Submitted:
02 April 2026
Posted:
02 April 2026
You are already at the latest version
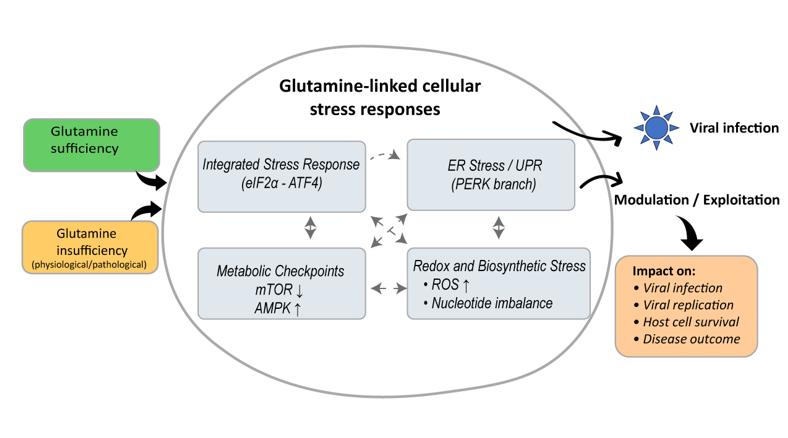
Abstract
Keywords:
1. Introduction
2. Glutamine Biology as a Regulator of Cellular Stress
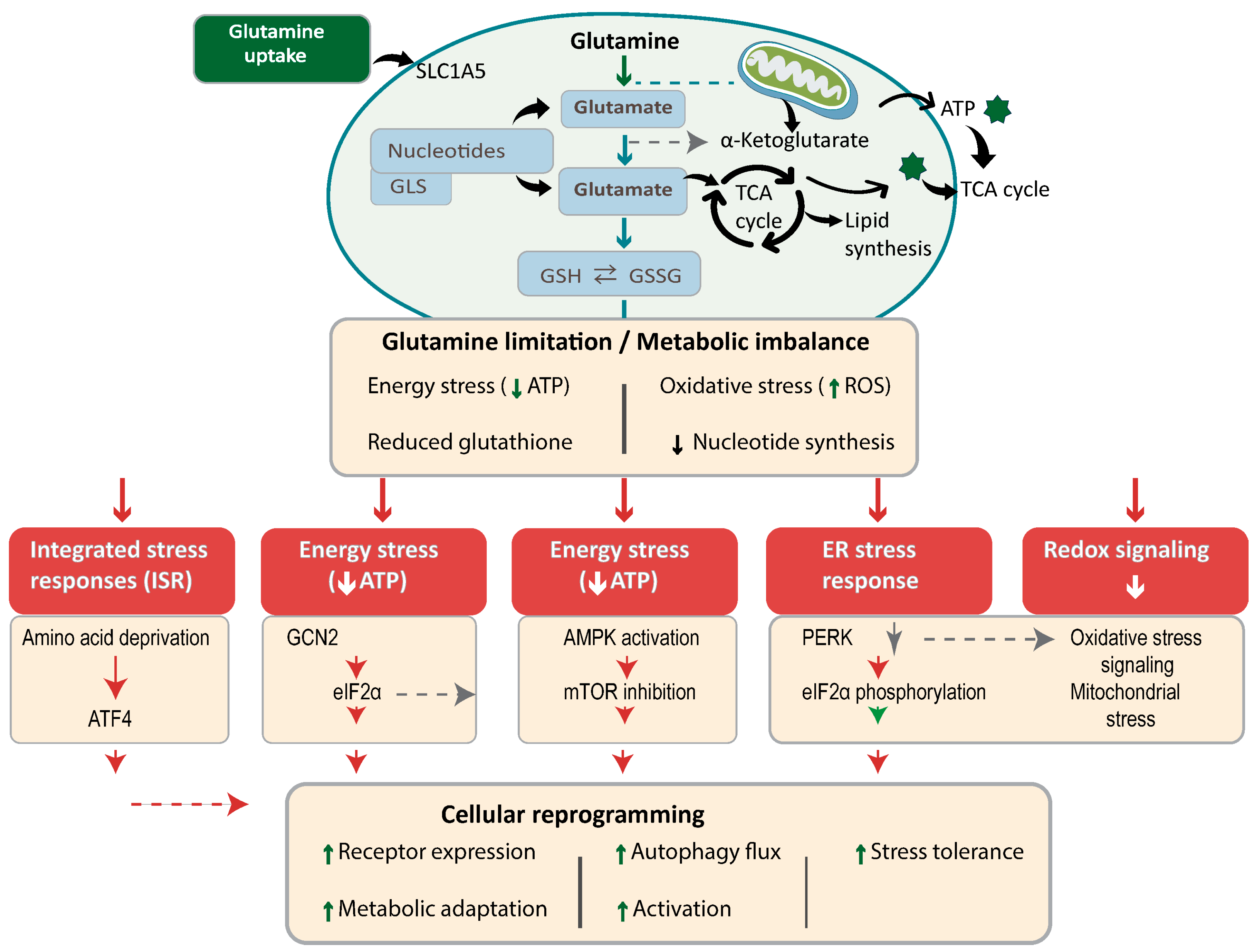
2.1. Glutamine Metabolism and Homeostasis
2.2. Glutamine Sensing and Activation of Stress Pathways
2.3. Redox and Biosynthetic Stress Induced by Glutamine Imbalance
3. Glutamine-Linked Cellular Stress Pathways
3.1. Integrated Stress Response
3.2. Endoplasmic Reticulum Stress and the Unfolded Protein Response
3.3. Metabolic Stress Signaling: AMPK and mTOR
3.4. Autophagy as an Adaptive Response
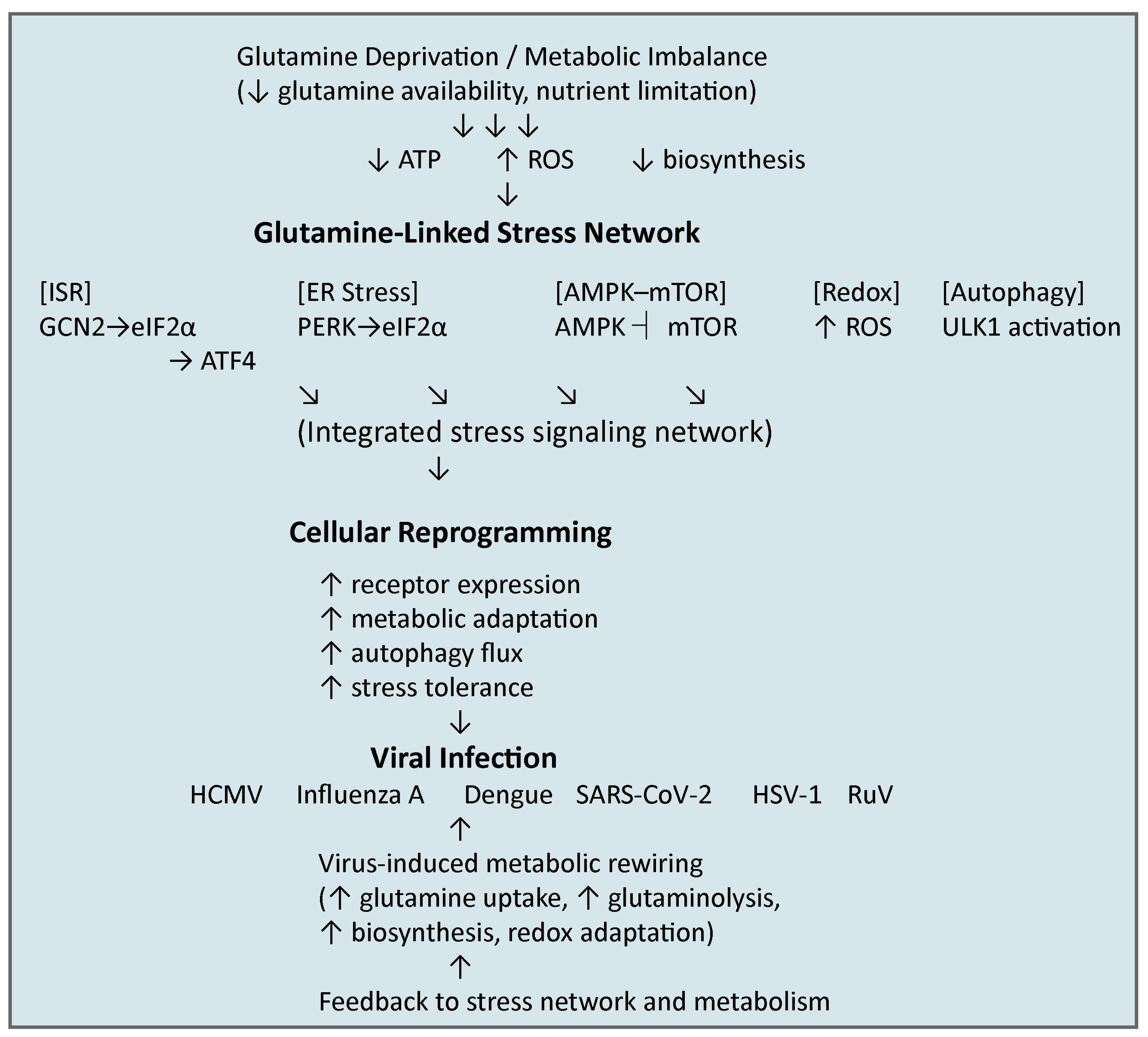
3.5. Integration of Stress Networks
4. Viral Exploitation of Glutamine-Linked Stress Networks
4.1. Viral Dependence on Host Glutamine Metabolism
4.2. Integrated Stress Response in Viral Infection
4.3. Endoplasmic Reticulum Stress in Viral Infection
4.4. Metabolic Stress Signaling (AMPK–mTOR Axis) in Viral Infection
4.5. Autophagy in Viral Infection
| Virus | Viral Type |
Metabolic/Glutamine Reprogramming |
Stress Pathways | Functional Outcome | References |
| HCMV | DNA | ↑ Glutamine uptake; ↑ TCA cycle flux | mTOR; ER stress; autophagy | Supports biosynthesis and replication | [47,62] |
| Dengue virus | RNA | ↑ Glutamine metabolism; ↑ lipid metabolism (lipophagy) | Autophagy; ER stress (PERK); ROS | Enhances replication complex formation | [46,65,66] |
| Influenza A virus | RNA | ↑ metabolic reprogramming (primarily glucose; glutamine supportive) | AMPK; ROS | Supports replication | [49,61,67,68] |
| SARS-CoV-2 | RNA | Altered metabolism; redox imbalance | ER stress; ISR; autophagy; GRP78 | Modulates replication; enhances entry | [52,53,69,70] |
| Hepatitis B virus | DNA | Metabolic reprogramming; ↑ amino acid metabolism (including glutamine) | ER stress (UPR); ROS | Supports replication; promotes viral protein production | [71,72,73,74] |
| Hepatitis C virus | RNA | Reprograms amino acid (including glutamine, indirect) and lipid metabolism | ER stress, autophagy | Promotes replication and assembly | [9,75,76,77] |
| KSHV |
DNA | ↑ Glutamine metabolism (latency/reactivation) | mTOR; redox pathways | Supports persistence and oncogenesis | [20] |
| ASFV | DNA | Redox remodeling; ↑ GSH | ER stress; PERK–eIF2α; ATF6–Ca2⁺; autophagy; ROS | Favors replication environment | [55,56,57,58] |
| Rubella virus | RNA | Nutrient stress sensitivity; redox imbalance | ER stress; ROS; ISR (proposed) | Enhances infection; ↑ susceptibility | [15,19] |
| Rotavirus | RNA | ↑ Glutamine catabolism; ↑ aspartate biosynthesis | ER stress; PERK–eIF2α; ROS; AMPK–Nrf2 | Supports replication; enhances biosynthesis | [78,79,80,81] |
5. Perspectives and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Newsholme, P.; Procopio, J.; Lima, M.M.; Pithon-Curi, T.C.; Curi, R. Glutamine and glutamate--their central role in cell metabolism and function. Cell Biochem Funct 2003, 21, 1-9. [CrossRef]
- DeBerardinis, R.J.; Cheng, T. Q's next: the diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene 2010, 29, 313-324. [CrossRef]
- Cruzat, V.; Macedo Rogero, M.; Noel Keane, K.; Curi, R.; Newsholme, P. Glutamine: Metabolism and Immune Function, Supplementation and Clinical Translation. Nutrients 2018, 10. [CrossRef]
- Altman, B.J.; Stine, Z.E.; Dang, C.V. From Krebs to clinic: glutamine metabolism to cancer therapy. Nat Rev Cancer 2016, 16, 749; ; Erratum in Nat Rev Cancer 2016, 16, 773, https://doi.org/10.1038/nrc.2016.131. [CrossRef]
- Lu, S.C. Glutathione synthesis. Biochim Biophys Acta 2013, 1830, 3143-3153. [CrossRef]
- Galluzzi, L.; Baehrecke, E.H.; Ballabio, A.; Boya, P.; Bravo-San Pedro, J.M.; Cecconi, F.; Choi, A.M.; Chu, C.T.; Codogno, P.; Colombo, M.I.; et al. Molecular definitions of autophagy and related processes. EMBO J 2017, 36, 1811-1836. [CrossRef] [PubMed]
- Hetz, C.; Zhang, K.; Kaufman, R.J. Mechanisms, regulation and functions of the unfolded protein response. Nat Rev Mol Cell Biol 2020, 21, 421-438. [CrossRef] [PubMed]
- Pakos-Zebrucka, K.; Koryga, I.; Mnich, K.; Ljujic, M.; Samali, A.; Gorman, A.M. The integrated stress response. EMBO Rep 2016, 17, 1374-1395. [CrossRef] [PubMed]
- Munz, C.; Campbell, G.R.; Esclatine, A.; Faure, M.; Labonte, P.; Lussignol, M.; Orvedahl, A.; Altan-Bonnet, N.; Bartenschlager, R.; Beale, R.; et al. Autophagy machinery as exploited by viruses. Autophagy Rep 2025, 4. [CrossRef]
- Saxton, R.A.; Sabatini, D.M. mTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960-976. [CrossRef]
- Sanchez, E.L.; Lagunoff, M. Viral activation of cellular metabolism. Virology 2015, 479-480, 609-618. [CrossRef]
- Thaker, S.K.; Ch'ng, J.; Christofk, H.R. Viral hijacking of cellular metabolism. BMC Biol 2019, 17, 59. [CrossRef]
- Goodwin, C.M.; Xu, S.; Munger, J. Stealing the Keys to the Kitchen: Viral Manipulation of the Host Cell Metabolic Network. Trends Microbiol 2015, 23, 789-798. [CrossRef] [PubMed]
- Stern-Ginossar, N.; Thompson, S.R.; Mathews, M.B.; Mohr, I. Translational Control in Virus-Infected Cells. Cold Spring Harb Perspect Biol 2019, 11. [CrossRef]
- Trinh, Q.D.; Takada, K.; Pham, N.T.K.; Takano, C.; Namiki, T.; Ito, S.; Takeda, Y.; Okitsu, S.; Ushijima, H.; Hayakawa, S.; et al. Oxidative Stress Enhances Rubella Virus Infection in Immortalized Human First-Trimester Trophoblasts. Int J Mol Sci 2025, 26. [CrossRef]
- Jackson, W.T. Viruses and the autophagy pathway. Virology 2015, 479-480, 450-456. [CrossRef]
- Codo, A.C.; Davanzo, G.G.; Monteiro, L.B.; de Souza, G.F.; Muraro, S.P.; Virgilio-da-Silva, J.V.; Prodonoff, J.S.; Carregari, V.C.; de Biagi Junior, C.A.O.; Crunfli, F.; et al. Elevated Glucose Levels Favor SARS-CoV-2 Infection and Monocyte Response through a HIF-1alpha/Glycolysis-Dependent Axis. Cell Metab 2020, 32, 437-446 e435. [CrossRef]
- Ke, P.Y. The Multifaceted Roles of Autophagy in Flavivirus-Host Interactions. Int J Mol Sci 2018, 19. [CrossRef]
- Trinh, Q.D.; Takada, K.; Pham, N.T.K.; Takano, C.; Namiki, T.; Ikuta, R.; Hayashida, S.; Okitsu, S.; Ushijima, H.; Komine-Aizawa, S.; et al. Enhancement of Rubella Virus Infection in Immortalized Human First-Trimester Trophoblasts Under Low-Glucose Stress Conditions. Front Microbiol 2022, 13, 904189. [CrossRef] [PubMed]
- Sanchez, E.L.; Pulliam, T.H.; Dimaio, T.A.; Thalhofer, A.B.; Delgado, T.; Lagunoff, M. Glycolysis, Glutaminolysis, and Fatty Acid Synthesis Are Required for Distinct Stages of Kaposi's Sarcoma-Associated Herpesvirus Lytic Replication. J Virol 2017, 91. [CrossRef]
- Scalise, M.; Pochini, L.; Console, L.; Losso, M.A.; Indiveri, C. The Human SLC1A5 (ASCT2) Amino Acid Transporter: From Function to Structure and Role in Cell Biology. Front Cell Dev Biol 2018, 6, 96. [CrossRef]
- Nicklin, P.; Bergman, P.; Zhang, B.; Triantafellow, E.; Wang, H.; Nyfeler, B.; Yang, H.; Hild, M.; Kung, C.; Wilson, C.; et al. Bidirectional transport of amino acids regulates mTOR and autophagy. Cell 2009, 136, 521-534. [CrossRef]
- Labow, B.I.; Souba, W.W.; Abcouwer, S.F. Mechanisms governing the expression of the enzymes of glutamine metabolism--glutaminase and glutamine synthetase. J Nutr 2001, 131, 2467S-2474S; discussion 2486S-2467S. [CrossRef]
- Yang, L.; Venneti, S.; Nagrath, D. Glutaminolysis: A Hallmark of Cancer Metabolism. Annu Rev Biomed Eng 2017, 19, 163-194. [CrossRef] [PubMed]
- Hensley, C.T.; Wasti, A.T.; DeBerardinis, R.J. Glutamine and cancer: cell biology, physiology, and clinical opportunities. J Clin Invest 2013, 123, 3678-3684. [CrossRef]
- Pavlova, N.N.; Thompson, C.B. The Emerging Hallmarks of Cancer Metabolism. Cell Metab 2016, 23, 27-47. [CrossRef]
- Wek, R.C. Role of eIF2alpha Kinases in Translational Control and Adaptation to Cellular Stress. Cold Spring Harb Perspect Biol 2018, 10. [CrossRef] [PubMed]
- Jewell, J.L.; Russell, R.C.; Guan, K.L. Amino acid signalling upstream of mTOR. Nat Rev Mol Cell Biol 2013, 14, 133-139. [CrossRef] [PubMed]
- Jewell, J.L.; Kim, Y.C.; Russell, R.C.; Yu, F.X.; Park, H.W.; Plouffe, S.W.; Tagliabracci, V.S.; Guan, K.L. Metabolism. Differential regulation of mTORC1 by leucine and glutamine. Science 2015, 347, 194-198. [CrossRef]
- Reshi, M.L.; Su, Y.C.; Hong, J.R. RNA Viruses: ROS-Mediated Cell Death. Int J Cell Biol 2014, 2014, 467452. [CrossRef]
- B'Chir, W.; Maurin, A.C.; Carraro, V.; Averous, J.; Jousse, C.; Muranishi, Y.; Parry, L.; Stepien, G.; Fafournoux, P.; Bruhat, A. The eIF2alpha/ATF4 pathway is essential for stress-induced autophagy gene expression. Nucleic Acids Res 2013, 41, 7683-7699. [CrossRef]
- Costa-Mattioli, M.; Walter, P. The integrated stress response: From mechanism to disease. Science 2020, 368. [CrossRef]
- Nandakumar, S.; Grmai, L.; Vasudevan, D. Emerging roles for integrated stress response signaling in homeostasis. FEBS J 2025, 292, 4418-4445. [CrossRef]
- Jeong, J.; Kim, J.; Kim, M.S. Dual Nature of Mitochondrial Integrated Stress Response: Molecular Switches from Protection to Pathology. Genes (Basel) 2025, 16. [CrossRef]
- Ryoo, H.D. The integrated stress response in metabolic adaptation. J Biol Chem 2024, 300, 107151. [CrossRef] [PubMed]
- Zhu, J.; Marciniak, S.J. GCN2 in proteostasis: structural logic, signalling networks and disease. FEBS J 2026. [CrossRef]
- Yuan, J.R.; Tang, J.; Sheng, R. The stress responsive transcription factor ATF4: from molecular structure to disease mechanisms. J Adv Res 2026. [CrossRef]
- Adjibade, P.; Mazroui, R. Regulation of Translation of ATF4 mRNA: A Focus on Translation Initiation Factors and RNA-Binding Proteins. Cells 2026, 15. [CrossRef]
- Yan, X.; Liu, C. The ATF4-glutamine axis: a central node in cancer metabolism, stress adaptation, and therapeutic targeting. Cell Death Discov 2025, 11, 390. [CrossRef]
- Ye, J.; Kumanova, M.; Hart, L.S.; Sloane, K.; Zhang, H.; De Panis, D.N.; Bobrovnikova-Marjon, E.; Diehl, J.A.; Ron, D.; Koumenis, C. The GCN2-ATF4 pathway is critical for tumour cell survival and proliferation in response to nutrient deprivation. EMBO J 2010, 29, 2082-2096. [CrossRef]
- Chan, S.W. Unfolded protein response in hepatitis C virus infection. Front Microbiol 2014, 5, 233. [CrossRef]
- Hetz, C.; Papa, F.R. The Unfolded Protein Response and Cell Fate Control. Mol Cell 2018, 69, 169-181. [CrossRef]
- Kwon, J.; Kim, J.; Kim, K.I. Crosstalk between endoplasmic reticulum stress response and autophagy in human diseases. Anim Cells Syst (Seoul) 2023, 27, 29-37. [CrossRef]
- Kroemer, G.; Marino, G.; Levine, B. Autophagy and the integrated stress response. Mol Cell 2010, 40, 280-293. [CrossRef]
- Dikic, I.; Elazar, Z. Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol 2018, 19, 349-364. [CrossRef]
- Fontaine, K.A.; Sanchez, E.L.; Camarda, R.; Lagunoff, M. Dengue virus induces and requires glycolysis for optimal replication. J Virol 2015, 89, 2358-2366. [CrossRef] [PubMed]
- Yu, Y.; Clippinger, A.J.; Alwine, J.C. Viral effects on metabolism: changes in glucose and glutamine utilization during human cytomegalovirus infection. Trends Microbiol 2011, 19, 360-367. [CrossRef] [PubMed]
- Thai, M.; Graham, N.A.; Braas, D.; Nehil, M.; Komisopoulou, E.; Kurdistani, S.K.; McCormick, F.; Graeber, T.G.; Christofk, H.R. Adenovirus E4ORF1-induced MYC activation promotes host cell anabolic glucose metabolism and virus replication. Cell Metab 2014, 19, 694-701. [CrossRef]
- Smallwood, H.S.; Duan, S.; Morfouace, M.; Rezinciuc, S.; Shulkin, B.L.; Shelat, A.; Zink, E.E.; Milasta, S.; Bajracharya, R.; Oluwaseum, A.J.; et al. Targeting Metabolic Reprogramming by Influenza Infection for Therapeutic Intervention. Cell Rep 2017, 19, 1640-1653. [CrossRef]
- Wu, Y.; Zhang, Z.; Li, Y.; Li, Y. The Regulation of Integrated Stress Response Signaling Pathway on Viral Infection and Viral Antagonism. Front Microbiol 2021, 12, 814635. [CrossRef] [PubMed]
- Wang, Q.; Chen, L.; Wang, J.; Chen, Z.; Ma, Z.; Xie, X.; Zeng, X.; Bie, Y.; Wang, Z. The integrated stress response suppresses antiviral RNA interference by autophagy-mediated degradation of the RNA-induced silencing complex. Proc Natl Acad Sci U S A 2025, 122, e2511857122. [CrossRef]
- Shin, W.J.; Ha, D.P.; Machida, K.; Lee, A.S. The stress-inducible ER chaperone GRP78/BiP is upregulated during SARS-CoV-2 infection and acts as a pro-viral protein. Nat Commun 2022, 13, 6551. [CrossRef]
- Fung, T.S.; Liu, D.X. Coronavirus infection, ER stress, apoptosis and innate immunity. Front Microbiol 2014, 5, 296. [CrossRef]
- Ambrose, R.L.; Mackenzie, J.M. West Nile virus differentially modulates the unfolded protein response to facilitate replication and immune evasion. J Virol 2011, 85, 2723-2732. [CrossRef]
- Wang, Y.; Li, J.; Cao, H.; Li, L.F.; Dai, J.; Cao, M.; Deng, H.; Zhong, D.; Luo, Y.; Li, Y.; et al. African swine fever virus modulates the endoplasmic reticulum stress-ATF6-calcium axis to facilitate viral replication. Emerg Microbes Infect 2024, 13, 2399945. [CrossRef]
- Liang, R.; Fu, Y.; Li, G.; Shen, Z.; Guo, F.; Shi, J.; Guo, Y.; Zhang, D.; Wang, Z.; Chen, C.; et al. EP152R-mediated endoplasmic reticulum stress contributes to African swine fever virus infection via the PERK-eIF2alpha pathway. FASEB J 2024, 38, e70187. [CrossRef]
- Gao, H.; Gu, T.; Gao, X.; Song, Z.; Liu, J.; Song, Y.; Zhang, G.; Sun, Y. African swine fever virus enhances viral replication by increasing intracellular reduced glutathione levels, which suppresses stress granule formation. Vet Res 2024, 55, 172. [CrossRef] [PubMed]
- Wang, Q.; Zhou, L.; Wang, J.; Su, D.; Li, D.; Du, Y.; Yang, G.; Zhang, G.; Chu, B. African Swine Fever Virus K205R Induces ER Stress and Consequently Activates Autophagy and the NF-kappaB Signaling Pathway. Viruses 2022, 14. [CrossRef]
- Khaminets, A.; Behl, C.; Dikic, I. Ubiquitin-Dependent And Independent Signals In Selective Autophagy. Trends Cell Biol 2016, 26, 6-16. [CrossRef] [PubMed]
- Mankouri, J.; Harris, M. Viruses and the fuel sensor: the emerging link between AMPK and virus replication. Rev Med Virol 2011, 21, 205-212. [CrossRef]
- Mayer, K.A.; Stockl, J.; Zlabinger, G.J.; Gualdoni, G.A. Hijacking the Supplies: Metabolism as a Novel Facet of Virus-Host Interaction. Front Immunol 2019, 10, 1533. [CrossRef]
- Clippinger, A.J.; Maguire, T.G.; Alwine, J.C. Human cytomegalovirus infection maintains mTOR activity and its perinuclear localization during amino acid deprivation. J Virol 2011, 85, 9369-9376. [CrossRef]
- Paul, P.; Munz, C. Autophagy and Mammalian Viruses: Roles in Immune Response, Viral Replication, and Beyond. Adv Virus Res 2016, 95, 149-195. [CrossRef]
- Deretic, V.; Saitoh, T.; Akira, S. Autophagy in infection, inflammation and immunity. Nat Rev Immunol 2013, 13, 722-737. [CrossRef]
- Datan, E.; Roy, S.G.; Germain, G.; Zali, N.; McLean, J.E.; Golshan, G.; Harbajan, S.; Lockshin, R.A.; Zakeri, Z. Dengue-induced autophagy, virus replication and protection from cell death require ER stress (PERK) pathway activation. Cell Death Dis 2016, 7, e2127. [CrossRef]
- Heaton, N.S.; Randall, G. Dengue virus-induced autophagy regulates lipid metabolism. Cell Host Microbe 2010, 8, 422-432. [CrossRef]
- Kohio, H.P.; Adamson, A.L. Glycolytic control of vacuolar-type ATPase activity: a mechanism to regulate influenza viral infection. Virology 2013, 444, 301-309. [CrossRef]
- Ritter, J.B.; Wahl, A.S.; Freund, S.; Genzel, Y.; Reichl, U. Metabolic effects of influenza virus infection in cultured animal cells: Intra- and extracellular metabolite profiling. BMC Syst Biol 2010, 4, 61. [CrossRef]
- Abedi Dorcheh, F.; Balmeh, N.; Hejazi, S.H.; Allahyari Fard, N. Investigation of the mutated antimicrobial peptides to inhibit ACE2, TMPRSS2 and GRP78 receptors of SARS-CoV-2 and angiotensin II type 1 receptor (AT1R) as well as controlling COVID-19 disease. J Biomol Struct Dyn 2025, 43, 1641-1664. [CrossRef]
- Shin, J.; Toyoda, S.; Fukuhara, A.; Shimomura, I. GRP78, a Novel Host Factor for SARS-CoV-2: The Emerging Roles in COVID-19 Related to Metabolic Risk Factors. Biomedicines 2022, 10. [CrossRef]
- Cheng, L.; Qiang, R.; Song, H.; Zhou, Q.; Lv, X.; Wu, M.; Xu, J.; Xu, P. Hepatitis B virus induces T cell exhaustion by increasing mitochondrial ROS accumulation. Microb Pathog 2026, 212, 108289. [CrossRef]
- Koyaweda, G.W.; Glitscher, M.; Schollmeier, A.; Bender, D.; Hildt, E. Isochlorogenic acid A impairs hepatitis B virus replication by interference with various steps of hepatitis B virus life cycle involving HO-1-mediated ROS modulation. Antiviral Res 2026, 245, 106323. [CrossRef]
- Lin, Y.; Wu, C.; Wang, X.; Liu, S.; Zhao, K.; Kemper, T.; Yu, H.; Li, M.; Zhang, J.; Chen, M.; et al. Glucosamine promotes hepatitis B virus replication through its dual effects in suppressing autophagic degradation and inhibiting MTORC1 signaling. Autophagy 2020, 16, 548-561. [CrossRef]
- Wing, P.A.C.; Liu, P.J.; Harris, J.M.; Magri, A.; Michler, T.; Zhuang, X.; Borrmann, H.; Minisini, R.; Frampton, N.R.; Wettengel, J.M.; et al. Hypoxia inducible factors regulate hepatitis B virus replication by activating the basal core promoter. J Hepatol 2021, 75, 64-73. [CrossRef]
- Levy, P.L.; Duponchel, S.; Eischeid, H.; Molle, J.; Michelet, M.; Diserens, G.; Vermathen, M.; Vermathen, P.; Dufour, J.F.; Dienes, H.P.; et al. Hepatitis C virus infection triggers a tumor-like glutamine metabolism. Hepatology 2017, 65, 789-803. [CrossRef]
- Ke, P.Y.; Yeh, C.T. Functional Role of Hepatitis C Virus NS5A in the Regulation of Autophagy. Pathogens 2024, 13. [CrossRef]
- Lee, J.; Ou, J.J. HCV-induced autophagy and innate immunity. Front Immunol 2024, 15, 1305157. [CrossRef]
- Guerrero, M.; Hernandez, J.; Gomez, L.; Guerrero, C. Oxidative stress enhances rotavirus oncolysis in breast cancer and leukemia, except in melanoma with abundant matrix. Virus Res 2024, 339, 199285. [CrossRef]
- Dong, X.; Wang, Y.; Zhu, X.; Shen, L.; Chen, L.; Niu, L.; Gan, M.; Zhang, S.; Zhang, M.; Jiang, J.; et al. Sodium butyrate protects against rotavirus-induced intestinal epithelial barrier damage by activating AMPK-Nrf2 signaling pathway in IPEC-J2 cells. Int J Biol Macromol 2023, 228, 186-196. [CrossRef]
- Zhao, Y.; Hu, N.; Jiang, Q.; Zhu, L.; Zhang, M.; Jiang, J.; Xiong, M.; Yang, M.; Yang, J.; Shen, L.; et al. Protective effects of sodium butyrate on rotavirus inducing endoplasmic reticulum stress-mediated apoptosis via PERK-eIF2alpha signaling pathway in IPEC-J2 cells. J Anim Sci Biotechnol 2021, 12, 69. [CrossRef]
- Guerrero, C.A.; Acosta, O. Inflammatory and oxidative stress in rotavirus infection. World J Virol 2016, 5, 38-62. [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




