Adaptation During Pregnancy and Postpartum
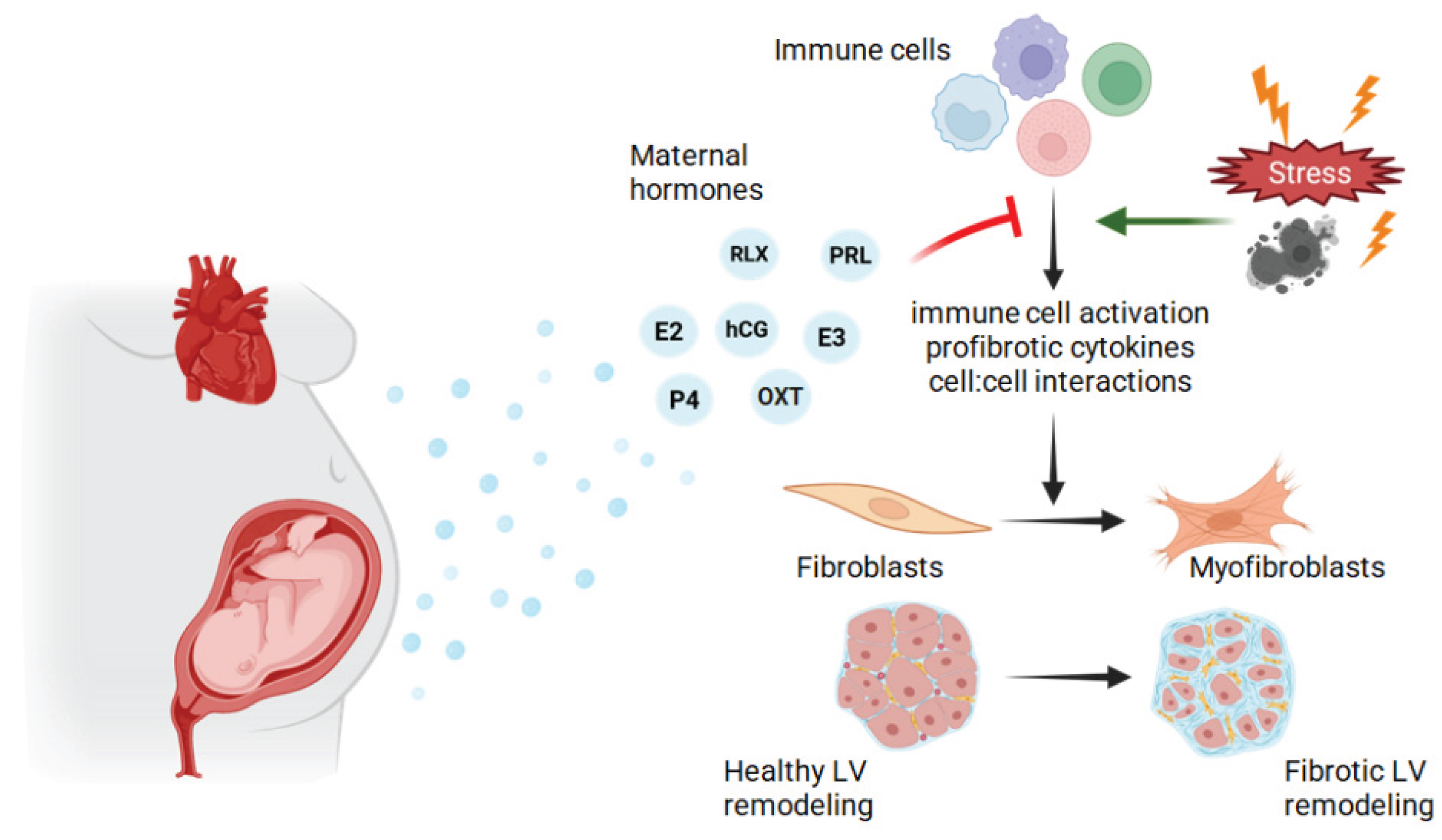
3. Etiology of Fibrosis and the Role of Fibroblasts
Cardiac Fibrosis
4. Cell-Cell Communication in Cardiac Remodeling & Immune Cells
4.1. Monocytes/Macrophages
4.2. Neutrophils
4.3. T Lymphocytes
4.4. B Cells
4.5. Mast Cells
5. Hormones in Cardiac Remodeling During Pregnancy and the Peripartum Period
5.1. Estrogen
5.2. Progesterone
5.3. Relaxin
5.4. Prolactin & Placental Lactogens
5.5. Human Chorionic Gonadotropin
5.6. Oxytocin
6. Future Directions
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADAM | a disintegrin and metalloproteinase |
| ADRB1 | beta-1 adrenergic receptor |
| ADRB2 | beta-2 adrenergic receptor |
| AngII | angiotensin II |
| CAV1 | caveolin-1 |
| CF | cardiac fibroblasts |
| CK | creatine kinase |
| cTMs | cardiac tissue macrophages |
| E1 | estrone |
| E2 | 17-beta estradiol |
| E3 | estriol |
| ECM | extracellular matrix |
| EDV | end-diastolic volume |
| EF | ejection fraction |
| eNOS | endothelial nitric oxide synthase |
| ERα | estrogen receptor alpha |
| ERβ | estrogen receptor beta |
| ESR1 | estrogen receptor 1 |
| ESR2 | estrogen receptor 2 |
| ESV | end-systolic volume |
| GPER | G protein-coupled estrogen receptor |
| hCG | human chorionic gonadotropin |
| HFpEF | heart failure with preserved ejection fraction |
| HFrEF | heart failure with reduced ejection fraction |
| hPL | human placental lactogen |
| LDH | lactate dehydrogenase |
| LH | luteinizing hormone |
| LV | left ventricle |
| MCs | mast cells |
| MCT | mast cell subtype with tryptase-only granules |
| MHCII | major histocompatibility complex class II |
| MI | myocardial infarction |
| miRNAs | microRNAs |
| MMP | matrix metalloproteinase |
| MMP-2 | matrix metalloproteinase 2 |
| MMP-9 | matrix metalloproteinase 9 |
| mPR | membrane progesterone receptor |
| mRNA | messenger RNA |
| NF-κB | nuclear factor kappa B |
| nPR-A | nuclear progesterone receptor A |
| nPR-B | nuclear progesterone receptor B |
| OXTRs | oxytocin receptors |
| P4 | progesterone |
| PDGF | platelet-derived growth factor |
| PPCM | peripartum cardiomyopathy |
| PR-M | mitochondrial progesterone receptor |
| PRs | progesterone receptors |
| RAAS | renin-angiotensin-aldosterone system |
| ROS | reactive oxygen species |
| RU486 | mifepristone |
| RXFP | relaxin family peptide receptor |
| scRNA-seq | single-cell RNA sequencing |
| SERCA | sarcoplasmic reticulum calcium ATPase |
| SPP1 | secreted phosphoprotein 1 (osteopontin) |
| TAC | transverse aortic constriction |
| TNF-α | tumor necrosis factor-alpha |
| TnI | troponin I |
| Treg | regulatory T cells |
| VEGF | vascular endothelial growth factor |
| VEGF-A | vascular endothelial growth factor A |
| VMSC(s) | vascular smooth muscle cell(s) |
| α-SMA | alpha-smooth muscle actin |
References
- Azevedo, P.S.; Polegato, B.F.; Minicucci, M.F.; Paiva, S.A.R.; Zornoff, L.A.M. Cardiac remodeling: concepts, clinical impact, pathophysiological mechanisms and pharmacologic treatment. Arq. Bras. Cardiol. 2016, 106, 62–69. [Google Scholar] [CrossRef]
- Gibb, A.A.; Hill, B.G. Metabolic coordination of physiological and pathological cardiac remodeling. Circ. Res. 2018, 123, 107–128. [Google Scholar] [CrossRef]
- Liaquat, M.T.; Makaryus, A.N. Cardiac electrical and structural remodeling. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Frangogiannis, N.G. Cardiac fibrosis. Cardiovasc. Res. 2020, 117, 1450–1488. [Google Scholar] [CrossRef] [PubMed]
- Collins, H.E.; Alexander, B.T.; Care, A.S.; Davenport, M.H.; Davidge, S.T.; Eghbali, M.; Giussani, D.A.; Hoes, M.F.; Julian, C.G.; LaVoie, H.A.; et al. Guidelines for assessing maternal cardiovascular physiology during pregnancy and postpartum. Am. J. Physiol. - Heart Circ. Physiol. 2024, 327, H191–H220. [Google Scholar] [CrossRef]
- Umar, S.; Nadadur, R.; Iorga, A.; Amjedi, M.; Matori, H.; Eghbali, M. Cardiac structural and hemodynamic changes associated with physiological heart hypertrophy of pregnancy are reversed postpartum. J. Appl. Physiol. 2012, 113, 1253–1259. [Google Scholar] [CrossRef]
- Goldsmith, J.G.; Goldsmith, E.C. Current perspectives in peripartum cardiomyopathy. Reproduction 2026, 171, xaag009. [Google Scholar] [CrossRef]
- Fan, D.; Takawale, A.; Lee, J.; Kassiri, Z. Cardiac fibroblasts, fibrosis and extracellular matrix remodeling in heart disease. Fibrogenesis Tissue Repair 2012, 5, 15. [Google Scholar] [CrossRef]
- Savarese, G.; Becher, P.M.; Lund, L.H.; Seferovic, P.; Rosano, G.M.C.; Coats, A.J.S. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc. Res. 2022, 118, 3272–3287. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hill, J.A. Electrophysiological remodeling in heart failure. J. Mol. Cell. Cardiol. 2010, 48, 619–632. [Google Scholar] [CrossRef] [PubMed]
- Shams, P.; Malik, A.; Chhabra, L. Heart failure (congestive heart failure). In StatPearls [Internet]; StatPearls Publishing, 2025. [Google Scholar]
- Bosch, L.; Lam, C.S.P.; Gong, L.; Chan, S.P.; Sim, D.; Yeo, D.; Jaufeerally, F.; Leong, K.T.G.; Ong, H.Y.; Ng, T.P.; et al. Right ventricular dysfunction in left-sided heart failure with preserved versus reduced ejection fraction. Eur. J. Heart Fail. 2017, 19, 1664–1671. [Google Scholar] [CrossRef] [PubMed]
- Hartupee, J.; Mann, D.L. Neurohormonal activation in heart failure with reduced ejection fraction. Nat. Rev. Cardiol. 2017, 14, 30–38. [Google Scholar] [CrossRef]
- Mihl, C.; Dassen, W.R.M.; Kuipers, H. Cardiac remodelling: concentric versus eccentric hypertrophy in strength and endurance athletes. Neth. Heart J. 2008, 16, 129–133. [Google Scholar] [CrossRef]
- Burke, R.M.; Villar, K.N.B.; Small, E.M. Fibroblast contributions to ischemic cardiac remodeling. Cell. Signal. 2021, 77, 109824. [Google Scholar] [CrossRef]
- Paolisso, P.; Gallinoro, E.; Belmonte, M.; Bertolone, D.T.; Bermpeis, K.; De Colle, C.; Shumkova, M.; Leone, A.; Caglioni, S.; Esposito, G.; et al. Coronary microvascular dysfunction in patients with heart failure: characterization of patterns in hfref versus hfpef. Circ. Heart Fail. 2024, 17, e010805. [Google Scholar] [CrossRef]
- Cokkinos, D.V.; Belogianneas, C. Left ventricular remodelling: a problem in search of solutions. Eur. Cardiol. Rev. 2016, 11, 29–35. [Google Scholar] [CrossRef]
- Jee, S.B.; Sawal, A. Physiological changes in pregnant women due to hormonal changes. Cureus 16 e55544. [CrossRef] [PubMed]
- Sanghavi, M.; Rutherford, J.D. Cardiovascular physiology of pregnancy. Circulation 2014, 130, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Alexander, B.T.; South, A.M.; August, P.; Bertagnolli, M.; Ferranti, E.P.; Grobe, J.L.; Jones, E.J.; Loria, A.S.; Safdar, B.; Sequeira-Lopez, M.L.S.; et al. Appraising the preclinical evidence of the role of the renin-angiotensin-aldosterone system in antenatal programming of maternal and offspring cardiovascular health across the life course: moving the field forward: a scientific statement from the american heart association. Hypertension 2023, 80, e75–e89. [Google Scholar] [CrossRef]
- de Haas, S.; Spaanderman, M.E.A.; van Kuijk, S.M.J.; van Drongelen, J.; Mohseni, Z.; Jorissen, L.; Ghossein-Doha, C. Adaptation of left ventricular diastolic function to pregnancy: a systematic review and meta-analysis. J. Hypertens. 2021, 39, 1934–1941. [Google Scholar] [CrossRef]
- Chung, E.; Leinwand, L.A. Pregnancy as a cardiac stress model. Cardiovasc. Res. 2014, 101, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Aljabri, M.B.; Songstad, N.T.; Lund, T.; Serrano, M.C.; Andreasen, T.V.; Al-Saad, S.; Lindal, S.; Sitras, V.; Acharya, G.; Ytrehus, K. Pregnancy protects against antiangiogenic and fibrogenic effects of angiotensin II in rat hearts. Acta Physiol. Oxf. Engl. 2011, 201, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Magaye, R.R.; Wang, B.H.; Liu, H.R.; Bailey, S.; Nuon, R.; Sutano, S.; Nyugen, M.; Kiriazis, H.; Grigolon, K.; Shan, L.; et al. Multiparity induces persistent myocardial structural, functional and transcriptomic remodelling in mice. Sci. Rep. 2025, 15, 24254. [Google Scholar] [CrossRef]
- Clapp, J.F., III; Capeless, E. Cardiovascular function before, during, and after the first and subsequent pregnancies. Am. J. Cardiol. 1997, 80. [Google Scholar] [CrossRef]
- Chung, E.; Yeung, F.; Leinwand, L.A. Akt and mapk signaling mediate pregnancy-induced cardiac adaptation. J. Appl. Physiol. 2012, 112, 1564–1575. [Google Scholar] [CrossRef]
- Fulghum, K.L.; Smith, J.B.; Chariker, J.; Garrett, L.F.; Brittian, K.R.; Lorkiewicz, P.K.; McNally, L.A.; Uchida, S.; Jones, S.P.; Hill, B.G.; et al. Metabolic signatures of pregnancy-induced cardiac growth. Am. J. Physiol.-Heart Circ. Physiol. 2022, 323, H146–H164. [Google Scholar] [CrossRef] [PubMed]
- Limon-Miranda, S.; Salazar-Enriquez, D.G.; Muñiz, J.; Ramirez-Archila, M.V.; Sanchez-Pastor, E.A.; Andrade, F.; Soñanez-Organis, J.G.; Moran-Palacio, E.F.; Virgen-Ortiz, A. Pregnancy differentially regulates the collagens types I and III in left ventricle from rat heart. BioMed Res. Int. 2014, 2014, 984785. [Google Scholar] [CrossRef] [PubMed]
- Chung, E.; Heimiller, J.; Leinwand, L.A. Distinct cardiac transcriptional profiles defining pregnancy and exercise. PLoS ONE 2012, 7, e42297. [Google Scholar] [CrossRef]
- Thurstin, A.A.; Egeli, A.N.; Goldsmith, E.C.; Spinale, F.G.; LaVoie, H.A. Tissue inhibitor of metalloproteinase-4 deletion in mice impacts maternal cardiac function during pregnancy and postpartum. Am. J. Physiol.-Heart Circ. Physiol. 2023, 324, H85–H99. [Google Scholar] [CrossRef]
- Younesi, F.S.; Miller, A.E.; Barker, T.H.; Rossi, F.M.V.; Hinz, B. Fibroblast and myofibroblast activation in normal tissue repair and fibrosis. Nat. Rev. Mol. Cell Biol. 2024, 25, 617–638. [Google Scholar] [CrossRef]
- Kong, P.; Christia, P.; Frangogiannis, N.G. The pathogenesis of cardiac fibrosis. Cell. Mol. Life Sci. CMLS 2014, 71, 549–574. [Google Scholar] [CrossRef]
- Jiang, W.; Xiong, Y.; Li, X.; Yang, Y. Cardiac fibrosis: cellular effectors, molecular pathways, and exosomal roles. Front. Cardiovasc. Med. 2021, 8, 715258. [Google Scholar] [CrossRef]
- Baum, J.; Duffy, H.S. Fibroblasts and myofibroblasts: what are we talking about? J. Cardiovasc. Pharmacol. 2011, 57, 376–379. [Google Scholar] [CrossRef]
- Hinz, B.; Dugina, V.; Ballestrem, C.; Wehrle-Haller, B.; Chaponnier, C. α-smooth muscle actin is crucial for focal adhesion maturation in myofibroblasts. Mol. Biol. Cell 2003, 14, 2508–2519. [Google Scholar] [CrossRef]
- Herum, K.M.; Lunde, I.G.; McCulloch, A.D.; Christensen, G. The soft- and hard-heartedness of cardiac fibroblasts: mechanotransduction signaling pathways in fibrosis of the heart. J. Clin. Med. 2017, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- González, A.; Schelbert, E.B.; Díez, J.; Butler, J. Myocardial interstitial fibrosis in heart failure: biological and translational perspectives. J. Am. Coll. Cardiol. 2018, 71, 1696–1706. [Google Scholar] [CrossRef] [PubMed]
- Travers, J.G.; Kamal, F.A.; Robbins, J.; Yutzey, K.E.; Blaxall, B.C. Cardiac fibrosis: the fibroblast awakens. Circ. Res. 2016, 118, 1021–1040. [Google Scholar] [CrossRef]
- Kanisicak, O.; Khalil, H.; Ivey, M.J.; Karch, J.; Maliken, B.D.; Correll, R.N.; Brody, M.J.; J Lin, S.-C.; Aronow, B.J.; Tallquist, M.D.; et al. Genetic lineage tracing defines myofibroblast origin and function in the injured heart. Nat. Commun. 2016, 7, 12260. [Google Scholar] [CrossRef]
- Klingberg, F.; Hinz, B.; White, E.S. The myofibroblast matrix: implications for tissue repair and fibrosis. J. Pathol. 2013, 229, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Nkennor, B.; Mastikhina, O.; Soon, K.; Nunes, S.S. Endothelium-mediated contributions to fibrosis. Semin. Cell Dev. Biol. 2020, 101, 78–86. [Google Scholar] [CrossRef]
- Kovacic, J.C.; Dimmeler, S.; Harvey, R.P.; Finkel, T.; Aikawa, E.; Krenning, G.; Baker, A.H. Endothelial to mesenchymal transition in cardiovascular disease: jacc state-of-the-art review. J. Am. Coll. Cardiol. 2019, 73, 190–209. [Google Scholar] [CrossRef]
- Zeisberg, E.M.; Tarnavski, O.; Zeisberg, M.; Dorfman, A.L.; McMullen, J.R.; Gustafsson, E.; Chandraker, A.; Yuan, X.; Pu, W.T.; Roberts, A.B.; et al. Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat. Med. 2007, 13, 952–961. [Google Scholar] [CrossRef]
- Trogisch, F.A.; Abouissa, A.; Keles, M.; Birke, A.; Fuhrmann, M.; Dittrich, G.M.; Weinzierl, N.; Wink, E.; Cordero, J.; Elsherbiny, A.; et al. Endothelial cells drive organ fibrosis in mice by inducing expression of the transcription factor soxSci. Transl. Med. 2024, 16, eabq4581. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, L.; Feng, Z.; Chen, W.; Yan, S.; Yang, R.; Xiao, J.; Gao, J.; Zhang, D.; Ke, X. EPC-derived exosomal mir-1246 and mir-1290 regulate phenotypic changes of fibroblasts to endothelial cells to exert protective effects on myocardial infarction by targeting elf5 and spFront. Cell Dev. Biol. 2021, 9, 647763. [Google Scholar] [CrossRef]
- Ke, X.; Yang, D.; Liang, J.; Wang, X.; Wu, S.; Wang, X.; Hu, C. Human endothelial progenitor cell-derived exosomes increase proliferation and angiogenesis in cardiac fibroblasts by promoting the mesenchymal-endothelial transition and reducing high mobility group box 1 protein b1 expression. DNA Cell Biol. 2017, 36, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Pellman, J.; Zhang, J.; Sheikh, F. Myocyte-fibroblast communication in cardiac fibrosis and arrhythmias: mechanisms and model systems. J. Mol. Cell. Cardiol. 2016, 94, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Herum, K.M.; Choppe, J.; Kumar, A.; Engler, A.J.; McCulloch, A.D. Mechanical regulation of cardiac fibroblast profibrotic phenotypes. Mol. Biol. Cell 2017, 28, 1871–1882. [Google Scholar] [CrossRef]
- Rao, W.; Li, D.; Zhang, Q.; Liu, T.; Gu, Z.; Huang, L.; Dai, J.; Wang, J.; Hou, X. Complex regulation of cardiac fibrosis: insights from immune cells and signaling pathways. J. Transl. Med. 2025, 23, 242. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Vannella, K.M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef]
- Pinto, A.R.; Ilinykh, A.; Ivey, M.J.; Kuwabara, J.T.; D’Antoni, M.L.; Debuque, R.; Chandran, A.; Wang, L.; Arora, K.; Rosenthal, N.; et al. Revisiting cardiac cellular composition. Circ. Res. 2016, 118, 400–409. [Google Scholar] [CrossRef]
- Dick, S.A.; Macklin, J.A.; Nejat, S.; Clemente-Casares, X.; Momen, A.; Kantores, C.; Hosseinzadeh, S.; Barbu, I.; Chen, J.; Althagafi, M.G.; et al. Self-renewing resident cardiac macrophages limit adverse remodeling following myocardial infarction. Nat. Immunol. 2019, 20, 29–39. [Google Scholar] [CrossRef]
- Ma, Y. Role of neutrophils in cardiac injury and repair following myocardial infarction. Cells 2021, 10, 1676. [Google Scholar] [CrossRef] [PubMed]
- Smolgovsky, S.; Bayer, A.L.; Kaur, K.; Sanders, E.; Aronovitz, M.; Filipp, M.E.; Thorp, E.B.; Schiattarella, G.G.; Hill, J.A.; Blanton, R.M.; et al. Impaired t cell ire1α/xbp1 signaling directs inflammation in experimental heart failure with preserved ejection fraction. J. Clin. Invest. 2023, 133. [Google Scholar] [CrossRef]
- Swirski, F.K.; Nahrendorf, M. Leukocyte behavior in atherosclerosis, myocardial infarction, and heart failure. Science 2013, 339, 161–166. [Google Scholar] [CrossRef]
- Dieterlen, M.-T.; John, K.; Reichenspurner, H.; Mohr, F.W.; Barten, M.J. Dendritic cells and their role in cardiovascular diseases: a view on human studies. J. Immunol. Res. 2016, 2016, 5946807. [Google Scholar] [CrossRef]
- Mildner, A.; Marinkovic, G.; Jung, S. Murine monocytes: origins, subsets, fates, and functions. Microbiol. Spectr. 2016, 4. [Google Scholar] [CrossRef]
- Jian, Y.; Zhou, X.; Shan, W.; Chen, C.; Ge, W.; Cui, J.; Yi, W.; Sun, Y. Crosstalk between macrophages and cardiac cells after myocardial infarction. Cell Commun. Signal. 2023, 21, 109. [Google Scholar] [CrossRef]
- Pinto, A.R.; Paolicelli, R.; Salimova, E.; Gospocic, J.; Slonimsky, E.; Bilbao-Cortes, D.; Godwin, J.W.; Rosenthal, N.A. An abundant tissue macrophage population in the adult murine heart with a distinct alternatively-activated macrophage profile. PLoS ONE 2012, 7, e36814. [Google Scholar] [CrossRef] [PubMed]
- Epelman, S.; Lavine, K.J.; Beaudin, A.E.; Sojka, D.K.; Carrero, J.A.; Calderon, B.; Brija, T.; Gautier, E.L.; Ivanov, S.; Satpathy, A.T.; et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity 2014, 40, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, G.; Bredemeyer, A.; Li, W.; Zaitsev, K.; Koenig, A.L.; Lokshina, I.; Mohan, J.; Ivey, B.; Hsiao, H.-M.; Weinheimer, C.; et al. Tissue resident ccr2− and ccr2+ cardiac macrophages differentially orchestrate monocyte recruitment and fate specification following myocardial injury. Circ. Res. 2019, 124, 263–278. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.-L.; Lin, Q.-Y.; Liu, Y.; Guan, X.-M.; Ma, X.-L.; Cao, H.-J.; Liu, Y.; Bai, J.; Xia, Y.-L.; et al. CXCL1–cxcr2 axis mediates angiotensin ii-induced cardiac hypertrophy and remodelling through regulation of monocyte infiltration. Eur. Heart J. 2018, 39, 1818–1831. [Google Scholar] [CrossRef]
- Wong, K.L.; Tai, J.J.-Y.; Wong, W.-C.; Han, H.; Sem, X.; Yeap, W.-H.; Kourilsky, P.; Wong, S.-C. Gene expression profiling reveals the defining features of the classical, intermediate, and nonclassical human monocyte subsets. Blood 2011, 118, e16–e31. [Google Scholar] [CrossRef]
- Bachelerie, F.; Ben-Baruch, A.; Burkhardt, A.M.; Combadiere, C.; Farber, J.M.; Graham, G.J.; Horuk, R.; Sparre-Ulrich, A.H.; Locati, M.; Luster, A.D.; et al. International union of pharmacology. lxxxix. update on the extended family of chemokine receptors and introducing a new nomenclature for atypical chemokine receptors. Pharmacol. Rev. 2014, 66, 1–79. [Google Scholar] [CrossRef]
- Chen, R.; Zhang, H.; Tang, B.; Luo, Y.; Yang, Y.; Zhong, X.; Chen, S.; Xu, X.; Huang, S.; Liu, C. Macrophages in cardiovascular diseases: molecular mechanisms and therapeutic targets. Signal Transduct. Target. Ther. 2024, 9, 1–44. [Google Scholar] [CrossRef]
- Yerra, V.G.; Advani, A. Role of ccr2-positive macrophages in pathological ventricular remodelling. Biomedicines 2022, 10, 661. [Google Scholar] [CrossRef]
- Revelo, X.S.; Parthiban, P.; Chen, C.; Barrow, F.; Fredrickson, G.; Wang, H.; Yücel, D.; Herman, A.; van Berlo, J.H. Cardiac resident macrophages prevent fibrosis and stimulate angiogenesis. Circ. Res. 2021, 129, 1086–1101. [Google Scholar] [CrossRef]
- Bolego, C.; Cignarella, A.; Staels, B.; Chinetti-Gbaguidi, G. Macrophage function and polarization in cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Hoeft, K.; Schaefer, G.J.L.; Kim, H.; Schumacher, D.; Bleckwehl, T.; Long, Q.; Klinkhammer, B.M.; Peisker, F.; Koch, L.; Nagai, J.; et al. Platelet-instructed spp1+ macrophages drive myofibroblast activation in fibrosis in a cxcl4-dependent manner. Cell Rep. 2023, 42, 112131. [Google Scholar] [CrossRef]
- Lafuse, W.P.; Wozniak, D.J.; Rajaram, M.V.S. Role of cardiac macrophages on cardiac inflammation, fibrosis and tissue repair. Cells 2020, 10, 51. [Google Scholar] [CrossRef] [PubMed]
- Kologrivova, I.; Shtatolkina, M.; Suslova, T.; Ryabov, V. Cells of the immune system in cardiac remodeling: main players in resolution of inflammation and repair after myocardial infarction. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Thomassen, J.Q.; Nordestgaard, B.G.; Tybjærg-Hansen, A.; Frikke-Schmidt, R. Neutrophil counts and cardiovascular disease. Eur. Heart J. 2023, 44, 4953–4964. [Google Scholar] [CrossRef]
- Antipenko, S.; Mayfield, N.; Jinno, M.; Gunzer, M.; Ismahil, M.A.; Hamid, T.; Prabhu, S.D.; Rokosh, G. Neutrophils are indispensable for adverse cardiac remodeling in heart failure. J. Mol. Cell. Cardiol. 2024, 189, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Puhl, S.-L.; Steffens, S. Neutrophils in post-myocardial infarction inflammation: damage vs. resolution? Front. Cardiovasc. Med. 2019, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Loh, W.; Vermeren, S. Anti-inflammatory neutrophil functions in the resolution of inflammation and tissue repair. Cells 2022, 11, 4076. [Google Scholar] [CrossRef]
- Cano, R.L.E.; Lopera, H.D.E. Introduction to T and B lymphocytes. In Autoimmunity: From Bench to Bedside [Internet]; El Rosario University Press, 2013. [Google Scholar]
- Hofmann, U.; Beyersdorf, N.; Weirather, J.; Podolskaya, A.; Bauersachs, J.; Ertl, G.; Kerkau, T.; Frantz, S. Activation of cd4+ t lymphocytes improves wound healing and survival after experimental myocardial infarction in mice. Circulation 2012, 125, 1652–1663. [Google Scholar] [CrossRef] [PubMed]
- Laroumanie, F.; Douin-Echinard, V.; Pozzo, J.; Lairez, O.; Tortosa, F.; Vinel, C.; Delage, C.; Calise, D.; Dutaur, M.; Parini, A.; et al. CD4+ t cells promote the transition from hypertrophy to heart failure during chronic pressure overload. Circulation 2014, 129, 2111–2124. [Google Scholar] [CrossRef]
- Radziszewska, A.; Moulder, Z.; Jury, E.C.; Ciurtin, C. CD8+ t cell phenotype and function in childhood and adult-onset connective tissue disease. Int. J. Mol. Sci. 2022, 23, 11431. [Google Scholar] [CrossRef]
- Ilatovskaya, D.V.; Pitts, C.; Clayton, J.; Domondon, M.; Troncoso, M.; Pippin, S.; DeLeon-Pennell, K.Y. CD8+ t-cells negatively regulate inflammation post-myocardial infarction. Am. J. Physiol.-Heart Circ. Physiol. 2019, 317, H581–H596. [Google Scholar] [CrossRef]
- Althwaiqeb, S.A.; Fakoya, A.O.; Bordoni, B. Histology, B-cell lymphocyte. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Goodchild, T.T.; Robinson, K.A.; Pang, W.; Tondato, F.; Cui, J.; Arrington, J.; Godwin, L.; Ungs, M.; Carlesso, N.; Weich, N.; et al. Bone marrow-derived B cells preserve ventricular function after acute myocardial infarction. JACC Cardiovasc. Interv. 2009, 2, 1005–1016. [Google Scholar] [CrossRef]
- Kallikourdis, M.; Martini, E.; Carullo, P.; Sardi, C.; Roselli, G.; Greco, C.M.; Vignali, D.; Riva, F.; Ormbostad Berre, A.M.; Stølen, T.O.; et al. T cell costimulation blockade blunts pressure overload-induced heart failure. Nat. Commun. 2017, 8, 14680. [Google Scholar] [CrossRef]
- Haghikia, A.; Kaya, Z.; Schwab, J.; Westenfeld, R.; Ehlermann, P.; Bachelier, K.; Oettl, R.; von Kaisenberg, C.S.; Katus, H.A.; Bauersachs, J.; et al. Evidence of autoantibodies against cardiac troponin i and sarcomeric myosin in peripartum cardiomyopathy. Basic Res. Cardiol. 2015, 110, 60. [Google Scholar] [CrossRef]
- Fong, M.; Crane, J.S. Histology, mast cells. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Jaggi, A.S.; Singh, M.; Sharma, A.; Singh, D.; Singh, N. Cardioprotective effects of mast cell modulators in ischemia-reperfusion-induced injury in rats. Methods Find. Exp. Clin. Pharmacol. 2007, 29, 593–600. [Google Scholar] [CrossRef]
- Levick, S.P.; McLarty, J.L.; Murray, D.B.; Freeman, R.M.; Carver, W.E.; Brower, G.L. Cardiac mast cells mediate left ventricular fibrosis in the hypertensive rat heart. Hypertension 2009, 53, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.S.; Kim, Y.S.; Cho, A.S.; Cho, H.H.; Kim, J.S.; Hong, M.H.; Jeong, S.Y.; Jeong, M.H.; Cho, J.G.; Park, J.C.; et al. The novel role of mast cells in the microenvironment of acute myocardial infarction. J. Mol. Cell. Cardiol. 2011, 50, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Magon, N. Hormones in pregnancy. Niger. Med. J. J. Niger. Med. Assoc. 2012, 53, 179–183. [Google Scholar] [CrossRef]
- Frohlich, J. Mice and Rats as Pets - Exotic and Laboratory Animals. Available online: https://www.merckvetmanual.com/exotic-and-laboratory-animals/rodents/mice-and-rats-as-pets (accessed on 2 March 2026).
- Murray, S.A.; Morgan, J.L.; Kane, C.; Sharma, Y.; Heffner, C.S.; Lake, J.; Donahue, L.R. Mouse gestation length is genetically determined. PloS One 2010, 5, e12418. [Google Scholar] [CrossRef]
- Jukic, A.M.; Baird, D.D.; Weinberg, C.R.; McConnaughey, D.R.; Wilcox, A.J. Length of human pregnancy and contributors to its natural variation. Hum. Reprod. Oxf. Engl. 2013, 28, 2848–2855. [Google Scholar] [CrossRef]
- Tal, R.; Taylor, H.S. Endocrinology of pregnancy. In Endotext; Feingold, K.R., Ahmed, S.F., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., Hofland, J., Kalra, S., Kaltsas, G., Kapoor, N., Koch, C., Kopp, P., Korbonits, M., Kovacs, C.S., Kuohung, W., Laferrère, B., Levy, M., McGee, E.A., McLachlan, R., Muzumdar, R., Purnell, J., Rey, R., Sahay, R., Shah, A.S., Singer, F., Sperling, M.A., Stratakis, C.A., Trence, D.L., Wilson, D.P., Eds.; MDText.com, Inc.: South Dartmouth (MA), 2000. [Google Scholar]
- Gruber, C.J.; Tschugguel, W.; Schneeberger, C.; Huber, J.C. Production and actions of estrogens. N. Engl. J. Med. 2002, 346, 340–352. [Google Scholar] [CrossRef] [PubMed]
- Parichatikanond, W.; Duangrat, R.; Kurose, H.; Mangmool, S. Regulation of β-adrenergic receptors in the heart: a review on emerging therapeutic strategies for heart failure. Cells 2024, 13, 1674. [Google Scholar] [CrossRef]
- Chu, S.H.; Goldspink, P.; Kowalski, J.; Beck, J.; Schwertz, D.W. Effect of estrogen on calcium-handling proteins, beta-adrenergic receptors, and function in rat heart. Life Sci. 2006, 79, 1257–1267. [Google Scholar] [CrossRef]
- Bupha-Intr, T.; Wattanapermpool, J. Regulatory role of ovarian sex hormones in calcium uptake activity of cardiac sarcoplasmic reticulum. Am. J. Physiol.-Heart Circ. Physiol. 2006, 291, H1101–H1108. [Google Scholar] [CrossRef]
- Matarrese, P.; Maccari, S.; Vona, R.; Gambardella, L.; Stati, T.; Marano, G. Role of β-adrenergic receptors and estrogen in cardiac repair after myocardial infarction: an overview. Int. J. Mol. Sci. 2021, 22, 8957. [Google Scholar] [CrossRef] [PubMed]
- Fliegner, D.; Schubert, C.; Penkalla, A.; Witt, H.; Kararigas, G.; Dworatzek, E.; Staub, E.; Martus, P.; Ruiz Noppinger, P.; Kintscher, U.; et al. Female sex and estrogen receptor-beta attenuate cardiac remodeling and apoptosis in pressure overload. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1597–1606. [Google Scholar] [CrossRef] [PubMed]
- Pedram, A.; Razandi, M.; O’Mahony, F.; Lubahn, D.; Levin, E.R. Estrogen receptor-beta prevents cardiac fibrosis. Mol. Endocrinol. Baltim. Md 2010, 24, 2152–2165. [Google Scholar] [CrossRef] [PubMed]
- Pelligrino, D.A.; Ye, S.; Tan, F.; Santizo, R.A.; Feinstein, D.L.; Wang, Q. Nitric-oxide-dependent pial arteriolar dilation in the female rat: effects of chronic estrogen depletion and repletion. Biochem. Biophys. Res. Commun. 2000, 269, 165–171. [Google Scholar] [CrossRef]
- Takahashi, K.; Ohmichi, M.; Yoshida, M.; Hisamoto, K.; Mabuchi, S.; Arimoto-Ishida, E.; Mori, A.; Tsutsumi, S.; Tasaka, K.; Murata, Y.; et al. Both estrogen and raloxifene cause g1 arrest of vascular smooth muscle cells. J. Endocrinol. 2003, 178, 319–329. [Google Scholar] [CrossRef]
- Fedotcheva, T.A. Clinical use of progestins and their mechanisms of action: present and future (review). Mod. Technol. Med. 2021, 13, 93–106. [Google Scholar] [CrossRef]
- dos Santos, R.L.; da Silva, F.B.; Ribeiro, R.F.; Stefanon, I. Sex hormones in the cardiovascular system. Horm. Mol. Biol. Clin. Investig. 2014, 18, 89–103. [Google Scholar] [CrossRef]
- Simoncini, T.; Mannella, P.; Fornari, L.; Caruso, A.; Willis, M.Y.; Garibaldi, S.; Baldacci, C.; Genazzani, A.R. Differential signal transduction of progesterone and medroxyprogesterone acetate in human endothelial cells. Endocrinology 2004, 145, 5745–5756. [Google Scholar] [CrossRef]
- Fedotcheva, T.A.; Fedotcheva, N.I.; Shimanovsky, N.L. Progesterone as an anti-inflammatory drug and immunomodulator: new aspects in hormonal regulation of the inflammation. Biomolecules 2022, 12, 1299. [Google Scholar] [CrossRef]
- Dubey, R.K.; Gillespie, D.G.; Jackson, E.K.; Keller, P.J. 17β-estradiol, its metabolites, and progesterone inhibit cardiac fibroblast growth. Hypertension 1998, 31, 522–528. [Google Scholar] [CrossRef]
- Forestiero, V.; Sconfienza, E.; Mulatero, P.; Monticone, S. Primary aldosteronism in pregnancy. Rev. Endocr. Metab. Disord. 2023, 24, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Likes, C.E.; Luz, A.L.; Mao, L.; Yeh, J.S.; Wei, Z.; Kuchibhatla, M.; Ilkayeva, O.R.; Koves, T.R.; Price, T.M. A mitochondrial progesterone receptor increases cardiac beta-oxidation and remodeling. J. Endocr. Soc. 2019, 3, 446–467. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.; Sites, C.K.; Toth, M.J. Progesterone stimulates cardiac muscle protein synthesis via receptor-dependent pathway. Fertil. Steril. 2004, 82, 430–436. [Google Scholar] [CrossRef]
- Sarwar, M.; Du, X.; Dschietzig, T.B.; Summers, R.J. The actions of relaxin on the human cardiovascular system. Br. J. Pharmacol. 2017, 174, 933–949. [Google Scholar] [CrossRef]
- Conrad, K.P. Maternal vasodilation in pregnancy: the emerging role of relaxin. Am. J. Physiol. - Regul. Integr. Comp. Physiol. 2011, 301, R267–R275. [Google Scholar] [CrossRef]
- Conrad, K.P.; Debrah, D.O.; Novak, J.; Danielson, L.A.; Shroff, S.G. Relaxin modifies systemic arterial resistance and compliance in conscious, nonpregnant rats. Endocrinology 2004, 145, 3289–3296. [Google Scholar] [CrossRef]
- Samuel, C.S.; Unemori, E.N.; Mookerjee, I.; Bathgate, R.A.D.; Layfield, S.L.; Mak, J.; Tregear, G.W.; Du, X.-J. Relaxin modulates cardiac fibroblast proliferation, differentiation, and collagen production and reverses cardiac fibrosis in vivo. Endocrinology 2004, 145, 4125–4133. [Google Scholar] [CrossRef]
- Al-Chalabi, M.; Bass, A.N.; Alsalman, I. Physiology, prolactin. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Freeman, G.J.; Long, A.J.; Iwai, Y.; Bourque, K.; Chernova, T.; Nishimura, H.; Fitz, L.J.; Malenkovich, N.; Okazaki, T.; Byrne, M.C.; et al. Engagement of the pd-1 immunoinhibitory receptor by a novel b7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 2000, 192, 1027–1034. [Google Scholar] [CrossRef]
- Vieira Borba, V.; Shoenfeld, Y. Prolactin, autoimmunity, and motherhood: when should women avoid breastfeeding? Clin. Rheumatol. 2019, 38, 1263–1270. [Google Scholar] [CrossRef]
- Borba, V.V.; Zandman-Goddard, G.; Shoenfeld, Y. Prolactin and autoimmunity. Front. Immunol. 2018, 9, 73. [Google Scholar] [CrossRef] [PubMed]
- Rasmi, Y.; Jalali, L.; Khalid, S.; Shokati, A.; Tyagi, P.; Ozturk, A.; Nasimfar, A. The effects of prolactin on the immune system, its relationship with the severity of covid-19, and its potential immunomodulatory therapeutic effect. Cytokine 2023, 169, 156253. [Google Scholar] [CrossRef] [PubMed]
- Patten, I.S.; Rana, S.; Shahul, S.; Rowe, G.C.; Jang, C.; Liu, L.; Hacker, M.R.; Rhee, J.S.; Mitchell, J.; Mahmood, F.; et al. Cardiac angiogenic imbalance leads to peripartum cardiomyopathy. Nature 2012, 485, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Ravi, R.; Sivakumar, R.K.; Chidambaram, V.; Majella, M.G.; Sinha, S.; Adamo, L.; Lau, E.S.; Al’Aref, S.J.; Asnani, A.; et al. Prolactin inhibition in peripartum cardiomyopathy: systematic review and meta-analysis. Curr. Probl. Cardiol. 2023, 48, 101461. [Google Scholar] [CrossRef]
- Haghikia, A.; Schwab, J.; Vogel-Claussen, J.; Berliner, D.; Pfeffer, T.; König, T.; Zwadlo, C.; Moulig, V.A.; Franke, A.; Schwarzkopf, M.; et al. Bromocriptine treatment in patients with peripartum cardiomyopathy and right ventricular dysfunction. Clin. Res. Cardiol. 2019, 108, 290–297. [Google Scholar] [CrossRef]
- Rassie, K.; Giri, R.; Joham, A.E.; Teede, H.; Mousa, A. Human placental lactogen in relation to maternal metabolic health and fetal outcomes: a systematic review and meta-analysis. Int. J. Mol. Sci. 2022, 23, 15621. [Google Scholar] [CrossRef]
- Garay, S.M.; Sumption, L.A.; John, R.M. Prenatal health behaviours as predictors of human placental lactogen levels. Front. Endocrinol. 2022, 13. [Google Scholar] [CrossRef]
- Herghelegiu, C.G.; Veduta, A.; Stefan, M.F.; Magda, S.L.; Ionascu, I.; Radoi, V.E.; Oprescu, D.N.; Calin, A.M. Hyperglycosylated-hcg: its role in trophoblast invasion and intrauterine growth restriction. Cells 2023, 12, 1647. [Google Scholar] [CrossRef]
- Cable, J.K.; Grider, M.H. Physiology, progesterone. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Gridelet, V.; Perrier d’Hauterive, S.; Polese, B.; Foidart, J.-M.; Nisolle, M.; Geenen, V. Human chorionic gonadotrophin: new pleiotropic functions for an “old” hormone during pregnancy. Front. Immunol. 2020, 11, 343. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Arrowsmith, S.; Wray, S. Oxytocin: its mechanism of action and receptor signalling in the myometrium. J. Neuroendocrinol. 2014, 26, 356–369. [Google Scholar] [CrossRef]
- Jankowski, M.; Broderick, T.L.; Gutkowska, J. The role of oxytocin in cardiovascular protection. Front. Psychol. 2020, 11, 2139. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, J.; Wang, L.; Wu, W.; Wang, Q.; Zhao, Y.; Qian, X.; Wang, Z.; Fu, N.; Wang, Y.; et al. Oxytocin attenuates cardiac hypertrophy by improving cardiac glucose metabolism and regulating oxtr/jak2/stat3 axis. Peptides 2024, 182, 171323. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Yao, M.; Zhou, R.; Qu, Y.; Yang, Y.; Wang, Z.; Song, N.; Chen, H.; Qian, J. Oxytocin ameliorates ischemia/reperfusion-induced injury by inhibiting mast cell degranulation and inflammation in the rat heart. Biomed. Pharmacother. Biomedecine Pharmacother. 2020, 128, 110358. [Google Scholar] [CrossRef] [PubMed]
- Lother, A.; Kohl, P. The heterocellular heart: identities, interactions, and implications for cardiology. Basic Res. Cardiol. 2023, 118, 30. [Google Scholar] [CrossRef]
- Lintao, R.C.V.; Kammala, A.K.; Radnaa, E.; Bettayeb, M.; Vincent, K.L.; Patrikeev, I.; Yaklic, J.; Bonney, E.A.; Menon, R. Characterization of fetal microchimeric immune cells in mouse maternal hearts during physiologic and pathologic pregnancies. Front. Cell Dev. Biol. 2023, 11. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



